1. Introduction
Climate change poses a significant challenge to sustainable agriculture, particularly in the context of drought stress. Drought stress, a result of water deficit, alters crops' physiological, morphological, biochemical, and molecular characteristics, impairing their growth and development. Sub Saharan Africa has been highlighted as a potential hotspot regarding climate change, with an increased frequency of drought incidence observed over the past few decades [
1,
2]. This is anticipated to have several negative effects on dryland crop production [
3,
4]. As a result, studies on plants drought stress tolerance in plants are becoming major focus of research in order to establish possible ways of adaptation.
To survive under water deficit conditions, plants must maintain their water status and maintain homeostasis. Therefore, plants have evolved various strategies, including drought escape, drought avoidance, and drought tolerance that enable them to prevent water loss and survive the onslaught of water deficit conditions. Stomatal regulation has been found to be important for drought stress tolerance mechanisms in plants through drought avoidance which involves sustaining of important physiological processes [
5]. Stomatal closure has been documented as one of the first responders to drought stress in plants [
5], as it aims to reduce water loss through transpiration, and it is mainly influenced by soil moisture content as compared to leaf water status. Another morpho-logical alteration attributed moisture deficit stress is a decrease in stomata number. Reduced stomatal opening reduces net CO2 uptake, transpiration and net photosynthesis rates as mechanisms by plants survive in variable conditions including water deficit [
6]. Under extreme drought stress, stomata can completely close, but this is highly influenced by the plant species. The wild relatives of different domesticated crops such as wheat and barley are more tolerant to harsh environmental conditions as they possess a unique collection of morphological, anatomical and physiological traits [
7]. Among the different traits, the efficient control of transpiration through stomata is one of the crucial features of wild relatives for balancing the efficiency of CO2 assimilation and transpiration under abiotic stress conditions. Xerophytic wild barley has low stomatal conductance [
8]. Additionally, com-pared to the kidney-type stomatal structure of dicotyledons, the stomatal structure and developmental peculiarities of Poaceae crops, which consist of two guard cells and two secondary guard cells, have great potential in improving water utilization in crops and overall drought tolerance [
9,
10]. Tolerant species therefore regulate the status of their stomata to enable carbon fixation and assimilation while also increasing water use efficiency. Most cultivated crop species have wild relatives that exhibit tolerance to abiotic stresses. Therefore, to come up with efficient crop improvement production methods wild species can be used as a good source for studying crop response to environmental stress.
Crop landraces and wild relatives have shown to be an important part in understanding most of the tolerant mechanisms as they have shown to have natural acquired this tolerance mechanisms and these can be harnessed and be used to domesticated crops species tolerance to both biotic and abiotic stress [
11,
12]. It is important to harness the natural patterns of diversity Wild watermelon (
Citrullus lanatus sp.) is a plant species of the Cucurbitaceae family which inhabits the Kalahari Desert in Botswana. This crop survives annually in the desert from spring to summer. The climate of the Kalahari Desert is very severe for plant growth [
13] with less rainfall which causes accumulation of salts on the soil surface and strong summer light and less available water derive plants into oxidative and heat damages. The two landraces used in this study are commonly cultivated by dryland subsistence farmers in Botswana and have shown to possess extensive tolerance to drought. The landraces have always yielded above average even in the worst rainy season. Thus, it calls for extensive studies on them towards understanding their survival mechanisms and exploit them for crop domestication. Therefore, the objective of this study was to evaluate the stomatal aperture in watermelon accession as a suggested tolerance mechanism which has been well documented as the first response mechanisms to drought stress. The evaluation involved morphological, physiological, and molecular response patterns of traits associated with stomatal aperture in accessions with different drought tolerance level. We hypnotized that the stomatal movement plays a significant role in the tolerance mechanism of plant and that the local landraces a superior stomatal response that aids tolerance response to drought stress as compared to the domesticated hybrid watermelon. By elucidating these important mechanisms, we expect our study to provide valuable insights and technical support for the incorporation of stomatal movement as an important trait in crop improvement for drought tolerance.
2. Materials and Methods
2.1. Study site and plant materials
This experimental research was carried out in Sebele at BUAN (Botswana University of Agriculture and Natural Resources) greenhouse, and it was repeated three times. A total of 4 watermelon accessions were used in this study (
Table 1). Among the accessions used 1 was a wild species, two were local landraces and last one was the commercial hybrid. The three landraces were sourced from Botswana National Plant Genetic Resource Center (BNPGRC) seed bank in Sebele while hybrid (crimson sweet) watermelon seeds were purchased from an agricultural shop in Gaborone.
2.2. Planting and design of experiment
The watermelon seeds were planted in drought stress wooden boxes in the greenhouse. One box was used for each moisture treatment. The seeds were planted in rows, with each row representing a single genotype.
All treatments were adequately irrigated every day for fourteen (14) days after planting to promote the development of a root system without water stress. Moisture stress treatment was imposed by complete withholding of irrigation for nine (9) days and a re-watering was done immediately to collect recovery data. Control plants were continually watered as per the pre-treatment schedule to maintain soil moisture to near field capacity throughout the experiment. Management practices particularly weeding, cultivation and pest control were carried out according to the needs of the crop.
Data was collected at 3 days intervals for a period of 15 days with day 0 being the control data prior to stress initiation and day 12 being post rewatering or recovery data.
Soil moisture content was taken at every data collection day using a Moisture Probe Meter (MPM-160-B) 12-bit Resolution, ICT International Pty Ltd, Australia. A calibrated soil moisture sensor was inserted at a depth of 10cm, and the reading taken and recorded [
14].
Stomatal conductance (mmolH2O m-2 s-1) was measured using a Leaf Porometer, Decagon Devices, INC (USA). Measurements were taken from three fully grown fresh leaves that were selected from three randomly selected plants from each watermelon genotype. And the measurements were recorded by placing the leaf porometer sensor on the leaf for 60 seconds.
RWC = 100 [FW DM]/ [TW DM] [
15].
Where FW = Fresh weight (g), Turgid weight (TW) and DM = dry weight.
Three fully grown fresh leaves were harvested from three randomly selected plants for each watermelon genotype. The leaves were weighed (FW) immediately after detaching from plants, then turgid weight (TW) was determined by soaking the leaves in sterile water overnight at 4 °C, and thereafter the leaves were oven dried at 70°C until they reached a constant weight then dry weight (DW) was recorded.
Chlorophyll fluorescence parameters, Fv/Fm, qL, PhiNPQ, PhiNO, Phi2 and PS1 Active Centres were measured between 10 00hrs and 12 00hrs on a cloudless weather. A MultiSpeQ (version 2.0) by PhotosynQ Inc. Michigan, United States, was used to take readings of all these fluorescence parameters under ambient greenhouse conditions [
16]. Chlorophyll content (SPAD) was also measured used the same instrument and procedure as mentioned above.
At 3-day intervals post the initiation on stress treatment, three leaves per treatment were harvested from different randomly selected plants for observation of stomatal morphology. Stomata imaging was done using nail polish method as described by [
17]. Leaf imprints were collected by applying nail polish on the adaxial and abaxial leaf surface of watermelon genotypes before detaching the leaves. Clear nail polish (Insta-Dri Top Coat, Sally Hansen, USA) was applied on the leaf surface in a thin layer, and left to air dry for 5 min. Dry nail polish was peeled off using clear tape (Crystal clear Office tapes, Winc, Australia) and was mounted on a microscope slide. Stomata on leaf imprints were observed using a (Carl ZEISS Axio Observer 5 inverted biological microscope, ZEISS, Germany) connected to an AxioCamMR5 digital camera and a 40 × objective. Images of leaf stomata imprints and measurements of the stomatal aperture were taken using ZEN imaging software.
2.5. Expression analysis of stomatal aperture genes and transcription factors (TFs)
Leaf tissues of four different planted watermelon genotypes were harvested for use in qRT-PCR analysis. One random leaf was harvested from each of the three randomly selected watermelon plants per each variety and treatment, with each leaf representing a biological replication. These leaves were collected in liquid nitrogen and immediately stored at −80 °C to until RNA extraction.
Total RNA was isolated from the frozen samples using Quick-RNA ™ Plant Miniprep kit, Zymo Research (USA). The integrity of the extracted RNA was verified using 2% agarose gel electrophoresis. The concentration and purity of RNA was also measured using NanoDrop 2000 spectrophotometer (Thermo Scientific, China).
The synthesis of DNA from RNA template via reverse transcription was carried out using cDNA synthesis kit with oligio DT primers using ProtoScript® II First Strand cDNA Synthesis Kit, New England BioLabs INC., (Ipswich, MA). The cDNA was checked for success of amplification using the designed primers and reference primers.
Primer sequences of the target genes and the reference genes used for normalization was designed with Primer3Plus program (
http://primer3plus.com/cgi-bin/dev/primer3plus.cgi) based on the sequences of important genes related to stomatal aperture in plants. First, the gene of interest of a model crop was blast searched into cucurbits genome database to identify homologs on watermelon gene sequence. The target sequence was then used to design primers using a primer3plus online tool to design specific primers with the set parameters.
2.6. Targeted genes
Table 1.
Plant transcriptional regulators with a function in stomatal movements and primers sequences were used for amplifying genes responsible for stomatal aperture and reference genes in wild watermelon.
Table 1.
Plant transcriptional regulators with a function in stomatal movements and primers sequences were used for amplifying genes responsible for stomatal aperture and reference genes in wild watermelon.
| Name |
Pathways |
Role |
Target TFs of stomatal aperture and reference genes in wild watermelon. |
| Cla017012 |
dark/light, ABA |
opening |
F_CCAATACTGGGTTGCTTAGATGTAG
R_GTTCTTTGTGGAAGGTATGAAGCTA |
| Cla017389 |
ABA |
opening |
F_TCTCTACACTGTTCCTGAAAATTCC
R _TGTTCTGACCAAAACCTAATCTCTC |
| Cla016849 |
ABA |
closure |
F_ACATCTTCATGCACTAAACAGAGTG
R:GATCCATTAGAGATGCTTGTGATCT |
| Cla004380 |
ABA |
closure |
F_GTGGAAGAAGCTCTACAAAGTCAAC
R_AAGAGTGTCTTCTTCCTGGTTGTAA |
| Glyceraldehyde 3-phosphate dehydrogenase(GAPDH) |
F_CTGGCAGTACTTTGCCAACA
R_AGGATTGGAGAGGAGGTCGT |
| Tubulin |
F_CAGCACTCCTAGCTTTGGTGA
R_CGGGGAAATGGGATTAGATT |
The analysis was run in a final volume of 20 μL, with 50ng cDNA samples with 0.5ng of each of the forward and reverse primer, 10 μL qPCR master mix and 5 μL water. The reactions were performed on a Bio-Rad CFX Connect real-time PCR detection system, Bio-Rad Laboratories, Inc., (USA) with two technical replicates for each of the three biological replicates. Negative controls which were PCR mixtures without cDNA templates were used. The PCR conditions were set as follows; initial denaturation at 95°C for 30s, followed by 45 cycles of denaturation at 95°C for 5 s, annealing at as per the melting temperature of the primers for 15 s, and extension at 72°C for 10 s. The melting curve analysis was performed at the end of amplification to verify its specificity of the primers used [
18]. The 2-∆∆CT method was used to estimate the relative RNA expression [
19].
1.7. Statistical Analysis
Data analyses were performed with R – 4.3.1 (2023) software. Analysis of variance (ANOVA) of data sets was used to determine the significance of the variations in treatment mean for morphological and physiological data analysis. Multiple means were compared using Fisher’s LSD test, and these were considered significant at p < 0.05. Relative gene expression was quantified using the Delta Delta (CT) formula as demonstrated in [
19].
3. Results
3.1. Soil water content
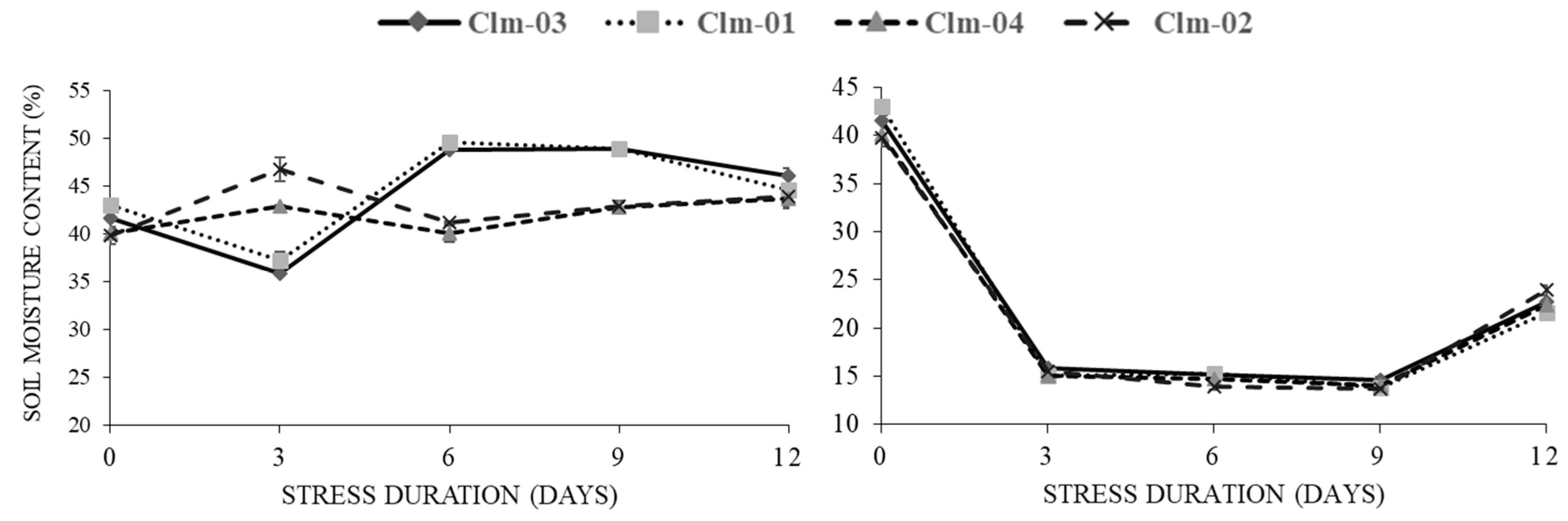
The water content in the soil decreased from the highest of 43% to 13.7% as early as 3 days after water stress treatment, irrespective of the plant species (
Figure 1). Thereafter, water continuously decreased at a slower rate until final day (day 9) of moisture stress where the lowest moisture content was recorded at 13.7 for Clm-02 plants. After re-watering, the soil water content significantly increased for all the genotypes to the highest of 25% at day 12.
3.2. Physiological parameters
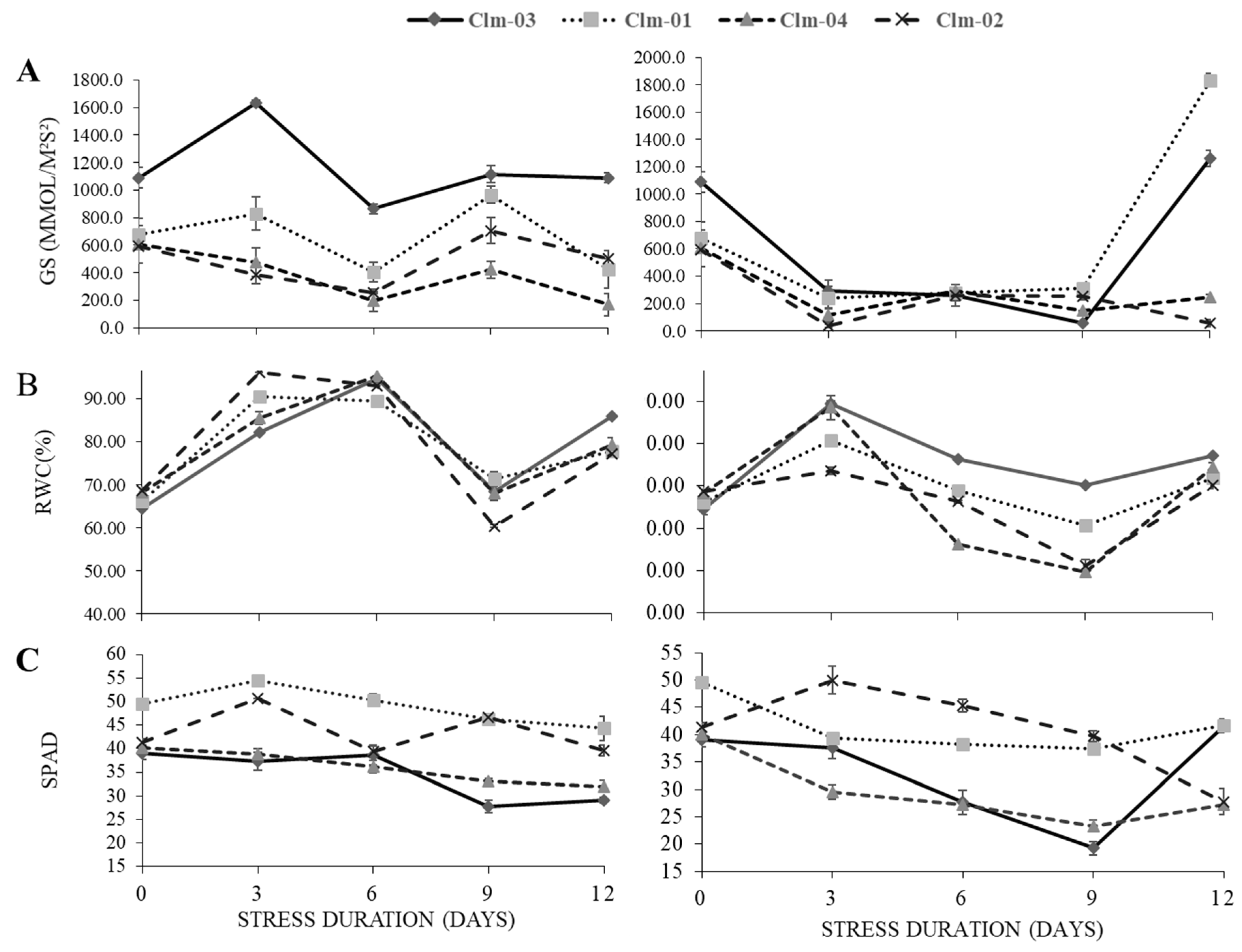
Effect of water deficit on physiological performance of watermelon cultivars is shown in
Figure 2. All cultivars manifested a significant decline in stomatal conductance (gs), leaf relative water content (RWC), and chlorophyll content (SPAD) with decrease in moisture level. Nonetheless, the cultivars differed significantly in their response at different stress levels. Highest RWC was observed in wild watermelon across all the stress days, with highest RWC recorded at 89± 0.82 % at day 3 (
Figure 2B).
The significant decline in stomatal conductance was observed as early as day 3 where the highest was recorded in Clm-03, 297.0± 73.2. The lowest stomatal conductance at this day was recorded in Clm-02 at 35.5 ± 14.4 mmol/m²s². At day 9 of drought stress, Clm-03 recorded the lowest stomatal conductance at 55.1 ± 6.3 mmol/m²s². Under drought stress, Clm-02 recorded the highest SPAD values, 50 ± 3 at day 3 in stress plants and Clm-03 recorded the lowest value 19 ± 1 at day 9. In general, Clm-02 had the highest SPAD values among all the 3 stress days, 50 ± 3, 45 ± 1 and 40 ± 1 respectively for day 3, 6 and 9 in drought stress plants. Clm-04 had the lowest SPAD values of 29 ± 1.34 and 27 ± 0.37 at day 3 and day 6.
3.2. Chlorophyll Fluorescence (CF) Parameters
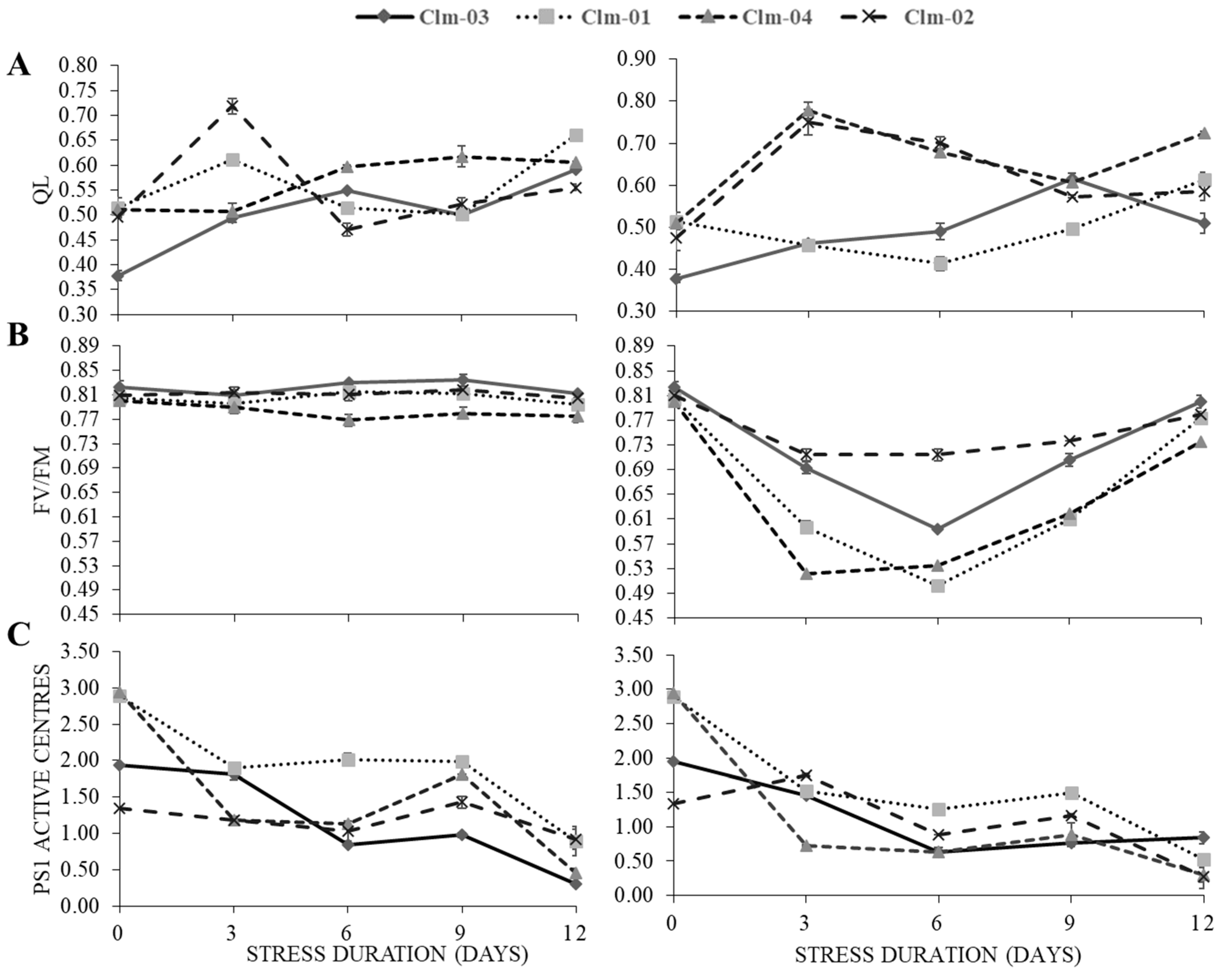
qL, Fv/Fm.and PS1 Active Centres
Chlorophyll fluorescence parameters qL and PS1 Active Centres decreased slightly during the initial period of drought stress while there was a significant decrease in Fv/Fm (
Figure 3). Under drought stress, Clm-01 recorded the lowest values of qL at all the three stress days, 0.45 ± 0.00, 0.41 ± 0.01 and 0.50 ± 0.01 for day 3, 6 and 9 respectively and at day 6 and 9 the values were significantly different from all other genotypes. The highest value 0.78 ± 0.02 was recorded for Clm-04 at day 3. In overall, the highest average qL was recorded for Clm-04 in drought stressed plants for all the treatment days.
The maximum quantum efficiency of PSII (Fv/Fm) values presented in
Figure 3B showed that non-stress plants had no significant differences among either the treatment days and the genotypes throughout the study. In overall, a reduction in Fv/Fm was observed for all the watermelon genotypes when subjected to drought stress conditions. The decline and recovery showed a similar trend for all the genotypes but varied in magnitude among the genotypes. Clm-02 had significantly higher Fv/Fm values which were maintained across all the stress days as it only recorded a slight decrease from day 0 were it recorded 0.83 ± 0.01, thereafter declined to 0.71 ± 0.01, 0.71 ± 0.01 and 0.74 ± 0.00 respectively for stress days 3, 6 and 9. The second genotype which showed slight decline was the Clm-03 recording 0.69 ± 0.01, 0.59 ± 0.00 and 0.71 ± 0.01 respectively for the three stress days. The lowest values were recorded at day 6, 0.50 ± 0.00 in in local watermelon.
Figure 3C shows that there was a less significant reduction in PS1 AC recorded values under drought stress. PSI active centres showed a fluctuating tendency as they declined at day 3 and 6 and recovery was observed at day 9, decreasing again at day 12 for all the watermelon genotypes under non stress conditions. Similarly, the pattern was observed under drought stress treatment, PSI active centres decreased at day 3 and 6 and increased at day 9, decreasing again at day 12.
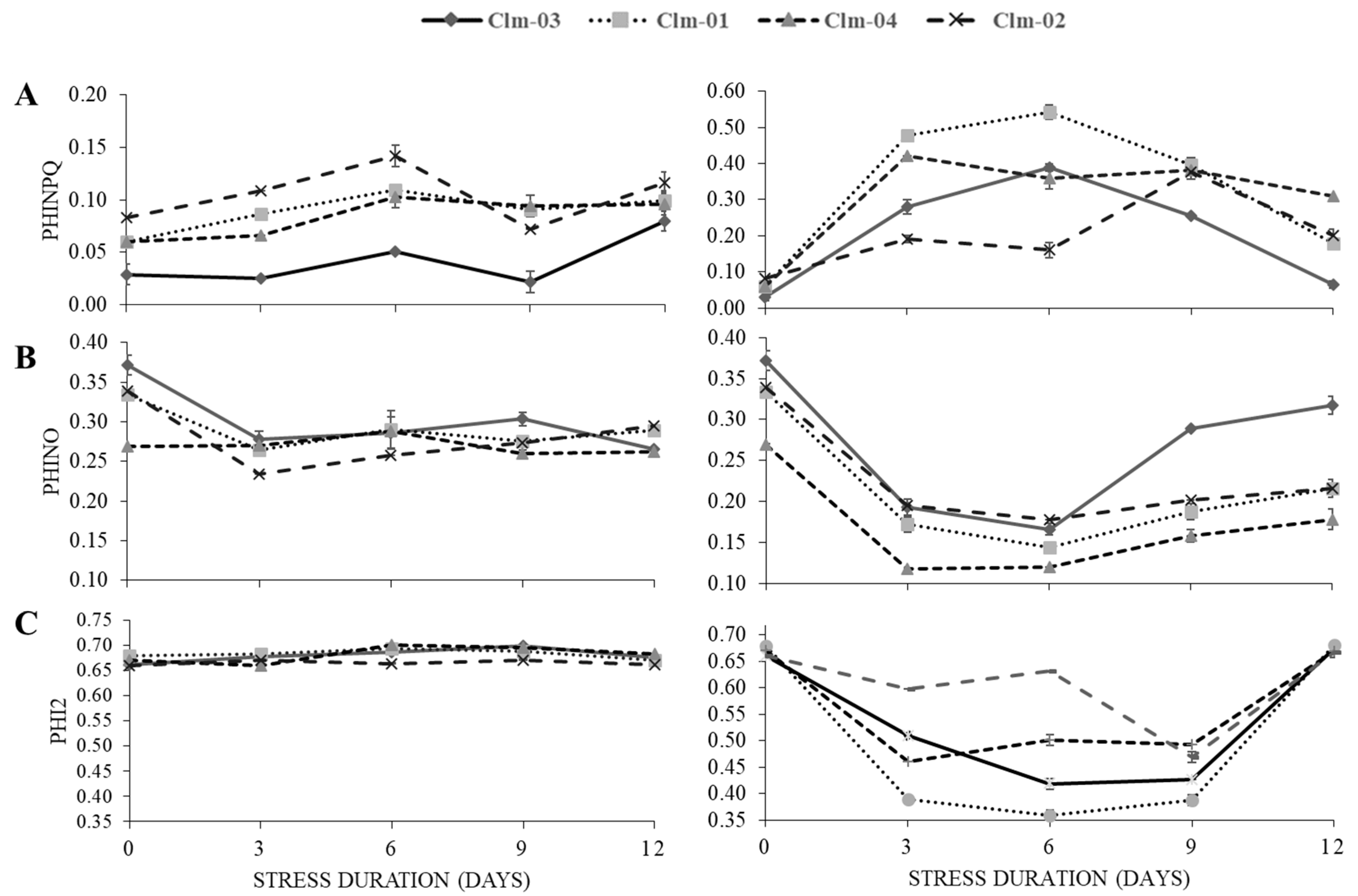
PhiNPQ, PhiNO and Phi2
In general, when exposed to drought stress, PSII efficiency and representative parameters of non-photochemical parameters (PhiNPQ) tended to increase while PhiNO and Phi2 were decreasing with drought stress.
Under drought stress treatment, Clm-01 had the highest PhiNPQ values across all the three stress days reaching 0.54 ± 0.02 the highest at day 6. Clm-02 had the lowest at day 3 and day 6 of stress, 0.19 ± 0.01 at day 3 and 0.16 ± 0.02 at day 6. At day 9 Clm-03 had the lowest at 0.26 ± 0.00. In overall, under drought stress the PhiNPQ values gradually increased at day 3 and at day 6 of stress treatment and decreased at day 9 of stress and continued to decrease at day 12 of stress for Clm-01 and Clm-03 while for Clm-02 and Clm-04 the values increased at day 3 and slight decreased at day 6, increasing at day 9 of stress and decreasing at day.
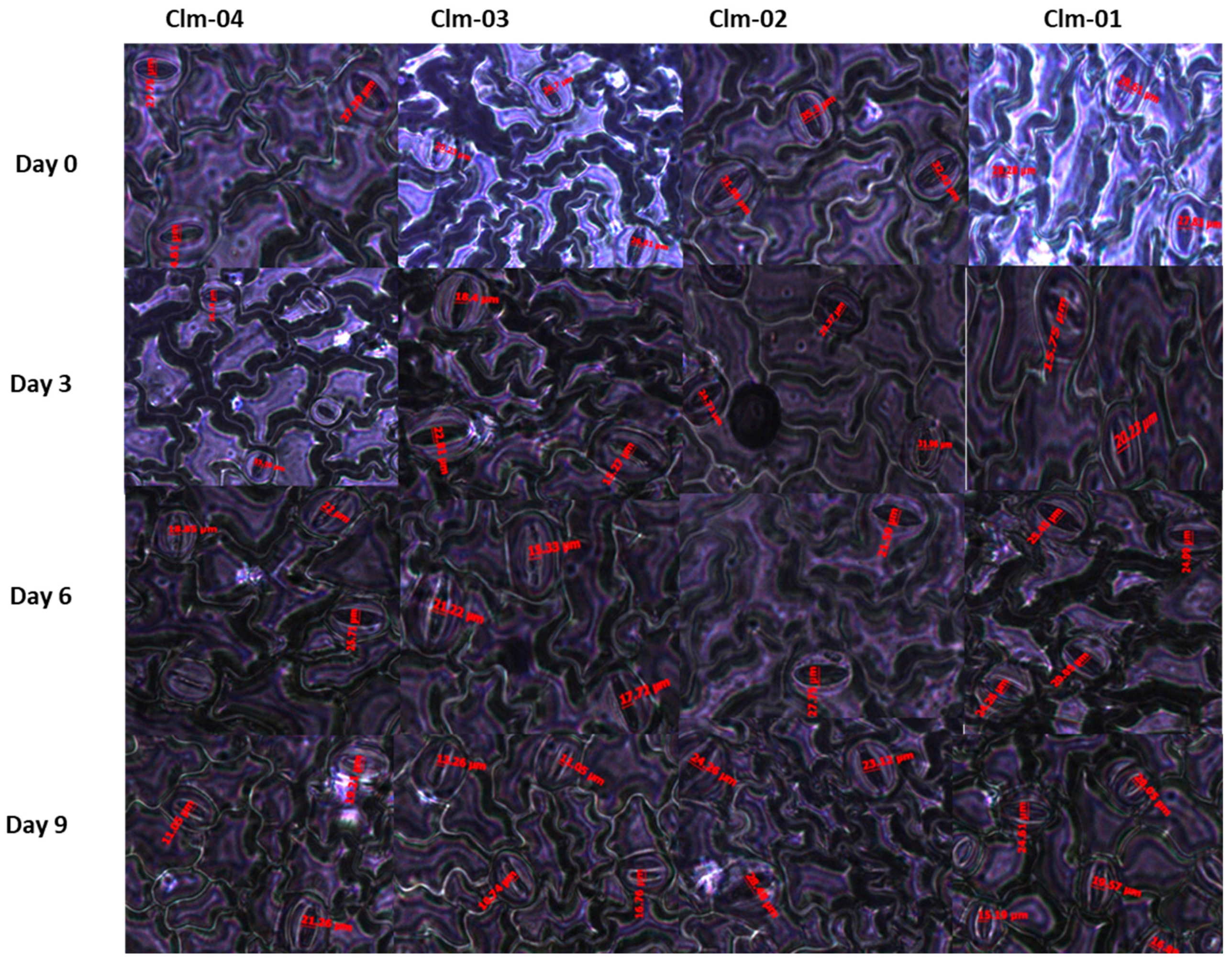
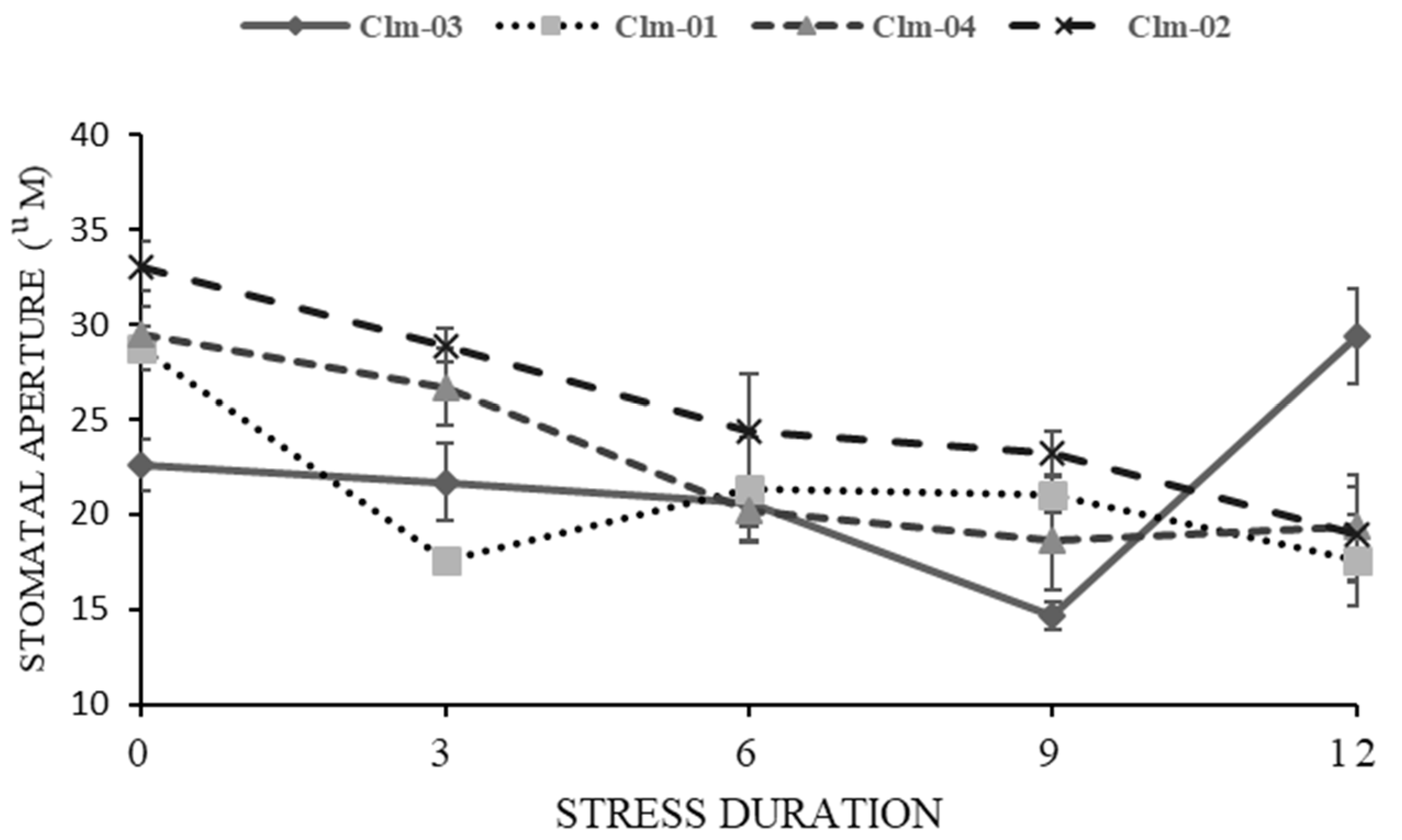
3.3. Stomatal morphology (stomatal aperture measurements)
In overall, drought stress led to a reduction in stomatal aperture in all the studied watermelon genotypes (
Figure 5 and
Figure 6). Clm-02 had the highest average stomatal size as compared to other genotypes across all the stress days and the highest was 33.0 ± 1.3 µm at day 0. Clm-03 recorded the lowest at day 9 of stress, 14.7 ± 0.7 µm while Clm-02 had the highest stomatal aperture value at 23.1 ± 1.2 µm. At day 12 of re-watering, Clm-03 had a highly significant rapid recovery than all the genotypes reaching 29.3 ± 2.5 µm at day 12 which was higher than all the experiment days. Clm-01 and Clm-02 recovery was delayed beyond day 12 as the stomatal size continued to decrease at day 12.
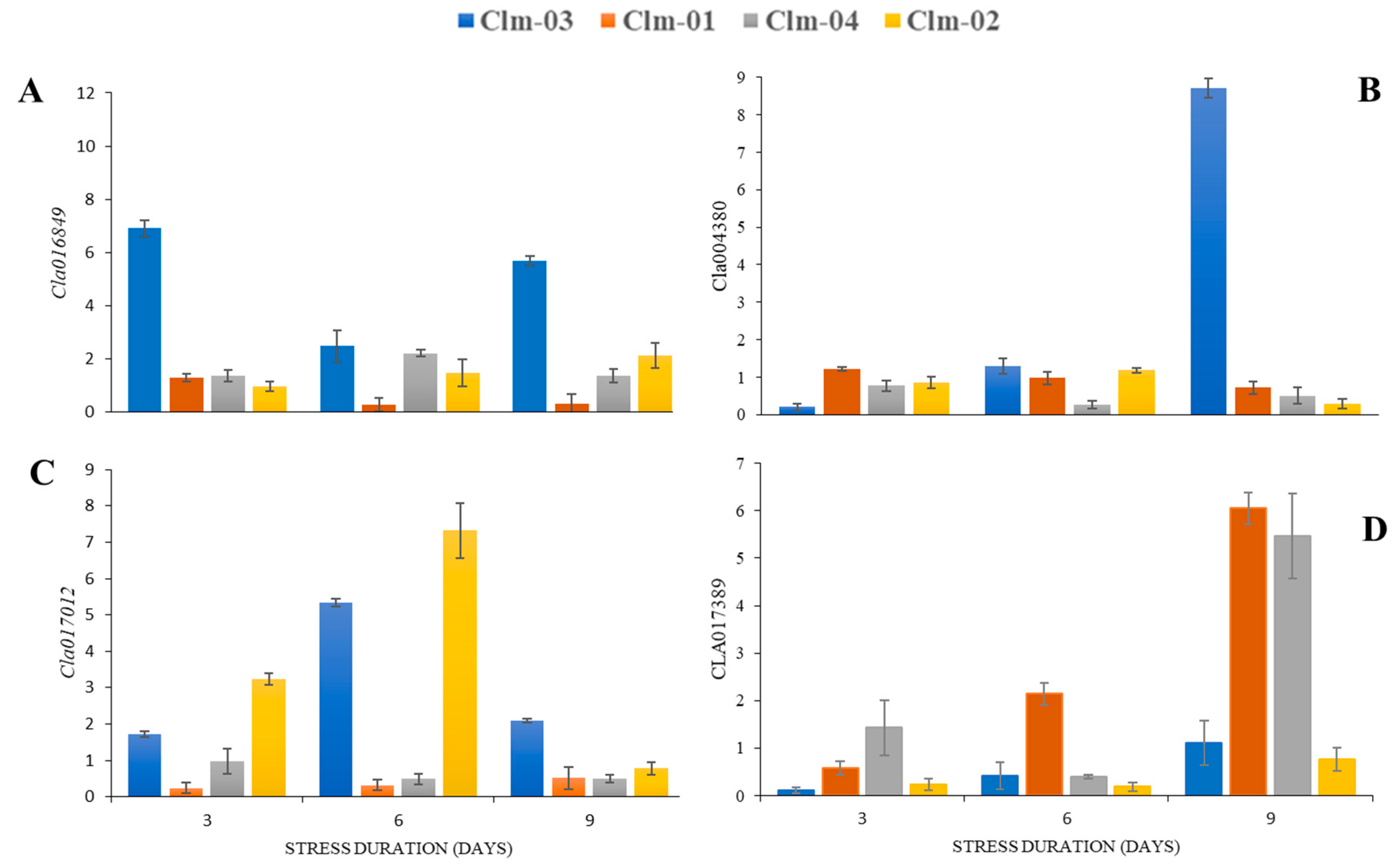
3.4. Relative expression of plant transcriptional regulators with a function in stomatal movement
Expression patterns of genes related to stomatal aperture showed that the genes studied were related to the stomata movements (
Figure 7). The two accessions (Clm0-02 and Clm-03) showed to limit their stomatal opening as drought stress progressed and shown by the expression of TF
Cla016489 and
Cla004380 (
Figure 7a and b) related to stomatal closure. Clm-03
Cla004380 showed the highest fold increase at day 9 of stress with about 8 fold increase as compared to the other genotypes. The Clm-01 and Clm-04had the lowest expression of both the TF studied for stomatal closure showing their stomatas keep open even in the extreme stress condition. Expression pattern on the TFs responsible for stomatal opening had contradictory results.
Cla017389 showed a higher expression in the Clm-01 and Clm-04 accession even in the extreme drought stress day while the Clm-03 and Clm-02 showed lesser expression thus suggesting that the opening was limited. A highest fold increase on
Cla017389 was observed in Clm-01 and Clm-04 at day 9 of drought stress with 5- and 6-fold increase respectively.
4. Discussion
3.1. Soil moisture content in response to imposition to drought stress
Root zone soil moisture plays a significant role in crop growth hence soil moisture can serve as an indicator of drought induced water stress in plants [
20]. However, the optimal range of soil moisture content for crops depends on specific plant species, and the range for most crops is between 20% and 60% [
21]. In this experiment drought stress resulted in highly significant reduction in soil moisture in all the studied watermelon genotypes to about 13.7 % recorded in Clm-02 at day 9 of stress while the highest was 14.6 % recorded in Clm-03. This is indicative of very high levels of moisture stress and the decreasing soil moisture triggered drought stress in watermelon plants as plant wilting was observed as early at day 3 of drought stress. The root zone moisture plays an important role in the stomatal movements as it regulates the root to shoot signals which the reduces the stomatal openings [
22]. The root-shot signals vary from plant to plant and this is very critical in the response of the plant to drought tolerance.
3.2. Physiological responses
Stomatal closure, one of the first plant responses to drought, is important in determining overall plant survival strategies [
23]. It is known to be a fast mechanism that reduces water loss and increases CO2 diffusion resistance to the mesophyll thus a crop that actively regulates its stomatas during stress has a greater chance of survival. In this study, the decrease in Gs was recorded for all the watermelon genotypes under drought stress, the decline in Gs, was much higher in Clm-03 than that in other genotypes under prolonged stress (
Figure 2A), which suggests that Clm-03 is better mechanisms to adapt to moisture stress conditions.
Relative water content, a key indicator of the degree of cell and tissue hydration, is crucial to optimum physiological functioning and growth processes [
24,
25]. In this study, RWC decreased in all watermelon genotypes with an increasing degree of water stress with wild watermelon having a higher RWC under all stress days (
Figure 2B). Thus, Clm-03 high level of RWC in the leaves exhibited greater drought stress tolerance as expected given the natural habitat of this crop. Our findings are consistent with the results of wild watermelon experimentally exposed to water deficit [
26]. Additionally, several studies on melon (
Cucumis melo L.), watermelon (
Citrullus lanatus) species have demonstrated parallel results on the effects of drought conditions on plant relative water contents [
27,
28,
29]. This suggests that drought tolerant plant species can maintain high leaf relative water content under drought stress conditions. The stomata have been noted to play an important role in in hydrological cycles which are responsible for regulating transpiration movement of water from plants to atmosphere and that limits their stomatal movement early in drought stress reduces moisture loss [
30]. The results have shown that the stomatal opening plays a significant role in tolerant plants maintaining the RWC as observed in the results where the wild watermelon experienced less changes in the RWC as the drought stress progressed. Thus, it can be suggested that stomatal closure plays an important role in this trait and subsequently aiding the wild watermelon to tolerate drought stress to some extent.
Chlorophyll contents (SPAD) determination is a critical indicator to evaluate plant performance under abiotic stress. Drought stress has been noted to accelerate chlorophyll decomposition [
29] in various plants. As expected, the results of this study show the negative impact of drought stress on Chl contents with a significant difference among all the genotypes (
Figure 2C). Similarly, to this experiment, [
31] reported that SPAD values significantly decreased from 35.5 to 22.4 in soybean [
Glycine max (L.) Merr.] under drought stress. The values significantly decreased with increasing water stress duration. However, Clm-02 had significantly higher SPAD values at all the stress days, highest of 50 at day 3 and the lowest was recorded in Clm-03, 19 at day 9. This result suggests Clm-02 seems to have a mechanism that protects chlorophyll from degradation under drought stress. Interestingly the Clm-03 which is thought to be more tolerant had lower values of chlorophyll during stress treatment but upon re-watering it showered greater recovery than other genotypes that showed recovery as indicated in
Figure 2C. However, it is possible that quick loss of chlorophyll under drought stress may also be an adaptive protective response to reduce photosynthesis which may be destructive when it takes place under drought stress [
32]. A decrease of total chlorophyll with drought stress implies a lowered capacity for light harvesting. Since the production of reactive oxygen species is mainly driven by excess energy absorption in the photosynthetic apparatus, when the balance between light absorption and carbon fixation is disrupted due to drought stress, this might be avoided by degrading the absorbing pigments [
33].
3.3. Chlorophyll fluorescence parameters
The Fv/Fm, PhiNPQ, qL and Phi2 values are the most important Chl fluorescence parameters which are broadly used in plant stress response studies [
34,
35,
36,
37]. With an inverse relationship between qL and PhiNPQ as well as Phi2 and PhiNPQ [
36]. In this study, the Phi2 and qL decreased with increasing value of PhiNPQ and were in line with the results reported by [
36,
38].
On the other hand, PhiNPQ which is the amount of dissipated excessive irradiation into heat as a protective measure. PhiNPQ represents an effective way by which photosynthetic organisms dissipate excessive irradiation into heat [
39]. As expected, values of PhiNPQ increased with an increase in drought stress duration. Results in fig. 4a show that under drought stress treatment local watermelon had the highest PhiNPQ values across all the three stress days, 0.48 ± 0.00, 0.54 ± 0.02 and 0.40 ± 0.02 respectively for day 3, 6 and 9 with the highest at day 6. Clm-02 had the lowest at day 3 and day 6 of stress, 0.19 ± 0.01 at day 3 and 0.16 ± 0.02 at day 6. Drought stress makes the electron transfer chain saturated and increase proton accumulation, therefore NPQ would increase [
40]. The higher value of NPQ indicates the ability to mitigate the negative effects of drought stress at the chloroplast level, as these organelles could dissipate the excess excitation energy [
41]. [
42], found out that the PhiNPQ was significantly high on a study on tomato (
Solanum lycopersicum). Based on the results it was suggested that it was unlikely that significant photo-inhibition occurred suggesting enhanced protection of the photosynthetic apparatus. This phenomenon has also been related to the observed increase in the antioxidant levels, especially carotenoids in the chloroplast [
43].
The Fv/Fm value as the ratio of variable fluorescence to maximal fluorescence measures the maximum efficiency of PSII (the efficiency if all PSII centers were open [
35]. Decrease in Fv/Fm reflects reduction in light use efficiency in plants [
44,
45]. In addition, increase in Fv/Fm is also correlated with reduced energy loss as heat [
46,
47,
48]. Thus, in this experiment it was observed that under drought stress Fv/Fm decreased with an increase in PhiNPQ,
Figure 3a and
Figure 4a. In this experiment the Fv/Fm fell to the lowest of 0.50 in local watermelon at day 6. The highest values were recorded in Clm-02 across all the three stress days, 0.71, 0.71 and 0.74 respectfully which was only 0.12% reduction compared to the average Fv/Fm values in control plants. This could possible present Clm-02 as having a very important mechanism in which it is able to reduce damage to the photosystem and ability to continue the photosynthetic process even under moisture stress. This could be true in that the Clm-02 usually produced large fruits even during extreme drought seasons [
49].
Water stress imposition caused a less significant reduction in Photosytem I Active Centres in this experiment. In contrast, in a study, proportion of PSI-active centres in three maize hybrids were higher under drought stress [
50]. This fortifies the idea that these additional PSI-active centres had a role in cyclic electron transport (CEF) to stimulate the proton-motive force that results in non-photochemical quenching (NPQ) to protect PS II [
51]. CEF also helps in avoiding over-reduction of PSI and generation of ROS under drought stress. In this study Clm-01 recorded the highest AC values at day 6 and 9 of intensified drought stress and this may present the ability to protect PS II under drought stress.
3.4. Stomatal movement in response to drought stress
As the survival of plants growing in drought conditions is associated to water economy, stomata in the leaves perform an important role in restricting water loss by transpiration [
52]. A reduction in stomatal index is an expected stomatal response in plants subject to drought stress [
53]. Drought stress resulted in reduction in stomatal aperture of all the watermelon genotypes under this study (
Figure 5 and
Figure 6). However, a highly significant reduction was recorded at extreme levels of drought stress (day 6 and 9). Clm-03 recorded the lowest value of stomatal aperture (14.7) at day 9 of drought stress and this has been associated to drought stress avoidance mechanism reported by [
54,
55]. [
54], reported that both medium and severe drought led to significant decreases in stomatal aperture in drought stressed maize (
Zea mays) plants compared with well-watered plants. It was suggested that by altering stomatal pore apertures, plants can optimize their CO2 uptake for photosynthesis while minimizing water loss which improves water use efficiency and ultimately lead to drought stress tolerance [
56]. The stomatal movement have been shown to play an important role under different environmental conditions and the stomata which are highly responsive might achieve important response to abiotic stress [
57]. In the current study the two probable tolerant landraces showed a rapid stomatal movement where they restricted the stomatal opening thus limiting moisture loss and aiding their tolerance. The hybrid watermelon which had a delayed stomatal closure showed signs of susceptibility to drought stress. This future proves that the stomata mainly its timely movement is important in the tolerance mechanisms of plants.
3.5. Relative expression of plant transcriptional regulators with a function in stomatal movement
Transcription factors naturally act as master regulators of cellular processes, they are expected to be excellent candidates for modifying complex traits in crop plants, such as response to abiotic stresses [
58]. The phytohormone abscisic acid (ABA) is an important regulator of plant responses to abiotic stresses [
59]. Drought stress results in accumulation of ABA, which initiates many adaptive responses [
60] including plant transcriptional regulators. Under water-deficit conditions, up regulated genes participating in the ABA signalling pathways for example, ABA-induced stomatal closure reduces transpirational water loss from plants [
61,
62].
Cla016849 and
Cla004380 transcription factors which have been documented to have an active role in guard cell ABA signalling and stomatal closure [
63] were studied. The highest expression of both of genes was recorded in wild watermelon (
Figure 7 A and B) at day 9 of stress and these results correlates with the morphological expression of stomatal aperture (
Figure 5 and 6). And this has been suggested as the most critical mechanisms the wild watermelon uses to manage the internal photosynthetic apparatus and also maintain internal moisture, this avoiding adverse effects of drought stress [
55,
64]. In a study, Arabidopsis ROC3 T-DNA mutants showed a reduced level of ABA-activated S-type anion currents, and stomatal closure than wild type [
63] thus showing the important role of ROC3 in regulating stomatal closure.
On the other hand, the
Cla016849 and
Cla017389 (
Figure 7 C and D) transcription factors involved in abscisic acid mediated stomatal opening were also studied. The results of
Cla016849 were not conclusive and thus were not discussed in this study.
Cla017389 which is a transcriptional factor involved in stomatal opening and it is ABA- dependent [
65]. In this study, a significantly high expression of
Cla017389 gene was observed in Clm-01 and Clm-04 at day 9 of drought stress. It was least expressed in Clm-03 at day 3 and 9. These results suggest that the two genotypes’ stomata remain open even at the extreme of drought stress something that could be associated with their susceptibility as continued photosynthesis during stress leads to damage to the photosynthetic apparatus thus leading to adverse effects on the plant. A study on yellow horn (
Xanthoceras sorbifolium) revealed that stomatal opening under drought stress may result in carbon dioxide diffusional limitations on leaf photosynthesis as well as unnecessary water loss when stomata continue to open after photosynthesis has reached saturation [
66].
Genetic factors have shown to play an important role in the stomatal movement as shown by several studies where over-expressions of genes [
67,
68,
69] related to stomatal movement results in significant changes that also aided plants to withstand abiotic stress like drought. Thus, genes from the tolerant species can play a significant role in improving the susceptible relatives or even other species. The expression of transcription factors related to stomatal closure was highly expressed in the two probable tolerant accessions in this study thus can be used for improving other crop species.
Conclusion
The four watermelon (Citrullus lanatus) accessions evaluated in this study, representing the major cultivated species of watermelon in Botswana and mostly southern Africa under varying climatic condition and having varying drought tolerance level as observed in most traditional farmer’s field. The result of this study further confirms the observations that of Clm-03 and Clm-02 are more tolerant than Clm-04 and Clm-01, which showed susceptibility. This is usually the case in traditional fields where even during the worst drought seasons there are always Clm-02 and Clm-03 plants and fruits available. This then presents the two landraces as an important research tool to fully understand their tolerance mechanisms and be harnessed to improve the susceptible crop species. Importantly the stomatal responses as observed in the morphological and molecular evaluations, shows that the closure plays a significant role in species tolerance as the tolerant landraces showed rapid closure when exposed to stress and response was a bit delayed when assessed in susceptible accession, and interestingly the inverse on stomatal aperture was also true on the accessions. This presents an important trait that needs to be fully studied and improve to successful develop climate smart crops that can survive in the extreme drought thus help in contributing to the dwindling food security.
Author Contributions
Conceptualization, K.M. and G.M.; methodology, K.M.; validation, G.M.; formal analysis, K.M.; investigation, K.M., L.T.S and M.N.N.; resources, G.M.; writing—original draft preparation, K.M.; writing—review and editing, K.M., G.M. and U.B..; supervision, G.M., U.B.; project administration, G.M.; funding acquisition, G.M . All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by SG-NAPI award supported by the German Ministry of Education and Research, BMBF through UNESCO-TWAS (4500454040) and BOTSWANA UNIVERSITY OF AGRICULTURE AND NATUAL RESOURCES.
Data Availability Statement
Data are available upon request.
Conflicts of Interest
The authors declare no conflict of interest
References
- Serdeczny, O.; Adams, S. Climate change impacts in Sub-Saharan Africa: from physical changes to their social repercussions. Reg. Environ. Change 2016, 16, 1–16. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Jacob, D.; Taylor, M. Impacts of 1.5°C global warming on natural and human systems. Global Warming 2018, 1.5, 175–311. [Google Scholar]
- Ramankutty, N.; Evan, A. T.; Monfreda, C. Farming the planet: 1. Geographic distribution of global agricultural lands in the year 2000. Glob. Biogeochem. Cycles 2002, 16, 1135. [Google Scholar] [CrossRef]
- Sloat, L.L.; Nagy, J.D.; Jones, C.S. Allometric scaling of gas exchange in crops and the implications for carbon balance. Glob. Change Biol 2020, 26, 2814–2828. [Google Scholar]
- Hadi, F.; Hassan, N.; Malik, A. Stomatal regulation for drought tolerance in plants. Int. J. Agric. Biol. 2016, 18, 282–289. [Google Scholar]
- Hajihashemi, S. Physiological and morphological alterations of stomata in response to drought stress. In Stomatal Function 2019, 63–85. [Google Scholar]
- Solis, C.A.; Machado, M.A.; Kozarewa, I. Genetic variation in the wild relatives of cereals: opportunities for crop improvement. Front. Plant Sci 2020, 11, 94. [Google Scholar]
- Guoxiong, C.; Yubao, L.; Shiping, W. Wild barley genetic diversity of drought tolerance. Plant Sci. 2002, 163, 837–843. [Google Scholar]
- Cai, C.; Zhu, J.; Lou, Y.; Guo, Z. Stomatal conductance of plant leaves responds to exogenous abscisic acid in a dosage-dependent manner. J. Plant Growth Regul 2017, 36, 1–10. [Google Scholar]
- Chen, S.L.; Chen, X.; Gao, L. The significance of volatile organic compounds in water relations of lower plants. Volatile Organic Compounds (VOCs) in Environ 2016, 1-12.
- Adhikari, K. N.; Buirchell, B. J.; Thomas, G. J.; Sweetingham, M. W.; Yang, H. Identification of anthracnose resistance in Lupinus albus L. and its transfer from landraces to modern cultivars. Crop and Pasture Science 2009, 60, 472. [Google Scholar] [CrossRef]
- Gupta, C.; Salgotra, R,K..; Mahajan, G. Future Threats and Opportunities Facing Crop Wild Relatives and Landrace Diversity. In: Salgotra, R., Zargar, S. (eds) Rediscovery of Genetic and Genomic Resources for Future Food Security. 2020. Springer, Singapore.
- Lovegrove, B.G. The burrows and burrowing behaviour of golden moles (Chrysochloridae). J. Zool 1993, 231, 529–542. [Google Scholar]
- Aninbon, C.; Jogloy, S.; Vorasoot, N.; Patanothai, A. Relationship between physiological and root traits of peanut genotypes under terminal drought stress. Int. J. Agric. Technol 2021, 17, 795–808. [Google Scholar]
- Smart, R.E.; Bingham, G,E. Rapid estimates of relative water content. Plant Physiol 1974, 53, 258–260. [Google Scholar] [CrossRef] [PubMed]
- Kuhlgert, S.; Austic, G.; Zegarac, R.; Osei-Bonsu, I.; Hoh, D.; Chilvers, M.; Roth, M.; Bi, K.; Teravest, D.; Weebadde, P.; Kramer, D. MultispeQ Beta: a tool for large-scale plant phenotyping connected to the open PhotosynQ network. R Soc Open Sci 2016, 3, 160592. [Google Scholar] [CrossRef] [PubMed]
- Pathoumthong, R.; Thammachuan, P.; Seifert, S. Rapid non-destructive method to phenotype stomatal traits. Plant Methods 2023, 19, 36. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q. S.; Yuan, J.; X.; Gao, L.Y.; Zhao, L.; Q.; Cheng, F.; Huang, Y.; et al. Evaluation of appropriate reference genes for gene expression normalization during watermelon fruit development. PLoS ONE 2015, 10, e0130865. [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Ajaz, A.; Taghvaeian, S.; Khand, K.; Gowda, P.; Moorhead, J. Development and Evaluation of an Agricultural Drought Index by Harnessing Soil Moisture and Weather Data. Water 2019, 11, 1375. [Google Scholar] [CrossRef]
- Reynolds-Henne, C.E.; Langenegger, A.; Mani, J.; Schenk, N.; Zumsteg, A.; Feller, U. Interactions between temperature, drought, and stomatal opening in legumes. Environ. Exp. Bot 2010, 68, 37–43. [Google Scholar] [CrossRef]
- Liu, X.; Wei, Z.; Manevski, K.; Liu, J.; Ma, Y.; Andersen, M.N.; Liu, F. Partial root-zone drying irrigation increases water-use efficiency of tobacco plants amended with biochar, Industrial Crops and Products, 2021, 166, 113487, ISSN 0926-6690. [CrossRef]
- Jamaux, I.; Steinmertz, A.; Belhassen, E. Looking for molecular and physiological markers of osmotic adjustment in sunflower. New Phytologist 1997, 137, 117–127. [Google Scholar] [CrossRef]
- Altinkut, A.; Kazan, K.; Ipekci, Z.; Gozukirmizi, N. Tolerance to paraquat is correlated with the traits associated with water stress tolerance in segregating F2 populations of barley and wheat. Euphytica 2001, 121, 81–86. [Google Scholar] [CrossRef]
- Kawasaki, S. M.; Chikahiro, K.; Takayuki, F.; Shinichiro, U.; Masato, Y.A. Responses of Wild Watermelon to Drought Stress: Accumulation of an ArgE Homologue and Citrulline in Leaves during Water Deficits. Plant & Cell Physiology 2000, 41, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Goodarzian, G.M.; Mansurifar, S; Taghizadeh-Mehrjardi, R.; Saeidi, M.; Jamshidi, A.M.; Ghasemi, E. Effects of drought stress and rewatering on antioxidant systems and relative water content in different growth stages of maize (Zea mays L.) hybrids. Archives of Agronomy and Soil Science 2015, 61, 493–506.
- Mo, Y.; Wang, Y.; Yang, R.; Zheng, J.; Liu, C.; Li, H.; Ma, J.; Zhang, Y.; Wei, C.; Zhang, X. Regulation of Plant Growth, Photosynthesis, Antioxidation and Osmosis by an Arbuscular Mycorrhizal Fungus in Watermelon Seedlings under Well-Watered and Drought Conditions. Front. Plant Sci 2016, 7, 644. [Google Scholar] [CrossRef]
- Liang, G.; Liu, J.; Zhang, J.; Guo, J. Effects of Drought Stress on Photosynthetic and Physiological Parameters of Tomato. J. Amer. Soc. Hort. Sci 2020, 145, 12–17. [Google Scholar] [CrossRef]
- Nankishore, A.; Farrell, A. The response of contrasting tomato genotypes to combined heat and drought stress. Journal of Plant Physiology 2016, 202. [Google Scholar] [CrossRef] [PubMed]
- Haworth, M.; Marino, G.; Loreto, F.; Centritto, M. Integrating stomatal physiology and morphology: evolution of stomatal control and development of future crops. Oecologia. 2021, 197, 867–883. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Kjaer, K.; Rosenqvist, E.; Yu, X.; Wu, Z.; Ottosen, C.O. Physiological Response to Heat Stress during Seedling and Anthesis Stage in Tomato Genotypes Differing in Heat Tolerance. Journal of Agronomy and crop science 2017, 203, 68–80. [Google Scholar] [CrossRef]
- Ergo, V.V.; Lascano, Ramiro, Vega, Claudia, R.C., Parola, R., Carrera, Constanza, S. Heat and water stressed field-grown soybean: A multivariate study on the relationship between physiological-biochemical traits and yield. Environmental and Experimental Botany 2017, 148, 1-11. [CrossRef]
- Mafakheri, A.; Siosemardeh, A.; Bahramnejad, B.; Struik, P.C.; Sohrabi, Y. Effect of drought stress on yield, proline and chlorophyll contents in three chickpea cultivars. AJCS 2010, 4, 580–585. [Google Scholar]
- Herbinger, K.; Tausz, M.; Wonisch, A.; Soja,G.; Sorger, A.; Grill, D. Complex interactive effects of drought and ozone stress on the antioxidant defence systems of two wheat cultivars. Plant Physiol. Biochem 2002, 40, 691–696. [CrossRef]
- Thomas, D.S.; Turner, D.W. Banana (Musa sp.) leaf gas exchange and chlorophyll fluorescence in response to soil drought, shading, and lamina folding. Sci. Hortic 2001, 90, 93–108. [Google Scholar]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. Journal of Experimental Botany 2004, 55, 1607–1621. [Google Scholar] [CrossRef]
- Fu, W.; Li, P.; Wu, Y. Effects of different light intensities on chlorophyll fluorescence characteristics and yield in lettuce. Scientia Horticulturae 2012, 135, 45–51. [Google Scholar] [CrossRef]
- Murchie, E.H.; Lawson, T. Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. J. Exp. Bot 2013, 64, 3983–3998. [Google Scholar] [CrossRef] [PubMed]
- Malambane, G.; Batlang, U.; Ramolekwa, K.; Tsujimoto, H.; Akashi, K. Growth chamber and field evaluation of physiological factors of two watermelon genotypes. Plant Stress 2021, 2, 100017. [Google Scholar] [CrossRef]
- Pinnola, A.; Dall’Osto, L.; Gerotto, C.; Morosinotto, T.; Bassi, R.; Alboresi, A. Zeaxanthin binds to light-harvesting complex stress-related protein to enhance nonphotochemical quenching in Physcomitrella patens. Plant Cell 2013, 25, 3519–3534. [Google Scholar] [CrossRef] [PubMed]
- Porcar-Castell, A.; Tyystj€arvi, E.; Atherton, J.; Van Der Tol, C.; Flexas, J.; Pfündel, E.E.; Moreno, J.; Frankenberg, C.; Berry, J.A. Linking chlorophyll a fluorescence to photosynthesis for remote sensing applications: mechanisms and challenges. J. Exp. Bot 2014, 65, 4065–4095. [Google Scholar] [CrossRef] [PubMed]
- Ismail, I. M.; Basahi, J.M.; Hassan, I. A. Gas exchange and chlorophyll fluorescence of pea (Pisum sativum L.) plants in response to ambient ozone at a rural site in Egypt. Science of the Total Environment 2014, 497, 585–593. [Google Scholar]
- Gitau, M.M.; Farkas, A.; Ördög, V.; Maróti, G. Evaluation of the biostimulant effects of two Chlorophyta microalgae on tomato (Solanum lycopersicum). Journal of Cleaner Production 2022, 364. [Google Scholar] [CrossRef]
- Farid, R.; Mutale-joan, C.; Redouane, B. et al. Effect of Microalgae Polysaccharides on Biochemical and Metabolomics Pathways Related to Plant Defense in Solanum lycopersicum. Appl Biochem Biotechnol 2019, 188, 225–240. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annual Review of Plant Biology 2008, 59, 89–113. [Google Scholar] [CrossRef]
- Li, S.; Yang, W.; Yang, T.; Chen, Y.; Ni, W. Effects of cadmium stress on leaf chlorophyll fluorescence and photosynthesis of Elsholtzia argyi da cadmium accumulating plant. Int. J. Phytoremediation 2015, 17, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, V.; Bhargava, S.; Streb, P.; Feierabend, J. Comparative effect of water, heat, and light stresses on photosynthetic reactions in Sorghum bicolor (L.) Moench. Journal of Experimental Botany 1998, 49, 1715–1721. [Google Scholar]
- Jeon, M.W.; Ali, M.B.; Hahn, E.J.; Paek, K.Y. Photosynthetic pigments, morphology, and leaf gas exchange during ex vitro acclimatization of micropropagated CAM doritaenopsis plantlets under relative humidity and air temperature. Environmental and Experimental Botany 2006, 55, 183–194. [Google Scholar] [CrossRef]
- Broetto, F.; Monteiro Duarte, H.; Lüttge, U. Responses of chlorophyll fluorescence parameters of the facultative halophyte and C3-CAM intermediate species Mesembryanthemum crystallinum to salinity and high irradiance stress. Journal of Plant Physiology 2007, 164, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Mandizvo, T.; Odindo, A.O.; Mashilo, J.; Magwaza, L.S. Drought tolerance assessment of citron watermelon (Citrullus lanatus var. citroides (L.H. Bailey) Mansf. ex Greb.) accessions based on morphological and physiological traits. Plant Physiol Biochem 2022, 180, 106–123. [Google Scholar] [CrossRef] [PubMed]
- Bashir, N.; Athar, H.; Hazem, M.; Kalaji, J. W.; Mahmood, S.; Zafar, Z. U.; Ashraf, M. Is Photoprotection of PSII One of the Key Mechanisms for Drought Tolerance in Maize? International Journal of Molecular Sciences 2021, 22, 13490. [Google Scholar] [CrossRef] [PubMed]
- Ruban, A.V.; Wilson, S. The Mechanism of Non-Photochemical Quenching in Plants: Localization and Driving Forces. Plant Cell Physiol 2021, 62, 1063–1072. [Google Scholar] [CrossRef]
- Bosabalidis, A. M.; Kofidis, G. Comparative effects of drought stress on leaf anatomy of two olive cultivars. Plant Science 2002, 163, 375–379. [Google Scholar] [CrossRef]
- Xu, Z.Z.; Zhou, G.S. Effects of water stress and high nocturnal temperature on photosynthesis and nitrogen level of a perennial grass Leymus chinensis. Plant Soil 2005, 269, 131–139. [Google Scholar] [CrossRef]
- Fang, Y.; Xiong, L. General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 2015, 72, 673–89. [Google Scholar] [CrossRef]
- Malambane, G.; Madumane, K.; Sewelo, L. T.; Batlang, U. Drought stress tolerance mechanisms and their potential common indicators to salinity, insights from the wild watermelon (Citrullus lanatus): A review. Front Plant Sci 2023, 13, 1074395. [Google Scholar] [CrossRef] [PubMed]
- Bertolino, L.T.; Caine, R.S.; Gray, J. E; Impact of stomatal density and morphology on water use efficiency in a changing world. Front Plant Sci 2019, 10. [Google Scholar] [CrossRef]
- Century, K.; Lynne, R.; Oliver, J. R. Regulating the Regulators: The Future Prospects for Transcription-Factor-Based Agricultural Biotechnology Products. Plant Physiol 2008, 147, 20–29. [Google Scholar] [CrossRef]
- Finkelstein, R.; Gampala, S.; Rock, C. Abscisic acid signaling in seeds and seedlings. Plant Cell 2002, 14, 15–45. [Google Scholar] [CrossRef]
- Leung, J.; Giraudat, J. Abscisic acid signal transduction. Annu Rev Plant Physiol Plant Mol Biol 1998, 49, 199–222. [Google Scholar] [CrossRef]
- Nilson, S.E.; Assman, S.M. The Control of transpiration. Insights from Arabidopsis. Plant Physiol 2007, 143, 19–27. [Google Scholar] [CrossRef]
- Schroeder, J.I.; Kwak, J.M.; Allen, G.J. Guard cell abscisic acid signaling and engineering drought hardiness in plants. Nature 2001, 410, 327–330. [Google Scholar] [CrossRef]
- Liu, H.; Shen, J.; Yuan, C, Lu, D.; Acharya, B.; Wang, M.; Chen, D.; Zhang, W. The Cyclophilin ROC3 Regulates ABA-Induced Stomatal Closure and the Drought Stress Response of Arabidopsis thaliana. Front Plant Sci 2021, 12. [CrossRef] [PubMed]
- Nanasato, Y.; Miyake, C.; Takahara, K.; Kohzuma, K.; Munekage, Y. N.; Yokota, A.; Akashi, K. Chapter 23 Mechanisms of Drought and High Light Stress Tolerance Studied in a Xerophyte, Citrullus lanatus (Wild Watermelon). Advances in Ph tosynthesis and Respiration 2010, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Eleonora, C.; Massimo, G.; Chiara, T. Transcription: Transcription factors controlling stomatal movements and drought tolerance. Transcription 2010, 1, 41–45. [Google Scholar] [CrossRef]
- Matthews, J.S.A.; Vialet-Chabrand, S.; Lawson, T. Role of blue and red light in stomatal dynamic behavior. J Exp Bot 2020, 71, 2253–2269. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qin, L.; Han, L.; Xiang, Y.; Zhao, D. Overexpression of maize SDD1 (ZmSDD1) improves drought resistance in Zea mays L. by reducing stomatal density. Plant Cell Tissue Organ Cult. 2015, 122, 147–159. [CrossRef]
- Hughes, J.; Hepworth, C.; Dutton, C.; Dunn, J,A.; Hunt, L.; Stephens, J., et al.. (2017). Reducing stomatal density in barley improves drought tolerance without impacting on yield. Plant Physiol. 2017, 174, 776–787. [CrossRef]
- Caine, R.S.; Yin, X.; Sloan, J.; Harrison, E.L.; Mohammed, U.; Fulton, T. et al... Rice with reduced stomatal density conserves water and has improved drought tolerance under future climate conditions. New Phytol. 2019, 221, 371–384. [CrossRef]
Figure 1.
Soil moisture content response to drought stress in non-stress (control) plants (left panel) and drought stressed plants (right panel). In stressed plants day 12 represents data after re-watering. The error bars show the standard deviation of means, for n=3.
Figure 1.
Soil moisture content response to drought stress in non-stress (control) plants (left panel) and drought stressed plants (right panel). In stressed plants day 12 represents data after re-watering. The error bars show the standard deviation of means, for n=3.
Figure 2.
The effects of water stress on (A) stomatal conductance (GS), (B) relative water content (RWC) and (C) chlorophyll content measured as SPAD in non-stress plants, (left panel) and drought stressed plants, (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means.
Figure 2.
The effects of water stress on (A) stomatal conductance (GS), (B) relative water content (RWC) and (C) chlorophyll content measured as SPAD in non-stress plants, (left panel) and drought stressed plants, (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means.
Figure 3.
The effects of water stress on (A) photochemical quenching of variable chlorophyll fluorescence (qL), (B) quantum yield of photosystem II (Fv/Fm), and (C) Photosystem I active centers in the non-stress plants (left panel) and water stressed plants (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means for n=3.
Figure 3.
The effects of water stress on (A) photochemical quenching of variable chlorophyll fluorescence (qL), (B) quantum yield of photosystem II (Fv/Fm), and (C) Photosystem I active centers in the non-stress plants (left panel) and water stressed plants (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means for n=3.
Figure 4.
The effects of drought stress on (A) quantum yield of regulated energy dissipation in PS II (PhiNPQ), (B) quantum yield of non-regulated energy dissipation in PS II (PhiNO) and (C) effective quantum yield of photochemical energy conversion in PS II (Phi2) in the non-stress plants (left panel) and water stressed plants (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means, for n=3. Wild watermelon (WW) is represented by solid lines with kites in both the non-stress and water stressed plants, and Local Watermelon is represented by broken lines with squares while hybrid Crimson Sweet (CS) is represented by broken lines with triangles and lastly the broken lines with crosses represent Cooking Watermelon (CW).
Figure 4.
The effects of drought stress on (A) quantum yield of regulated energy dissipation in PS II (PhiNPQ), (B) quantum yield of non-regulated energy dissipation in PS II (PhiNO) and (C) effective quantum yield of photochemical energy conversion in PS II (Phi2) in the non-stress plants (left panel) and water stressed plants (right panel). In stressed plants day 12 and 15 represent data after re-watering. The error bars show the standard deviation of means, for n=3. Wild watermelon (WW) is represented by solid lines with kites in both the non-stress and water stressed plants, and Local Watermelon is represented by broken lines with squares while hybrid Crimson Sweet (CS) is represented by broken lines with triangles and lastly the broken lines with crosses represent Cooking Watermelon (CW).
Figure 5.
The images show the effects of drought stress on stomatal morphology of four watermelon genotypes. The images were taken under the inverted biological microscope. Day 0 represents the first day before drought stress imposition, whereas day 3 to day 9 are the days of drought stress duration and lastly day 12 is the day after re-watering.
Figure 5.
The images show the effects of drought stress on stomatal morphology of four watermelon genotypes. The images were taken under the inverted biological microscope. Day 0 represents the first day before drought stress imposition, whereas day 3 to day 9 are the days of drought stress duration and lastly day 12 is the day after re-watering.
Figure 6.
The effects of drought stress on stomatal aperture response of four watermelon genotypes. Day 0 represents the first day before drought stress imposition, whereas day 3 to day 9 are the days of drought stress duration and lastly day 12 is the day after re-watering. The error bars show the standard deviation of means, for n=5.
Figure 6.
The effects of drought stress on stomatal aperture response of four watermelon genotypes. Day 0 represents the first day before drought stress imposition, whereas day 3 to day 9 are the days of drought stress duration and lastly day 12 is the day after re-watering. The error bars show the standard deviation of means, for n=5.
Figure 7.
Results from the gene expression analysis of four watermelon genotypes under three drought stress days (Day 3, 6 and 9). The Delta-Delta CT relative quantification method was used to estimate mRNA levels of targeted genes A (Cla016489) and B (Cla004380) which are involved in stomatal closure through abscisic acid mediated pathway. Similarly, targeted genes C (Cla017012) and D (Cla017389) which are involved in stomatal opening through abscisic acid mediated pathway were quantified. The quantification for all the targeted genes was relative to an average of two reference genes (Tublin and GAPDH).. The error bars show the standard deviation of means.
Figure 7.
Results from the gene expression analysis of four watermelon genotypes under three drought stress days (Day 3, 6 and 9). The Delta-Delta CT relative quantification method was used to estimate mRNA levels of targeted genes A (Cla016489) and B (Cla004380) which are involved in stomatal closure through abscisic acid mediated pathway. Similarly, targeted genes C (Cla017012) and D (Cla017389) which are involved in stomatal opening through abscisic acid mediated pathway were quantified. The quantification for all the targeted genes was relative to an average of two reference genes (Tublin and GAPDH).. The error bars show the standard deviation of means.
Table 1.
Information on the Citrullus lanatus (watermelon) accessions used in this study.
Table 1.
Information on the Citrullus lanatus (watermelon) accessions used in this study.
| Code |
Accession type |
Cultivation |
Source |
Type of use |
Flesh colour |
| Clm-01 |
Landrace |
Domesticated |
BNPGRC |
Fresh eating |
Red |
| Clm-02 |
Landrace |
Domesticated |
BNPGRC |
Cooking |
Yellow |
| Clm-03 |
landrace |
Wild |
BNPGRC |
Livestock feed |
White |
| Clm-04 |
Hybrid |
Domesticated |
Commercial shops |
Fresh eating |
Red |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).