Submitted:
08 December 2023
Posted:
11 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Association of m6A and ncRNAs in various liver diseases
4. Attention to YTHDF2, an m6A reader molecule, in the pathogenesis of obesity
5. Relationship between YTHDF2 and Diabetes mellitus
6. Future perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Liu, H.; Hao, W.; Yang, J.; Zhang, Y.; Wang, X.; Zhang, C. Emerging roles and potential clinical applications of translatable circular RNAs in cancer and other human diseases. Genes Dis. 2023, 10, 1994–2012. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Gorospe, M.; Panda, A.C. The coding potential of circRNAs. Aging 2018, 10, 2228–2229. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Chen, S.; Han, J.-X.; Qian, B.; Wang, X.-R.; Zhong, W.-L.; Qin, Y.; Zhang, H.; Gao, W.-F.; Lei, Y.-Y.; et al. Twist1 Regulates Vimentin through Cul2 Circular RNA to Promote EMT in Hepatocellular Carcinoma. Cancer Res 2018, 78, 4150–4162. [Google Scholar] [CrossRef] [PubMed]
- Memczak, S.; Jens, M.; Elefsinioti, A.; Torti, F.; Krueger, J.; Rybak, A.; Maier, L.; Mackowiak, S.D.; Gregersen, L.H.; Munschauer, M.; et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 2013, 495, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Rybak-Wolf, A.; Stottmeister, C.; Glažar, P.; Jens, M.; Pino, N.; Giusti, S.; Hanan, M.; Behm, M.; Bartok, O.; Ashwal-Fluss, R.; et al. Circular RNAs in the Mammalian Brain Are Highly Abundant, Conserved, and Dynamically Expressed. Mol. Cell 2015, 58, 870–885. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Liu, C.-H.; Wu, D.; Jiang, W.; Zhang, N.; Tang, H. LncRNA and circRNA in Patients with Non-Alcoholic Fatty Liver Disease: A Systematic Review. Biomolecules 2023, 13, 560. [Google Scholar] [CrossRef]
- Kopp, F.; Mendell, J.T. Functional Classification and Experimental Dissection of Long Noncoding RNAs. Cell 2018, 172, 393–407. [Google Scholar] [CrossRef]
- Zhang, Y.; Mao, Q.; Xia, Q.; Cheng, J.; Huang, Z.; Li, Y.; Chen, P.; Yang, J.; Fan, X.; Liang, Y.; et al. Noncoding RNAs link metabolic reprogramming to immune microenvironment in cancers. J. Hematol. Oncol. 2021, 14, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Yuan, X.; Wu, S.; Yuan, Y.; Cui, L.; Lin, D.; Peng, X.; Liu, X.; Wang, F. Effects of writers.; erasers and readers within miRNA-related m6A modification in cancers. Cell Prolif. 2023, 56, e13340.
- Xu, K.; Sun, Y.; Sheng, B.; Zheng, Y.; Wu, X.; Xu, K. Role of identified RNA N6-methyladenosine methylation in liver. Anal. Biochem. 2019, 578, 45–50. [Google Scholar] [CrossRef]
- Lee, Y.; Choe, J.; Park, OH.; Kim, YK. Molecular Mechanisms Driving mRNA Degradation by m6A Modification. Trends Genet. 2020, 36, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Dominissini, D.; Rechavi, G.; He, C. Gene expression regulation mediated through reversible m6A RNA methylation. Nat. Rev. Genet. 2014, 15, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Kholodenko, IV.; Yarygin, KN. Hepatic Macrophages as Targets for the MSC-Based Cell Therapy in Non-Alcoholic Steatohepatitis. Biomedicines. 2023, 11, 3056. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Yu, F.; Yuan, G.; Jia, J. Update on N6-methyladenosine methylation in obesity-related diseases. Obesity 2023, 32, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Tian, X.; Yan, T.; Wang, H.; Zhang, D.; Lin, C.; Liu, Q.; Jiang, S. Insights into the role of nucleotide methylation in metabolic-associated fatty liver disease. Front. Immunol. 2023, 14, 1148722. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Feng, Y.; Zhang, L.; Jia, Y.; Cai, D.; Qian, S.-B.; Du, M.; Zhao, R. GR-mediated FTO transactivation induces lipid accumulation in hepatocytes via demethylation of m6A on lipogenic mRNAs. RNA Biol. 2020, 17, 930–942. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Tian, S.; Zheng, X.; Zhang, M.; Zhou, X.; Shang, Y.; Han, Y. N6-methyladenosine RNA methylation in liver diseases: from mechanism to treatment. J. Gastroenterol. 2023, 58, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Meyer, KD.; Jaffrey, SR. Rethinking m6A Readers. ; Writers.; and Erasers. Annu Rev Cell Dev Biol. 2017, 33, 319–42. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Zhang, Q.; Xu, Y.; Ma, S.; Wang, T.; Huang, Y.; Ju, S. m6A-modified circRNAs: detections, mechanisms, and prospects in cancers. Mol. Med. 2022, 28, 1–17. [Google Scholar] [CrossRef]
- Yang, Y.; Shen, F.; Huang, W.; Qin, S.; Huang, J.-T.; Sergi, C.; Yuan, B.-F.; Liu, S.-M. Glucose Is Involved in the Dynamic Regulation of m6A in Patients With Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2018, 104, 665–673. [Google Scholar] [CrossRef]
- Li, Z.; Weng, H.; Su, R.; Weng, X.; Zuo, Z.; Li, C.; Huang, H.; Nachtergaele, S.; Dong, L.; Hu, C.; et al. FTO Plays an Oncogenic Role in Acute Myeloid Leukemia as a N 6 -Methyladenosine RNA Demethylase. Cancer Cell 2016, 31, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, S.; Nakano, M.; Fukami, T.; Nakajima, M. m6A modification impacts hepatic drug and lipid metabolism properties by regulating carboxylesterase 2. Biochem. Pharmacol. 2021, 193, 114766. [Google Scholar] [CrossRef] [PubMed]
- Bansal, H.; Yihua, Q.; Iyer, S.P.; Ganapathy, S.; Proia, D.; O Penalva, L.; Uren, P.J.; Suresh, U.; Carew, J.S.; Karnad, A.B.; et al. WTAP is a novel oncogenic protein in acute myeloid leukemia. Leukemia 2014, 28, 1171–1174. [Google Scholar] [CrossRef] [PubMed]
- Patop, IL.; Wüst, S.; Kadener, S. Past. ; present.; and future of circRNAs. EMBO J. 2019, 38, e100836. [Google Scholar] [PubMed]
- Lee, C.Q.E.; Kerouanton, B.; Chothani, S.; Zhang, S.; Chen, Y.; Mantri, C.K.; Hock, D.H.; Lim, R.; Nadkarni, R.; Huynh, V.T.V.T.; et al. Coding and non-coding roles of MOCCI (C15ORF48) coordinate to regulate host inflammation and immunity. Nat. Commun. 2021, 12, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Liu, B.; Nie, Z.; Duan, L.; Xiong, Q.; Jin, Z.; Yang, C.; Chen, Y. The role of m6A modification in the biological functions and diseases. Signal Transduct. Target. Ther. 2021, 6, 1–16. [Google Scholar] [CrossRef]
- Shao, Y.; Liu, Z.; Song, X.; Sun, R.; Zhou, Y.; Zhang, D.; Sun, H.; Huang, J.; Wu, C.; Gu, W.; Zheng, X.; Jiang, J. ALKBH5/YTHDF2-mediated m6A modification of circAFF2 enhances radiosensitivity of colorectal cancer by inhibiting Cullin neddylation. Clin Transl Med. 2023, 13, e1318. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Zhang, X.; Cong, H. Advances in the protein-encoding functions of circular RNAs associated with cancer (Review). Oncol. Rep. 2023, 50, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Zheng, G.; Ning, Q.; Zheng, J.; Dong, D. Translation and functional roles of circular RNAs in human cancer. Mol. Cancer 2020, 19, 1–9. [Google Scholar] [CrossRef]
- Wu, Y.; Zeng, Y.; Ren, Y.; Yu, J.; Zhang, Q.; Xiao, X. Insights into RNA N6-methyladenosine in Glucose and Lipid Metabolic Diseases and Their Therapeutic Strategies. Endocrinology 2023, 165. [Google Scholar] [CrossRef]
- Wang, W.; Yan, J.; Han, L.; Zou, Z.-L.; Xu, A.-L. Silencing METTL14 alleviates liver injury in non-alcoholic fatty liver disease by regulating mitochondrial homeostasis. Biomol. Biomed. 2024, 24, 505–519. [Google Scholar] [CrossRef] [PubMed]
- Farías, C.; Cisternas, C.; Gana, J.C.; Alberti, G.; Echeverría, F.; Videla, L.A.; Mercado, L.; Muñoz, Y.; Valenzuela, R. Dietary and Nutritional Interventions in Nonalcoholic Fatty Liver Disease in Pediatrics. Nutrients 2023, 15, 4829. [Google Scholar] [CrossRef] [PubMed]
- Gómez de Cedrón, M.; Moreno Palomares, R.; Ramírez de Molina, A. Metabolo-epigenetic interplay provides targeted nutritional interventions in chronic diseases and ageing. Front Oncol. 2023, 13, 1169168. [Google Scholar] [CrossRef] [PubMed]
- Farsetti, A.; Illi, B.; Gaetano, C. How epigenetics impacts on human diseases. Eur. J. Intern. Med. 2023, 114, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Chen, X.; Zhu, S.; Wang, J.; Diao, S.; Liu, J.; Xu, J.; Li, X.; Sun, Y.; Huang, C.; et al. Decoding m6A mRNA methylation by reader proteins in liver diseases. Genes Dis. 2024, 11, 711–726. [Google Scholar] [CrossRef]
- Moghadam, S.K.; Bakhshinejad, B.; Khalafizadeh, A.; Hussen, B.M.; Babashah, S. Non-coding RNA-associated competitive endogenous RNA regulatory networks: Novel diagnostic and therapeutic opportunities for hepatocellular carcinoma. J. Cell. Mol. Med. 2021, 26, 287–305. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Tan, QQ.; Tan, XR.; Li, SJ.; Zhang, XX. Circ_0057558 promotes nonalcoholic fatty liver disease by regulating ROCK1/AMPK signaling through targeting miR-206. Cell Death Dis. 2021, 12, 809. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Zheng, H.; Wu, Z.; Chen, M.; Huang, Y. Circular RNA-protein interactions: functions, mechanisms, and identification. Theranostics 2020, 10, 3503–3517. [Google Scholar] [CrossRef] [PubMed]
- Larsson, O.; Perlman, D.M.; Fan, D.; Reilly, C.S.; Peterson, M.; Dahlgren, C.; Liang, Z.; Li, S.; Polunovsky, V.A.; Wahlestedt, C.; et al. Apoptosis resistance downstream of eIF4E: posttranscriptional activation of an anti-apoptotic transcript carrying a consensus hairpin structure. Nucleic Acids Res. 2006, 34, 4375–4386. [Google Scholar] [CrossRef]
- Karim, M.M.; Svitkin, Y.V.; Kahvejian, A.; De Crescenzo, G.; Costa-Mattioli, M.; Sonenberg, N. A mechanism of translational repression by competition of Paip2 with eIF4G for poly(A) binding protein (PABP) binding. Proc. Natl. Acad. Sci. 2006, 103, 9494–9499. [Google Scholar] [CrossRef]
- Yang, M.; Qi, M.; Xu, L.; Huang, P.; Wang, X.; Sun, J.; Shi, J.; Hu, Y. Differential host circRNA expression profiles in human lung epithelial cells infected with SARS-CoV-2. Infect Genet Evol. 2021, 93, 104923. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Wang, X.; Sun, H.; Xu, B.; Song, R.; Tian, Y.; Zhao, L.; Xu, Y.; Zhao, Y.; Yang, F.; et al. Oxidant stress-sensitive circRNA Mdc1 controls cardiomyocyte chromosome stability and cell cycle re-entry during heart regeneration. Pharmacol. Res. 2022, 184, 106422. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, Y.; Morikawa, S.; Nakashima, M.; Yoshikawa, S.; Taniguchi, K.; Sawamura, H.; Suga, N.; Tsuji, A.; Matsuda, S. CircRNAs and RNA-Binding Proteins Involved in the Pathogenesis of Cancers or Central Nervous System Disorders. Non-Coding RNA 2023, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Winkler, G.S. The mammalian anti-proliferative BTG/Tob protein family. J. Cell. Physiol. 2009, 222, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Ezzeddine, N.; Chang, T.-C.; Zhu, W.; Yamashita, A.; Chen, C.-Y.A.; Zhong, Z.; Yamashita, Y.; Zheng, D.; Shyu, A.-B. Human TOB, an Antiproliferative Transcription Factor, Is a Poly(A)-Binding Protein-Dependent Positive Regulator of Cytoplasmic mRNA Deadenylation. Mol. Cell. Biol. 2007, 27, 7791–7801. [Google Scholar] [CrossRef] [PubMed]
- Tirone, F. The gene PC3TIS21/BTG2, prototype member of the PC3/BTG/TOB family: Regulator in control of cell growth, differentiation, and DNA repair? J. Cell. Physiol. 2001, 187, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Doidge, R.; Mittal, S.; Aslam, A.; Winkler, G.S. The Anti-Proliferative Activity of BTG/TOB Proteins Is Mediated via the Caf1a (CNOT7) and Caf1b (CNOT8) Deadenylase Subunits of the Ccr4-Not Complex. PLOS ONE 2012, 7, e51331. [Google Scholar] [CrossRef]
- Lim, N.S.; Kozlov, G.; Chang, T.-C.; Groover, O.; Siddiqui, N.; Volpon, L.; De Crescenzo, G.; Shyu, A.-B.; Gehring, K. Comparative Peptide Binding Studies of the PABC Domains from the Ubiquitin-protein Isopeptide Ligase HYD and Poly(A)-binding Protein. J. Biol. Chem. 2006, 281, 14376–14382. [Google Scholar] [CrossRef] [PubMed]
- Horiuchi, M.; Takeuchi, K.; Noda, N.; Muroya, N.; Suzuki, T.; Nakamura, T.; Kawamura-Tsuzuku, J.; Takahasi, K.; Yamamoto, T.; Inagaki, F. Structural basis for the antiproliferative activity of the Tob-hCaf1 complex. J. Biol. Chem. 2009, 284, 13244–13255. [Google Scholar] [CrossRef]
- Ezzeddine, N.; Chen, C.-Y.A.; Shyu, A.-B. Evidence Providing New Insights into TOB-Promoted Deadenylation and Supporting a Link between TOB's Deadenylation-Enhancing and Antiproliferative Activities. Mol. Cell. Biol. 2012, 32, 1089–1098. [Google Scholar] [CrossRef]
- Stupfler, B.; Birck, C.; Séraphin, B.; Mauxion, F. BTG2 bridges PABPC1 RNA-binding domains and CAF1 deadenylase to control cell proliferation. Nat. Commun. 2016, 7, 10811. [Google Scholar] [CrossRef]
- Hwang, S.S.; Lim, J.; Yu, Z.; Kong, P.; Sefik, E.; Xu, H.; Harman, C.C.D.; Kim, L.K.; Lee, G.R.; Li, H.-B.; et al. mRNA destabilization by BTG1 and BTG2 maintains T cell quiescence. Science 2020, 367, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Flamand, M.N.; Wu, E.; Vashisht, A.; Jannot, G.; Keiper, B.D.; Simard, M.J.; Wohlschlegel, J.; Duchaine, T.F. Poly(A)-binding proteins are required for microRNA-mediated silencing and to promote target deadenylation in C. elegans. Nucleic Acids Res. 2016, 44, 5924–5935. [Google Scholar] [CrossRef] [PubMed]
- Fabian, M.R.; Mathonnet, G.; Sundermeier, T.; Mathys, H.; Zipprich, J.T.; Svitkin, Y.V.; Rivas, F.; Jinek, M.; Wohlschlegel, J.; Doudna, J.A.; et al. Mammalian miRNA RISC recruits CAF1 and PABP to affect PABP-dependent deadenylation. Mol. Cell 2009, 35, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Li, Q.; Xu, L.; Jiang, H. Exosomal microRNA-301a-3p promotes the proliferation and invasion of nasopharyngeal carcinoma cells by targeting BTG1 mRNA. Mol. Med. Rep. 2021, 23, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yuan, Y.; Teng, F.; Li, K.; Luo, S.; Zhang, P.; Liu, D.; Zhang, H.; Zhang, J. Obesity-induced upregulation of microRNA-183-5p promotes hepatic triglyceride accumulation by targeting the B-cell translocation gene 1. Life Sci. 2021, 268, 119011. [Google Scholar] [CrossRef]
- Sun, G.; Wang, Y.; Yang, L.; Zhang, Z.; Zhao, Y.; Shen, Z.; Han, X.; Du, X.; Jin, H.; Li, C.; et al. Rebalancing liver-infiltrating CCR3+ and CD206+ monocytes improves diet-induced NAFLD. Cell Rep. 2023, 42, 112753. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; and Lim, I.K. Regulation of Btg2(/TIS21/PC3) expression via reactive oxygen species-protein kinase C-NF kappa B pathway under stress conditions. Cell Signal. 2013, 25, 2400–2412. [Google Scholar] [CrossRef] [PubMed]
- Abdelgwad, M.; Zakaria, R.; Marzouk, S.; Sabry, D.; Ahmed, R.; Badary, H.A.; Samir, M. The Emerging Role of Circular RNA Homeodomain Interacting Protein Kinase 3 and Circular RNA 0046367 through Wnt/Beta-Catenin Pathway on the Pathogenesis of Nonalcoholic Steatohepatitis in Egyptian Patients. Rep. Biochem. Mol. Biol. 2023, 11, 614–625. [Google Scholar] [CrossRef]
- Fu, L.; Jiang, Z.; Li, T.; Hu, Y.; Guo, J. Circular RNAs in hepatocellular carcinoma: Functions and implications. Cancer Med. 2018, 7, 3101–3109. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Y.; Lin, Y.; Wei, B.; Hu, X.; Xu, L.; Zhang, W.; Lu, J. N6 -methyladenosine-modified circRNA RERE modulates osteoarthritis by regulating β-catenin ubiquitination and degradation. Cell Prolif. 2023, 56, e13297. [Google Scholar] [CrossRef]
- Rehman, S.; Varma, A.; Gupta, A.; Storey, K.B. The regulation of m6A-related proteins during whole-body freezing of the freeze-tolerant wood frog. Biochem. Cell Biol. 2023, 101, 77–86. [Google Scholar] [CrossRef]
- Qin, Y.; Li, B.; Arumugam, S.; Lu, Q.; Mankash, S.M.; Li, J.; Sun, B.; Li, J.; Flavell, R.A.; Li, H.-B.; et al. m6A mRNA methylation-directed myeloid cell activation controls progression of NAFLD and obesity. Cell Rep. 2021, 37, 109968–109968. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Zhao, Y.; He, J.; Zhang, Y.; Xi, H.; Liu, M.; Ma, J.; Wu, L. YTHDF2 destabilizes m6A-containing RNA through direct recruitment of the CCR4–NOT deadenylase complex. Nat. Commun. 2016, 7, 12626. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, B.; Jiang, Q.; Wu, R.; Cai, M.; Yao, Y.; Liu, Q.; Shi, H.; Feng, J.; Wang, Y. mRNA m6A plays opposite role in regulating UCP2 and PNPLA2 protein expression in adipocytes. Int. J. Obes. 2018, 42, 1912–1924. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wan, J.; Gao, X.; Zhang, X.; Jaffrey, SR.; Qian, SB. Dynamic m(6)A mRNA methylation directs translational control of heat shock response. Nature. 2015, 526, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, Y.; Yu, J.; Gan, Z.; Wei, W.; Wang, C.; Zhang, L.; Wang, T.; Zhong, X. Resveratrol Attenuates High-Fat Diet Induced Hepatic Lipid Homeostasis Disorder and Decreases m6A RNA Methylation. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Gebeyew, K.; Yang, C.; Mi, H.; Cheng, Y.; Zhang, T.; Hu, F.; Yan, Q.; He, Z.; Tang, S.; Tan, Z. Lipid metabolism and m6A RNA methylation are altered in lambs supplemented rumen-protected methionine and lysine in a low-protein diet. J. Anim. Sci. Biotechnol. 2022, 13, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Boo, SH.; Ha, H.; Lee, Y.; Shin, MK.; Lee, S.; Kim, YK. UPF1 promotes rapid degradation of m6A-containing RNAs. Cell Rep. 2022, 39, 110861. [Google Scholar] [CrossRef]
- Yu, H.; Yang, X.; Tang, J.; Si, S.; Zhou, Z.; Lu, J.; Han, J.; Yuan, B.; Wu, Q.; Lu, Q.; et al. ALKBH5 Inhibited Cell Proliferation and Sensitized Bladder Cancer Cells to Cisplatin by m6A-CK2α-Mediated Glycolysis. Mol. Ther. - Nucleic Acids 2020, 23, 27–41. [Google Scholar] [CrossRef]
- Bhattacharjee, J.; Borra, V.J.; Salem, E.S.B.; Zhang, C.; Murakami, K.; Gill, R.K.; Kim, A.; Kim, J.K.; Salazar-Gonzalez, R.-M.; Warren, M.; et al. Hepatic Ago2 Regulates PPARα for Oxidative Metabolism Linked to Glycemic Control in Obesity and Post Bariatric Surgery. Endocrinology 2021, 162. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Guo, J.; Wu, Y.; Yang, L.; Wang, X.; Du, J.; Dai, J.; Chen, W.; Gong, K.; Miao, S.; et al. m6A demethylase ALKBH5 inhibits tumor growth and metastasis by reducing YTHDFs-mediated YAP expression and inhibiting miR-107/LATS2-mediated YAP activity in NSCLC. Mol Cancer. 2020, 19, 40. [Google Scholar] [CrossRef] [PubMed]
- Park, OH.; Ha, H.; Lee, Y.; Boo, SH.; Kwon, DH.; Song, HK.; Kim, YK. Endoribonucleolytic Cleavage of m6A-Containing RNAs by RNase P/MRP Complex. Mol Cell. 2019, 74, 494–507e8. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Chen, B.; Liu, Z.; Cai, Y.; Wan, Y.; Zhang, G.; Fan, Y.; Zhang, Y.; Wang, F. YTHDF2 Regulates Maternal Transcriptome Degradation and Embryo Development in Goat. Front. Cell Dev. Biol. 2020, 8, 580367. [Google Scholar] [CrossRef] [PubMed]
- Khalifa, O.; Errafii, K.; Al-Akl, N.S.; Arredouani, A. Noncoding RNAs in Nonalcoholic Fatty Liver Disease: Potential Diagnosis and Prognosis Biomarkers. Dis. Markers 2020, 2020, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pirola, C.J.; Sookoian, S. Epigenetics factors in nonalcoholic fatty liver disease. Expert Rev. Gastroenterol. Hepatol. 2020, 16, 521–536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Song, J.; Liu, S.; Zhang, Y.; Qiu, T.; Jiang, L.; Bai, J.; Yao, X.; Wang, N.; Yang, G.; et al. m6A methylation-mediated PGC-1α contributes to ferroptosis via regulating GSTK1 in arsenic-induced hepatic insulin resistance. Sci. Total. Environ. 2023, 905, 167202. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Sun, J.; Zhang, Z.; Xiao, M.; Ren, D.; Liu, S.-M. Reduction of mRNA m6A associates with glucose metabolism via YTHDC1 in human and mice. Diabetes Res. Clin. Pr. 2023, 198, 110607. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dou, X.; Zheng, Z.; Ye, C.; Lu, TX.; Liang, HL.; Wang, L.; Weichselbaum, RR.; He, C. YTHDF2/m6 A/NF-κB axis controls anti-tumor immunity by regulating intratumoral Tregs. EMBO J. 2023, 42, e113126. [Google Scholar] [CrossRef]
- Yang, B.; Lu, L.; Zhou, D.; Fan, W.; Barbier-Torres, L.; Steggerda, J.; Yang, H.; Yang, X. Regulatory network and interplay of hepatokines, stellakines, myokines and adipokines in nonalcoholic fatty liver diseases and nonalcoholic steatohepatitis. Front. Endocrinol. 2022, 13, 1007944. [Google Scholar] [CrossRef]
- Ha, M.; Kim, VN. Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Li, J.; Feng, G.; Gao, S.; Wang, Y.; Zhang, S.; Liu, Y.; Ye, L.; Li, Y.; Zhang, X. MicroRNA-145 Modulates N6-Methyladenosine Levels by Targeting the 3′-Untranslated mRNA Region of the N6-Methyladenosine Binding YTH Domain Family 2 Protein. J. Biol. Chem. 2017, 292, 3614–3623. [Google Scholar] [CrossRef]
- Li, H.; Zhang, N.; Jiao, X.; Wang, C.; Sun, W.; He, Y.; Ren, G.; Huang, S.; Li, M.; Chang, Y.; et al. Downregulation of microRNA-6125 promotes colorectal cancer growth through YTHDF2-dependent recognition of N6-methyladenosine-modified GSK3β. Clin Transl Med. 2021, 11, e602. [Google Scholar] [CrossRef] [PubMed]
- Saeki, C.; Matsuzaki, J.; Kuroda, M.; Fujita, K.; Ichikawa, M.; Takizawa, S.; Takano, K.; Oikawa, T.; Nakanuma, Y.; Saruta, M.; et al. Identification of circulating microRNAs as potential biomarkers for hepatic necroinflammation in patients with autoimmune hepatitis. BMJ Open Gastroenterol. 2022, 9, e000879. [Google Scholar] [CrossRef] [PubMed]
- Jensen, A.-S.H.; Ytting, H.; Winther-Sørensen, M.; Burisch, J.; Bergquist, A.; Gluud, L.L.; Albrechtsen, N.J.W. Autoimmune liver diseases and diabetes. Eur. J. Gastroenterol. Hepatol. 2023, 35, 938–947. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhao, X.; Wang, W.; Shi, H.; Pan, Q.; Lu, Z.; Perez, S.P.; Suganthan, R.; He, C.; Bjørås, M.; et al. Ythdf2-mediated m6A mRNA clearance modulates neural development in mice. Genome Biol. 2018, 19, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Burgdorf, K.S.; Grarup, N.; Justesen, J.M.; Harder, M.N.; Witte, D.R.; Jørgensen, T.; Sandbæk, A.; Lauritzen, T.; Madsbad, S.; Hansen, T.; et al. Studies of the Association of Arg72Pro of Tumor Suppressor Protein p53 with Type 2 Diabetes in a Combined Analysis of 55,521 Europeans. PLOS ONE 2011, 6, e15813. [Google Scholar] [CrossRef] [PubMed]
- Kung, C.-P.; Leu, J.I.-J.; Basu, S.; Khaku, S.; Anokye-Danso, F.; Liu, Q.; George, D.L.; Ahima, R.S.; Murphy, M.E. The P72R Polymorphism of p53 Predisposes to Obesity and Metabolic Dysfunction. Cell Rep. 2016, 14, 2413–2425. [Google Scholar] [CrossRef]
- Paramasivam, A.; Priyadharsini, J.V. The emerging role of m6A modification in autophagy regulation and its implications in human disease. Epigenomics 2022, 14, 565–568. [Google Scholar] [CrossRef]
- Zhuang, S.; Zhou, X.; Yang, X.; Chang, D.; Chen, T.; Sun, Y.; Wang, C.; Zhang, C.; Jiang, J.; Chen, Y.; et al. Dendrobium mixture ameliorates hepatic injury induced by insulin resistance in vitro and in vivo through the downregulation of AGE/RAGE/Akt signaling pathway. Heliyon 2023, 9, e22007. [Google Scholar] [CrossRef]
- Chen, M.; Wei, L.; Law, C.; Tsang, F.H.; Shen, J.; Cheng, C.L.; Tsang, L.; Ho, D.W.; Chiu, D.K.; Lee, J.M.; et al. RNA N6-methyladenosine methyltransferase-like 3 promotes liver cancer progression through YTHDF2-dependent posttranscriptional silencing of SOCS2. J. Hepatol. 2017, 67, 2254–2270. [Google Scholar] [CrossRef]
- de Bock, C.E.; Demeyer, S.; Degryse, S.; Verbeke, D.; Sweron, B.; Gielen, O.; Vandepoel, R.; Vicente, C.; Bempt, M.V.; Dagklis, A.; et al. HOXA9 Cooperates with Activated JAK/STAT Signaling to Drive Leukemia Development. Cancer Discov. 2018, 8, 616–631. [Google Scholar] [CrossRef] [PubMed]
- Aziz, F. The emerging role of miR-223 as novel potential diagnostic and therapeutic target for inflammatory disorders. Cell. Immunol. 2016, 303, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Huang, S.; Zhuang, H.; Ruan, S.; Zhou, Z.; Huang, K.; Ji, F.; Ma, Z.; Hou, B.; He, X. YTHDF2 promotes the liver cancer stem cell phenotype and cancer metastasis by regulating OCT4 expression via m6A RNA methylation. Oncogene 2020, 39, 4507–4518. [Google Scholar] [CrossRef]
- Liu, F.; Cai, Z.; Yang, Y.; Plasko, G.; Zhao, P.; Wu, X.; Tang, C.; Li, D.; Li, T.; Hu, S.; et al. The adipocyte-enriched secretory protein tetranectin exacerbates type 2 diabetes by inhibiting insulin secretion from β cells. Sci Adv. 2022, 8, eabq1799. [Google Scholar] [CrossRef] [PubMed]
- Morishita, A.; Oura, K.; Tadokoro, T.; Fujita, K.; Tani, J.; Kobara, H.; Ono, M.; Himoto, T.; Masaki, T. MicroRNAs and Nonalcoholic Steatohepatitis: A Review. Int. J. Mol. Sci. 2023, 24, 14482. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, L.; Li, M.; Zhang, Y.; Sun, M.; Wang, L.; Lin, J.; Cui, Y.; Chen, Q.; Jin, C.; et al. Fusobacterium nucleatum reduces METTL3-mediated m6A modification and contributes to colorectal cancer metastasis. Nat. Commun. 2022, 13, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Fardi, F.; Khasraghi, L.B.; Shahbakhti, N.; Naseriyan, A.S.; Najafi, S.; Sanaaee, S.; Alipourfard, I.; Zamany, M.; Karamipour, S.; Jahani, M.; et al. An interplay between non-coding RNAs and gut microbiota in human health. Diabetes Res. Clin. Pr. 2023, 201, 110739. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Huang, J.; Li, X.; Xing, J.; Chen, Q.; Liu, R.; Hua, F.; Qiu, Z.; Song, Y.; Bai, C.; et al. Gut microbiota regulate tumor metastasis via circRNA/miRNA networks. Gut Microbes 2020, 12. [Google Scholar] [CrossRef]
- Marra, F.; Svegliati-Baroni, G. Lipotoxicity and the gut-liver axis in NASH pathogenesis. J. Hepatol. 2018, 68, 280–295. [Google Scholar] [CrossRef]
- Makri, E.; Goulas, A.; Polyzos, S.A. Epidemiology, Pathogenesis, Diagnosis and Emerging Treatment of Nonalcoholic Fatty Liver Disease. Arch. Med Res. 2020, 52, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Gadecka, A.; Bielak-Zmijewska, A. Slowing Down Ageing: The Role of Nutrients and Microbiota in Modulation of the Epigenome. Nutrients 2019, 11, 1251. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; Vétizou, M.; Daillère, R.; Roberti, M.P.; Yamazaki, T.; Routy, B.; Lepage, P.; Boneca, I.G.; Chamaillard, M.; Kroemer, G.; et al. Resistance Mechanisms to Immune-Checkpoint Blockade in Cancer: Tumor-Intrinsic and -Extrinsic Factors. Immunity 2016, 44, 1255–1269. [Google Scholar] [CrossRef]
- Blaser, M.J. Antibiotic use and its consequences for the normal microbiome. Science 2016, 352, 544–545. [Google Scholar] [CrossRef] [PubMed]
- Vesely, M.D.; Kershaw, M.H.; Schreiber, R.D.; Smyth, M.J. Natural Innate and Adaptive Immunity to Cancer. Annu. Rev. Immunol. 2011, 29, 235–271. [Google Scholar] [CrossRef]
- Chen, Y.G.; Kim, M.V.; Chen, X.; Batista, P.J.; Aoyama, S.; Wilusz, J.E.; Iwasaki, A.; Chang, H.Y. Sensing Self and Foreign Circular RNAs by Intron Identity. Mol. Cell 2017, 67, 228–238. [Google Scholar] [CrossRef]
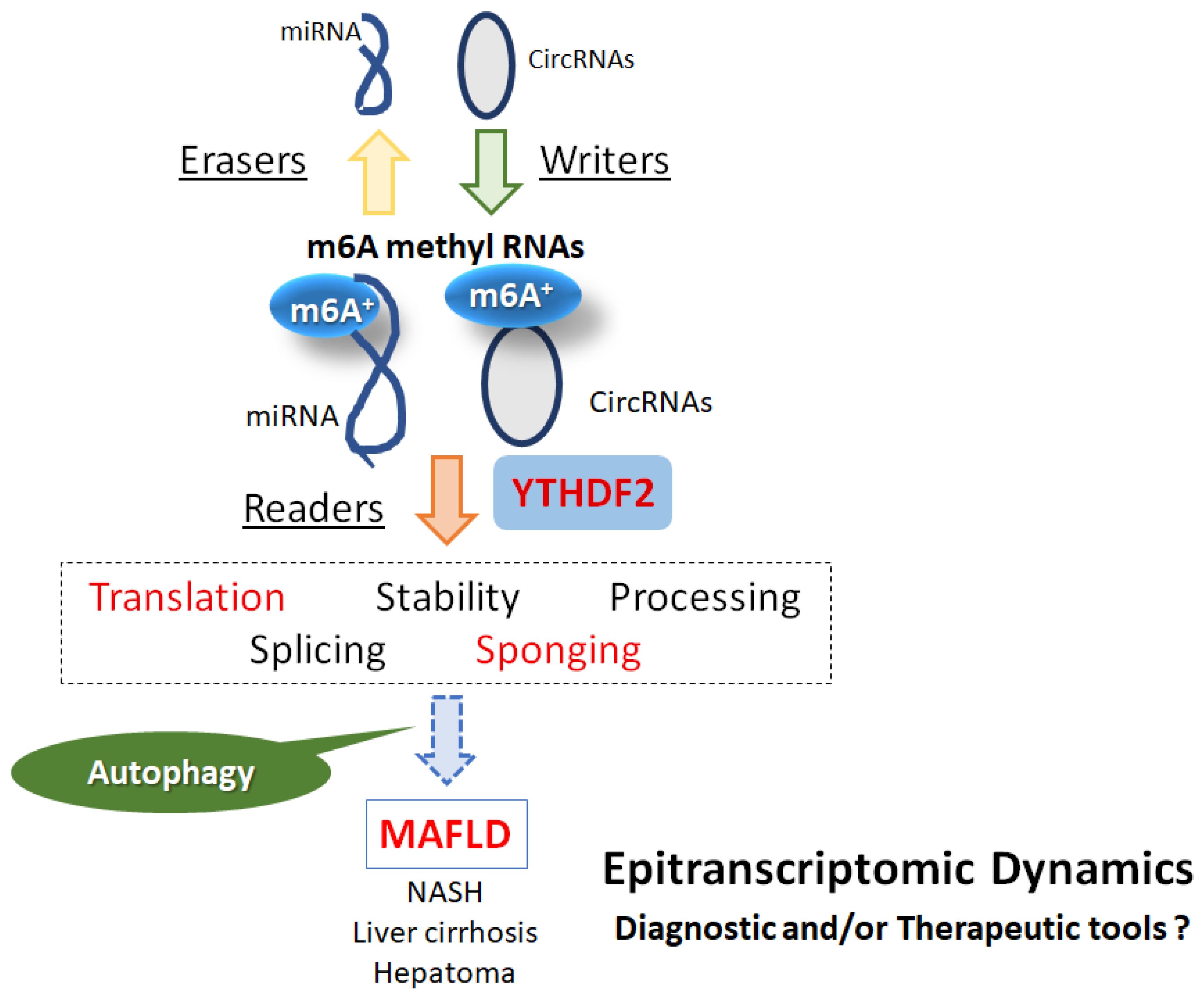
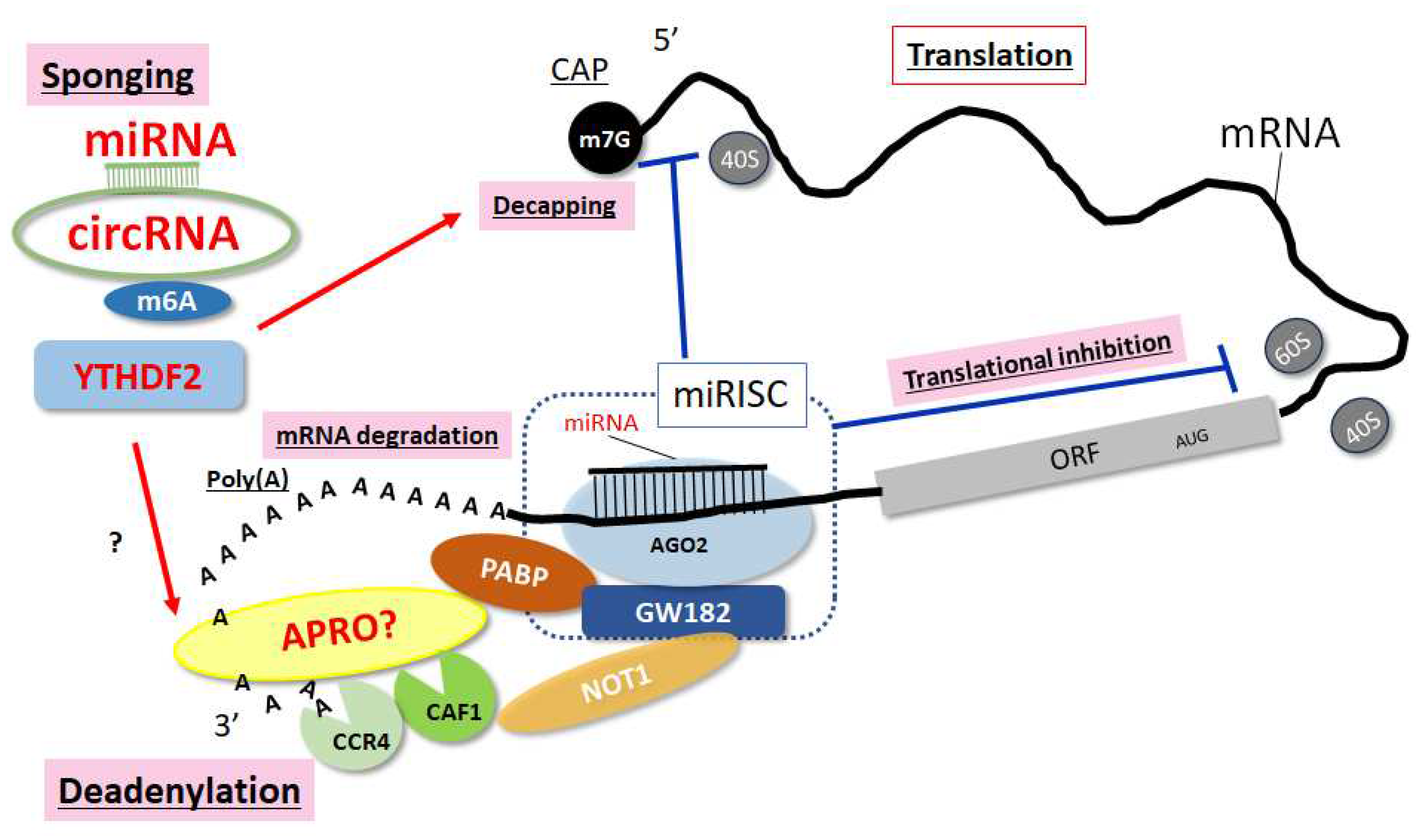
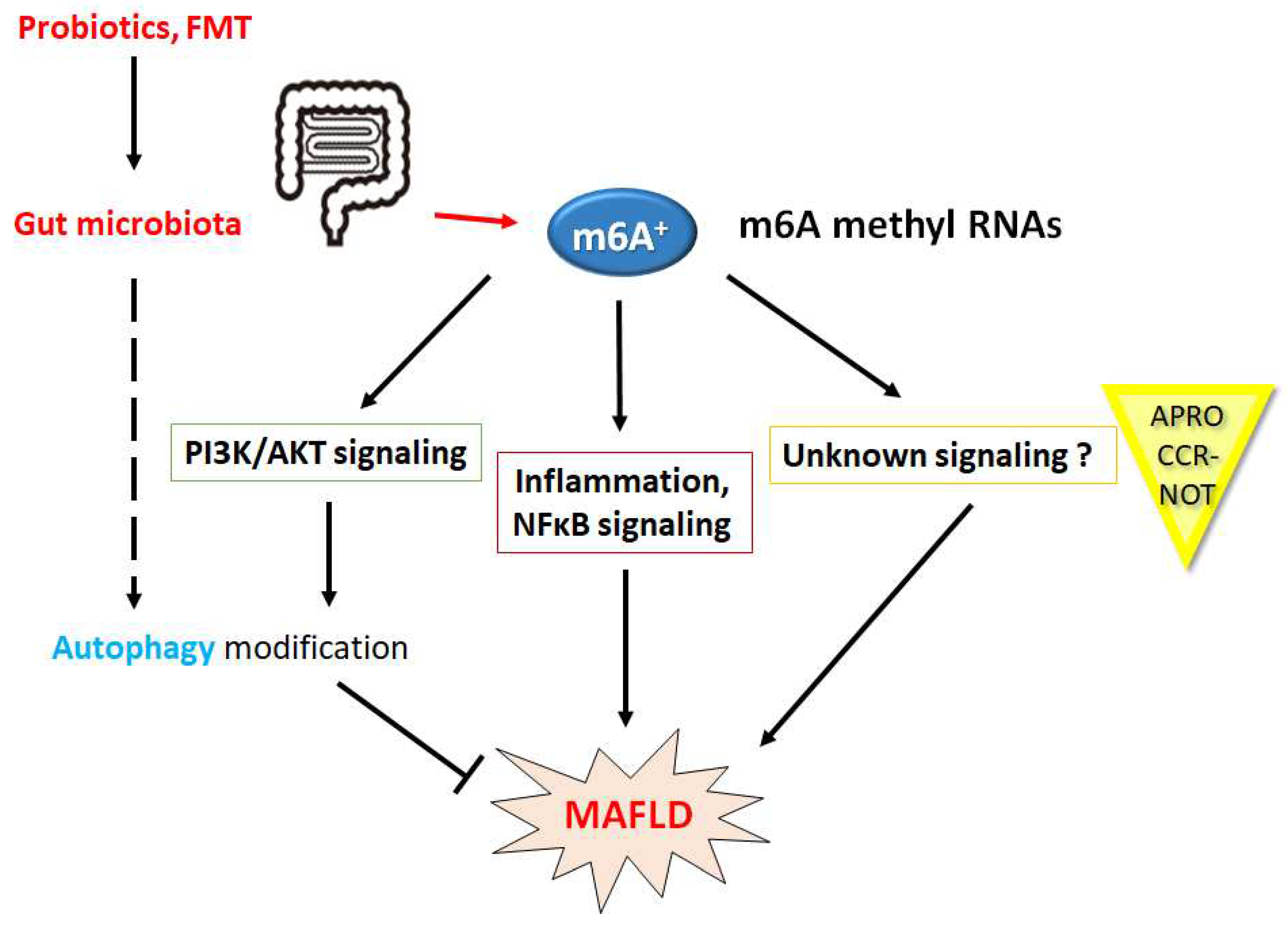
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




