1. Introduction
To enhance quality of life, maintaining optimal oral health is paramount. Regrettably, according to the World Health Organization (WHO), approximately 3.5 billion individuals globally are afflicted by oral diseases [
1,
2]. These oral conditions arise due to inadequate management of biofilm, commonly referred to as plaque [
3]. Plaque is characterized as a deposit forming a biofilm composed of adherent bacteria within the oral cavity [
4]. This biofilm can trigger inflammation in periodontal tissues. It has been identified as a significant contributor to gingivitis and periodontitis [
5]. Notably, deposit of plaque within the oral cavity can manifest clinical indications within 10 days [
6,
7]. Hence, to avert or manage periodontal diseases, it becomes imperative to either remove or impede the formation of plaque [
8]. Efficient plaque removal proves pivotal in regulating periodontal diseases.
Notably, plaque has direct correlations with systemic illnesses such as stroke [
9], diabetes [
10], cardiovascular disease [
11], and Alzheimer’s disease [
12]. Consequently, effective removal of plaque is important for both public and private healthcare. Plaque is removed by a mechanical method using a toothbrush or oral products as a typical method [
13,
14]. In the 1960s, electric toothbrushes were developed that increased plaque removal efficiency [
15]. Nevertheless, with 3.5 billion people worldwide suffering from oral diseases, typical plaque removal methods might not be effective. Therefore, a more efficient means of removing plaque is needed.
Toothpaste is commonly employed during teeth brushing routines. While toothpaste could effectively aid in plaque removal, ongoing discussions regarding its safety arise due to the presence of abrasives, surfactants, and fluoride [
16,
17,
18]. Overdosing of fluoride has been associated with adverse effects such as skeletal deformities, cancer, and gastric mucosa damage [
18,
19,
20]. Among pregnant women, certain studies have indicated potential risks of birth defects [
21]. Additionally, concerns are being raised, particularly concerning children, regarding potential seriousness such as teeth and skeletal underdevelopment [
17,
22,
23].
The durability of a biofilm extends to the point where antibiotic resistance becomes 500 to 5000 times more pronounced than that exhibited by individual bacteria [
8,
24,
25]. Biofilms consist of polysaccharides and electrically polarized bacterial cells [
26,
27]. External electrical stimuli can disrupt biofilm’s structural integrity and impact the metabolic state of bacteria [
28,
29,
30,
31]. Utilizing an alternating current (AC) at a specific frequency can enhance the porosity of the biofilm’s structure by inducing vibrations [
32,
33]. Conversely, a direct current (DC) can induce changes in the electrolyte state [
28,
34]. As demonstrated in a recent study, combined application of an electrical signal known as the bioelectric effect (BE) along with both DC and AC is remarkably effective for biofilm removal [
28,
29,
30,
31].
Recently, our group has developed a toothbrush with a bioelectric effect and demonstrated its effectiveness in reducing plaque-induced dental gingival index in clinical trials [
8,
35]. However, clinical trials have the disadvantage of being difficult to quantitatively evaluate due to variations among subjects [
36]. In vitro tests are more convenient and highly reproducible than in vivo tests. Therefore, they are widely used in research related to the human body [
36,
37,
38]. Likewise, in relation to toothbrushes, studies using various in vitro brushing simulators have been reported [
39,
40,
41,
42].
In this study, we quantitatively compared plaque removal efficiency of a microcurrent toothbrush and a regular toothbrush to observe the effect of bioelectrical effect on plaque. We performed a quantitative plaque removal efficiency test using a developed brushing simulator for four types of toothbrushes (two types of regular toothbrushes and proposed toothbrush (with or without BE) under two conditions (with only saline and with toothpaste slurry). Based on experimental results, we investigated plaque removal efficiency of BE and toothpaste dependence for each.
2. Materials and Methods
2.1. Tested Toothbrushes and Design of BE Toothbrush
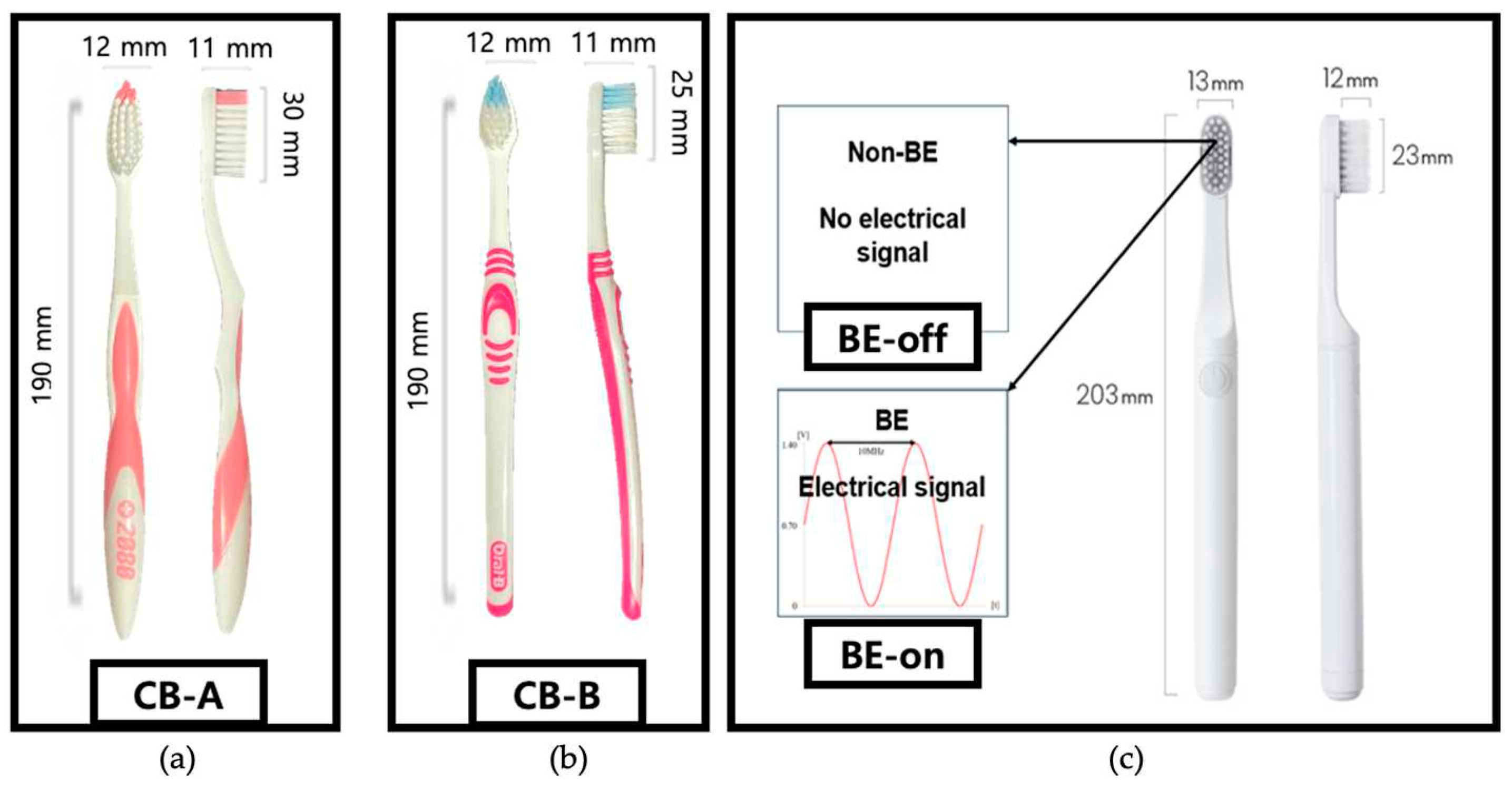
Tested toothbrushes and their abbreviations are listed in
Table 1 and
Figure 1. We aimed to compare plaque removal efficiency of a toothbrush with BE applied. Two commonly used toothbrushes and two developed toothbrushes were tested under two conditions: BE off and on. This condition was specified to investigate effect of bristles (typical toothbrush versus proposed toothbrush) on plaque removal.
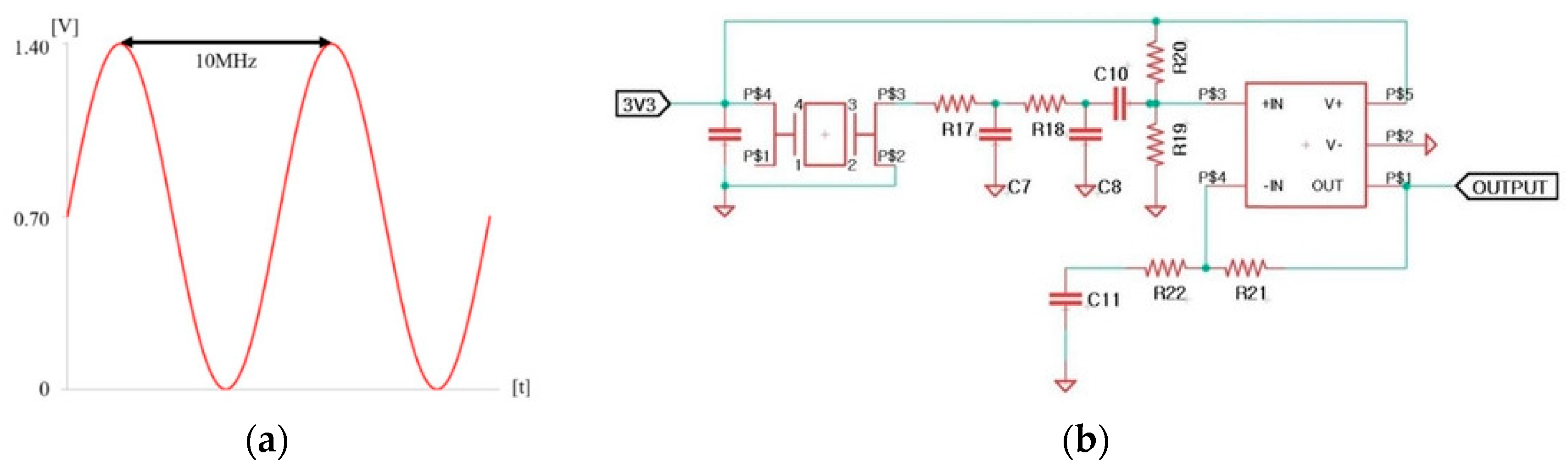
BE applies a combination of electrical signals to remove biofilm. The electrical signal was a combination of a 0.7 V sinusoidal signal at a frequency of 10 MHz with a DC offset of 0.7 V as shown in
Table 2 [
8,
35].
The frequency of the electrical signal was selected based on previous studies. DC offset was set below the electrolysis threshold of 0.82 V [
28]. Our previous study has revealed that this signal does not induce electrolysis [
35]. The electrical signal is shown in
Figure 2a. An electronic circuit for generating the output signal was developed, as shown in
Figure 2b. Electronic circuits were embedded in the proposed toothbrush. A stainless steel electrode was chosen due to its corrosion resistance and conductivity essential for supplying an electric field. The signal for BE is output through these two electrodes.
The current from the signal output was measured using a multimeter (EDU34450A, Keysight Technologies, Santa Rosa, CA, USA) in 0.9% saline condition. The measured current was about 40.7 ± 1.5 μA (N = 3). The current of BE was investigated to be safe because it was within the biocompatible current range [
43].
2.2. Development of Brushing Simulator
The brushing simulator consists of five linear actuators with a stepper motor (Scipia, Gwangju, Korea), five motor drivers (TB6600, DFRobot, Shanghai, China), four piezoelectric pressure sensors (QA3040P, Marveldex Inc., Bucheon, Korea), a switching mode power supply (HU10748-13002E, Yueqing Hengwei Electric Co., YueQing, China), and a construct of typodonts and toothbrushes. The construct was produced using a 3D printer (X1-carbon, Bambu lab, China) and PolyLactic Acid (PLA). A standard typodonts was located construct of brushing simulator. For brushing control and pressure exerted by the toothbrush data acquisition (DAQ), we used an Arduino Uno (Arduino.cc, Ivrea, Italy). The toothbrushing simulator was capable of simulating toothbrushing at up to 150 strokes/min in a 5 cm driving range. The brushing simulator is shown in
Figure 3.
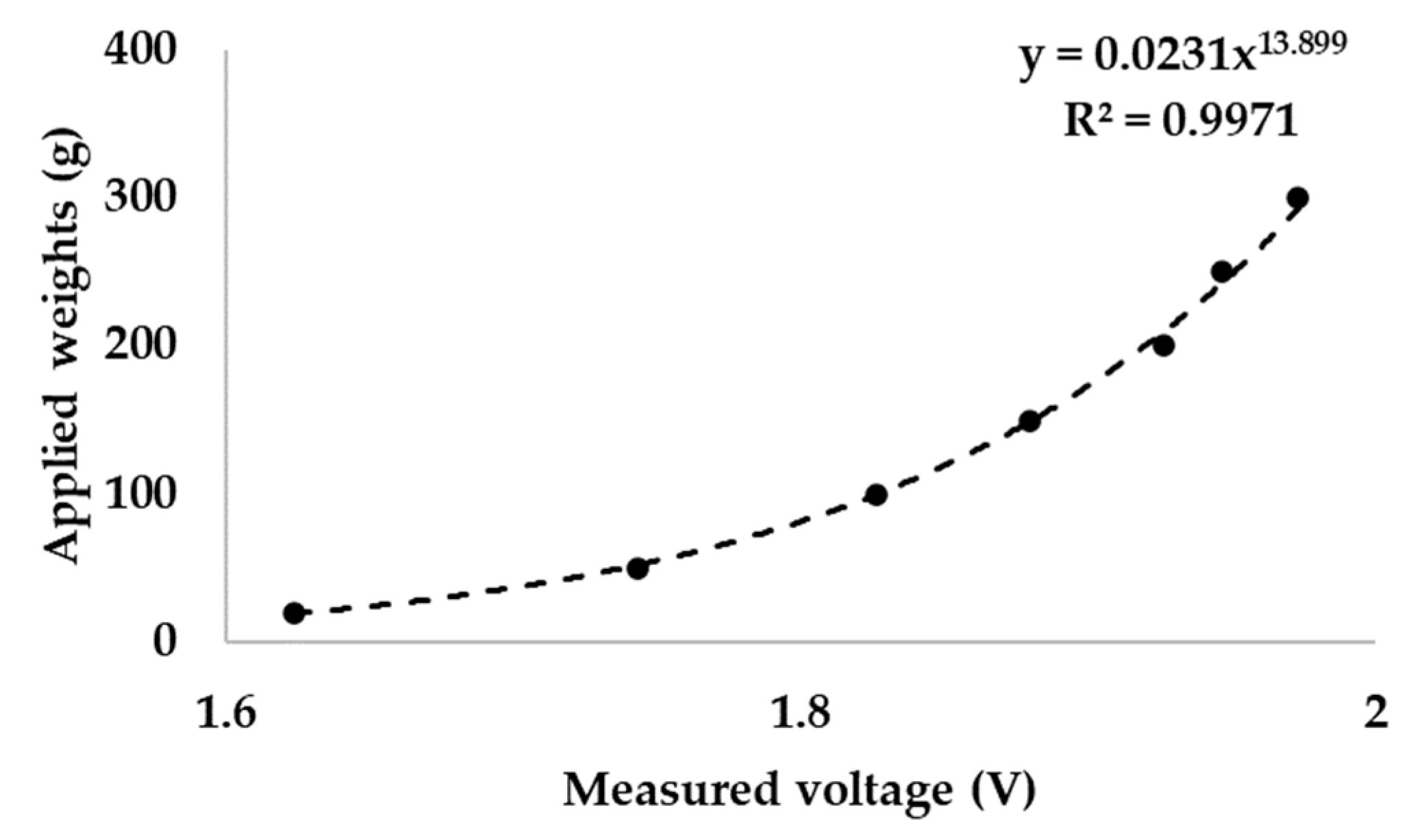
The pressure sensor of the simulator was calibrated by applying various mass from 20 to 300 g. The result showed a standard correlation of piezoelectric material (
Figure 4). A piezoelectric pressure sensor can be calibrated using a power function [
44]. A standard weight was applied to the pressure sensor and the output voltage was recorded (N = 3). According to ISO/TR 14569-1, the pressure that the toothbrush presses on the artificial tooth is between 0.5 and 2.5 N [
45]. The measured weight was converted to newton (N).
2.3. Plaque Culture
Streptococcus mutans (KCTC 3065, Korean Collection for Type Cultures (KCTC), Jeongeup, Korea) used in this research were distributed from KCTC. It was cultured in a growth media (LB Broth, Ambrothia Inc., Daejeon, Korea) at 37 °C for 48 hours to provide sufficient time for maturation.
2.4. Experiment of Plaque Removal
Typodonts were evenly coated with the cultured
S. mutans. Green marker spray (OccludeTM, Pascal, Bellevue, WA, USA) was used to visualize
S. mutans. (plaques) [
41]. The coated typodont was mounted on the brushing simulator. The force that the toothbrush pressed on the tooth was set to be 0.5 N and 2.5 N. According to ISO/TR 14569-1, the force that a toothbrush presses on the teeth is between 0.5 N and 2.5 N [
45]. The molar area was brushed [
41]. The brushing frequency was 120 strokes/minute [
45] for 30 seconds. The recommended time for brushing is generally 2 minutes [
46,
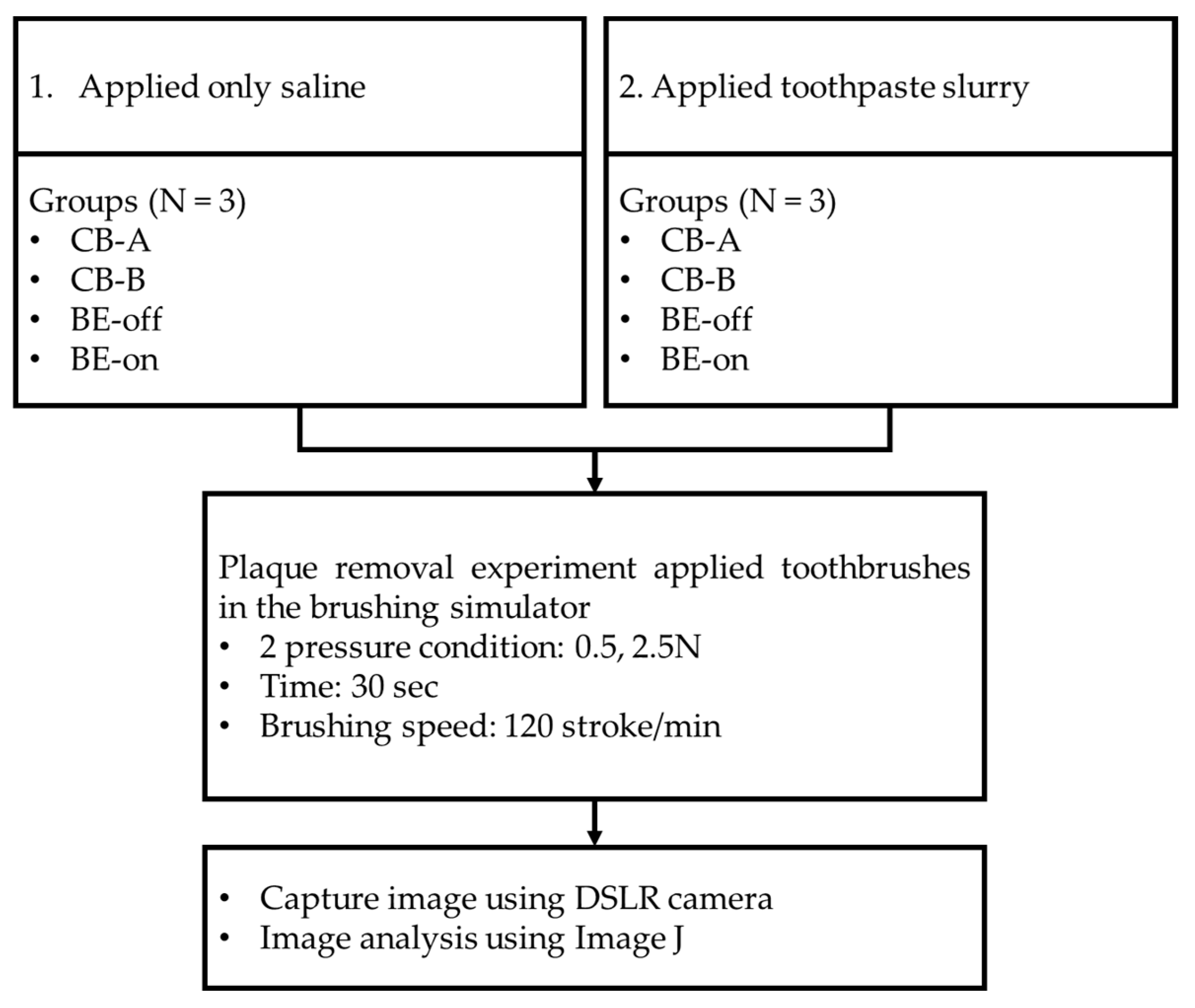
47]. The target area was approximately 1/4 of the entire tooth. The driving range was set to 4 cm as a range of molar area approximately. After brushing, tested typodonts were eluted in tab water for 30 seconds. This process was used to remove the plaque that was separated by brushing the teeth so that only the residual plaque would be quantified. According to our preliminary test, it was determined that this process did not cause unintended plaque elution. Plaques of typodonts before and after the experiment were captured under controlled lighting conditions using a DSLR camera (A350, Sony Co., Tokyo, Japan). Image analysis was performed using ImageJ (National Institutes of Health, Bethesda, MD, USA). Two experiments were conducted (N = 3). The experimental design is shown in
Figure 5.
The first experiment (applied only saline) was conducted to determine the biofilm removal effect of using only BE without a toothpaste. Saline solution has been used in several studies to simulate saliva [
48,
49]. The second experiment (applied toothpaste slurry) was designed to have typical brushing conditions. The toothpaste used was a toothpaste containing fluorine at 1450 ppm (Grate regular flavor toothpaste, Colgate, New York, USA).
In our previous work, we have found that a toothbrush with applied BE in a clinical trial could significantly reduce gingivitis index [
8,
35]. Therefore, an additional comparison was made between a typical toothbrush using toothpaste and a BE toothbrush without toothpaste.
2.5. Statistical Analysis
Residual plaques are presented as means and standard deviations (SDs). A two-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test was used to determine the significance of differences between the experimental condition and the CB-A at 0.5 N as control. Paired t-tests were used to compare differences of BE-on between saline and toothpaste slurry. All statistical analyses were performed using R-studio version 4.3.1 (Posit, Boston, MA, USA). P-values of less than 0.05 were considered significant.
3. Results
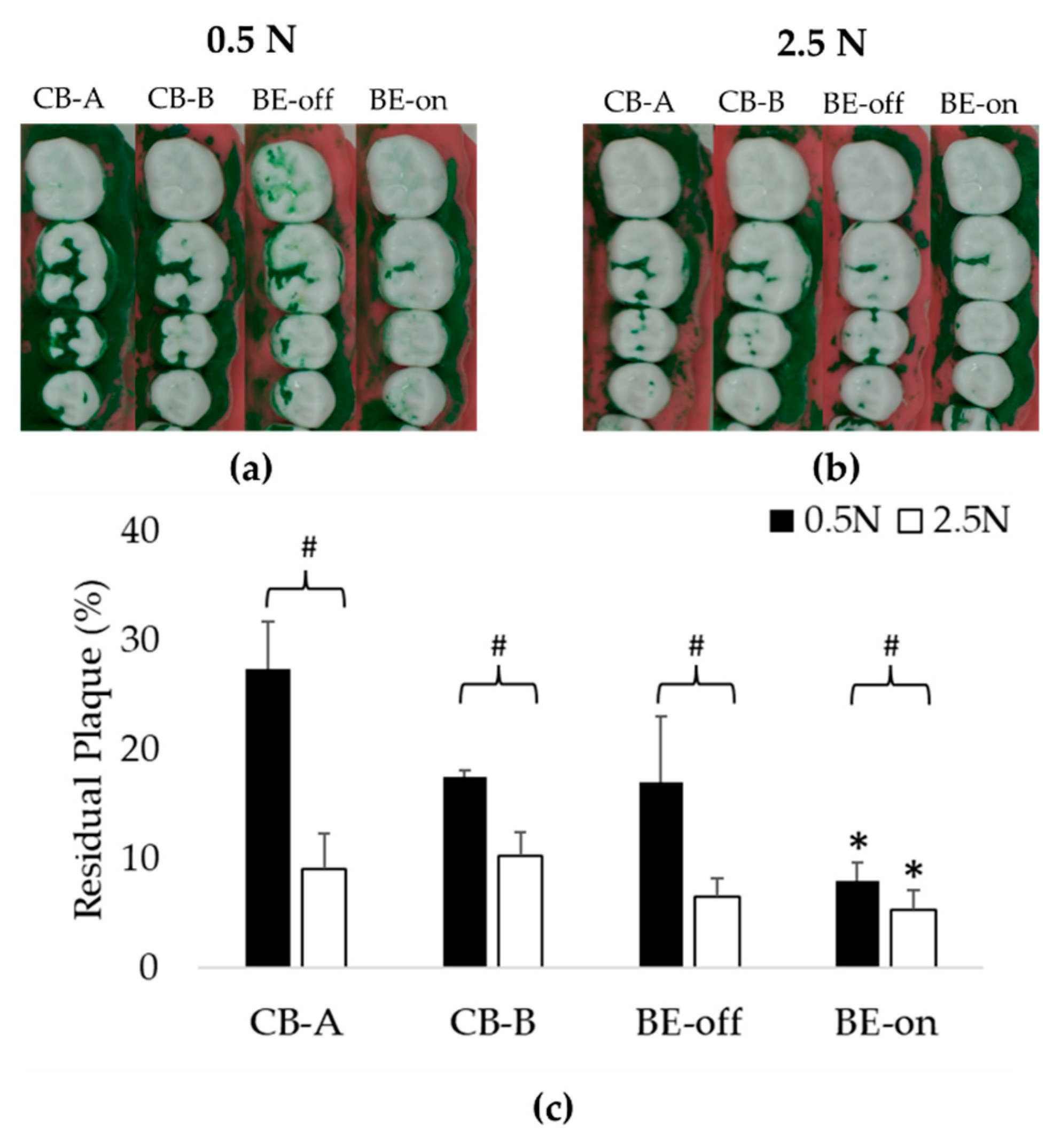
3.1. Experiment Using Only Saline
This experiment was designed to evaluate the plaque removal efficiencies to observe the effect of BE without using a toothpaste. In all pressure conditions, the residual plaque using BE-on toothbrush was significantly lower than that using other toothbrushes. Toothbrushes except for BE-on showed no significant difference in residual plaque as shown in
Figure 6. A previous study has reported that there is no significant difference in plaque removal rate according to the shape of bristles [
50]. This result suggests that the high plaque removal rate might be due to the application of BE. The order of residual plaque was 0.5 N > 2.5 N. The higher the pressure with which the toothbrush presses against the teeth, the higher the plaque removal rate. These results are consistent with those of previous reports [
51].
At 0.5 N, BE-on showed a 7.96 ± 1.67% of residual plaque, while CB-A, CB-B, and BE-off showed 27.42 ± 4.30%, 17.45 ± 0.59%, and 17.03 ± 5.99% of residual plaque, respectively. At 2.5 N, BE-on showed a 5.24 ± 1.86% of residual plaque, while CB-A, CB-B, and BE-off showed 9.08 ± 3.18%, 10.20 ± 2.18%, and 6.55 ± 1.59% of residual plaque, respectively. The highest plaque removal efficiency was observed with BE-on at 2.5 N. On average, the plaque removal rate with BE-on was 215.43 ± 89.92% higher at 0.5 N and 116.77% ± 47.02% higher at 2.5 N than those using other toothbrushes.
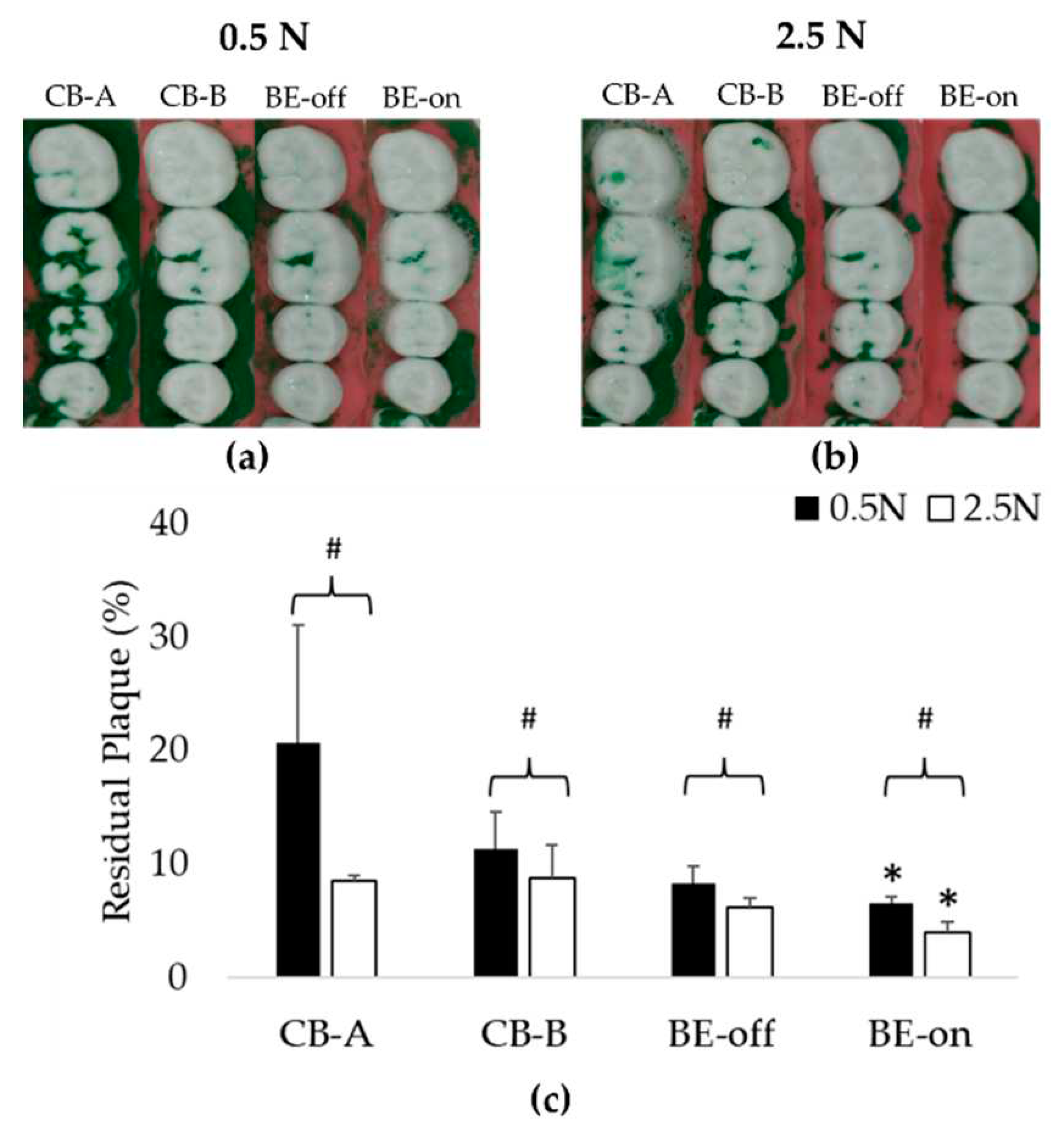
3.2. Experiement Appling a Toothpaste Slurry
This experiment was designed to evaluate plaque removal efficiencies in typical situations. As observed in the experiment applying only saline, the residual plaque using BE-on was significantly lower than those using other toothbrushes under all pressure conditions as shown in
Figure 7. Using toothpaste can increase plaque removal efficiency [
16,
17,
18]. Therefore, the residual plaque in this experiment using a toothpaste slurry was lower than that when using only saline.
The order of residual plaque was 0.5 N > 2.5 N as shown in
Figure 7. At 0.5 N, BE-on showed a 6.54 ± 0.59% of residual plaque, while CB-A, CB-B, and BE-off showed 20.66 ± 10.38%, 11.32 ± 3.24%, and BE-off: 8.23 ± 1.54% of residual plaque, respectively. At 2.5 N, BE-on showed a 3.97 ± 0.97% of residual plaque, while CB-A, CB-B, and BE-off showed 8.51 ± 0.53%, 8.69 ± 2.95%, and 6.19 ± 0.84% of residual plaque, respectively. BE-on was the most efficient in all pressure conditions. The highest plaque removal efficiency was observed for BE-on at 2.5 N. On average, the plaque removal rate using BE-on was 104.96 ± 98.93% higher at 0.5 N and 96.23% ± 35.16% higher at 2.5 N than those using other toothbrushes.
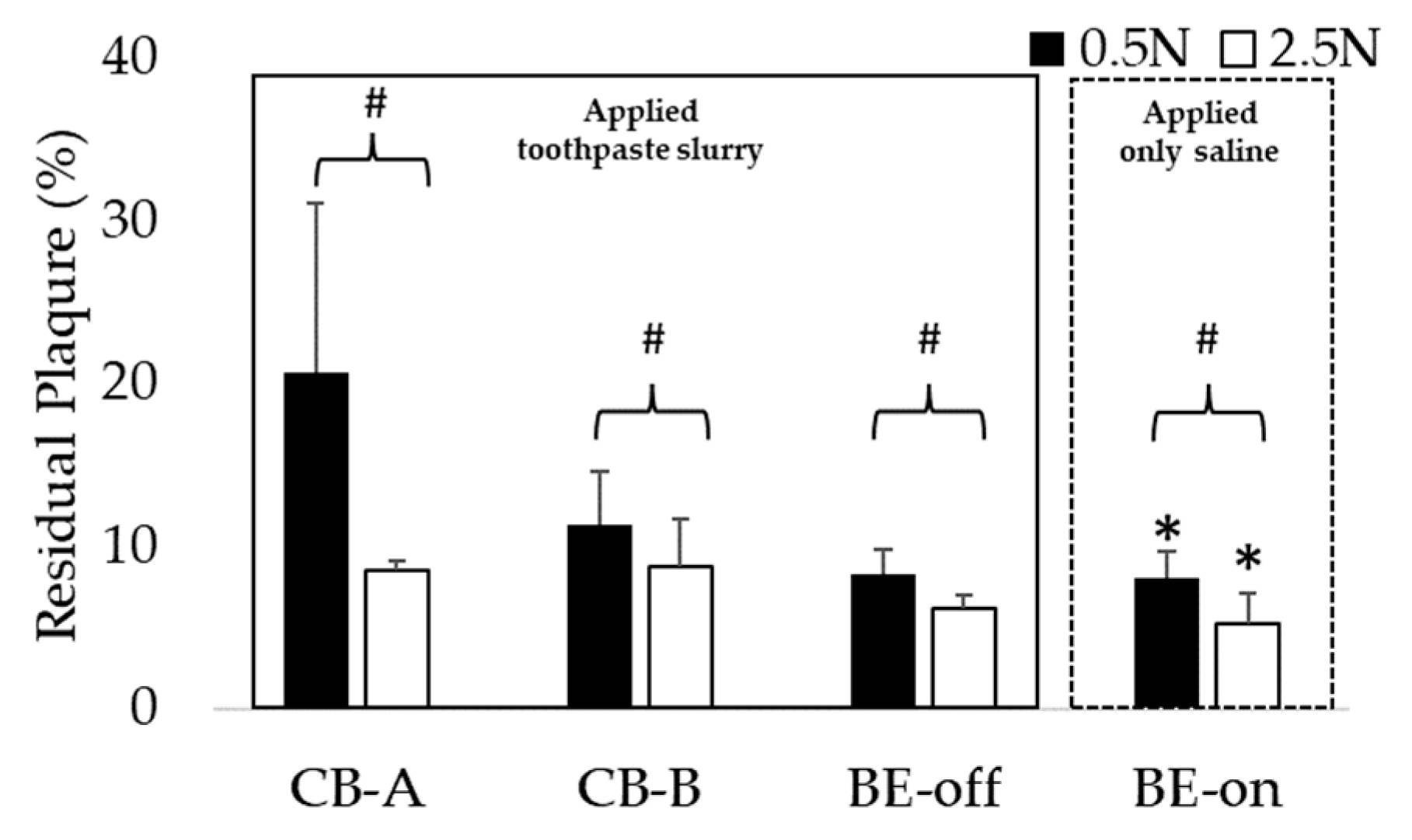
3.3. Comparison of Residual Plaque in Applied Saline with BE Versus in Toothpaste Slurry without BE
Through this comparison, we investigated whether BE could replace toothpaste. We investigated whether using BE-on with only saline had significantly lower residual plaque than using other toothbrushes with a toothpaste and saline slurry under all pressure conditions. As shown in
Figure 8, the highest plaque removal efficiency was observed for BE-on without a toothpaste at 2.5 N. These results suggest that applying BE to a toothbrush can replace toothpaste.
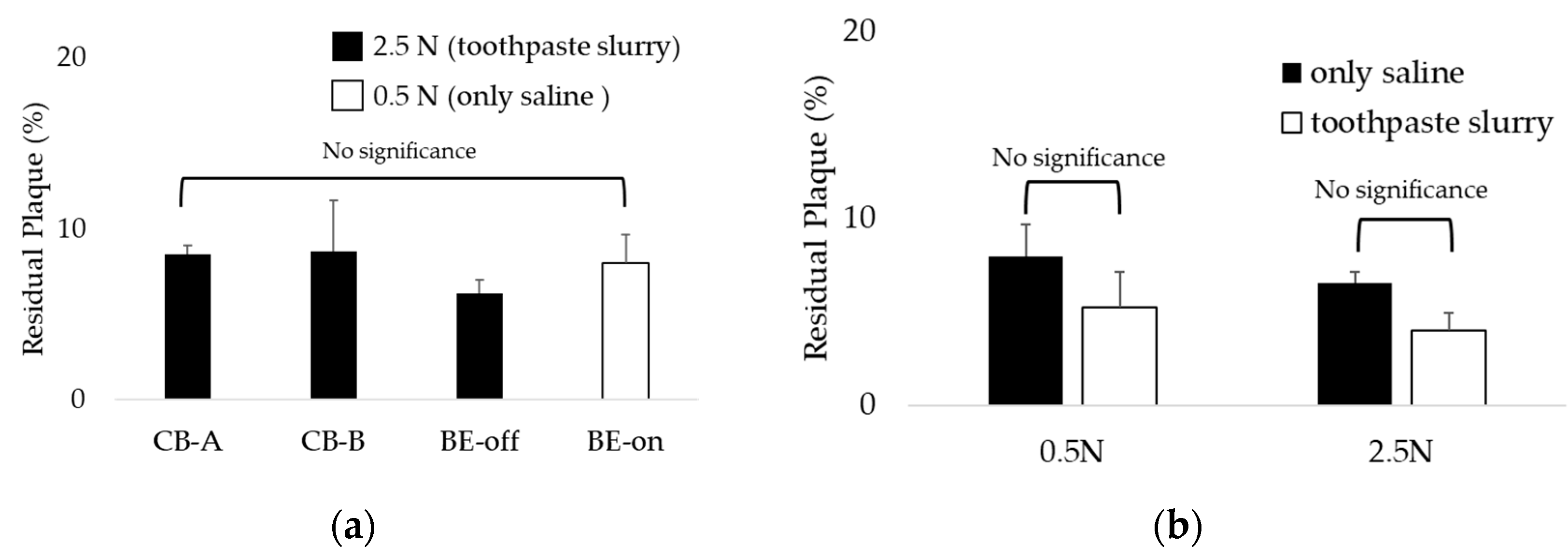
Interestingly, the BE-on under the 0.5N condition using only saline showed residual plaque similar to other toothbrushes under the 2.5N condition using toothpaste and saline slurry as shown in
Figure 9a. This result suggests that even if brushing is weak without a toothpaste, plaque can be removed similarly to general brushing conditions if BE is applied. Also, as a result of comparing the residual plaque of BE-on using only saline or a toothpaste and saline slurry, there was no significant difference as shown in
Figure 9b. Therefore, if BE is applied, the dependence on toothpaste for plaque removal is expected to decrease.
4. Discussion
In both only saline and toothpaste slurry experiments, there was no significant difference in residual plaque among toothbrushes except for BE-on. According to a previous report, there was no significant difference in plaque removal efficiency according to toothbrush bristles alone [
50], similar to results of the present study. There is a report showing that brushing under high pressure conditions is more effective in removing plaque than under a low pressure condition [
51]. Similar results were observed in this study as residual plaque was lower under high pressure (2.5 N) conditions. The use of toothpaste also affected the plaque removal efficiency [
16,
17,
18]. The condition of using toothpaste was 34.67 ± 33.10% higher in plaque removal on average than without a toothpaste. This is similar to a previously reported study [
52].
BE-on had a significantly lower residual plaque than other toothbrushes without BE under the same condition. Plaque is a type of biofilm. Previous studies have reported that the treatment efficiency of biofilm is high when BE is applied [
28,
29,
30,
31]. BE is caused by propagation of electromagnetic currents, waves, and voltages. The experimental condition is an electrically dielectric state due to only saline or toothpaste slurry used. Saline has been used in several studies to simulate saliva [
48,
49]. It seems that effective propagation of the electric field can increase the biofilm removal effect. Therefore, a toothbrush applied with BE is expected to have a higher plaque removal efficiency.
Interestingly, it was observed that the residual plaque using BE-on without a toothpaste was significantly lower those using toothbrushes with a toothpaste under the same conditions. Based on these results, we propose BE as a possible substitute for toothpaste. Regardless of toothpaste application, no significant difference was observed in the residual plaque of BE-on. This result indicates that if BE is applied, the dependence on toothpaste for teeth brushing can be reduced. There is controversy regarding the safety of toothpaste due to abrasives, surfactants, and fluoride [
16,
17,
18]. Generally, it includes potential carcinogenic risk, skeletal deformity, and gastric mucosal damage [
18,
19,
20]. In child and infants, this includes developmental disorders in the fetus and bone un-development in children [
17,
22,
23]. We anticipate that BE will be a good option for people who find it difficult to use toothpaste because of their controversial points.
Studies have reported that brushing teeth under high pressure conditions can accelerate tooth wear [
40,
41]. In this study, we found that there was no significant difference in residual plaque between BE-on at low pressure (0.5 N) without toothpaste and other toothbrushes with a high pressure (2.5 N) and a toothpaste. Therefore, it can be proposed that the application of BE can effectively remove plaque even without toothpaste or aggressive brushing. In addition, under the condition of the lowest pressure, brushing with only BE applied without toothpaste showed a plaque removal efficiency similar to that using the highest pressure and a toothpaste. This means that a toothbrush using BE can show good plaque removal efficiency without excessive brushing or toothpaste. Hard brushing can increase tooth wear [
40,
41]. Therefore, it is expected that the wear of permanent teeth can be reduced if BE is applied. We believe that the application of BE will reduce not only the potential risk of toothpaste, but also the potential risk of tooth wear caused by strong brushing.
The use of an in vitro simulator not only has high experimental reproducibility compared to clinical trials, but also enables quantitative testing [
36]. In this study, we quantitatively compared the plaque removal efficiency of BE toothbrushes and other toothbrushes using an in vitro simulator. In our previous clinical study, BE-applied toothbrush significantly reduced the gingival index, but not the plaque index [
8]. This might be because there was variation among subjects in the clinical study. It was possible that a significant plaque index was not observed due to bias of human. However, since this study conducted an in vitro experiment using brushing simulator capable of quantitative evaluation, we were able to observe significant plaque removal efficiency.
Based on these research results, we intend to additionally develop various oral care products including toothbrushes. Plaque can cause various problems in the oral cavity, not only on the teeth, but also on parts such as the tongue [
53]. Not only people, but also pets have various problems due to plaque [
54]. Therefore, we plan to develop oral care products with BE, including tongue cleaners and pet toothbrushes, to solve various problems related to plaque.
In this study, we evaluated the plaque removal efficiency of a BE toothbrush in a simulated brushing environment. However, our study has some limitations. First, we used consistent brushing speed and pressure, which might not reflect the variability in brushing pattern that occurs in real life [
55]. Second, we used the same typodont for our tests, which does not account for differences in oral cavity structure that exist from person to person [
56]. Third, we only evaluated plaque removal in one area of the teeth, although the structure of teeth varies from region to region. Therefore, additional studies are needed to determine the plaque removal efficiency of BE in more realistic conditions. These studies should investigate effects of different brushing patterns, pressure conditions, and oral cavity structures.
5. Conclusions
In this study, we developed a BE toothbrush and investigated the plaque removal effect of BE through quantitative plaque removal experiments using a brushing simulator. A BE toothbrush was observed to be effective in removing plaque. We believe that the application of BE will contribute to effective oral care. In addition, the application of BE not only shows the possibility of replacing toothpaste, but also shows the ability to reduce the intensity of brushing. Thus, it is expected that the wear of permanent teeth can also be reduced. In the future, we will develop various oral care products applying BE, including tongue scrapper, toothbrushes for pets, and so on.
Author Contributions
H.M.P., and Y.W.K. planned experiments; H.M.P., S.J.R., E.A.J., performed experiments; H.M.P. and Y.W.K. analyzed and calculated the data; H.M.P., E.A.J., S.J.R., S.K.Y., and Y.W.K. wrote, reviewed, and revised the paper. All authors read and approved the final manuscript.
Funding
This work was supported by the Technology development Program (No. S3301078) funded by the Ministry of SMEs and Startups (MSS, Korea).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lorenzo-Pouso, A.I.; Pérez-Sayáns, M.; Bravo, S.B.; López-Jornet, P.; García-Vence, M.; Alonso-Sampedro, M.; Carballo, J.; García-García, A. Protein-Based Salivary Profiles as Novel Biomarkers for Oral Diseases. Dis. Markers 2018, 2018, 6141845. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Available online: https://www.who.int/health-topics/oral-health#tab=tab_1 (accessed on 8 September 2023).
- Allaker, R.P.; Douglas, C.I. Novel anti-microbial therapies for dental plaque-related diseases. Int. J. Antimicrob. Agents 2009, 33, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Marsh, P.D. Dental plaque: Biological significance of a biofilm and community life-style. J. Clin Periodontol. 2005, 32, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Abebe, G.M. Oral Biofilm and Its Impact on Oral Health, Psychological and Social Interaction. Int. J. Oral Dent. Health 2021, 7, 127. [Google Scholar]
- Löe, H.; Theilade, E.; Jensen, S.B. Experimental Gingivitis in Man. J. Periodontol. 1965, 36, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Lovdal, A.; Arno, A.; Waerhaug, J. Incidence of clinical manifestations of periodontal disease in light of oral hygiene and calculus formation. J. Amer. Dent. Assoc. 1958, 56, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Lee, J.; Lee, T.H. Bioelectric effect utilized a healthcare device for effective management of dental biofilms and gingivitis. Med. Eng. Phys. 2022, 104, 103804. [Google Scholar] [CrossRef]
- Alhadainy, H.A.; Keefe, T.; Abdel-Karim, A.H.; Abdulrab, S.; Halboub, E. Association between dental diseases and history of stroke in the United States. Clin. Exp. Dent. Res. 2021, 7, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Albert, D.A.; Ward, A.; Allweiss, P. Diabetes and oral disease: Implications for health professionals. Ann. N. Y. Acad. Sci. 2012, 1255, 1–15. [Google Scholar] [CrossRef]
- Kotronia, E.; Brown, H.; Papacosta, A.O.; Lennon, L.T.; Weyant, R.J.; Whincup, P.H.; Wannamethee, S.G.; Ramsay, S.E. Oral health and all-cause, cardiovascular disease, and respiratory mortality in older people in the UK and USA. Sci. Rep. 2021, 11, 16452. [Google Scholar] [CrossRef]
- Ming, Y.; Hsu, S.W.; Yen, Y.; Lan, S.J. Association of oral health–related quality of life and Alzheimer disease: A systematic review. J. Prosthet. Dent. 2020, 124, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Sälzer, S.; Slot, D.E.; Van der Weijden, F.A.; Dörfer, C.E. Efficacy of inter-dental mechanical plaque control in managing gingivitis—A meta-review. J. Clin. Periodontol. 2015, 42, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Westfelt, E. Rationale of mechanical plaque control. J. Clin. Periodontol. 1996, 23, 263–267. [Google Scholar] [CrossRef]
- Jepsen, S. Role of m toothbrushes in effective plaque control: Advantages and limitations. In Proceedings of European Workship on Mechanical Plaque Control, 1st ed.; Lang, N.P., Attstrom, R., Love, H., Eds.; Quintessence Publishing: Batavia, IL, USA; pp. 121–137.
- Hara, A.T.; Turssi, C.P. Baking soda as an abrasive in toothpastes: Mechanism of action and safety and effectiveness considerations. J. Am. Dent. Assoc. 2017, 148, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Stovell, A.G.; Newton, B.M.; Lynch, R.J.M. Important considerations in the development of toothpaste formulations for children. Int. Dent. J. 2013, 63, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Ullah, R.; Zafar, M.S.; Shahani, N. Potential fluoride toxicity from oral medicaments: A review. Iran. J. Basic Med Sci. 2017, 20, 841–848. [Google Scholar]
- Mohideen, H.; Dahiya, D.S.; Parsons, D.; Hussain, H.; Ahmed, R.S. Skeletal Fluorosis: A Case of Inhalant Abuse Leading to a Diagnosis of Colon Cancer. J. Investig. Med. High Impact Case Rep. 2022, 10, 23247096221084919. [Google Scholar] [CrossRef]
- Pashley, D. H.; Allison, N. B.; Easmann, R. P.; McKinney, R. V.; Horner, J. A.; Whitford, G. M. The effects of fluoride on the gastric mucosa of the rat. J. Oral Path. Med. 1984, 13, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, L.; Zhao, Y. Awareness of the Benefits and Risks Related to Using Fluoridated Toothpaste Among Doctors: A Population-Based Study. Med. Sci. Monit 2019, 25, 6397–6404. [Google Scholar] [CrossRef]
- Basch, C. H.; Rajan, S. Marketing strategies and warning labels on children’s toothpaste. J. Am. Dent. Hyg. Assoc. 2014, 88, 316–319. [Google Scholar]
- Buzalaf, M.A.; Levy, S.M. Fluoride intake of children:considerations for dental caries and dental fluorosis. Monogr. Oral. Sci. 2011, 22, 1–19. [Google Scholar] [PubMed]
- Hathroubi, S.; Mekni, M.A.; Domenico, P.; Nguyen, D.; Jacques, M. Biofilms: Microbial shelters against antibiotics. Microb. Drug Resist. 2017, 23, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Costerton, J.W.; Ellis, B.; Lam, K.; Johnson, F.; Khoury, A.E. Mechanism of electrical enhancement of efficacy of antibiotics in killing biofilm bacteria. Antimicrob. Agents Chemother. 1994, 38, 2803–2809. [Google Scholar] [CrossRef] [PubMed]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Daubert, D.M.; Weinstein, B.F. Biofilm as a risk factor in implant treatment. Periodontology 2000 2019, 81, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Subramanian, S.; Gerasopoulos, K.; Ben-Yoav, H.; Wu, H.C.; Quan, D.; Carter, K.; Meyer, M.T.; Bentley, W.E.; Ghodssi, R. Effect of Electrical Energy on the Efficacy of Biofilm Treatment Using the Bioelectric Effect. NPJ Biofilms Microbiomes 2015, 1, 15016. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Meyer, M.T.; Berkovich, A.; Subramanian, S.; Iliadis, A.A.; Bentley, W.E.; Ghodssi, R. A surface acoustic wave biofilm sensor integrated with a treatment method based on the bioelectric effect. Sens. Actuators A Phys. 2016, 238, 140–149. [Google Scholar] [CrossRef]
- Pareilleux, A.; Sicard, N. Lethal effects of electric current on Escherichia coli. Appl. Microbiol. 1970, 19, 421–424. [Google Scholar] [CrossRef]
- Blenkinsopp, S.A.; Khoury, A.; Costerton, J. Electrical enhancement of biocide efficacy against Pseudomonas aeruginosa biofilms. Appl. Environ. Microbiol. 1992, 58, 3770–3773. [Google Scholar] [CrossRef]
- Caubet, R.; Pedarros-Caubet, F.; Chu, M.; Freye, E.; de Belém Rodrigues, M.; Moreau, J.M.; Ellison, W.J. A radio frequency electric current enhances antibiotic efficacy against bacterial biofilms. Antimicrob. Agents Chemother. 2004, 48, 4662–4664. [Google Scholar] [CrossRef]
- del Pozo, J.L.; Rouse, M.S.; Mandrekar, J.N.; Steckelberg, J.M.; Patel, R. The electricidal effect: Reduction of staphylococcus and pseudomonas biofilms by prolonged exposure to low-intensity electrical current. Antimicrob. Agents Chemother. 2009, 53, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, P.; DeBeer, D.; Lappin-Scott, H.M. Influence of Electric Fields and pH on Biofilm Structure as Related to the Bioelectric Effect. Antimicrob. Agents Chemother. 1997, 41, 1876–1879. [Google Scholar] [CrossRef]
- Kim, Y.W.; Lee, J.; Han, S.K.; Koo, B.-S.; Park, T.; Park, H.M.; Lee, B. A Non-Electrolysis Bioelectric Effect for Gingivitis and Hygiene Contamination Biofilm Removal. Appl. Microbiol. 2023, 3, 675–686. [Google Scholar] [CrossRef]
- Park, H.M.; Chang, K.H.; Moon, S.H.; Park, B.J.; Yoo, S.K.; Nam, K.C. In vitro delivery efficiencies of nebulizers for different breathing patterns. Biomed. Eng. OnLine 2021, 20, 59. [Google Scholar] [CrossRef]
- Wisotzki, M.; Mair, A.; Schlett, P.; Lindner, B.; Oberhardt, M.; Bernhard, S. In Vitro Major Arterial Cardiovascular Simulator to Generate Benchmark Data Sets for In Silico Model Validation. Data 2022, 7, 145. [Google Scholar] [CrossRef]
- Szalma, J.; Lovasz, B.V.; Vajta, L.; Soos, B.; Lempel, E.; Mohlhenrich, S.C. The influence of the chosen in vitro bone simulation model on intraosseous temperatures and drilling times. Sci. Rep. 2019, 9, 11817. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, S.-H.; Han, J.-S.; Yeo, I.-S.L.; Yoon, H.-I. Optical and Surface Properties of Monolithic Zirconia after Simulated Toothbrushing. Materials 2019, 12, 1158. [Google Scholar] [CrossRef] [PubMed]
- Bizhang, M.; Schmidt, I.; Chun, Y.P.; Arnold, W.H.; Zimmer, S. Toothbrush abrasivity in a long-term simulation on human dentin depends on brushing mode and bristle arrangement. PLoS ONE 2017, 12, e0172060. [Google Scholar] [CrossRef] [PubMed]
- Ledder, R.G.; Latimer, J.; Forbes, S.; Penney, J.L.; Sreenivasan, P.K.; Mcbain, A.J. Visualization and quantification of the oral hygiene effects of brushing, dentifrice use, and brush wear using a tooth brushing stimulator. Front. Public Health 2019, 7, 1–7. [Google Scholar] [CrossRef]
- Lang, T.; Staufer, S.; Jennes, B.; Gaengler, P. Clinical validation of robot simulation of toothbrushing-comparative plaque removal efficacy. BMC Oral Health 2014, 14, 1–9. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Lee, S.G.; Go, S.J.; An, S.; Kim, Y.E.; Kim, Y.I.; Hyun, K.-Y.; Cho, D.S.; Choi, G.E. Anti-inflammatory Effects of Low-frequency Stimulator using Superposition of Alternating Microcurrent Wave in the Animal Models. Biomed. Sci. Letters 2021, 27, 99–104. [Google Scholar] [CrossRef]
- Hao, L.; Li, C.; Wang, L.; Bai, B.; Zhao, Y.; Luo, C. Design and Fabrication of a High-Temperature SOI Pressure Sensor with Optimized Crossbeam Membrane. Micromachines 2023, 14, 1045. [Google Scholar] [CrossRef] [PubMed]
- ISO TR 14569-1: dental materials-guidance on testing of wear resistance. Part 1: wear by tooth brushing.
- van Leeuwen, M.P.C.; van der Weijden, F.A.; Slot, D.E.; Rosema, M.A.M. Toothbrush wear in relation to toothbrushing effectiveness. Int. J. Dent. Hyg. 2019, 17, 77–84. [Google Scholar] [CrossRef] [PubMed]
- García-Godoy, F.; Villalta, P.; Bartizek, R.D.; Barker, M.L.; Biesbrock, A.R. Tooth whitening effects of an experimental power whitening toothbrush relative to an 8.7% hydrogen peroxide paint-on gel control. Am. J. Dent. 2004, 17, 25A–30A. [Google Scholar] [PubMed]
- Addy, M.; Jenkins, S.; Newcombe, R. The effect of triclosan, stannous fluoride and chlorhexidine products on:(I) Plaque regrowth over a 4-day period. J. Clin. Periodontol. 1990, 17, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Binney, A.; Addy, M.; McKeown, S.; Everatt, L. The effect of a commercially available triclosan-containing toothpaste compared to a sodium-fluoride-containing toothpaste and a chlorhexidine rinse on 4-day plaque regrowth. J. Clin. Periodontol. 1995, 22, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Sripriya, N.; and Shaik Hyder Ali, K.H. A comparative study of the efficacy of four different bristle designs of tooth brushes in plaque removal. J. Indian Soc. Pedod. Prev. Dent. 2007, 25, 76–81. [Google Scholar] [CrossRef]
- Pujarern, P.; Rodanant, P.; Warinsiriruk, E.; Rattanasuwan, K. Evaluation of the optimum brushing force on dental plaque removal: an in vitro study. M. Dent. J. 2021, 41, 75–82. [Google Scholar]
- Mankodi, S.; Berkowitz, H.; Durbin, K.; Nelson, B. Evaluation of the effects of brushing on the removal of dental plaque, J. Clin. Dent. 1998, 9, 57–60. [Google Scholar]
- Ozdemir, A.; Mas, M.R.; Sahin, S.; Sağlamkaya, U.; Ateşkan, U. Detection of Helicobacter pylori colonization in dental plaques and tongue scrapings of patients with chronic gastritis. Quintessence Int. 2001, 32, 131–134. [Google Scholar] [PubMed]
- Pérez-Serrano, R.M.; Domínguez-Pérez, R.A.; Ayala-Herrera, J.L.; Luna-Jaramillo, A.E.; De Larrea, G.Z.-L.; Solís-Sainz, J.C.; García-Solís, P.; Loyola-Rodríguez, J.P. Dental plaque microbiota of pet owners and their dogs as a shared source and reservoir of antimicrobial resistance genes. J. Glob. Antimicrob. Resist. 2020, 21, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Essalat, M.; Morrison, D.; Kak, S.; Chang, E.J.; Penso, I.R.; Kulchar, R.J.; Padilla, O.H.M.; Shetty, V. A Naturalistic Study of Brushing Patterns Using Powered Toothbrushes. PLoS ONE 2022, 17, e0263638. [Google Scholar] [CrossRef] [PubMed]
- Collins, L.M.; Dawes, C. The surface area of the adult human mouth and thickness of the salivary film covering the teeth and oral mucosa. J. Dent. Res. 1987, 66, 1300–1302. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).