Submitted:
17 October 2023
Posted:
19 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Detection and Quantification of Serum Antibodies
2.3. ELISpot Assay
2.4. Statistical Analyses
3. Results
3.1. Cohorts and Patient Characteristics at 6 and 12 Months
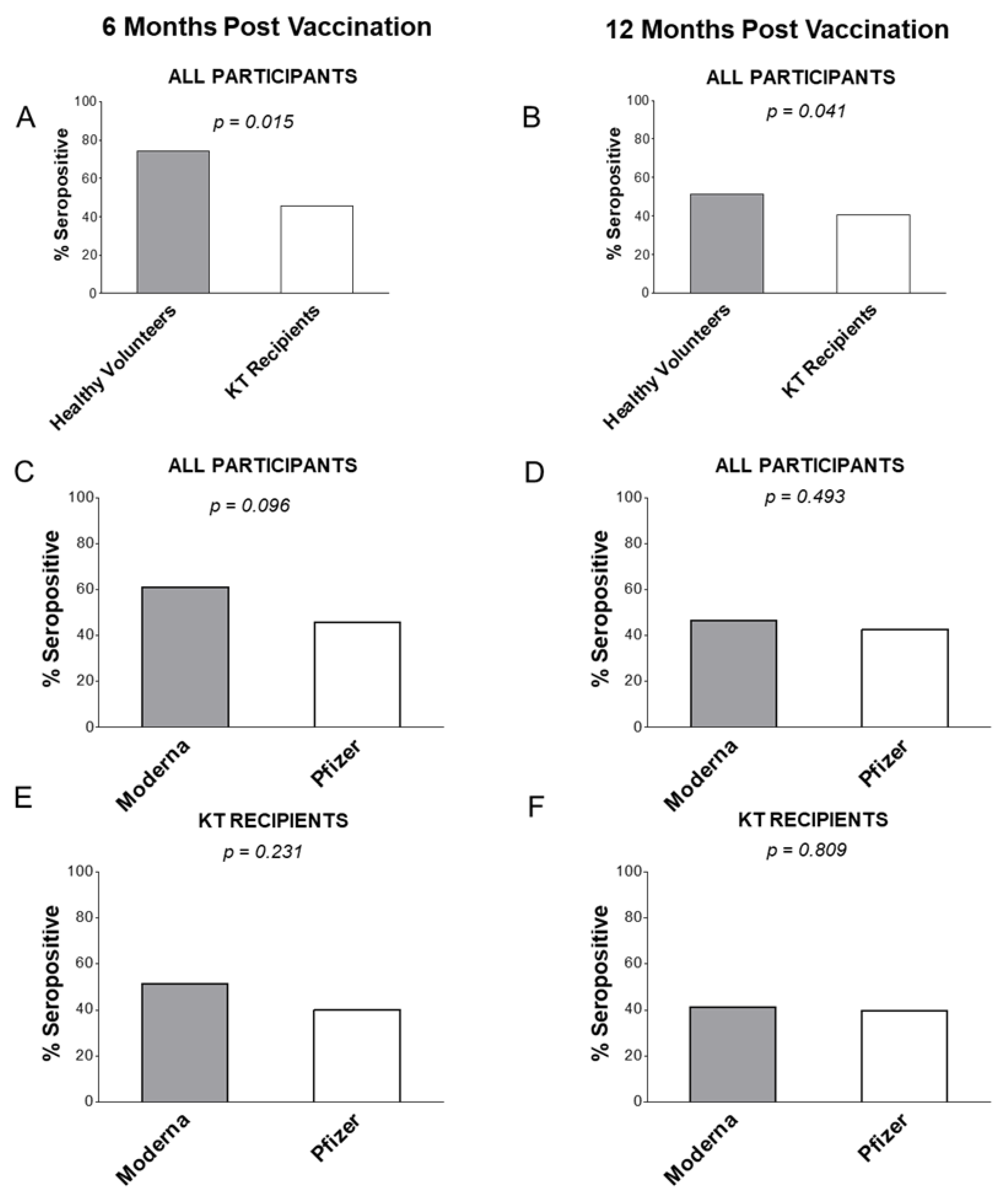
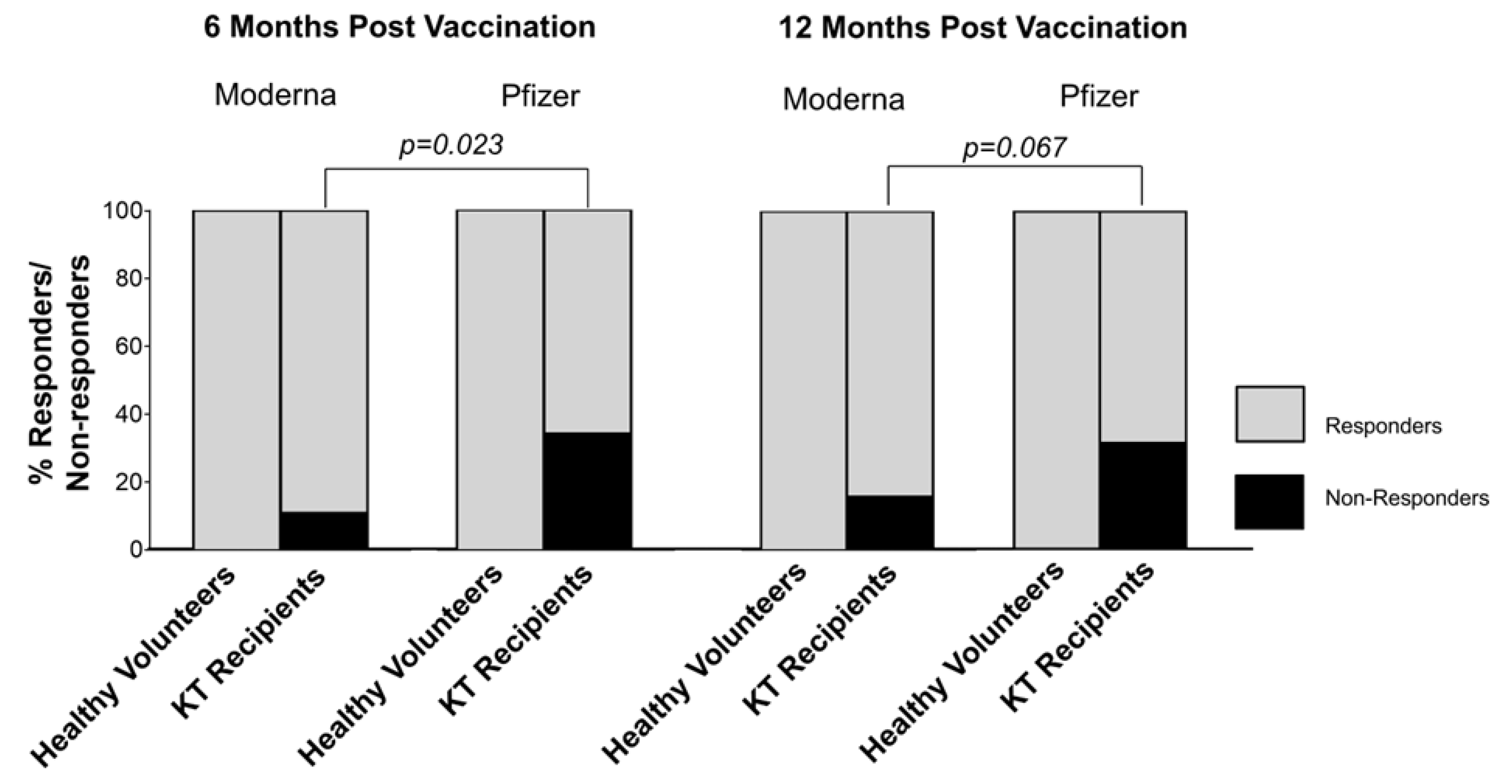
3.2. Anti-SARS-CoV-2 Seropositivity Rates at 6 and 12 Months post Vaccination
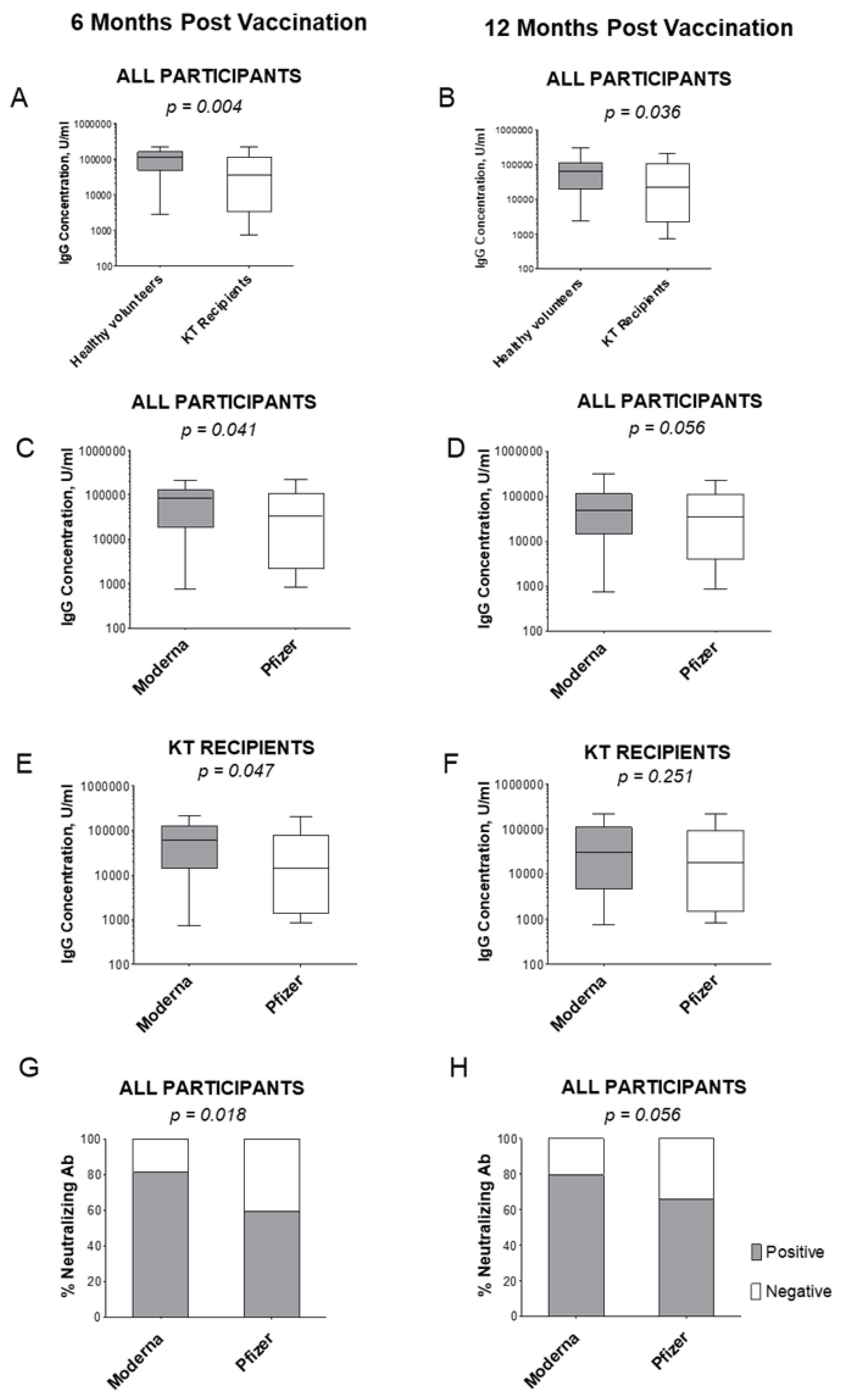
3.3. Quantitative IgG Levels at 6 and 12 Months post Vaccination
3.4. Neutralizing Antibody Frequency at 6 and 12 Months post Vaccination
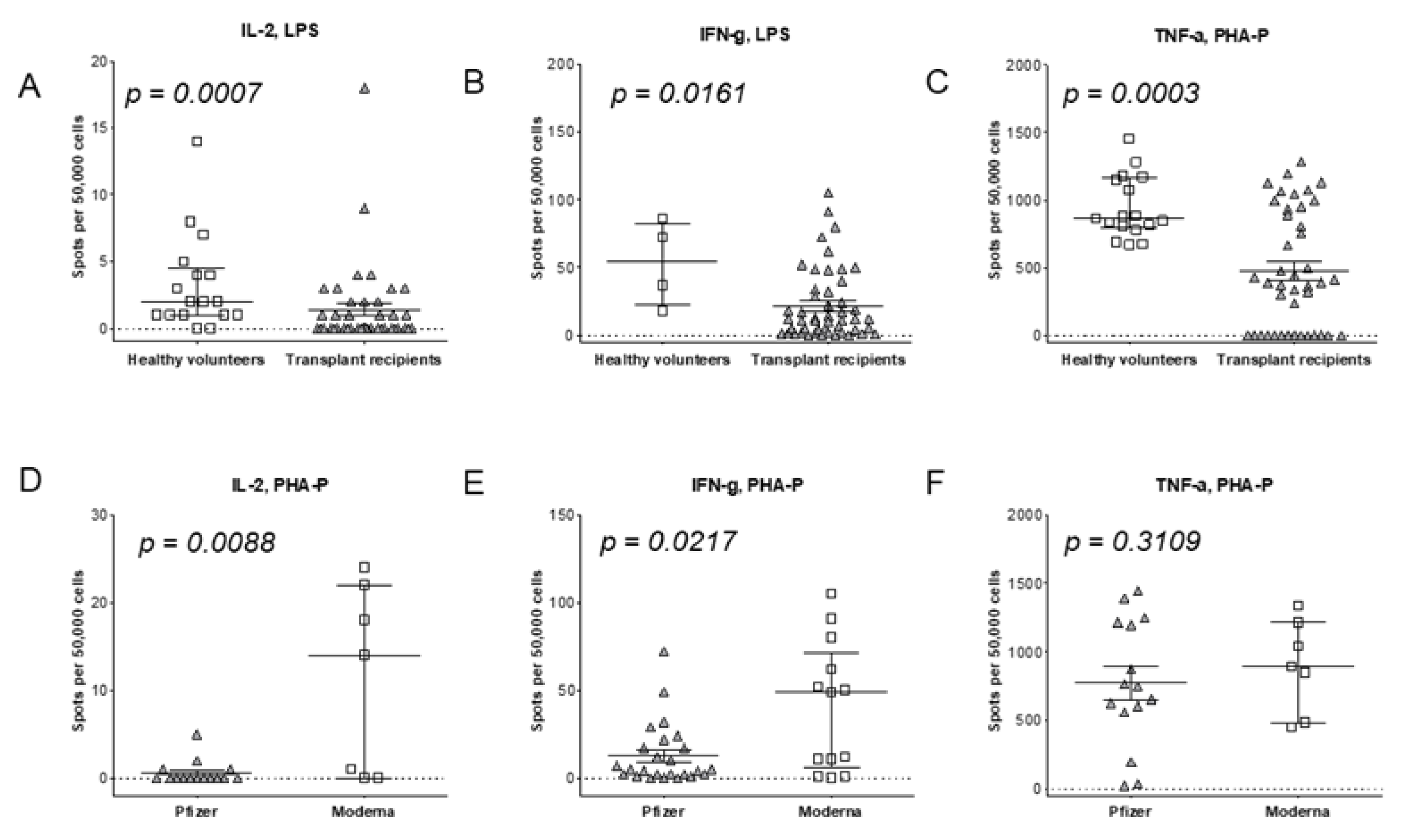
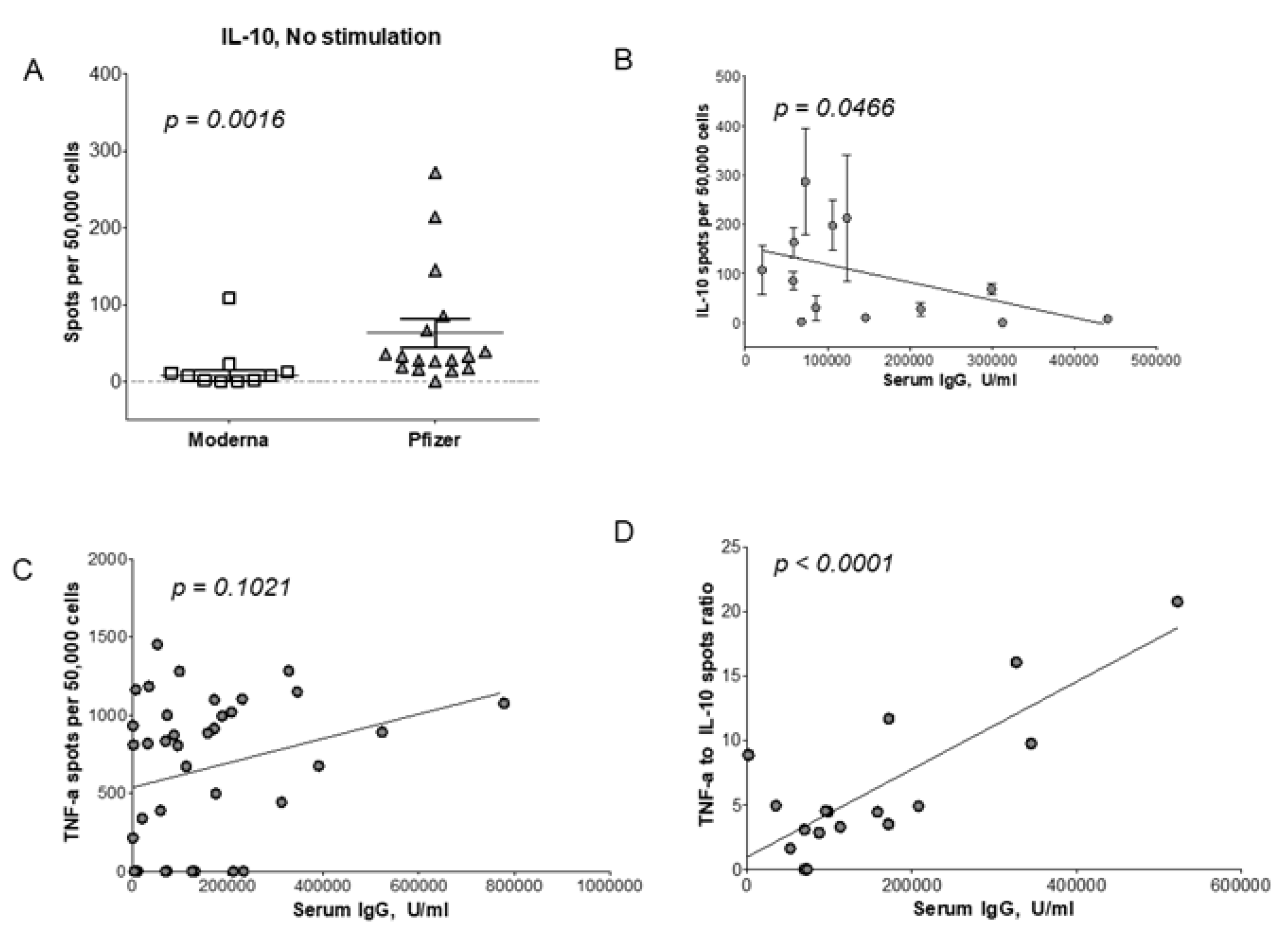
3.5. T Cell Pro-Inflammatory Response to Vaccination
3.6. T cell Regulatory Response to Vaccination
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pollard CA, Morran MP, Nestor-Kalinoski AL. The COVID-19 pandemic: a global health crisis. Physiological Genomics. 2020;52:549-57. [CrossRef]
- Merad M, Blish CA, Sallusto F, Iwasaki A. The immunology and immunopathology of COVID-19. Science. 2022;375:1122-+. [CrossRef]
- Harvey WT, Carabelli AM, Jackson B, Gupta RK, Thomson EC, Harrison EM, et al. SARS-CoV-2 variants, spike mutations and immune escape. Nature Reviews Microbiology. 2021;19:409-24. [CrossRef]
- Fan Y, Li X, Zhang L, Wan S, Zhou FF. SARS-CoV-2 Omicron variant: recent progress and future perspectives. Signal Transduction and Targeted Therapy. 2022;7.
- V'Kovski P, Kratzel A, Steiner S, Stalder H, Thiel V. Coronavirus biology and replication: implications for SARS-CoV-2. Nature Reviews Microbiology. 2021;19:155-70.
- Jackson CB, Farzan M, Chen B, Choe H. Mechanisms of SARS-CoV-2 entry into cells. Nature Reviews Molecular Cell Biology. 2022;23:3-20.
- Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271-+.
- Szabo GT, Mahiny AJ, Vlatkovic I. COVID-19 mRNA vaccines: Platforms and current developments. Molecular Therapy. 2022;30:1850-68. [CrossRef]
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. New England Journal of Medicine. 2020;383:2603-15.
- Sahin U, Muik A, Vogler I, Derhovanessian E, Kranz LM, Vormehr M, et al. BNT162b2 vaccine induces neutralizing antibodies and poly-specific T cells in humans. Nature. 2021;595:572-+. [CrossRef]
- Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. New England Journal of Medicine. 2021;384:403-16.
- Kremer D, Pieters TT, Verhaar MC, Berger SP, Bakker SJL, van Zuilen AD, et al. A systematic review and meta-analysis of COVID-19 in kidney transplant recipients: Lessons to be learned. American Journal of Transplantation. 2021;21:3936-45. [CrossRef]
- Nair V, Jandovitz N, Hirsch JS, Nair G, Abate M, Bhaskaran M, et al. COVID-19 in kidney transplant recipients. American Journal of Transplantation. 2020;20:1819-25.
- Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430-+. [CrossRef]
- Aziz H, Lashkari N, Yoon YC, Kim J, Sher LS, Genyk Y, et al. Effects of Coronavirus Disease 2019 on Solid Organ Transplantation. Transplantation Proceedings. 2020;52:2642-53. [CrossRef]
- Aslam S, Buggs J, Wyatt K, Kumar A, Rogers E, Watson R. The Impact of Virtual Crossmatch on Cold Ischemic Times and Outcomes Following Kidney Transplantation. American Surgeon. 2021;87:109-13. [CrossRef]
- Chandran S, Stock PG. COVID-19 Vaccination in Kidney Transplant Recipients: An Ounce Pre-Transplant is Worth a Pound Post-Transplant. Journal of the American Society of Nephrology. 2021;32:2977-8. [CrossRef]
- Diseases NCfIaR. Interim Clinical Considerations for Use of COVID-19 Vaccines Currently Approved or Authorized in the United States. CDC2022.
- Embi PJ, Levy ME, Naleway AL, Patel P, Gaglani M, Natarajan K, et al. Effectiveness of 2-Dose Vaccination with mRNA COVID-19 Vaccines Against COVID-19-Associated Hospitalizations Among Immunocompromised Adults - Nine States, January-September 2021. Mmwr-Morbidity and Mortality Weekly Report. 2021;70:1553-9.
- Danziger-Isakov L, Kumar D, Practice AIC. Vaccination of solid organ transplant candidates and recipients: Guidelines from the American society of transplantation infectious diseases community of practice. Clinical Transplantation. 2019;33. [CrossRef]
- Dos Santos G, Haguinet F, Cohet C, Webb D, Logie J, Ferreira GLC, et al. Risk of solid organ transplant rejection following vaccination with seasonal trivalent inactivated influenza vaccines in England: A self-controlled case-series. Vaccine. 2016;34:3598-606. [CrossRef]
- Grupper A, Rabinowich L, Schwartz D, Schwartz IF, Ben-Yehoyada M, Shashar M, et al. Reduced humoral response to mRNA SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients without prior exposure to the virus. American Journal of Transplantation. 2021;21:2719-26.
- Boyarsky BJ, Werbel WA, Avery RK, Tobian AAR, Massie AB, Segev DL, et al. Antibody Response to 2-Dose SARS-CoV-2 mRNA Vaccine Series in Solid Organ Transplant Recipients. Jama-Journal of the American Medical Association. 2021;325:2204-6.
- Sattler A, Schrezenmeier E, Weber UA, Potekhin A, Bachmann F, Straub-Hohenbleicher H, et al. Impaired humoral and cellular immunity after SARS-CoV-2 BNT162b2 (tozinameran) prime-boost vaccination in kidney transplant recipients. Journal of Clinical Investigation. 2021;131.
- Korth J, Jahn M, Dorsch O, Anastasiou OE, Sorge-Hadicke B, Eisenberger U, et al. Impaired Humoral Response in Renal Transplant Recipients to SARS-CoV-2 Vaccination with BNT162b2 (Pfizer-BioNTech). Viruses-Basel. 2021;13.
- Danthu C, Hantz S, Dahlem A, Duval M, Ba B, Guibbert M, et al. Humoral Response after SARS-CoV-2 mRNA Vaccination in a Cohort of Hemodialysis Patients and Kidney Transplant Recipients. Journal of the American Society of Nephrology. 2021;32:2154-9.
- Benotmane I, Gautier-Vargas G, Cognard N, Olagne J, Heibel F, Braun-Parvez L, et al. Weak anti-SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients. Kidney International. 2021;99:1487-9.
- Steensels D, Pierlet N, Penders J, Mesotten D, Heylen L. Comparison of SARS-CoV-2 Antibody Response Following Vaccination With BNT162b2 and mRNA-1273. Jama-Journal of the American Medical Association. 2021;326:1533-5.
- Cucchiari D, Egri N, Bodro M, Herrera S, Del Risco-Zevallos J, Casals-Urquiza J, et al. Cellular and humoral response after MRNA-1273 SARS-CoV-2 vaccine in kidney transplant recipients. American Journal of Transplantation. 2021;21:2727-39.
- Jordan SC. Innate and adaptive immune responses to SARS-CoV-2 in humans: relevance to acquired immunity and vaccine responses. Clinical and Experimental Immunology. 2021;204:310-20.
- Wisnewski AV, Luna JC, Redlich CA. Human IgG and IgA responses to COVID-19 mRNA vaccines. Plos One. 2021;16. [CrossRef]
- Meo SA, Bukhari IA, Akram J, Meo AS, Klonoff DC. COVID-19 vaccines: comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. European Review for Medical and Pharmacological Sciences. 2021;25:1663-9. [CrossRef]
- Couper KN, Blount DG, Riley EM. IL-10: The master regulator of immunity to infection. Journal of Immunology. 2008;180:5771-7. [CrossRef]
- Gregori S, Amodio G, Passerini L, de Sio FRS. Alteration of interleukin-10-producing Type 1 regulatory cells in autoimmune diseases. Current Opinion in Hematology. 2022;29:218-24. [CrossRef]
- Bahabayi A, Zeng XY, Tuerhanbayi B, Zhang YY, Hasimu A, Guo SY, et al. Changes in circulating TCF1-and GARP-associated regulatory T cell subsets reflect the clinical status of patients with chronic HBV infection. Medical Microbiology and Immunology. [CrossRef]
- Luke E, Swafford K, Shirazi G, Venketaraman V. TB and COVID-19: An Exploration of the Characteristics and Resulting Complications of Co-infection. Frontiers in Bioscience (Schol Ed). 2022;14:1-11. [CrossRef]
- Safar HA, Mustafa A, Amoudy HA, El-Hashim A. The effect of adjuvants and delivery systems on Th1, Th2, Th17 and Treg cytokine responses in mice immunized with Mycobacterium tuberculosis-specific proteins. Plos One. 2020;15. [CrossRef]
- Elizondo DM, Andargie TE, Haddock NL, da Silva RLL, de Moura TR, Lipscomb MW. IL-10 producing CD8(+) CD122(+) PD-1(+) regulatory T cells are expanded by dendritic cells silenced for Allograft Inflammatory Factor-1. Journal of Leukocyte Biology. 2019;105:123-30. [CrossRef]
- Degner KR, Wilson NA, Reese SR, Parajuli S, Aziz F, Garg N, et al. Short-term Immunopathological Changes Associated with Pulse Steroids/IVIG/Rituximab Therapy in Late Kidney Allograft Antibody Mediated Rejection. Kidney360. 2020;1:389-98.
- Chen W, Bai J, Huang HY, Bi LL, Kong XR, Gao Y, et al. Low proportion of follicular regulatory T cell in renal transplant patients with chronic antibody-mediated rejection. Scientific Reports. 2017;7. [CrossRef]
- Shao CY, Chen YH, Nakao T, Amouzegar A, Yin J, Tahvildari M, et al. Local Delivery of Regulatory T Cells Promotes Corneal Allograft Survival. Transplantation. 2019;103:182-90. [CrossRef]
- Dai HL, Peng FH, Lin MJ, Xia JJ, Yu SJ, Lan GB, et al. Anti-OX40L monoclonal antibody prolongs secondary heart allograft survival based on CD40/CD40L and LFA-1/ICAM-1 blockade. Transplant Immunology. 2015;32:84-91.
- McCallion O, Bilici M, Hester J, Issa F. Regulatory T-cell therapy approaches. Clinical and Experimental Immunology. [CrossRef]
| Variable | Moderna | Pfizer | p-value1 |
|---|---|---|---|
| All Participants (n=99) | |||
| Race Caucasian Other |
36 (73.5%) 13 (26.5%) |
28 (57.1%) 22 (44.9%) |
0.069 |
| Gender Male Female |
28 (57.1%) 21 (42.9%) |
32 (64.0%) 18 (36.0%) |
0.485 |
| Age at consent 35 or less 36 or more Unknown |
15 (30.6%) 34 (69.4%) 0 (0.0%) |
16 (32.0%) 33 (66.0%) 1 (2.0%) |
0.828 |
| Age at consent, years (average) | 59.4 | 56.3 | 0.177 |
| Vaccine doses 2 + boosting dose |
19 (38.8%) 30 (61.2%) |
22 (44.0%) 28 (56.0%) |
0.598 |
| Healthy Volunteers (n=27) | |||
| Race Caucasian Other |
8 (50.0%) 4 (36.4%) |
8 (50.0%) 7 (63.6%) |
0.276 |
| Gender Male Female |
4 (36.4%) 8 (50.0%) |
7 (63.6%) 8 (50.0%) |
0.484 |
| Age at consent 50 or less 51 or more Unknown |
5 (38.5%) 7 (53.9%) |
8 (61.5%) 6 (46.1%) 1 (100.0%) |
0.431 |
| Vaccine doses 2 + boosting dose |
4 (44.4%) 8 (44.4%) |
5 (55.6%) 10 (55.6%) |
0.986 |
| Transplant Recipients (n=72) | |||
| Race Caucasian Other |
28 (75.7%) 9 (24.3%) |
20 (57.1%) 15 (42.9%) |
0.095 |
| Race African American Other |
7 (18.9%) 30 (81.1%) |
9 (25.7%) 26 (74.3%) |
0.488 |
| Donor type Living Deceased |
6 (15.8%) 31 (84.2%) |
3 (8.6%) 32 (91.4%) |
0.349 |
| Gender Male Female |
24 (64.9%) 13 (35.1%) |
25 (71.4%) 10 (28.6%) |
0.551 |
| BMI 29 or less 30 or more Unknown |
20 (55.1%) 15 (40.5%) 2 (5.4%) |
17 (48.6%) 17 (48.6%) 1 (2.8%) |
0.642 |
| Age at consent 35 or less 36 or more |
10 (27.0%) 27 (73.0%) |
8 (22.9%) 27 (77.1%) |
0.683 |
| Age at consent, years (average) |
58.9 |
58.8 |
0.490 |
| Vaccine doses 2 + boosting dose |
15 (40.5%) 22 (59.5%) |
17 (48.6%) 18 (51.5%) |
0.493 |
| Antimetabolites use Yes No Unknown |
30 (81.1%) 6 (16.2%) 1 (2.6%) |
29 (82.9%) 5 (14.3%) 1 (2.8%) |
0.592 |
| CNI use Yes No |
34 (89.5%) 4 (10.5%) |
34 (100%) 0 (0%) |
0.052 |
| Prednisone use Yes No Unknown |
30 (81.1%) 6 (16.2%) 1 (2.6%) |
29 (82.9%) 5 (14.3%) 1 (2.8%) |
0.636 |
| Group | Positive | Negative* | p-value* |
|---|---|---|---|
| All participants (n=99) | |||
| Gender Male Female |
30 (50.0%) 23 (60.0%) |
30 (50.0%) 16 (40.0%) |
0.396 |
| Age 50 years or younger 51 years or older |
18 (58.1%) 35 (50.8%) |
13 (41.9%) 33 (49.2%) |
0.408 |
| Race Caucasian Other |
33 (51.6%) 20 (54.1%) |
29 (48.4%) 17 (45.9%) |
0.600 |
|
Healthy volunteers (n=27) | |||
| Gender Male Female |
9 (81.8%) 11 (68.8%) |
2 (18.2%) 5 (31.2%) |
0.614 |
| Age 50 years or younger 51 years or older |
8 (61.5%) 12 (85.7%) |
5 (38.5%) 2 (14.3%) |
0.343 |
| Race Caucasian Other |
11 (68.8%) 9 (56.3%) |
5 (31.2%) 7 (43.7%) |
0.888 |
|
Transplant recipients (n=72) | |||
| Gender Male Female |
21 (43.8%) 12 (52.1%) |
28 (56.2%) 11 (47.9%) |
0.555 |
| Age 35 years or younger 36 years or older |
23 (42.6%) 10 (55.6%) |
31 (57.4%) 8 (44.4%) |
0.407 |
| Race Caucasian Other |
22 (45.8%) 11 (44.0%) |
26 (54.2%) 13 (56.0%) |
0.936 |
| Vaccine doses 2 + boosting dose |
13 (52.0%) 20 (54.1%) |
22 (48.0%) 17 (45.9%) |
0.220 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




