1. Introduction
The purpose of organisms is ultimately the successful transmission of a specific DNA from one generation to the next. This is true of both single celled species and multicellular eukaryotes. Natural selection allows the appearance of species that are increasingly adapted to their environment. In animal phyla with more complex structure, the inheritance and transmission of these modifications forms the basis of Darwinian transmutation of species. Such active driving forces are only relevant during that portion of the lifespan where production and maintenance of the next generation is involved. Beyond this maturational stage, the power of trans-generational selective forces becomes irrelevant and is gradually lost. In the case of human, medical developments have led to the lifespan often being considerably longer than the fertility-span and the period of essential sustenance of offspring. After this, organisms are no longer subject to evolutionary influences. The consequent generational irrelevance means that the biology of the aged is not forced into an increasing optimal configuration. As a result, those metabolic processes, vital for organismic success are no longer subject to selective pressures and are more free to deviate from their originally tightly circumscribed limits. A more direct route taken by a specific metabolic path may increase its efficiency in isolation but not that of the organism as a whole. Such “short cuts” are prevented earlier in the life cycle when the effectiveness of the entire organism is paramount. However, in the absence of trans-generational relevance, there is nothing to prevent their increasing appearance.
This review discusses how, in the absence of selective forces, three key networks vital for defense and maintenance of nervous system integrity, can become degraded and ultimately injurious rather than protective. The basis of such adverse age-related deviation is attributed in large part to diminishing competence of mitochondrial function.
2. The Decline of Immune System Function with Age
The continual surveillance of tissues within the body by both circulating immune system and the immune responsivity generated intrinsically by many cell types is an important contributor to organismic survival. Such protection is by recognition and destruction of extrinsic bacteria and viruses together with endogenous abnormal or dead cells. In order to ensure effective functioning, the elimination of undesirable materials has to take place in a focused manner, with a minimal penumbra that would involve proximal healthy cells. This system develops during maturation when the protective effect of maternal antibodies in the young, progressively decreases. Innate immunity depends on neutrophils and macrophages and these proliferate shortly after birth. Adaptive immunity requires the development of appropriate antibodies for a range of antigens, Both T cells and B cells require maturation and also experience, in order to become fully functional by allowing T cells to present antigens to B cells which can then produce specifically directed antibodies. This developmental process is promoted by some desirable constituents of the intestinal microbiome, notably Bifidobacterium infantis (Luo et al., 2023).
Several autoimmune disorders can develop early in life, or in adulthood with earlier onset generally being associated with elevated disease severity (Amador-Patorroyo et al., 2012). These include such diseases as type 1 diabetes and asthma, multiple sclerosis, and rheumatoid arthritis. These diseases have a significant genetic component but environmental factors also determine whether and when the disease will be expressed.
A different type of inflammatory disturbance is found in many age-related diseases. This consists of a low level of sustained systemic inflammation which, unlike a targeted immune response, this inflammation is independent of any provocative stimulus. Such a chronic condition has a wide variety of adverse effects and plays a significant role in the pathology of a wide range of diseases whose prevalence rises with aging. These include ischemic heart disease, vascular disease, cancer, Type II diabetes, and fatty liver disease. Accelerated appearance of the condition has been attributed to many underlying dietary and lifestyle factors (Furman et al., 2019). Aging is likely to be a key but not an exclusive factor. Such extended inflammation forms a significant component of nearly all neurodegenerative disorders including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease and amyotrophic lateral sclerosis (Bondy, 2020, Kwon and Koh. 2020). Neuroinflammation may well a primary mechanism of Alzheimer’s disease rather than a secondary consequence (Kinney et al., 2018) and may be the factor that drives the formation of tangled tau proteins and amyloid plaque (Pascoal et al., 2021). This age-related malfunction seems attributable to both innate and adaptive immune mechanisms (Li et al., 2021).
3. The Excessive Presence of Oxidant Free Radicals with Aging.
Hydrogen peroxide (H2O2),the superoxide anion radical (O2·-), and nitric oxide (NO.) are key signaling agents. They are generated by a many enzymes notably by the mitochondrial respiratory chain. Redox signaling is a critical mechanism in the regulation of many metabolic processes and adaptive responses to various stressors (Sies and Jones, 2020). This signaling is especially prominent in the brain due to its high energy demand (Yin et al., 2016). Any excess content or diffusion of these more stable precursors of highly reactive short lived oxidant free radicals, can be effectively regulated by the presence of the several antioxidant enzymes, such as catalase, superoxide dismutase and peroxidases.
With senescence, the efficiency of underlying processes is diminished and a redox imbalance emerges between the synthesis and degradation of these species by antioxidant enzymes and chemicals (Goldsteins et al., 2022). The failure of homeostasis leads to free radical-induced damage to macromolecules. This has been linked to the aging process and also to the emergence of many age-associated disease classes including neurodegenerative disease, cardiovascular disease and cancer (Jomova et al., 2023). The issue of whether oxidative stress is the primary cause or secondary consequence of the disease remains unclarified.
4. The Emergence of a Chronic State of Low Level Excitatory Activity
The aging brain is characterized by a minor but permanent presence of undirected hyperexcitation. The basis for this includes heightened content of free calcium ions within the cell which can be attributed to failure of their mitochondrial sequestration. This results in an excessive level of calcium-effected neurotransmitter release and overstimulation of glutaminergic NMDA receptors. The prolonged activation of glutamate receptors leads to enhanced calcium influx, further mitochondrial failure, and oxidative stress (Sanghai and Tranmer, 2023). In addition to initiating excitotoxicity, excess intracellular calcium causes the activation of calpain (Ameen et al., 2022). This protease can disrupt intracellular architecture, leading to the emergence of a range of metabolic defects and ultimately to apoptosis. Calpain also contributes to the emergence of several inflammatory events including activation of NF-kB and inflammatory, cytokines. Calpains participate in many age-related non-resolving inflammatory diseases, including atherosclerosis and rheumatoid arthritis (Ji et al., 2016). Calpain can effect the activation of astrocytes from the resting to the reactive fibrous astrocytes, and can increase microglial reactivity. These chronic neuroinflammatory responses lead to neuronal loss (Mahaman et al., 2019, Ko et al., 2020). Inhibition of calpain diminishes microglial and astroglial responsivity and reduces the extent of the neuropathological changes and behavioral decline of aged transgenic mice modeling Alzheimer’s disease (Medeirios et al., 2012) and Parkinson’s disease (Metwally et al., 2023). Glutamate receptors that respond to excess activation are also found in oligodendrocytes which can be damaged by excitotoxic events (Bayón-Cordero et al., 2022). NMDA receptor stimulation also induces activation of NADPH oxidase 2, magnitude relation enzyme that leads to production of reactive oxygen species which can harm surrounding cells (Reyes et al., 2012). The transition from the ability of activation of the NMDA receptor as a means to initiate plasticity and stimulate cell survival, to such activation leading to advancement of cell death in Alzheimer’s disease, may reside in the shift of this receptor shift from a germane intrasynaptic location to irrelevant extrasynaptic sites (Wang et al., 2017). The use of NMDA antagonists may protect against age-related neurodegenerative disease (Companys-Alemany et al., 2022).
5. The Mitochondrial Basis for Age-Related Deviation of Essential Processes toward Injurious Configurations
The inevitability of the distortion of the three key metabolic processes described must ultimately be ascribed to failure of repair and maintenance programs that were previously functional. There is evidence that most of these deficits are triggered by the onset and accumulation of mitochondrial flaws during senescence. Effective mitochondrial functioning is vital for organismic function. Mitochondrial quality which is a prime determining factor in the health of cerebral cell, declines with aging (Ragupathy et al., 2020) and even more so in neurodegenerative disease (Shen et al., 2022). The three adverse transformations described above are all found in aging mitochondria and this organelle may trigger effects throughout the entire cell. Mitochondrial dynamics of fusion, fission and mitophagy are reduced with age, and the consequent reduction of mitochondrial quality can lead to a series of adverse consequences (Grimm and Eckert, 2017).
A wide range of changes can be observed in mitochondria of aged subjects. These include greater leakage of free radicals during oxidative phosphorylation and an accumulation of mutations in mtDNA (Lee and Wei, 2012). A higher rate of single point mutations in mtDNA has been found in brains from the elderly in comparison with those from the young (Lin et al., 2002). These mutations often involve deletion of DNA and consequent failure of productive oxidative phosphorylation (Fontana and Gahlon, 2020). Such ineffective mitochondria can be clonally amplified along with healthy mitochondria. In younger healthy animals, mitochondrial dynamics of division and fusion serve to minimize the accumulation of such unproductive organelles but the efficiency of this process is diminished with age (Grimm and Eckert, 2017).
The harmful changes in mitochondria that are emphasized with age, involvs not only deficits in mtDNA replication but also release of nuclear chromatin fragments into the cytoplasm induced by mitochondrial factors (Vizioli et al., 2020). These changes lead to a hypometabolic state in the aged brain (Yin et al., 2016). In a genetic mouse model where mitochondrial polymerase-γ is blocked, premature aging occurs and longevity is drastically reducedxxx (Sanchez-Contreras and Kennedy, 2022).
5.1. Free Radical Production
Deficits in mitochondrial ATP production are accompanied by elevated leakage of reactive oxygen species as aging proceeds (Rosa et al., 2023). ROS are components of key intracellular signaling systems can modulate the activity of several Ca2+ channels (Godoy et al., 2022).
Paradoxically, there is also contrary evidence that indicates that a mild diminution of mitochondrial effectiveness can result in lengthening of the lifespan. Since this effect is blocked by antioxidants, it seems that this effect is due to the hormetic effect of low levels of reactive oxygen species (Sun et al., 2016, Grimm and Eckert, 2017). Moderate activation of stress responses may restore the effectiveness of clearance of improperly folded proteins that accumulate with aging (Dutta et al.,2022).
Potentially reparable mitochondrial changes include reversal of transient mtDNA deletions that reduce the effective functioning of oxidative phosphorylation. Permanent loss of the intact and functional mitochondrial genome following oxidative stress may be due to the excessive and harmful prolongation of processes that could efficiently repair mtDNA in response to transient moderate stressors. Prolonged excess production of reactive oxygen species leads irreversible mitochondrial damage rather than leading to a reversible and useful adaptation (Stenberg et al., 2022). This dysregulation is likely to grow with age where chronic stressors tend to accumulate (Schosserer et al., 2019).
5.2. Undesirable Autoimmune Responses
Unlike nuclear DNA, mitochondrial DNA is not methylated. Since unmethylated DNA is also found in bacteria, the appearance of mtDNA in the cytoplasm can invoke an autoimmune response by way of the innate immune system (Sun et al., 2016). mtDNA can be sensed by several moieties including Toll-like receptor 9, and the NLRP3 inflammasome. When activated, all these will lead to the production of inflammatory cytokines (Zanini et al., 2023).
The mtDNA fragments released by damaged aging mitochondria contain bacterial-like CpG segments (Pica et al., 2021). These are recognized and interpreted as a threat by the innate immune system, and a response is triggered by way of the cGAS-STING signaling pathway a regulator of senescence-associated secretory phenotype (SASP). Cyclic GMP-AMP synthase (cGAS) recognizes cytoplasmic DNA fragments and this effects the stimulation of interferon genes (STING) (Kim et al., 2023). This in turn promotes formation of inflammatory cytokines such as TNF-a. Such untargeted inflammation leads to neurodegeneration (Gulen et al., 2023). The leakage of mitochondrial DNA into the cytosol, may thus be a key factor in accounting for many age related diseases as being based on autoimmune responses. Such a condition is typical of cells that have lost functionality but have evaded the apoptotic pathway (Homolak, 2023). Inhibition of excessive mitochondrial permeability in aged mice, decreases escape of miDNA fragments and calcium into the cytosol and this leads to reduction of inflammation and reduces the progression of markers of senescence (Victorelli et al., 2023).
5.3. Persistent Hyperexcitation
Following electrical activity, the cytosolic calcium level is increased as the wave of axonal depolarization arrives at the synapse. The mitochondrion rapidly takes up this excess calcium by way of the calcium uniporter complex. This prevents further calcium-initiated repetitive transmitter release. Calcium can then be more gradually released into the cytosol and thence out of the cell by Na+/Ca2+ and H+/Ca2+ exchangers. By this means, the mitochondrion is critical for optimal regulation of synaptic transmission. Oxidative stress can be a secondary consequence of mitochondrial calcium overload in excitotoxicity (Depp et al., 2018, Neves et al., 2023) and can promote the opening of the non-selective mitochondrial permeability transition pore (mPTP). This can lead to mitochondrial swelling, reduced capacity for oxidative phosphorylation and ultimately, apoptic cell death. Such changes are prominent in aging but causality is difficult to pinpoint due to the reciprocal interactions between these systems (Jia and Du, 2021). Most of the energy requirement of the brain is concerned with maintenance of ion gradients and their rebuilding after neural activity. Diminution of ATP production by aged mitochondria can lead to increased glutamatergic excitotoxicity (Baltan et al., 2014). It is noteworthy that synapses with few mitochondria in their vicinity exhibit hyperexcitability (Verma et al., 2022).
Several age-related neurodegenerative disorders are characterized by a common feature, namely excessive calcium levels within mitochondria which inevitably leads to heightened cytoplasmic calcium content and thence to elevated glutamatergic activity (Walters and Usachev, 2023). Rather than resulting in overt excitotoxicity, this lesser level of rising cytosolic calcium may lead to a prolonged extension of excitatory postsynaptic potentials (Verma et al., 2022). Failure of mitochondrial calcium buffering may have similar deficits leading to excitotoxicity in normal aged (Baltan, 2014).
6. Importance of Mitophagy
A major factor in maintaining the quality of mitochondria, is the clearance of non-functioning variants by mitophagy. However, the vigor of mitophagy is reduced as the brain ages. An important aspect of cellular quality control is removal of ineffective mitochondria by mitophagy. However, the intensity of mitophagy is reduced with brain aging. Both mitogenesis and phagocytic removal of aberrant mitochondria are both reduced with age (Gaziev et al., 2014).
The cellular content of ineffective mitochondria is gradually increased for several reasons. Firstly, while the DNA deletions can cause loss of genes required for oxidative phosphorylation, such diminution in size may enable faster replication than that of normal mitochondria.
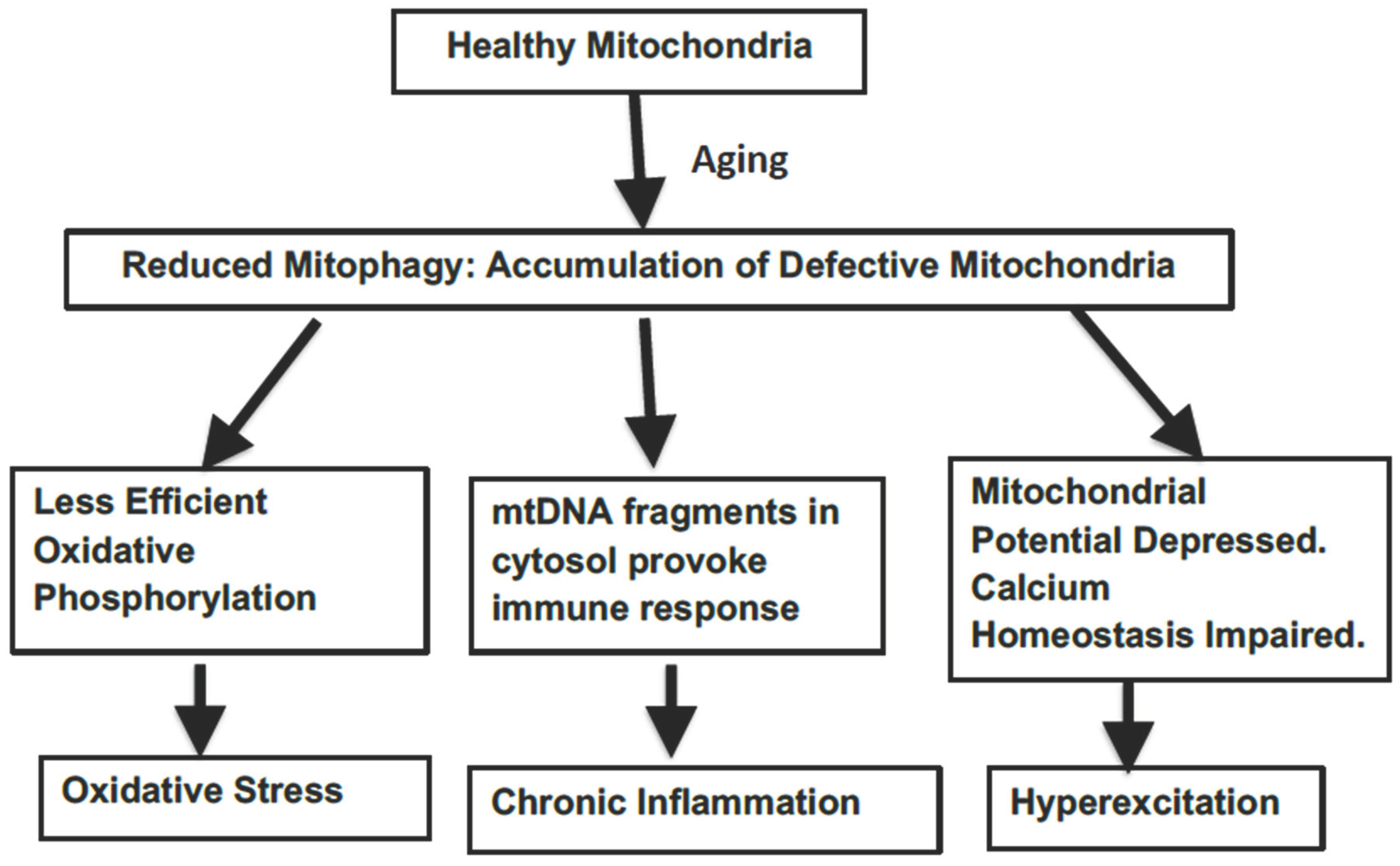
Secondly, there is evidence that mitochondria can be transferred between cells of the nervous system (Fairley et al., 2022). This could allow dissemination of flawed mitochondria and induction of an inflammatory response, thereby contributing to neurodegeneration (Joshi et al., 2019). Alternatively, damaged mitochondria can be transferred from neurons to astrocytes where they can be effectively degraded (Morales et al., 2020). Reduced mitophagy may lead to an excessive content of defective mitochondria in AD. Depression of levels of glucose consumption characterize cerebral senescence. Age-related mitochondrial dysfunction may be an early component of the development of AD, predating the onset of clinical symptoms. (Atlante et al., 2022).
The potential means by which defective mitophagy could lead to the age-related warping of normal biological processes is illustrated in
Figure 1.
7. Therapeutic Moderation of the Rate of Aging
Effective mitophagy has been associated with longevity in several species (Swerdlow and Wilkins, 2020). In a variety of experimental systems, stimulation of mitophagy has found to promote overall organismic health, to extend the lifespan and to retard cognitive decline (Schmid et al., 2022). Human trials are currently in progress (Caponio et al., 2022, Rappe and Williams, 2022).The mitochondrion may thus be a suitable target for developing new therapeutic strategies for treatment not only of AD but for slowing brain aging in general (Onyango et al., 2021) A wide range of bioactive dietary constituents may stimulate mitophagy within the brain. While the utility of most of these agents have been ascribed to their antioxidant properties, the use of broad spectrum antioxidants has not been demonstrated to effectively extend the lifespan or to retard the rate of onset of cognitive or neurological evidence of senescence (Gebert et al., 2023). The ability of these micronutrients to promote degradation of ineffective mitochondria may involve more specific mechanisms by way of the stress-responsive neurotrophic factor BDNF, mTOR and the sirtuin pathway. In addition to dietary constituents, there is increasing evidence that the moderate challenges presented by activities such as caloric restriction and exercise can significantly decelerate the aging process (Camandola et al., 2017, Wan et al., 2022, Varghese et al., 2023).
8. Conclusion
All of the systems described above are interrelated, and deficits in one can rapidly have a reciprocal impact on others. Declining energy production by less efficient mitochondria disrupts Ca
2+ buffering which leads to excitotoxicity and enhances generation of reactive oxygen species. Excess ROS lead to increased accretion of mutations in mtDNA (Godoy et al., 2021). Fragments of mtDNA entering the cytosol can then provoke inflammatory responses. The role of the mitochondrion in being causal in the deformation of several vital intracellular activities is summarized in
Table 1.
The key metabolic operations described initially, which are essentially adaptive in nature, and their competence is generally maintained through maturity but inevitably begins to decline with senescence. The reason for this broad based failure may relate to the evolutionary forces driving optimal metabolic efficiency. Presumably these are in effect for around the first forty years of human life when reproduction and nurturing of offspring are cardinal. The maintenance of the best survival strategies over this time period may be at the expense of later biological competence. The lack of evolutionary pressure in later life may account for the onset of neurodegenerative disorders due to absence of corrective imperatives. The deficits discussed all involve a falling off of the proportion of a meaningful signal relative to irrelevant background activity. The failure of effective mitochondrial function may underlie all three of the age-related modulations described and may lead to the onset of several neurodegenerative diseases (Cilleros-Holgado et al., 2023). The basis for such progressive failure may relate to the lack of a regulating evolutionary track-line after mid-life. Any significant deficits occurring prior to this would be subject to strict pruning by evolutionary forces. In the absence of such focusing imperatives, a certain drift takes places. Receptors diffuse from their functional sites to extraneous locations where they act in a less directed in a manner that is often harmful. Rather than accurately targeting invasive species or abnormal cells, the immune system maintains a more dispersed but less purposeful presence, leading to autoimmune incursions. There is increased production of reactive oxygen species which can be randomly destructive, consequent to less efficient metabolic transformations.
The mitochondrion is active throughout life and undergoes many replications. The mitochondrial genome has 100 fold higher mutation rate than the nuclear genome due to replication and repair errors and the absence of protective histones. This is due to mitochondria being the site of high levels of reactive oxygen species due to leakage from the electron transport respiratory chain, causing single or double-strand breakage of mtDNA. In addition, the fidelity of mitochondrial DNA polymerase is much lower than that of nuclear DNA polymerases can further contribute to heteroplasmy of mtDNA (Wang et al., 2023). Any independent evolutionary appearance of less competent variants will be curtailed in the young where the pressure of natural selection is borne by the entire organism. With age this selection for efficiency of the whole eukaryote dissipates, leading to greater mtDNA diversity (Nadalutti et al., 2022). Combined with the large number of replications undertaken by mitochondria, this implies that these organelles are subject to intense competitive influences within the cell. This allows mitochondrial variants free rein for contest among themselves with the most rapidly dividing variants gaining a selective advantage. However, such pressures made in the best interest of individual mitochondrial survival, may not always align with the best interests of the whole organism. However, while this drift may be unavoidable, there are several means by which the rate of drift toward the age-related appearance of inoperative mitochondria may be delayed.
References
- Amador-Patarroyo, M.J.; Rodriguez-Rodriguez, A.; Montoya-Ortiz, G. How does age at onset influence the outcome of autoimmune diseases? Autoimmune Dis. 2012, 2012, 251730. [Google Scholar] [CrossRef]
- Ameen, S.S.; Griem-Krey, N.; Dufour, A.; Hossain, M.I.; Hoque, A.; Sturgeon, S.; Nandurkar, H.; Draxler, D.F.; Medcalf, R.L.; Kamaruddin, M.A.; Lucet, I.S.; Leeming, M.G.; Liu, D.; Dhillon, A.; Lim, J.P.; Basheer, F.; Zhu, H.J.; Bokhari, L.; Roulston, C.L.; Paradkar, P.N.; Kleifeld, O.; Clarkson, A.N.; Wellendorph, P.; Ciccotosto, G.D.; Williamson, N.A.; Ang, C.S.; Cheng, H.C. N-Terminomic Changes in Neurons During Excitotoxicity Reveal Proteolytic Events Associated With Synaptic Dysfunctions and Potential Targets for Neuroprotection. Mol Cell Proteomics. 2023, 22, 100543. [Google Scholar] [CrossRef]
- Atlante, A.; Amadoro, G.; Latina, V.; Valenti, D. Therapeutic Potential of Targeting Mitochondria for Alzheimer’s Disease Treatment. J. Clin. Med. 2022, 11, 6742. [Google Scholar] [CrossRef] [PubMed]
- Baltan, S. Excitotoxicity and mitochondrial dysfunction underlie age-dependent ischemic white matter injury. Adv. Neurobiol. 2014, 11, 151–70. [Google Scholar] [CrossRef]
- Bayón-Cordero, L.; Ochoa-Bueno, B.I.; Ruiz, A.; Ozalla, M.; Matute, C.; Sánchez-Gómez, M.V. GABA Receptor Agonists Protect From Excitotoxic Damage Induced by AMPA in Oligodendrocytes. Front Pharmacol. 2022, 13, 897056. [Google Scholar] [CrossRef] [PubMed]
- Bondy, S.C. Aspects of the immune system that impact brain function. J. Neuroimmunol. 2020, 340, 577167. [Google Scholar] [CrossRef] [PubMed]
- Camandola, S.; Mattson, M.P. Brain metabolism in health, aging, and neurodegeneration. EMBO J. 2017, 36, 1474–1492. [Google Scholar] [CrossRef]
- Caponio, D.; Veverová, K.; Zhang, S.Q.; Shi, L.; Wong, G.; Vyhnalek, M.; Fang, E.F. Compromised autophagy and mitophagy in brain ageing and Alzheimer’s diseases. Aging Brain 2022, 2, 100056. [Google Scholar] [CrossRef]
- Cilleros-Holgado, P.; Gómez-Fernández, D.; Piñero-Pérez, R.; Romero-Domínguez, J.M.; Reche-López, D.; López-Cabrera, A.; Álvarez-Córdoba, M.; Munuera-Cabeza, M.; Talaverón-Rey, M.; Suárez-Carrillo, A.; Romero-González, A.; Sánchez-Alcázar, J.A. Mitochondrial Quality Control via Mitochondrial Unfolded Protein Response (mtUPR) in Ageing and Neurodegenerative Diseases. Biomolecules 2023, 13, 1789. [Google Scholar] [CrossRef]
- Companys-Alemany, J.; Turcu, A.L.; Schneider, M.; Müller, C.E.; Vázquez, S.; Griñán-Ferré, C.; Pallàs, M. NMDA receptor antagonists reduce amyloid-β deposition by modulating calpain-1 signaling and autophagy, rescuing cognitive impairment in 5XFAD mice. Cell Mol Life Sci. 2022, 79, 408. [Google Scholar] [CrossRef]
- Depp, C.; Bas-Orth, C.; Schroeder, L.; Hellwig, A.; Bading, H. Synaptic Activity Protects Neurons Against Calcium-Mediated Oxidation and Contraction of Mitochondria During Excitotoxicity. Antioxid Redox Signal. 2018, 29, 1109–1124. [Google Scholar] [CrossRef] [PubMed]
- Drew, B.; Leeuwenburgh, C. Ageing and subcellular distribution of mitochondria: role of mitochondrial DNA deletions and energy production. Acta Physiol Scand. 2004, 182, 333–41. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.; Garcia, G.; Higuchi-Sanabria, R. Hijacking Cellular Stress Responses to Promote Lifespan. Front. Aging 2022, 3, 860404. [Google Scholar] [CrossRef] [PubMed]
- Fairley, L.H.; Grimm, A.; Eckert, A. Mitochondria Transfer in Brain Injury and Disease. Cells 2022, 11, 3603. [Google Scholar] [CrossRef] [PubMed]
- Fontana, G.A.; Gahlon, H.L. Mechanisms of replication and repair in mitochondrial DNA deletion formation. Nucleic Acids Res. 2020, 48, 11244–11258. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; Miller, A.H.; Mantovani, A.; Weyand, C.M.; Barzilai, N.; Goronzy, J.J.; Rando, T.A.; Effros, R.B.; Lucia, A.; Kleinstreuer, N.; Slavich, G.M. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Gao, A.; McCoy, H.M.; Zaman, V.; Shields, D.C.; Banik, N.L.; Haque, A. Calpain activation and progression of inflammatory cycles in Parkinson’s disease. Front. Biosci. (Landmark Ed). 2022, 27, 20. [Google Scholar] [CrossRef]
- Gaziev, A.I.; Abdullaev, S.; Podlutsky, A. Mitochondrial function and mitochondrial DNA maintenance with advancing age. Biogerontology 2014, 15, 417–38. [Google Scholar] [CrossRef]
- Gebert, M.; Sławski, J.; Kalinowski, L.; Collawn, J.F.; Bartoszewski, R. The Unfolded Protein Response: A Double-Edged Sword for Brain Health. Antioxidants 2023, 12, 1648. [Google Scholar] [CrossRef]
- Godoy, J.A.; Rios, J.A.; Picón-Pagès, P.; Herrera-Fernández, V.; Swaby, B.; Crepin, G.; Vicente, R.; Fernández- Fernández, J.M.; Muñoz, F.J. Mitostasis, Calcium and Free Radicals in Health, Aging and Neurodegeneration. Biomolecules 2021, 11, 1012. [Google Scholar] [CrossRef]
- Goldsteins, G.; Hakosalo, V.; Jaronen, M.; Keuters, M.H.; Lehtonen, Š.; Koistinaho, J. CNS Redox Homeostasis and Dysfunction in Neurodegenerative Diseases. Antioxidants 2022, 11, 405. [Google Scholar] [CrossRef]
- Grimm, A.; Eckert, A. Brain aging and neurodegeneration: from a mitochondrial point of view. J. Neurochem. 2017, 143, 418–431. [Google Scholar] [CrossRef]
- Gulen, M.F.; Samson, N.; Keller, A.; Schwabenland, M.; Liu, C.; Glück, S.; Thacker, V.V.; Favre, L.; Mangeat, B.; Kroese, L.J.; Krimpenfort, P.; Prinz, M.; Ablasser, A. cGAS-STING drives ageing-related inflammation and neurodegeneration. Nature 2023, 620, 374–380. [Google Scholar] [CrossRef]
- Homolak, J. Targeting the microbiota-mitochondria crosstalk in neurodegeneration with senotherapeutics. Adv. Protein Chem. Struct. Biol. 2023, 136, 339–383. [Google Scholar] [CrossRef]
- Ji, J.; Su, L.; Liu, Z. Critical role of calpain in inflammation. Biomed Rep 5: 647-652, 2016 Jomova K, Raptova R, Alomar SY, Alwasel SH, Nepovimova, E., Kuca, K., Valko, M. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Arch. Toxicol. 2023, 97, 2499–2574. [Google Scholar] [CrossRef]
- Joshi, A.U.; Minhas, P.S.; Liddelow, S.A.; Haileselassie, B.; Andreasson, K.I.; Dorn, G.W., 2nd; Mochly-Rosen, D. Fragmented mitochondria released from microglia trigger A1 astrocytic response and propagate inflammatory neurodegeneration. Nat Neurosci. 2019, 22, 1635–1648, doi: 10.1038/s41593-019-0486-0. Erratum in: Nat Neurosci. 2021 Feb;24(2):289. [Google Scholar] [CrossRef]
- Kim, J.; Kim, H.S.; Chung, J.H. Molecular mechanisms of mitochondrial DNA release and activation of the cGAS-STING pathway. Exp. Mol. Med. 2023, 55, 510–519. [Google Scholar] [CrossRef]
- Kinney, J.W.; Bemiller, S.M.; Murtishaw, A.S.; Leisgang, A.M.; Salazar, A.M.; Lamb, B.T. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimers Dement. (N Y). 2018, 4, 575–590. [Google Scholar] [CrossRef]
- Kwon, H.S.; Koh, S.H. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl. Neurodegener. 2020, 9, 42. [Google Scholar] [CrossRef]
- Lee, H.C.; Wei, Y.H. Mitochondria and aging. Adv. Exp. Med. Biol. 2012, 942, 311–27. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Laws, S.M.; Miles, L.A.; Wiley, J.S.; Huang, X.; Masters, C.L.; Gu, B.J. Genomics of Alzheimer’s disease implicates the innate and adaptive immune systems. Cell Mol. Life Sci. 2021, 78, 7397–7426. [Google Scholar] [CrossRef]
- Lin, M.T.; Simon, D.K.; Ahn, C.H.; Kim, L.M.; Beal, M.F. High aggregate burden of somatic mtDNA point mutations in aging and Alzheimer’s disease brain. Hum. Mol. Genet. 2002, 11, 133–45. [Google Scholar] [CrossRef]
- Luo, Y.; Zhang, Y.; Yang, Y.; Wu, S.; Zhao, J.; Li, Y.; Kang, X.; Li, Z.; Chen, J.; Shen, X.; He, F.; Cheng, R. Bifidobacterium infantis and 2’-fucosyllactose supplementation in early life may have potential long-term benefits on gut microbiota, intestinal development, and immune function in mice. J. Dairy Sci. 2023, 106, 7461–7476. [Google Scholar] [CrossRef] [PubMed]
- Mahaman, Y.A.R.; Huang, F.; Kessete Afewerky, H.; Maibouge, T.M.S.; Ghose, B.; Wang, X. Involvement of calpain in the neuropathogenesis of Alzheimer’s disease. Med. Res. Rev. 2019, 39, 608–630. [Google Scholar] [CrossRef]
- Medeiros, R.; Kitazawa, M.; Chabrier, M.A.; Cheng, D.; Baglietto-Vargas, D.; Kling, A.; Moeller, A.; Green, K.N.; LaFerla, F.M. Calpain inhibitor A-705253 mitigates Alzheimer’s disease-like pathology and cognitive decline in aged 3xTgAD mice. Am. J. Pathol. 2012, 181, 616–25. [Google Scholar] [CrossRef]
- Metwally, E.; Al-Abbadi, H.A.; Hussain, T.; Murtaza, G.; Abdellatif, A.M.; Ahmed, M.F. Calpain signaling: from biology to therapeutic opportunities in neurodegenerative disorders. Front. Vet. Sci. 2023, 10, 1235163. [Google Scholar] [CrossRef]
- Morales, I.; Sanchez, A.; Puertas-Avendaño, R.; Rodriguez-Sabate, C.; Perez-Barreto, A.; Rodriguez, M. Neuroglial transmitophagy and Parkinson’s disease. Glia 2020, 68, 2277–2299. [Google Scholar] [CrossRef]
- Nadalutti, C.A.; Ayala-Peña, S.; Santos, J.H. Mitochondrial DNA damage as driver of cellular outcomes. Am. J. Physiol. Cell Physiol. 2022, 322, C136–C150. [Google Scholar] [CrossRef] [PubMed]
- Neves, D.; Salazar, I.L.; Almeida, R.D.; Silva, R.M. Molecular mechanisms of ischemia and glutamate excitotoxicity. Life Sci. 2023, 328, 121814. [Google Scholar] [CrossRef] [PubMed]
- Onyango, I.G.; Bennett, J.P.; Stokin, G.B. Mitochondrially-Targeted Therapeutic Strategies for Alzheimer’s Disease. Curr. Alzheimer Res. 2021, 18, 753–771. [Google Scholar] [CrossRef]
- Pascoal, T.A.; Benedet, A.L.; Ashton, N.J.; Kang, M.S.; Therriault, J.; Chamoun, M.; Savard, M.; Lussier, F.Z.; Tissot, C.; Karikari, T.K.; Ottoy, J.; Mathotaarachchi, S.; Stevenson, J.; Massarweh, G.; Schöll, M.; de Leon, M.J.; Soucy, J.P.; Edison, P.; Blennow, K.; Zetterberg, H.; Gauthier, S.; Rosa-Neto, P. Microglial activation and tau propagate jointly across Braak stages. Nat. Med. 2021, 27, 1592–1599, doi: 10.1038/s41591-021-01456-w. Erratum in: Nat Med. 2021 Nov;27(11):2048–2049. [Google Scholar] [CrossRef]
- Picca, A.; Calvani, R.; Coelho-Junior, H.J.; Marzetti, E. Cell Death and Inflammation: The Role of Mitochondria in Health and Disease. Cells 2021, 10, 537. [Google Scholar] [CrossRef]
- Rappe, A.; McWilliams, T.G. Mitophagy in the aging nervous system. Front. Cell Dev. Biol. 2022, 10, 978142. [Google Scholar] [CrossRef]
- Reyes, R.C.; Brennan, A.M.; Shen, Y.; Baldwin, Y.; Swanson, R.A. Activation of neuronal NMDA receptors induces superoxide-mediated oxidative stress in neighboring neurons and astrocytes. J. Neurosci. 2012, 32, 12973–8. [Google Scholar] [CrossRef] [PubMed]
- Rosa, F.L.L.; de Souza, I.I.A.; Monnerat, G.; Campos de Carvalho, A.C.; Maciel, L. Aging Triggers Mitochondrial Dysfunction in Mice. Int. J. Mol. Sci. 2023, 24, 10591. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Contreras, M.; Kennedy, S.R. The Complicated Nature of Somatic mtDNA Mutations in Aging. Front. Aging 2022, 2, 805126. [Google Scholar] [CrossRef] [PubMed]
- Sanghai, N.; Tranmer, G.K. Biochemical and Molecular Pathways in Neurodegenerative Diseases: An Integrated View. Cells 2023, 12, 2318. [Google Scholar] [CrossRef] [PubMed]
- Schmid, E.T.; Pyo, J.H.; Walker, D.W. Neuronal induction of BNIP3-mediated mitophagy slows systemic aging in Drosophila. Nat. Aging 2022, 2, 494–507. [Google Scholar] [CrossRef] [PubMed]
- Schosserer, M.; Banks, G.; Dogan, S.; Dungel, P.; Fernandes, A.; Marolt Presen, D.; Matheu, A.; Osuchowski, M.; Potter, P.; Sanfeliu, C.; Tuna, B.G.; Varela-Nieto, I.; Bellantuono, I. Modelling physical resilience in ageing mice. Mech. Ageing Dev. 2019, 177, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Sun, P.; Zhang, H.; Yang, H. Mitochondrial quality control in the brain: The physiological and pathological roles. Front. Neurosci. 2022, 16, 1075141. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef] [PubMed]
- Stenberg, S.; Li, J.; Gjuvsland, A.B.; Persson, K.; Demitz-Helin, E.; González Peña, C.; Yue, J.X.; Gilchrist, C.; Ärengård, T.; Ghiaci, P.; Larsson-Berglund, L.; Zackrisson, M.; Smits, S.; Hallin, J.; Höög, J.L.; Molin, M.; Liti, G.; Omholt, S.W.; Warringer, J. Genetically controlled mtDNA deletions prevent ROS damage by arresting oxidative phosphorylation. Elife 2022, 11, e76095. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Youle, R.J.; Finkel, T. The Mitochondrial Basis of Aging. Mol. Cell. 2016, 61, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, N.S.; Wilkins, H.M. Mitophagy and the Brain. Int. J. Mol. Sci. 2020, 21, 9661. [Google Scholar] [CrossRef]
- Varghese, N.; Werner, S.; Grimm, A.; Eckert, A. Dietary Mitophagy Enhancer: A Strategy for Healthy Brain Aging? Antioxidants 2020, 9, 932. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.; Lizama, B.N.; Chu, C.T. Excitotoxicity, calcium and mitochondria: a triad in synaptic neurodegeneration. Transl. Neurodegener. 2022, 11, 3. [Google Scholar] [CrossRef]
- Victorelli, S.; Salmonowicz, H.; Chapman, J.; Martini, H.; Vizioli, M.G.; Riley, J.S.; Cloix, C.; Hall-Younger, E.; Machado Espindola-Netto, J.; Jurk, D.; Lagnado, A.B.; Sales Gomez, L.; Farr, J.N.; Saul, D.; Reed, R.; Kelly, G.; Eppard, M.; Greaves, L.C.; Dou, Z.; Pirius, N.; Szczepanowska, K.; Porritt, R.A.; Huang, H.; Huang, T.Y.; Mann, D.A.; Masuda, C.A.; Khosla, S.; Dai, H.; Kaufmann, S.H.; Zacharioudakis, E.; Gavathiotis, E.; LeBrasseur, N.K.; Lei, X.; Sainz, A.G.; Korolchuk, V.I.; Adams, P.D.; Shadel, G.S.; Tait, S.W.G.; Passos, J.F. Apoptotic stress causes mtDNA release during senescence and drives the SASP. Nature 2023, 622, 627–636. [Google Scholar] [CrossRef]
- Vizioli, M.G.; Liu, T.; Miller, K.N.; Robertson, N.A.; Gilroy, K.; Lagnado, A.B.; Perez-Garcia, A.; Kiourtis, C.; Dasgupta, N.; Lei, X.; Kruger, P.J.; Nixon, C.; Clark, W.; Jurk, D.; Bird, T.G.; Passos, J.F.; Berger, S.L.; Dou, Z.; Adams, P.D. Mitochondria-to-nucleus retrograde signaling drives formation of cytoplasmic chromatin and inflammation in senescence. Genes Dev. 2020, 34, 428–445. [Google Scholar] [CrossRef]
- Walters, G.C.; Usachev, Y.M. Mitochondrial calcium cycling in neuronal function and neurodegeneration. Front. Cell Dev. Biol. 2023, 11, 1094356. [Google Scholar] [CrossRef]
- Wan, W.; Hua, F.; Fang, P.; Li, C.; Deng, F.; Chen, S.; Ying, J.; Wang, X. Regulation of Mitophagy by Sirtuin Family Proteins: A Vital Role in Aging and Age-Related Diseases. Front. Aging Neurosci. 2022, 14, 845330. [Google Scholar] [CrossRef]
- Wang, R.; Reddy, P.H. Role of Glutamate and NMDA Receptors in Alzheimer’s Disease. J. Alzheimers Dis. 2017, 57, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Lin, L.; Zhang, Q.; Yang, J.; Kamili, E.; Chu, J.; Li, X.; Yang, S.; Xu, Y. Heteroplasmy and Individual Mitogene Pools: Characteristics and Potential Roles in Ecological Studies. Biology 2023, 12, 1452. [Google Scholar] [CrossRef]
- Yingze, Y.; Zhihong, J.; Tong, J.; et al. NOX2-mediated reactive oxygen species are double-edged swords in focal cerebral ischemia in mice. J. Neuroinflammation 2022, 19, 184. [Google Scholar] [CrossRef] [PubMed]
- Zanini, G.; Selleri, V.; Lopez Domenech, S.; Malerba, M.; Nasi, M.; Mattioli, A.V.; Pinti, M. Mitochondrial DNA as inflammatory DAMP: a warning of an aging immune system? Biochem. Soc. Trans. 2023, 51, 735–745. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).