Submitted:
23 October 2023
Posted:
23 October 2023
Read the latest preprint version here
Abstract
Keywords:
Main Concept
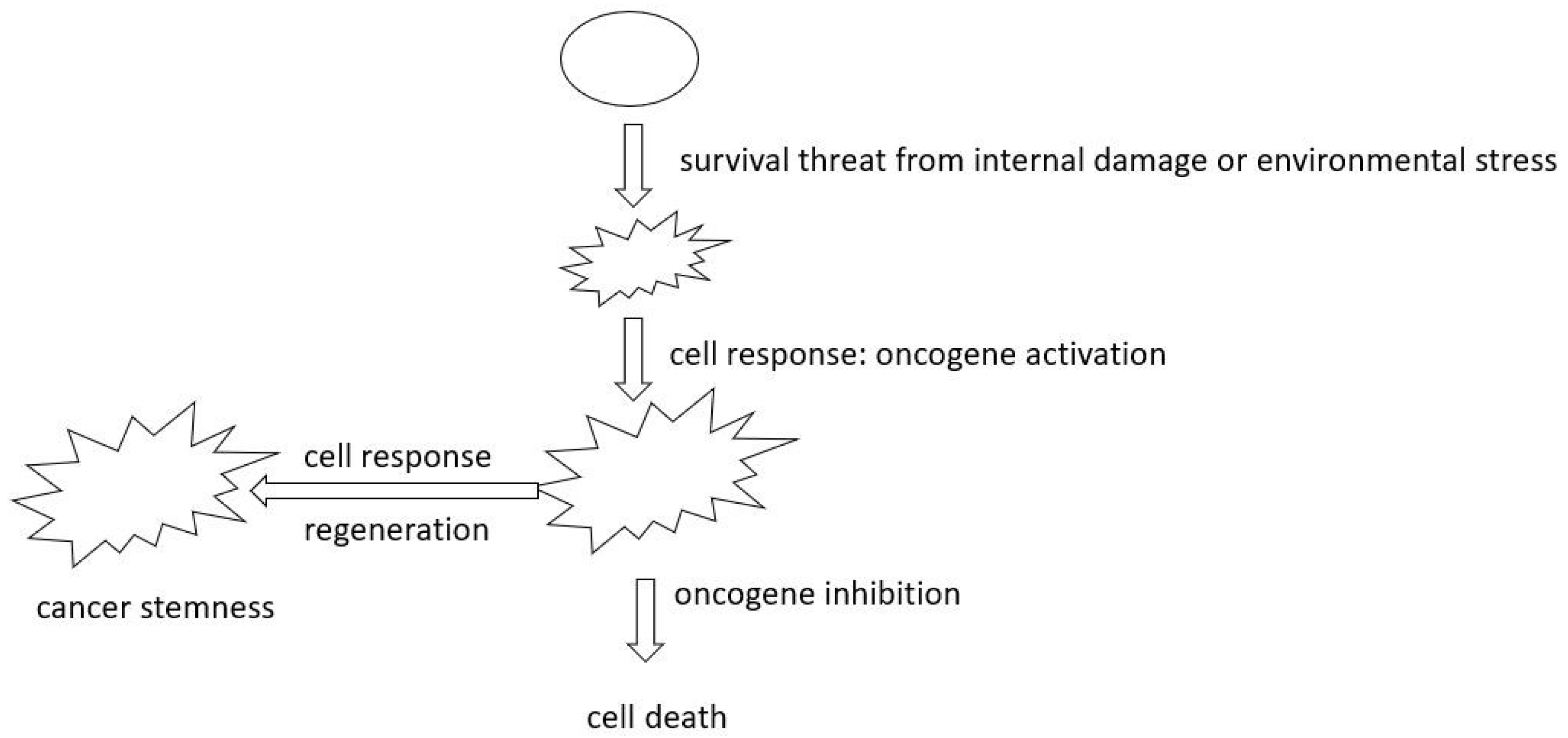
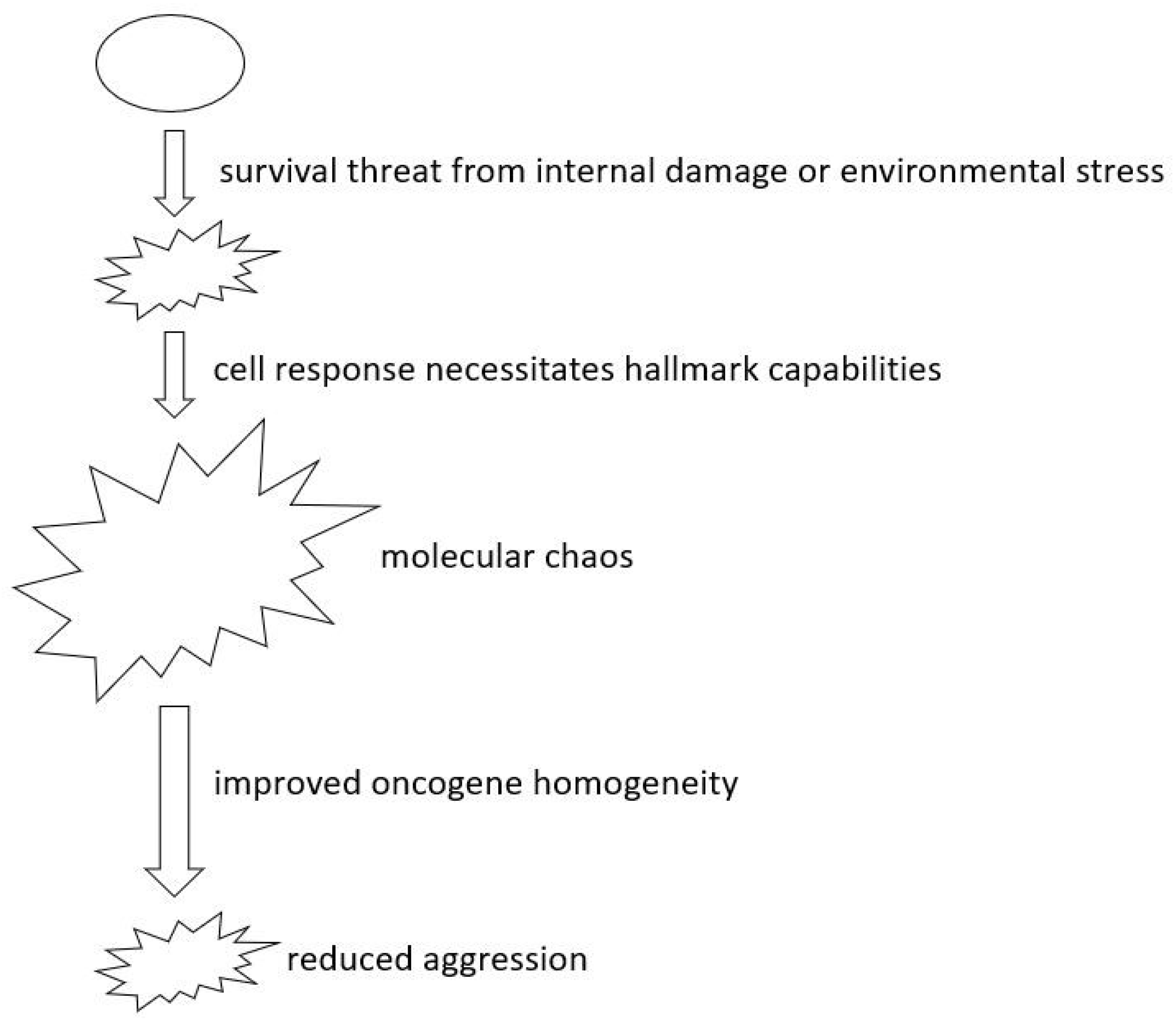
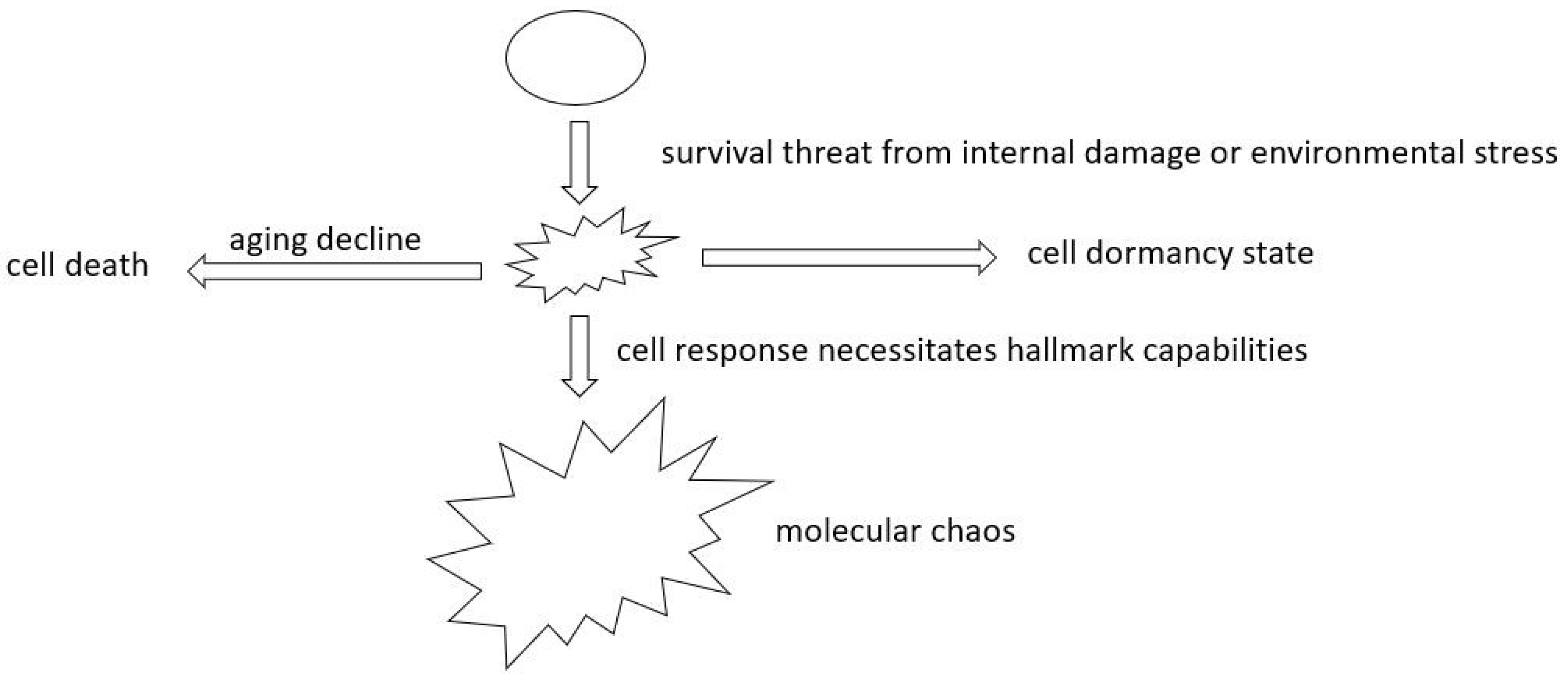
- Rethinking Oncogene Addiction: As our grasp of cellular evolution and tumor heterogeneity deepens, the necessity to re-evaluate oncogene addiction becomes evident. The phenomena of oncogene addiction might be better understood as a cell fate outcome, occurring when cells are detached from the evolutionary process when the target oncogene is inhibited. This fresh perspective could herald innovative treatment paradigms.
- Deepening Insights into Oncogenes and Tumor Suppressors: Reclassifying genes according to their heterogeneity levels has promise. Treatment strategies resulting from this approach could focus on amplifying the homogeneity of critical genes and specifying precise levels of homogeneity, which in turn is likely to enhance the predictability and efficacy of therapeutic outcomes.
- Exploring Aging’s Role in Cancer Development: A better understanding of aging’s role in cancer will allow us to probe the links between aging and cancer onset. A focus here should be distinguishing between normal aging decline and tumor formation. Such insights will contribute to innovative strategies to enhance broader human health.
Future Perspectives
- 1)
- rethinking oncogene addiction.
- 2)
- Deepening Insights into Oncogenes and Tumor Suppressors.
- 3)
- Exploring Aging’s Role in Cancer Development.
Final remark
Acknowledgment
Author information
Contributions
Footnote
References
- Dagogo-Jack, I.; Shaw, A.T. Tumour heterogeneity and resistance to cancer therapies. Nat Rev Clin Oncol 2018, 15, 81–94. [Google Scholar] [CrossRef]
- Vogelstein, B.; Papadopoulos, N.; Velculescu, V.E.; Zhou, S.; Diaz, L.A.; Kinzler, K.W. Cancer Genome Landscapes. Science 2013, 339, 1546–1558. [Google Scholar] [CrossRef]
- Blanke, C.D.; Demetri, G.D.; Von Mehren, M.; Heinrich, M.C.; Eisenberg, B.; et al. Long-Term Results From a Randomized Phase II Trial of Standard- Versus Higher-Dose Imatinib Mesylate for Patients With Unresectable or Metastatic Gastrointestinal Stromal Tumors Expressing KIT. JCO 2008, 26, 620–625. [Google Scholar] [CrossRef]
- Juric, D.; Castel, P.; Griffith, M.; Griffith, O.L.; Won, H.H.; et al. Convergent loss of PTEN leads to clinical resistance to a PI(3)Kα inhibitor. Nature 2015, 518, 240–244. [Google Scholar] [CrossRef]
- Kwak, E.L.; Ahronian, L.G.; Siravegna, G.; Mussolin, B.; Borger, D.R.; et al. Molecular Heterogeneity and Receptor Coamplification Drive Resistance to Targeted Therapy in MET-Amplified Esophagogastric Cancer. Cancer Discovery 2015, 5, 1271–1281. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The Hallmarks of Cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Sun, X.; Yu, Q. Intra-tumor heterogeneity of cancer cells and its implications for cancer treatment. Acta Pharmacol Sin 2015, 36, 1219–1227. [Google Scholar] [CrossRef]
- Andor, N.; Graham, T.A.; Jansen, M.; Xia, L.C.; Aktipis, C.A.; et al. Pan-cancer analysis of the extent and consequences of intratumor heterogeneity. Nat Med 2016, 22, 105–113. [Google Scholar] [CrossRef]
- Bakhoum, S.F.; Landau, D.A. Chromosomal Instability as a Driver of Tumor Heterogeneity and Evolution. Cold Spring Harb Perspect Med 2017, 7, a029611. [Google Scholar] [CrossRef]
- Rao, C.V.; Asch, A.S.; Yamada, H.Y. Frequently mutated genes/pathways and genomic instability as prevention targets in liver cancer. CARCIN 2017, 38, 2–11. [Google Scholar] [CrossRef]
- Li, M.; Zhang, Z.; Li, L.; Wang, X. An algorithm to quantify intratumor heterogeneity based on alterations of gene expression profiles. Commun Biol 2020, 3, 1–19. [Google Scholar] [CrossRef]
- Geiger, T.R.; Peeper, D.S. Metastasis mechanisms. Biochimica et Biophysica Acta (BBA)-Reviews on Cancer 2009, 1796, 293–308. [Google Scholar] [CrossRef]
- Ha, N.-H.; Faraji, F.; Hunter, K.W. Mechanisms of Metastasis. In Cancer Targeted Drug Delivery: An Elusive Dream; Bae, Y.H., Mrsny, R.J., Park, K., Eds.; Springer: New York, NY, USA, 2013; pp. 435–458. [Google Scholar]
- Yachida, S.; Jones, S.; Bozic, I.; Antal, T.; Leary, R.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef]
- Yokota, J. Tumor progression and metastasis. Carcinogenesis 2000, 21, 497–503. [Google Scholar] [CrossRef]
- Lee, W.-C.; Kopetz, S.; Wistuba, I.I.; Zhang, J. Metastasis of cancer: When and how? Annals of Oncology 2017, 28, 2045–2047. [Google Scholar] [CrossRef]
- Patel, S.A.; Rodrigues, P.; Wesolowski, L.; Vanharanta, S. Genomic control of metastasis. Br J Cancer 2021, 124, 3–12. [Google Scholar] [CrossRef]
- Reiter, J.G.; Makohon-Moore, A.P.; Gerold, J.M.; Heyde, A.; Attiyeh, M.A.; et al. Minimal functional driver gene heterogeneity among untreated metastases. Science 2018, 361, 1033–1037. [Google Scholar] [CrossRef]
- Sherwood, J.; Dearden, S.; Ratcliffe, M.; Walker, J. Mutation status concordance between primary lesions and metastatic sites of advanced non-small-cell lung cancer and the impact of mutation testing methodologies: A literature review. Journal of Experimental & Clinical Cancer Research 2015, 34, 92. [Google Scholar]
- Xie, T.; Cho, Y.B.; Wang, K.; Huang, D.; Hong, H.K.; et al. Patterns of somatic alterations between matched primary and metastatic colorectal tumors characterized by whole-genome sequencing. Genomics 2014, 104, 234–241. [Google Scholar] [CrossRef]
- Townson, J.L.; Chambers, A.F. Dormancy of Solitary Metastatic Cells. Cell Cycle 2006, 5, 1744–1750. [Google Scholar] [CrossRef]
- Giancotti, F.G. Mechanisms Governing Metastatic Dormancy and Reactivation. Cell 2013, 155, 750–764. [Google Scholar] [CrossRef]
- Park, S.-Y.; Nam, J.-S. The force awakens: Metastatic dormant cancer cells. Exp Mol Med 2020, 52, 569–581. [Google Scholar] [CrossRef]
- Gomis, R.R.; Gawrzak, S. Tumor cell dormancy. Molecular Oncology 2017, 11, 62–78. [Google Scholar] [CrossRef]
- Summers, M.A.; McDonald, M.M.; Croucher, P.I. Cancer Cell Dormancy in Metastasis. Cold Spring Harb Perspect Med 2020, 10, a037556. [Google Scholar] [CrossRef]
- Yadav, A.S.; Pandey, P.R.; Butti, R.; Radharani, N.N.V.; Roy, S.; et al. The Biology and Therapeutic Implications of Tumor Dormancy and Reactivation. Frontiers in Oncology 2018, 8. [Google Scholar] [CrossRef]
- Pradhan, S.; Sperduto, J.L.; Farino, C.J.; Slater, J.H. Engineered In Vitro Models of Tumor Dormancy and Reactivation. J Biol Eng 2018, 12, 37. [Google Scholar] [CrossRef]
- Singh, D.K.; Patel, V.G.; Oh, W.K.; Aguirre-Ghiso, J.A. Prostate Cancer Dormancy and Reactivation in Bone Marrow. Journal of Clinical Medicine 2021, 10, 2648. [Google Scholar] [CrossRef]
- Chen, Q.; Sun, L.; Chen, Z.J. Regulation and function of the cGAS–STING pathway of cytosolic DNA sensing. Nat Immunol 2016, 17, 1142–1149. [Google Scholar] [CrossRef]
- Janssen, L.M.E.; Ramsay, E.E.; Logsdon, C.D.; Overwijk, W.W. The immune system in cancer metastasis: Friend or foe? J Immunother Cancer 2017, 5, 79. [Google Scholar] [CrossRef]
- Whiteside, T.L. Immune responses to malignancies. J Allergy Clin Immunol 2010, 125, S272–S283. [Google Scholar] [CrossRef]
- Caspi, R.R. Immunotherapy of autoimmunity and cancer: The penalty for success. Nat Rev Immunol 2008, 8, 970–976. [Google Scholar] [CrossRef]
- Ofuji, K.; Hiramatsu, K.; Nosaka, T.; Naito, T.; Takahashi, K.; et al. Pembrolizumab-induced autoimmune side effects of colon and pancreas in a patient with lung cancer. Clin J Gastroenterol 2021, 14, 1692–1699. [Google Scholar] [CrossRef]
- Amos, S.M.; Duong, C.P.M.; Westwood, J.A.; Ritchie, D.S.; Junghans, R.P.; et al. Autoimmunity associated with immunotherapy of cancer. Blood 2011, 118, 499–509. [Google Scholar] [CrossRef]
- Yang, S.; Yu, K.; Palmer, N.; Fox, K.; Kou, S.C.; Kohane, I.S. Autoimmune Effects of Lung Cancer Immunotherapy Revealed by Data-Driven Analysis on a Nationwide Cohort. Clin Pharma and Therapeutics 2020, 107, 388–396. [Google Scholar] [CrossRef]
- Cheng, F.; Loscalzo, J. Autoimmune Cardiotoxicity of Cancer Immunotherapy. Trends in Immunology 2017, 38, 77–78. [Google Scholar] [CrossRef]
- Weinstein, I.B.; Joe, A. Oncogene Addiction. Cancer Research 2008, 68, 3077–3080. [Google Scholar] [CrossRef]
- Weinstein, I.B.; Joe, A.K. Mechanisms of Disease: Oncogene addiction—A rationale for molecular targeting in cancer therapy. Nat Rev Clin Oncol 2006, 3, 448–457. [Google Scholar] [CrossRef]
- McCormick, F. Cancer therapy based on oncogene addiction. Journal of Surgical Oncology 2011, 103, 464–467. [Google Scholar] [CrossRef]
- Lee, H.-J.; Zhuang, G.; Cao, Y.; Du, P.; Kim, H.-J.; Settleman, J. Drug resistance via feedback activation of Stat3 in oncogene-addicted cancer cells. Cancer Cell 2014, 26, 207–221. [Google Scholar] [CrossRef]
- Pagliarini, R.; Shao, W.; Sellers, W.R. Oncogene addiction: Pathways of therapeutic response, resistance, and road maps toward a cure. EMBO Reports 2015, 16, 280–296. [Google Scholar] [CrossRef]
- Zimmermann, G.; Papke, B.; Ismail, S.; Vartak, N.; Chandra, A.; et al. Small molecule inhibition of the KRAS–PDEδ interaction impairs oncogenic KRAS signalling. Nature 2013, 497, 638–642. [Google Scholar] [CrossRef]
- Kopp, F.; Wagner, E.; Roidl, A. The proto-oncogene KRAS is targeted by miR-200c. Oncotarget 2013, 5, 185–195. [Google Scholar] [CrossRef]
- Kim, Y.; Yoon, J.W.; Xiao, X.; Dean, N.M.; Monia, B.P.; Marcusson, E.G. Selective down-regulation of glioma-associated oncogene 2 inhibits the proliferation of hepatocellular carcinoma cells. Cancer Res 2007, 67, 3583–3593. [Google Scholar] [CrossRef]
- Felsher, D.W.; Bishop, J.M. Reversible tumorigenesis by MYC in hematopoietic lineages. Mol Cell 1999, 4, 199–207. [Google Scholar] [CrossRef]
- Schmidt, A.V.; Monga, S.P.; Prochownik, E.V.; Goetzman, E.S. A Novel Transgenic Mouse Model Implicates Sirt2 as a Promoter of Hepatocellular Carcinoma. Int J Mol Sci 2023, 24, 12618. [Google Scholar] [CrossRef]
- Pollack, R.; Wolman, S.; Vogel, A. Reversion of Virus-transformed Cell Lines: Hyperploidy accompanies Retention of Viral Genes. Nature 1970, 228, 938–938. [Google Scholar] [CrossRef]
- Dhanyamraju, P.K.; Schell, T.D.; Amin, S.; Robertson, G.P. Drug-Tolerant Persister Cells in Cancer Therapy Resistance. Cancer Res 2022, 82, 2503–2514. [Google Scholar] [CrossRef]
- De Conti, G.; Dias, M.H.; Bernards, R. Fighting Drug Resistance through the Targeting of Drug-Tolerant Persister Cells. Cancers 2021, 13, 1118. [Google Scholar] [CrossRef]
- Álvarez-Varela, A.; Novellasdemunt, L.; Barriga, F.M.; Hernando-Momblona, X.; Cañellas-Socias, A.; et al. Mex3a marks drug-tolerant persister colorectal cancer cells that mediate relapse after chemotherapy. Nat Cancer 2022, 3, 1052–1070. [Google Scholar] [CrossRef]
- Kumar, U.; Castellanos-Uribe, M.; May, S.T.; Yagüe, E. Adaptive resistance is not responsible for long-term drug resistance in a cellular model of triple negative breast cancer. Gene 2023, 850, 146930. [Google Scholar] [CrossRef]
- Sánchez-Romero, M.A.; Casadesús, J. Contribution of phenotypic heterogeneity to adaptive antibiotic resistance. Proc. Natl. Acad. Sci. USA 2014, 111, 355–360. [Google Scholar] [CrossRef]
- Lim, Z.-F.; Ma, P.C. Emerging insights of tumor heterogeneity and drug resistance mechanisms in lung cancer targeted therapy. Journal of Hematology & Oncology 2019, 12, 134. [Google Scholar]
- Ayob, A.Z.; Ramasamy, T.S. Cancer stem cells as key drivers of tumour progression. Journal of Biomedical Science 2018, 25, 20. [Google Scholar] [CrossRef]
- Klein, C.A.; Blankenstein, T.J.F.; Schmidt-Kittler, O.; Petronio, M.; Polzer, B.; et al. Genetic heterogeneity of single disseminated tumour cells in minimal residual cancer. Lancet 2002, 360, 683–689. [Google Scholar] [CrossRef]
- Berger, N.; Kim-Schulze, S.; Parekh, S. Minimal Residual Disease in Multiple Myeloma: Impact on Response Assessment, Prognosis and Tumor Heterogeneity. In Biological Mechanisms of Minimal Residual Disease and Systemic Cancer; Aguirre-Ghiso, J.A., Ed.; Springer International Publishing: Cham, 2018; pp. 141–159. [Google Scholar]
- Raghavendra, N.M.; Pingili, D.; Kadasi, S.; Mettu, A.; Prasad, S.V.U.M. Dual or multi-targeting inhibitors: The next generation anticancer agents. European Journal of Medicinal Chemistry 2018, 143, 1277–1300. [Google Scholar] [CrossRef]
- Eder, J.P.; Shapiro, G.I.; Appleman, L.J.; Zhu, A.X.; Miles, D.; et al. A phase I study of foretinib, a multi-targeted inhibitor of c-Met and vascular endothelial growth factor receptor 2. Clin Cancer Res 2010, 16, 3507–3516. [Google Scholar] [CrossRef]
- Petrelli, A.; Giordano, S. From single- to multi-target drugs in cancer therapy: When aspecificity becomes an advantage. Curr Med Chem 2008, 15, 422–432. [Google Scholar]
- Foulkes, W.D. Inherited Susceptibility to Common Cancers. N Engl J Med 2008, 359, 2143–2153. [Google Scholar] [CrossRef]
- Fearon, E.R. Human Cancer Syndromes: Clues to the Origin and Nature of Cancer. Science 1997, 278, 1043–1050. [Google Scholar] [CrossRef]
- Hall, J.M.; Lee, M.K.; Newman, B.; Morrow, J.E.; Anderson, L.A.; et al. Linkage of Early-Onset Familial Breast Cancer to Chromosome 17q21. Science 1990, 250, 1684–1689. [Google Scholar] [CrossRef]
- Moore, N.; Lyle, S. Quiescent, Slow-Cycling Stem Cell Populations in Cancer: A Review of the Evidence and Discussion of Significance. Journal of Oncology 2010, 2011, e396076. [Google Scholar] [CrossRef]
- Makena, M.R.; Ranjan, A.; Thirumala, V.; Reddy, A.P. Cancer stem cells: Road to therapeutic resistance and strategies to overcome resistance. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2020, 1866, 165339. [Google Scholar] [CrossRef]
- Shiokawa, D.; Sakai, H.; Ohata, H.; Miyazaki, T.; Kanda, Y.; et al. Slow-Cycling Cancer Stem Cells Regulate Progression and Chemoresistance in Colon Cancer. Cancer Res 2020, 80, 4451–4464. [Google Scholar] [CrossRef]
- Robinson, N.J.; Taylor, D.J.; Schiemann, W.P. Stem cells, immortality, and the evolution of metastatic properties in breast cancer: Telomere maintenance mechanisms and metastatic evolution. J Cancer Metastasis Treat 2019, 5, 39. [Google Scholar] [CrossRef]
- Shay, J.W.; Wright, W.E. Telomeres and telomerase in normal and cancer stem cells. FEBS Letters 2010, 584, 3819–3825. [Google Scholar] [CrossRef]
- Friedmann-Morvinski, D.; Verma, I.M. Dedifferentiation and reprogramming: Origins of cancer stem cells. EMBO Reports 2014, 15, 244–253. [Google Scholar] [CrossRef]
- Visvader, J.E. Cells of origin in cancer. Nature 2011, 469, 314–322. [Google Scholar] [CrossRef]
- Visvader, J.E.; Lindeman, G.J. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat Rev Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef]
- Jolly, M.K.; Celià-Terrassa, T. Dynamics of Phenotypic Heterogeneity Associated with EMT and Stemness during Cancer Progression. Journal of Clinical Medicine 2019, 8, 1542. [Google Scholar] [CrossRef]
- Zhou, F.; Aroua, N.; Liu, Y.; Rohde, C.; Cheng, J.; et al. A Dynamic rRNA Ribomethylome Drives Stemness in Acute Myeloid Leukemia. Cancer Discov 2023, 13, 332–347. [Google Scholar] [CrossRef]
- Jain, P.; Duddu, A.S.; Jolly, M.K. Stochastic population dynamics of cancer stemness and adaptive response to therapies. Essays Biochem 2022, 66, 387–398. [Google Scholar]
- Ma, H.; He, M.; Wei, M. [Research progress on targeting effect and regulating mechanisms of the stemness of cancer stem cells]. Yao Xue Xue Bao 2016, 51, 189–196. [Google Scholar]
- Takahashi, K.; Okita, K.; Nakagawa, M.; Yamanaka, S. Induction of pluripotent stem cells from fibroblast cultures. Nat Protoc 2007, 2, 3081–3089. [Google Scholar] [CrossRef] [PubMed]
- Chico, M.A.; Mesas, C.; Doello, K.; Quiñonero, F.; Perazzoli, G.; et al. Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review. Cancers 2023, 15, 2449. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, T.; Gu, W.; Li, P.; Cheng, X.; et al. The ALDH1+ subpopulation of the human NMFH-1 cell line exhibits cancer stem-like characteristics. Oncol Rep 2015, 33, 2291–2298. [Google Scholar] [CrossRef]
- Feng, B.-H.; Liu, A.-G.; Gu, W.-G.; Deng, L.; Cheng, X.-G.; et al. CD133+ subpopulation of the HT1080 human fibrosarcoma cell line exhibits cancer stem-like characteristics. Oncol Rep 2013, 30, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Cell Dedifferentiation—An overview | ScienceDirect Topics. www.sciencedirect.com.
- Li, M.-M.; Tang, Y.-Q.; Gong, Y.-F.; Cheng, W.; Li, H.-L.; et al. Development of an oncogenic dedifferentiation SOX signature with prognostic significance in hepatocellular carcinoma. BMC Cancer 2019, 19, 851. [Google Scholar] [CrossRef]
- Komarova, N.L.; Wodarz, D. Drug resistance in cancer: Principles of emergence and prevention. Proc. Natl. Acad. Sci. USA 2005, 102, 9714–9719. [Google Scholar] [CrossRef] [PubMed]
- Colombo, P.-E.; Fabbro, M.; Theillet, C.; Bibeau, F.; Rouanet, P.; Ray-Coquard, I. Sensitivity and resistance to treatment in the primary management of epithelial ovarian cancer. Critical Reviews in Oncology/Hematology 2014, 89, 207–216. [Google Scholar] [CrossRef]
- Zhu, P.; Fan, Z. Cancer stem cells and tumorigenesis. Biophys Rep 2018, 4, 178–188. [Google Scholar] [CrossRef]
- Eramo, A.; Lotti, F.; Sette, G.; Pilozzi, E.; Biffoni, M.; et al. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 2008, 15, 504–514. [Google Scholar] [CrossRef]
- Ma, S.; Chan, K.; Hu, L.; Lee, T.K.; Wo, J.Y.; et al. Identification and Characterization of Tumorigenic Liver Cancer Stem/Progenitor Cells. Gastroenterology 2007, 132, 2542–2556. [Google Scholar] [CrossRef]
- Cheung, K.J.; Gabrielson, E.; Werb, Z.; Ewald, A.J. Collective invasion in breast cancer requires a conserved basal epithelial program. Cell 2013, 155, 1639–1651. [Google Scholar] [CrossRef]
- Wakimoto, H.; Mohapatra, G.; Kanai, R.; Curry, W.T.; Yip, S.; et al. Maintenance of primary tumor phenotype and genotype in glioblastoma stem cells. Neuro Oncol 2012, 14, 132–144. [Google Scholar] [CrossRef]
- Huang, E.H.; Hynes, M.J.; Zhang, T.; Ginestier, C.; Dontu, G.; et al. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res 2009, 69, 3382–3389. [Google Scholar] [CrossRef]
- Sherr, C.J. Principles of tumor suppression. Cell 2004, 116, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Oncogene—An overview | ScienceDirect Topics. www.sciencedirect.com.
- Endo, Y.; Lyon, S.; Shen, Y.; Mohan, N.; Wu, W.J. Cell proliferation and invasion are regulated differently by EGFR and MRP1 in T-DM1-resistant breast cancer cells. Sci Rep 2019, 9, 16383. [Google Scholar] [CrossRef]
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev 2010, 29, 405–434. [Google Scholar] [CrossRef]
- De Donatis, A.; Ranaldi, F.; Cirri, P. Reciprocal control of cell proliferation and migration. Cell Communication and Signaling 2010, 8, 20. [Google Scholar] [CrossRef]
- Swami, P.; Thiyagarajan, S.; Vidger, A.; Indurthi, V.S.K.; Vetter, S.W.; Leclerc, E. RAGE Up-Regulation Differently Affects Cell Proliferation and Migration in Pancreatic Cancer Cells. International Journal of Molecular Sciences 2020, 21, 7723. [Google Scholar] [CrossRef]
- Yachida, S.; Jones, S.; Bozic, I.; Antal, T.; Leary, R.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Huysentruyt, L.C. On the Origin of Cancer Metastasis. Crit Rev Oncog 2013, 18, 43–73. [Google Scholar] [CrossRef]
- Matassa, D.S.; Agliarulo, I.; Avolio, R.; Landriscina, M.; Esposito, F. TRAP1 Regulation of Cancer Metabolism: Dual Role as Oncogene or Tumor Suppressor. Genes 2018, 9, 195. [Google Scholar] [CrossRef]
- Robbs, B.K.; Cruz, A.L.S.; Werneck, M.B.F.; Mognol, G.P.; Viola, J.P.B. Dual Roles for NFAT Transcription Factor Genes as Oncogenes and Tumor Suppressors. Molecular and Cellular Biology 2008, 28, 7168–7181. [Google Scholar] [CrossRef] [PubMed]
- Uribesalgo, I.; Benitah, S.A.; Di Croce, L. From oncogene to tumor suppressor: The dual role of Myc in leukemia. Cell Cycle 2012, 11, 1757–1764. [Google Scholar] [CrossRef]
- Shen, L.; Shi, Q.; Wang, W. Double agents: Genes with both oncogenic and tumor-suppressor functions. Oncogenesis 2018, 7, 1–14. [Google Scholar] [CrossRef]
- Ayob, A.Z.; Ramasamy, T.S. Cancer stem cells as key drivers of tumour progression. Journal of Biomedical Science 2018, 25, 20. [Google Scholar] [CrossRef]
- Damen, M.P.F.; Van Rheenen, J.; Scheele, C.L.G.J. Targeting dormant tumor cells to prevent cancer recurrence. The FEBS Journal 2021, 288, 6286–6303. [Google Scholar] [CrossRef]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The Hallmarks of Aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef]
- Mc Auley, M.T.; Guimera, A.M.; Hodgson, D.; Mcdonald, N.; Mooney, K.M.; et al. Modelling the molecular mechanisms of aging. Biosci Rep 2017, 37, BSR20160177. [Google Scholar] [CrossRef]
- Campisi, J. Aging, Cellular Senescence, and Cancer. Annu Rev Physiol 2013, 75, 685–705. [Google Scholar] [CrossRef]
- Laconi, E.; Marongiu, F.; DeGregori, J. Cancer as a disease of old age: Changing mutational and microenvironmental landscapes. Br J Cancer 2020, 122, 943–952. [Google Scholar] [CrossRef]
- Rufini, A.; Tucci, P.; Celardo, I.; Melino, G. Senescence and aging: The critical roles of p53. Oncogene 2013, 32, 5129–5143. [Google Scholar] [CrossRef]
- Mori, H.; Funahashi, Y.; Yoshino, Y.; Kumon, H.; Ozaki, Y.; et al. Blood CDKN2A Gene Expression in Aging and Neurodegenerative Diseases. J Alzheimers Dis 2021, 82, 1737–1744. [Google Scholar] [CrossRef]
- Zeng, H.; Jorapur, A.; Shain, A.H.; Lang, U.E.; Torres, R.; et al. Bi-allelic loss of CDKN2A initiates melanoma invasion via BRN2 activation. Cancer Cell 2018, 34, 56–68.e9. [Google Scholar] [CrossRef]
- Ozaki, T.; Nakagawara, A. Role of p53 in Cell Death and Human Cancers. Cancers 2011, 3, 994–1013. [Google Scholar] [CrossRef]
- Endo, H.; Inoue, M. Dormancy in cancer. Cancer Sci 2019, 110, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Dormancy—An overview | ScienceDirect Topics. www.sciencedirect.com.
- Neophytou, C.M.; Kyriakou, T.-C.; Papageorgis, P. Mechanisms of Metastatic Tumor Dormancy and Implications for Cancer Therapy. Int J Mol Sci 2019, 20, 6158. [Google Scholar] [CrossRef]
- Park, S.-Y.; Nam, J.-S. The force awakens: Metastatic dormant cancer cells. Exp Mol Med 2020, 52, 569–581. [Google Scholar] [CrossRef]
- Blasco, M.T.; Espuny, I.; Gomis, R.R. Ecology and evolution of dormant metastasis. Trends Cancer 2022, 8, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Afifi, N.; Barrero, C.A. Understanding Breast Cancer Aggressiveness and Its Implications in Diagnosis and Treatment. J Clin Med 2023, 12, 1375. [Google Scholar] [CrossRef]
- Beckwith, J.B.; Zuppan, C.E.; Browning, N.G.; Moksness, J.; Breslow, N.E. Histological analysis of aggressiveness and responsiveness in Wilms’ tumor. Med. Pediatr. Oncol. 1996, 27, 422–428. [Google Scholar] [CrossRef]
- El Zein, D.; Hughes, M.; Kumar, S.; Peng, X.; Oyasiji, T.; et al. Metaplastic Carcinoma of the Breast Is More Aggressive Than Triple-negative Breast Cancer: A Study From a Single Institution and Review of Literature. Clinical Breast Cancer 2017, 17, 382–391. [Google Scholar] [CrossRef]
- Kumar, R.; Angelini, S.; Snellman, E.; Hemminki, K. BRAF Mutations Are Common Somatic Events in Melanocytic Nevi. Journal of Investigative Dermatology 2004, 122, 342–348. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




