1. Introduction
Already, in 1939, Ide et al. suggested that tumors may release specific factors with the aim to stimulate the growth of blood vessels [
1]. After 32 years, Dr. Folkmann, a renowned scientist known as "the father of angiogenesis”, proposed in the esteemed "New England Journal of Medicine" that cancer cells may be effectively treated by blocking their blood supply, depriving them of essential nutrients. This idea of anti-angiogenesis as a cancer treatment approach focuses on inhibiting the entry of new blood vessels into the early tumor implantation. Dr Folkmann has already known that without new blood vessels, tumors cannot grow from a microscopic size of 1-2 mm
3 [
2]. However, was he aware that it would become possible and used in clinical practice within three decades?
Bevacizumab's development required the diligent efforts of numerous researchers. In 1983, Senger et al. identified the factor present in tumor ascites fluid from guinea pigs, hamsters, and mice increasing microvascular permeability [
3]. In 1989, Ferrara and Hanzel observed that pituitary follicular cells secrete a growth factor specific for vascular endothelial cells. They proposed to name it vascular endothelial growth factor (VEGF), which remains to this day [
4]. In 1993, Kim et al. published a study in the scientific journal “Nature” reporting that the treatment with a monoclonal antibody specific for VEGF inhibited the growth of the tumors in laboratory conditions. Moreover, it was observed that the density of the vessels in the treated group was lower [
5]. Therefore, subsequent research was more and more advanced… until the first registration of the new drug by the FDA in 2004 [
6].
An antiangiogenic medicine, bevacizumab, is the treatment option which changed and is still changing today’s oncology. Bevacizumab is an antibody, which is used in more and more indications. In 2011 it was approved by the European Medicines Agency (EMA) and in 2018 [
7] by the FDA as a treatment option for patients with stage III or IV epithelial ovarian cancer [
8]. In this indication, it still raises a lot of controversies. One of these aspects concerns the impact of residual disease location of ovarian cancer on bevacizumab treatment. Bevacizumab acts by specifically binding VEGF-A, which is the isoform that plays a crucial role in forming new blood vessels [
7,
9,
10]. Meanwhile, in tumors located in lymphatic organs such as lymph nodes, nutrients are delivered also by lymphatic vessels. The formation of those vessels is regulated by the other isoforms from the VEGF family, namely VEGF-C and VEGF-D. It is worth noting that these isoforms are not targeted by bevacizumab. Could this be the reason that limits the efficacy of bevacizumab treatment? However, is it truly of significant importance?
2. Materials and Methods
The studies cited in the presented review were selected from PUBMED, Google Scholar and Science Direct databases. Terms used by us were created by combining all words connected with bevacizumab and different types of gynecological cancers, which we were describing in our manuscript, by using boolean operator “OR”. Among these terms were for example: “bevacizumab”, "bev", “antiangiogenic treatment”. Thereafter we were adding new words of interest with the use of the boolean operator "AND". These words were changing depending on the part of the manuscript, we were working on. Among them were for example: “ovarian cancer”, “endometrial cancer”. Moreover, we used also more specific terms relating to the mechanism of action of bevacizumab including: “residual disease” or “lymph nodes” and others, which are connected to selected sections. Even if the paper was designed as narrative review, we applied the rules of paper selection listed below.
Inclusion criteria:
- -
The types of included studies: clinical trials, retrospective studies, reviews, metanalyses;
- -
More than one patient described in the study
- -
No limitations of the year of published study were used
Exclusion criteria:
- -
Articles not written in English
- -
Conference abstracts only
- -
Study cases
- -
Duplicated papers
3. Process of angiogenesis
In order to grow and progress, solid tumors must constantly need nutrients and oxygen. Angiogenesis, which refers to forming new blood vessels from pre-existing ones, is essential for the development of solid tumors [
9,
11]. Physiologically, angiogenesis is an important process during embryogenesis. In an adult, in turn, it is a process that is usually active in wound healing [
10]. Moreover, enhanced angiogenesis has been demonstrated in the process of cancerogenesis, as the formation of new blood vessels is crucial to the metabolic demands of the tumor [
11]. Moreover, angiogenesis is involved in the development of various diseases and pathological conditions, including vascular inflammation, rheumatoid arthritis, psoriasis, gastritis, inflammatory bowel disease, ischemic heart disease, atherosclerosis, cyanotic heart defects, and diabetes [
10,
12,
13].
The creation of new blood vessels is possible thanks to pro-angiogenic factors secreted by tumors, i.e., VEGFs. These molecules interact with endothelial cells and stimulate angiogenesis by binding to VEGF receptor tyrosine kinases (VEGFR1-3) [
14,
15]. Moreover, study results showed increased VEGF expression within solid tumors associated with a poor prognosis, including metastatic breast cancer [
16], renal cell carcinoma [
17], metastatic colorectal cancer [
18], ovarian cancer, and various other types [
19]. Taking into account that the overexpression of VEGF in cancers or metastases, promotes the formation of new blood vessels and, hence, tumor development, it seemed necessary to develop therapeutic methods targeting the anti-angiogenic mechanism.
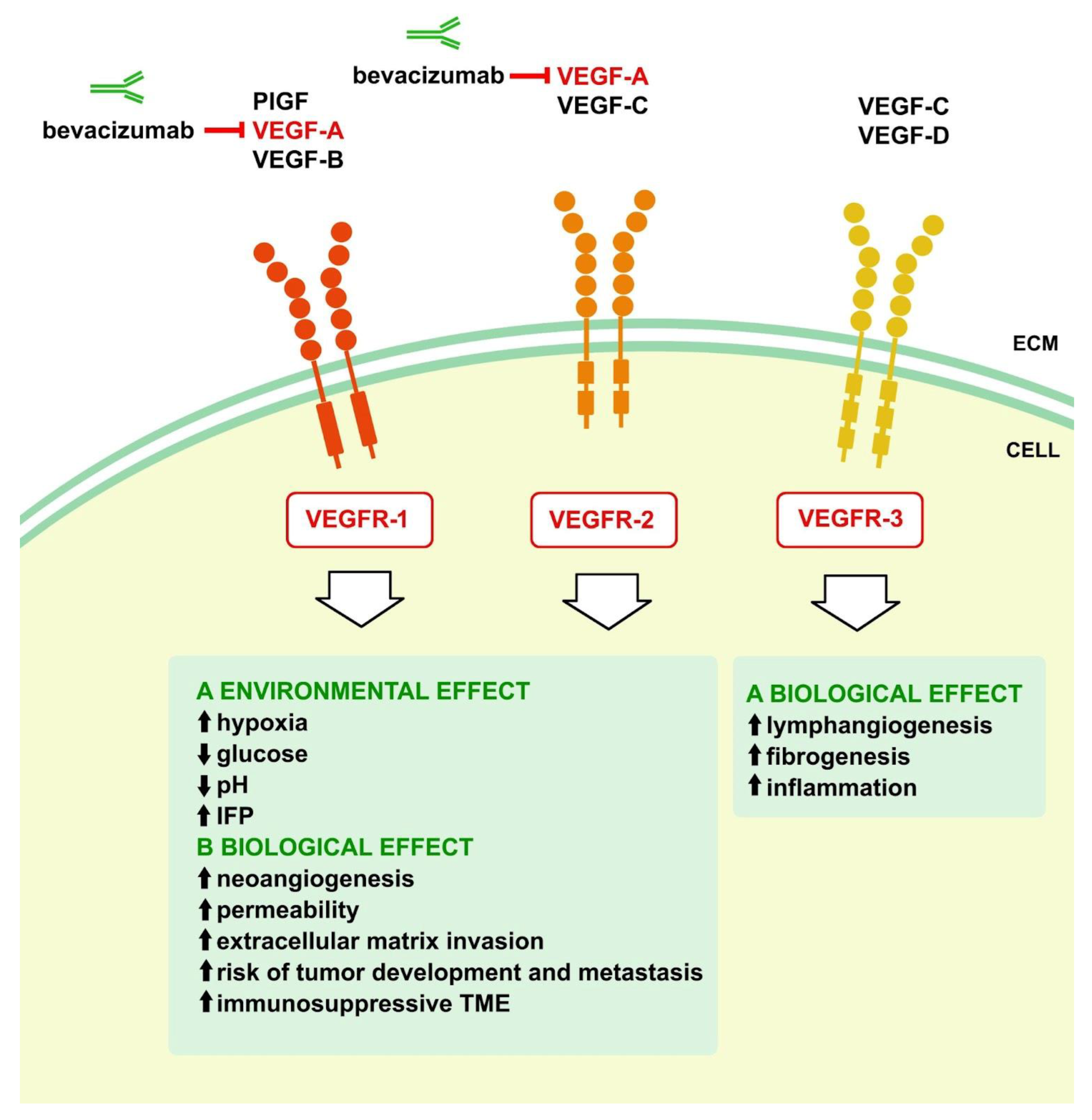
Solid tumors consist of neoplastic cells and the surrounding stromal cells, such as tumor-associated fibroblasts, endothelial cells, pericytes, and vascular smooth muscle cells, making them intricate structures [
20,
21,
22]. These cells play a crucial role in the growth and spread of tumors and their susceptibility to chemotherapy [
20,
21,
23,
24]. Furthermore, immune system cells, such as tumor-associated macrophages, are crucial in creating the inflammatory milieu within tumors. Cancer and stromal cells can release excessive proangiogenic substances, including VEGF, which in turn promote environmental, vascular biological, and immunological effects [
23,
25,
26] (
Figure 1).
Due to reduced blood flow, tumors frequently experience hypoxia, leading to the continuous excessive synthesis of VEGF and persistent development of aberrant tumor blood vessel patterns [
27]. The distribution of tumor blood vessels is dispersed, lacking a well-organized pattern of arterioles, capillaries, and venules. These vessels are characterized by variable and often enlarged diameters, irregular forms, leakage, and disordered blood flow [
27]. The blood arteries in tumors are also poorly developed, with few loosely connected pericytes and smooth muscle cells, and lack complete basement membranes. The combination of these features and insufficient lymphatic drainage frequently results in impaired blood circulation and elevated interstitial fluid pressure (IFP) within the tumors [
27]. Furthermore, VEGF triggers an immunosuppressive tumor microenvironment by impairing the maturation of dendritic cells, leading to the deactivation of cytotoxic T lymphocytes, promoting the production of regulatory T cells, tumor-associated macrophages, and myeloid-derived suppressor cells. In addition, VEGF increases the level of Programmed Death-1 in cytotoxic T lymphocytes and regulatory T cells [
26,
28].
4. Process of lymphangiogenesis
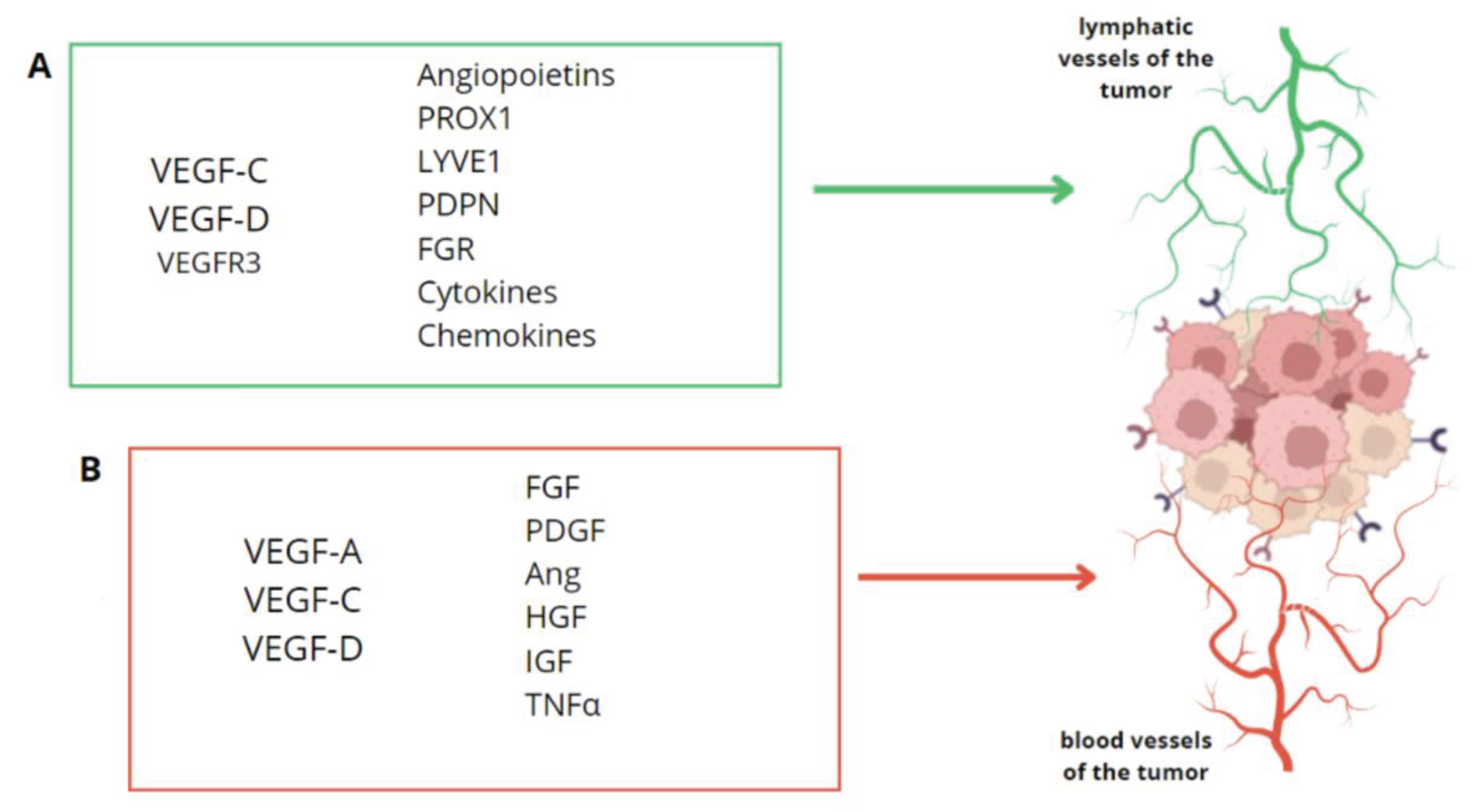
The creation of new lymphatic vessels from an existing system is known as lymphangiogenesis. This process depends on the interaction of the vascular endothelium with signaling molecules originating from serum and extracellular matrix (ECM) [
29]. In conditions such as inflammation, tumor growth, or tissue repair, pathogenic lymphatic vessels originate from pre-existing vessels [
30]. Lymphatic vessels are also responsible for cancer spread. Furthermore, they actively drive tumor cell recruitment to lymph nodes (LN), cancer stem cell survival and immunological regulation. Malignant tumors can produce factors that stimulate lymphatic vessel growth. In the study by Sopo et al., the significance of blood and lymphatic vessels in the prognosis of ovarian cancer was analyzed. The authors observed that the small size of lymph vessels was connected with 26% shorter 5-year survival. Moreover, the more lymphatic vessels were in the tumor, the more lymph node metastasis were present [
31]. It has been proven that lymphangiogenesis is primarily mediated by the growth factors VEGF-C and VEGF-D, as well as the particular receptor VEGFR-3. This phenomenon is also induced by the endothelial lymphatic vascular hyaluronate receptor 1 (LYVE1), the prospero homeobox-related transcription factor 1 (PROX1), and the membrane glycoprotein - podoplanin (PDPN). Surprisingly, the latter two components were discovered to be expressed only in lymphatic vessels but not in blood vessels, so that they may be more specific for the lymphatic system [
32]. Proving that hypoxia-induced regulation of lymphangiogenic factors made it possible to consider tumor hypoxia and lymphangiogenesis as closely related phenomena [
33]. Lymphangiogenesis is regulated not only by VEGF-C and VEGF-D, which act directly on lymphendothelial progenitor cells (LECs), but also by other cytokines that may act indirectly by promoting VEGF-C expression. These include, for example, angiopoietins, which act directly or familial fibroblast growth factor (FGR), pro-inflammatory cytokines such as IL-1β and hepatocyte growth factor, platelet-derived growth factors, insulin-like growth factor 1 and 2, adrenomedullin and endothelin-1. Therefore, VEGF-C stands out as a key regulator of lymphangiogenesis (
Figure 2).
As seen in ovarian cancer patients, tumor-associated lymphangiogenesis may increase metastasis and, as a consequence, decrease survival. Lymphatic capillaries at the tumor site let tumor cells metastasize from tumor foci to draining lymph nodes and enter the systemic circulation via nodal lymphatic vessels or blood vessels. The newly generated lymphatic capillaries promote the tumor's inflammatory milieu and signal lymphatic vessel formation in this multi-step process. Tumor-associated fibroblasts at the primary tumor site can secrete lymphangiogenic factors like VEGF-C, COX-2, HIF1 alpha chemokines like CCL21, CXCL12, which affect lymphatic endothelial cell permeability, adhesion molecule expression, migration to form nascent capillaries, and ECM remodeling [
34].
Despite the fact that there are more factors that contribute to lymphangiogenesis, the two most important are VEGF-C and VEGF-D. All of these factors act as lymphangiogenic growth factors [
29]. Overexpression of VEGF-C or VEGF-D has been found in studies describing tumor lymphangiogenesis to stimulate the development of tumor-associated lymphatic vessels, increase LN metastasis, and poor prognosis. VEGF-C or VEGF-D expression in primary tumors has been linked to LN metastasis in malignancies of the ovary, breast, colon, rectum, prostate, esophagus, stomach, lung, cervix, and endometrial [
32]. Sopo et al. conducted a study that compared the expression levels of VEGF-C and VEGF-D in primary and metastatic cancers. VEGF-C expression was shown to be much higher in primary tumors, while VEGF-D expression was found to be higher in metastatic tumors. (31) A similar conclusion comes from a study on endometrial cancer, which demonstrates that only VEGF-D expression increases with cancer stage [
35]. In well-differentiated colon cancer, there was equal overexpression of VEGF-C and VEGF-D. A significant association was discovered between the expression of lymphatic vessel growth factors and higher vessel density in the stroma surrounding the tumor, as well as a better prognosis [
36].
5. Bevacizumab - role in a treatment
In 2004, the US FDA approved bevacizumab, the first anti-angiogenic drug for the treatment of metastatic colon cancer [
6]. This new anti-angiogenic medicine is a humanized monoclonal antibody that has the ability to bind to all VEGF-A isoforms. This mechanism of bevacizumab prevents the activation of VEGF signaling pathways that would promote neovascularization. This prevents the formation of new blood vessels, which leads to the maturation of the tumor's vascularization, preventing its excessive growth. Moreover, the anti-angiogenic effect of bevacizumab has a synergistic effect with chemotherapy with carboplatin [
41,
42]. Since the first study on bevacizumab was published, numerous subsequent studies have been conducted describing the effects of bevacizumab in combination with cytotoxic chemotherapy in the treatment of solid tumors [
43,
44], including colorectal, lung, breast, or kidney cancer [
45,
46,
47]. Since then, the literature reports that over 3,500,000 patients with severe cancer have been treated with bevacizumab [
9].
Regarding the side effects of bevacizumab, the most common ones observed among patients are hypertension, weakness, abdominal pain and diarrhea [
7]. Due to the risk of hypertension in patients, constant monitoring of blood pressure is necessary during bevacizumab therapy. Some patients with kidney cancer have developed proteinuria during treatment with this drug [
9]. Studies have also described the occurrence of thromboembolism and an increased risk of hemorrhage in patients. In patients with colon or cervical cancer, there is a risk of gastrointestinal perforation. Most side effects occurred at the beginning of the treatment cycle when bevacizumab was administered together with chemotherapy. However, effects such as hypertension and proteinuria have been observed with long-term use of this drug [
48,
49].
The results of studies using bevacizumab in combination with other drugs and the immunomodulatory effect of bevacizumab seem to be promising. A phase III study evaluated bevacizumab in combination with atezolizumab in patients with non-small cell lung cancer. Stratified OS was 64% in the bevacizumab plus atezolizumab group and 48% in the atezolizumab plus chemotherapy group. Regarding side effects, most were grade 1/2 immunological in nature [
50]. To our knowledge, this is the first such study to evaluate the combination of bevacizumab with a drug that inhibits PD-L1/PD-1. It turned out that the addition of bevacizumab to atezolizumab is probably associated with a more favorable effect of atezolizumab at the level of PD-L1/PD-1 blockade in those patients with metastases who have not been previously treated with chemotherapy. A phase III study of the combination of bevacizumab with interferon α-2a in patients with metastatic renal cell carcinoma also showed an improvement in PFS in patients treated with this combination method compared to patients receiving interferon α-2a alone (10.2 months vs. 5.4 months). It seems interesting that the doses of interferon alfa in patients were reduced in order to avoid toxic side effects. The study results showed an improvement in PFS in those patients who received interferon alfa at a reduced dose [
51]. The study results indicate that immune modulation with bevacizumab acting at the level of VEGF inhibition may be important in solid tumors with increased VEGF expression or liver metastases. Moreover, it turned out that the use of bevacizumab in combination with interferon alfa may lead to a reduction in interferon doses, which may prevent long-term side effects associated with it.
Taking into account numerous studies on bevacizumab, its anti-angiogenic effect, effectiveness in cancer treatment, high safety profile and potential side effects, this drug still seems to be crucial in the treatment of patients with solid tumors.
6. Bevacizumab in the treatment of gynecological cancers
Each year, approximately 2 millions of women are diagnosed with gynecological and breast cancer all over the world [
52]. Taking into account the high number of patients diagnosed with those types of cancer, new drugs are still investigated. Bevacizumab is one of the drugs which seems to be a really helpful treatment option. The studies describing this direct VEGF inhibitor are described below.
6.1. Ovarian cancer
Bevacizumab was approved by EMA in December 2011 and in June 2018 by the FDA as a treatment for patients with stage III or IV epithelial ovarian cancer [
53]. Patients may undergo chemotherapy with carboplatin, paclitaxel, and bevacizumab, as well as surgical resection. Standard treatment of patients with ovarian cancer with bevacizumab lasts 15 months [
54,
55].
There are two most important studies describing the use of bevacizumab in the treatment of ovarian cancer, which guaranteed the approval of the FDA: GOG-218 and ICON7 [
54,
56]. GOG-218, a double-blind, placebo-controlled Phase III study, enrolled 1,873 women. Patients were randomly assigned to 3 treatments. In each group, patients received chemotherapy in the form of paclitaxel and carboplatin. The control group received chemotherapy plus a placebo in cycles 2 through 22. In the bevacizumab initiation group, patients received chemotherapy plus bevacizumab at a dose of 15 mg/kg b.w. in cycles 2 through 6 and placebo in cycles 7 through 22. In the third group, patients received bevacizumab in cycles 2 to 22. The median progression-free survival (PFS) in patients taking bevacizumab was 14.1 months in the group receiving bevacizumab continuously compared with PFS of 11.2 months in the group receiving bevacizumab at the beginning and 10.3 months in the control group [
54].
ICON7 was an international, open-label, randomized Phase III trial that demonstrated improved PFS when bevacizumab was added to chemotherapy in patients with high-risk early-stage or advanced-stage ovarian cancer. 1,528 women in the study were randomized to chemotherapy alone (n=764) or chemotherapy plus bevacizumab 7.5 mg per kg intravenously every 3 weeks for 12 3-week maintenance cycles (n=764). Overall survival was 45.4 months in the chemotherapy plus bevacizumab group and 44.6 months in the chemotherapy alone group. However, in the group of 502 patients with poor prognosis, the mean PFS was 34.5 months with standard chemotherapy and 36.3 months with chemotherapy in combination with bevacizumab. Although the use of bevacizumab did not increase the mean PFS in patients in the entire group, an increase in PFS was observed in patients with a poor prognosis. The study results emphasize a significant relationship between a higher stage of disease and a more beneficial effect of bevacizumab. There is a group of women who will not benefit from treatment with bevacizumab - these are mainly patients in FIGO stage I/II. Given the effects of bevacizumab on the tumor microenvironment, it appears that bevacizumab will be effective in residual physical tumor that produces VEGF [
56].
Interestingly, bevacizumab has shown synergy with other treatments in patients with ovarian cancer in many studies. Studies indicate that bevacizumab added to carboplatin in combination with paclitaxel contributed to the prolongation of PFS [
54,
56]. The combination of bevacizumab with PARP inhibitors is also interesting. In 2023, the final results of the randomized, double-blind, phase III trial PAOLA-1 were published. The aim of the study was to evaluate the addition of olaparib to bevacizumab in patients with newly diagnosed ovarian cancer. 537 patients were assigned to olaparib (300 mg twice daily for up to 24 months) plus bevacizumab (15 mg/kg every 3 weeks for 15 months) and 269 patients were assigned to placebo plus bevacizumab. Median OS was 56.5 months in the olaparib plus bevacizumab group and 51.6 months in the placebo group. Moreover, benefits have been demonstrated in patients with BRCAm knockout or homologous recombination deficiency genes (HRD). Significant improvement in 5-year OS was demonstrated in patients with HRD-positive ovarian cancer (65.5%) compared to patients with HRD-negative ovarian cancer (48.4%) [
57]. Taking into account the fact that the addition of olaparib to bevacizumab resulted in prolonged OS in patients, there is a need to study biomarkers in patients with ovarian cancer to determine an appropriate treatment regimen.
Another study was the open-label randomized phase III trial AGO-OVAR 17 BOOS/GINECO OV118/ENGOT Ov-15, the aim of which was to compare standard of 15 dosages and extended duration of bevacizumab treatment with chemotherapy in 927 patients with newly diagnosed stage IIB-IV ovarian cancer. The patients first underwent cytoreductive surgery with 6 cycles of chemotherapy in the form of paclitaxel at a dose of 175 mg/m2 with carboplatin area under the curve 5 times every 3 weeks and bevacizumab at a dose of 15 mg/kg once every 3 weeks. Patients received bevacizumab for 15 or 30 months. The median PFS was 24.2 months in patients treated with the standard duration and 26.0 months in patients treated with the extended duration. Regarding OS, no difference was found between patient groups [
58]. The study results showed that the extension of bevacizumab treatment in patients with ovarian cancer did not significantly affect PFS and OS.
In 2023, the results of a study assessing the effectiveness and safety of the combination of mirvetuximab soravtensine and bevacizumab in patients with platinum-resistant ovarian cancer were published, the aim of which was to. 94 patients were qualified to the group receiving mirvetuximab soravtensine at a dose of 6 mg/kg adjusted ideal body weight and bevacizumab at a dose of 15 mg/kg intravenously once every 3 weeks. It seems significant that 52% of patients had previously received ≥3 therapies, 59% had previously received bevacizumab, and 27% had previously received PARP therapy. The median PFS was 8.2 months and the median duration of response (DOR) was 9.7 months. Given the good results with this combination, treatment with bevacizumab and mirvetuximab soravtensine appears to be promising. These results were favorable regardless of the level of folate receptor alpha (FRα) expression or previous treatment with bevacizumab [
59].
Although bevacizumab, as an antiangiogenic drug, has been described in many studies, its use in patients with ovarian cancer is still controversial. This is evidenced by the results of the GOG-218 study, in which a significant improvement in PFS was observed in the group of patients with stage IV cancer. In 2022, You et al. conducted an external validation study in the GOG-218 study using the KELIM scale to determine factors influencing bevacizumab treatment. To assess the benefits using the KELIM scale, it is important to measure at least three CA125 values during the first three cycles of chemotherapy. 1662 patients with ≥ CA125 marker values were examined using the scale. The study showed that PFS and OS were higher in high-risk patients with poorly chemosensitive disease [
60]. All clinical trials with bevacizumab in ovarian cancer are presented in the
Table 1. When qualifying a patient for bevacizumab treatment, it is necessary to take into account not only the risk of disease progression and response to previous treatment, but also the value of markers that may be important.
6.2. Cervical cancer
Thanks to constant cytological tests and the increase in vaccinations against the human papillomavirus (HPV), during recent years a decrease in the number of cervical cancer cases has been observed. However, despite available diagnostic methods, 500,000 women worldwide will be diagnosed with cervical cancer each year [
61,
62]. Moreover, radical surgery and radiotherapy, to which patients in the early stages of cervical cancer respond well, do not provide such good treatment results in women with metastases or recurrent disease [
63]. Chronic HPV infection may lead to inactivation of cancer suppressor genes, including: p53 and pRb. This causes the accumulation of HIF-1 protein, which in turn induces an increase in VEGF expression by the tumor tissue. Therefore, bevacizumab, by inhibiting VEGF, has been used in the treatment of cervical cancer [
64,
65].
The GOG 227C, the phase II trial from 2009, assessed the treatment of 46 patients with recurrent squamous cell cervical cancer with bevacizumab at a single dose of 15 mg/kg body weight. every 3 weeks. The median overall survival was 7.3 months, and the median progression-free survival was 3.4 months. In the study there was no control group, but the results were compared with other studies [
66,
67,
68,
69,
70,
71]. 24% of patients treated with bevacizumab did not progress after 6 months on protocol, which was the highest score in all analyzed researches [
72].
In a 2014 study, Tewari et al (GOG 240 study) evaluated the effectiveness of combination therapy with platinum-free chemotherapy and bevacizumab in 452 patients with recurrent (325 patients), persistent (51 patients), or metastatic (76 patients) cervical cancer. Patients were randomly assigned to receive chemotherapy with bevacizumab (227 women) or without (225 women). Chemotherapy as cisplatin and paclitaxel, or topotecan and paclitaxel was administered in 1 day. Cycles were repeated every 21 days. The study results showed that treatment with chemotherapy together with bevacizumab was associated with increased overall survival of patients (17.0 months) compared to treatment with chemotherapy alone (13.3 months) [
73].
GOG 240 is a randomized, controlled, open-label, phase III trial that assessed the effectiveness of bevacizumab in 452 patients with advanced cervical cancer. Patients were assigned in a 1:1:1:1 ratio to receive chemotherapy with cisplatin plus paclitaxel or topotecan plus paclitaxel with or without bevacizumab administered intravenously at a dose of 15 mg/kg on day 1 in 21-day cycles. The median OS was 16.8 months in the chemotherapy plus bevacizumab group and 13.3 months in the chemotherapy alone group. OS after progression was 8.4 months in patients in the group receiving chemotherapy plus bevacizumab and 7.1 months in the group receiving chemotherapy alone. The study results confirmed the effectiveness of bevacizumab in patients with advanced cervical cancer [
74].
The results of available studies unanimously indicate the beneficial effects of bevacizumab in patients with cervical cancer. For the moment, bevacizumab has been approved as an antiangiogenic therapy in patients with advanced tumors. Given the low effectiveness of bevacizumab in early stages of cancer, it does not seem necessary to perform it in patients with benign tumors. Moreover, it may be beneficial to combine bevacizumab with other new drugs. The BEATcc trial is currently being conducted - a randomized, open-label, multicenter phase III clinical trial. Its aim is to determine whether adding atezolizumab - the immune checkpoint inhibitor - to bevacizumab will improve survival in patients with cervical cancer [
75]. The results of KEYNOTE-826 - a multicentre, randomized, phase III trial, seem to be also interesting, in which the authors assessed the effectiveness and improvement of quality of life after adding pembrolizumab to chemotherapy with or without bevacizumab. This study was conducted on 617 patients with persistent, recurrent or metastatic cervical cancer not previously treated with systemic chemotherapy. Patients were assigned to receive pembrolizumab 200 mg or placebo intravenously every 3 weeks and chemotherapy (paclitaxel or carboplatin) with or without bevacizumab 15 mg/kg every 3 weeks intravenously. GHS quality of life improved in 122 of 290 patients treated in the pembrolizumab group compared to 85 of 297 patients treated in the placebo group. The study results indicate an improvement in the quality of life after adding pembrolizumab to treatment, which indicates the benefits of treatment with immunotherapy [
76]. All studies describing clinical trials with bevacizumab in the cervical cancer are presented in the
Table 2.
6.3. Endometrial cancer
Endometrial cancer continues to be the most common cancer among women in the developed countries, and its incidence is increasing each year compared with other gynecological cancers, which are decreasing in number [
77]. Chemotherapeutics, such as paclitaxel and carboplatin, are used as initial treatment for advanced endometrial cancer. When it comes to adjuvant therapy, it mainly includes surgical, hormonal and chemotherapy methods used depending on the histological grade and stage of the tumor [
78]. Therefore, antiangiogenic drugs seemed to be an excellent option, but did it turn out to be true?
In 2007, Wright et al. retrospectively analyzed 11 patients with recurrent endometrial cancer treated with bevacizumab. Among the group of patients, 9 women were patients with epithelial endometrial cancer, and 2 were patients with globular cell sarcoma. 3 patients were in stage I, 1 patient in stage II, 2 patients in stage III and 5 patients in stage IV. All patients were previously treated with 3 chemotherapy regimens and received bevacizumab in combination with a chemotherapy agent. The median progression-free period for all patients was 5.4 months, including 8.7 months for 5 patients. Unfortunately, the study was performed on a small group of patients and there was no control group, therefore it is difficult to compare the results [
79].
Already in 2011, Aghajanian et al. conducted a phase II study to determine the tolerability of bevacizumab in the treatment of recurrent or persistent endometrial cancer. 52 patients were treated with bevacizumab at a dose of 15 mg/kg administered intravenously every 3 weeks. In 21 patients (40.4%), PFS was at least 6 months. The median survival time was 4.2 months and the median overall survival time was 10.5 months. At that stage, it was recognized that due to prolonged PFS, this drug is worthy of further investigation in endometrial cancer [
80].
Alvarez et al. conducted a phase II study, whose aim was to evaluate the combination of temsirolimus, an inhibitor of mTOR, and bevacizumab in patients with recurrent or persistent endometrial cancer. 53 patients were enrolled in the study, of which 40 patients had previously received treatment with one chemotherapy regimen, 9 patients with two regimens, and 20 patients had been previously treated with radiotherapy. Patients received treatment with bevacizumab 10 mg/kg every other week and temsirolimus 25 mg intravenously weekly until disease progression. The median progression-free survival was 5.6 months and the median overall survival was 16.9 months. In this study no control group was included. Moreover, toxicity seemed to be a great issue, because it was present in nearly 40% of patients [
81].
Rubinstein et al., in a retrospective study, showed moderate effectiveness of bevacizumab in monotherapy in the treatment of advanced endometrial cancer. 101 patients were included in the study, including 46% of cases in the stage IV according to FIGO and 24% in stage III. In addition, patients had differential histology, the most common of which was serous histology in 44%. Patients were assigned to appropriate cohorts based on prior treatment: cohort I included 14 patients who had previously received 1 line of treatment, cohort II included 29 patients who had previously received 2 lines of treatment, cohort III included patients who had previously received 3 lines of treatment, and cohort IV included patients who had received ≥ 4 lines of treatment. What is interesting, the group who received ≥ 4 lines of treatment had the highest median PFS: 4.9 months compared to cohort I: 3.5 months, cohort II: 2.6 months, cohort III 3.5 months. Therefore, the authors of the study opt that bevacizumab, due to an excellent result in the group after other lines of treatment, should be considered as a palliative treatment [
82].
However, no randomized study reported prolonged PFS. In 2018, the first results of the GOG 209 trial were published - a randomized phase II trial. The study included 349 patients with chemotherapy-naïve stage III/IVA and IVB or recurrent endometrial cancer. Patients were randomly assigned to receive paclitaxel and carboplatin with bevacizumab, carboplatin with temsirolimus, or ixabepilone and carboplatin with bevacizumab. The study results did not show an increase in PFS in the group of patients treated with bevacizumab compared to the group of patients treated with carboplatin plus temsirolimus and the group of patients treated with ixabepilone and carboplatin. The hazard ratios for the groups were 0.81, 1.22 and 0.87, respectively [
83].
For the moment, no significant profit of bevacizumab in the endometrial cancer treatment was observed, therefore this drug is not used in daily clinical practice. It is also probably connected with the fact that generally endometrial cancer is diagnosed in not advanced stages and surgery is the most effective treatment option, so the potential benefit from antiangiogenic treatment might be seen only in advanced/recurrent cases. A meta-analysis from 2020 showed that conventional chemotherapy in combination with bevacizumab improved median PFS and OS in patients with advanced or recurrent endometrial cancer [
84]. All clinical trials with bevacizumab in the endometrial cancer are analyzed in the
Table 3.
7. Primary ovarian cancer. The presence and location of residual disease.
Already, in 2011 bevacizumab was approved by EMA in the treatment of patients with epithelial ovarian, fallopian tube or primary peritoneal cancer in combination with carboplatin and paclitaxel for stage III or IV disease after initial surgical resection by the FDA [
8]. As it was proven, this antiangiogenic drug did not increase overall survival (OS) in the whole population of cancer patients, but the difference in OS was observed in the poor-prognosis patients [
56,
85]. The definition of high-risk patients, meaning the patients with the worst prognosis, in ICON7 and GOG-128 studies have some differences. In the ICON7 study, the definition was as follows: stage IV or inoperable stage III disease, or suboptimally debulked (> 1 cm) stage III ovarian cancer [
56]. The GOG-218 study included patients in stage III with optimal (=<1 cm) and suboptimal (>1 cm) cytoreduction and stage IV [
85]. However, in the aim to facilitate the comparison of both studies, in the GOG-218 study some additional exploratory analyses were done, and as a consequence both studies had the same result: no OS benefit and PFS benefit [
56,
85]. The question is why bevacizumab brings the best results when it is used in patients with the worst prognosis? In general, the best prognosis for patients with ovarian cancer is ensured due to complete surgical resection. The fact that the optimal residual disease is the best alternative in the cases when complete surgical resection is not possible is also reported by many studies. But what does it mean that resection is optimal? The definition says that the largest disease diameter is less than 1 cm, independent of the total volume of disease [
86,
87,
88,
89,
90,
91,
92,
93]. The study of Manning-Geist et al. showed that the median progression-free survival for the following groups: with complete resection, with ≤1 cm residual disease in a single anatomic location, ≤1 cm residual disease in a multiple anatomic location and in the group suboptimally debulked were accordingly 14, 12, 10 and 6 months. The median overall survival in the above-mentioned groups were respectively: 58, 37, 26 and 33 months [
94]. Taking into account aforementioned information, in the cases of ovarian cancer the maximum surgical effort is required to achieve the lowest number of visible lesions. The minimal residual disease is clinically undetectable disease, a small number of cancer cells left in the body after treatment. Those cells are not connected with vessels and by vessels, and this is one of the reasons why they are less sensitive to anti-VEGF therapy. No vessels, no drug’s action point and in the consequence - no effect of bevacizumab treatment. Especially compared to the lesions which are visible and connected by vessels – macroscopic disease [
13,
95,
96]. This is the reason why bevacizumab may not change OS or PFS in the non-high risk patients. However, in patients with no visible lesions, bevacizumab is used as angioprevention, meaning interruption the creation of new blood vessels in tumors, whose cell foci are in an indolent state. In the study by Albini et al. bevacizumab is one of the treatments which should be considered in patients who have achieved cancer remission. In this regard, the main aim of medication is to prevent disease recurrences [
97]. It is described as the suppression of the “soil”, meaning microenvironment, so that the “seed” (microscopic metastases) will not survive [
98,
99].
To our best knowledge, none of the studies analyzed the impact of the location of residual disease of ovarian cancer on bevacizumab treatment. According to the mechanism of bevacizumab [
7,
9,
41,
42,
43,
44,
45,
46,
47,
48,
49], we know that this treatment acts by binding VEGF-A present in blood vessels, without binding VEGF-C and VEGF-D present in lymphatic vessels [
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40]. In this context, we would like to divide the presence of residual disease into nodal and not-nodal types. Potentially, bevacizumab should act well in the cases of not-nodal residual disease. Why? This is more probable that in the process of spreading the tumor, the new blood vessels will be formed. Blood vessels, which are more susceptible to the action of bevacizumab due to the presence of VEGF-A receptors. Compared to the nodal residual disease, in which the action of bevacizumab will be limited, due to the presence of lymphangiogenesis regulated predominantly by VEGF-C and VEGF-D.
8. Second line treatment. Recurrences.
There are three studies analyzing the efficacy of bevacizumab in the second line treatment: OCEANS, AURELIA and GOG213. First of aforementioned, OCEANS analyzed the efficacy of bevacizumab in the dose of 15 mg/kg with gemcitabine and carboplatin in platinum-sensitive recurrent ovarian, primary peritoneal, or fallopian tube cancer. Median PFS was 12.4 in the bevacizumab-treated group vs 8.4 months in the control group [
100]. The study did not describe the location of the tumor recurrences, therefore we cannot state if this location has an impact on the bevacizumab treatment efficacy. The same conclusion relates to the AURELIA study, which analyzed the effectiveness of bevacizumab (10 mg/kg every 2 weeks or 15 mg/kg every 3 weeks) combined with chemotherapy in platinum-resistant ovarian cancer. Median PFS was almost two times longer in the bevacizumab group compared to the control group, 6.7 vs 3.4 months, respectively [
101]. GOG213 study analyzed the role of bevacizumab in the dosage of 15 mg/kg every 3 weeks in patients with recurrent platinum-sensitive ovarian cancer, who performed secondary surgical cytoreduction. The study showed the median OS in the bevacizumab treated group was 42.2 months and 37.3 in the chemotherapy alone group [
102]. The question if it is better to use bevacizumab in the first or in the second line was asked by many researchers, including Sznurkowski. In his review he suggested that bevacizumab should be preserved for the second line maintenance therapy, because it is more efficacious than in the first line treatment [
103]. It is consistent with the results of a meta-analysis, which implies that, compared to first line treatment, in second line treatment bevacizumab shows significant survival benefit in PFS and OS in high-risk patients and patients with recurrences [
104].
From our perspective, there is a gap of knowledge in understanding why bevacizumab acts more successfully in patients with recurrences. At the beginning, we should recall the “traditional” recurrence patterns in ovarian cancer in the bevacizumab-non-treated group. Paik et al. conducted a study on the group of 303 patients with the epithelial ovarian cancer with no gross residual disease analyzed patterns of recurrence after primary debulking surgery. Firstly, the location of recurrence was analyzed. In the stages III-IV, distant recurrences were more frequent that locoregional recurrences - 83,3% vs 16,7%, accordingly. Secondly, discrete recurrences were more common than diffuse recurrences. The definition of diffuse carcinomatosis was as follows: nodules larger than four centimeters or constituting plaques on the peritoneum or mesentery. In stages III-IV, 39 patients (65%) had discrete recurrences and 21 (35%) had diffuse recurrences [
105]. So, what are the consequences of bevacizumab use in the context of recurrences?
Taking into consideration the mechanism of bevacizumab, meaning inhibition of VEGF-A without affecting VEGF-C and VEGF-D, it seems to be clear that some differences in the patterns of recurrences, especially in the lymphatic system, may occur. The study by Petrillo et al. aimed to evaluate the timing and pattern of relapse in patients with advanced ovarian cancer treated in the first line by standard chemotherapy with or without bevacizumab. The research was performed on the group of 74 patients in the bevacizumab group and 148 patients in the chemotherapy alone group. What is interesting, even if the PFS was longer in the bevacizumab group, the recurrence pattern was different, especially in the context of lymph nodes. What seems very attaching, patients treated with bevacizumab were more likely to relapse in lymph nodes – in the bevacizumab group metastases were present in 51.3% of patients compared to the control group: 31.1% of patients (p=0.004). Moreover, peritoneal recurrence described as diffuse disease was also more frequent in patients treated in the first line by bevacizumab (96.8%) compared to the chemotherapy group [
106]. The results of the study were consistent with previous study by conducted Dao et al. which showed, that patients treated with bevacizumab in the first line had higher probability to recur in extra-visceral sites (p=0.04) or in lymph nodes, especially those located extraperitoneally (p=0.0002) [
107]. In the comparison, the study by Kim et al. performed on the group of 52 patients with epithelial ovarian cancer treated by platinum-based doublet chemotherapy with bevacizumab as a second-line treatment and 104 patients in the control group only treated by chemotherapy did not show the same dependence. Bevacizumab treated patients had lower probability to develop disease recurrence in retroperitoneal lymph nodes (p=0.001), pelvis (p=0.003) and abdomen (p=0.001), but the same for distant metastases (p=0.32) compared to the control group. The authors of the study mentioned the heterogeneity in study design, study populations, and disease settings as the reason for the differences [
108]. To our best knowledge, this is the only study in which the lower frequency of nodal metastases was observed.
The next question which appears during the analysis of the recurrence patterns after bevacizumab treatment is the following: how this process runs in other types of neoplasm. The systematic review and meta-analysis of 17 articles describing the recurrence patterns in high-grade glioma showed that in the bevacizumab-treated patients the non-local recurrences were more frequent than in the standard-treated patients [
109]. However, the results of the studies are not consistent, because the other two studies suggest that in glioma local progression after bevacizumab treatment is more common [
110,
111]. Anyway the recurrence patterns in glioma, in our opinion, do not help us in understanding the ways of recurrence in ovarian cancer. How about other gynecological cancers? The analysis of the bevacizumab recurrence patterns in persistent recurrent or metastatic cervical cancer treated by bevacizumab in combination with cisplatin and paclitaxel showed that loco-regional recurrence was more frequent [
112]. To our best knowledge, there are no studies describing the patterns of recurrence after bevacizumab treatment in endometrial cancer, which indicates the lack of knowledge in this area. From our point of view, the recurrence patterns after bevacizumab treatment in gliomas do not have influence on the understanding of the recurrence patterns after bevacizumab treatment in ovarian cancer or other gynecologic neoplasms, because even without the antiangiogenic treatment those types of cancer metastasize differently.
9. Discussion and conclusions
Bevacizumab, as an antiangiogenic drug, was created to interrupt angiogenesis by binding to VEGF-A isoform. As a consequence, a reduction in microvascular growth of tumor blood vessels is observed, which limits the blood supply to tumor tissues and ensuing tumor growth [
7,
9,
41,
42,
43,
44,
45,
46,
47,
48,
49. However, the angiogenesis is not the only one process, which leads to the tumor growth and metastases. Equally important is lymphangiogenesis, which is also dependent on the VEGF family. According to the Karaman et al. the process of lymphangiogenesis is mediated by other isoforms of VEGF family: VEGF-C and VEGF-D [
32]. Those whose expression is elevated especially in lymph nodes, are not affected by the bevacizumab activity [
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40,
41,
42]. In consequence, even if the creation of new blood vessels is interrupted by bevacizumab, the presence of lymphatic vessels is not reduced. Therefore, our first hypothesis is born: if the lymph nodes in the primary ovarian cancers are metastatic, the outcome of bevacizumab treatment is worsened. The second one is connected with the first one, but it concerns second-line treatment. In our opinion, bevacizumab is going to act in a weakened manner if the recurrence will be in lymph nodes compared to local recurrences. To this day, there are no studies analyzing the relationship between the presence of metastases in lymph nodes in primary ovarian cancer and the outcome of bevacizumab treatment. The bevacizumab approval in the treatment of patients with ovarian cancer refers to patients in stage III or IV disease after initial surgical resection. It is a very heterogeneous group, which may have both positive or negative lymph nodes. Moreover, there is a gap of knowledge regarding the differences of bevacizumab efficacy in the second line treatment depending on the location of recurrence locations. Our third hypothesis, which is partially confirmed, is the following: patients treated by bevacizumab are more likely to have recurrences in lymph nodes. Two out of free studies, performed by Petrillo et al. and Dao et al., showed that the patterns of recurrences are different when patients are treated by bevacizumab [
106,
107]. More precisely, in the Petrillo et al. study, in the bevacizumab treated group metastases in lymph nodes were present in 51.3% of patients and in the control group they were present in 31.1% of patients [
106]. Those conclusions were not in accordance with Kim et al., who observed that bevacizumab treated patients had lower probability to develop disease recurrence in retroperitoneal lymph nodes [
108].
Author Contributions
Conceptualization, K.Ż. and M.B.; methodology; K.Ż, M.B., M.S.; software, K.Ż, M.S..; validation, K.Ż, M.B., M.O-L., and R.T..; formal analysis, K.Ż., M.S., I.S.;; investigation, K.Ż., M.S., I.S.; resources, K.Ż., M.S., I.S.; data curation, K.Ż., M.S., I.S.; writing—original draft preparation, K.Ż., M.S., I.S.; writing—review and editing, K.Ż., M.S., I.S., M.B., M.O-L.; visualization, K.Ż., M.B..; supervision, M.B, R.T. and M.O-L..; project administration, K.Ż., M.B. All authors have read and agreed to the published version of the manuscript.
References
- Ide, A.G.; Baker, N.H.; Warren, S.L. Vascularization of the Brown-Pearce rabbit epithelioma transplant as seen in the transparent ear chamber. Am. J. Roentg. 1939, 32, 891–899. [Google Scholar]
- Folkman, J. Tumor angiogenesis: Therapeutic implications. N. Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Senger, D.R.; Galli, S.J.; Dvorak, A.M.; Perruzzi, C.A.; Harvey, V.S.; Dvorak, H.F. Tumor Cells Secrete a Vascular Permeability Factor That Promotes Accumulation of Ascites Fluid. Science 1983, 219, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Henzel, W.J. Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem. Biophys. Res. Commun. 1989, 161, 851–858. [Google Scholar] [CrossRef]
- Kim, K.J.; Li, B.; Winer, J.; Armanini, M.; Gillett, N.; Phillips, H.S.; Ferrara, N. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 1993, 362, 841–844. [Google Scholar] [CrossRef] [PubMed]
- Drug Approval Package: Avastin (Bevacizum) NDA #125085 [Internet]. [cited 2024 Jan 26]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2004/STN-125085_Avastin.cfm?fbclid=IwAR0pRz9CF2adjMNJX8bifA9O43GSDWVB2zXqUpeE4DN9hFBwZDcXoyAEjl8.
- Avastin | European Medicines Agency [Internet]. [cited 2024 Jan 26]. Available from: https://www.ema.europa.eu/en/medicines/human/EPAR/avastin.
- Research C for DE and. FDA approves bevacizumab in combination with chemotherapy for ovarian cancer. FDA [Internet]. 2019 Sep 2 [cited 2023 Dec 9]; Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-bevacizumab-combination-chemotherapy-ovarian-cancer.
- Garcia, J.; Hurwitz, H.I.; Sandler, A.B.; Miles, D.; Coleman, R.L.; Deurloo, R.; Chinot, O.L. Bevacizumab (Avastin®) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat. Rev. 2020, 86, 102017. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Hillan, K.J.; Gerber, H.-P.; Novotny, W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat. Rev. Drug Discov. 2004, 3, 391–400. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Xu, L.; Kanasaki, K.; Kitada, M.; Koya, D. Diabetic angiopathy and angiogenic defects. Fibrogenesis Tissue Repair 2012, 5, 13–13. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Li, Y.L.; Zhao, H.; Ren, X.B. Relationship of VEGF/VEGFR with immune and cancer cells: staggering or forward? Cancer Biol Med. 2016, 13, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef] [PubMed]
- A Foekens, J.; A Peters, H.; Grebenchtchikov, N.; Look, M.P.; Gelder, M.E.M.-V.; Geurts-Moespot, A.; Van Der Kwast, T.H.; Sweep, C.G.; Klijn, J.G. High tumor levels of vascular endothelial growth factor predict poor response to systemic therapy in advanced breast cancer. Cancer Res. 2001, 61, 5407–5414. [Google Scholar] [PubMed]
- Gao, X.; Chen, W.; Li, J.; Shen, C.; Zhou, P.; Che, X.; Li, X.; Xie, R. The protective effect of alpha-lipoic acid against brain ischemia and reperfusion injury via mTOR signaling pathway in rats. Neurosci. Lett. 2018, 671, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Guetz, G.D.; Uzzan, B.; Nicolas, P.; Cucherat, M.; Morere, J.-F.; Benamouzig, R.; Breau, J.-L.; Perret, G.-Y. Microvessel density and VEGF expression are prognostic factors in colorectal cancer. Meta-analysis of the literature. Br. J. Cancer 2006, 94, 1823–1832. [Google Scholar] [CrossRef] [PubMed]
- Paley, P.J.; Staskus, K.A.; Gebhard, K.; Mohanraj, D.; Twiggs, L.B.; Carson, L.F.; Ramakrishnan, S. Vascular endothelial growth factor expression in early stage ovarian carcinoma. Cancer 1997, 80, 98–106. [Google Scholar] [CrossRef]
- Cho, A.; Howell, V.M.; Colvin, E.K. The Extracellular Matrix in Epithelial Ovarian Cancer – A Piece of a Puzzle. Front. Oncol. 2015, 5, 245. [Google Scholar] [CrossRef] [PubMed]
- Balduit, A.; Agostinis, C.; Mangogna, A.; Maggi, V.; Zito, G.; Romano, F.; Romano, A.; Ceccherini, R.; Grassi, G.; Bonin, S.; et al. The Extracellular Matrix Influences Ovarian Carcinoma Cells’ Sensitivity to Cisplatinum: A First Step towards Personalized Medicine. Cancers 2020, 12, 1175. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, Y.; Yang, J.; Zhao, X.; Wei, X. Tumor Microenvironment in Ovarian Cancer: Function and Therapeutic Strategy. Front. Cell Dev. Biol. 2020, 8, 758. [Google Scholar] [CrossRef]
- Ostrowska-Lesko, M.; Rajtak, A.; Moreno-Bueno, G.; Bobinski, M. Scientific and clinical relevance of non-cellular tumor microenvironment components in ovarian cancer chemotherapy resistance. Biochim. et Biophys. Acta (BBA) - Rev. Cancer 2023, 1879, 189036. [Google Scholar] [CrossRef]
- Jurj, A.; Ionescu, C.; Berindan-Neagoe, I.; Braicu, C. The extracellular matrix alteration, implication in modulation of drug resistance mechanism: friends or foes? J Exp Clin Cancer Res. 2022, 41, 276. [Google Scholar] [CrossRef]
- Ribatti, D. Immunosuppressive effects of vascular endothelial growth factor (Review). Oncol. Lett. 2022, 24, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tamura, R.; Tanaka, T.; Akasaki, Y.; Murayama, Y.; Yoshida, K.; Sasaki, H. The role of vascular endothelial growth factor in the hypoxic and immunosuppressive tumor microenvironment: perspectives for therapeutic implications. Med. Oncol. 2019, 37, 2. [Google Scholar] [CrossRef]
- Saharinen, P.; Eklund, L.; Pulkki, K.; Bono, P.; Alitalo, K. VEGF and angiopoietin signaling in tumor angiogenesis and metastasis. Trends Mol. Med. 2011, 17, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Gabrilovich, D.; Ishida, T.; Oyama, T.; Ran, S.; Kravtsov, V.; Nadaf, S.; et al. Vascular endothelial growth factor inhibits the development of dendritic cells and dramatically affects the differentiation of multiple hematopoietic lineages in vivo. Blood 1998, 92, 4150–4166. [Google Scholar] [CrossRef]
- McCOLL, B.K.; Stacker, S.A.; Achen, M.G. Molecular regulation of the VEGF family – inducers of angiogenesis and lymphangiogenesis. APMIS 2004, 112, 463–480. [Google Scholar] [CrossRef] [PubMed]
- Sáinz-Jaspeado, M.; Claesson-Welsh, L. Cytokines regulating lymphangiogenesis. Curr. Opin. Immunol. 2018, 53, 58–63. [Google Scholar] [CrossRef]
- Sopo, M.; Anttila, M.; Muukkonen, O.-T.; Ylä-Herttuala, S.; Kosma, V.-M.; Keski-Nisula, L.; Sallinen, H. Microvessels in Epithelial Ovarian Tumors: High Microvessel Density Is a Significant Feature of Malignant Ovarian Tumors. Anticancer. Res. 2020, 40, 6923–6931. [Google Scholar] [CrossRef]
- Karaman, S.; Detmar, M. Mechanisms of lymphatic metastasis. J. Clin. Investig. 2014, 124, 922–928. [Google Scholar] [CrossRef]
- Morfoisse, F.; Renaud, E.; Hantelys, F.; Prats, A.-C.; Garmy-Susini, B. Role of hypoxia and vascular endothelial growth factors in lymphangiogenesis. Mol. Cell. Oncol. 2015, 2, e1024821. [Google Scholar] [CrossRef]
- Pal, S.; Bhowmick, S.; Sharma, A.; Sierra-Fonseca, J.A.; Mondal, S.; Afolabi, F.; Roy, D. Lymphatic vasculature in ovarian cancer. Biochim. et Biophys. Acta (BBA) - Rev. Cancer 2023, 1878, 188950. [Google Scholar] [CrossRef] [PubMed]
- Oplawski, M.; Dziobek, K.; Zmarzły, N.; Grabarek, B.; Halski, T.; Januszyk, P.; Kuś-Kierach, A.; Adwent, I.; Dąbruś, D.; Kiełbasiński, K.; et al. Expression Profile of VEGF-C, VEGF-D, and VEGFR-3 in Different Grades of Endometrial Cancer. Curr. Pharm. Biotechnol. 2019, 20, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Szajewski, M.; Kruszewski, W.J.; Lakomy, J.; Ciesielski, M.; Kawecki, K.; Jankun, J.; Buczek, T.; Szefel, J. VEGF-C and VEGF-D overexpression is more common in left-sided and well-differentiated colon adenocarcinoma. Oncol. Rep. 2013, 31, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Wada, I.; Nakao, S.; Yamaguchi, M.; Kaizu, Y.; Arima, M.; Sawa, S.; Sonoda, K.-H. Retinal VEGF-A Overexpression Is Not Sufficient to Induce Lymphangiogenesis Regardless of VEGF-C Upregulation and Lyve1+ Macrophage Infiltration. Investig. Opthalmology Vis. Sci. 2021, 62, 17–17. [Google Scholar] [CrossRef] [PubMed]
- Zajkowska, M.; Lubowicka, E.; Malinowski, P.; Szmitkowski, M.; Ławicki, S. Plasma levels of VEGF-A, VEGF B, and VEGFR-1 and applicability of these parameters as tumor markers in diagnosis of breast cancer. Acta Biochim. Pol. 2018, 65, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Bock, F.; Onderka, J.; Dietrich, T.; Bachmann, B.; Kruse, F.E.; Paschke, M.; Zahn, G.; Cursiefen, C. Bevacizumab as a Potent Inhibitor of Inflammatory Corneal Angiogenesis and Lymphangiogenesis. Investig. Opthalmology Vis. Sci. 2007, 48, 2545–2552. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Palma, M.B.; Verine, J.; Miriuka, S.; Inda, A.M.; Errecalde, A.L.; Desgrandchamps, F.; Carosella, E.D.; Tronik-Le Roux, D. The immune-checkpoint HLA-G/ILT4 is involved in the regulation of VEGF expression in clear cell renal cell carcinoma. BMC Cancer 2020, 20, 624. [Google Scholar] [CrossRef] [PubMed]
- Wild, R.; Dings, R.P.; Subramanian, I.; Ramakrishnan, S. Carboplatin selectively induces the VEGF stress response in endothelial cells: Potentiation of antitumor activity by combination treatment with antibody to VEGF. Int. J. Cancer 2004, 110, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Brady, M.F.; Penson, R.T.; Huang, H.; Birrer, M.J.; Walker, J.L.; DiSilvestro, P.A.; Rubin, S.C.; Martin, L.P.; Davidson, S.A.; et al. Weekly vs. Every-3-Week Paclitaxel and Carboplatin for Ovarian Cancer. New Engl. J. Med. 2016, 374, 738–748. [Google Scholar] [CrossRef]
- Saltz, L.B.; Clarke, S.; Diaz-Rubio, E.; Scheithauer, W.; Figer, A.; Wong, R.; Koski, S.; Lichinitser, M.; Yang, T.-S.; Rivera, F.; et al. Bevacizumab in Combination With Oxaliplatin-Based Chemotherapy As First-Line Therapy in Metastatic Colorectal Cancer: A Randomized Phase III Study. J. Clin. Oncol. 2008, 26, 2013–2019. [Google Scholar] [CrossRef]
- Bennouna, J.; Sastre, J.; Arnold, D.; Österlund, P.; Greil, R.; Van Cutsem, E.; von Moos, R.; Viéitez, J.M.; Bouché, O.; Borg, C.; et al. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013, 14, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, H.; Fehrenbacher, L.; Novotny, W.; Cartwright, T.; Hainsworth, J.; Heim, W.; Berlin, J.; Baron, A.; Griffing, S.; Holmgren, E.; et al. Bevacizumab plus Irinotecan, Fluorouracil, and Leucovorin for Metastatic Colorectal Cancer. New Engl. J. Med. 2004, 350, 2335–2342. [Google Scholar] [CrossRef] [PubMed]
- Eskens, F.A.L.M.; Sleijfer, S. The use of bevacizumab in colorectal, lung, breast, renal and ovarian cancer: where does it fit? Eur J Cancer. 2008, 44, 2350–2356. [Google Scholar] [CrossRef] [PubMed]
- Sandler, A.B.; Gray, R.; Brahmer, J.; Dowlati, A.; Schiller, J.H.; Perry, M.C.; Johnson, D.H. Randomized phase II/III trial of paclitaxel (P) plus carboplatin (C) with or without bevacizumab (NSC #704865) in patients with advanced non-squamous non-small cell lung cancer (NSCLC): An Eastern Cooperative Oncology Group (ECOG) Trial - E4599. J. Clin. Oncol. 2005, 23, LBA4–LBA4. [Google Scholar] [CrossRef]
- Previs, R.; Bevis, K.; Huh, W.; Tillmanns, T.; Perry, L.; Moore, K.; Chapman, J.; McClung, C.; Kiet, T.; Java, J.; et al. A prognostic nomogram to predict overall survival in women with recurrent ovarian cancer treated with bevacizumab and chemotherapy. Gynecol. Oncol. 2014, 132, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.S.; Zhang, Z.; Lee, S.I.; Marques, H.S.; Burgers, K.; Su, F.; Bauza, J.; Mannel, R.S.; Walker, J.L.; Huh, W.K.; et al. CT Perfusion as an Early Biomarker of Treatment Efficacy in Advanced Ovarian Cancer: An ACRIN and GOG Study. Clin. Cancer Res. 2017, 23, 3684–3691. [Google Scholar] [CrossRef]
- Socinski, M.A.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous, N.S.C.L.C. N Engl J Med. 2018, 378, 2288–2301. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Pluzanska, A.; Koralewski, P.; Ravaud, A.; Bracarda, S.; Szczylik, C.; Chevreau, C.; Filipek, M.; Melichar, B.; Bajetta, E.; et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet 2007, 370, 2103–2111. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Li, T.; Niu, M.; Luo, S.; Chu, Q.; Wu, K. Epidemiological trends of women’s cancers from 1990 to 2019 at the global, regional, and national levels: a population-based study. Biomark Res. 2021, 9, 55. [Google Scholar] [CrossRef]
- Marchetti, C.; Muzii, L.; Romito, A.; Panici, P.B. First-line treatment of women with advanced ovarian cancer: focus on bevacizumab. OncoTargets Ther. 2019, 12, 1095–1103. [Google Scholar] [CrossRef]
- Burger, R.A.; Brady, M.F.; Bookman, M.A.; Fleming, G.F.; Monk, B.J.; Huang, H.; Mannel, R.S.; Homesley, H.D.; Fowler, J.; Greer, B.E.; et al. Incorporation of Bevacizumab in the Primary Treatment of Ovarian Cancer. N. Engl. J. Med. 2011, 365, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Perren, T.J.; Swart, A.M.; Pfisterer, J.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; Kurzeder, C.; et al. A Phase 3 Trial of Bevacizumab in Ovarian Cancer. N. Engl. J. Med. 2011, 365, 2484–2496. [Google Scholar] [CrossRef] [PubMed]
- Oza, A.M.; Cook, A.D.; Pfisterer, J.; Embleton, A.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): Overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015, 16, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Ray-Coquard, I.; Leary, A.; Pignata, S.; Cropet, C.; González-Martín, A.; Marth, C.; et al. Olaparib plus bevacizumab first-line maintenance in ovarian cancer: final overall survival results from the PAOLA-1/ENGOT-ov25 trial. Ann Oncol. 2023, 34, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Pfisterer, J.; Joly, F.; Kristensen, G.; Rau, J.; Mahner, S.; Pautier, P.; et al. Optimal Treatment Duration of Bevacizumab as Front-Line Therapy for Advanced Ovarian Cancer: AGO-OVAR 17 BOOST/GINECO OV118/ENGOT Ov-15 Open-Label Randomized Phase III Trial. J Clin Oncol. 2023, 41, 893–902. [Google Scholar] [CrossRef]
- Gilbert, L.; Oaknin, A.; Matulonis, U.A.; Mantia-Smaldone, G.M.; Lim, P.C.; Castro, C.M.; Provencher, D.; Memarzadeh, S.; Method, M.; Wang, J.; et al. Safety and efficacy of mirvetuximab soravtansine, a folate receptor alpha (FRα)-targeting antibody-drug conjugate (ADC), in combination with bevacizumab in patients with platinum-resistant ovarian cancer. Gynecol. Oncol. 2023, 170, 241–247. [Google Scholar] [CrossRef]
- You, B.; Purdy, C.; Copeland, L.J.; Swisher, E.M.; Bookman, M.A.; Fleming, G.; Coleman, R.; Randall, L.M.; Tewari, K.S.; Monk, B.J.; et al. Identification of Patients With Ovarian Cancer Experiencing the Highest Benefit From Bevacizumab in the First-Line Setting on the Basis of Their Tumor-Intrinsic Chemosensitivity (KELIM): The GOG-0218 Validation Study. J. Clin. Oncol. 2022, 40, 3965–3974. [Google Scholar] [CrossRef] [PubMed]
- Michaud, S.E.; Ménard, C.; Guy, L.G.; Gennaro, G.; Rivard, A. Inhibition of hypoxia-induced angiogenesis by cigarette smoke exposure: impairment of the HIF-1alpha/VEGF pathway. FASEB J. 2003, 17, 1150–1152. [Google Scholar] [CrossRef]
- DiSaia, P.J.; Creasman, W.T.; Mannel, R.S.; McMeekin, D.S.; Mutch, D.G. Clinical Gynecologic Oncology; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017; 893p. [Google Scholar]
- Monk, B.J.; Tewari, K.S.; Koh, W.-J. Multimodality Therapy for Locally Advanced Cervical Carcinoma: State of the Art and Future Directions. J. Clin. Oncol. 2007, 25, 2952–2965. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, H.S.; Jung, J.J.; Lee, M.C.; Park, C.S. Expression of vascular endothelial growth factor in adenocarcinomas of the uterine cervix and its relation to angiogenesis and p53 and c-erbB-2 protein expression. Gynecol Oncol. 2002, 85, 469–475. [Google Scholar] [CrossRef]
- Wright, J.D.; Viviano, D.; Powell, M.A.; Gibb, R.K.; Mutch, D.G.; Grigsby, P.W.; Rader, J.S. Bevacizumab combination therapy in heavily pretreated, recurrent cervical cancer. Gynecol. Oncol. 2006, 103, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Bookman, M.A.; Blessing, J.A.; Hanjani, P.; Herzog, T.J.; Andersen, W.A. Topotecan in Squamous Cell Carcinoma of the Cervix: A Phase II Study of the Gynecologic Oncology Group. Gynecol. Oncol. 2000, 77, 446–449. [Google Scholar] [CrossRef] [PubMed]
- Look, K.Y.; Blessing, J.A.; Nelson, B.E.; Johnson, G.A.; Fowler, W.C.; Reid, G.C. A phase II trial of isotretinoin and alpha interferon in patients with recurrent squamous cell carcinoma of the cervix: a Gynecologic Oncology Group study. Am J Clin Oncol. 1998, 21, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Mannel, R.S.; Blessing, J.A.; Boike, G. Cisplatin and Pentoxifylline in Advanced or Recurrent Squamous Cell Carcinoma of the Cervix: A Phase II Trial of the Gynecologic Oncology Group. Gynecol. Oncol. 2000, 79, 64–66. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.G.; Blessing, J.A.; Arseneau, J. Phase II Evaluation of Altretamine for Advanced or Recurrent Squamous Cell Carcinoma of the Cervix: A Gynecologic Oncology Group Study. Gynecol. Oncol. 1996, 62, 100–102. [Google Scholar] [CrossRef]
- Schilder, R.J.; Blessing, J.A.; Morgan, M.; Mangan, C.E.; Rader, J.S. Evaluation of Gemcitabine in Patients with Squamous Cell Carcinoma of the Cervix: A Phase II Study of the Gynecologic Oncology Group. Gynecol. Oncol. 2000, 76, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.T.; Ng, T.-H. Optimal flexible designs in phase II clinical trials. Stat. Med. 1998, 17, 2301–2312. [Google Scholar] [CrossRef]
- Monk, B.J.; Sill, M.W.; Burger, R.A.; Gray, H.J.; Buekers, T.E.; Roman, L.D. Phase II Trial of Bevacizumab in the Treatment of Persistent or Recurrent Squamous Cell Carcinoma of the Cervix: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2009, 27, 1069–1074. [Google Scholar] [CrossRef]
- Tewari, K.S.; Sill, M.W.; Long, H.J., III; Penson, R.T.; Huang, H.; Ramondetta, L.M.; Landrum, L.M.; Oaknin, A.; Reid, T.J.; Leitao, M.M.; et al. Improved Survival with Bevacizumab in Advanced Cervical Cancer. N. Engl. J. Med. 2014, 370, 734–743. [Google Scholar] [CrossRef]
- Tewari, K.S.; Sill, M.W.; Penson, R.T.; Huang, H.; Ramondetta, L.M.; Landrum, L.M.; Oaknin, A.; Reid, T.J.; Leitao, M.M.; E Michael, H.; et al. Bevacizumab for advanced cervical cancer: final overall survival and adverse event analysis of a randomised, controlled, open-label, phase 3 trial (Gynecologic Oncology Group 240). Lancet 2017, 390, 1654–1663. [Google Scholar] [CrossRef]
- Grupo Español de Investigación en Cáncer de Ovario. A Randomized Phase III Trial of Platinum Chemotherapy Plus Paclitaxel With Bevacizumab and Atezolizumab Versus Platinum Chemotherapy Plus Paclitaxel and Bevacizumab in Metastatic (Stage IVB), Persistent, or Recurrent Carcinoma of the Cervix [Internet]. clinicaltrials.gov; 2023 Dec [cited 2024 Jan 1]. Report No.: NCT03556839. Available from: https://clinicaltrials.gov/study/NCT03556839.
- Monk, B.J.; Tewari, K.S.; Dubot, C.; Caceres, M.V.; Hasegawa, K.; Shapira-Frommer, R.; Salman, P.; Yañez, E.; Gümüş, M.; de Mendoza, M.O.H.; et al. Health-related quality of life with pembrolizumab or placebo plus chemotherapy with or without bevacizumab for persistent, recurrent, or metastatic cervical cancer (KEYNOTE-826): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2023, 24, 392–402. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Kuku, S.; Williams, M.; McCormack, M. Adjuvant therapy in stage III endometrial cancer: treatment outcomes and survival. a single-institution retrospective study. Int J Gynecol Cancer. 2013, 23, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.D.; A Powell, M.; Rader, J.S.; Mutch, D.G.; Gibb, R.K. Bevacizumab therapy in patients with recurrent uterine neoplasms. Anticancer Res. 2007, 27, 3525–3528. [Google Scholar] [PubMed]
- Aghajanian, C.; Sill, M.W.; Darcy, K.M.; Greer, B.; McMeekin, D.S.; Rose, P.G.; Rotmensch, J.; Barnes, M.N.; Hanjani, P.; Leslie, K.K. Phase II Trial of Bevacizumab in Recurrent or Persistent Endometrial Cancer: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2011, 29, 2259–2265. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, E.A.; Brady, W.E.; Walker, J.L.; Rotmensch, J.; Zhou, X.C.; Kendrick, J.E.; Yamada, S.D.; Schilder, J.M.; Cohn, D.E.; Harrison, C.R.; et al. Phase II trial of combination bevacizumab and temsirolimus in the treatment of recurrent or persistent endometrial carcinoma: A Gynecologic Oncology Group study. Gynecol. Oncol. 2013, 129, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, M.M.; Dickinson, S.; Narayan, P.; Zhou, Q.; Iasonos, A.; Ma, W.; Lakhman, Y.; Makker, V. Bevacizumab in advanced endometrial cancer. Gynecol. Oncol. 2021, 161, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Aghajanian, C.; Filiaci, V.; Dizon, D.S.; Carlson, J.W.; Powell, M.A.; Secord, A.A.; Tewari, K.S.; Bender, D.P.; O'Malley, D.M.; Stuckey, A.; et al. A phase II study of frontline paclitaxel/carboplatin/bevacizumab, paclitaxel/carboplatin/temsirolimus, or ixabepilone/carboplatin/bevacizumab in advanced/recurrent endometrial cancer. Gynecol. Oncol. 2018, 150, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Liang, M.; Min, J. Efficacy and Safety of Bevacizumab-Combined Chemotherapy for Advanced and Recurrent Endometrial Cancer: A Systematic Review and Meta-analysis. Balkan Med J. 2021, 38, 7–12. [Google Scholar] [CrossRef]
- Tewari, K.S.; Burger, R.A.; Enserro, D.; Norquist, B.M.; Swisher, E.M.; Brady, M.F.; Bookman, M.A.; Fleming, G.F.; Huang, H.; Homesley, H.D.; et al. Final Overall Survival of a Randomized Trial of Bevacizumab for Primary Treatment of Ovarian Cancer. J. Clin. Oncol. 2019, 37, 2317–2328. [Google Scholar] [CrossRef]
- May, T.; Altman, A.; McGee, J.; Lu, L.; Xu, W.; Lane, K.; Ghatage, P.; Rosen, B. Examining Survival Outcomes of 852 Women With Advanced Ovarian Cancer: A Multi-institutional Cohort Study. Int. J. Gynecol. Cancer 2018, 28, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Colombo, P.-E.; Mourregot, A.; Fabbro, M.; Gutowski, M.; Saint-Aubert, B.; Quenet, F.; Gourgou, S.; Rouanet, P. Aggressive surgical strategies in advanced ovarian cancer: A monocentric study of 203 stage IIIC and IV patients. Eur. J. Surg. Oncol. 2009, 35, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, C.K.; Tadesse, W.; Timmermans, M.; Kruitwagen, R.F.; Walsh, T. Only complete tumour resection after neoadjuvant chemotherapy offers benefit over suboptimal debulking in advanced ovarian cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 219, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Rauh-Hain, J.A.; Rodriguez, N.; Growdon, W.B.; Goodman, A.K.; Boruta, D.M.; Horowitz, N.S.; del Carmen, M.G.; Schorge, J.O. Primary Debulking Surgery Versus Neoadjuvant Chemotherapy in Stage IV Ovarian Cancer. Ann. Surg. Oncol. 2011, 19, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Muraji, M.; Sudo, T.; Iwasaki, S.-I.; Ueno, S.; Wakahashi, S.; Yamaguchi, S.; Fujiwara, K.; Nishimura, R. Histopathology predicts clinical outcome in advanced epithelial ovarian cancer patients treated with neoadjuvant chemotherapy and debulking surgery. Gynecol. Oncol. 2013, 131, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Rosen, B.; Laframboise, S.; Ferguson, S.; Dodge, J.; Bernardini, M.; Murphy, J.; Segev, Y.; Sun, P.; Narod, S.A. The impacts of neoadjuvant chemotherapy and of debulking surgery on survival from advanced ovarian cancer. Gynecol. Oncol. 2014, 134, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Bian, C.; Yao, K.; Li, L.; Yi, T.; Zhao, X. Primary debulking surgery vs. neoadjuvant chemotherapy followed by interval debulking surgery for patients with advanced ovarian cancer. Arch. Gynecol. Obstet. 2015, 293, 163–168. [Google Scholar] [CrossRef]
- Bryant, A.; Hiu, S.; Kunonga, P.T.; Gajjar, K.; Craig, D.; Vale, L.; et al. Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery. Cochrane Database Syst Rev. 2022, 9, CD015048. [Google Scholar] [PubMed]
- Manning-Geist, B.L.; Hicks-Courant, K.; Gockley, A.A.; Clark, R.M.; Del Carmen, M.G.; Growdon, W.B.; Horowitz, N.S.; Berkowitz, R.S.; Muto, M.G.; Worley, M.J. A novel classification of residual disease after interval debulking surgery for advanced-stage ovarian cancer to better distinguish oncologic outcome. Am. J. Obstet. Gynecol. 2019, 221, 326–e1. [Google Scholar] [CrossRef]
- Tong, R.T.; Boucher, Y.; Kozin, S.V.; Winkler, F.; Hicklin, D.J.; Jain, R.K. Vascular Normalization by Vascular Endothelial Growth Factor Receptor 2 Blockade Induces a Pressure Gradient Across the Vasculature and Improves Drug Penetration in Tumors. Cancer Res 2004, 64, 3731–3736. [Google Scholar] [CrossRef]
- Jain, R.K. Normalization of Tumor Vasculature: An Emerging Concept in Antiangiogenic Therapy. Science 2005, 307, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Albini, A.; Tosetti, F.; Li, V.W.; Noonan, D.M.; Li, W.W. Cancer prevention by targeting angiogenesis. Nat. Rev. Clin. Oncol. 2012, 9, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Albini, A.; Noonan, D.M. Angiopoietin2 and Tie2: Tied to Lymphangiogenesis and Lung Metastasis. New Perspectives in Antimetastatic Antiangiogenic Therapy. JNCI J. Natl. Cancer Inst. 2012, 104, 429–431. [Google Scholar] [CrossRef] [PubMed]
- Albini, A.; Noonan, D.M. The ‘chemoinvasion’ assay, 25 years and still going strong: the use of reconstituted basement membranes to study cell invasion and angiogenesis. Curr. Opin. Cell Biol. 2010, 22, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Aghajanian, C.; Blank, S.V.; Goff, B.A.; Judson, P.L.; Teneriello, M.G.; Husain, A.; Sovak, M.A.; Yi, J.; Nycum, L.R. OCEANS: A Randomized, Double-Blind, Placebo-Controlled Phase III Trial of Chemotherapy With or Without Bevacizumab in Patients With Platinum-Sensitive Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer. J. Clin. Oncol. 2012, 30, 2039–2045. [Google Scholar] [CrossRef] [PubMed]
- Pujade-Lauraine, E.; Hilpert, F.; Weber, B.; Reuss, A.; Poveda, A.; Kristensen, G.; Sorio, R.; Vergote, I.; Witteveen, P.; Bamias, A.; et al. Bevacizumab Combined With Chemotherapy for Platinum-Resistant Recurrent Ovarian Cancer: The AURELIA Open-Label Randomized Phase III Trial. J. Clin. Oncol. 2014, 32, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.L.; Brady, M.F.; Herzog, T.J.; Sabbatini, P.; Armstrong, D.K.; Walker, J.L.; Kim, B.G.; Fujiwara, K.; Tewari, K.S.; O’Malley, D.M.; et al. Bevacizumab and paclitaxel–carboplatin chemotherapy and secondary cytoreduction in recurrent, platinum-sensitive ovarian cancer (NRG Oncology/Gynecologic Oncology Group study GOG-0213): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017, 18, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Sznurkowski, J.J. To Bev or Not to Bev during Ovarian Cancer Maintenance Therapy? Cancers 2023, 15, 2980. [Google Scholar] [CrossRef]
- Wu, Y.S.; Shui, L.; Shen, D.; Chen, X. Bevacizumab combined with chemotherapy for ovarian cancer: an updated systematic review and meta-analysis of randomized controlled trials. Oncotarget 2016, 8, 10703–10713. [Google Scholar] [CrossRef]
- Paik, E.S.; Lee, Y.-Y.; Shim, M.; Choi, H.J.; Kim, T.-J.; Choi, C.H.; Lee, J.-W.; Kim, B.-G.; Bae, D.-S. Timing and patterns of recurrence in epithelial ovarian cancer patients with no gross residual disease after primary debulking surgery. Aust. New Zealand J. Obstet. Gynaecol. 2016, 56, 639–647. [Google Scholar] [CrossRef]
- Petrillo, M.; Amadio, G.; Salutari, V.; Paris, I.; Di Stefano, M.; Ferandina, G.; Scambia, G.; Fagotti, A. Impact of bevacizumab containing first line chemotherapy on recurrent disease in epithelial ovarian cancer: A case-control study. Gynecol. Oncol. 2016, 142, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.D.; Alwan, L.M.; Gray, H.J.; Tamimi, H.K.; Goff, B.A.; Liao, J.B. Recurrence patterns after extended treatment with bevacizumab for ovarian, fallopian tube, and primary peritoneal cancers. Gynecol. Oncol. 2013, 130, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.I.; Lee, E.J.; Lee, M.; Chung, H.; Kim, J.-W.; Park, N.H.; Song, Y.-S.; Kim, H.S. Recurrence patterns after bevacizumab in platinum-sensitive, recurrent epithelial ovarian cancer. Int. J. Gynecol. Cancer 2020, 30, 1943–1950. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.J.; Kim, H.S.; Suh, C.H.; Park, J.E. Radiological Recurrence Patterns after Bevacizumab Treatment of Recurrent High-Grade Glioma: A Systematic Review and Meta-Analysis. Korean J. Radiol. 2020, 21, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Mamo, A.; Baig, A.; Azam, M.; Rho, Y.; Sahebjam, S.; Muanza, T.; Owen, S.; Petrecca, K.; Guiot, M.; Al-Shami, J.; et al. Progression Pattern and Adverse Events with Bevacizumab in Glioblastoma. Curr. Oncol. 2016, 23, 468–471. [Google Scholar] [CrossRef] [PubMed]
- Christ, S.M.; Youssef, G.; Tanguturi, S.K.; Cagney, D.; Shi, D.; McFaline-Figueroa, J.R.; Chukwueke, U.; Lee, E.Q.; Hertler, C.; Andratschke, N.; et al. Re-irradiation of recurrent IDH-wildtype glioblastoma in the bevacizumab and immunotherapy era: Target delineation, outcomes and patterns of recurrence. Clin. Transl. Radiat. Oncol. 2024, 44, 100697. [Google Scholar] [CrossRef]
- Lee, N.; Kim, S.I.; Lee, M.; Kim, H.S.; Kim, J.W.; Park, N.H.; Song, Y.S. Bevacizumab Efficacy and Recurrence Pattern of Persistent and Metastatic Cervical Cancer. In Vivo 2019, 33, 863–868. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).