1. Introduction
Burning mouth syndrome (BMS) is defined in the International Classification of Orofacial Pain, 2020 as “idiopathic orofacial pain with intraoral burning or dysesthesia recurring daily for more than 2 hours per day and more than 3 months, without any identifiable causative lesions, with or without somatosensory changes” [
1,
2]. The development of research diagnostic criteria for BMS was published in 2021 [
3]. Previously, it was divided into primary and secondary [
3]. Intraoral burning is a result of a range of underlying causes of lesions, such as candida infection, oral lichen planus, hyposalivation, contact mucosal reactivity, medications, anemia, deficiencies in vitamin B12 or folic acid, Sjögren's syndrome, diabetes, and hypothyroidism [
3]. However, the correct diagnosis of BMS is a diagnosis of exclusion, and only primary cases are diagnosed as true BMS. Tests have false positives and false negatives. Even if the algorithm is followed, misclassification bias is inevitable. In our hospital, it is common to diagnose BMS by excluding other diseases whenever possible.
There are various reports on effective ways to treat BMS patients [
4,
5,
6,
7,
8,
9]. One of the treatment options for BMS are topical and systemic application of clonazepam [
4,
5,
6,
7,
8,
9]. For another treatment, there are a variety of options available such as systemic the selective serotonin reuptake inhibitors (SSRIs), zinc replacement therapy, alpha-lipoic acid, and aloe vera, hormone replacement therapy, cognitive behavior therapy, and acupuncture [
5,
6]. However, some suggest that cautious use of clonazepam and alpha-lipoid acid be used in the treatment of burning mouth syndrome [
8,
9,
10]. Y Cui et al. conducted a meta-analysis of clonazepam, but the heterogeneity was large [
7], making it undesirable to pool the results. Therefore, the current treatment management is debatable [
7,
8,
9,
10]. There is the fact that the management of BMS is still not efficacious with the traditional management options [
8,
9,
10]. There are many studies on quantitative assessment, based on the amount of change in visual analogue scale (VAS), but there are no reports of high evidence for the effectiveness [
7,
8,
9].
One of our previous studies evaluated quantitatively, which evaluate the change of the pain intensity using numerical rating scale (NRS) [
11]. After using rikkosan for one month, the average change in NRS was an approximately 2-point reduction in pain intensity [
11]. The other our previous studies evaluated qualitatively, which evaluate the improvement rate defined as reduced subjective VAS to <50% of baseline before rikkosan treatment [
12]. The sample sizes for each study were 32 and 48 people [
11,
12]. Rikkosan is a traditional Japanese (kampo) medicine used to control oral pain [
12,
13]. Rikkosan has anti-inflammatory effects, but the exact mechanism is still unknown [
12,
13]. The surface anesthetic action of saishin, one of the components of rikkosan, may reduce the pain [
12,
13]. In addition, rikkosan contains two ingredients with analgesic effect (ryutan: glycyrrhiza and kanzou: Japanese gentian) [
12]. The sedative action of shoma and kanzou might reduce pain [
13].
A minimum of 3-point NRS reduction is usually considered clinically meaningful in the pain field. A change of 3-point NRS reduction was statistically a large difference, and the difference can be detected with a small number of cases tested. However, if there are fewer than 20 people, the sampling becomes more biased, increasing the bias. The study design had a limit of 2-point NRS reduction. Therefore, the purpose of this study was to confirm the hypothesis that rikkosan leads to a 2-point decrease in pain intensity. The working hypothesis was based on our previous preliminary research [
11].
2. Materials and Methods
Patients Selection
Patients were examined and diagnosed in our department according to the criteria of The International Classification of Orofacial Pain, 1st edition (ICOP) [
2,
3]. Patients were considered to have secondary BMS if systemic and psychosocial factors associated with secondary BMS and structural disorder in the oral cavity were found during examination. Patients with residual symptoms after antifungal therapy and replacement therapy for deficiency factors such as trace metals and vitamin B12 were diagnosed as BMS when blood tests were normal. In Japan, oral medicine has been established as a subspecialty of oral surgery. At our facility, multiple oral medicine and oral surgeons specialists with over 10 years of experience and accredited by the Japanese Society of Oral and Maxillofacial Surgeons make final diagnoses and provide treatment.
The patients visited at the Department of Oral Medicine, Hokkaido University Hospital between August 2019 and March 2023. Patients with an underlying medical condition, based on an interview, or any abnormalities found through our diagnostic algorithm were diagnosed secondary BMS. The classification of secondary BMS details was based on the one proposed by Currie et al. [
3] Twenty female primary BMS patients who had clear medical records of their treatment were enrolled in this study. All of them was diagnosed by the multiple oral surgeon specialists with over 10 years of experience and accredited by the Japanese Society of Oral and Maxillofacial Surgeons (S.K.-i. and J.S.).
Procedure
The patients were diagnosed primary BMS and initially treated with single agent rikkosan (2.5 g rikkosan [Tsumura, Tokyo, Japan]) three times daily (7.5 g/day). After drug therapy, the patients’ compliance in taking the medication was confirmed. There were no changes in the medication status of other drugs during treatment with rikkosan.
Study Variables
Various factors, such as patient characteristics (age, sex) and clinical parameters (dosing period, treatment outcome, and side effects), were retrospectively examined.
The patients were 43–80 (63.3 ± 13.4) years old and the median duration of pain complaints was 14 months (range: 3 months to more than 10 years). 90% of them were undertaking other medications for other diseases. The medication status was the same during this study period. In other words, rikkosan was just added to the medication. They were treated approximately 4 weeks (29.5 ± 6.5 days) for the initial treatment.
The effectiveness of the treatment was assessed by referring to changes in NRS or VAS/10 scores. NRS or VAS/10 scores were evaluated by asking patients to assess the degree of pain they were currently experiencing, with 0 being no pain and 10 or 100 being the worst possible pain. NRS or VAS/10 scores were measured at the time of the initiation of rikkosan and at 1 month after.
Statistical Analysis
Statistical analyses were performed using Excel (Microsoft
® Excel
® for Microsoft 365MSO(version 2306 build 16.0.16529.20164, 64 bit) and R version 4.0.3 (2020-10-10) (Copyright © 2020, The R Foundation for Statistical Computing). The effectiveness of the pain evaluation index was evaluated based on the presence or absence of ceiling and floor effects. The ceiling effect was determined when the mean plus exceeded the maximum value of the measurement, and the floor effect was determined when the mean minus one standard deviation (SD) exceeded the minimum value of the measurement. Based on our previous exploratory research [
11], the average effect of rikkosan was estimated to be a two-level reduction in NRS, and the sample size was designed with a standard deviation of 2.6, alpha error of 5%, and power of 90%. The calculated sample size was 20 people. The 20 selected patients were the 20 patients with the lowest patient identification number at our hospital who met this study criteria. A
paired t test was performed to assess significant differences in mean NRS scores at approximately a one-month interval. As a secondary analysis, changes in pain intensity were evaluated using
Kendall's coefficient of concordance and
Friedman's test was performed to compare the center of grade distribution before and after administration.
IRB Approval and Ethics
This retrospective study was conducted with the approval of the Hokkaido University Hospital Independent Clinical Research Review Committee (Approval No. 023-0331). All the study procedures were performed in accordance with the principles of the Declaration of Helsinki.
3. Results
As summarized in
Table 1, the ceiling effect and the floor effect were not observed. Therefore, the assessment of pain worked well.
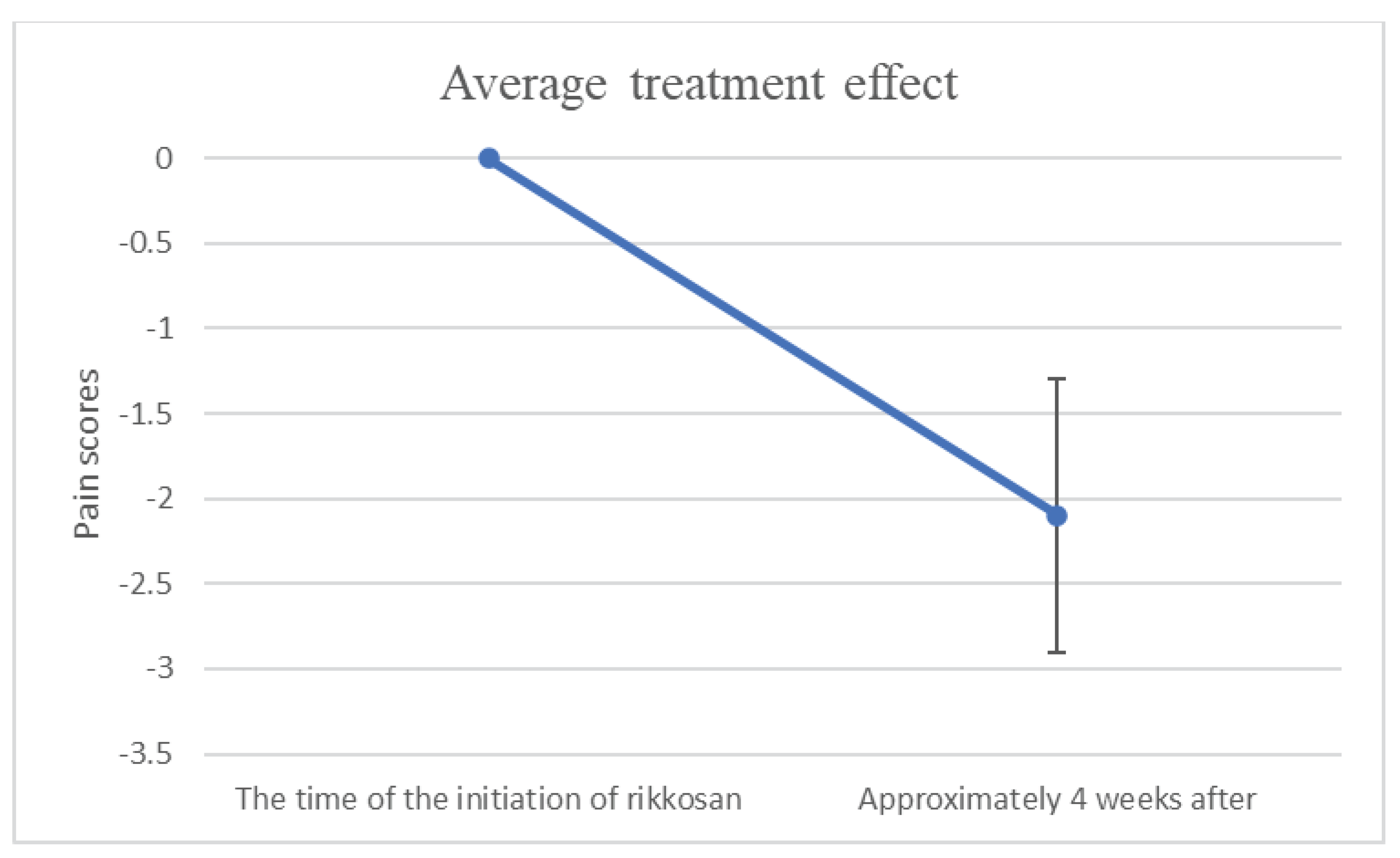
Figure 1 showed that changes in NRS or VAS/10 scores between the time of the initiation of rikkosan and one month after.
As summarized in
Table 2, the changes in pain scores for average treatment effect treated with rikkosan in 4 weeks and qualitative assessment of the change. A significant difference was observed in pain evaluation before and after treatment (
p < 0.05). Kendall's coefficient of concordance was 0.85. As summarized in
Table 2, 85% improvement rate was achievable (
p = 3.7 × 10
-5) if a slight improvement is set as a cutoff. However, if the cutoff is an improvement of 2 or more, it was 50% in this study.
4. Discussion
This is the first study to assess the working hypothesis which rikkosan leads to a 2-point reduction of pain in patients with BMS. Rikkosan treatment showed two-level reduction in NRS or VAS/10 scores for BMS. This result indicated that rikkosan could be a first good therapeutic option for BMS in the short term. Our results statistically showed rikkosan produced a 2-point reduction in pain intensity. The response rate may be used to indicate the therapeutic effect. However, this does not depend on how effective they are individually, but rather they are the same if they are effective above a certain level. When the improvement rate is used as an evaluation index, there is a problem with the cutoff value. On the other hand, this study performed quantitative evaluation using NRS or VAS. Consequently, the average treatment effect could be predicted. Due to the study design, 90% reproducibility is expected under similar conditions. The statistics regarding the change in NRS were consistent with the results of our previous study [
11].
There are three pain classifications for chronic pain [
14]. There is a theory that BMS is neuropathic pain, but the evidence for this is unreliable [
15]. The reason for this is that the average age of the control and BMS patient groups differed by 22 years, and age-related neurodegeneration was not considered [
15]. If BMS is neuropathic pain, treatments for neuropathic pain should be effective, but there are almost no clinical trials. Nociceptive pain often involves obvious damage, which is easy to see on the body surface. BMS does not show obvious damage, so it is unlikely to be nociceptive pain, but the possibility of nociplastic pain that develops afterwards cannot be ruled out. BMS is probably nociplastic pain. There is no established treatment for nociplastic pain. The results of this study suggest that rikkosan is a candidate drug for the treatment of nociplastic pain or BMS.
Traditional Chinese medicine developed the use of medicinal plants [
16]. Traditional Japanese medicine, also called Kampo medicine, derives from traditional Chinese medicine [
16]. In recent years, kampo medicine has been attracting attention in the West as well [
16]. Rikkosan contains licorice. Licorice contains glycyrrhizic acid, which is hydrolyzed to glycyrrhetinic acid (GRA) in the intestine [
16]. Therefore, a side effect of rikkosan is pseudoaldosteronism, so regular blood tests are required. Electropharmacological profile of licorice can be explained by Na
+, Ca
2+ and K
+ channels blockade. Ion channels exist in pain receptors [
17]. These ion channels may be one of the targets of rikkosan.
This study has several limitations. This study was a single-arm study. A single-arm study is prone to bias and false positives, so a comparative study with a control group is preferable. An important limitation was the short-term follow-up. Rikkosan has a bitter taste, so some people don't like it. Taste preferences may be used to determine responders. Some patients did not want further treatment, but others still required treatment. The duration of treatment effects may vary depending on the patients. Therefore, further research on additional treatments is needed. However, careful consideration should be given to evaluating the quantitative effect of rikkosan before conducting the next trial.
5. Conclusions
This study quantitatively evaluated the effects of rikkosan, including some bias. The results of our study suggested with statistical reproducibility that rikkosan can reduce two-level in pain intensity of patients with BMS.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Table S1: Raw data. Excel File: Our calculation processes.
Author Contributions
Conceptualization, T.I., K.N. and K.-i.S.; methodology, T.I. and K.N.; software, T.I.; validation, T.I., J.S., and Y.K.; formal analysis, T.I. and T.S.; investigation, T.I. and T.T.; resources, T.I.; data curation, T.I.; writing—original draft preparation, T.I. and K.N.; writing—review and editing, T.I., K.-i.S., J.S. and Y.K.; visualization, T.I. and Y.N.; supervision, K.-i.S., and Y.K.; project administration, T.T. and K.-i.S.; funding acquisition, K.-i.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This retrospective study was conducted with the approval of the Hokkaido University Hospital Independent Clinical Research Review Committee (Approval No. 023-0331). All the study procedures were performed in accordance with the principles of the Declaration of Helsinki.
Informed Consent Statement
This article does not disclose identifiable information of any of the participants in any form. Hence, consent for publication is not applicable in this case.
Data Availability Statement
Acknowledgments
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Russo, M.; Crafa, P.; Guglielmetti, S.; Franzoni, L.; Fiore, W.; Di Mario, F. Burning Mouth Syndrome Etiology: A Narrative Review. J Gastrointestin Liver Dis 2022, 31, 223–228. [Google Scholar] [CrossRef] [PubMed]
- International Classification of Orofacial Pain, 1st edition (ICOP). Cephalalgia 2020, 40, 129–221. [CrossRef] [PubMed]
- Currie, C.C.; Ohrbach, R.; De Leeuw, R.; Forssell, H.; Imamura, Y.; Jääskeläinen, S.K.; Koutris, M.; Nasri-Heir, C.; Huann, T.; Renton, T.; et al. Developing a research diagnostic criteria for burning mouth syndrome: Results from an international Delphi process. J Oral Rehabil 2021, 48, 308–331. [Google Scholar] [CrossRef] [PubMed]
- Amos, K.; Yeoh, S.C.; Farah, C.S. Combined topical and systemic clonazepam therapy for the management of burning mouth syndrome: a retrospective pilot study. J Orofac Pain 2011, 25, 125–130. [Google Scholar] [PubMed]
- Imamura, Y.; Okada-Ogawa, A.; Noma, N.; Shinozaki, T.; Watanabe, K.; Kohashi, R.; Shinoda, M.; Wada, A.; Abe, O.; Iwata, K. A perspective from experimental studies of burning mouth syndrome. Journal of Oral Science 2020, 62, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Felipe, C.; Tvarijonaviciute, A.; López-Arjona, M.; Pardo-Marin, L.; Pons-Fuster, E.; López-Jornet, P. Response to Treatment with Melatonin and Clonazepam versus Placebo in Patients with Burning Mouth Syndrome. Journal of Clinical Medicine 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Xu, H.; Chen, F.M.; Liu, J.L.; Jiang, L.; Zhou, Y.; Chen, Q.M. Efficacy evaluation of clonazepam for symptom remission in burning mouth syndrome: a meta-analysis. Oral Dis 2016, 22, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Farag, A.M.; Kuten-Shorrer, M.; Natto, Z.; Ariyawardana, A.; Mejia, L.M.; Albuquerque, R.; Carey, B.; Chmieliauskaite, M.; Miller, C.S.; Ingram, M.; et al. WWOM VII: Effectiveness of systemic pharmacotherapeutic interventions in the management of BMS: A systematic review and meta-analysis. Oral Diseases 2023, 29, 343–368. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, S.; Carey, B.; Farag, A.M.; Kuten-Shorrer, M.; Natto, Z.S.; Ariyawardana, A.; Mejia, L.M.; Chmieliauskaite, M.; Miller, C.S.; Ingram, M.; et al. WWOM VII: Effectiveness of topical interventions in the management of burning mouth syndrome: A systematic review. Oral Diseases 2023, 29, 3016–3033. [Google Scholar] [CrossRef] [PubMed]
- Suga, T.; Tu, T.T.H.; Nagamine, T.; Toyofuku, A. Careful use of clonazepam and alpha lipoid acid in burning mouth syndrome treatment. In Oral Dis; Denmark, 2022; Volume 28, pp. 846–847. [Google Scholar] [CrossRef]
- Hato, H.; Sakata, K.-i.; Sato, J.; Asaka, T.; Ohga, N.; Yamazaki, Y.; Kitagawa, Y. Efficacy of rikkosan for primary burning mouth syndrome: a retrospective study. BioPsychoSocial Medicine 2021, 15, 19. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, S.; Okada, K.; Matsushita, T.; Hegozaki, S.; Sakata, K.-i.; Kitagawa, Y.; Yamazaki, Y. Effectiveness of rikkosan gargling for burning mouth syndrome. Traditional & Kampo Medicine 2017, 4, 121–123. [Google Scholar] [CrossRef]
- Sakata, K.-i.; Yamazaki, Y.; Ohga, N.; Sato, J.; Asaka, T.; Yoshikawa, K.; Nakazawa, S.; Sato, C.; Nakamura, Y.; Kitagawa, Y. Clinical efficacy of a traditional Japanese (kampo) medicine for burning mouth syndrome. Traditional & Kampo Medicine 2016, 3, 120–122. [Google Scholar] [CrossRef]
- Nijs, J.; Lahousse, A.; Kapreli, E.; Bilika, P.; Saraçoğlu, İ.; Malfliet, A.; Coppieters, I.; De Baets, L.; Leysen, L.; Roose, E.; et al. Nociplastic Pain Criteria or Recognition of Central Sensitization? Pain Phenotyping in the Past, Present and Future. Journal of Clinical Medicine 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, Z.; Renton, T.; Yiangou, Y.; Zakrzewska, J.; Chessell, I.P.; Bountra, C.; Anand, P. Burning mouth syndrome as a trigeminal small fibre neuropathy: Increased heat and capsaicin receptor TRPV1 in nerve fibres correlates with pain score. J Clin Neurosci 2007, 14, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Veilleux, M.-P.; Moriyama, S.; Yoshioka, M.; Hinode, D.; Grenier, D. A Review of Evidence for a Therapeutic Application of Traditional Japanese Kampo Medicine for Oral Diseases/Disorders. Medicines 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Izumi-Nakaseko, H.; Chiba, K.; Goto, A.; Kambayashi, R.; Matsumoto, A.; Takei, Y.; Kawai, S.; Sugiyama, A. Electropharmacological Characterization of Licorice Using the Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes Sheets and the Chronic Atrioventricular Block Dogs. Cardiovascular Toxicology 2023, 23, 207–217. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).