Submitted:
08 October 2023
Posted:
10 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Model development
3. Results
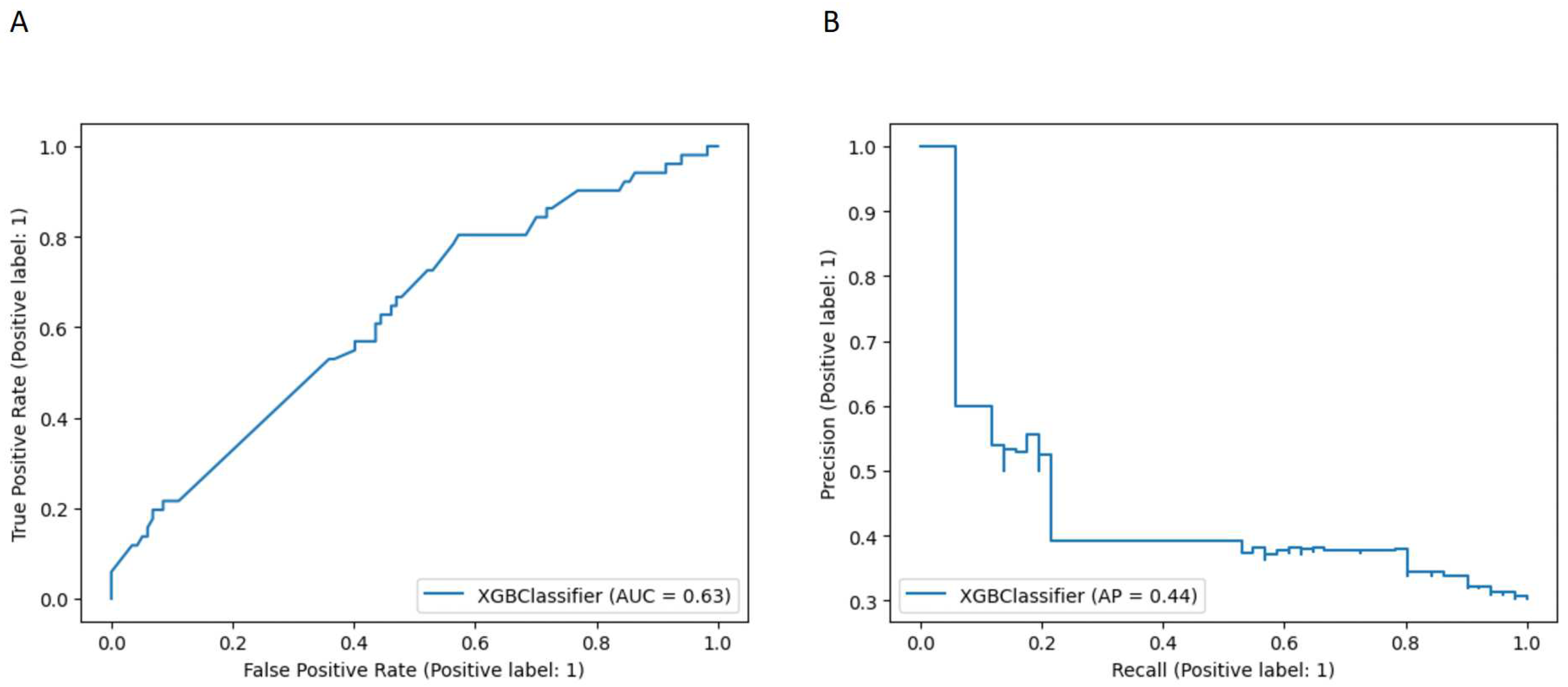
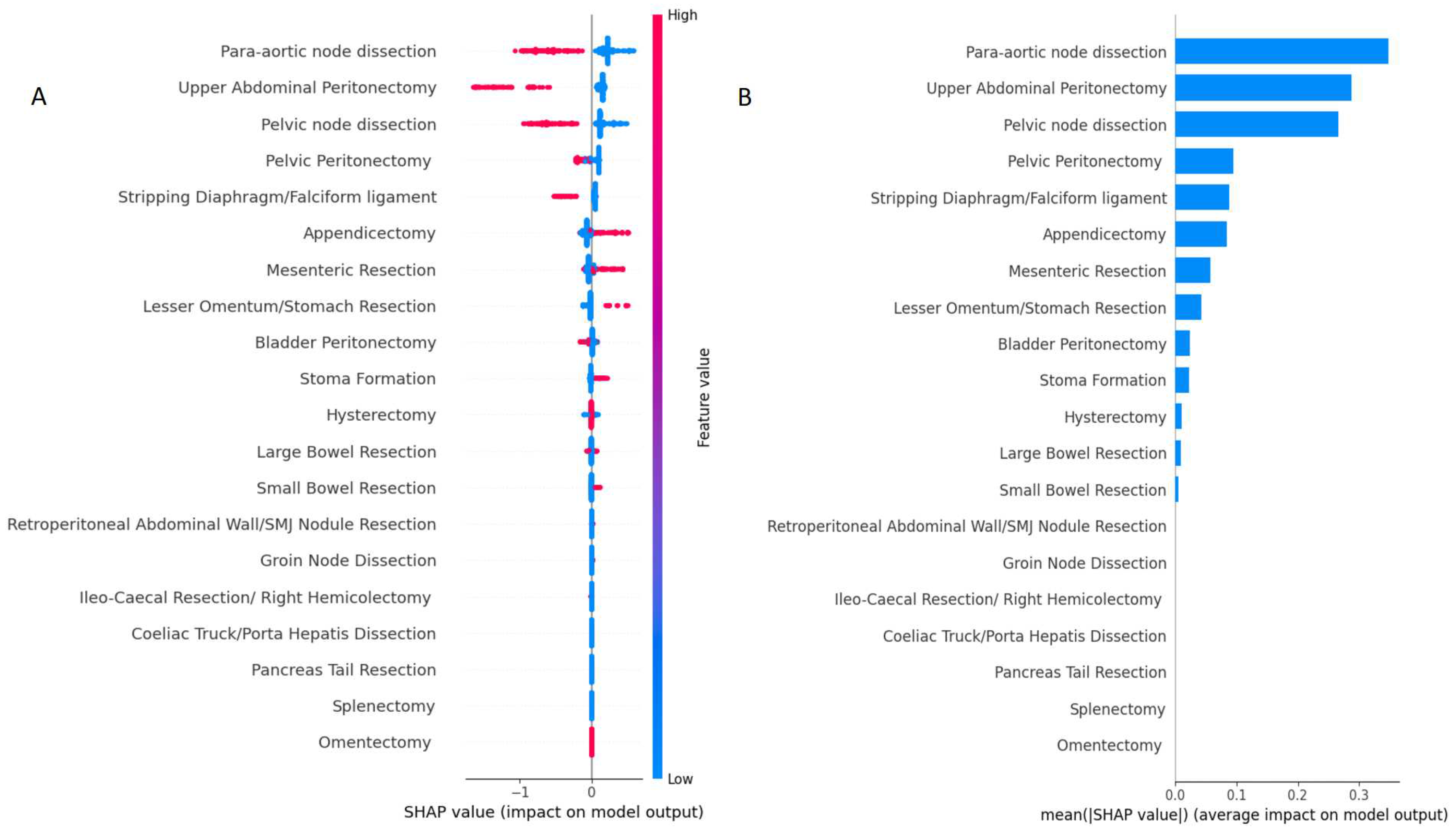
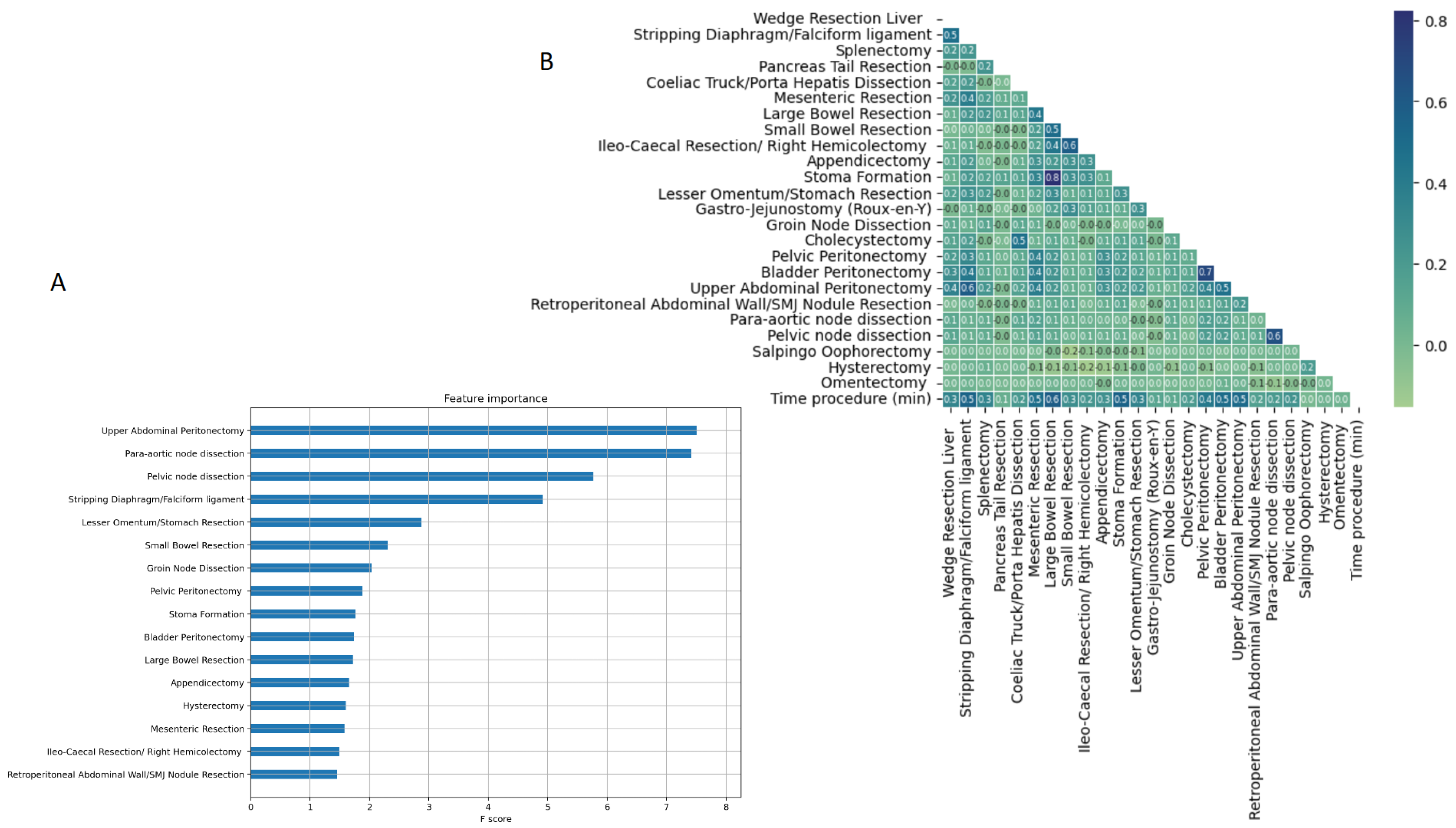
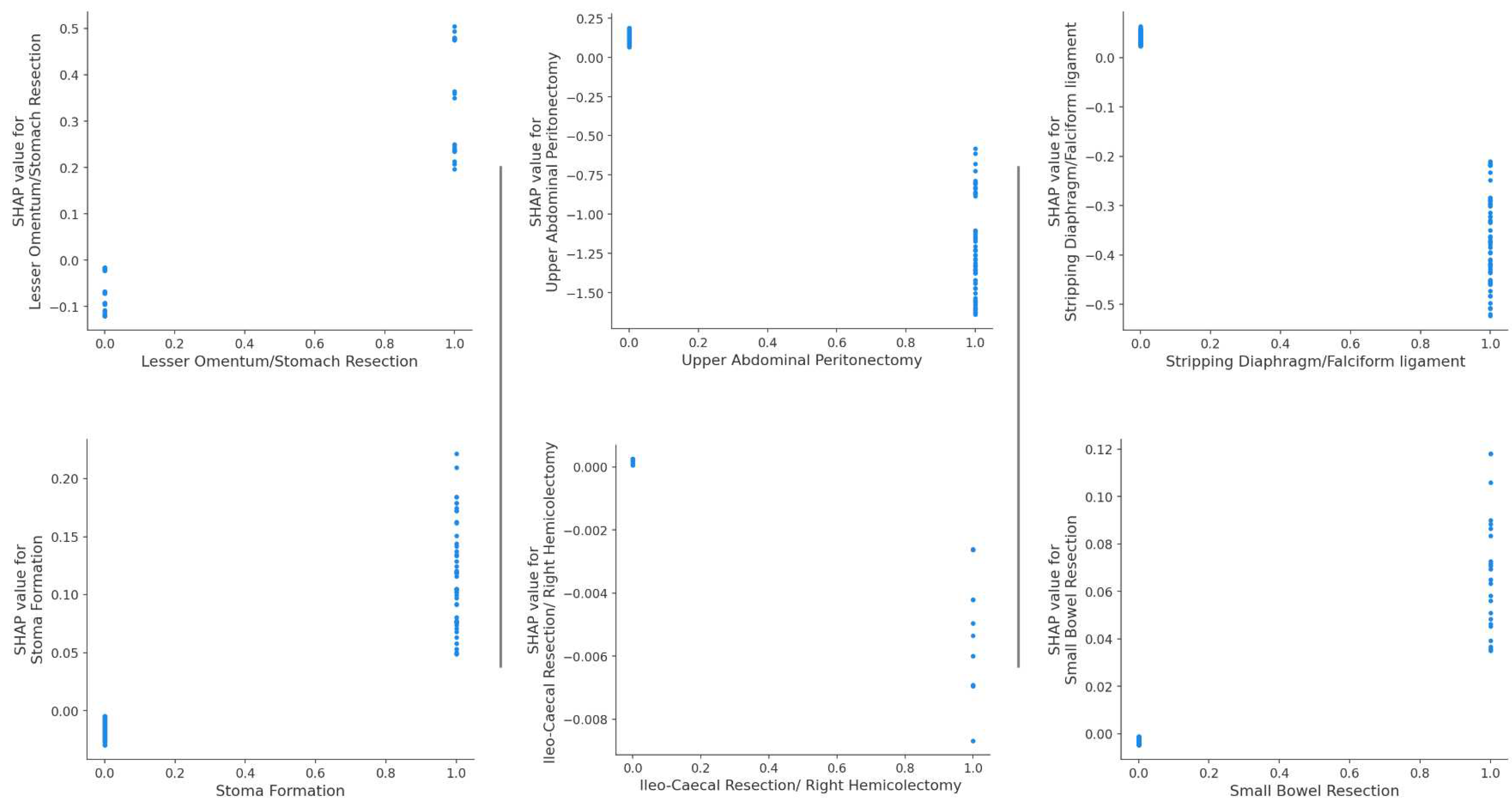
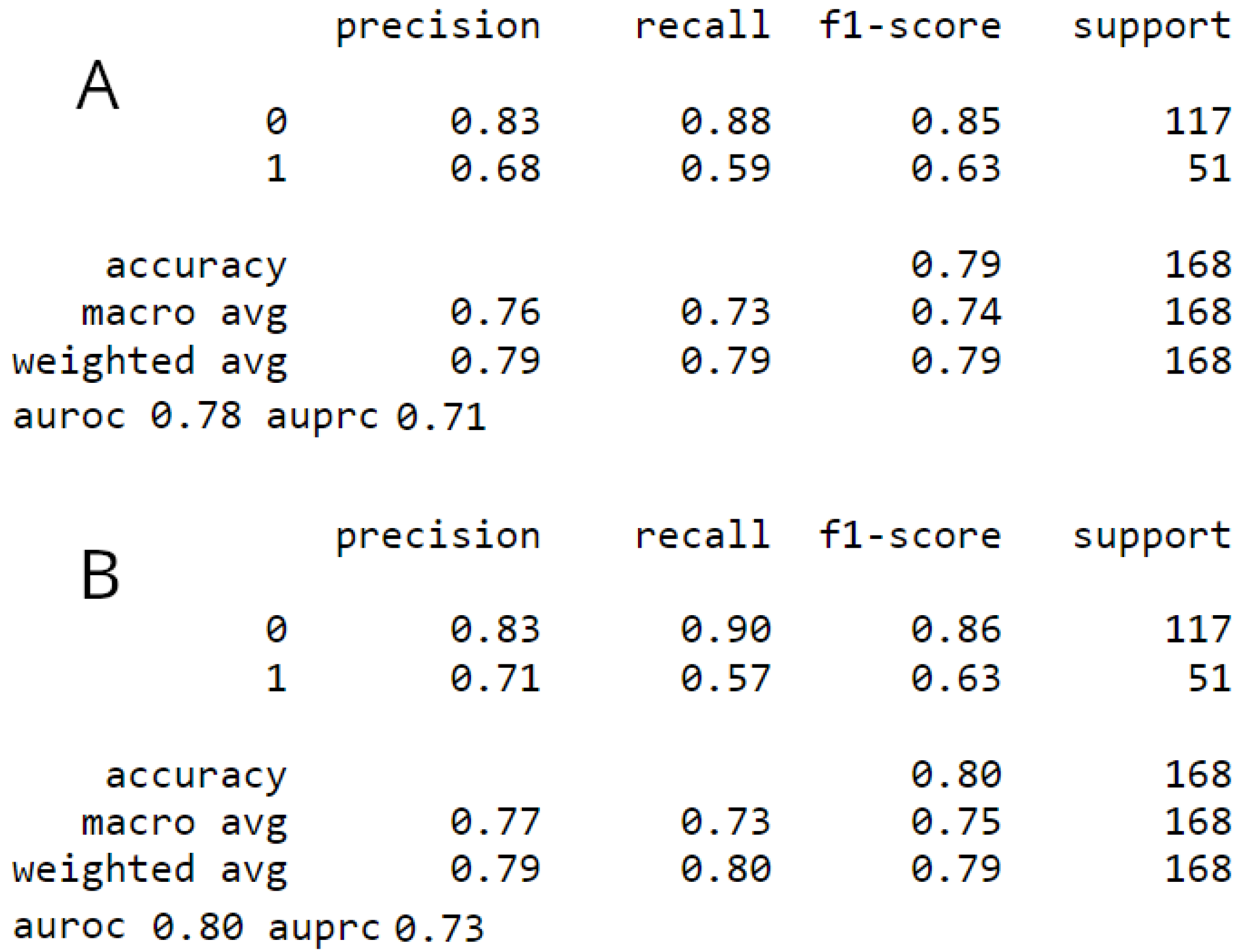
3.1. Model comparison
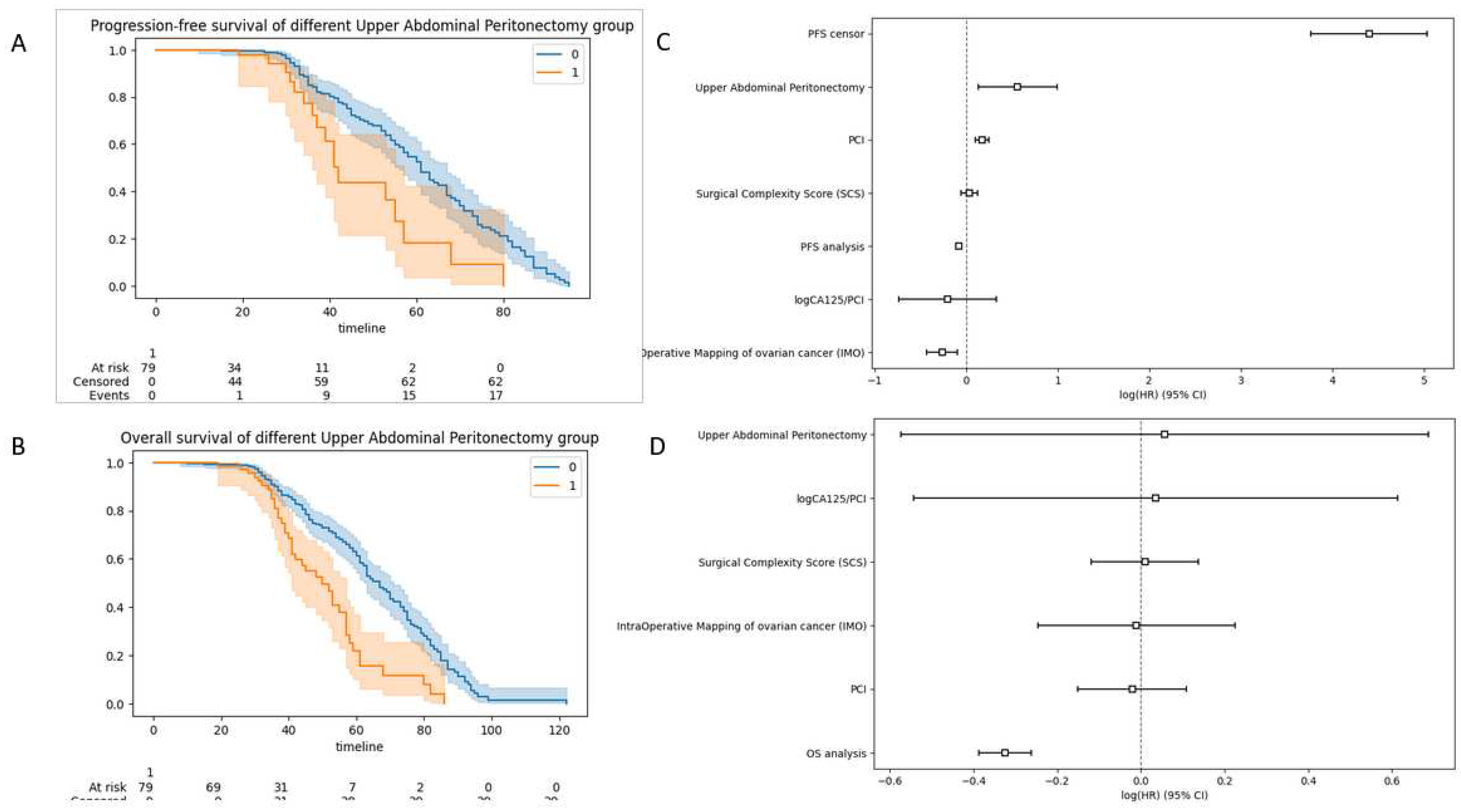
3.2. Survival data
4. Discussion
5. Strengths and limitations
6. Conclusions
References
- Du Bois, A.; Quinn, M.; Thigpen, T.; Vermorken, J.; Avall-Lundqvist, E.; Bookman, M.; Bowtell, D.; Brady, M.; Casado, A.; Cervantes, A.; others. 2004 consensus statements on the management of ovarian cancer: final document of the 3rd International Gynecologic Cancer Intergroup Ovarian Cancer Consensus Conference (GCIG OCCC 2004). Annals of oncology 2005, 16, viii7–viii12. [CrossRef]
- Monk, B.J.; Coleman, R.L.; Fujiwara, K.; Wilson, M.K.; Oza, A.M.; Oaknin, A.; O’Malley, D.M.; Lorusso, D.; Westin, S.N.; Safra, T.; others. ATHENA (GOG-3020/ENGOT-ov45): A randomized, phase III trial to evaluate rucaparib as monotherapy (ATHENA–MONO) and rucaparib in combination with nivolumab (ATHENA–COMBO) as maintenance treatment following frontline platinum-based chemotherapy in ovarian cancer. International Journal of Gynecologic Cancer 2021, 31. [CrossRef]
- Dahm-Kähler, P.; Palmqvist, C.; Staf, C.; Holmberg, E.; Johannesson, L. Centralized primary care of advanced ovarian cancer improves complete cytoreduction and survival-A population-based cohort study. Gynecologic oncology 2016, 142, 211–216. [CrossRef]
- Palmqvist, C.; Staf, C.; Mateoiu, C.; Johansson, M.; Albertsson, P.; Dahm-Kähler, P. Increased disease-free and relative survival in advanced ovarian cancer after centralized primary treatment. Gynecologic oncology 2020, 159, 409–417. [CrossRef]
- Elattar, A.; Bryant, A.; Winter-Roach, B.A.; Hatem, M.; Naik, R. Optimal primary surgical treatment for advanced epithelial ovarian cancer. Cochrane database of systematic reviews 2011. [CrossRef]
- Aletti, G.D.; Dowdy, S.C.; Podratz, K.C.; Cliby, W.A. Relationship among surgical complexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. American journal of obstetrics and gynecology 2007, 197, 676–e1. [CrossRef]
- Querleu, D.; Planchamp, F.; Chiva, L.; Fotopoulou, C.; Barton, D.; Cibula, D.; Aletti, G.; Carinelli, S.; Creutzberg, C.; Davidson, B.; others. European Society of Gynaecologic Oncology quality indicators for advanced ovarian cancer surgery. International Journal of Gynecologic Cancer 2016, 26. [CrossRef]
- Préfontaine, M.; Gelfand, A.T.; Donovan, J.T.; Powell, J.L. Reproducibility of tumor measurements in ovarian cancer: a study of interobserver variability. Gynecologic oncology 1994, 55, 87–90. [CrossRef]
- Fotopoulou, C.; Concin, N.; Planchamp, F.; Morice, P.; Vergote, I.; du Bois, A.; Querleu, D. Quality indicators for advanced ovarian cancer surgery from the European Society of Gynaecological Oncology (ESGO): 2020 update. International Journal of Gynecological Cancer 2020, 30, 436–440. [CrossRef]
- Kawakami, E.; Tabata, J.; Yanaihara, N.; Ishikawa, T.; Koseki, K.; Iida, Y.; Saito, M.; Komazaki, H.; Shapiro, J.S.; Goto, C.; others. Application of artificial intelligence for preoperative diagnostic and prognostic prediction in epithelial ovarian cancer based on blood biomarkers. Clinical cancer research 2019, 25, 3006–3015. [CrossRef]
- Laios, A.; Gryparis, A.; DeJong, D.; Hutson, R.; Theophilou, G.; Leach, C. Predicting complete cytoreduction for advanced ovarian cancer patients using nearest-neighbor models. Journal of Ovarian Research 2020, 13, 1–8. [CrossRef]
- Laios, A.; Kalampokis, E.; Johnson, R.; Thangavelu, A.; Tarabanis, C.; Nugent, D.; De Jong, D. Explainable artificial intelligence for prediction of complete surgical cytoreduction in advanced-stage epithelial ovarian cancer. Journal of personalized medicine 2022, 12, 607. [CrossRef]
- Laios, A.; Kalampokis, E.; Johnson, R.; Munot, S.; Thangavelu, A.; Hutson, R.; Broadhead, T.; Theophilou, G.; Nugent, D.; De Jong, D. Development of a Novel Intra-Operative Score to Record Diseases’ Anatomic Fingerprints (ANAFI Score) for the Prediction of Complete Cytoreduction in Advanced-Stage Ovarian Cancer by Using Machine Learning and Explainable Artificial Intelligence. Cancers 2023, 15, 966. [CrossRef]
- Jacquet, P.; Sugarbaker, P.H. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Peritoneal carcinomatosis: principles of management 1996, pp. 359–374. [CrossRef]
- Sehouli, J.; Könsgen, D.; Mustea, A.; Oskay-Özcelik, G.; Katsares, I.; Weidemann, H.; Lichtenegger, W. „IMO”-Intraoperatives Mapping des Ovarialkarzinoms. Zentralblatt für Gynäkologie 2003, 125, 129–135. [CrossRef]
- Freund, Y.; Schapire, R.E. A decision-theoretic generalization of on-line learning and an application to boosting. Journal of computer and system sciences 1997, 55, 119–139. [CrossRef]
- Dottino, J.A.; He, W.; Sun, C.C.; Zhao, H.; Fu, S.; Rauh-Hain, J.A.; Suidan, R.S.; Lu, K.H.; Giordano, S.H.; Meyer, L.A. National trends in bowel and upper abdominal procedures in ovarian cancer surgery. International Journal of Gynecologic Cancer 2020, 30. [CrossRef]
- Slack, D.; Hilgard, S.; Jia, E.; Singh, S.; Lakkaraju, H. Fooling lime and shap: Adversarial attacks on post hoc explanation methods. Proceedings of the AAAI/ACM Conference on AI, Ethics, and Society, 2020, pp. 180–186. [CrossRef]
- NICE. Overview: Maximal cytoreductive surgery for advanced ovarian cancer: Guidance.
- Stepanyan, A.; Malakyan, Z.; Alaverdyan, A.; Davtyan, H.; Hovhannisyan, T. Right upper quadrant peritonectomy. Answering frequently asked questions. International Journal of Gynecological Cancer 2021, 31, 1305–1306. [CrossRef]
- Zivanovic, O.; Eisenhauer, E.L.; Zhou, Q.; Iasonos, A.; Sabbatini, P.; Sonoda, Y.; Abu-Rustum, N.R.; Barakat, R.R.; Chi, D.S. The impact of bulky upper abdominal disease cephalad to the greater omentum on surgical outcome for stage IIIC epithelial ovarian, fallopian tube, and primary peritoneal cancer. Gynecologic oncology 2008, 108, 287–292. [CrossRef]
- Kehoe, S.M.; Eisenhauer, E.L.; Chi, D.S. Upper abdominal surgical procedures: liver mobilization and diaphragm peritonectomy/resection, splenectomy, and distal pancreatectomy. Gynecologic oncology 2008, 111, S51–S55. [CrossRef]
- Bogani, G.; Ditto, A.; Martinelli, F.; Lorusso, D.; Chiappa, V.; Donfrancesco, C.; Di Donato, V.; Indini, A.; Aletti, G.; Raspagliesi, F. Surgical techniques for diaphragmatic resection during cytoreduction in advanced or recurrent ovarian carcinoma: a systematic Review and meta-analysis. International Journal of Gynecologic Cancer 2016, 26. [CrossRef]
- Sugarbaker, P.H. Peritonectomy procedures. Annals of surgery 1995, 221, 29. [CrossRef]
- Lago, V.; Domingo, S.; Matute, L.; Padilla-Iserte, P.; Gurrea, M.; others. Radical en bloc peritonectomy in advanced ovarian cancer 2018. [CrossRef]
- Fotopoulou, C.; Taskiran, C. The principles of safe and efficacious upper abdominal surgery. Gynecol Pelvic Med 2021, 4. [CrossRef]
- Spirtos, N.M.; Gross, G.M.; Freddo, J.L.; Ballon, S.C. Cytoreductive surgery in advanced epithelial cancer of the ovary: the impact of aortic and pelvic lymphadenectomy. Gynecologic oncology 1995, 56, 345–352. [CrossRef]
- Harter, P.; Sehouli, J.; Lorusso, D.; Reuss, A.; Vergote, I.; Marth, C.; Kim, J.W.; Raspagliesi, F.; Lampe, B.; Aletti, G.; others. A randomized trial of lymphadenectomy in patients with advanced ovarian neoplasms. New England Journal of Medicine 2019, 380, 822–832. [CrossRef]
- Straubhar, A.M.; Filippova, O.T.; Cowan, R.A.; Lakhman, Y.; Sarasohn, D.M.; Nikolovski, I.; Torrisi, J.M.; Ma, W.; Abu-Rustum, N.R.; Gardner, G.J.; others. A multimodality triage algorithm to improve cytoreductive outcomes in patients undergoing primary debulking surgery for advanced ovarian cancer: a Memorial Sloan Kettering Cancer Center team ovary initiative. Gynecologic oncology 2020, 158, 608–613. [CrossRef]
- Ren, Y.; Jiang, R.; Yin, S.; You, C.; Liu, D.; Cheng, X.; Tang, J.; Zang, R. Radical surgery versus standard surgery for primary cytoreduction of bulky stage IIIC and IV ovarian cancer: an observational study. BMC cancer 2015, 15, 1–12. [CrossRef]
- Chi, D.S.; Eisenhauer, E.L.; Zivanovic, O.; Sonoda, Y.; Abu-Rustum, N.R.; Levine, D.A.; Guile, M.W.; Bristow, R.E.; Aghajanian, C.; Barakat, R.R. Improved progression-free and overall survival in advanced ovarian cancer as a result of a change in surgical paradigm. Gynecologic oncology 2009, 114, 26–31. [CrossRef]
- Spruance, S.L.; Reid, J.E.; Grace, M.; Samore, M. Hazard ratio in clinical trials. Antimicrobial agents and chemotherapy 2004, 48, 2787–2792. [CrossRef]
- Chi, D.S.; Eisenhauer, E.L.; Zivanovic, O.; Sonoda, Y.; Abu-Rustum, N.R.; Levine, D.A.; Guile, M.W.; Bristow, R.E.; Aghajanian, C.; Barakat, R.R. Improved progression-free and overall survival in advanced ovarian cancer as a result of a change in surgical paradigm. Gynecologic oncology 2009, 114, 26–31. [CrossRef]
- Laios, A.; De Jong, D.; Kalampokis, E. Beauty is in the explainable artificial intelligence (XAI) of the “agnostic” beholder. Translational Cancer Research 2023, 12, 226. [CrossRef]
- Coleridge, S.L.; Bryant, A.; Kehoe, S.; Morrison, J. Neoadjuvant chemotherapy before surgery versus surgery followed by chemotherapy for initial treatment in advanced ovarian epithelial cancer. Cochrane Database of Systematic Reviews 2021. [CrossRef]
- Chang, S.J.; Bristow, R.E.; Chi, D.S.; Cliby, W.A. Role of aggressive surgical cytoreduction in advanced ovarian cancer. Journal of gynecologic oncology 2015, 26, 336–342. [CrossRef]
- Pannu, H.K.; Bristow, R.E.; Montz, F.J.; Fishman, E.K. Multidetector CT of peritoneal carcinomatosis from ovarian cancer. Radiographics 2003, 23, 687–701. [CrossRef]
- Bhatt, A.; Yonemura, Y.; Mehta, S.; Benzerdjeb, N.; Kammar, P.; Parikh, L.; Shah, M.Y.; Shaikh, S.; Prabhu, A.; Mishra, S.; others. Target region resection in patients undergoing cytoreductive surgery for peritoneal metastases-is it necessary in absence of visible disease? European Journal of Surgical Oncology 2020, 46, 582–589. [CrossRef]
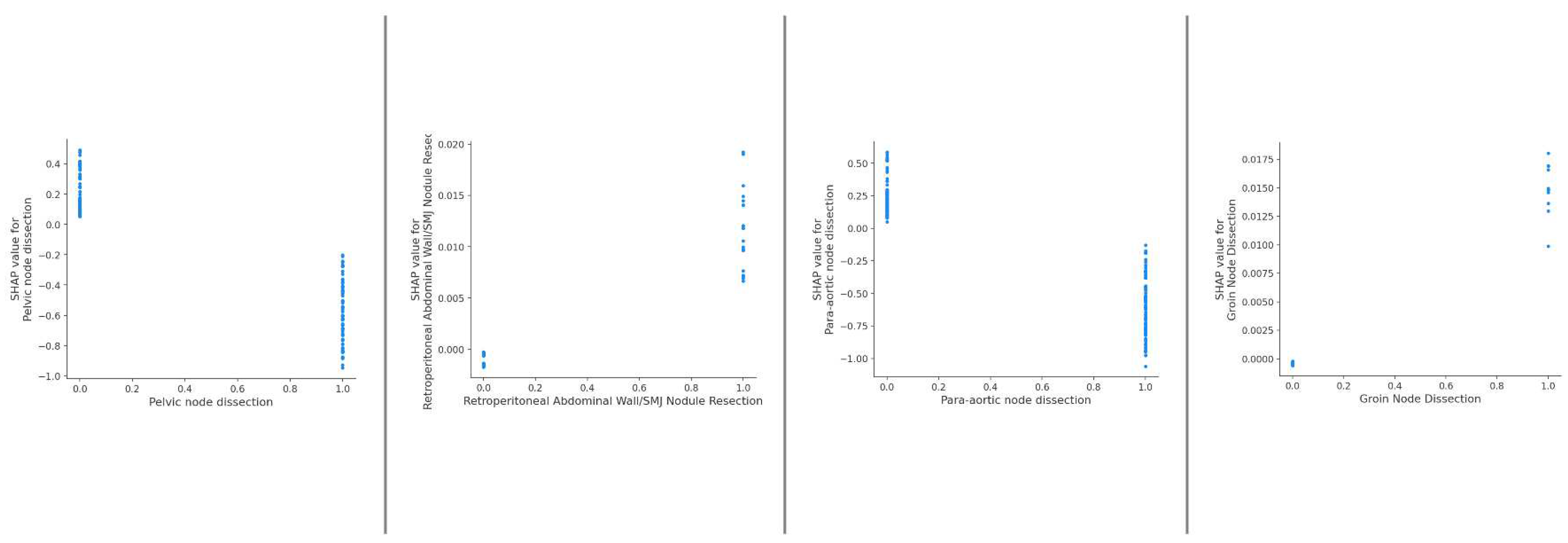
| Variable | Overall | Training Set | Testing Set | pvalue (training) | No UAP | UAP | pvalue (UAP) | |
|---|---|---|---|---|---|---|---|---|
| Wedge Resection Liver | 0 | 537 (95.89) | 428 (95.54) | 109 (97.32) | 0.558 | 475 (98.75) | 62 (78.48) | <0.001 |
| 1 | 23 (4.11) | 20 (4.46) | 3 (2.68) | 0.558 | 6 (1.25) | 17 (21.52) | <0.001 | |
| Stripping Diaphragm/Falciform ligament | 0 | 484 (86.43) | 385 (85.94) | 99 (88.39) | 0.6 | 455 (94.59) | 29 (36.71) | <0.001 |
| 1 | 76 (13.57) | 63 (14.06) | 13 (11.61) | 0.6 | 26 (5.41) | 50 (63.29) | <0.001 | |
| Splenectomy | 0 | 543 (96.96) | 435 (97.1) | 108 (96.43) | 0.951 | 474 (98.54) | 69 (87.34) | <0.001 |
| 1 | 17 (3.04) | 13 (2.9) | 4 (3.57) | 0.951 | 7 (1.46) | 10 (12.66) | <0.001 | |
| Pancreas Tail Resection | 0 | 559 (99.82) | 447 (99.78) | 112 (100.0) | 1.0 | 480 (99.79) | 79 (100.0) | 1 |
| 1 | 1 (0.18) | 1 (0.22) | 0 (0.0) | 1.0 | 1 (0.21) | 0 (0.0) | 1 | |
| Coeliac Truck/Porta Hepatis Dissection | 0 | 554 (98.93) | 443 (98.88) | 111 (99.11) | 1.0 | 479 (99.58) | 75 (94.94) | 0.002 |
| 1 | 6 (1.07) | 5 (1.12) | 1 (0.89) | 1.0 | 2 (0.42) | 4 (5.06) | 0.002 | |
| Mesenteric Resection | 0 | 427 (76.25) | 340 (75.89) | 87 (77.68) | 0.785 | 399 (82.95) | 28 (35.44) | <0.001 |
| 1 | 133 (23.75) | 108 (24.11) | 25 (22.32) | 0.785 | 82 (17.05) | 51 (64.56) | <0.001 | |
| Large Bowel Resection | 0 | 496 (88.57) | 399 (89.06) | 97 (86.61) | 0.572 | 440 (91.48) | 56 (70.89) | <0.001 |
| 1 | 64 (11.43) | 49 (10.94) | 15 (13.39) | 0.572 | 41 (8.52) | 23 (29.11) | <0.001 | |
| Small Bowel Resection | 0 | 537 (95.89) | 430 (95.98) | 107 (95.54) | 1.0 | 464 (96.47) | 73 (92.41) | 0.168 |
| 1 | 23 (4.11) | 18 (4.02) | 5 (4.46) | 1.0 | 17 (3.53) | 6 (7.59) | 0.168 | |
| Ileo-Caecal Resection/ Right Hemicolectomy | 0 | 539 (96.25) | 432 (96.43) | 107 (95.54) | 0.868 | 465 (96.67) | 74 (93.67) | 0.326 |
| 1 | 21 (3.75) | 16 (3.57) | 5 (4.46) | 0.868 | 16 (3.33) | 5 (6.33) | 0.326 | |
| Appendicectomy | 0 | 439 (78.39) | 352 (78.57) | 87 (77.68) | 0.939 | 398 (82.74) | 41 (51.9) | <0.001 |
| 1 | 121 (21.61) | 96 (21.43) | 25 (22.32) | 0.939 | 83 (17.26) | 38 (48.1) | <0.001 | |
| Stoma Formation | 0 | 509 (90.89) | 407 (90.85) | 102 (91.07) | 1.0 | 449 (93.35) | 60 (75.95) | <0.001 |
| 1 | 51 (9.11) | 41 (9.15) | 10 (8.93) | 1.0 | 32 (6.65) | 19 (24.05) | <0.001 | |
| Lesser Omentum/Stomach Resection | 0 | 534 (95.36) | 427 (95.31) | 107 (95.54) | 1.0 | 468 (97.3) | 66 (83.54) | <0.001 |
| 1 | 26 (4.64) | 21 (4.69) | 5 (4.46) | 1.0 | 13 (2.7) | 13 (16.46) | <0.001 | |
| Gastro-Jejunostomy (Roux-en-Y) | 0 | 558 (99.64) | 447 (99.78) | 111 (99.11) | 0.859 | 480 (99.79) | 78 (98.73) | 0.658 |
| 1 | 2 (0.36) | 1 (0.22) | 1 (0.89) | 0.859 | 1 (0.21) | 1 (1.27) | 0.658 | |
| Groin Node Dissection | 0 | 549 (98.04) | 440 (98.21) | 109 (97.32) | 0.819 | 473 (98.34) | 76 (96.2) | 0.407 |
| 1 | 11 (1.96) | 8 (1.79) | 3 (2.68) | 0.819 | 8 (1.66) | 3 (3.8) | 0.407 | |
| Cholecystectomy | 0 | 553 (98.75) | 442 (98.66) | 111 (99.11) | 1.0 | 479 (99.58) | 74 (93.67) | <0.001 |
| 1 | 7 (1.25) | 6 (1.34) | 1 (0.89) | 1.0 | 2 (0.42) | 5 (6.33) | <0.001 |
| Variable | Overall | Training Set | Testing Set | pvalue (training) | No UAP | UAP | pvalue (UAP) | |
|---|---|---|---|---|---|---|---|---|
| Pelvic Peritonectomy | 0 | 277 (49.46) | 216 (48.21) | 61 (54.46) | 0.281 | 273 (56.76) | 4 (5.06) | <0.001 |
| 1 | 283 (50.54) | 232 (51.79) | 51 (45.54) | 0.281 | 208 (43.24) | 75 (94.94) | <0.001 | |
| Bladder Peritonectomy | 0 | 358 (63.93) | 284 (63.39) | 74 (66.07) | 0.676 | 351 (72.97) | 7 (8.86) | <0.001 |
| 1 | 202 (36.07) | 164 (36.61) | 38 (33.93) | 0.676 | 130 (27.03) | 72 (91.14) | <0.001 | |
| Upper Abdominal Peritonectomy | 0 | 481 (85.89) | 383 (85.49) | 98 (87.5) | 0.693 | 481 (100.0) | 0 (0.0) | <0.001 |
| 1 | 79 (14.11) | 65 (14.51) | 14 (12.5) | 0.693 | 0 (0.0) | 79 (100.0) | <0.001 | |
| Retroperitoneal Abdominal Wall/SMJ Nodule Resection | 0 | 537 (95.89) | 432 (96.43) | 105 (93.75) | 0.312 | 470 (97.71) | 67 (84.81) | <0.001 |
| 1 | 23 (4.11) | 16 (3.57) | 7 (6.25) | 0.312 | 11 (2.29) | 12 (15.19) | <0.001 | |
| Para-aortic node dissection | 0 | 381 (68.04) | 303 (67.63) | 78 (69.64) | 0.768 | 333 (69.23) | 48 (60.76) | 0.172 |
| 1 | 179 (31.96) | 145 (32.37) | 34 (30.36) | 0.768 | 148 (30.77) | 31 (39.24) | 0.172 | |
| Pelvic node dissection | 0 | 414 (73.93) | 335 (74.78) | 79 (70.54) | 0.427 | 363 (75.47) | 51 (64.56) | 0.056 |
| 1 | 146 (26.07) | 113 (25.22) | 33 (29.46) | 0.427 | 118 (24.53) | 28 (35.44) | 0.056 | |
| Salpingo Oophorectomy | 0 | 6 (1.07) | 3 (0.67) | 3 (2.68) | 0.182 | 6 (1.25) | 0 (0.0) | 0.683 |
| 1 | 554 (98.93) | 445 (99.33) | 109 (97.32) | 0.182 | 475 (98.75) | 79 (100.0) | 0.683 | |
| Hysterectomy | 0 | 56 (10.0) | 41 (9.15) | 15 (13.39) | 0.245 | 49 (10.19) | 7 (8.86) | 0.871 |
| 1 | 504 (90.0) | 407 (90.85) | 97 (86.61) | 0.245 | 432 (89.81) | 72 (91.14) | 0.871 | |
| Omentectomy | 0 | 7 (1.25) | 4 (0.89) | 3 (2.68) | 0.296 | 7 (1.46) | 0 (0.0) | 0.594 |
| 1 | 553 (98.75) | 444 (99.11) | 109 (97.32) | 0.296 | 474 (98.54) | 79 (100.0) | 0.594 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





