Submitted:
26 January 2024
Posted:
29 January 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Cell Culture and Transfection
Plasmids, Site-Directed Mutagenesis, and in vitro RNA synthesis
Zebrafish Housing and Morpholino (MO) Injection
Structure Refinement, Molecular Modeling, and Interaction Analysis
Cloning, Expression, and Purification of LRRs
Cell proliferation Assays
Protein Co-Purification Assay
Statistical Analysis
Results
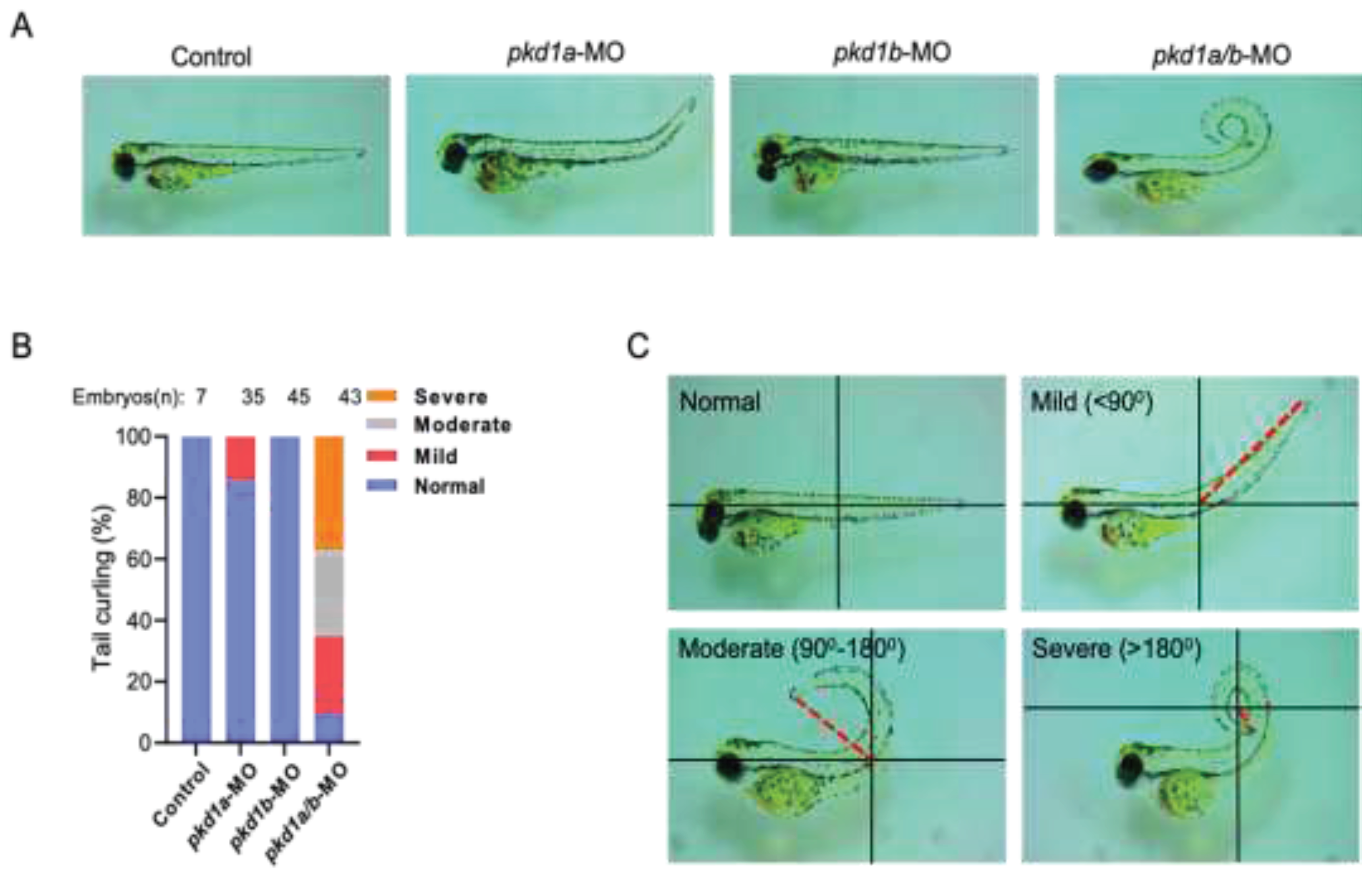
Zebrafish Human PKD1 Paralogs, pkd1a and pkd1b, Have Redundant Functions
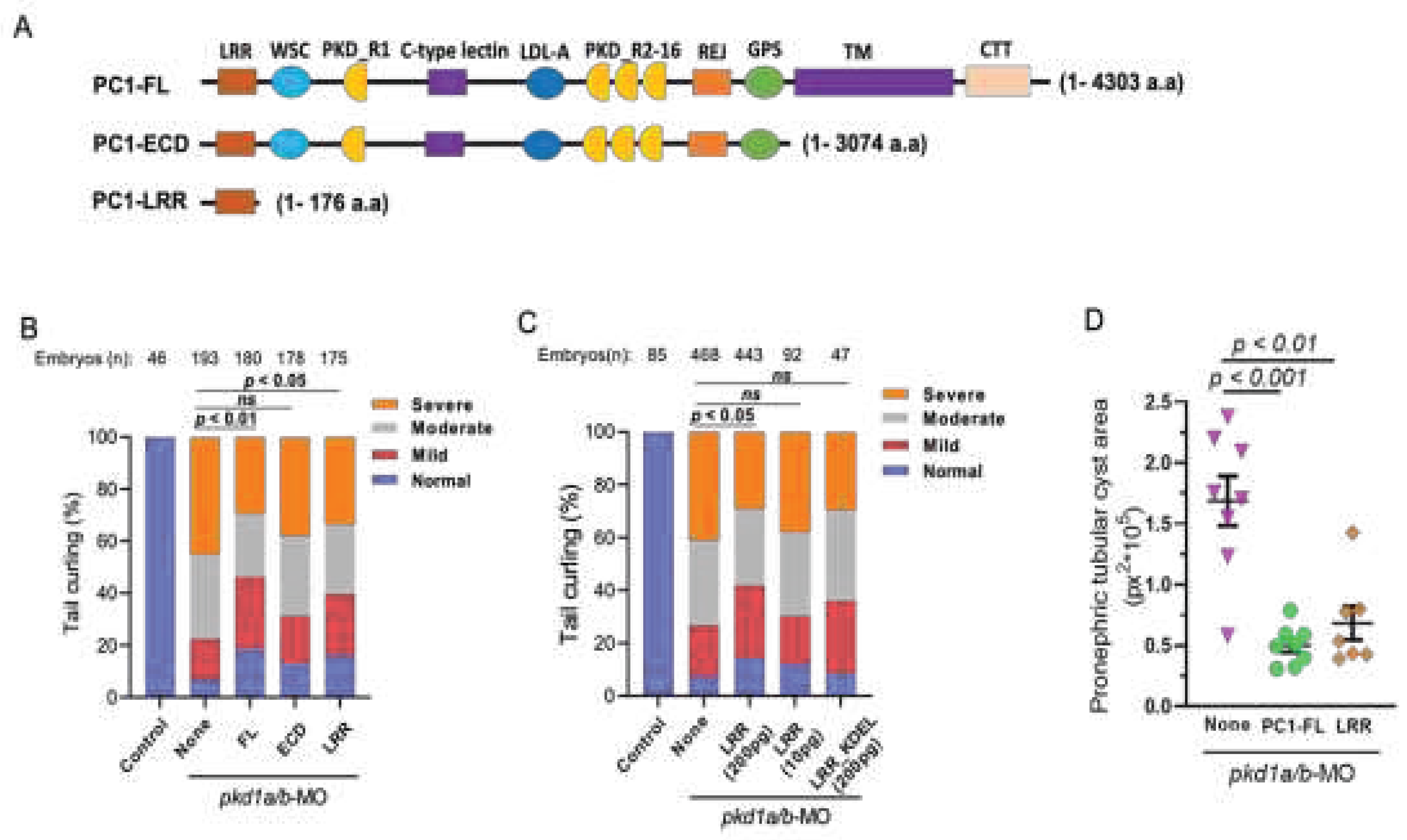
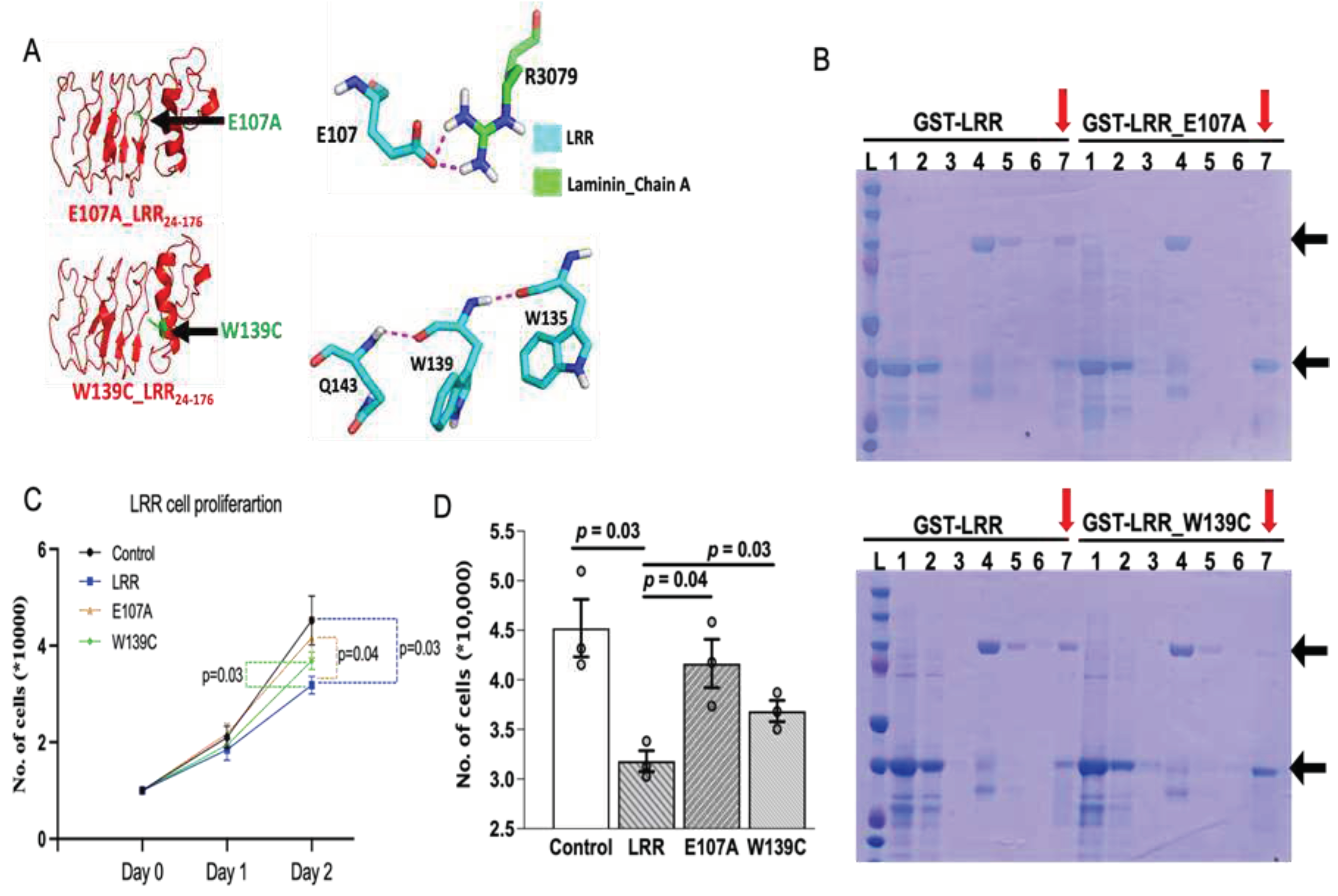
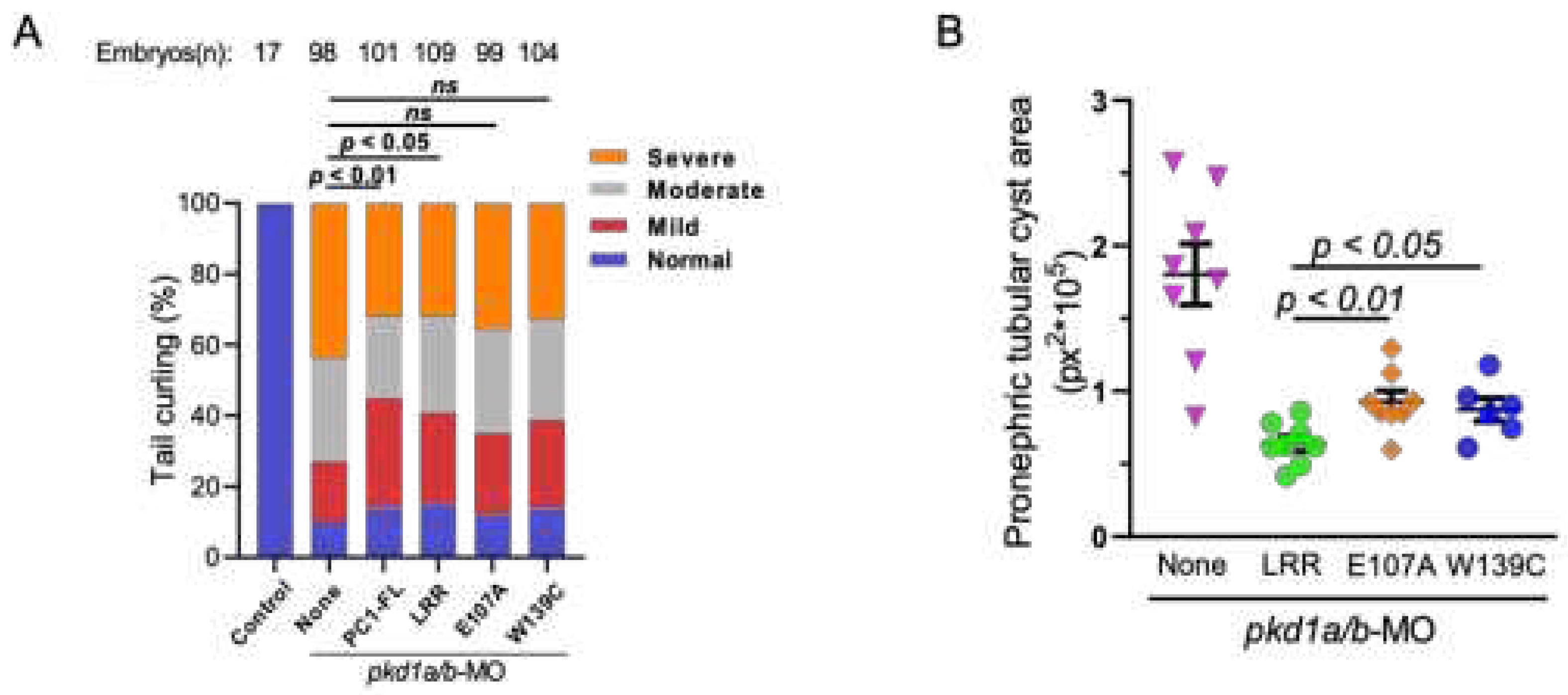
Overexpression of N-Terminal Leucine-Rich Repeats Suppresses Dorsal Axis Curvature and Pronephric Cystic Phenotypes in pkd1a/b Morphants
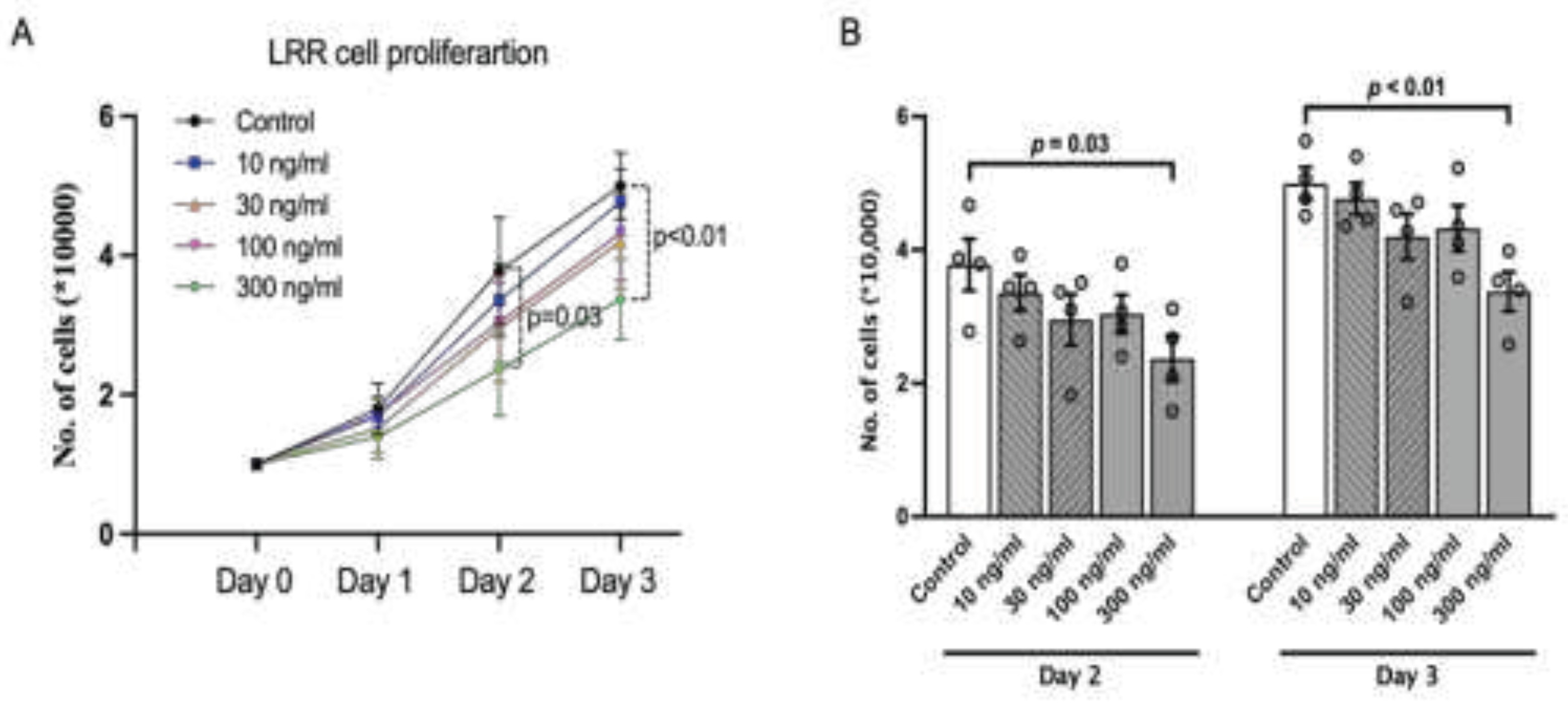
LRR Plays an Anti-Proliferative Role in Cell Proliferation
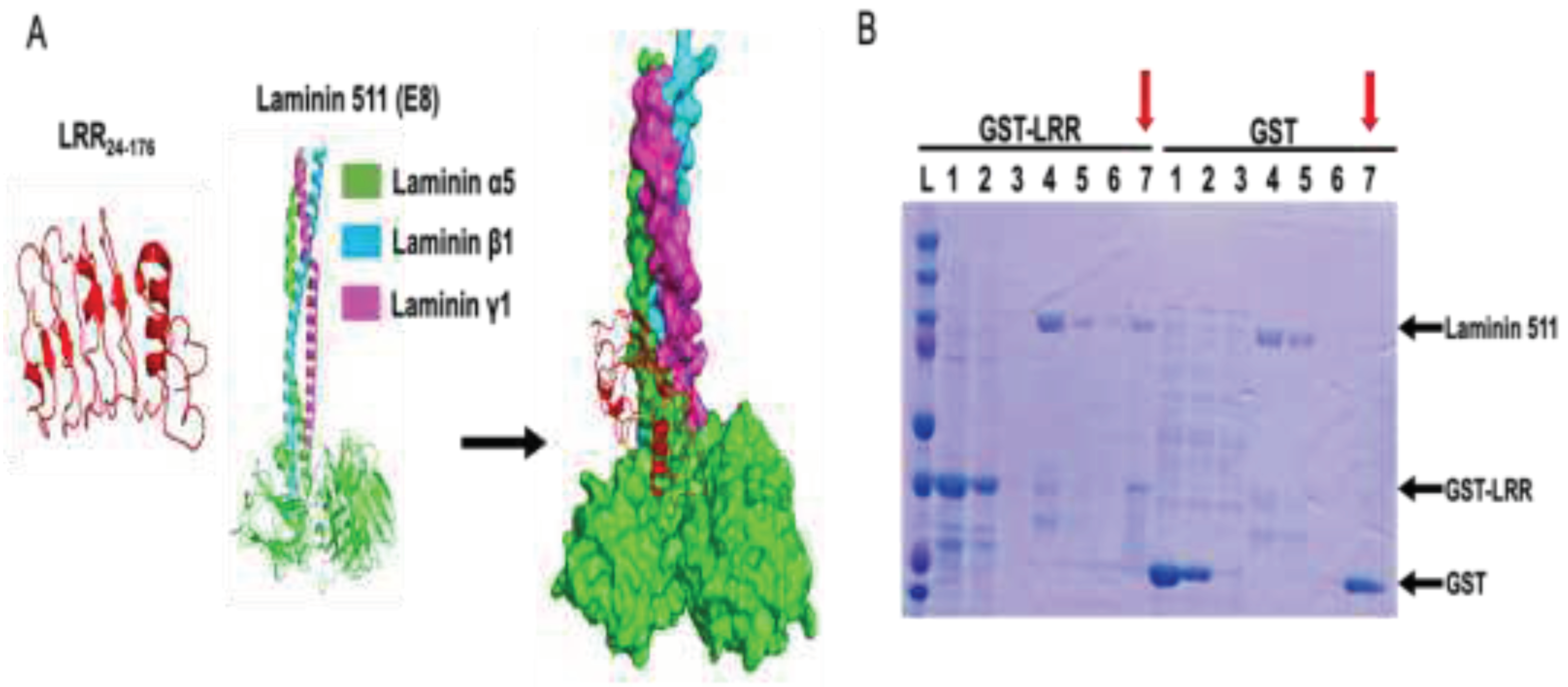
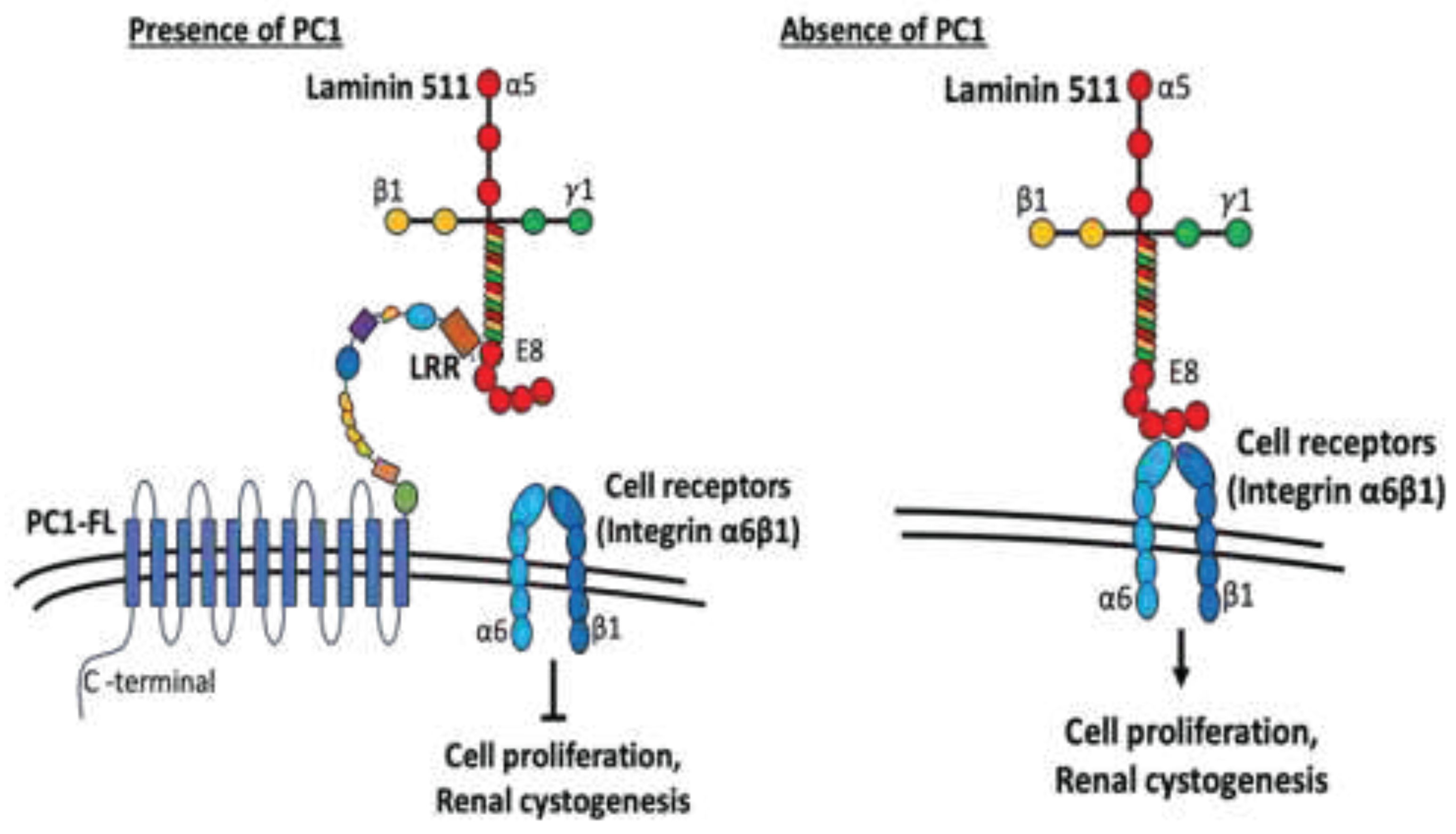
LRR Binds to Extracellular Matrix Protein, Laminin-511
Binding of LRR to Laminin-511 Suppresses Renal Cystogenesis
Discussion
Author Contributions
Funding
Data availability statement
Conflicts of Interest
References
- Luciano, R.L.; Dahl, N.K. Extra-renal manifestations of autosomal dominant polycystic kidney disease (ADPKD): Considerations for routine screening and management. Nephrol. Dial. Transplant. 2014, 29, 247–254. [Google Scholar] [CrossRef]
- Igarashi, P.; Somlo, S. Genetics and pathogenesis of polycystic kidney disease. J. Am. Soc. Nephrol. 2002, 13, 2384–2398. [Google Scholar] [CrossRef]
- Grimm, D.H.; Cai, Y.; Chauvet, V.; Rajendran, V.; Zeltner, R.; Geng, L.; Avner, E.D.; Sweeney, W.; Somlo, S.; Caplan, M.J. Polycystin-1 distribution is modulated by polycystin-2 expression in mammalian cells. J. Biol. Chem. 2003, 278, 36786–36793. [Google Scholar] [CrossRef]
- Lin, C.C.; Kurashige, M.; Liu, Y.; Terabayashi, T.; Ishimoto, Y.; Wang, T.; Choudhary, V.; Hobbs, R.; Liu, L.K.; Lee, P.H.; et al. A cleavage product of Polycystin-1 is a mitochondrial matrix protein that affects mitochondria morphology and function when heterologously expressed. Sci. Rep. 2018, 8, 2743. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Hu, F.; Ge, X.; Lei, J.; Yu, S.; Wang, T.; Zhou, Q.; Mei, C.; Shi, Y. Structure of the human PKD1-PKD2 complex. Science 2018, 361. [Google Scholar] [CrossRef] [PubMed]
- Low, S.H.; Vasanth, S.; Larson, C.H.; Mukherjee, S.; Sharma, N.; Kinter, M.T.; Kane, M.E.; Obara, T.; Weimbs, T. Polycystin-1, STAT6, and P100 function in a pathway that transduces ciliary mechanosensation and is activated in polycystic kidney disease. Dev. Cell 2006, 10, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Parnell, S.C.; Magenheimer, B.S.; Maser, R.L.; Rankin, C.A.; Smine, A.; Okamoto, T.; Calvet, J.P. The polycystic kidney disease-1 protein, polycystin-1, binds and activates heterotrimeric G-proteins in vitro. Biochem. Biophys. Res. Commun. 1998, 251, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Arnould, T.; Kim, E.; Tsiokas, L.; Jochimsen, F.; Gruning, W.; Chang, J.D.; Walz, G. The polycystic kidney disease 1 gene product mediates protein kinase C alpha-dependent and c-Jun N-terminal kinase-dependent activation of the transcription factor AP-1. J. Biol. Chem. 1998, 273, 6013–6018. [Google Scholar] [CrossRef] [PubMed]
- Casuscelli, J.; Schmidt, S.; DeGray, B.; Petri, E.T.; Celic, A.; Folta-Stogniew, E.; Ehrlich, B.E.; Boggon, T.J. Analysis of the cytoplasmic interaction between polycystin-1 and polycystin-2. Am. J. Physiol. Renal Physiol. 2009, 297, F1310–F1315. [Google Scholar] [CrossRef] [PubMed]
- Giamarchi, A.; Feng, S.; Rodat-Despoix, L.; Xu, Y.; Bubenshchikova, E.; Newby, L.J.; Hao, J.; Gaudioso, C.; Crest, M.; Lupas, A.N.; et al. A polycystin-2 (TRPP2) dimerization domain essential for the function of heteromeric polycystin complexes. EMBO J. 2010, 29, 1176–1191. [Google Scholar] [CrossRef]
- Weston, B.S.; Malhas, A.N.; Price, R.G. Structure-function relationships of the extracellular domain of the autosomal dominant polycystic kidney disease-associated protein, polycystin-1. FEBS Lett. 2003, 538, 8–13. [Google Scholar] [CrossRef]
- Harris, P.C.; Torres, V.E. Polycystic kidney disease. Annu. Rev. Med. 2009, 60, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Vien, T.; Duan, J.; Sheu, S.H.; DeCaen, P.G.; Clapham, D.E. Polycystin-2 is an essential ion channel subunit in the primary cilium of the renal collecting duct epithelium. Elife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Caplan, M.J. Holding open the door reveals a new view of polycystin channel function. EMBO Rep. 2019, 20, e49156. [Google Scholar] [CrossRef] [PubMed]
- Padhy, B.; Xie, J.; Wang, R.; Lin, F.; Huang, C.L. Channel Function of Polycystin-2 in the Endoplasmic Reticulum Protects against Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2022, 33, 1501–1516. [Google Scholar] [CrossRef] [PubMed]
- Mangos, S.; Lam, P.Y.; Zhao, A.; Liu, Y.; Mudumana, S.; Vasilyev, A.; Liu, A.; Drummond, I.A. The ADPKD genes pkd1a/b and pkd2 regulate extracellular matrix formation. Dis. Model. Mech. 2010, 3, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Sullivan-Brown, J.; Bisher, M.E.; Burdine, R.D. Embedding, serial sectioning and staining of zebrafish embryos using JB-4 resin. Nat. Protoc. 2011, 6, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, M.; Arimori, T.; Taniguchi, Y.; Kitago, Y.; Yamashita, E.; Takagi, J.; Sekiguchi, K. Mechanistic basis for the recognition of laminin-511 by alpha6beta1 integrin. Sci. Adv. 2017, 3, e1701497. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, Y. I-TASSER server: New development for protein structure and function predictions. Nucleic Acids Res. 2015, 43, W174–W181. [Google Scholar] [CrossRef]
- Yang, J.; Yan, R.; Roy, A.; Xu, D.; Poisson, J.; Zhang, Y. The I-TASSER Suite: Protein structure and function prediction. Nat. Methods 2015, 12, 7–8. [Google Scholar] [CrossRef]
- Kozakov, D.; Hall, D.R.; Xia, B.; Porter, K.A.; Padhorny, D.; Yueh, C.; Beglov, D.; Vajda, S. The ClusPro web server for protein-protein docking. Nat. Protoc. 2017, 12, 255–278. [Google Scholar] [CrossRef]
- Kaur, J.; Reinhardt, D.P. Immobilized metal affinity chromatography co-purifies TGF-beta1 with histidine-tagged recombinant extracellular proteins. PLoS ONE 2012, 7, e48629. [Google Scholar] [CrossRef]
- Ray, D.; Laverty, K.U.; Jolma, A.; Nie, K.; Samson, R.; Pour, S.E.; Tam, C.L.; von Krosigk, N.; Nabeel-Shah, S.; Albu, M.; et al. RNA-binding proteins that lack canonical RNA-binding domains are rarely sequence-specific. Sci. Rep. 2023, 13, 5238. [Google Scholar] [CrossRef]
- Arif Pavel, M.; Lv, C.; Ng, C.; Yang, L.; Kashyap, P.; Lam, C.; Valentino, V.; Fung, H.Y.; Campbell, T.; Moller, S.G.; et al. Function and regulation of TRPP2 ion channel revealed by a gain-of-function mutant. Proc. Natl. Acad. Sci. USA 2016, 113, E2363–E2372. [Google Scholar] [CrossRef] [PubMed]
- Malhas, A.N.; Abuknesha, R.A.; Price, R.G. Interaction of the leucine-rich repeats of polycystin-1 with extracellular matrix proteins: Possible role in cell proliferation. J. Am. Soc. Nephrol. 2002, 13, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Weston, B.S.; Bagneris, C.; Price, R.G.; Stirling, J.L. The polycystin-1 C-type lectin domain binds carbohydrate in a calcium-dependent manner, and interacts with extracellular matrix proteins in vitro. Biochim. Biophys. Acta 2001, 1536, 161–176. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M.; Virtanen, I.; Palgi, J.; Korsgren, O.; Otonkoski, T. Proliferation and plasticity of human beta cells on physiologically occurring laminin isoforms. Mol. Cell Endocrinol. 2012, 355, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Ido, H.; Harada, K.; Futaki, S.; Hayashi, Y.; Nishiuchi, R.; Natsuka, Y.; Li, S.; Wada, Y.; Combs, A.C.; Ervasti, J.M.; et al. Molecular dissection of the alpha-dystroglycan- and integrin-binding sites within the globular domain of human laminin-10. J. Biol. Chem. 2004, 279, 10946–10954. [Google Scholar] [CrossRef] [PubMed]
- Kunneken, K.; Pohlentz, G.; Schmidt-Hederich, A.; Odenthal, U.; Smyth, N.; Peter-Katalinic, J.; Bruckner, P.; Eble, J.A. Recombinant human laminin-5 domains. Effects of heterotrimerization, proteolytic processing, and N-glycosylation on alpha3beta1 integrin binding. J. Biol. Chem. 2004, 279, 5184–5193. [Google Scholar] [CrossRef] [PubMed]
- Puri, S.; Magenheimer, B.S.; Maser, R.L.; Ryan, E.M.; Zien, C.A.; Walker, D.D.; Wallace, D.P.; Hempson, S.J.; Calvet, J.P. Polycystin-1 activates the calcineurin/NFAT (nuclear factor of activated T-cells) signaling pathway. J. Biol. Chem. 2004, 279, 55455–55464. [Google Scholar] [CrossRef]
- Lal, M.; Song, X.; Pluznick, J.L.; Di Giovanni, V.; Merrick, D.M.; Rosenblum, N.D.; Chauvet, V.; Gottardi, C.J.; Pei, Y.; Caplan, M.J. Polycystin-1 C-terminal tail associates with beta-catenin and inhibits canonical Wnt signaling. Hum. Mol. Genet. 2008, 17, 3105–3117. [Google Scholar] [CrossRef]
- Ibraghimov-Beskrovnaya, O.; Bukanov, N.O.; Donohue, L.C.; Dackowski, W.R.; Klinger, K.W.; Landes, G.M. Strong homophilic interactions of the Ig-like domains of polycystin-1, the protein product of an autosomal dominant polycystic kidney disease gene, PKD1. Hum. Mol. Genet. 2000, 9, 1641–1649. [Google Scholar] [CrossRef]
- Joly, D.; Morel, V.; Hummel, A.; Ruello, A.; Nusbaum, P.; Patey, N.; Noel, L.H.; Rousselle, P.; Knebelmann, B. Beta4 integrin and laminin 5 are aberrantly expressed in polycystic kidney disease: Role in increased cell adhesion and migration. Am. J. Pathol. 2003, 163, 1791–1800. [Google Scholar] [CrossRef]
- van Adelsberg, J. Murine polycystic kidney epithelial cell lines have increased integrin-mediated adhesion to collagen. Am. J. Physiol. 1994, 267, F1082–F1093. [Google Scholar] [CrossRef]
- von der Mark, K.; Kuhl, U. Laminin and its receptor. Biochim. Biophys. Acta 1985, 823, 147–160. [Google Scholar] [CrossRef]
- Yap, L.; Tay, H.G.; Nguyen, M.T.X.; Tjin, M.S.; Tryggvason, K. Laminins in Cellular Differentiation. Trends Cell Biol. 2019, 29, 987–1000. [Google Scholar] [CrossRef]
- Nakashima, Y.; Kariya, Y.; Miyazaki, K. The beta3 chain short arm of laminin-332 (laminin-5) induces matrix assembly and cell adhesion activity of laminin-511 (laminin-10). J. Cell Biochem. 2007, 100, 545–556. [Google Scholar] [CrossRef]
- Bodary, S.C.; McLean, J.W. The integrin beta 1 subunit associates with the vitronectin receptor alpha v subunit to form a novel vitronectin receptor in a human embryonic kidney cell line. J. Biol. Chem. 1990, 265, 5938–5941. [Google Scholar] [CrossRef]
- Yarwood, S.J.; Woodgett, J.R. Extracellular matrix composition determines the transcriptional response to epidermal growth factor receptor activation. Proc. Natl. Acad. Sci. USA 2001, 98, 4472–4477. [Google Scholar] [CrossRef]
- Miner, J.H. Renal basement membrane components. Kidney Int. 1999, 56, 2016–2024. [Google Scholar] [CrossRef]
- Shannon, M.B.; Patton, B.L.; Harvey, S.J.; Miner, J.H. A hypomorphic mutation in the mouse laminin alpha5 gene causes polycystic kidney disease. J. Am. Soc. Nephrol. 2006, 17, 1913–1922. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Boctor, S.; Barisoni, L.M.; Gusella, G.L. Inactivation of integrin-beta1 prevents the development of polycystic kidney disease after the loss of polycystin-1. J. Am. Soc. Nephrol. 2015, 26, 888–895. [Google Scholar] [CrossRef]
- Wilson, P.D.; Geng, L.; Li, X.; Burrow, C.R. The PKD1 gene product, "polycystin-1," is a tyrosine-phosphorylated protein that colocalizes with alpha2beta1-integrin in focal clusters in adherent renal epithelia. Lab. Invest. 1999, 79, 1311–1323. [Google Scholar] [PubMed]
- Drummond, I.A. Polycystins, focal adhesions and extracellular matrix interactions. Biochim. Biophys. Acta 2011, 1812, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



