Submitted:
04 October 2023
Posted:
05 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sampling locations and geological background
2.2. Polarised light microscopy (PLM)
2.3. Crushing
2.4. X-Ray Diffraction (XRD)
2.5. X-Ray Fluorescence (XRF)
2.6. BET-N2 specific surface area determination
2.7. Bio-durability tests
3. Results
3.1. Bulk material description
3.2. Polarised light microscopy (PLM)
3.3. X-Ray Diffraction (XRD)
3.4. X-Ray Fluorescence (XRF) major and trace elemental analysis
3.5. BET-N2 specific surface area
3.6. Bio-durability tests
4. Discussion
4.1. Mineralogical impact
4.2. Geochemical impact
4.3. Geographic impact and rehabilitation
5. Conclusions
Supplementary Materials
Conflicts of Interest
References
- Cornelissen, H.; Watson, I.; Adam, E.; Malefetse, T. Challenges and strategies of abandoned mine rehabilitation in South Africa: The case of asbestos mine rehabilitation. J. Geochem. Explor. 2019, 205. [Google Scholar] [CrossRef]
- Harris, L.V.; Kahwa, I.A. Asbestos: old foe in 21st century developing countries. Sci. Total. Environ. 2003, 307, 1–9. [Google Scholar] [CrossRef]
- Ndlovu, N.; Naude, J.T.; Murray, J. Compensation for environmental asbestos-related diseases in South Africa: a neglected issue. Glob. Heal. Action 2013, 6, 19410. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Dell, L.; Adams, R.; Rose, T.; Van Orden, D.R. State-of-the-science assessment of non-asbestos amphibole exposure: Is there a cancer risk? Environmental Geochemistry and Health 2013, 35, 357–377. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.A. Nature and paragenesis of asbestos minerals. Philosophical Transactions Royal Society of London A, 1977, 286, 611–624. [Google Scholar]
- Okayasu, R.; Takahashi, S.; Yamada, S.; Hei, T.K.; Ullrich, R.L. Asbestos and DNA double strand breaks. . 1999, 59, 298–300. [Google Scholar]
- Kilburn, K.H. Indoor air effects after building renovation and in manufactured homes. The American Journal of Medical Sciences 2000, 320(4), 249–54. [Google Scholar] [CrossRef]
- Unfried, K.; Schürkes, C.; Abel, J. Distinct spectrum of mutations induced by crocidolite asbestos: clue for 8-hydroxydeoxyguanosine-dependent mutagenesis in vivo. . 2002, 62, 99–104. [Google Scholar]
- Mossman, B.T.; Lippmann, M.; Hesterberg, T.W.; Kelsey, K.T.; Barchowsky, A.; Bonner, J.C. Pulmonary Endpoints (Lung Carcinomas and Asbestosis) Following Inhalation Exposure to Asbestos. J. Toxicol. Environ. Heal. Part B 2011, 14, 76–121. [Google Scholar] [CrossRef]
- Pugnaloni, A.; Giantomassi, F.; Lucarini, G.; Capella, S.; Bloise, A.; Di Primio, R.; Belluso, E. Cytotoxicity induced by exposure to natural and synthetic tremolite asbestos: An in vitro pilot study. Acta Histochem. 2013, 115, 100–112. [Google Scholar] [CrossRef]
- Bloise, A.; Barca, D.; Gualtieri, A.F.; Pollastri, S.; Belluso, E. Trace elements in hazardous mineral fibres. Environ. Pollut. 2016, 216, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Bloise, A.; Punturo, R.; Catalano, M.; Miriello, D.; Cirrincione, R. Naturally occurring asbestos (NOA) in rock and soil and relation with human activities: the monitoring example of selected sites in Calabria (southern Italy). Ital. J. Geosci. 2016, 135, 268–279. [Google Scholar] [CrossRef]
- Harper, M. 10th Anniversary Critical Review: Naturally occurring asbestos. J. Environ. Monit. 2008, 10, 1394–1408. [Google Scholar] [CrossRef] [PubMed]
- Culley, M.R.; Zorland, J.; Freire, K. Community responses to naturally occurring asbestos: implications for public health practice. Heal. Educ. Res. 2010, 25, 877–891. [Google Scholar] [CrossRef]
- Bloise, A.; Belluso, E.; Critelli, T.; Catalano, M.; Apollaro, C. ; Miriello, D; Barrese, E. Amphibole asbestos and other fibrous minerals in the meta-basalt of the gimigliano-mount reventino unit (Calabria, South-Italy). Rendiconti online della Società Geologica Italiana 2012, 21, 847–848. [Google Scholar]
- Bloise, A.; Critelli, T.; Catalano, M.; Apollaro, C.; Miriello, D.; Croce, A.; Barrese, E.; Liberi, F.; Piluso, E.; Rinaudo, C.; et al. Asbestos and other fibrous minerals contained in the serpentinites of the Gimigliano-Mount Reventino Unit (Calabria, S-Italy). Environ. Earth Sci. 2014, 71, 3773–3786. [Google Scholar] [CrossRef]
- Punturo, R.; Ricchiuti, C.; Mengel, K.; Apollaro, C.; De Rosa, R.; Bloise, A. Serpentinite-derived soils in southern Italy: potential for hazardous exposure. J. Mediterr. Earth Sci 2018, 10, 51–61. [Google Scholar]
- Apollaro, C.; Fuoco, I.; Vespasiano, G.; De Rosa, R.; Cofone, F.; Miriello, D.; Bloise, A. Geochemical and mineralogical characterization of tremolite asbestos contained in the Gimigliano-Monte Reventino Unit (Calabria, south Italy). J. Mediterr. Earth Sci 2018, 10, 5–15. [Google Scholar]
- Braun, L. and Kisting, S. Asbestos-Related Disease in South Africa: The Social Production of an Invisible Epidemic. American Journal of Public Health 2006, 96 (8), 1386 – 1396.
- Rees, D., Myers, J.E., Goodman, K., Fourie, E., Blignaut, C., Chapman, R., Bachmann, M.O. Case-Control Study of Mesothelioma in South Africa. American journal of industrial medicine. 1999, 35, 213–222.
- Ramazzini, C. The global health dimensions of asbestos and asbestos-related diseases. J. Occup. Heal. 2016, 58, 220–223. [Google Scholar] [CrossRef]
- Gualtieri, A.F.; Viani, A.; Sgarbi, G.; Lusvardi, G. In vitro biodurability of the product of thermal transformation of cement–asbestos. J. Hazard. Mater. 2012, 205-206, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Fubini, B.; Areán, C.O. Chemical aspects of the toxicity of inhaled mineral dusts. Chem. Soc. Rev. 1999, 28, 373–381. [Google Scholar] [CrossRef]
- Fubini, B.; Bolis, V.; Cavenago, A.; Volante, M. Physicochemical properties of crystalline silica dusts and their possible implication in various biological responses. Scandinavian Journal of Work, Environment and Health 1995, 9–14.
- Higueras, P.; Oyarzun, R.; Iraizoz, J.M.; Lorenzo, S.; Esbrí, J.M.; Martinezcoronado, A. Low-cost geochemical surveys for environmental studies in developing countries: Testing a field portable XRF instrument under quasi-realistic conditions. J. Geochem. Explor. 2012, 113, 3–12. [Google Scholar] [CrossRef]
- Anhaeusser, C.R. The nature of chrysotile asbestos occurrences in southern Africa; a review. Econ. Geol. 1976, 71, 96–116. [Google Scholar] [CrossRef]
- Hunter, D.R.; Jones, D.H. (1969): Geological Map Series (1:25,000). Sheet 2, Piggs Peak: Swaziland Geol. Survey.
- Viljoen, R.P.; Viljoen, M. J. The geology and geochemistry of the layered ultramafic bodies of the Kaapmuiden area, Barberton Mountain Land. Geological Society of South Africa Special Publication 1969, 1, 661–688. [Google Scholar]
- Van Biljon, (1964): The chrysotile deposits of the eastern Transvaal and Swaziland, in Haughton, S. H., ed., The geology of some ore deposits in southern Africa: Johannesburg. Geological Society of South Africa, p. 625-669.
- Abbott, P. A review of asbestos resources. Dissertation, Master of Science (Mineral Exploration) Degree, Rhodes University, Grahamstown, South Africa, January 1983. 19 January.
- Hart, H.P. Asbestos in South Africa. Journal – South African Institute of Mining and Metallurgy 1988, 88 (6), 185-198.
- Riordon, P.H. The genesis of asbestos in ultrabasic rocks [Quebec]. Econ. Geol. 1955, 50, 67–81. [Google Scholar] [CrossRef]
- Cairncross, B. (2004): Field Guide to Rocks and Minerals of Southern Africa. Struik Nature, Cape Town, South Africa.
- Dreyer, C.J.B. and Sohnge, A.P.G. (1992): The Crocidolite and Amosite Deposits of the Republic of South Africa and Bophuthatswana. Pretoria: Geological Survey of South Africa, Handbook, 12, 126 pp.
- Miyano, T.; Beukes, N.J. Mineralogy and Petrology of the Contact Metamorphosed Amphibole Asbestos-bearing Penge Iron Formation, Eastern Transvaal, South Africa. Journal of Petrology 1997, 38 (5), 651 – 676.
- Genis, J.H. (1961): The genesis of the blue amphibole asbestos of the union of South Africa. PhD thesis, University of Cape Town.
- Reinecke, L.; McClure, L. Variations in the quality of amosite asbestos at Penge, Transvaal. Transactions of the geological society of South Africa 1933, 36 (1), 29 – 39.
- Howlling, G.E. (1937): Asbestos. London: Imperial Institute. Pp. 88.
- Strohmeier, B.R.; Huntington, J.C.; Bunker, K.L.; Sanchez, M.S.; Allison, K.; Lee, R.J. What is asbestos and why is it important? Challenges of defining and characterizing asbestos. Int. Geol. Rev. 2010, 52, 801–872. [Google Scholar] [CrossRef]
- Plumlee, G.S.; Morman, S.A.; Ziegler, T.L. The toxicological geochemistry of earth minerals: An overview of processes and the interdisciplinary methods used to understand them. Reviews in Mineralogy and Geochemistry 2006, 64, 5–57. [Google Scholar] [CrossRef]
- Ilgren, E. Review: The Fiber Length of Coalinga Chrysotile: Enhanced Clearance due to Its Short Nature in Aqueous Solution with a Brief Critique on ``Short Fiber Toxicity''. Indoor Built Environ. 2008, 17, 5–26. [Google Scholar] [CrossRef]
- Norrish, K.; Hutton, J.T. An accurate X-ray spectrographic method for the analysis of geologic samples. Geochemica et Cosmochimica Acta 1969, 33, 431–454. [Google Scholar] [CrossRef]
- Wilson, A.H. A Chill Sequence to the Bushveld Complex: Insight into the First Stage of Emplacement and Implications for the Parental Magmas. J. Pet. 2012, 53, 1123–1168. [Google Scholar] [CrossRef]
- Bismarck, A.; Aranberri-Askargorta, I.; Springer, J.; Lampke, T.; Wielage, B.; Stamboulis, A.; Shenderovich, I.; Limbach, H.-H. Surface characterization of flax, hemp and cellulose fibers; Surface properties and the water uptake behavior. Polym. Compos. 2002, 23, 872–894. [Google Scholar] [CrossRef]
- Boulanger, G.; Andujar, P.; Pairon, J.-C.; Billon-Galland, M.-A.; Dion, C.; Dumortier, P.; Brochard, P.; Sobaszek, A.; Bartsch, P.; Paris, C.; et al. Quantification of short and long asbestos fibers to assess asbestos exposure: a review of fiber size toxicity. Environ. Heal. 2014, 13, 59–59. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, A.F. Bridging the gap between toxicity and carcinogenicity of mineral fibres by connecting the fibre crystal-chemical and physical parameters to the key characteristics of cancer. Curr. Res. Toxicol. 2021, 2, 42–52. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. Journal of the American Chemical Society 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Aust, A.E.; Cook, P.M.; Dodson, R.F. Morphological and Chemical Mechanisms of Elongated Mineral Particle Toxicities. J. Toxicol. Environ. Heal. Part B 2011, 14, 40–75. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.J.; Liang, M.; Toth, I.; Monteiro, M.J.; Minchin, R.F. Molecular Interaction of Poly(acrylic acid) Gold Nanoparticles with Human Fibrinogen. ACS Nano 2012, 6, 8962–8969. [Google Scholar] [CrossRef]
- Di Giuseppe, D. Characterization of Fibrous Mordenite: A First Step for the Evaluation of Its Potential Toxicity. Crystals 2020, 10, 769. [Google Scholar] [CrossRef]
- Kaszuba, J.; Yardley, B.; Andreani, M. Experimental perspectives of mineral dissolution and precipitation due to carbon dioxide-water-rock interactions. Reviews in Mineralogy and Geochemistry 2013, 77 (1), 153 - 188.
- Oze, C.; Solt, K. Biodurability of chrysotile and tremolite asbestos in simulated lung and gastric fluids. American Mineralogist 2010, 95, 825–831. [Google Scholar] [CrossRef]
- Rozalen, M.; Huertas, F.J. Comparative effect of chrysotile leaching in nitric, sulfuric and oxalic acids at room temperature. Chem. Geol. 2013, 352, 134–142. [Google Scholar] [CrossRef]
- Rozalen, M.; Ramos, M.; Gervilla, F.; Kerestedjian, T.; Fiore, S.; Huertas, F. Dissolution study of tremolite and anthophyllite: pH effect on the reaction kinetics. Appl. Geochem. 2014, 49, 46–56. [Google Scholar] [CrossRef]
- Karunaratne, P.C.T.; Fernando, G.W.A.R. Characterisation and radiation impact of corrugated asbestos roofing sheets in Sri Lanka. Journal of Geological Society of Sri Lanka 2015, 17, 31–40. [Google Scholar]
- Fischer, C.; Kurganskaya, I.; Schäfer, T.; Lüttge, A. Variability of crystal surface reactivity: what do we know? Applications in Geochemistry 2014, 43, 132–157. [Google Scholar] [CrossRef]
- Donaldson, K.; Murphy, F.A.; Duffin, R.; Poland, C.A. Asbestos, carbon nanotubes and the pleural mesothelium: a review of the hypothesis regarding the role of long fibre retention in the parietal pleura, inflammation and mesothelioma. Fibre Tox. 2010, 7, 5. [Google Scholar] [CrossRef]
- Gualtieri, A.F. Towards a quantitative model to predict the toxicity/pathogenicity potential of mineral fibers. Toxicol. Appl. Pharmacol. 2018, 361, 89–98. [Google Scholar] [CrossRef]
- McCrone, W. , McCrone, L. and Delly, J. (1978): Polarized light microscopy. Ann Arbor, MI: Ann Arbor Science Publishers, Inc.
- Middleton, A.P. (1979): The identification of asbestos in solid materials. In: Michaels L, Chissick SS, eds. Asbestos: properties applications and hazards. Chichester, UK: John Wiley and Sons, Inc.
- Vaughan, N.P.; Rooker, S.J.; LE Guen, J.M.M. IN SITU IDENTIFICATION OF ASBESTOS FIBRES COLLECTED ON MEMBRANE FILTERS FOR COUNTING. Ann. Occup. Hyg. 1981, 24, 281–290. [Google Scholar] [CrossRef]
- Bernstein, D.; Dunnigan, J.; Hesterberg, T.; Brown, R.; Velasco, J.A.L.; Barrera, R.; Hoskins, J.; Gibbs, A. Health risk of chrysotile revisited. Crit. Rev. Toxicol. 2013, 43, 154–183. [Google Scholar] [CrossRef]
- Walton, W.H. THE NATURE, HAZARDS AND ASSESSMENT OF OCCUPATIONAL EXPOSURE TO AIRBORNE ASBESTOS DUST: A REVIEW. Ann. Occup. Hyg. 1982, 25, 117–119. [Google Scholar] [CrossRef]
- Stanton, M.F.; Layard, M.; Tegeris, A.; Miller, E.; May, M.; Morgan, E.; Smith, A. Relation of Particle Dimension to Carcinogenicity in Amphibole Asbestoses and Other Fibrous Minerals2. JNCI J. Natl. Cancer Inst. 1981, 67, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Churg, A. (1993): Asbestos lung burden and disease patterns in man. In: Guthrie, G.D. and Mossman, B.T. (Eds), Health Effects of Mineral Dust. Review in Mineralogy and Geochemistry, 28, Mineralogical Society of America, Chantilly, Virginia, USA, pp. 409 – 426.
- Pollastri, S.; Gualtieri, A.F.; Gualtieri, M.L.; Hanuskova, M.; Cavallo, A.; Gaudino, G. The zeta potential of mineral fibers. Journal of Hazardous Materials 2014, 276, 469–479. [Google Scholar] [CrossRef]
- Pollastri, S.; D’Acapito, F.; Trapananti, A.; Colantoni, I.; Andreozzi, G.B.; Gualtieri, A.F. The chemical environment of iron in mineral fibers. A combined X-ray absorption and Mossbauer spectroscopic study. Journal of Hazardous Materials 2015, 298, 282–293. [Google Scholar] [PubMed]
- Vigliaturo, R.; Della Ventura, G.; Choi, J.K.; Marengo, A.; Lucci, F.; O'Shea, M.J.; Pérez-Rodríguez, I.; Gieré, R. Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy). Minerals 2018, 8, 557. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G. TOXICOKINETICS AND EFFECTS OF FIBROUS AND NONFIBROUS PARTICLES. Inhal. Toxicol. 2002, 14, 29–56. [Google Scholar] [CrossRef]
- Singer, A.; Kirsten, W.F.A.; Buhmann, C. Occurance of sepiolite in the northern Transvaal, South Africa. South African Journal of Geology 1992, 95 (5/6), 165 – 170.
- Turci, T.; Tomatis, M.; Pacella, A. Surface and bulk properties of mineral fibres relevant to toxicity. European Mineralogical Union Notes in Mineralogy 2017, 18, 171–214. [Google Scholar]
- Iqbal, J.; Shah, M.H. Occurrence, risk assessment, and source apportionment of heavy metals in surface sediments from Khanpur Lake, Pakistan. J. Anal. Sci. Technol. 2014, 5, 28. [Google Scholar] [CrossRef]
- Gualtieri, A.F.; Pollastri, S.; Gandolfi, N.B.; Gualtieri, M.L. In vitro acellular dissolution of mineral fibres: A comparative study. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Nemery, B. Metal toxicity and the respiratory tract. Eur. Respir. J. 1990, 3, 202–219. [Google Scholar] [CrossRef]
- Fubini, B.; Mollo, L. Role of iron in the reactivity of mineral fibers. Toxicol. Lett. 1995, 82-83, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Gazzano, E.; Foresti, E.; Lesci, I.G.; Tomatis, M.; Riganti, C.; Fubini, B.; Roveri, N.; Ghigo, D. Different cellular responses evoked by natural and stoichiometric synthetic chrysotile asbestos. Toxicol. Appl. Pharmacol. 2005, 206, 356–364. [Google Scholar] [CrossRef]
- Gazzano, E.; Turci, F.; Foresti, E.; Putzu, M.G.; Aldieri, E.; Silvagno, F.; Lesci, I.G.; Tomatis, M.; Riganti, C.; Romano, C.; et al. Iron-Loaded Synthetic Chrysotile: A New Model Solid for Studying the Role of Iron in Asbestos Toxicity. Chem. Res. Toxicol. 2007, 20, 380–387. [Google Scholar] [CrossRef]
- Shimizu, Y.; Dobashi, K.; Kusakbe, T.; Nagamine, T.; Oikawa, M.; Satoh, T.; Haga, J.; Ishii, Y.; Ohkubo, T.; Kamiya, T.; et al. In-Air Micro-Particle Induced X-ray Emission Analysis of Asbestos and Metals in Lung Tissue. Int. J. Immunopathol. Pharmacol. 2008, 21, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Makishima, A.; Hagino, K.; Okabe, K. Accumulation of radium in ferruginous protein bodies formed in lung tissue: association of resulting radiation hotspots with malignant mesothelioma and other malignancies. Proceedings of the Japan Academy Series B Physical and Biological Sciences 2009, 85, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Hamilton Jr., R. F. Silica binding and toxicity in alveolar macrophages. Free Radical Biology and Medicine 2008, 44(7), 1246–1258. [Google Scholar] [PubMed]
- Barabasz, W.; Albinska, D. ; Jaskowska, M; Lipiec, J. Ecotoxicology of Aluminium. Polish Journal of Environmental Studies 2002, 11(3), 199–203. [Google Scholar]
- Gupta, N.; Gaurav, S.S.; Kumar, A. Molecular Basis of Aluminium Toxicity in Plants: A Review. Am. J. Plant Sci. 2013, 04, 21–37. [Google Scholar] [CrossRef]
- Deer, W.A. , Howie, R.A. and Zussman, J. (2009): Rock Forming Minerals. 3B. Layered Silicates Excluding Micas and Clay Minerals, second ed. The Geological Society, London.
- Wicks, F.J.; Plant, A.G. Electron-Microprobe and X-Ray-Microbeam studies of serpentine textures. Canadian Mineralogist 1979, 17, 785–830. [Google Scholar]
- Oberti, R. , Hawthorne, F.C., Cannillo, E. and Camara, F. (2007): Longrange order in amphiboles. In: Hawthorne, F.C., Oberti, R., Della Ventura, G., Mottana, A. (Eds.), Amphiboles: Crystal Chemistry, Occurrence, and Health Issues. Mineralogical Society of America, Chantilly, VA, USA, 125-172.
- Hudnell, H.K. Effects from environmental Mn exposures: a review of the evidence from non-occupational exposure studies. Neurotoxicology 1999, 20, 379–397. [Google Scholar]
- Aschner, J.L.; Aschner, M. Nutritional aspects of manganese homeostasis. Mol. Asp. Med. 2005, 26, 353–362. [Google Scholar] [CrossRef]
- Fukuda, H.; Ebara, M.; Yamada, H.; Arimoto, M.; Okabe, S.; Obu, M.; Yoshikawa, M.; Sugiura, N.; Saisho, H. Trace elements and cancer. Japan Medical Association Journal 2004, 47, 391–395. [Google Scholar]
- Bargagli, E.; Monaci, F.; Bianchi, N.; Bucci, C.; Rottoli, P. Analysis of Trace Elements in Bronchoalveolar Lavage of Patients with Diffuse Lung Diseases. Biol. Trace Element Res. 2008, 124, 225–235. [Google Scholar] [CrossRef]
- Wei, B.; Yang, L.; Zhu, O.; Yu, J.; Jia, X.; Dong, T.; Lu, R. Multivariate Analysis of Trace Elements Distribution in Hair of Pleural Plaques Patients and Health Group in a Rural Area from China. Hair: Ther. Transplant. 2014, 4. [Google Scholar] [CrossRef]
- IARC, (2012): A Review of Human Carcinogens. Part C: Arsenic, Metals, Fibers, and Dusts. International Agency for Research on Cancer, Lyon, France.
- Bloise, A.; Barrese, E.; Apollaro, C. Hydrothermal alteration of Ti-doped forsterite to chrysotile and characterization of the resulting chrysotile fibers. Neuses Jahrbuch fur Mineralogie Abhandlungen 2009, 185, 297–304. [Google Scholar] [CrossRef]
- Bloise, A.; Belluso, E.; Fornero, E.; Rinaudo, C.; Barrese, E.; Capella, S. Influence of synthesis conditions on growth of Ni-doped chrysotile. Microporous Mesoporous Mater. 2010, 132, 239–245. [Google Scholar] [CrossRef]
- Wicks, F.J.; O’Hanley, D.S. Serpentine minerals; structures and petrology. Reviews in Mineralogy and Geochemistry 1988, 19, 91–167. [Google Scholar]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.-Q. Heavy Metals and Pesticides Toxicity in Agricultural Soil and Plants: Ecological Risks and Human Health Implications. Toxics 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Mathew, B.B.; Tiwari, A.; Jatawa, S.K. Free radicals and antioxidants: A review. Journal of Pharmacy Research 2011, 4(12), 4340–4343. [Google Scholar]
- Wadhwa, N.; Mathew, B.B.; Jatawa, S.; Tiwari, A. Lipid peroxidation: mechanism, models and significance. International Journal of Current Science 2012, 3, 29–38. [Google Scholar]
- Oller, A.R.; Costa, M.; Oberdörster, G. Carcinogenicity Assessment of Selected Nickel Compounds. Toxicol. Appl. Pharmacol. 1997, 143, 152–166. [Google Scholar] [CrossRef]
- McGregor, D.; Baan, R.; Partensky, C.; Rice, J.; Wilbourn, J. Evaluation of the carcinogenic risks to humans associated with surgical implants and other foreign bodies — a report of an IARC Monographs Programme Meeting. Eur. J. Cancer 2000, 36, 307–313. [Google Scholar] [CrossRef]
- Seilkop, S.K.; Oller, A.R. Respiratory cancer risks associated with low-level nickel exposure: an integrated assessment based on animal, epidemiological, and mechanistic data. Regul. Toxicol. Pharmacol. 2003, 37, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Horie, M.; Nishio, K.; Fujita, K.; Kato, H.; Nakamura, A.; Kinugasa, S.; Endoh, S.; Miyauchi, A.; Yamamoto, K.; Murayama, H.; et al. Ultrafine NiO Particles Induce Cytotoxicity in Vitro by Cellular Uptake and Subsequent Ni(II) Release. Chem. Res. Toxicol. 2009, 22, 1415–1426. [Google Scholar] [CrossRef]
- Caicedo, M.; Jacobs, J.J.; Reddy, A.; Hallab, N.J. Analysis of metal ion-induced DNA damage, apoptosis, and necrosis in human (Jurkat) T-cells demonstrates Ni2+ and V3+ are more toxic than other metals: Al3+, Be2+, Co2+, Cr3+, Cu2+, Fe3+, Mo5+, Nb5+, Zr2+. Journal of Biomedical Materials Research Part A 2007, 84(4), 905–913. [Google Scholar] [CrossRef] [PubMed]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020, 6, e04691. [Google Scholar] [CrossRef] [PubMed]
- Scharf, B.; Clement, C.C.; Zolla, V.; Perino, G.; Yan, B.; Elci, S.G.; Purdue, E.; Goldring, S.; Macaluso, F.; Cobelli, N.; et al. Molecular analysis of chromium and cobalt-related toxicity. Sci. Rep. 2014, 4, srep05729. [Google Scholar] [CrossRef]
- Gualtieri, A.F.; Andreozzi, G.B.; Tomatis, M.; Turci, F. Iron from a geochemical viewpoint. Understanding toxicity/pathogenicity mechanisms in iron-bearing minerals with a special attention to mineral fibers. Free. Radic. Biol. Med. 2018, 133, 21–37. [Google Scholar] [CrossRef] [PubMed]
- O'Flaherty, E.J. A Physiologically Based Model of Chromium Kinetics in the Rat. Toxicol. Appl. Pharmacol. 1996, 138, 54–64. [Google Scholar] [CrossRef]
- Beaumont, J.J.; Sedman, R.M.; Reynolds, S.D.; Sherman, C.D.; Li, L.-H.; Howd, R.A.; Sandy, M.S.; Zeise, L.; Alexeeff, G.V. Cancer Mortality in a Chinese Population Exposed to Hexavalent Chromium in Drinking Water. Epidemiology 2008, 19, 12–23. [Google Scholar] [CrossRef]
- Shekhawat, K.; Chatterjee, S.; Joshi, B. Chromium Toxicity and its Health Hazards. International Journal of Advanced Research 2015, 3, 167–172. [Google Scholar]
- Gualtieri, A.F.; Lusvardi, G.; Zoboli, A.; Di Giuseppe, D.; Gualtieri, M.L. Biodurability and release of metals during the dissolution of chrysotile, crocidolite and fibrous erionite. Environ. Res. 2019, 171, 550–557. [Google Scholar] [CrossRef]
- Rodríguez-Mercado, J.J.; Mateos-Nava, R.A.; Altamirano-Lozano, M.A. DNA damage induction in human cells exposed to vanadium oxides in vitro. Toxicol. Vitr. 2011, 25, 1996–2002. [Google Scholar] [CrossRef] [PubMed]
- WHO (1990): Environmental health criteria 81: vanadium. 1-35. Geneva.
- WHO (2001): Vanadium Pentoxide and other Inorganic Vanadium Compounds. Concise International Chemical Assessment Document: 29.
- ATSDR (Agency for Toxic substances and Disease Registry) (2002): Draft toxicological profile for several trace elements. US Dept. Health Human Services, Agency for Toxic Substances and Disease Registry, Atlanta. GA, USA.
- Crans, D.C.; Smee, J.J.; Gaidamauskas, E.; Yang, L. The Chemistry and Biochemistry of Vanadium and the Biological Activities Exerted by Vanadium Compounds. Chem. Rev. 2004, 104, 849–902. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.C.; Herbert, R.A.; Roycroft, J.H.; Haseman, J.K.; Grumbein, S.L.; Miller, R.A; Chouf, B.J. Lung Tumor Induction by Inhalation Exposure to Molybdenum Trioxide in Rats and Mice. Toxicological Sciences 1998, 45, 58–65. [Google Scholar] [CrossRef]
- Ott, H.C.; Prior, C.; Herold, M.; Riha, M.; Laufer, G.; Ott, G. Respiratory symptoms and bronchoalveolar lavage abnormalities in molybdenum exposed workers. . 2004, 25–30. [Google Scholar]
- Ballirano, P.; Bloise, A.; Gualtieri, A.F.; Lezzerini, M.; Pacella, A.; Perchiazzi, N.; Dogan, M.; Dogan, A.U. The crystal structure of mineral fibres. In A.F. Gualtieri, Ed., Mineral fibres: Crystal chemistry, chemical physical properties, biological interaction and toxicity 2017, 18, 17–64. European Mineralogical Union and the Mineralogical Society of Great Britain & Ireland, UK.
- Andreozzi, G.B.; Ballirano, P.; Gianfagna, A.; Mazziotti-Tagliani, S.; Pacella, A. Structural and spectroscopic characterization of a suite of fibrous amphiboles with high environmental and health relevance from Biancavilla (Sicily, Italy). 2009; 94, 1330–1340. [Google Scholar] [CrossRef]
- Bloise, A.; Ricchiuti, C.; Punturo, R.; Pereira, D. Potentially toxic elements (PTEs) associated with asbestos chrysotile, tremolite and actinolite in the Calabria region (Italy). Chem. Geol. 2020, 558. [Google Scholar] [CrossRef]
- Lu, S.; Wang, Y.; Teng, Y.; Yu, X. Heavy metal pollution and ecological risk assessment of the paddy soils near a zinc-lead mining area in Hunan. Environ. Monit. Assess. 2015, 187. [Google Scholar] [CrossRef]
- Vanoeteren, C.; Cornelis, R.; Verbeeck, P. Evaluation of trace elements in human lung tissue III. Correspondence analysis. Sci. Total. Environ. 1986, 54, 237–245. [Google Scholar] [CrossRef]
- Lippmann, M. Asbestos Exposure Indices. Environmental Research 1988, 48 (1), 86 – 106.
- Dixon, J.R.; Lowe, D.B.; E Richards, D.; Cralley, L.J.; E Stokinger, H. The role of trace metals in chemical carcinogenesis: asbestos cancers. . 1970, 30, 1068–74. [Google Scholar]
- Visonà, S.D.; Capella, S.; Bodini, S.; Borrelli, P.; Villani, S.; Crespi, E.; Colosio, C.; Previderè, C.; Belluso, E. Evaluation of Deposition and Clearance of Asbestos (Detected by SEM-EDS) in Lungs of Deceased Subjects Environmentally and/or Occupationally Exposed in Broni (Pavia, Northern Italy). Front. Public Heal. 2021, 9. [Google Scholar] [CrossRef]
- Scambelluri, M.; Piccardo, G.B.; Philippot, P.; Robbiano, A.; Negretti, L. High salinity fluid inclusions formed from recycled seawater in deeply subducted alpine serpentinite. Earth Planet. Sci. Lett. 1997, 148, 485–499. [Google Scholar] [CrossRef]
- Tiepolo, M.; Oberti, R.; Zanetti, A.; Vannucci, R.; Foley, S.F. (2007): Trace-element partitioning between amphibole and silicate melt. In: Hawthorne, F.C., Oberti, R., Della Ventura, G., Mottana, A. (Eds.), Amphiboles: Crystal Chemistry, Occurrence, and Health Issues. Mineralogical Society of America, Chantilly, pp. 417–452. [CrossRef]
- Kumar, A.; Maiti, S.K. Assessment of potentially toxic heavy metal contamination in agricultural fields, sediment, and water from an abandoned chromite-asbestos mine waste of Roro hill, Chaibasa, India. Environ. Earth Sci. 2015, 74, 2617–2633. [Google Scholar] [CrossRef]
- Holmes, A.; Morgan, A.; Sandalls, F.J. Determination of Iron, Chromium, Cobalt, Nickel, and Scandium in Asbestos by Neutron Activation Analysis. Am. Ind. Hyg. Assoc. J. 1971, 32, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Tashakor, M.; Yaacob, W.Z.W.; Mohamad, H.; Ghani, A.A. Geochemical characteristics of serpentinite soils from Malaysia. Malays. J. Soil Sci. 2014, 18, 35–49. [Google Scholar]
- Punturo, R.; Bloise, A.; Critelli, T.; Catalano, M.; Fazio, E.; Apollaro, C. Environmental implications related to natural asbestos occurrences in the ophiolites of the Gimigliano-Mount Reventino Unit (Calabria, Southern Italy). International Journal of Environmental Research 2015, 9, 405–418. [Google Scholar]
- Mendez, M.O.; Maier, R.M. Phytoremediation of mine tailings in temperate and arid environments. Rev. Environ. Sci. Bio/Technology 2007, 7, 47–59. [Google Scholar] [CrossRef]
- Huang, L.; Baumgartl, T.; Mulligan, D. Is rhizosphere remediation sufficient for sustainable revegetation of mine tailings? Ann. Bot. 2012, 110, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Kumaresan, D.; Cross, A.T.; Moreira-Grez, B.; Kariman, K.; Nevill, P.; Stevens, J.; Allcock, R.J.N.; O’donnell, A.G.; Dixon, K.W.; Whiteley, A.S. Microbial Functional Capacity Is Preserved Within Engineered Soil Formulations Used In Mine Site Restoration. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Schimmer, C. (2017): Synergistic use of soil microbes and plants to facilitate rehabilitation on gold tailings materials. Dissertation (MSc) School for Geo- and Spatial Sciences, North-West University, Potchefstroom.
- Northwatch, Mining Watch Canada and Associates. 2008. The Boreal Below: Mining Issues and Activities in Canada’s Boreal Forest. Pp. 205.
- Pentreath, R.J. (1994): The Discharge of Waters from Active and Abandoned Mines. In: Mining and its Environmental Impact. Edited By: R.E. Hester and R.M. Harrison. Royal Society of Chemistry (Great Britain), p. 121-131.
- Lamb, D.; Erskine, P.D.; Fletcher, A. Widening gap between expectations and practice in Australian mine site rehabilitation. Ecological Management and Restoration 2015, 16, 186–195. [Google Scholar] [CrossRef]
- Cross, A.T.; Stevens, J.C.; Sadler, R.; Moreira-Grez, B.; Ivanov, D.; Zhong, H.; Dixon, K.W.; Lambers, H. Compromised root development constrains the establishment potential of native plants in unamended alkaline post-mining substrates. Plant Soil 2018, 461, 163–179. [Google Scholar] [CrossRef]
| Chrysotile | Amosite | Crocidolite | |
|---|---|---|---|
| Sample number | Ch1 | Am2 | Cr3 |
| Sampling locations |
Havelock Mine, Bulembu, Swaziland (Eswatini) | Penge, Sekhukhune District, Limpopo, South Africa | Prieska Division, Northern Cape, South Africa |
| Location coordinates |
25°57’21’’S 31°07’51’’E | 24°25’07’’S 30°20’14’’E | 28°19’01’’S 23°06’05’’E |
| Climate |
Humid sub-tropical | Sub-tropical | Semi-arid |
| Biome |
Grassland | Savanna | Savanna |
| Occurrence |
Cross-vein fibres (growth of fibres at right angles to the walls of cracks) | Cross-fibre seams in banded ironstones | Cross-fibre seams in banded ironstones |
| Number of samples collected | Three | Two | Two |
| Chrysotile | Amosite | Crocidolite | ||||
| Counts | Length (cm) | Width (µm) | Length (cm) | Width (µm) | Length (cm) | Width (µm) |
| 1 | 2.5 | 12.5 | 20 | 28.5 | 6 | 6.25 |
| 2 | 0.5 | 12 | 5 | 26.5 | 10 | 6.2 |
| 3 | 1.2 | 12.2 | 15 | 28 | 11 | 6.3 |
| 4 | 1.8 | 12.2 | 10 | 23.8 | 8 | 6 |
| 5 | 2.2 | 12.6 | 9 | 25.5 | 8 | 6 |
| 6 | 0.9 | 12.1 | 13 | 28 | 6 | 6 |
| 7 | 1.36 | 12.3 | 19 | 25 | 10 | 6.2 |
| 8 | 2.4 | 12.3 | 7 | 26.5 | 10 | 6.2 |
| 9 | 2.5 | 12.4 | 18 | 28 | 9 | 6.2 |
| 10 | 1.9 | 12.5 | 18 | 22.3 | 6 | 6.2 |
| 11 | 1.7 | 12 | 16 | 20.5 | 6 | 6.3 |
| 12 | 2.1 | 12.1 | 15 | 28.1 | 8 | 6 |
| 13 | 0.8 | 12.4 | 13 | 28.2 | 9 | 6.1 |
| 14 | 2 | 12.6 | 8 | 28 | 10 | 6 |
| 15 | 2 | 12.4 | 15 | 20.2 | 10 | 6.2 |
| 16 | 1.1 | 12.3 | 5 | 17 | 10 | 6.2 |
| 17 | 0.6 | 12.2 | 8 | 20 | 6 | 6.2 |
| 18 | 2.5 | 12.2 | 11 | 18.9 | 8 | 6.3 |
| 19 | 2.3 | 12.2 | 19 | 20 | 10 | 6.1 |
| 20 | 1.7 | 12.2 | 20 | 28.5 | 6 | 6.2 |
| 21 | 2 | 12.6 | 20 | 27.1 | 6 | 6.2 |
| Average | 1.72 | 12.3 | 13.52 | 24.7 | 8.24 | 6.16 |
| Minimum | 0.5 | 12 | 5 | 17 | 6 | 6 |
| Maximum | 2.5 | 12.6 | 20 | 28.57 | 11 | 6.3 |
| Variance | 0.40 | 0.30 | 25.11 | 13.87 | 3.13 | 0.01 |
| Standard dev. | 0.63 | 0.18 | 5.01 | 3.72 | 1.77 | 0.10 |
| Peak # | Chrysotile rock sample | Amosite rock sample | Crocidolite rock sample | |||||||
| Amosite (grunerite) | Quartz low | Sepiolite | ||||||||
| I/I1 | d (Ǻ) | I/I1 | d (Ǻ) | I/I1 | d (Ǻ) | I/I1 | d (Ǻ) | I/I1 | d (Ǻ) | |
| 1 | 100 | 7.34 | 47.4 | 9.3 | 100 | 3.35 | 63.5 | 22.8 | 15 | 8.9 |
| 2 | 20 | 4.6 | 100 | 8.6 | 48.9 | 1.8 | 100 | 15.3 | 100 | 8.3 |
| 3 | 43.33 | 4.4 | 42.1 | 3.5 | 5 | 5.15 | ||||
| 4 | 26.7 | 3.9 | 47.4 | 3.3 | 15 | 5.15 | ||||
| 5 | 53.3 | 3.6 | 64.2 | 3.1 | 7.5 | 3.8 | ||||
| 6 | 19.3 | 3.35 | 21.1 | 2.97 | 9 | 3.6 | ||||
| 7 | 13.3 | 2.9 | 74.7 | 2.7 | 30 | 3.4 | ||||
| 8 | 8 | 2.7 | 54.7 | 2.5 | 12.5 | 3.25 | ||||
| 9 | 20 | 2.6 | 32.6 | 2.3 | 60 | 3.1 | ||||
| 10 | 20.7 | 2.5 | 42.1 | 2.2 | 47.5 | 2.7 | ||||
| Asbestos rock sample | Phases detected (% composition) |
|---|---|
| Chrysotile | Chrysotile (100 %) |
| Amosite | Amosite (94.5 %) >> Quarts low (4.1 %) > Sepiolite (1.4 %) |
| Crocidolite | Magnesio-riebeckite (100 %) |
| % abundance represents the modal amounts of minerals (quantitative analysis) present in asbestos rock samples | |
| Oxides (wt%) | Chrysotile | Amosite | Crocidolite |
|---|---|---|---|
| SiO2 | 42.08 | 48.93 | 51.52 |
| Al2O3 | 0.59 | 0.36 | 0.07 |
| Fe2O3 | 2.00 | 41.37 | 38.31 |
| MnO | 0.03 | 0.64 | 0.06 |
| MgO | 40.83 | 5.84 | 2.23 |
| CaO | 0.06 | 2.02 | 0.35 |
| Na2O | 0.07 | 0.00 | 6.22 |
| K2O | 0.01 | 0.24 | 0.10 |
| TiO2 | 0.03 | 0.03 | 0.03 |
| P2O5 | 0.01 | 0.02 | 0.01 |
| Cr2O3 | 0.01 | 0.02 | 0.01 |
| NiO | 0.19 | 0.01 | 0.01 |
| LOI | 12.18 | 0.48 | 1.24 |
| Total | 100.08 | 99.87 | 100.15 |
| Element (ppm) | Chrysotile | Amosite | Crocidolite |
|---|---|---|---|
| Sc | 6.4 | 4.64 | D.L. |
| V | 16.42 | 3.51 | 3.91 |
| Cr | 83.4 | 4.61 | D.L. |
| Co | 52.55 | D.L. | D.L. |
| Ni | 1518.54 | 51.24 | 11.86 |
| Cu | 21.99 | 36.77 | 35 |
| Zn | 16.19 | 41.62 | 12.66 |
| Ga | D.l. | D.L. | 1.63 |
| Rb | D.L. | 20.77 | 1.02 |
| Sr | 0.74 | 26.63 | 0.86 |
| Y | 0.77 | 3.78 | 1.82 |
| Zr | 0.49 | 3.7 | 0.38 |
| Nb | D.L. | 0.85 | D.L. |
| Mo | D.L. | 0.62 | D.L. |
| Ba | D.L. | 28.17 | 1.69 |
| Pb | 6.62 | 5.32 | 5.06 |
| Th | D.L. | D.L. | D.L. |
| U | D.L. | D.L. | D.L. |
| D.L. – detection limit | |||
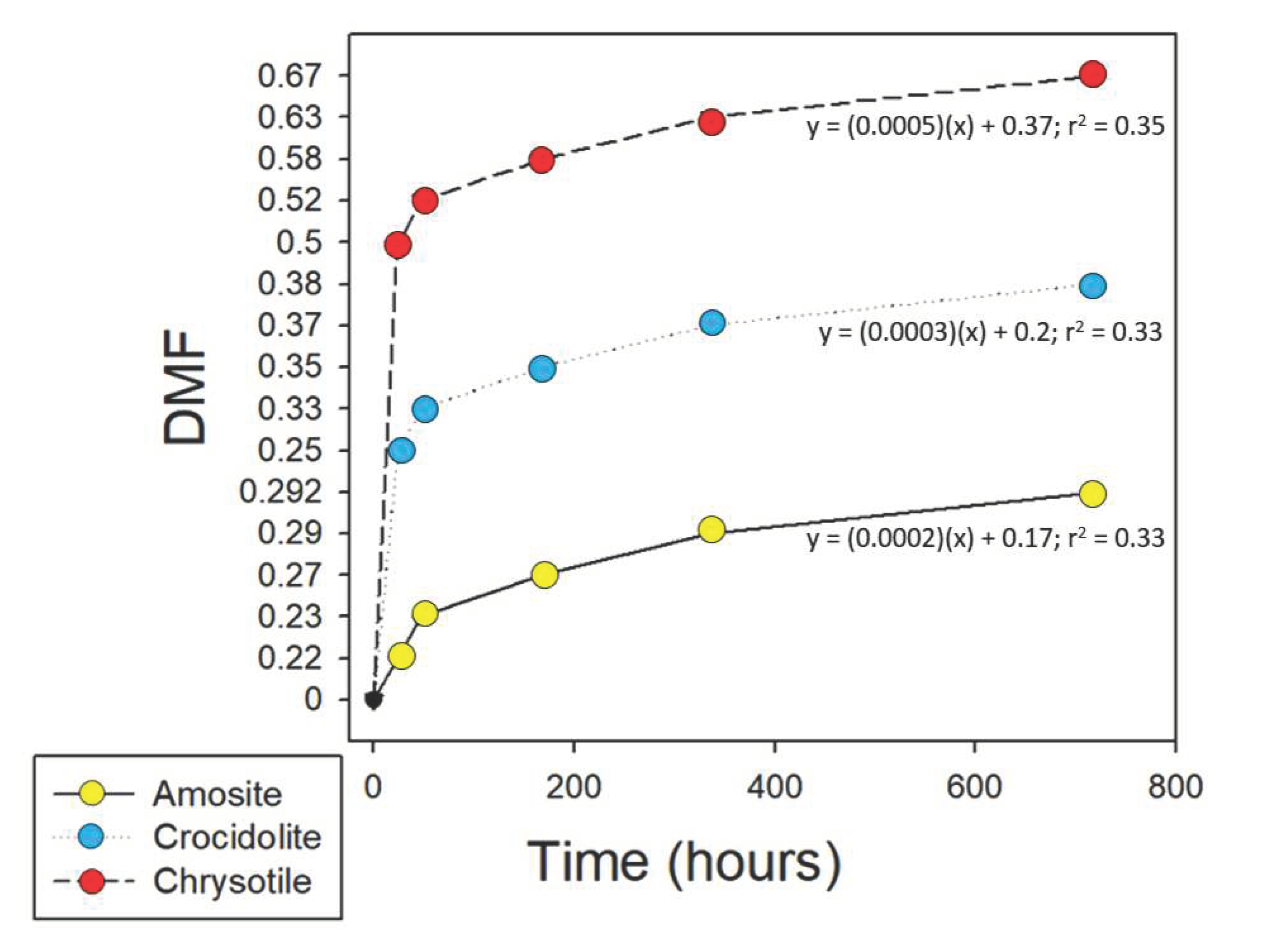
| Time (hours) | Chrysotile | Amosite | Crocidolite |
|---|---|---|---|
| 0 | 50 | 50 | 50 |
| 24 | 25.2 | 39.1 | 37.7 |
| 48 | 24 | 38.6 | 33.6 |
| 168 | 21 | 36.3 | 32.3 |
| 334 | 18.6 | 35.5 | 31.7 |
| 720 | 16.6 | 35.4 | 31.1 |
| Total mass loss (mg) | 33.4 | 14.6 | 18.9 |
| Metals (ppm) | Chrysotile | Amosite | Crocidolite | Concentration range of trace elements in normal human lungs (ppm) [122] |
|---|---|---|---|---|
| Al | 11147.8 | 6802.1 | 1322.6 | |
| Fe | 13988 | 289340 | 267940 | 40 – 500 |
| Mn | 230 | 4987 | 465 | 0.01 – 3 |
| Mg | 246200 | 35220 | 13449 | |
| Cr | 83.4 | 4.61 | D.L. | 0.002 – 0.50 |
| Co | 52.55 | D.L. | D.L. | 0.002 – 0.1 |
| Ni | 1518.54 | 51.24 | 11.86 | 0.01 – 1.00 |
| Cu | 21.99 | 36.77 | 35 | 1 – 5.00 |
| Zn | 16.19 | 41.62 | 12.66 | 1 – 30.00 |
| Zr | 0.49 | 3.7 | 0.38 | |
| Ba | D.L. | 28.17 | 1.69 | > 1.10 |
| Pb | 6.62 | 5.32 | 5.06 | 0.02 – 0.50 |
| [122] Vanoeteren et al., 1986 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





