1. Introduction
1.1. Brief overview of Type 1 Diabetes (T1D)
Type 1 diabetes (T1D) is a chronic autoimmune disorder characterized by the destruction of pancreatic beta cells in the pancreas responsible for insulin production, a hormone crucial for glucose catabolism [
1]. Over the past two decades, the prevalence of T1D has steadily increased, with an estimated 8.75 million people with T1D in 2022 [
2]; of these, there were 530000 incident cases of T1D in 2022; 35000, on the other hand, is the estimated number of patients under 25 who died due to missed diagnosis of T1D [
2]. This is of particular concern because patients with T1D have a life expectancy on average 11 years shorter than non-diabetic patients, mainly due to the macroangiopathic complications of T1D [
3,
4,
5].
1.2. Importance of understanding T1D and its microangiopathic complications
Although the use of intensive insulin therapy since the DCCT trial has greatly reduced the incidence of micro- and macroangiopathic complications of diabetes [
6], in these patients a reduced life expectancy and an increased prevalence of micro- and macroangiopathic complications remain even when glycemic compensation (estimated with Hba1c) appears optimal [
3,
4,
7]. Chronic glycemic alteration, in addition to the frequent long disease duration in patients with T1D, can also lead to microangiopathic complications, including neuropathy, nephropathy, and diabetic retinopathy (DR) [
8]. DR is a leading cause of blindness in adults, and diabetic nephropathy is a leading cause of end-stage renal disease [
9,
10]. In 2004, the direct costs associated with DR were estimated to be
$493 million per year, although it should be noted that this figure was before the introduction of antiangiogenic drugs, which also have a significant impact on the cost of this disease [
11]. In the meantime the prevalence of macular edema is projected to increase by 25 percent over the next 10 years, placing an even greater economic burden on international systems [
12]. In this context, it is essential to better understand the etiopathogenesis of this disease to improve its prevention. The high prevalence of DR and diabetic nephropathy in patients with T1D cannot be explained solely by the role of hyperglycemia: instead, multiple studies point to a multifactorial etiopathogenesis, which frequently leads to the association between retinopathy and diabetic nephropathy, so much so that the term "renal-retinal syndrome" [
13,
14], in which microvascular endothelial dysfunction is a common pathogenetic factor in the early development of the two diseases: the endothelial cells of the microcirculation are the main target of hyperglycemia-related damage through a series of pathways including the accumulation of Advanced glycation end products (AGEs) and the increase of receptors for them, activation of the polyol pathway, stimulation of PKC subtypes and aminohexose signal pathway [13, 14]. This review will mainly focus on the role of glycemic alterations in the etiopathogenesis of DR and how new models of glycemic management may play a role in the progression and prognosis of this disease.
2. Diabetic Retinopathy: Clinical Features and Stages
2.1. Non-proliferative diabetic retinopathy (NPDR) and Proliferative diabetic retinopathy (PDR)
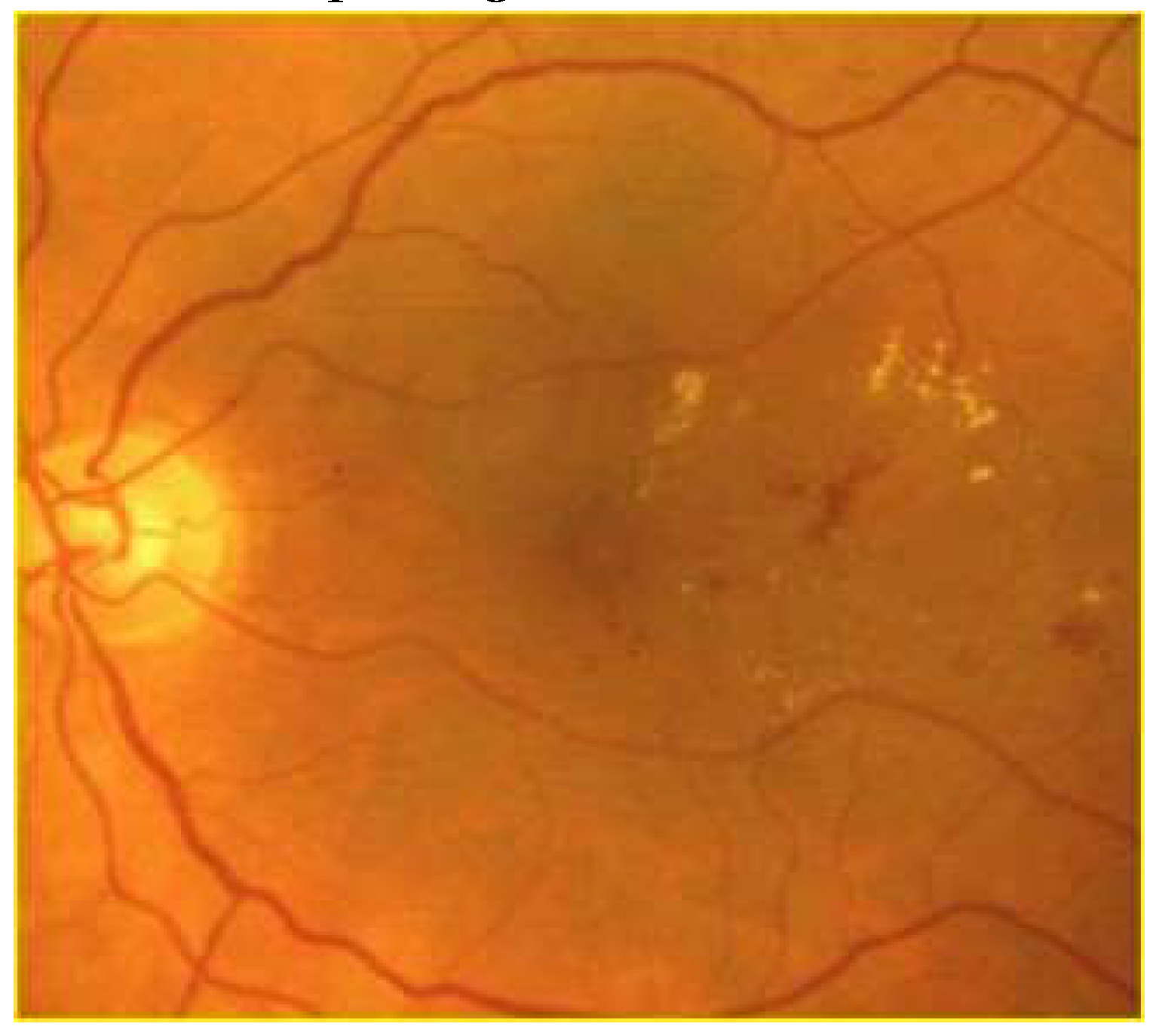
Clinically, DR is categorized into two distinct stages. The initial stage, known as non-proliferative DR (NPDR), is characterized by increased vascular permeability and the occlusion of retinal capillaries (
Figure 1). During funduscopic examinations in this stage, microaneurysms, hemorrhages, and hard exudates may be observed, though patients typically do not experience symptoms at this point [
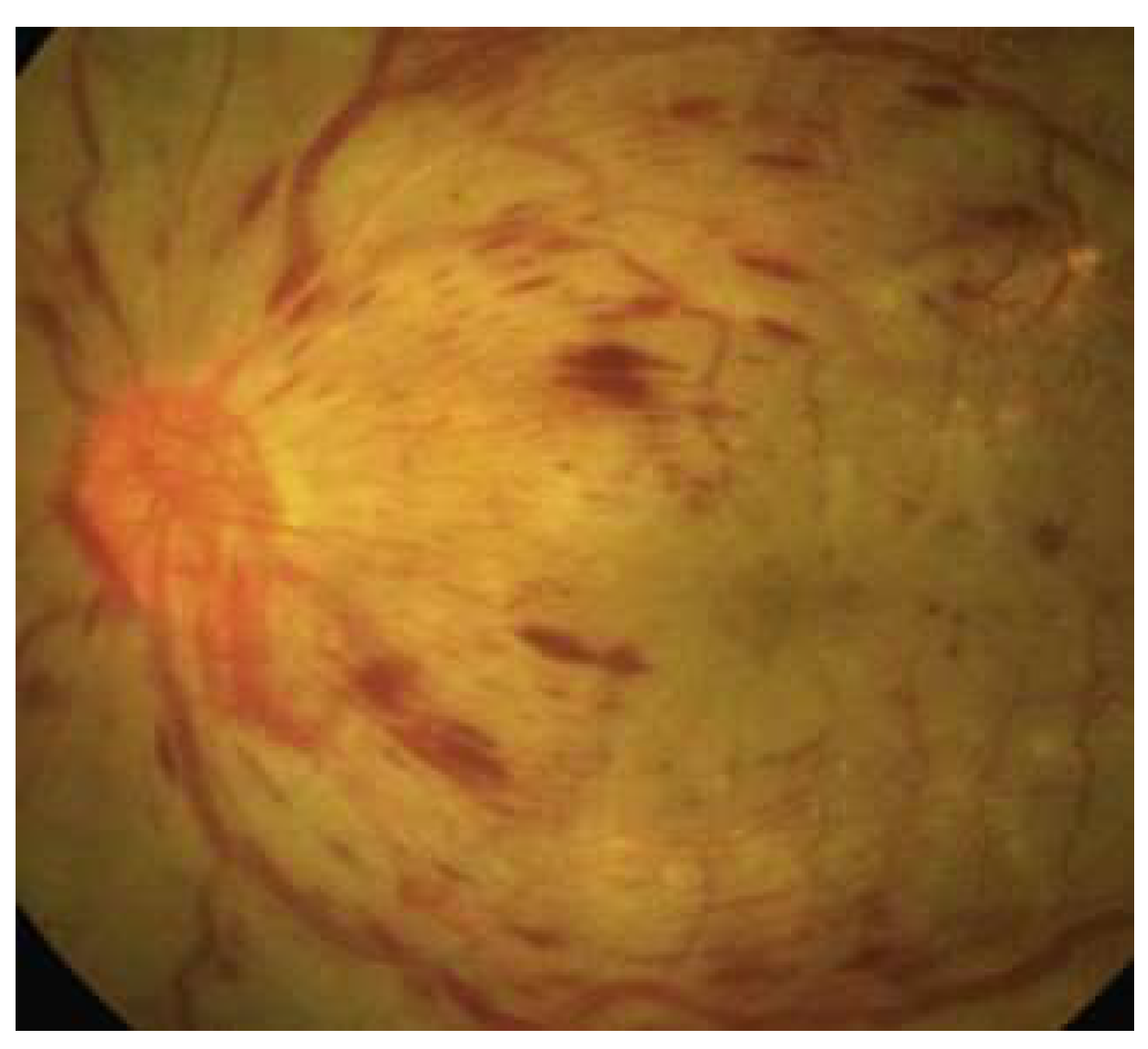
15]. Progressing to the more advanced stage, proliferative DR (PDR) is marked by a critical phenomenon: neovascularization (
Figure 2). This stage is a significant escalation in severity, as the growth of abnormal new blood vessels can lead to severe visual impairment. These vessels, forming in the vitreous or on the retina, are prone to causing complications such as vitreous hemorrhage or even retinal detachment, posing a serious threat to vision [
16].
2.2. Diabetic macular edema (DME)
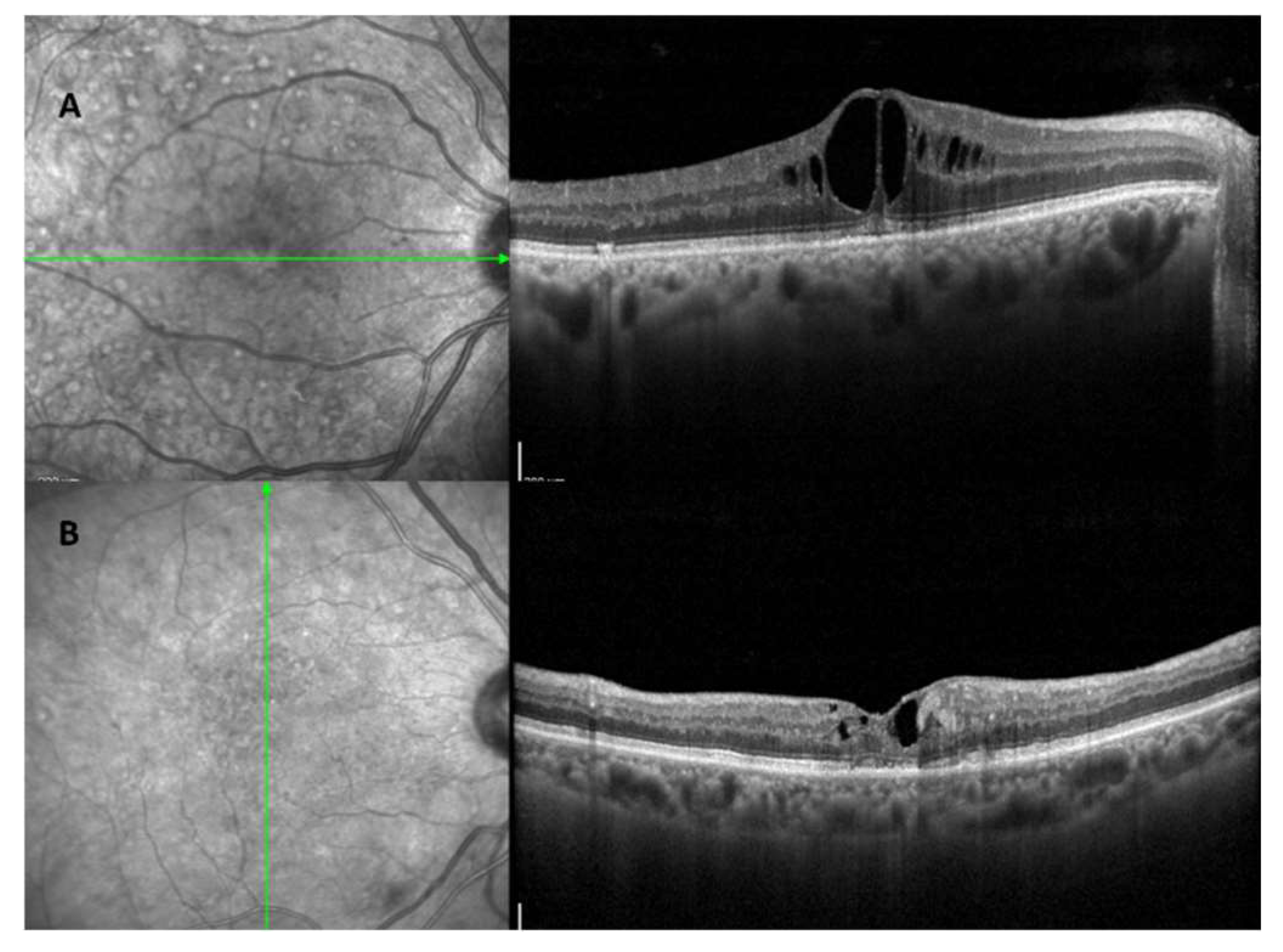
Diabetic macular edema (DME) is the primary cause of vision loss in diabetic retinopathy (DR)[
17]. It involves the macula swelling and thickening due to fluid buildup, which is linked to blood-retinal barrier (BRB) dysfunction [
17] (
Figure 3). The Early Treatment Diabetic Retinopathy Study (ETDRS) describes DME as either retinal thickening or the presence of hard exudates near the center of the macula [
18]. DME can develop at any DR stage, leading to decreased visual acuity or visual distortion. It's especially prevalent in working-age people with type 2 diabetes. Globally, DME affects about 6.8% of individuals [
19], with its prevalence increasing to about 30% in those who have had diabetes for over 20 years, and up to 71% in those with proliferative DR [
20].
2.3. Role of dysglycemia in the pathogenesis of Diabetic Retinopathy
The pathogenesis of DR is complex and multifactorial, with the hyperglycemic state associated with diabetes playing a pivotal role in inducing microangiopathy. Hyperglycemia activates different metabolic pathways leading to increased hypoxia, reactive oxygen species formation, and inflammation [
21,
22,
23] . Hyperglycemia leads to altered retinal vascular flow and dilation, probably adapted to compensate for the metabolic deficit in the diabetic patient [
24]. At the same time, hyperglycemia is responsible for pericyte apoptosis, both in vitro and in vivo [
25] . The lack of structural support to the capillaries provided by pericytes results in the process of aneurysmal dilatation of the capillaries, one of the first clinical signs of DR [
26]. At the same time, hyperglycemia is associated with endothelial cell apoptosis and basement membrane thickening, leading to BRB dysfunction and subsequent retinal ischemia secondary to capillary occlusion [
27]. Ischemia is followed by upregulation of VEGF through activation of hypoxia-inducible factor 1 [
28]. VEGF promotes increased vascular permeability and endothelial cell proliferation, and its elevated levels have been documented in the retina of diabetic rats and in diabetic patients with DME and PDR [
29,
30]. In vitro studies have shown that hyperglycemia also plays a role in the induction of mitochondrial dysfunction leading to apoptosis of retinal neurons [
31]. Hypoglycemia and glycemic variability are also thought to play a role in promoting the development of DR [
32,
33,
34], with increasing support for the idea that it is not so much necessary to only lower blood glucose levels as to bring them as close as possible to those of a euglycemic patient but to increase the general time spent in euglycemia by the patient, taking also into account the time spent in hypoglicemia and the glycemic variance [
33,
34]. The use of artificial pancreas systems has been shown to reduce the prevalence and severity of hypoglycemia and glycemic variance [
35,
36,
37] and is a potential tool for improving overall prognosis in diabetes and specifically in DR.
2.4. Role of inflammation and retinal degeneration in the pathogenesis of Diabetic Retinopathy
Inflammation significantly contributes to the development and continuation of DR [
23]. It occurs when retinal cells, like pigment epithelium cells, Müller cells, and activated microglia, release inflammatory substances such as cytokines, chemokines, and growth factors. These substances disrupt the connections between endothelial cells, cause the loss of pericytes (cells that support capillaries), and lead to leukostasis (a slowing or stopping of blood flow), all of which affect the blood-retinal barrier and increase vascular permeability. This process is driven by an elevated production of the vascular endothelial growth factor (VEGF), culminating in the development of DME [21, 22,
38]. Another key factor in the progression of DR is retinal neurodegeneration, which occurs from the earliest stages of the disease [
39]. As early as one month after induction of diabetes in streptozotocin-treated rats, apoptosis of retinal neurons can be observed [
39], and increased levels of pro-apoptotic molecules and increased production of reactive oxygen species (ROS), which are involved in retinal neuronal apoptosis, have been demonstrated [
40]. Although retinal neurodegeneration is associated with DR, recent evidence suggests that in diabetic patients it precedes the typical changes observed at the ocular fundus in the retinal microcirculation [
41]and thus represents a potential therapeutic target for early management of the disease.
3. Advancements in Diagnostic Techniques of Diabetic Retinopathy
Recent advances in retinal imaging have greatly improved the early detection and management of diabetic retinopathy (DR). Color fundus photography, which is useful for screening and monitoring DR, shows key disease indicators such as microaneurysms, retinal hemorrhages, and changes in intraretinal capillaries and veins [
42]. Fluorescein angiography (FA), although invasive and time-consuming, is effective in detecting capillary occlusion, new vessel growth, macular ischemia, and distinguishing between localized and widespread retinal edema [
43]. This method distinguishes between focal macular edema, characterized by localized retinal thickening, and diffuse macular edema, characterized by generalized leakage and accumulation of dye in the retina. However, due to the invasive nature of FA, many ophthalmologists now rely solely on optical coherence tomography (OCT) for the management of DME [
44]. OCT, which provides faster and more accurate assessments, measures central retinal thickness (CRT) to evaluate DME severity and treatment response. It also identifies different patterns of DME, such as diffuse retinal thickening, cystoid macular edema, and serous retinal detachment [
45]. Recently, OCT has distinguished two types of DME based on unique biomarkers and treatment response: the vascular DME phenotype (
Figure 3A), which is mainly due to inner blood-retinal barrier dysfunction and responds to anti-VEGF therapy, and the inflammatory DME phenotype (
Figure 3B), which typically responds better to steroids [
44,
46,
47]. The latter is linked to elevated cytokine levels in eye fluids, contributing to retinal changes [
44]. Features like multiple hyperreflective foci and serous retinal detachment, observed in 36.5% of DME eyes, are considered inflammation indicators, associated with increased hyperreflective foci and interleukin-6 levels [
44,
48].
4. Advanced Hybrid Closed Loop (AHCL) systems and T1D
4.1. Intensive insulin therapy and use of continuous glucose monitors
In recent years, the care of patients with T1D has undergone a remarkable revolution. Intensive use of exogenous insulin has been shown to slow the progression of hyperglycemia-related complications in patients with T1D, improving quality of life and reducing associated costs; however, it increases the risk of hypoglycemia [
49]. Thanks to major advances in blood glucose monitoring and insulin delivery technologies, a remarkable leap forward has been made in the management of diabetes. These breakthroughs have paved the way for a significant transformation in the management of DMT1, offering patients new opportunities to achieve glycemic control goals and improve their overall quality of life. The introduction of CGM systems has shown that glycate alone is not sufficient to assess the adequacy of glycemic compensation, but great importance should also be given to time spent in hypoglycemia (<70 and <54), time spent in hyperglycemia (<180 and >300) and glycemic variability 13 14: the use of these systems allows us not only to improve our ability to monitor diabetes, but also to improve the compensation of diabetes itself [
50].
4.2. Continuous Subcutaneous Insulin Infusion (CSII)
The use of continuous subcutaneous insulin delivery (CSII) devices has revolutionized insulin therapy in recent years; compared to multiple injection therapy (MDI), in which the patient takes three prandial ultra-rapid insulins and a daily basal insulin of approximately 24 hours' duration [
51], the pump allows the exclusive delivery of ultra-rapid insulin at rates programmed by the diabetologist[
52], which vary according to the different circadian needs of patients, in addition to the prandial boluses that are routinely administered before meals by the CSII [
52]. In these systems, it is often possible to schedule different daily insulin infusion profiles according to the different needs of the patient [
52] (days when the patient works vs. weekends, days when menstruation occurs, days when the patient is sick). In addition, the CSII allows prandial insulin boluses to be delivered at different rates depending on the macronutrient composition of the meal and based on different rates of nutrient absorption by patients (e.g., due to gastropathy associated with autonomic neuropathy) [
52]. A major advance in prandial bolus management has been the introduction of pump-integrated bolus calculators[
52], which allow accurate calculation of the recommended bolus for the patient by taking into account the insulin still in circulation delivered by the CSII, the patient's current measured blood glucose, target blood glucose, and patient-specific factors (periodically modified by the diabetologist) such as insulin sensitivity, insulin-to-carbohydrate ratio, and duration of insulin use [
52]. Commercially available pumps can deliver insulin subcutaneously through a cannula attached to a catheter connected to the CSII reservoir, or they can be catheterless, with the reservoir connected directly to the subcutis through a needle and monitored by an external PDA or cell phone [
52]. A major limitation of pumps, the increased incidence of diabetic ketoacidosis (DKA) in patients with DM1, has been overcome by the introduction of continuous glycemic monitoring sensors, which have have the capacity to alert the patient of CSII malfunctions through continuous detection of hypo/hyperglycemia in addition to the progressive improvements in patient education [
53]. Supported by international guidelines, pump therapies have emerged as a highly recommended approach for people with DMT[
52]1.
4.3. HCLS systems and glycemic variability
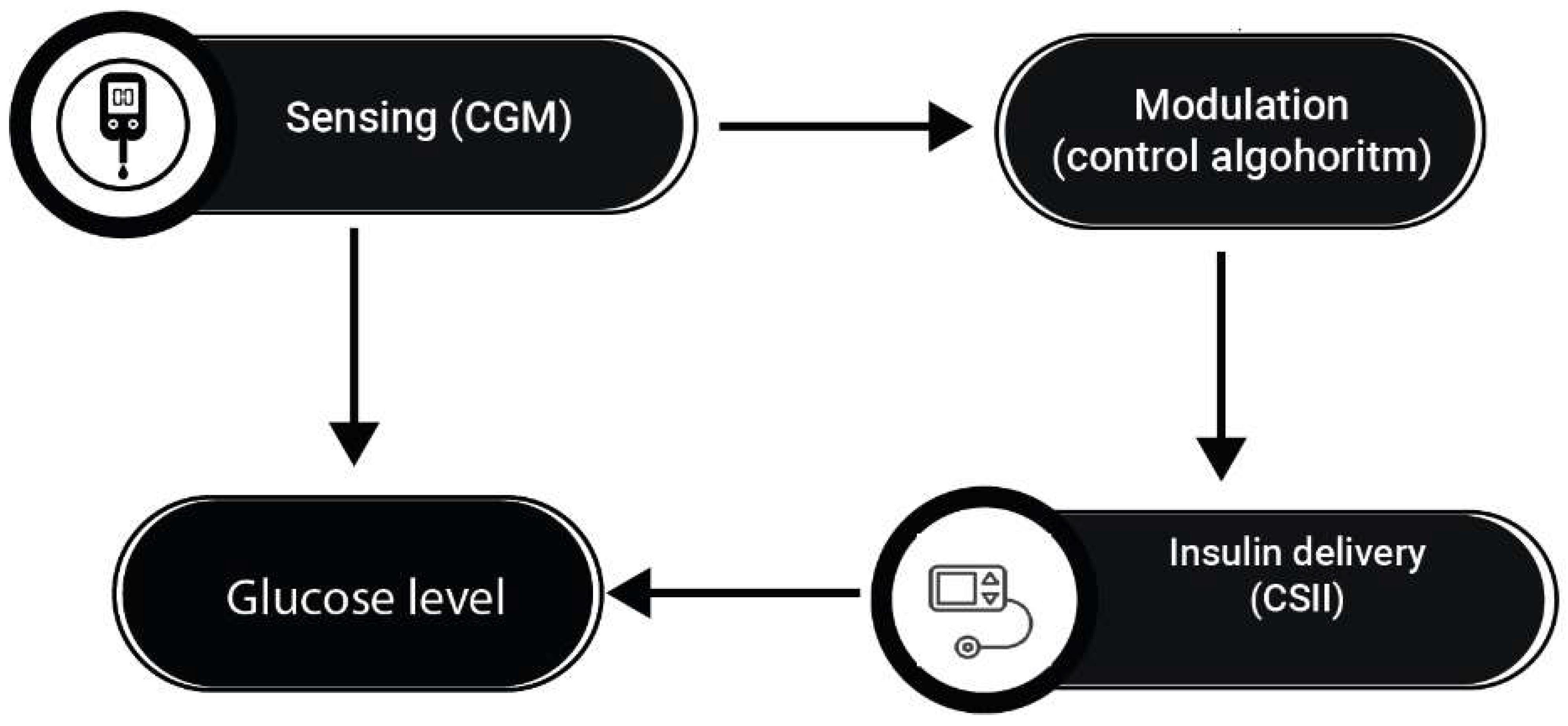
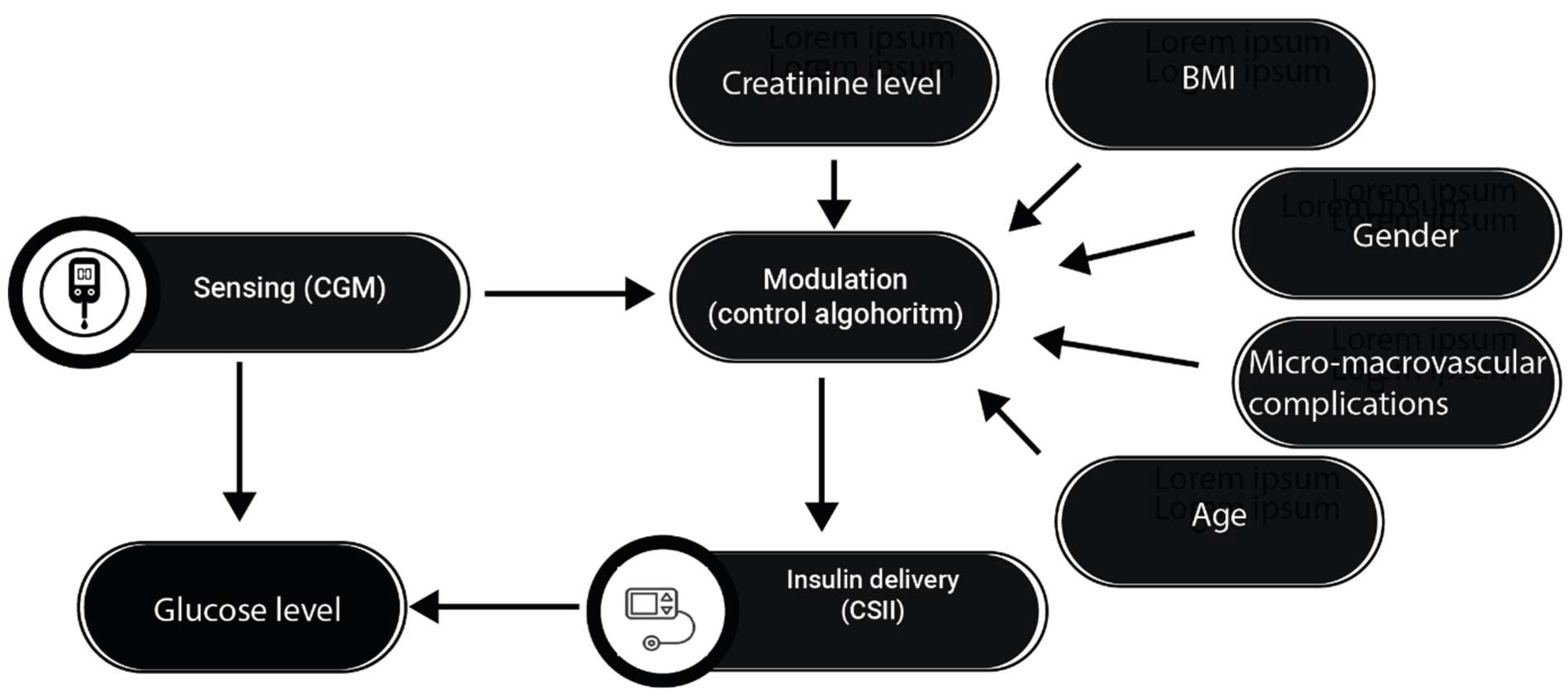
Recently, Hybrid Closed loop systems (HCLS), also known as hybrid artificial pancreas or closed-loop glucose control systems, have been developed to safely and automatically control blood glucose levels by integrating a subcutaneous insulin infusion pump, a subcutaneous continuous glucose monitor (CGM), and a control algorithm that continuously calculates the amount of insulin to infuse into the patient (
Figure 4)[
52]. Use of the subcutaneous route for both glucose sensing and insulin delivery is preferred over the intravenous (IV) and intraperitoneal (IP) routes because of the convenience and safety of outpatient use [
54]. The IV route can lead to frequent catheter problems such as intravenous migration and fibrin clot occlusion, while the IP route requires implantation and may increase the production of anti-insulin antibodies [
54]. Based on CGM measurements and the microdoses that the pump can handle, an HCLS is able to deliver the amount of insulin that the patient needs at any given time. In addition to insulin, glucagon and amylin analog hormones have been considered to improve the performance and safety of HCLS [
55]. These systems have revolutionized glycemic control by automating insulin delivery in response to continuous glucose monitoring data. However, the complexity and heterogeneity of diabetes require a more personalized approach to further improve metabolic control and prevent dysglycemia. Currently, no HCLS considers the exact macronutrient composition of meals, only their carbohydrate content, which limits the effectiveness of the bolus predictor. Another important limitation of the algorithms that run these systems is that they do not take into account a number of anamnestic, anthropometric and laboratory parameters to improve the pump's ability to match insulin delivery to the patient's blood glucose. No HCLS system considers specific patient characteristics that may modify insulin sensitivity, such as the presence of dual diabetes, the sex of the patient, the presence of menopause or hypogonadism, the presence of renal failure and/or liver cirrhosis. All of these factors, which are already measured to monitor the status of the diabetic patient and which affect the patient's insulin sensitivity in different ways, are currently ignored by all commercially available HCLS systems.
5. Emerging Research and Future Perspectives
5.1. Open source HCLS systems
When we talk about open source HCLS, we are essentially referring to systems where the brain that controls the interaction between CGM and CSII is an Android application, AndroidAps [
56]. In these systems, whose primary goal is to reduce the cost of HCLS and enable interoperability between CGM and CSII, the system algorithm is open and potentially modifiable by a team of experienced researchers [
56]. Although open-source artificial pancreases may appear to be equivalent in operation to traditional advanced HCLS, the potential of the latter becomes apparent when considering the possibilities offered by modern big data analytics. In fact, with these specific pumps, it is not only possible to perform retrospective analyses to see how a series of parameters known to affect blood glucose levels can modify the circadian glycemic patterns of patients, but it is also possible to hypothesize that not only the classical parameters underlying pump operation (basal insulin, insulin sensitivity, insulin-carbohydrate ratio, insulin duration) could be modified by the diabetologist: the entire algorithm of the HCLS could potentially be modified to tailor it to each specific patient (
Figure 5).
5.2. Prospects for future research and development
It is then possible to hypothesize that the pattern of insulin release in the dialyzed patient may be modified on days when he or she is further from dialysis, and that the system's response may be differentiated considering the patient's lean and fat mass, age, hormonal profile, and comorbidities (e.g., in patients undergoing chemotherapy on days when they are taking cortisone). The innovation that these systems offer us is the possibility of directing clinical research towards using the complex interplay between patient history, anthropometric parameters and instrumental studies in diabetes management in a structured way, to build algorithms that not only respond to changes in blood glucose levels to correct insulin delivery, but are also able, in the manner of a human diabetologist, to take into account all of the patient's comorbidities to stitch together a specific therapy that takes into account all of the patient's specific characteristics (
Figure 5).
6. Conclusions
The exploration of T1D and its associated complications in this review highlights the pivotal role of technological innovations, particularly in the form of Android applications and personalized Hybrid Closed Loop System (HCLS) algorithms. These advances are at the forefront of a revolution in diabetes management, offering new ways to mitigate the long-term complications of T1D. Central to this evolution is the integration of personalized algorithms into diabetes management. These are often only possible in open-source HCLS research frameworks today, but in the future will mark a significant leap toward individualized care, enabling more precise insulin dosing and reducing the risks associated with hyperglycemia and hypoglycemia. The potential of these applications to increase patient autonomy and engagement in their own care cannot be overstated. In addition, the development of personalized HCLS algorithms represents a major advance in the prevention of diabetes complications. By considering factors such as a patient's activity level, dietary habits and even hormonal fluctuations, these systems will be able to fine-tune insulin delivery to minimize the occurrence of glucose extremes (
Figure 5). This level of personalization is also critical to reducing the incidence of micro- and macrovascular complications associated with T1D, ultimately improving patient outcomes and quality of life. In addition, the integration of these advanced technologies into diabetes management aligns with the broader shift toward precision medicine. By harnessing the power of big data analytics and machine learning, these systems can continuously evolve to adapt to changing patient needs and incorporate new research findings. This dynamic approach is key to staying ahead of disease progression and mitigating its complications.
In conclusion, the integration personalized HCLS algorithms into the management of T1D is a game changer. It represents a new era of diabetes care that is more responsive, adaptive and patient-centered. As these technologies continue to evolve, their role in the prevention and management of T1D complications will undoubtedly grow, opening the way for more effective and personalized healthcare solutions.
Author Contributions
Conceptualization, G.C.,M.C and N.T..; methodology, writing—original draft preparation,; C.C., A.C. writing—review, visualization, supervision, validation. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Atkinson, M.A., G.S. Eisenbarth, and A.W. Michels, Type 1 diabetes. Lancet, 2014. 383(9911): p. 69-82.
- Patterson, C.C., et al., Worldwide estimates of incidence, prevalence and mortality of type 1 diabetes in children and adolescents: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract, 2019. 157: p. 107842.
-
Mortality in Type 1 Diabetes in the DCCT/EDIC Versus the General Population. Diabetes Care, 2016. 39(8): p. 1378-83.
- Petrie, D., et al., Recent trends in life expectancy for people with type 1 diabetes in Sweden. Diabetologia, 2016. 59(6): p. 1167-76.
- Rawshani, A., et al., Mortality and Cardiovascular Disease in Type 1 and Type 2 Diabetes. N Engl J Med, 2017. 376(15): p. 1407-1418.
-
Intensive Diabetes Treatment and Cardiovascular Outcomes in Type 1 Diabetes: The DCCT/EDIC Study 30-Year Follow-up. Diabetes Care, 2016. 39(5): p. 686-693.
- Lee, Y.-B., et al., Risk of early mortality and cardiovascular disease in type 1 diabetes: a comparison with type 2 diabetes, a nationwide study. Cardiovascular Diabetology, 2019. 18(1).
-
The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus. New England Journal of Medicine, 1993. 329(14): p. 977-986.
- Fong, D.S., et al., Retinopathy in diabetes. Diabetes Care, 2004. 27 Suppl 1: p. S84-7.
- Afkarian, M., et al., Kidney disease and increased mortality risk in type 2 diabetes. J Am Soc Nephrol, 2013. 24(2): p. 302-8.
- Rein, D.B., et al., The Economic Burden of Major Adult Visual Disorders in the United States. Archives of Ophthalmology, 2006. 124(12): p. 1754-1760.
- Teo, Z.L., et al., Global Prevalence of Diabetic Retinopathy and Projection of Burden through 2045: Systematic Review and Meta-analysis. Ophthalmology, 2021. 128(11): p. 1580-1591.
- Saini, D.C., A. Kochar, and R. Poonia, Clinical correlation of diabetic retinopathy with nephropathy and neuropathy. Indian J Ophthalmol, 2021. 69(11): p. 3364-3368.
- Yang, J. and Z. Liu, Mechanistic Pathogenesis of Endothelial Dysfunction in Diabetic Nephropathy and Retinopathy. Front Endocrinol (Lausanne), 2022. 13: p. 816400.
- Rudnisky, C.J., et al., High-resolution stereoscopic digital fundus photography versus contact lens biomicroscopy for the detection of clinically significant macular edema. Ophthalmology, 2002. 109(2): p. 267-74.
- Wang, W. and A.C.Y. Lo, Diabetic Retinopathy: Pathophysiology and Treatments. Int J Mol Sci, 2018. 19(6).
- Yau, J.W., et al., Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care, 2012. 35(3): p. 556-64.
- 18. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol, 1985. 103(12): p. 1796-806.
- Ogurtsova, K., et al., IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract, 2017. 128: p. 40-50.
- Varma, R. , et al., Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol, 2014. 132(11): p. 1334-40.
- Aiello, L.P. , Diabetic retinopathy and other ocular findings in the diabetes control and complications trial/epidemiology of diabetes interventions and complications study. Diabetes Care, 2014. 37(1): p. 17-23.
- Kohner, E.M. , Microvascular disease: what does the UKPDS tell us about diabetic retinopathy? Diabet Med, 2008. 25 Suppl 2: p. 20-4.
- Tang, J. and T.S. Kern, Inflammation in diabetic retinopathy. Prog Retin Eye Res, 2011. 30(5): p. 343-58.
- Bek, T. , Diameter Changes of Retinal Vessels in Diabetic Retinopathy. Curr Diab Rep, 2017. 17(10): p. 82.
- Romeo, G. , et al., Activation of nuclear factor-kappaB induced by diabetes and high glucose regulates a proapoptotic program in retinal pericytes. Diabetes, 2002. 51(7): p. 2241-8.
- Ejaz, S. , et al., Importance of pericytes and mechanisms of pericyte loss during diabetes retinopathy. Diabetes Obes Metab, 2008. 10(1): p. 53-63.
- Beltramo, E. and M. Porta, Pericyte loss in diabetic retinopathy: mechanisms and consequences. Curr Med Chem, 2013. 20(26): p. 3218-25.
- Huang, H. , et al., Deletion of placental growth factor prevents diabetic retinopathy and is associated with Akt activation and HIF1α-VEGF pathway inhibition. Diabetes, 2015. 64(1): p. 200-12.
- Li, J. , et al., Inhibition of reactive oxygen species by Lovastatin downregulates vascular endothelial growth factor expression and ameliorates blood-retinal barrier breakdown in db/db mice: role of NADPH oxidase 4. Diabetes, 2010. 59(6): p. 1528-38.
- Aiello, L.P. , et al., Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med, 1994. 331(22): p. 1480-7.
- Tien, T. , et al., High Glucose Induces Mitochondrial Dysfunction in Retinal Müller Cells: Implications for Diabetic Retinopathy. Invest Ophthalmol Vis Sci, 2017. 58(7): p. 2915-2921.
- Guo, C. , et al., HIF-1α accumulation in response to transient hypoglycemia may worsen diabetic eye disease. Cell Rep, 2023. 42(1): p. 111976.
- Park, J.Y. , et al., EFFECTS OF GLYCEMIC VARIABILITY ON THE PROGRESSION OF DIABETIC RETINOPATHY AMONG PATIENTS WITH TYPE 2 DIABETES. Retina, 2021. 41(7): p. 1487-1495.
- Hsu, C.-R., Y. -T. Chen, and W.H.H. Sheu, Glycemic variability and diabetes retinopathy: A missing link. Journal of Diabetes and its Complications, 2015. 29(2): p. 302-306.
- Bombaci, B. , et al., One-Year Real-World Study on Comparison among Different Continuous Subcutaneous Insulin Infusion Devices for the Management of Pediatric Patients with Type 1 Diabetes: The Supremacy of Hybrid Closed-Loop Systems. Int J Environ Res Public Health, 2022. 19(16).
- Keyu, G. , et al., Comparing the effectiveness of continuous subcutaneous insulin infusion with multiple daily insulin injection for patients with type 1 diabetes mellitus evaluated by retrospective continuous glucose monitoring: A real-world data analysis. Front Public Health, 2022. 10: p. 990281.
- Lunati, M.E. , et al., Hybrid Close-Loop Systems Versus Predictive Low-Glucose Suspend and Sensor-Augmented Pump Therapy in Patients With Type 1 Diabetes: A Single-Center Cohort Study. Frontiers in Endocrinology, 2022. 13.
- Lin, Y., Y. Tang, and F. Wang, The Protective Effect of HIF-1α in T Lymphocytes on Cardiac Damage in Diabetic Mice. Ann Clin Lab Sci, 2016. 46(1): p. 32-43.
- Barber, A.J. , et al., Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest, 1998. 102(4): p. 783-91.
- Sasaki, M. , et al., Neurodegenerative influence of oxidative stress in the retina of a murine model of diabetes. Diabetologia, 2010. 53(5): p. 971-9.
- Sohn, E.H. , et al., Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci U S A, 2016. 113(19): p. E2655-64.
- Mahapatra, S. , et al., A Review on Retinal Blood Vessel Enhancement and Segmentation Techniques for Color Fundus Photography. Crit Rev Biomed Eng, 2024. 52(1): p. 41-69.
- Kylstra, J.A. , et al., The importance of fluorescein angiography in planning laser treatment of diabetic macular edema. Ophthalmology, 1999. 106(11): p. 2068-73.
- Vujosevic, S. , et al., Hyperreflective intraretinal spots in diabetics without and with nonproliferative diabetic retinopathy: an in vivo study using spectral domain OCT. J Diabetes Res, 2013. 2013: p. 491835.
- Chung, Y.R. , et al., Role of Inflammation in Classification of Diabetic Macular Edema by Optical Coherence Tomography. J Diabetes Res, 2019. 2019: p. 8164250.
- Otani, T., S. Kishi, and Y. Maruyama, Patterns of diabetic macular edema with optical coherence tomography. Am J Ophthalmol, 1999. 127(6): p. 688-93.
- Sonoda, S. , et al., Retinal morphologic changes and concentrations of cytokines in eyes with diabetic macular edema. Retina, 2014. 34(4): p. 741-8.
- Vujosevic, S. , et al., Diabetic Macular Edema With and Without Subfoveal Neuroretinal Detachment: Two Different Morphologic and Functional Entities. Am J Ophthalmol, 2017. 181: p. 149-155.
-
Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. Diabetes Control and Complications Trial Research Group. J Pediatr, 1994. 125(2): p. 177-88.
- Pratley, R.E. , et al., Effect of Continuous Glucose Monitoring on Hypoglycemia in Older Adults With Type 1 Diabetes: A Randomized Clinical Trial. Jama, 2020. 323(23): p. 2397-2406.
- Liu, W., X. Yang, and J. Huang, Efficacy and Safety of Insulin Degludec versus Insulin Glargine: A Systematic Review and Meta-Analysis of Fifteen Clinical Trials. Int J Endocrinol, 2018. 2018: p. 8726046.
- Sherr, J.L. , et al., Automated insulin delivery: benefits, challenges, and recommendations. A Consensus Report of the Joint Diabetes Technology Working Group of the European Association for the Study of Diabetes and the American Diabetes Association. Diabetologia, 2023. 66(1): p. 3-22.
- Hoshina, S. , et al., Treatment Modality-Dependent Risk of Diabetic Ketoacidosis in Patients with Type 1 Diabetes: Danish Adult Diabetes Database Study. Diabetes Technol Ther, 2018. 20(3): p. 229-234.
- Cobelli, C., E. Renard, and B. Kovatchev, Artificial Pancreas: Past, Present, Future. Diabetes, 2011. 60(11): p. 2672-2682.
- Infante, M. , et al., Dual-hormone artificial pancreas for management of type 1 diabetes: Recent progress and future directions. Artif Organs, 2021. 45(9): p. 968-986.
- Kesavadev, J. , et al., The Do-It-Yourself Artificial Pancreas: A Comprehensive Review. Diabetes Ther, 2020. 11(6): p. 1217-1235.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).