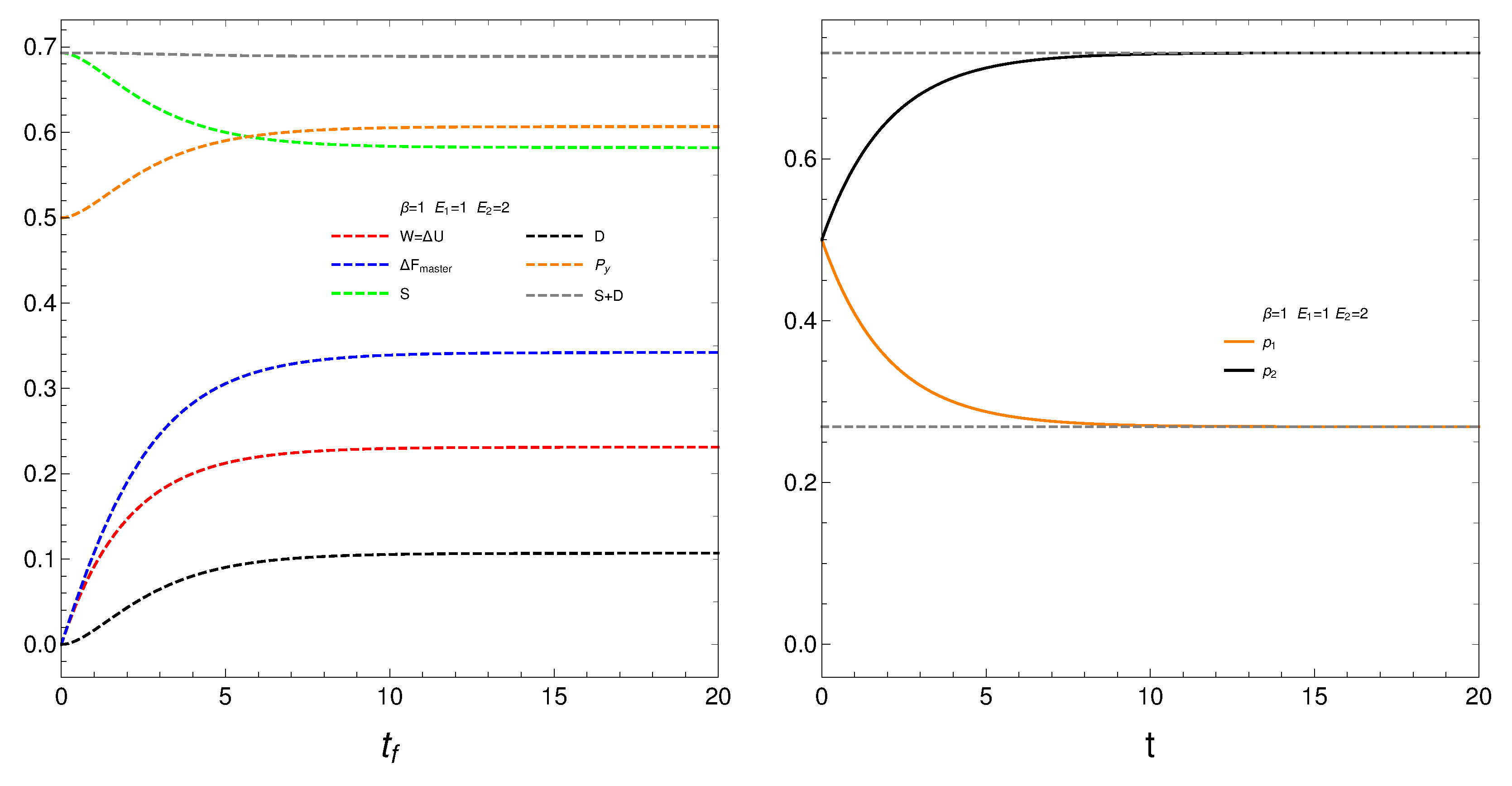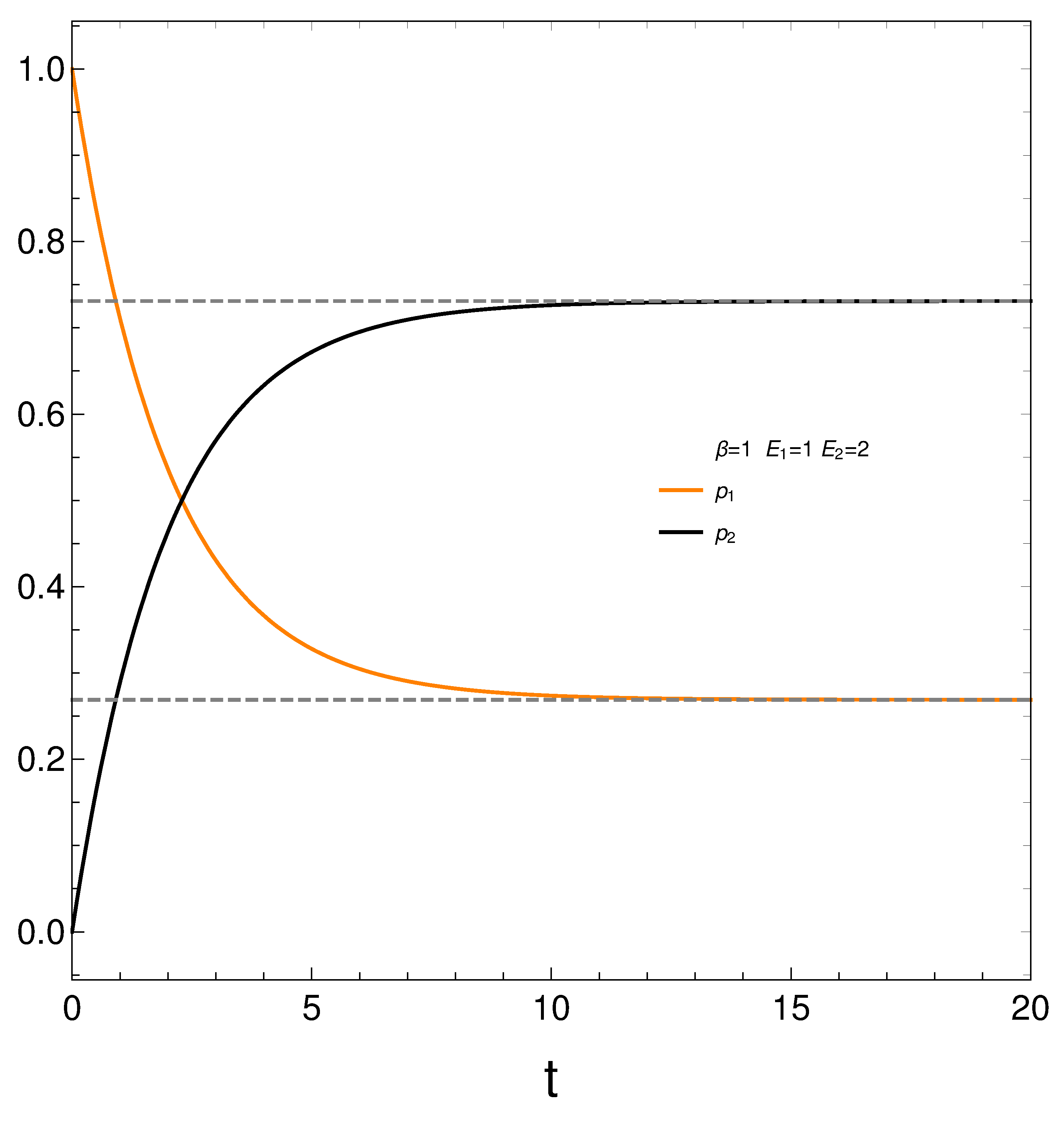1. Preliminaries
Encountering phase transitions in an elementary exactly solvable master equation can be of significant interest in various scientific fields, particularly in statistical physics and complex systems. Phase transitions are, of course, crucial phenomena that mark abrupt changes in the behavior of a system as external parameters, such as temperature or density, are varied. These transitions have been extensively studied because they reveal important insights into the collective behavior of particles and the emergence of novel properties in many physical and non-physical systems. This is why the findings to be described below may be noteworthy.
1.1. Master equations
Investigating master equations is of significant importance in the fields of quantum mechanics, quantum optics, quantum information theory, and various other areas of physics and science. Below we list several key reasons why the study of master equations is essential:
1) Modeling Quantum Systems: Master equations provide a mathematical framework for modeling the dynamics of open quantum systems. They allow scientists to describe how a quantum system evolves over time when it is interacting with its environment. This is crucial for understanding and predicting the behavior of complex quantum systems.
2) Quantum Open Systems: Many physical systems are not isolated but interact with their surroundings. Master equations are indispensable for describing and simulating open quantum systems, where information and energy can flow between the system and its environment. Examples include atoms interacting with light, electrons in a conducting material, and qubits in a quantum computer.
3) Decoherence and Dissipation: Understanding how quantum coherence is lost due to interactions with the environment (decoherence) and how energy dissipates in quantum systems is crucial for quantum technologies. Master equations allow researchers to quantify these processes, which is essential for designing and maintaining quantum devices.
4) Quantum Information and Quantum Computing: In the field of quantum information theory, master equations are fundamental for understanding and mitigating errors in quantum computation and communication. They help researchers develop error-correction codes and quantum error-correcting schemes.
5) Quantum Optics and Photonics: In quantum optics and photonics, master equations are used to study the behavior of light and quantum states of light in various physical systems. This has applications in quantum communication, quantum cryptography, and quantum sensing.
1.2. Very simple master equations
Studying a very simple master equation with all parameters having analytic expressions can be relevant and beneficial in several ways:
Pedagogical Purposes: Simple master equations with analytic solutions are excellent for educational purposes. They help students and researchers gain a fundamental understanding of the key concepts in quantum mechanics and open quantum systems without the added complexity of numerical or approximate methods.
Theoretical Foundations Simple master equations serve as building blocks for more complex models. By starting with analytic solutions, you can develop a solid theoretical foundation for understanding quantum systems. This can be particularly valuable when working with more complicated systems, as you can use the insights gained from the simple case to inform your approach.
Benchmarking: Simple models can be used as benchmarks for numerical or approximate methods. By comparing the results of more sophisticated techniques to the exact analytical solutions of a simple master equation, you can verify the accuracy of your computational methods and identify any potential sources of error.
Intuition and Insight: Analytic solutions provide deep insight into the behavior of quantum systems. They allow for a clear and intuitive understanding of how different parameters affect the system’s dynamics, which can be especially useful for physical intuition and insight into more complex systems.
Generalizations: Simple models can serve as a starting point for more generalized models. Once you understand the basic dynamics of a simple system, you can extend your knowledge to tackle more complex models by adding additional features, interactions, or degrees of freedom.
Real-world Applications: While the simplest models may not directly correspond to real-world physical systems, they can serve as a foundation for understanding and modeling more complex and realistic scenarios. Real systems can often be understood as perturbations or extensions of these idealized cases.
Exploration of Fundamental Principles: Simple models can be used to explore fundamental principles in quantum mechanics and open quantum systems. This can lead to new insights and discoveries, even if the model itself is highly idealized.
In summary, studying a simple master equation with analytic solutions is a valuable starting point in quantum mechanics and open quantum systems. It can provide foundational knowledge, benchmarking capabilities, and a clear understanding of how fundamental parameters affect the system’s behavior. This understanding can then be applied to more complex and realistic scenarios, making it a relevant and useful exercise in various research and educational contexts.
2. Present concerns
A master equation (ME) is a phenomenological set of first-order differential equations specifying the temporal evolution of the probability of a given (let us call it A) system to occupy each one of a discrete set of states. The variable is the time t. We speak of an equation of motion for the occupation probabilities of A with the underlying supposition that A interacts with a usually much larger system B.
2.1. Order-disorder disjunction
Analyzing the order-disorder disjunction is of fundamental importance in various scientific disciplines, including physics, chemistry, material science, and even in broader contexts like philosophy and social sciences. Here are several reasons why analyzing the order-disorder disjunction is significant. 1) Understanding the transition between ordered and disordered states is central to thermodynamics. It plays a crucial role in understanding phase transitions, like the transition between solid, liquid, and gas phases. The study of order-disorder transitions is essential for predicting and controlling these transitions. 2) In material science, the arrangement of atoms and molecules in a material can profoundly impact its properties. Analyzing the order-disorder disjunction is vital for the development of new materials with tailored properties, including semiconductors, superconductors, and advanced polymers. 3) In the realm of statistical mechanics, the order-disorder disjunction is a key consideration. This field provides a framework for understanding how macroscopic properties emerge from the behavior of individual particles. Understanding order and disorder at the microscale is vital in statistical mechanics. 4) Biological systems are rife with examples of the order-disorder disjunction. For instance, the folding of proteins into specific, ordered structures is crucial for their function. Understanding how biological systems maintain order and manage disorder is essential in fields like biochemistry and molecular biology. 5) In information theory and coding, error correction codes and data compression techniques rely on managing the trade-off between order and disorder in data transmission and storage. Analyzing this disjunction is critical for reliable communication and data storage. 6) The emergence of order from disorder and vice versa is a topic of interest in understanding emergent properties in complex systems. Emergence is a fundamental concept in many fields, including philosophy, physics, and biology.
2.2. Entropy, disequilibrium, and our ME
Entropy is a common synonym from disorder. On the other hand , for almost three decades the notion of disequilibrium, to be explained below, has come to represent the degree of order. We wish here to analyze using a simple ME the order-disorder question.
Possibly the simplest ME faces two states with (1) probabilities (
P) components
and
and (2) energies
and
. We will here study a ME whose final stationary state is a Gibbs’ canonical one at the inverse temperature
. Some valuable insight will be gained. In this respect an interesting order-indicator is the disequilibrium
D, which, in probability space, represents the Euclidean distance between
P and the uniform distribution
with
This uniform distribution is associated with maximum disorder. The larger the distance between
P and
, the larger the degree of order, according to references [
1,
2,
3,
4,
5,
6,
7,
8].
Our very simple ME reads [
9,
10]
with
,
, and
.
is the inverse temperature
. We set Boltzmann’s constant equal to unity.
The exact solution of the system is
where
is an appropriate constant. It depends on the initial conditions at
. For any other
t one has
.
The stationary solution for large
t has the form
where
Z denotes the partition function, that is [
11]
We clearly see that a canonical Gibbs’ distribution is eventually attained with our p’s.
Let
Note that, in general, this master equation represents a process that is not stationary. A process that is not stationary implies that there are ongoing changes in the system. For example, the system’s properties may be changing with time, or there may be gradients in temperature, pressure, or other thermodynamic variables within the system. Such a system is not in thermodynamic equilibrium. However, the we will use here leads eventually to stationarity.
3. Time evolution of our quantifiers
In analyzing our ME, time is measured always in arbitrary units.
We begin looking at the time evolution of our two probabilities in
Figure 1. We see that they eventually cross each other, which anticipates an interesting thermodynamics.
We begin with
and
.
Figure 1 depicts the behavior of our two probabilities for the initial conditions
and
versus time in arbitrary units. They cross at a time
. Here we have
,
,
, and
. Note that stationarity is eventually reached.
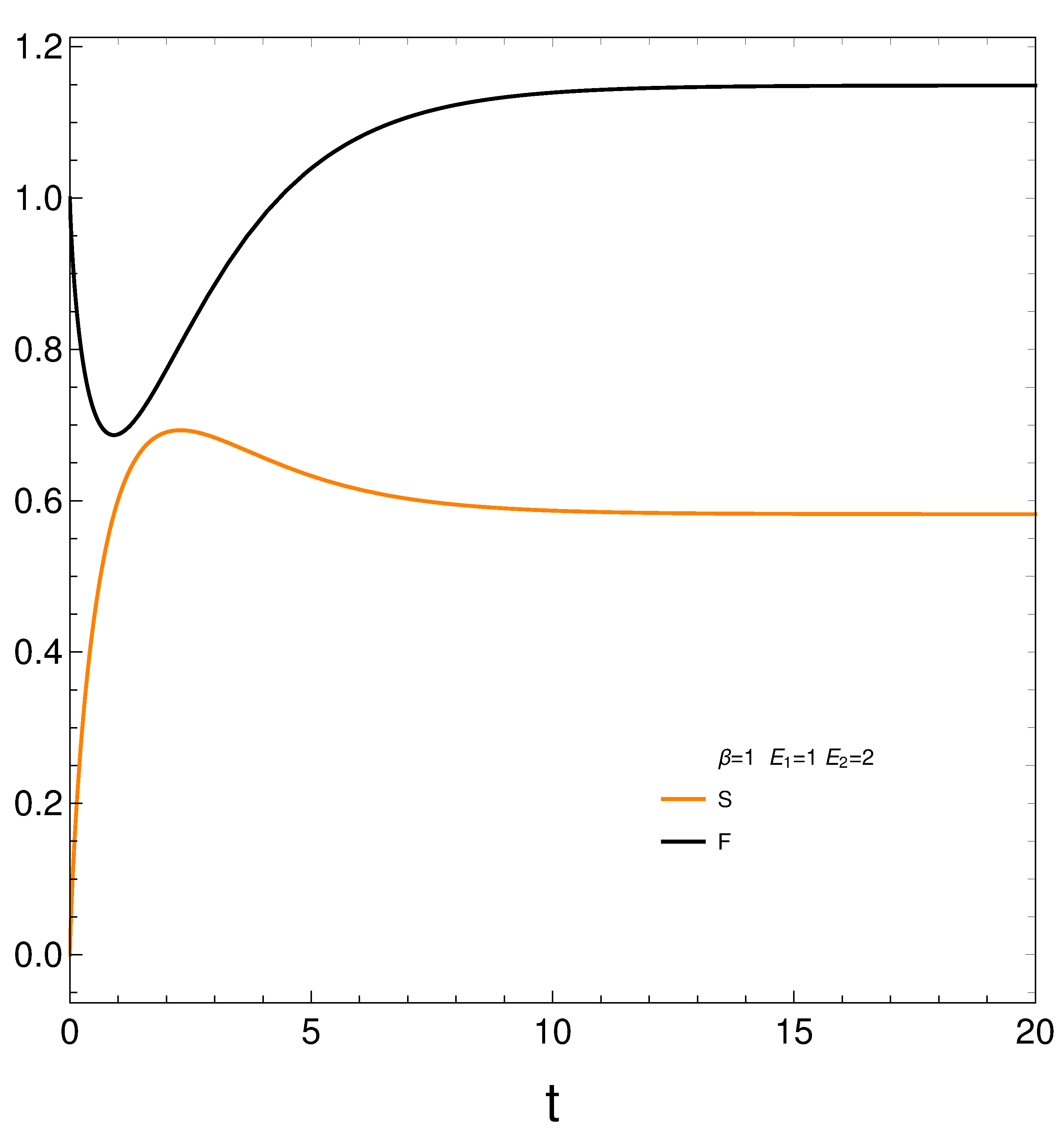
We compute next the entropy
S , Helmholtz free energy
F, and mean energy
U from their definitions and then plot the first two of them in
Figure 2. We have [
11]
Remind that the lower the free energy of a system, the more stable it tends to be since spontaneous changes involve a decrease in
F. Results are given in
Figure 2. Notice that the system reaches a minimum value for
F in black (maximum stability) at a certain time
. However, the master equation mechanism takes the system away from the minimum till it reaches stationarity.We discover that our master equation mechanism prefers stationarity over stability. Not surprisingly, the system reaches maximum entropy
S (color orange, disorder) at a time close to
(see above).
It seems that our master equation describes a process of initially growing of the entropy and the useful available energy followed by one of equilibration at higher values of both S and F.
the mean energy difference ,
the difference of the free energy ,
the disequilibrium ,
the purity (degree of mixing in the quantum sense) [, with for a pure state,
the entropy S, and
the sum .
Remark that is almost constant as we approach the stationary zone, which reinforces our assertions above about D-representing order. Thus, we note that the sum of order plus disorder indicators approaches a constant value as we get near stationarity.
The entropic and free energy behaviors are those displayed in
Figure 2 above. The disequilibrium (order indicator) attains its minium when
S gets maximal, as expected. The purity starts being maximal at
, descends first afterwards, and then slightly increased till it reaches stationarity. Thus, our ME transforms a pure state into a stationary mixed one. The ME is able to yield useful free energy only for a very short time in which it creates disorder.
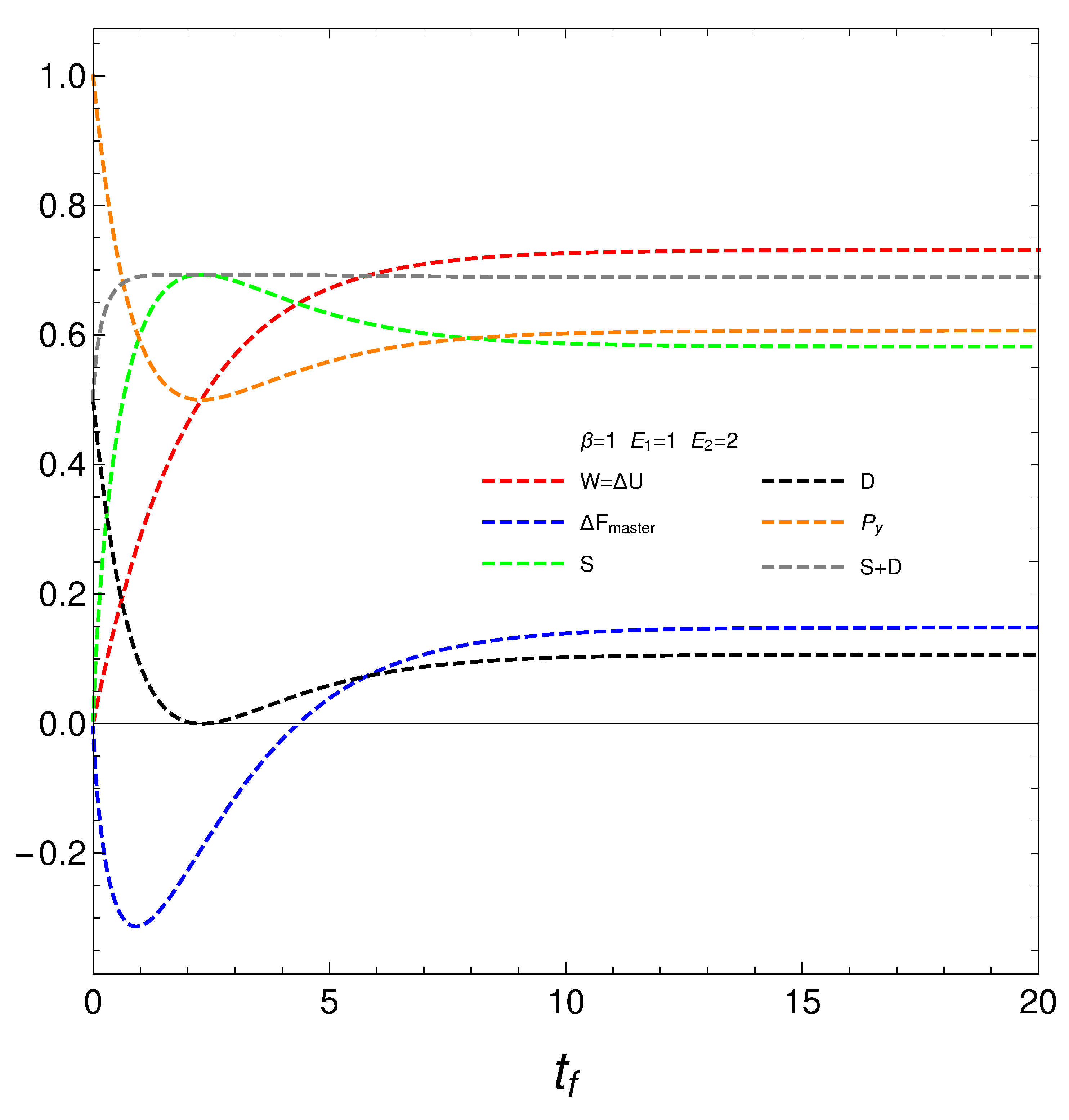
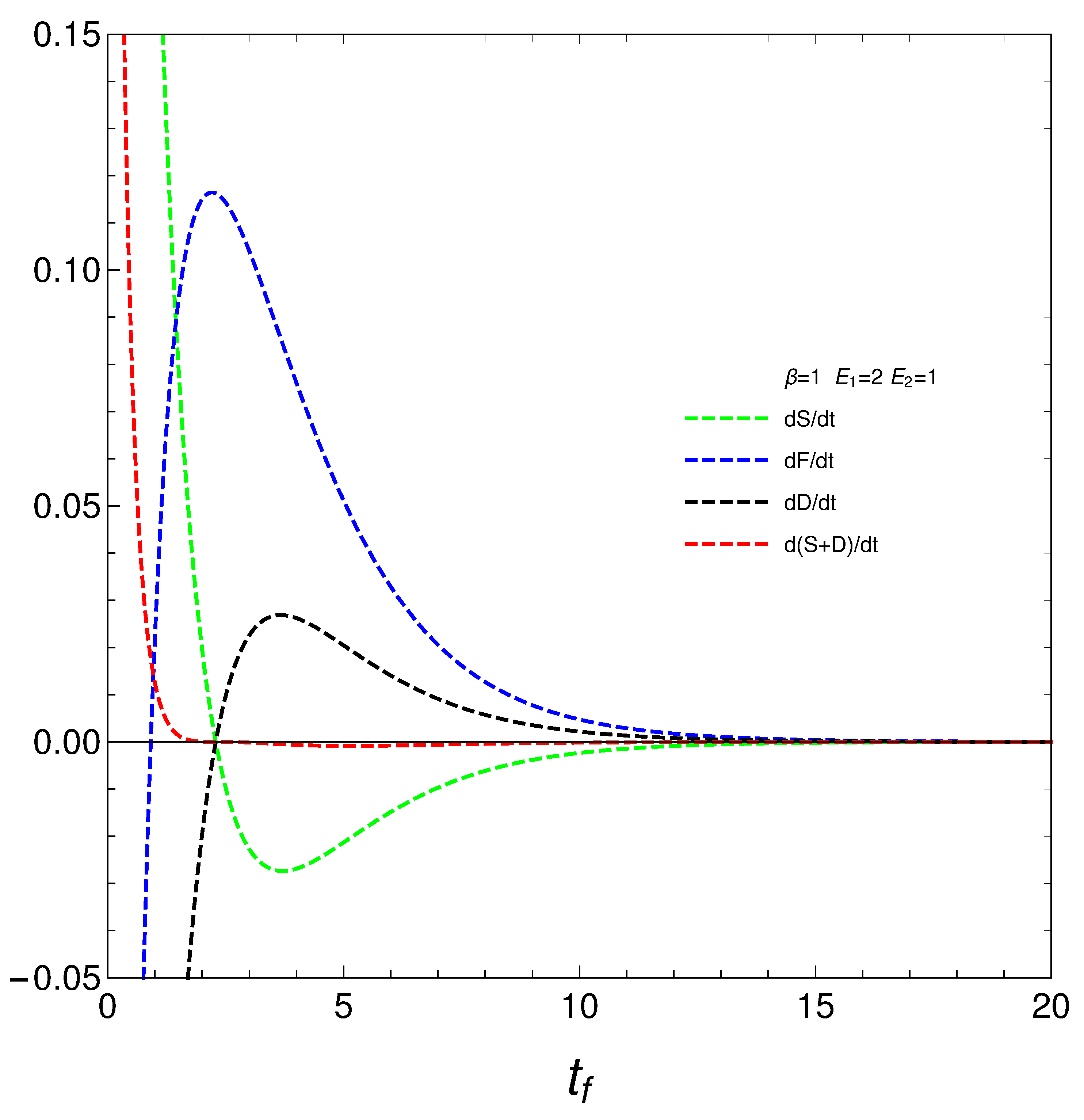
We pass now to plot in
Figure 4, versus final time
, the derivatives
,
,
.
4. Phase transitions
We reach the core of our presentation at this stage. Consider now the novel quantities
that determine how much free energy is expended o received so as to change either the disequilibrium or the entropy. We encounter a surprise in
Figure 5. Our
’s, that the free energies required to effect our changes diverge at the time at which
where the entropy is a maximum, that we called
. The divergence at this time indicate a temporary instability or singularity, and the
’s settle into a new, well-behaved regime after this time. They stabilize at a finite value, representing a new equilibrium or steady state. What happens at
is that the system was evolving to reach energetic equilibrium and maximum entropy before this time. The master equation, however, forces the system to leave that equilibrium to evolve toward statioarity. Thus, we face two distinct regimes.
At the time of regime-change we have a phase transition. Our simple master equation hides a powerful mechanism that is able to produce such regime-change at considerable energetic cost.
Our master equation displays a phase transition in our novel quantities when the entropy attains a maximum.
5. Initial conditions of maximum entropy
Given its great importance, consider in more detail the solutions of our master equation for at . The results for these maximum entropy initial conditions (MSIC) are plotted below. The entropy, of course, descends and the remaining quantifiers increase till reaching stationarity.
Figure 6.
MSIC. Thermal quantities versus time. One sets , and . The entropy diminishes. The remaining quaifiers incresae. Stationarity is reached.
Figure 6.
MSIC. Thermal quantities versus time. One sets , and . The entropy diminishes. The remaining quaifiers incresae. Stationarity is reached.
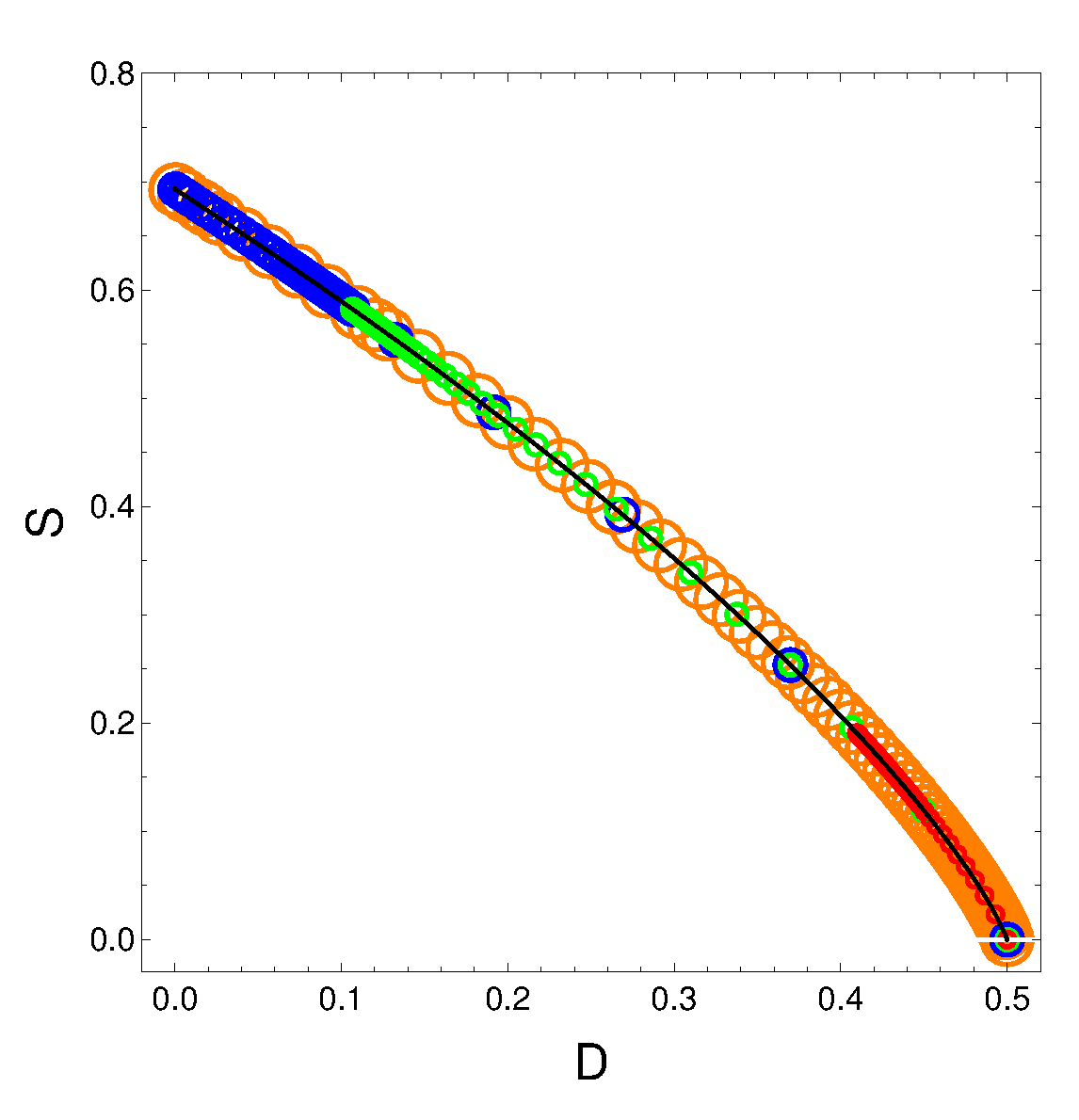
6. The order(D)-disorder(S) equation generated by the master equation
From its definition
and from the relation
, we gather that
and
, with
From the entropic definition one then finds
SvsD which turns out to be, for any values of
,
, and
the rather beautiful expression that expresses the entropy solely in terms of the order quantifier:
The above relation is independent of the and of . We plot versus with the time as parameter. The solid curve represents the equation . Of course, if the order is maximal the entropy vanishes. The circles represent specific parameters’ values of the energies in the following fashion:
orange: ;
blue: ;
green: ;
red: .
It is clear that S decreases as D grows. But this does not happen neither linearly nor in complicated manner.
Figure 7.
SvsD for variegated selection of the parameters that is detailed in the text. Also, and .
Figure 7.
SvsD for variegated selection of the parameters that is detailed in the text. Also, and .
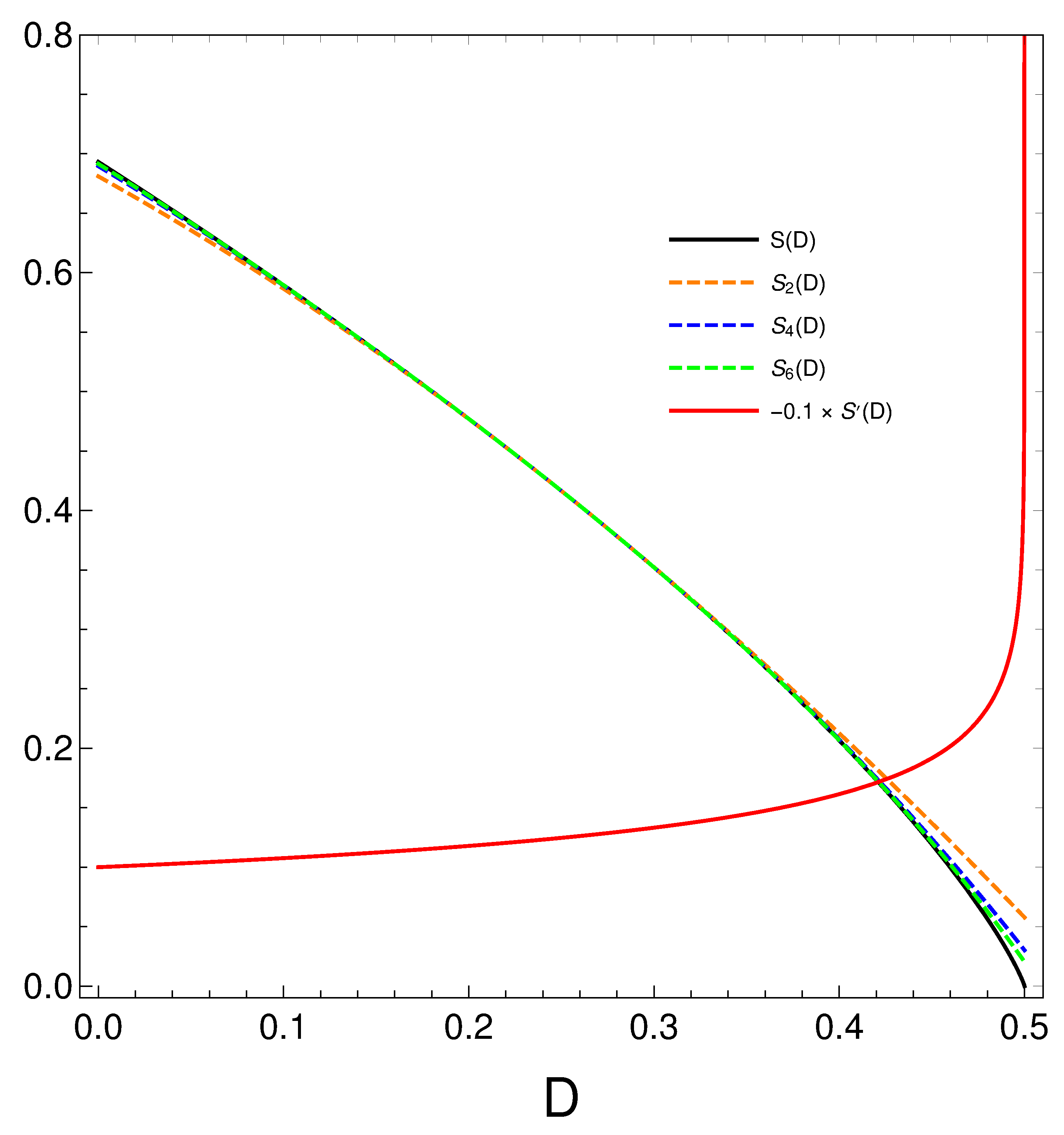
6.1. Polynomial approximations for the relation S versus D
We pay now to a closer look to the order-disorder relation that our master equation displays. We proceed thus to make a Taylor expansion of
around
and obtain
We plot below (black) and minus its derivative (red) [in fact, )]. Orange, blue, and green dashed curves represent, respectively, approximations to of degree two, four, and six. Note that the relation disorder versus order looks quasi linear in some region, but not quite of course. When the order-degree gets near its maximum value of one half, the disorder diminishes in a quite rapid fashion, as the derivative plot illustrates. Summing up, the order-disorder relation for our master equation displays some quirky traits.
Figure 8.
Polynomial approximations to the relation of orders two, four, and six in different colors. The exact is given in black. We also depict in orange.
Figure 8.
Polynomial approximations to the relation of orders two, four, and six in different colors. The exact is given in black. We also depict in orange.
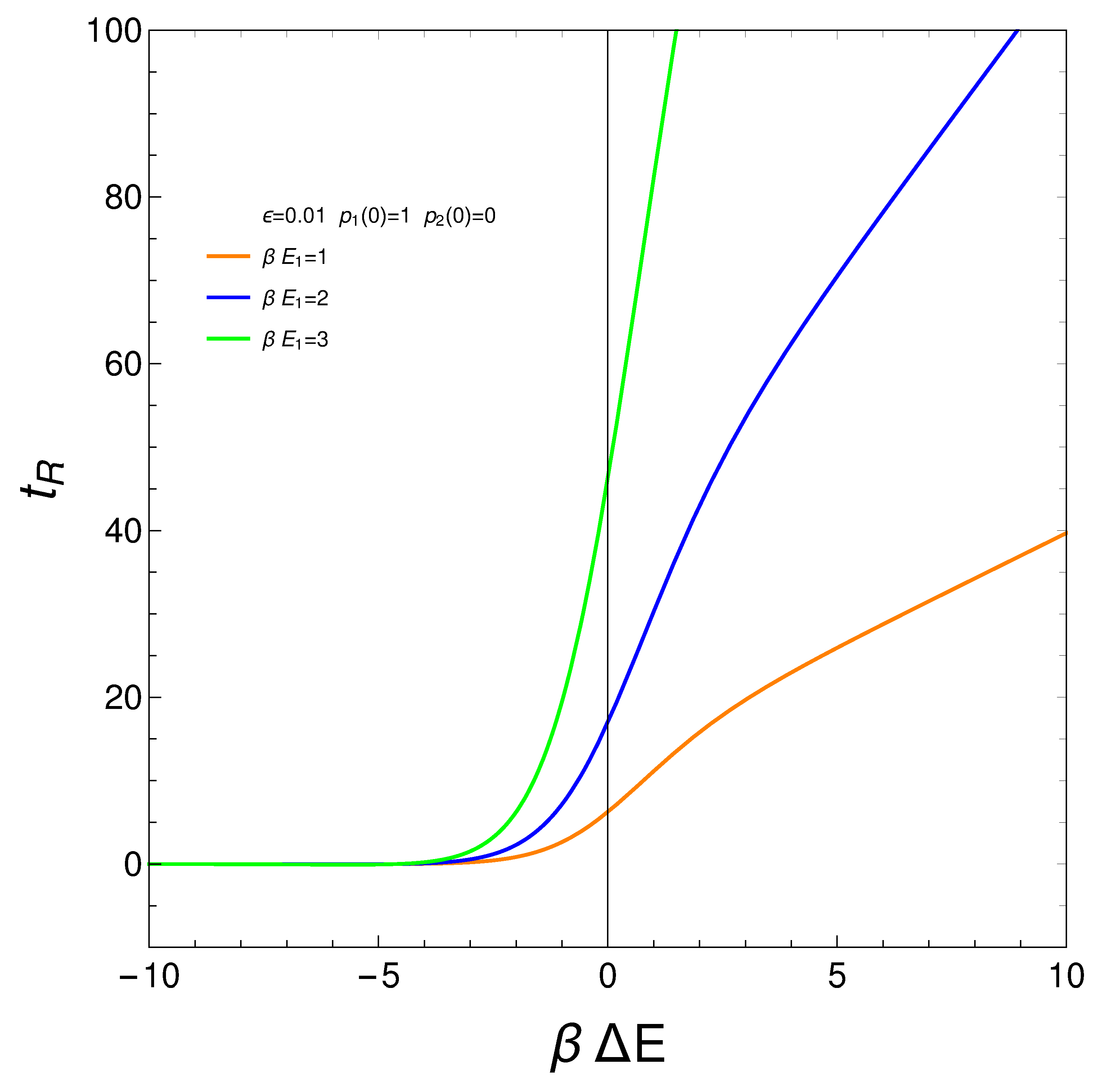
7. Relaxation time
We define our system’s relaxation
as the time-interval needed to reach stationarity so that the
no longer display significant changes. We can require, for instance, that
, with
. Thus, from Eqs. (3) and (4), we gather that
where
. The relation is displayed next for different values of the pre factor
. The initial conditions are
. For a considerable range of
the relaxation time is independent of this quantity.
Figure 9.
vs for . is the approximation’s degree (see text). Save for very small (negative) values of the relaxation time is constant. If g becomes small augments. This fact is of interest for high temperatures, of course. It makes sense that for such temperatures it should become more difficult to reach equilibration. Positive values of make little sense in the context of our master equation.
Figure 9.
vs for . is the approximation’s degree (see text). Save for very small (negative) values of the relaxation time is constant. If g becomes small augments. This fact is of interest for high temperatures, of course. It makes sense that for such temperatures it should become more difficult to reach equilibration. Positive values of make little sense in the context of our master equation.
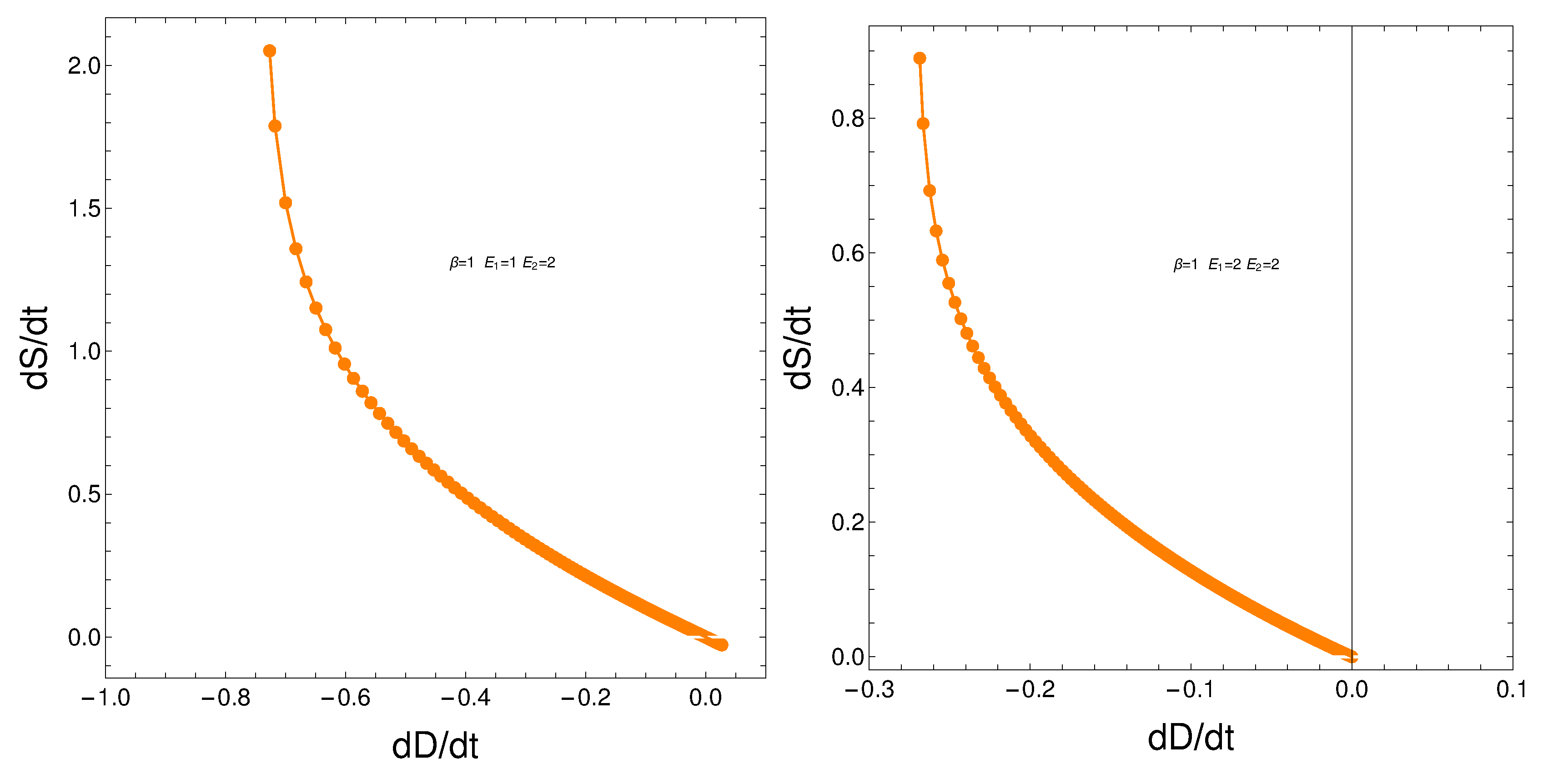
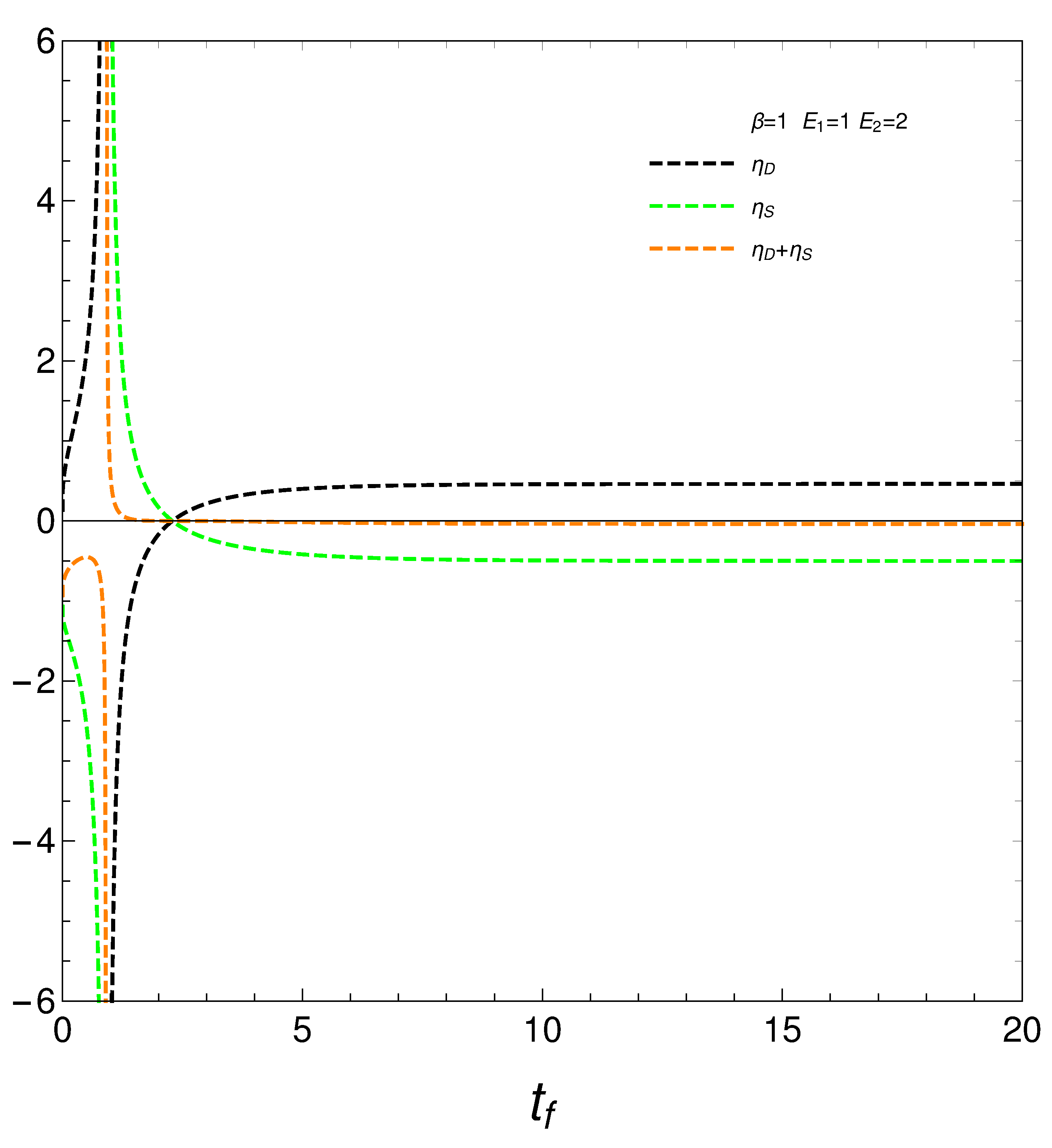
8. Relating the derivatives and
From our formalism we immediately obtain the important relation
This relation between the rates of change of order and disorder is illustrated next. These rates are positively correlated, as one intuitively expects. We consider two situations: a) , and b) . No important differences ensue between a) and b).
Figure 10.
versus for and - . Left: and . Right: and .
Figure 10.
versus for and - . Left: and . Right: and .
9. Conclusions
We have studied in detail a very simple master equation (ME) that displays a complex physics, as the divergences depicted in
Figure 5 illustrate. Our present ME hides a powerful mechanism that is able to produce a phase transition associated to a regime-change at considerable energetic cost.. This ME simplicity allows for easy access to recondite facts of the pertinent process.
Our master equation describes a process of initial growth of both the entropy and the useful available energy followed by one of equilibration at higher values of both S and F. Note that near stationarity order balances disorder.
We represent the notion of order by the disequilibrium D and (of course) disorder by the entropy S. Surprisingly enough we find that their sum tends to be a constant for much of the time under a variety of conditions. Since the meaning of S has been established more than a century ago, or results give new validity to the newer notion of disequilibrium.
Figure 8 shows that the degree of disorder greatly accelerates itself as D tends towards its maximum possible value. For smaller D values the rate of S growth is rather uniform.
Simple exactly solvable models are often used as starting points for understanding more complex systems. Our discovery of phase transitions here can serve as a model system to investigate the nature of phase transitions, their critical exponents, and scaling behavior. This knowledge might be applied to more intricate systems in the future. Discovering phase transitions in an elementary exactly solvable master equation can also have methodological importance. It demonstrates that our analytical and modeling techniques are capable of capturing non-trivial collective behaviors and can be applied to other systems.
References
- R. López-Ruiz, H. Mancini, X. Calbet, A statistical measure of complexity. Phys. Lett. A 209 (1995) 321-326.
- R. López-Ruiz, Complexity in some physical systems, International Journal of Bifurcation and Chaos 11 (2001) 2669-2673.
- M.T. Martin, A. Plastino, O.A. Rosso, Statistical complexity and disequilibrium, Phys. Lett. A 311 (2003) 126-132.
- L. Rudnicki, I.V. Toranzo, P. Sánchez-Moreno, J.S. Dehesa, Monotone measures of statistical complexity, Phys. Lett. A 380 (2016) 377-380.
- R. López-Ruiz, A Statistical Measure of Complexity in Concepts and recent advances in generalized infpormation measures and statistics, A. Kowalski, R. Rossignoli, E.M.C. Curado (Eds.), Bentham Science Books, pp. 147-168, New York, 2013.
- K.D. Sen (Editor), Statistical Complexity, Applications in elctronic structure, Springer, Berlin 2011.
- M.T. Martin, A. Plastino, O.A. Rosso, Generalized statistical complexity measures: Geometrical and analytical properties, Physica A 369 (2006) 439-462.
- C. Anteneodo, A.R. Plastino, Some features of the López-Ruiz-Mancini-Calbet (LMC) statistical measure of complexity, Phys. Lett. A 223 (1996) 348-354.
- Kryszewski, S. and Czechowska-Kryszk, J. (2008) Master Equation—Tutorial Approach.
- Takada, A. Conradt, R. and Richet, P. (2013) Journal of Non-Crystalline Solids, 360, 13. https://doi.org/10.1016/j.jnoncrysol.2012.10.002; Angel Ricardo Plastino1, Gustavo Luis Ferri, Angelo Plastino, Journal of Modern Physics 11 (2020) 1312-1325.
- R.K. Pathria, Statistical Mechanics, 2nd., Butterworth-Heinemann, Oxford, UK, 1996.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).