3. Results
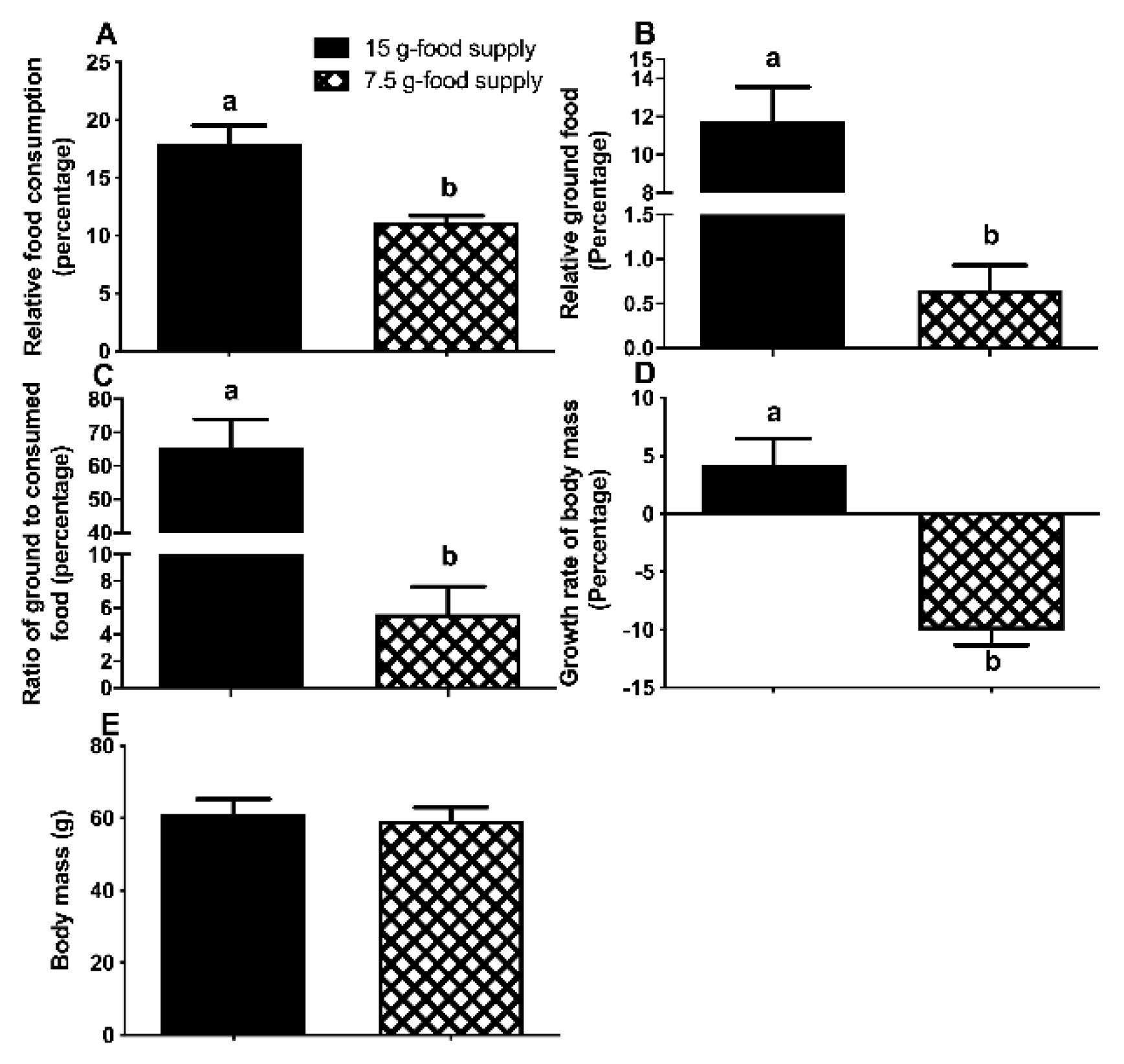
The relative food consumption and relative amount of ground food, the ratio of ground to consumed food, and the body mass growth rate significantly decreased for the high-level food-grinding voles when the food supply was reduced from 15 to 7.5 g (
P = 0.028, for all cases;
Figure 1A-C, E). However, body weight did not sharply differ between the 15- and 7.5 g-food supply groups (
P = 0.116;
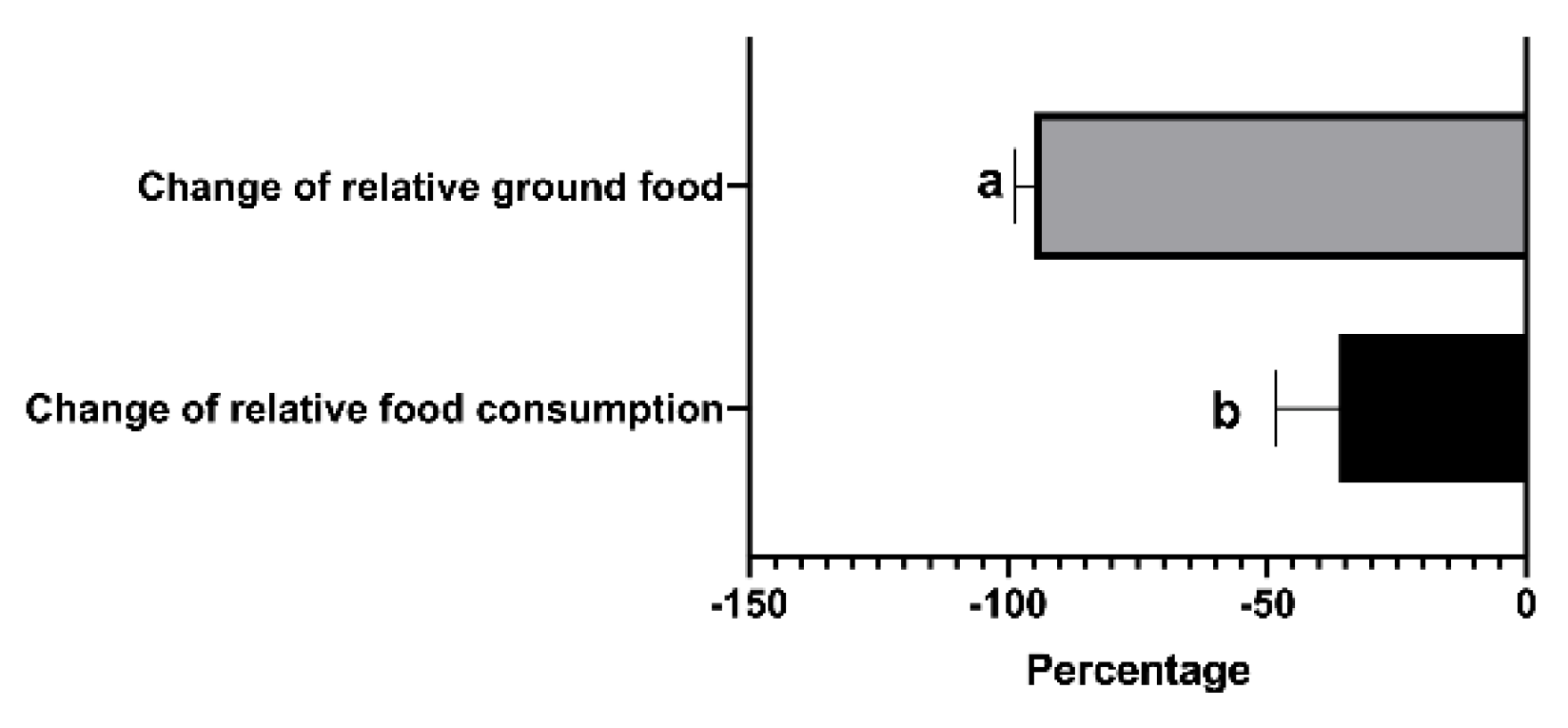
Figure 1D). The change in relative ground food was greater than the change in the relative food consumption (
P = 0.028) when the food supply was reduced from 15 to 7.5 g (
Figure 2).
In both the 15- and 7.5 g-food supply groups, the values of the average Goods coverage were as high as 99.75%, indicating that the16S rDNA sequencing method could identify the vast majority of taxa among the microbiota present in the fecal samples (
Table 1). The numbers of total tags and taxon tags for the 7.5 g-food supply group were not significantly different from those for the 15 g-food supply group (
P = 0.116 and 0.917, respectively;
Table 1). However, the number of OTUs for the 15 g-food supply group was significantly lower than that for the 7.5 g-food supply group (
P = 0.028;
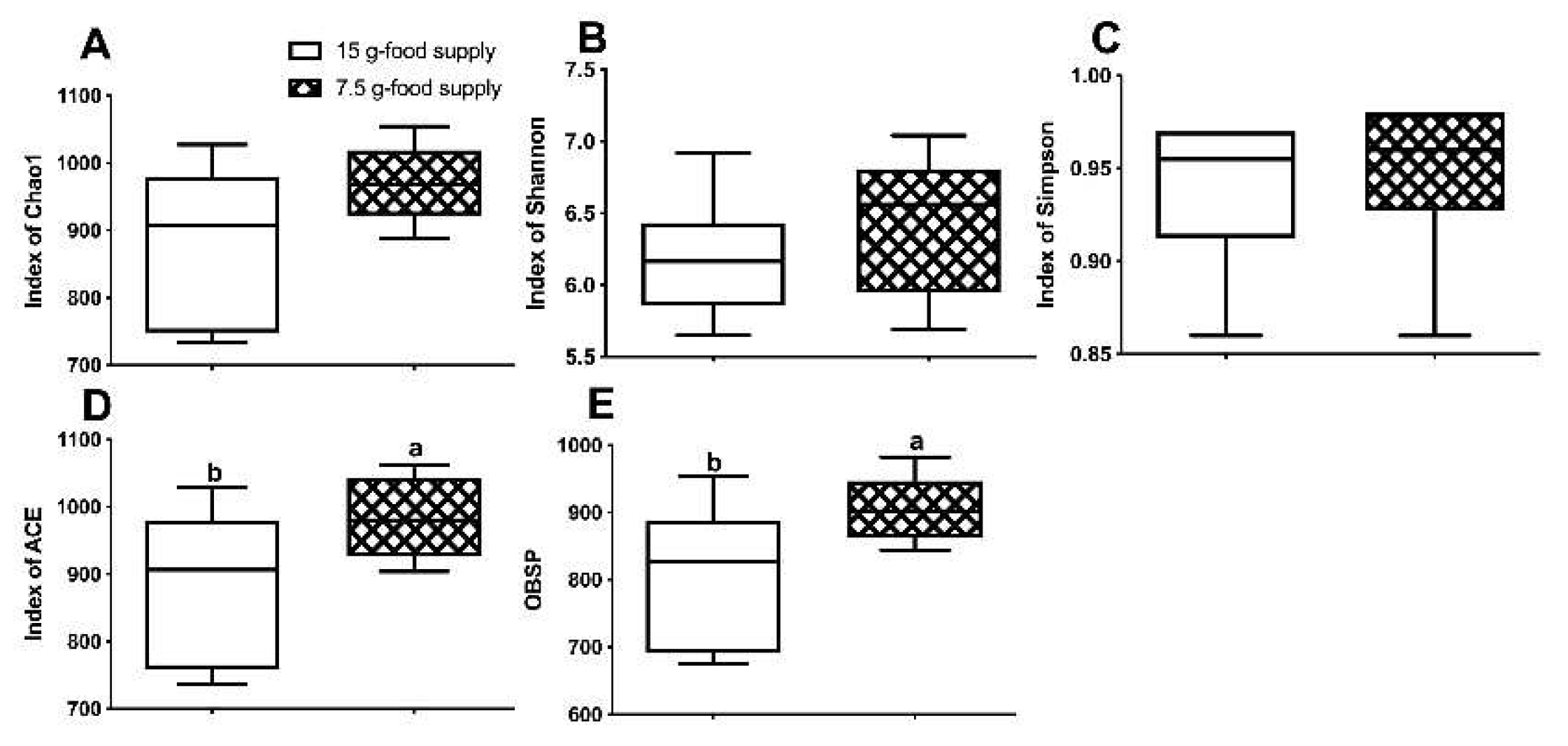
Table 1). Although no obvious differences between the two food supply groups were detected with respect to the Chao1 (
P = 0.116), Shannon (
P = 0.249), or Simpson (
P = 0.753) indices (
Figure 3A, D-E), the ACE index (
P = 0.046) and OBSP values (
P = 0.028) (
Figure 3B-C) for the 15 g-food supply group were lower than those for the 7.5 g-food supply group. The beta diversity of the fecal microbiota did not sharply vary after the food supply was reduced to 7.5 g (
F = 1.070,
P = 0.34).
Figure 1.
Differences of relative food consumption (A), relative ground food (B), food ground to consumption ratio (C), body mass (D), and growth rate of body mass (E) between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 1.
Differences of relative food consumption (A), relative ground food (B), food ground to consumption ratio (C), body mass (D), and growth rate of body mass (E) between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
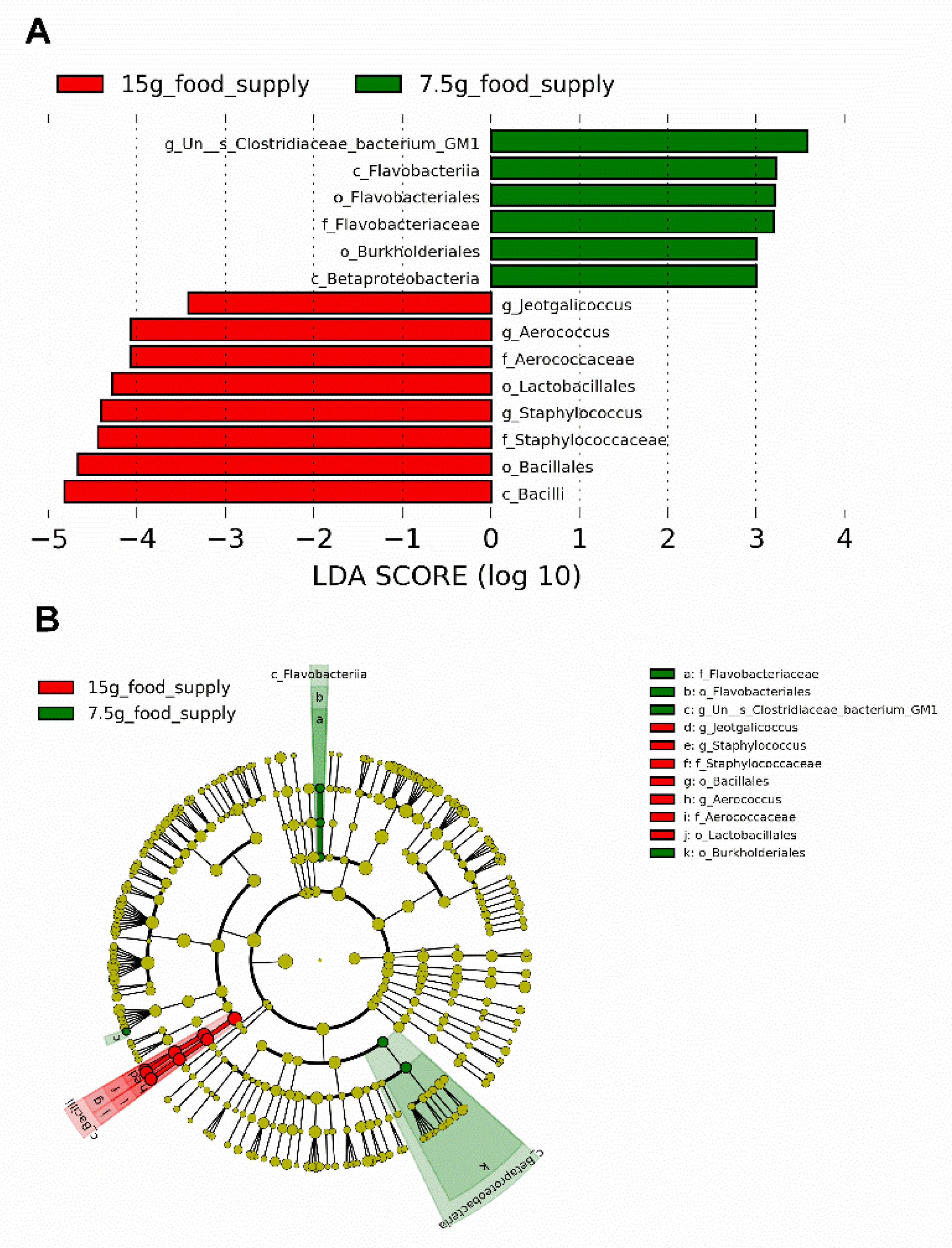
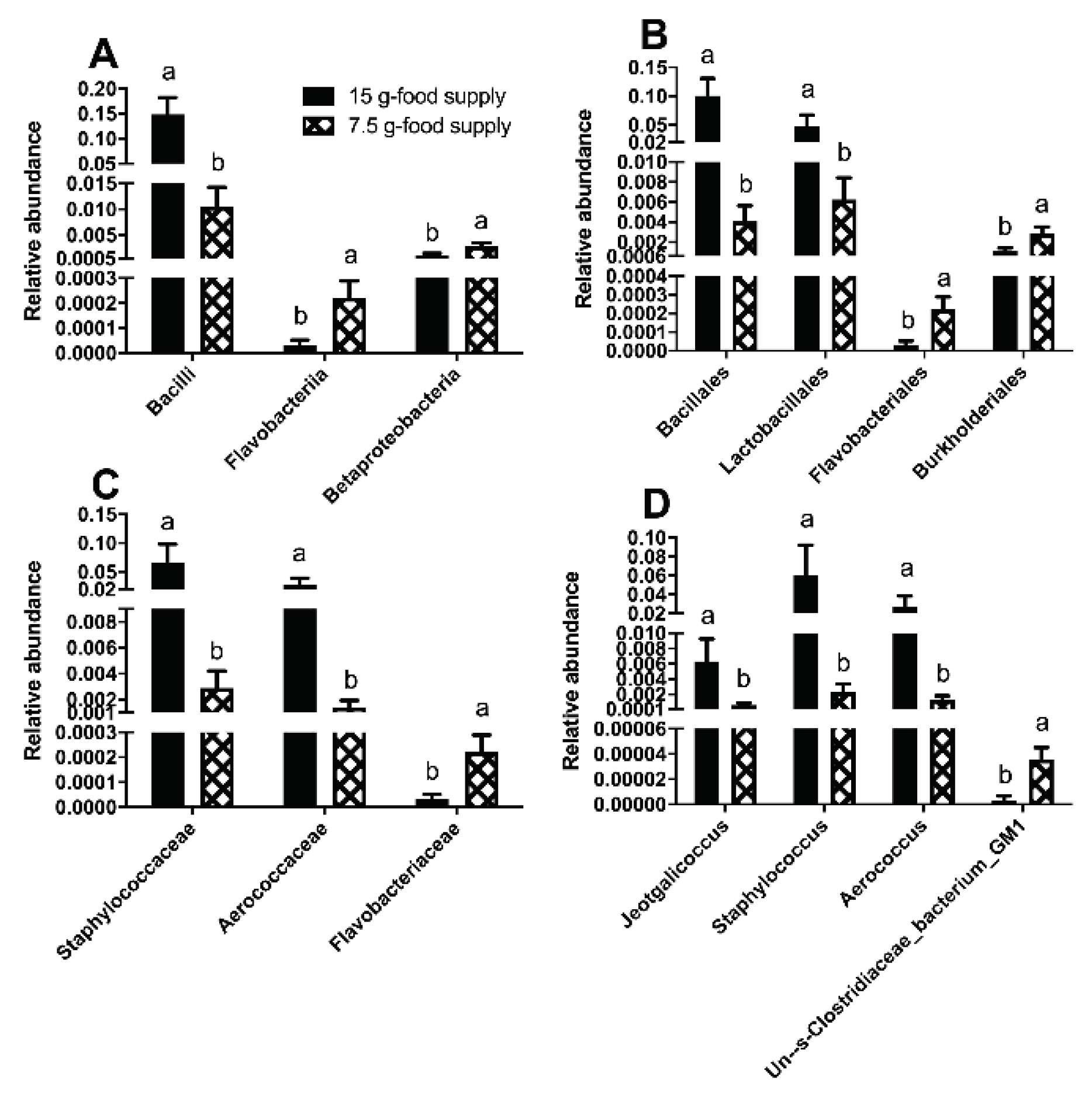
The biomarkers for the Bacilli class, Bacillales and Lactobacillales orders, Staphylococcaceae and Aerococcaceae families, and
Staphylococcus,
Aerococcus, and
Jeotgalicoccus genera (
Figure 4A-B) were enriched for the 15 g-food supply group. The biomarkers for the Flavobacteriia and Betaproteobacteria classes, Flavobacteriales and Burkholderiales orders, Flavobacteriaceae family, and
Un--s-Clostridiaceae bacterium GM1 genus (
Figure 4A-B) were enriched for the 7.5 g-food supply group. The relative abundances of Bacilli (
P = 0.028), Bacillales (
P = 0.028), Lactobacillales (
P = 0.046), Staphylococcaceae (
P = 0.028), Aerococcaceae (
P = 0.028),
Staphylococcus (
P = 0.028),
Aerococcus (
P = 0.028), and
Jeotgalicoccus (
P = 0.028) for the 15 g-food supply group were all higher than those for the 7.5 g-food supply group (
Figure 5A-D). Moreover, the relative abundances of Flavobacteriia (
P = 0.028), Betaproteobacteria (
P = 0.028), Flavobacteriales (
P = 0.028), Burkholderiales (
P = 0.028), Flavobacteriaceae (
P = 0.028), and
Un--s-Clostridiaceae bacterium GM1 (
P = 0.039) were all higher for the 7.5 g-food supply group than for the 15 g-food supply group (
Figure 5A-D).
Figure 2.
Differences between the changes of relative food consumption and relative ground food in Brandt’s vole when food supply reduced from 15 g to 7.5 g. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 2.
Differences between the changes of relative food consumption and relative ground food in Brandt’s vole when food supply reduced from 15 g to 7.5 g. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 3.
Differences in the fecal microbiota composition between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. (A) Chao1; (B) abundance-based coverage estimator (ACE); (C) observed species (OBSP); (D) Shannon index; (E) Simpson index. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 3.
Differences in the fecal microbiota composition between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. (A) Chao1; (B) abundance-based coverage estimator (ACE); (C) observed species (OBSP); (D) Shannon index; (E) Simpson index. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
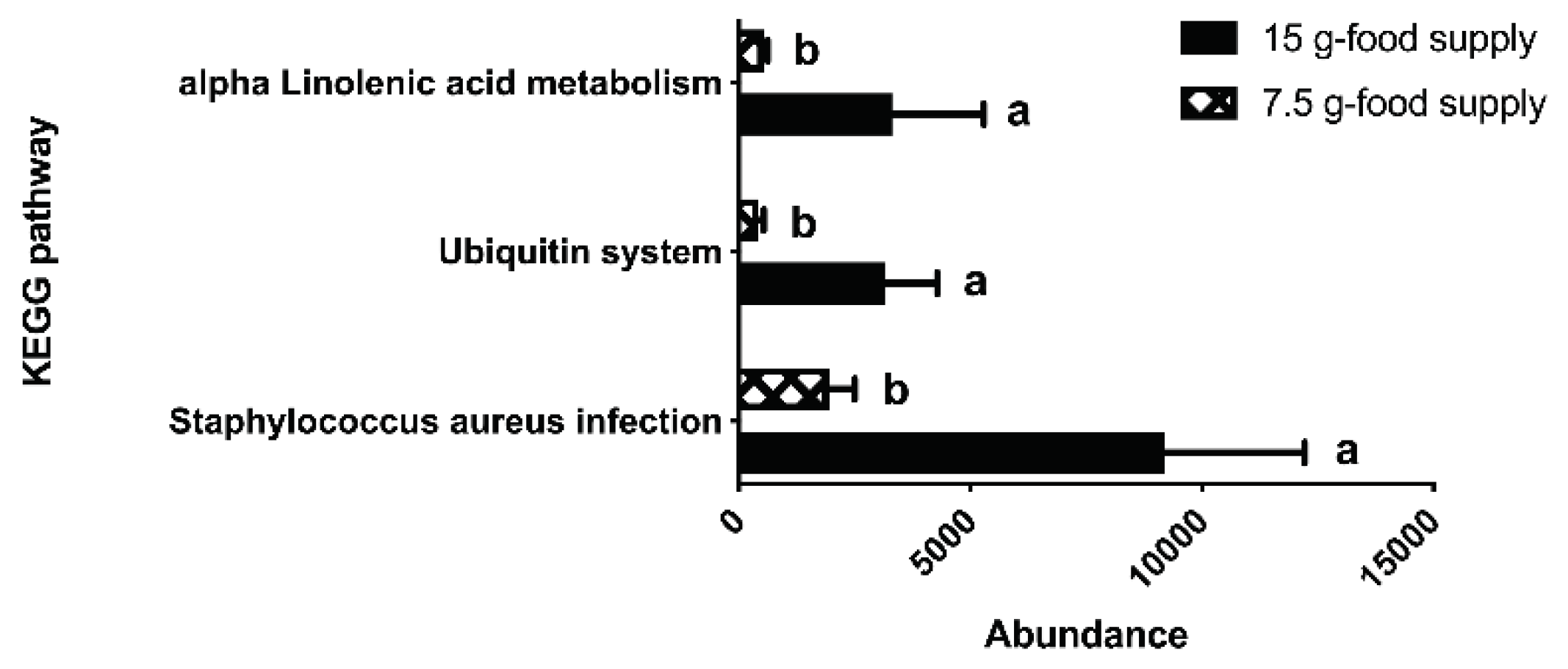
The KEGG pathways associated with
Staphylococcus aureus infection, the ubiquitin system, and alpha-linolenic acid metabolism (
P = 0.046, 0.028, and 0.028, respectively) were more enriched for the 15 g-food supply group compared to that for the 7.5 g-food supply group (
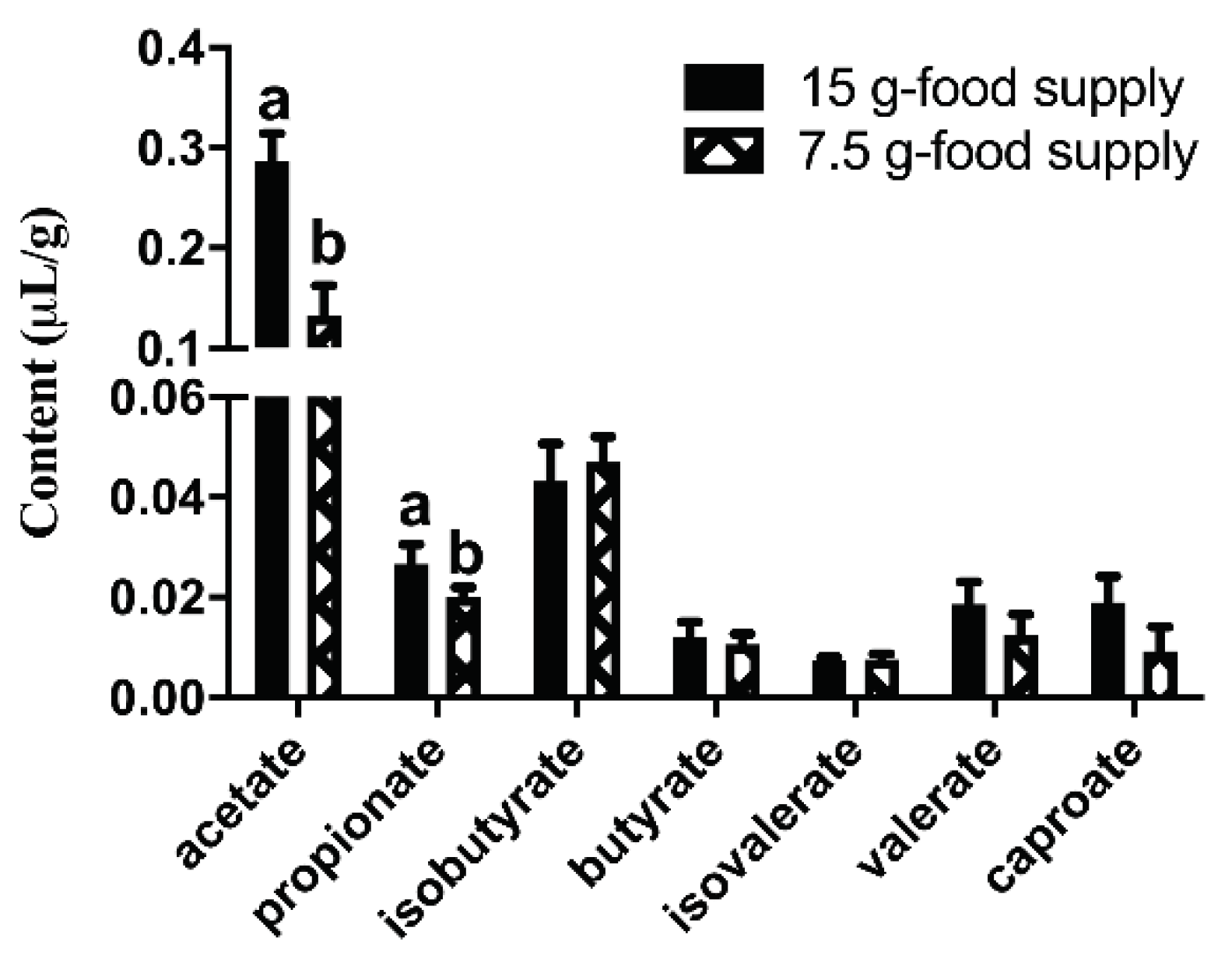
Figure 6). Further, the acetate and propionate content were lower for the 7.5 g-food supply group than for the 15 g-food supply group (
P = 0.028 and 0.042, respectively;
Figure 7). Meanwhile, the isobutyrate, butyrate, isovalerate, valerate, and caproate content did not significantly differ between the 15 g- and 7.5 g-food supply groups (
P = 0.600, 0.416, 0.581, 0.172, and 0.173, respectively;
Figure 7).
Figure 4.
Biomarker of taxa with statistically significant differences in relative abundance selected in the fecal microbiota between the 15 g- and 7.5 g-food supply groups of Brandt’s voles (n = 6). (A) Histogram displaying the differential taxa using the linear discriminant analysis (LDA) effect size (LEfSe) analysis with a LDA score significant threshold > 3; (B) Cladogram displaying the phylogenetic position of differential taxa.
Figure 4.
Biomarker of taxa with statistically significant differences in relative abundance selected in the fecal microbiota between the 15 g- and 7.5 g-food supply groups of Brandt’s voles (n = 6). (A) Histogram displaying the differential taxa using the linear discriminant analysis (LDA) effect size (LEfSe) analysis with a LDA score significant threshold > 3; (B) Cladogram displaying the phylogenetic position of differential taxa.
Figure 5.
Differences in taxa relative abundances of fecal microbiota at different taxonomic levels between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. (A) class level; (B) order level; (C) family level; (D) genus level. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 5.
Differences in taxa relative abundances of fecal microbiota at different taxonomic levels between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. (A) class level; (B) order level; (C) family level; (D) genus level. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 6.
Differences in the abundances of fecal microbial KEGG pathways between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 6.
Differences in the abundances of fecal microbial KEGG pathways between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 7.
Difference of fecal short-chain fatty acids (SCFAs: acetate, propionate, isobutyrate, butyrate, isovalerate, valerate, and caproate) content between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
Figure 7.
Difference of fecal short-chain fatty acids (SCFAs: acetate, propionate, isobutyrate, butyrate, isovalerate, valerate, and caproate) content between the 15 g- and 7.5 g-food supply groups of Brandt’s voles. Error bars indicate standard errors. Different letters connect bars with significant differences at P < 0.05 (n = 6).
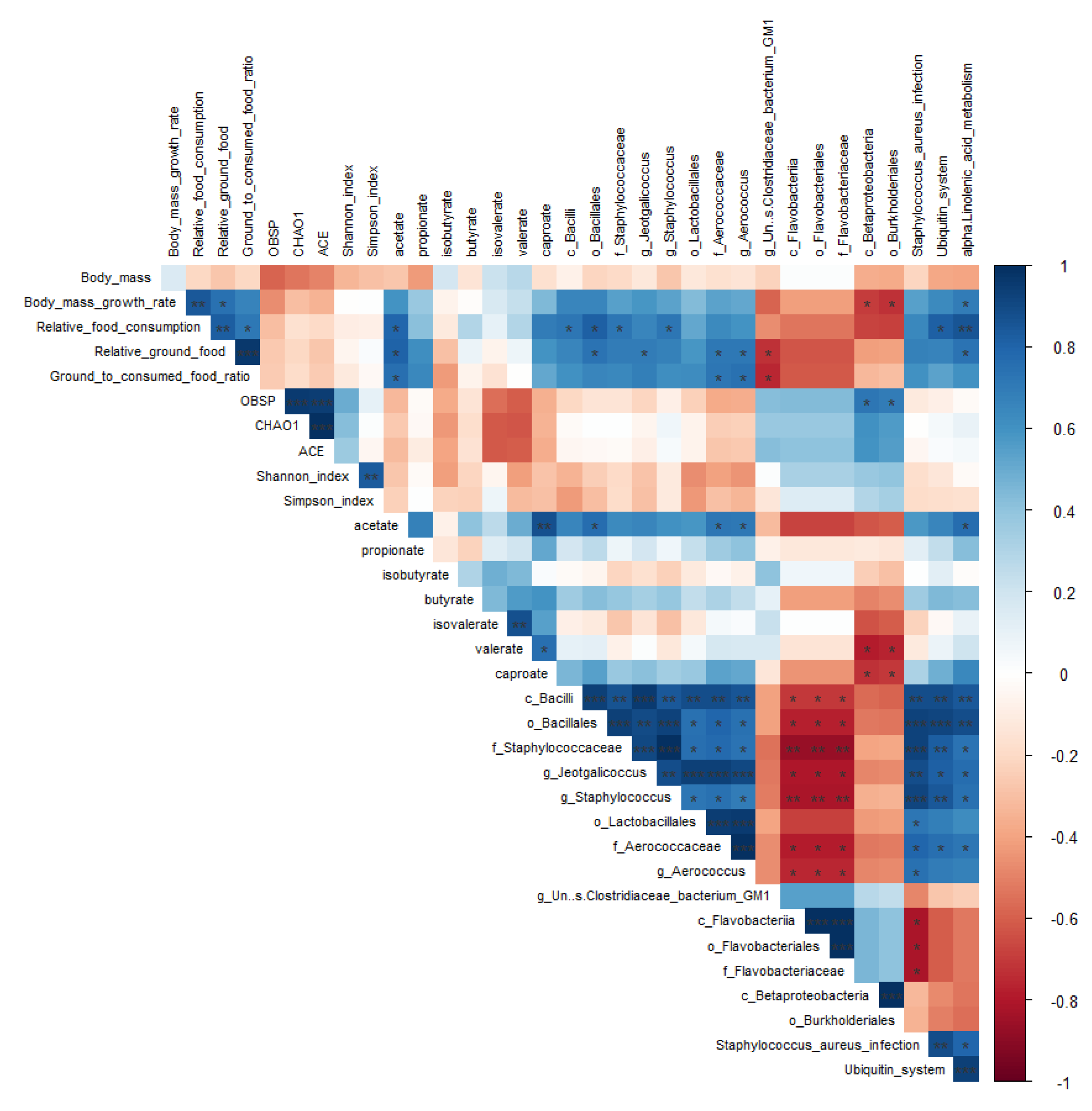
The body mass growth rate was positively correlated with relative food consumption, relative amount of ground food, and alpha-linolenic acid metabolism (
P = 0.004, 0.022, and 0.037, respectively), and negatively correlated with the relative abundance of the Betaproteobacteria class and Burkholderiales order (
P = 0.037 and 0.028, respectively;
Figure 8). Relative food consumption was positively correlated with the relative amount of ground food, ground-to-consumed food ratio, acetate and caproate contents, relative abundances of the Bacilli class, Bacillales order, Staphylococcaceae family,
Staphylococcus genus, and pathway enrichment of the ubiquitin system and alpha-linolenic acid metabolism (
P = 0.008, 0.041, 0.015, 0.050, 0.038, 0.011, 0.038, 0.035, 0.011, and 0.004, respectively;
Figure 8). Relative ground food was positively correlated with the ground-to-consumed food ratio (
P < 0.001), acetate content, relative abundances of the Bacillales order, Aerococcaceae family,
Aerococcus and
Jeotgalicoccus genera, and pathway enrichment of alpha-linolenic acid metabolism (
P = 0.011, 0.033, 0.039, 0.042, 0.049, and 0.034, respectively; Figure 8), but negatively correlated with the relative abundance of
Un-s-Clostridiaceae bacterium GM1 (
P = 0.038). The ratio of ground to consumed food was positively correlated with acetate content and the relative abundances of the Aerococcaceae family and
Aerococcus genus (
P = 0.026, 0.035, and 0.027, respectively), but negatively correlated with the
Un-s-Clostridiaceae bacterium GM1 genus (
P = 0.027) (
Figure 8). The OBSP was positively correlated with the relative abundances of Betaproteobacteria and Burkholderiales (
P = 0.035 and 0.048, respectively; Figure 8). Meanwhile, acetate content was positively correlated with the relative abundances of the Bacillales order, Aerococcaceae family, and
Aerococcus genus (
P = 0.021, 0.038, and 0.041, respectively), and pathway enrichment of alpha-linolenic acid metabolism (
P = 0.024) (Figure 8). Moreover, the relative abundances of the Betaproteobacteria class and Burkholderiales order were negatively correlated with the valerate (
P = 0.013 and 0.017, respectively) and caproate (
P = 0.030 and 0.037, respectively) content (
Figure 8), whereas the acetate (
P = 0.002) and valerate (
P = 0.025) content was positively correlated with the caproate content. Correlations among the abundances of microbiota biomarkers were either negative or positive (
Figure 8). Pathway enrichment related to
S. aureus infection, the ubiquitin system, and alpha-linolenic acid metabolism was also positively or negatively correlated with biomarkers of the microbiota (
Figure 8).
4. Discussion
In this study, we investigated the effect of food supply changes on food grinding in male Brandt’s voles and its potential underlying mechanisms with respect to the gut microbiota. When the food supply for Brandt’s voles was restricted to 7.5 g, which was below the average daily food intake, the body mass growth rate, food consumption, amount of ground food, and the ground-to-consumed food ratio decreased. Some alpha diversity indices and the abundances of some fecal microbiota, along with the content of some SCFAs, varied; however, the fecal microbial structure did not change after reducing the food supply. Although body mass did not differ remarkably between the 15- and 7.5 g-supply groups when the food supply was reduced to 7.5 g, food consumption decreased, and the body mass growth rate was negative and significantly lower than that observed when the food supply was 15 g. A positive correlation was detected between the relative food consumption and body mass growth rate. This indicates that food restriction (7.5 g) can reduce food consumption and restrict vole growth. The ratio of ground to consumed food also decreased sharply, consistent with the higher amplitude of variation in relative ground food than in relative food consumption, indicating that the quantity of ground food decreased more than food consumption under a reduced food supply. Ort production in mice decreases to zero when they are offered 80% of their food intake [
1]. Therefore, we concluded that the food-grinding behavior could be influenced by the quantity of food supplied. This suggests that when vegetation in grasslands is plentiful, food waste by wild Brandt’s voles is more extensive. Moreover, the inhibitive effect of food restrictions on food grinding could be explained by the hypothesis of an optimal food intake strategy [
19] with food components having high energy per gram selected through grinding, to render the energy intake as large as possible [
1]. When the food supply was reduced to less than the average food intake and the body mass growth rate was negative, food grinding would further reduce the available food to levels which could not meet the daily energy demand. Additionally, food grinding is an energy-cost behavior. Thus, no advantage would be gained regarding food selectivity or food grinding. Therefore, voles would reduce their food-grinding behavior and ingest as much food as possible to maximize energy intake and save energy. In our previous study, no significant correlation was detected between food consumption and relative ground food [
9], whereas in this study, a positive correlation was observed. This inconsistency might be attributed to the negative effects of food restriction on food consumption and grinding. Relative ground food was positively correlated with the body mass growth rate, and the ground-to-consumed food ratio was nearly significantly and positively correlated with this parameter (
P = 0.057), suggesting that food-grinding behavior might have other beneficial effects for voles, other than maximizing energy intake, which requires further research. The reason behind the body weight not being significantly different between the 15- and 7.5 g-food supply groups is that for the 15 g-food supply group, the body weights of voles increased from day 1 to day 14, whereas for the 7.5 g-food supply group, the body weights decreased from day 1 to day 14, and the food restriction experiment was conducted immediately following the 15 g-food supply experiment. Thus, the mean body weights were not obviously different between the 15- and 7.5 g-food supply groups. This also resulted in the lack of a pronounced correlation between the body weight and ground food or the ground-to-consumed food ratio.
In this study, the ACE index of the gut microbiota was high when the food supply was decreased, whereas the Shannon and Simpson indices did not change significantly, which is consistent with previously observed results for Chinese individuals after fasting [
20]. Increases in the numbers of OTUs as well as the OBSP and ACE indices, suggested that food restriction enhances the number of species in the gut microbiota of Brandt’s voles. The increase in the number of gut bacterial species could be due to the enrichment of biomarkers for Betaproteobacteria and Burkholderiales caused by the positive correlations between OBSP and the relative abundances of these taxa. The acetate content was also lower for the 7.5 g-food supply group than for the 15 g-food supply group and was positively correlated with the ground food and ground-to-consumed food ratio, supporting our speculation that acetate could promote food-grinding behavior [
9]. Therefore, the decreased acetate content might have restrained food-grinding behavior in this study. Assessing the ratio of ground to consumed food eliminated the influence of food intake on ground food. Given the strong negative or positive correlations between the ground-to-consumed food ratio and the
Un--s-Clostridiaceae bacterium GM1 and
Aerococcus genera between the 15- and 7.5 g-food supply groups, it is likely that these two genera could contribute to the regulation of food-grinding behavior, with the genus
Un--s-Clostridiaceae bacterium GM1 inhibiting and the genus
Aerococcus promoting it. The genera
Aerococcus and
Un--s-Clostridiaceae bacterium GM1 were the common biomarkers observed, based on two LEfSe analyses, that could differentiate the high- and low-level food-grinding groups in our previous study [
9], as well as the 15- and 7.5 g-food supply groups in this study. This indicates a strong relationship between the
Aerococcus or
Un--s-Clostridiaceae bacterium GM1 genera and food-grinding behavior, which supports our previous speculation that gut microbiota participate in the regulation of food-grinding behavior [
9]. The positive correlation between
Aerococcus and the acetate content indicates that the effect of this genus on food-grinding behavior might occur through alterations to the acetate content. Therefore, we inferred that changes in the abundance of the gut microbiome components and their metabolite SCFAs due to food restriction would be helpful in inhibiting food grinding in Brandt’s voles.
An increase in
S. aureus has been observed for obese individuals [
21]. Further, reductions in the body weight,
Staphylococcus abundance in the rectum, and Staphylococcaceae levels in the cecum have been observed for Kunming mice [
22]. In this study, the abundance of the genus
Staphylococcus, along with an increase in the body mass, was consistently higher for the 15 g-food supply group. Staphylococcaceae species are pathogens [
23], and
S. aureus infects a variety of tissues, organs, and systems in humans [
24,
25,
26]. The pathway associated with
S. aureus infection was more enriched for the 15 g-food supply group than for the 7.5 g-food supply group owing to the abundance of the genus
Staphylococcus and the family Staphylococcaceae. Therefore, it is likely that food restriction can help reduce the abundance of pathogenic Staphylococcaceae species. Alpha-linolenic acid is an antibacterial substance that inhibits
S. aureus [
27,
28]. Moreover, autophagy, mediated by ubiquitin receptors, is essential for zebrafish resistance to
S. aureus [
29]. Therefore, the enrichment of pathways related to the ubiquitin system and alpha-linolenic acid metabolism for the 15 g-food supply group could be associated with resistance to
Staphylococcus bacteria. In Rex rabbits with an increased weight, the concomitant enrichment of pathways related to alpha-linolenic acid metabolism, and increased
S. aureus colonization among the fecal microbiota was observed [
30]. Further, strongly negative correlations between the abundances of taxa within Bacilli and Flavobacteriia indicate that these bacterial classes might restrain each other in Brandt’s voles.
In this study, differences in the amount of ground food, the ground-to-consumed food ratio, the abundances and diversity of fecal microbiota components, and the metabolite content between voles with different food supplies were observed. Based on these findings, we speculated on the effects of food restriction on food-grinding behavior, as well as the correlation between the gut microbiota, in addition to their metabolites, and food-grinding behavior. This study, along with our previous research [
9], preliminarily clarifies the factors influencing food-grinding behavior and the mechanisms underlying this characteristic from the perspective of the gut microbiota. However, we could not distinguish whether the direct effect of food restriction, owing to the insufficient energy supply, or indirect effect of food restriction, via the gut microbiota, on food-grinding behavior was predominantly important. Food restriction in association with gut microbiota transplantation experiments should help to address this ambiguity. Additionally, the precise gut microbiota-associated mechanisms underlying food-grinding behavior and the functions of food-grinding behavior in voles require further research.