1. Introduction
Pathogenic microorganisms, particularly infectious viral diseases, continue to threaten human society worldwide [
1,
2,
3]. The majority of these diseases are caused by RNA viruses, including influenza A virus (IAV), which have resulted in catastrophic damage to the national economy and public health [
4,
5]. The high genetic diversity of RNA viruses accelerates evolutionary adaptation, leading to the acquisition of drug resistance against previously developed vaccines and drugs [
6]. Therefore, exploring novel antiviral agents sourced from natural substances has been a critical focus of research, as numerous studies in history have supported their safety and effectiveness [
7].
Natural compounds from marine microbes have recently garnered significant attention in drug development due to their anti-inflammatory, antitumor, antimicrobial, antiviral, antimalarial, and antioxidant activities [
7,
8,
9,
10,
11,
12]. Every year, a large number of natural products with antiviral potential are discovered, some of which have even advanced to clinical trials and commercial use [
9,
13]. Alkaloids, peptides, polyketides, and terpenoids with diverse biological functions have been found in the secondary metabolites of marine bacteria. For instance, cyanobacterial-derived cyanovirin N has been evaluated as an anti-HIV agent in preclinical studies [
14]. However, natural compounds of marine microbes have been minimally studied in veterinary science due to a lack of understanding of their mechanism of action in preclinical research.
The identification of antiviral agents derived from marine microbes has emerged as a promising approach to controlling a broad spectrum of animal viral infections. Prominently, an extract of
Mameliella sp. has been found to exhibit significant protection from IAV infection, presenting a potential broad anti-influenza virus activity [
8]. A comprehensive analysis of marine microbes has led to the identification of a novel bacterium,
Pseudomonas sp. M20A4R8, which exhibits potent antiviral activity. The pre-and post-treatment of its extract showed a tremendous therapeutic index against influenza virus A/Puerto Rico/8/34 (H1N1) (A/PR8), with a half maximal inhibitory concentration (IC
50) of 1.2 µg/mL and selectivity index
(SI) of 1186.
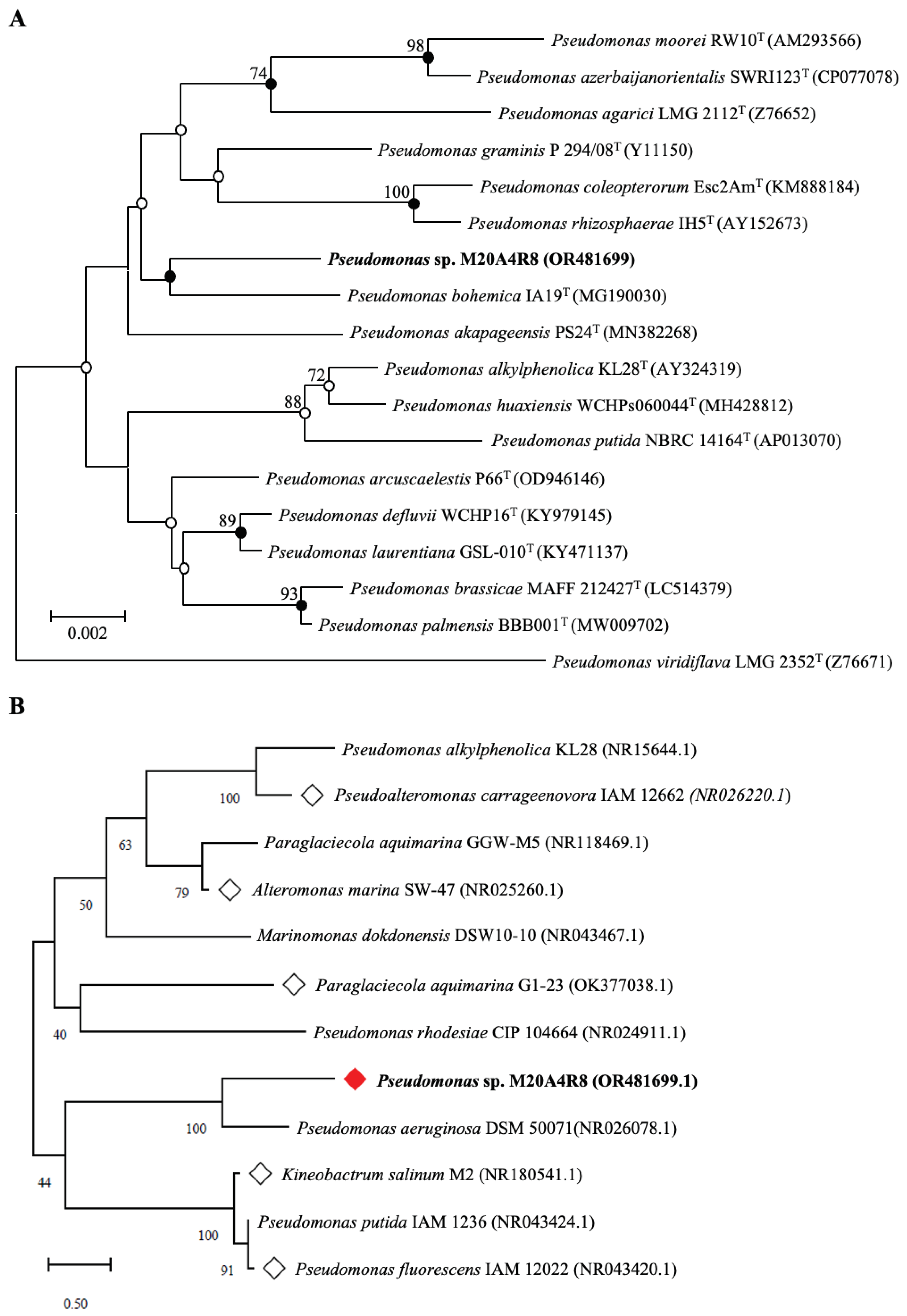
Phylogenetic analysis of the 16S rRNA gene indicated that M20A4R8 strain belonged to the genus Pseudomonas of the family Pseudomonadaceae, showing the highest similarity to Pseudomonas bohemica (98.56%). Notably, the post-treatment of the extract demonstrated remarkable inhibition of viral replication, protein synthesis, and progeny production, while pre- or co-treatment did not have a sufficient inhibitory effect. This observation suggests that the antiviral response was induced during the post-entry stages of viral replication. It further highlights the potential of post-treatment therapy as a practical approach against viral infections.
In the context of the molecular replication of the influenza virus, it is crucial to induce apoptosis by counteracting anti-apoptotic host proteins or stimulating autophagy-mediated apoptosis [
37,
47]. Furthermore, virus-induced apoptosis is imperative to promote virus propagation and the development of severe lung injuries such as acute respiratory disease complex (ARDS) [
48,
49,
50]. Significantly, the M20A4R8 extract has suppressed apoptotic cell death, and its extract has exhibited remarkable antiviral activity and safety against multiple influenza virus infections, including A/PR8 (IC
50 = 1.4 µg/mL, SI = 823.6), influenza virus A/Wisconsin/15/2009 (H3N2) (A/Wisconsin) (IC
50 = 8.1 µg/mL, SI = 146.2), and B/Florida/78/2015 Victoria lineage (B/Florida) (IC
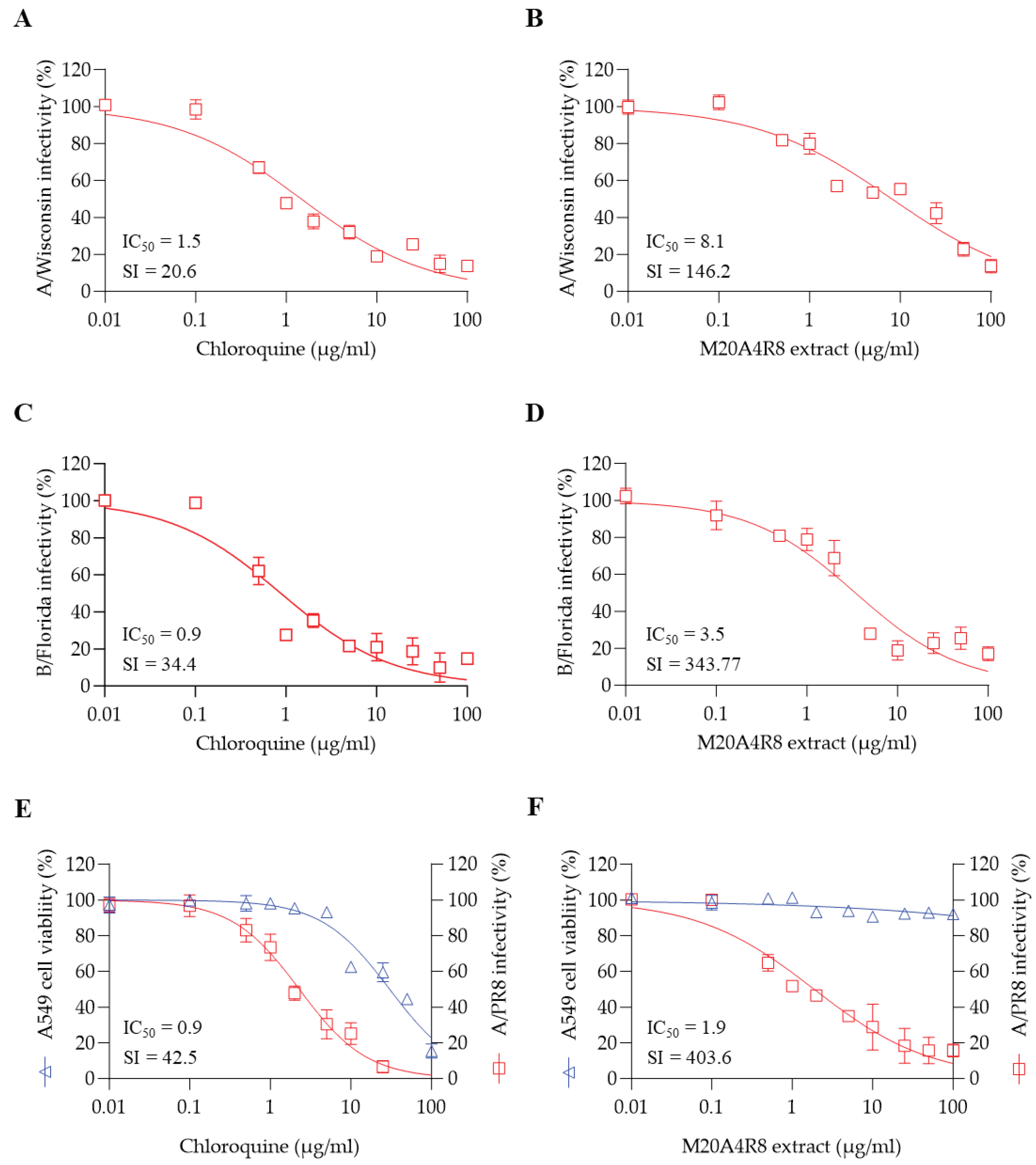
50 = 3.5 µg/mL, SI = 343.8), suggesting a broad anti-influenza virus activity. Therefore, it is plausible that the antiviral mechanism that attenuates apoptosis could be a potential target for disease control and better prognosis.
This study aims to investigate the antiviral properties of a novel strain of Pseudomonas and elucidate its therapeutic potential, which could provide new insights into preventing a broad spectrum of viral infections. After characterizing the single or combined compounds within the extract and explaining the anti-apoptotic mechanism, the therapeutic use of M20A4R8 strain could be warranted for preclinical treatment to develop a broad-spectrum antiviral drug. Future studies on the single or combined compound from the extract will be promising, encouraging its use in preclinical challenge tests with various influenza virus strains.
2. Materials and Methods
2.1. Isolation of the Bacterial Strain and Culture Conditions
Bacteria, including Pseudomonas, Pseudoalteromonas, Paraglaciecola, Alteromonas, Marinomonas, and Kineobactrum species, were procured from the coastal seawater collected in Dangjin (Chungnam Province, KR) on March 16th, 2020. For strain isolation, small sections of R. crispus roots were homogenized, diluted with sterile 0.85% saline, and spread on 1/10 diluted ZoBell medium (0.5 g peptone, 0.1 g yeast extract, and 0.001 g FePO4 per liter of 20% distilled water and 80% aged seawater). The inoculated plates were then incubated at 25°C for 5 days, followed by streaking of individual colonies based on morphological differences on marine agar 2216 (MA; Difco). After primary isolation and purification, the strain was routinely cultured on MA at 25°C and preserved with 20% (v/v) glycerol at -80°C. The bacterial isolates were deposited under the numbers MI0000628, MI00006266, MI00006269, MI00006270, MI00006279, and MI00006281, respectively, at the Microbial Marine BioBank (MMBB) of the National Marine Biodiversity Institute of Korea (MABIK).
2.2. Phylogeny Analysis of 16S rRNA Gene
Genomic DNA was extracted using an Exgene DNA extraction kit (Gene All, Seoul, KR) as per the manufacturer's instructions for amplifying the 16S rRNA gene with the bacteria-specific universal primers 27F and 1492R [
15]. The amplified partial 16S rRNA gene sequences were performed with an Applied Biosystems automated sequencer (ABI 3730XL) at Macrogen Co. Ltd. (Seoul, KR) and assembled by Geneious program v9.0.5 to obtain a nearly full-length 16S rRNA gene sequence. Identification of the phylogenetic position of M20A4R8 strain was searched using the EzBioCloud server (ezbiocloud.net/identify) [
16], and the 16S rRNA gene sequence (1,494 nucleotides) was compared with validly published species in the server. The 16S rRNA gene sequence has been deposited into GenBank under the accession OR481699. Phylogenetic trees based on 1,419 unambiguously aligned sequences were reconstructed using the neighbor-joining (NJ), maximum-likelihood (ML), and maximum-parsimony (MP) algorithms [
17,
18,
19] in the MEGA X [
20]. To evaluate the robustness of the tree topologies, the 1,000 bootstrap resampled datasets [
21] were performed in the three algorithms. M20A4R8 strain and test samples from other gammaproteobacterial classes were also compared via 16S rRNA gene sequences. Phylogenetic trees were performed in MEGA the 1,000 bootstrap resampled datasets were performed in the three algorithms as above.
2.3. Preparation of the Bacterial Extracts
Bacterial extracts were prepared using a slight modification of the method from Choi et al [
22]. Bacteria, including
Pseudoalteromonas, Alteromonas, Paraglaciecola, Pseudomonas and Kineobactrum species, were first cultured in 2.5 L Erlenmeyer flasks each containing 1 L of marine broth (total 5 L) under 60 µmol m-2 s-1 LED light at 25℃. Subsequently, 20 L of culture using a panel or column-type photobioreactor was inoculated with the initial inoculum at a concentration of 10
4 CFU mL-1 in the same culture condition for 10-20 days with shaking at 150 rpm. At the end of the culture period, culture broth was extracted twice with the same volume of ethyl acetate (EtOAc). The EtOAc soluble component of bacterial culture broth was combined and dried using a vacuum evaporator. A crude total of 150 mg of bacterial extract was obtained from each species. For the antiviral evaluation, the extracts were dissolved in dimethyl sulfoxide (DMSO).
2.4. Cells and Virus
The influenza virus A/Puerto Rico/8/34 (H1N1) (A/PR8), A/Wisconsin/15/2009 (H3N2) (A/Wisconsin) strain, and B/Florida/78/2015 Victoria lineage (B/Florida) were amplified in MDCK cells. The viruses were cultured for 2-3 days in a culture medium supplemented with 1 µg/mL TPCK-treated trypsin. After collecting centrifuged supernatant, the titer was measured using a cell culture immunofluorescence (CCIF) assay with a monoclonal antibody (Mab) specific for IAV nucleoprotein (NP) protein and expressed as fluorescence focus units per milliliter (FFU/mL) [
23].
2.5. Cytotoxicity Assessment
A water-soluble tetrazolium salt (WST) assay was used to determine the viability of the cells with CellVia cell viability assay kit (AbFrontier, Seoul, KR) as per the product guide protocol. MDCK cells were seeded in 96-well microplates and treated with each Pseudomonas sp. the next day. M20A4R8 extract at the following non-cytotoxic concentrations in triplication: 1000, 500, 250, 100, 50, 25, 10, 5, 2, 1, 0.5, 0.1, 0.01 µg/mL. After 48 hours of incubation at 37℃, the treated cells were washed with DPBS (Lonza, Basel, CH) and 50 µL CellVia solution diluted 10 times to DMEM in each well. For 30 minutes, the microplate was incubated at 37℃. The wells' Optical density (OD) was measured at 450 nm, and the dose-response curve was plotted using GraphPad Prism 9.5.1 (GraphPad Software Inc., San Diego, CA). Extract cytotoxicity was assessed by the formula: cytotoxicity (%) = (O.Dextract – O.DBlank)/ (O.Dmock – ODBlank) x100.
2.6. Antiviral Screening Assay
The bacterial extract was applied before and after IAV inoculation at an MOI of 0.1 in MDCK cells to confirm general antiviral activity, as described elsewhere [
8,
24]. Before virus infection, the cells were incubated with serially diluted
Pseudomonas sp. M20A4R8 extract (starting from concentrations of 500, 250, 125, 62.5, 31.3, 15.6, 5, 1, and 0.1 µg/mL, diluted with DMEM) at 37℃ for 1 h. Subsequently, cells were washed with DPBS and infected with IAV (0.1 MOI) at 37℃ for 1 h. Following virus adsorption, cells were washed and treated with the same concentration of bacterial extract as previously described, supplemented with 1 µg/mL TPCK-treated trypsin (Thermo Fisher Scientific).
2.7. Time-of-Addition (Application of Drug at Different Time Points) Antiviral Experiment
A set of time-of-addition with different time points of drug treatment was carried out as follows: treatment before (pre-treatment), during (co-treatment), or after (post-treatment) viral infection at 1 h intervals as previously reported [
8,
25]. For pre-treatment, the cell was incubated with extract for 1 h, washed with DMEM, and infected with IAV (0.1 MOI). After 1 h, the cell was cleaned and replaced with DMEM supplemented with 1 µg/mL TPCK-treated trypsin. For co-treatment, MDCK cells were treated simultaneously with virus and extracts, incubated at 37℃ for 1 h, washed, and replaced with DMEM supplemented with 1 µg/mL TPCK-treated trypsin. For post-treatment, the cell was infected with the virus. At 1 h, the cell was washed, and different concentrations of
Pseudomonas sp. were added.
Pseudomonas sp. M20A4R8 extract diluent with DMEM supplemented with 1 µg/mL TPCK-treated trypsin. Each group was examined in triplicate in 96-well plates, and the supernatants were collected by extract concentration at 48 hours post-infection (hpi) for further investigation.
Concerning the antiviral experiment, the half maximal inhibitory concentration (IC
50) was measured at 570 nm through MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (Sigma-Aldrich) as previously reported [
26,
27]. After 48 hpi, cells were washed with DPBS and then incubated with 200 µL of fresh 0.4 mg/mL of MTT solution dissolved in PBS (pH 7.4) at 37℃ for 4 h. Subsequently, supernatants were removed, DMSO was added to dissolve formazan crystals. The plate was placed on a shaker for 10 minutes, and absorbance was measured. The inhibition effect was calculated using the following formula: Inhibition effect (%) = (OD
extract – OD
virus mock)/(OD
mock – OD
virus mock)*100 [
28].
The half cytotoxic concentration (CC50) and IC50 of the Pseudomonas sp. M20A4R8 extract was calculated after the generation of the dose-response curve using GraphPad Prism 9.5.1. SI was determined as the ratio of CC50 to IC50 for each group.
2.8. Virus Attachment and Penetration Assays
As previously reported, MDCK cells were seeded in 24 healthy plates to determine the effect on virus attachment and penetration into cells [
29]. In the attachment assay, cells were treated with 50 µg/mL
Pseudomonas sp. M20A4R8 extract at 4℃ for 30 min. After removing the extract and washing to DPBS, the cells were infected with the virus an MOI 0.1 and kept at 4℃ for 1 h. The unattached virus was removed by washing with DPBS, and after incubation at 37℃ for 20 hours, the inhibitory activity of the extracts on virus attachment to cells was measured via relative genome copies by RT-qPCR. An antibody against HA of influenza A virus was used as a positive drug for comparison. In the penetration assay, pre-chilled cells were exposed to the virus while maintained at 4℃ for 1 h. The unattached virus was removed by washing three times with cold DPBS, and the 50 µg/mL extract was treated at 37℃ for 10 min. To remove the unpenetrated virus, the cells were washed with Tris buffer at a low pH of 3.0; a medium was added and incubated at 37℃ for 20 h. Likewise, the virus in the supernatant was detected through RT-qPCR to measure relative genome copies, and the anti-penetrate effect of the extract was determined. As a favorable drug for comparison, chloroquine, a well-known commercial drug, was used.
2.9. Real-Time Quantitative PCR
The reduction of genome copy numbers was confirmed by RT-qPCR quantification of influenza and coronavirus treated with extracts as previously described [
30,
31]. RNA was extracted from the supernatant of each group of antiviral tests using the QIAamp Viral RNA Mini kit following the handbook's instructions (QIAGEN, Hilden, DE). RT-qPCR was performed through the SensiFAST SYBR Lo-ROX One-Step kit (Meridian Bioscience, CIN, OH, US) using the following primer pairs; IAV PB1 (Forward, 5’-GGC CCT TCA GTT CAT C-3', Reverse 5’-GCA GAC TTC AGG AAT GTG-3'). The reaction mixture used consists of 2 µL of RNA template, 10 µL of 2X One-step mix, 1.6 µL of primer (forward + reverse), 0.2 µL of Reverse transcriptase, and 0.4 µL RNase inhibitor. Water was filled up to the final total reaction volume for each sample of 20 µL. The cycling condition was performed as follows: 45°C for 30 m and 95°C for 10 m, and then the three-step reaction of 95°C for 10 s, 60°C for 10 s, and 72°C for 15 s was repeated 40 times.
2.10. TCID50 Assay
To measure infection viral progeny to MDCK cells, a tissue culture infective dose (TCID
50) was computed using the Reed and Muench method [
32]. After washing MDCK cells in a 96-well plate with DMEM, the supernatants of each treatment were re-adapted with 10-fold serial dilution with DMEM supplemented 1 µg/mL TPCK-treated trypsin and incubated at 37℃ with 5% M. At 5 days post-infection, DMEM was removed, cells were fixed with 4% PFA buffer (Bio-solution, Seoul, KR) for 10 minutes at room temperature, and the supernatant was discarded and washed with tap water. Control groups were treated with only DMEM without trypsin. For staining, a 5% crystal violet solution (DUKSAN) was used, and the titer of influenza A virus was expressed as TCID
50/mL value.
2.11. Immunofluorescence Assay (IFA)
IFA was performed to determine the protein of viral progeny. MDCK monolayers grown on 8-well chamber glass slides were infected or not with influenza virus A/PR8 at an MOI 1 with 1 µg/mL TPCK-treated trypsin medium as previously reported [
23]. After incubating at 37℃ with a 5% CO
2 atmosphere for 24 h, the cell was fixed with cold 4% paraformaldehyde for 10 min and permeabilized for 10 min with 0.2% Triton X-100. The cell was blocked at room temperature for 1 h with 0.5% BSA in PBS and incubated 4℃ overnight with the primary antibody on a shaker. After washing twice with PBS, anti-mouse conjugated to AF488 (diluted 1:200), the secondary antibody was incubated for 1 h at room temperature in the dark. DAPI was used to stain the nucleus, and the cells were observed under an LSM 510 confocal microscope.
2.12. Flow Cytometry of DNA Strand Break
The levels of apoptosis were quantified to determine the impact of treatment with
Pseudomonas sp. M20A4R8 extract and IAV infection on cell death and viral replication. To the previous report that apoptosis-related endonucleases cause DNA cleavage, dUTP labeling was used to detect DNA with 3′hydroxyl-terminal [
33,
34]. Cells were prepared in a 6-well plate at 90% confluence and incubated for 24 h with or without virus and drug at 37℃. Cells were detached from the plate by trypsin treatment, and the pellet was harvested by centrifugation at 3,000 rpm for 2 min. After washing with PBS, it was fixed with cold 4% paraformaldehyde for 10 min and permeabilized for 10 min with 0.2% Triton X-100. The cell was blocked at room temperature for 1 h with 0.5% BSA in PBS, incubated 4℃ overnight with primary antibody and apoptotic markers on a shaker. After washing twice with PBS, anti-rabbit conjugated to AF647 (diluted 1:200), the secondary antibody was incubated at room temperature for 1 h. Each sample data was digitized under the same conditions of measurement using AttuneTM NxT flow cytometer and AttuneTM NxT software v3.1.2 as previously reported [
23].
2.13. Statistical Analysis
All data were analyzed using the GraphPad Prism software program version 9.5.1, and results are presented as mean ± the standard error of the mean (SEM). The statistical analysis was carried out by one-way analysis of variance (ANOVA) with Tukey's correction for multiple comparisons or independent sample t-tests (and nonparametric tests), individually annotated in the figure legend. Statistical significance of data was marked as follows: * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
3. Results
3.1. DNA-Based Identification and Phylogenetic
Comparative analysis of the 16S rRNA gene showed that M20A4R8 strain was closely related to Pseudomonas bohemica IA19T with a similarity of 98.56%. Phylogenetic analysis of the 16S rRNA gene using the NJ, ML, and MP algorithms showed that M20A4R8 strain formed a monophyletic clade with other members of the genus Pseudomonas (
Figure 1A). Consequently, sequence comparisons based on almost complete 16S rRNA gene sequences showed a clear affiliation of the isolate to the genus Pseudomonas.
3.2. In Vitro Antiviral Screening for Extract of Pseudomonas sp. M20A4R8 Extract
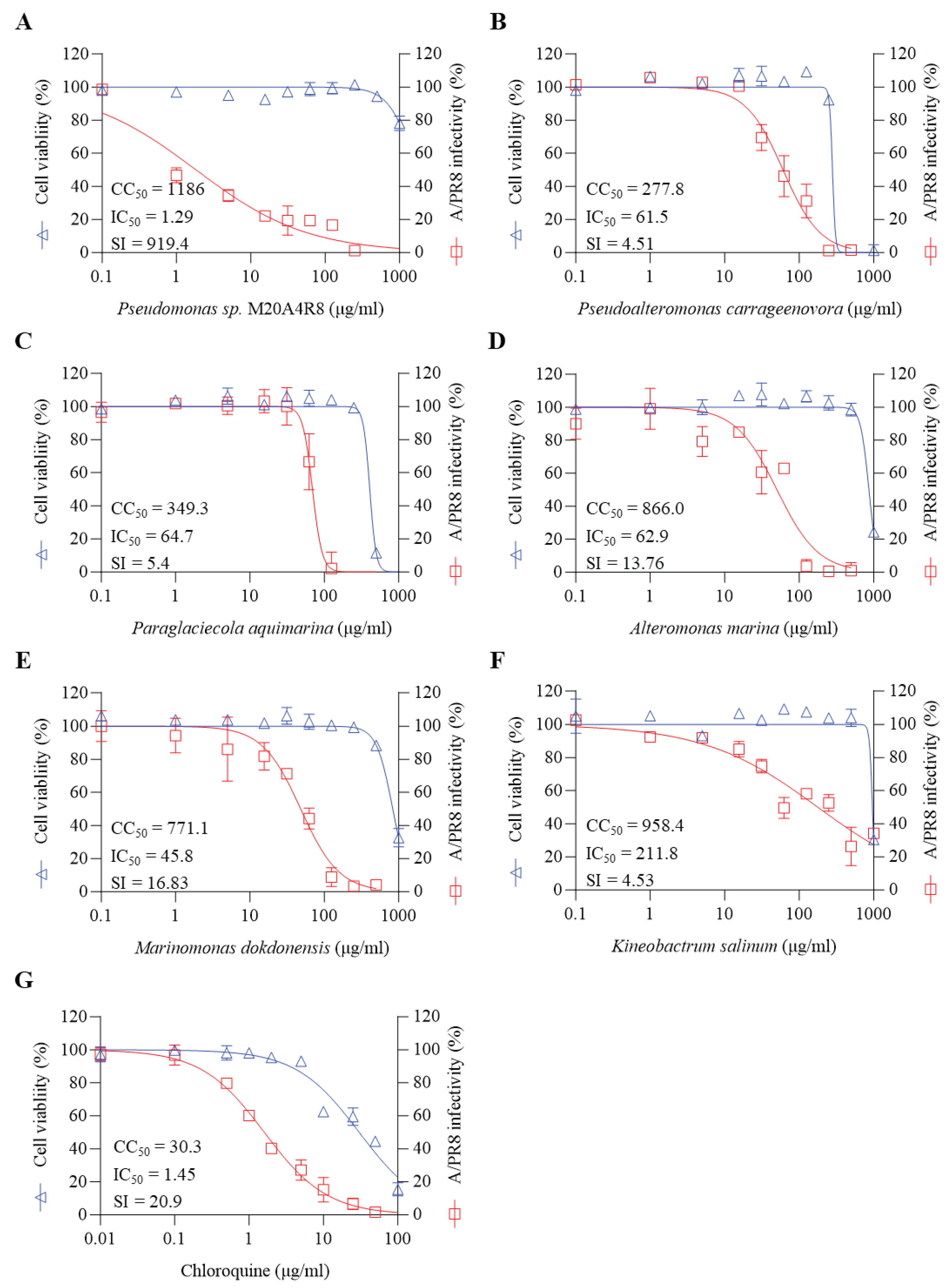
We investigated the inhibitory efficacy of extracts of the same gammaproteobacteria class related to Pseudomonas sp. M20A4R8 strain against influenza A virus(A/PR8 strain) (
Figure 1B,
Figure 2). Antiviral efficacy was mainly assessed using two main indices: CC
50 and IC
50. Briefly, vehicle, chloroquine, an FDA-approved antiviral drug, or one of the bacterial extracts were applied before and after either mock or IAV (A/PR8) inoculation at an MOI of 0.1. Antiviral activity against the A/PR8 strain was defined by an inhibitory effect on cytopathic effect (CPE) using the MTT method with slight modification after 48 h incubation [
26,
35]. As a result, IC
50 and SI of chloroquine were calculated as 1.45 and 20.9, respectively. Pseudoalteromonas carrageenovora, Paraglaciecola aquimarina and Kineobactrum salinum extracts showed low SI, whereas Alteromonas marina and Marinomonas dokdonensis extract had low cytoxicity and relatively high SI over 10. In particular, Pseudomonas sp. M20A4R8 extracts showed high safety and antiviral effectiveness, showing dramatic SI over 900 (
Figure 2).
3.3. Antiviral Response of M20A4R8 Extract according to extract treatment time against influenza virus
Pseudomonas sp. M20A4R8 extract was applied in three different approaches: pre-treatment (
Figure S1A-C), co-treatment (
Figure S1D-F) and post-treatment (
Figure 3). To determine whether the extract had a preventive effect by affecting the viral receptors in cells, the extract was pretreated 1 hr before viral infection (
Figure S1A). Subsequently, the cells were infected with IAV at an MOI of 0.1 and antiviral evaluation was performed at 48 hr after infection. However, the antiviral effect was not observed at any concentration (
Figure S1B,C).
To determine whether the extract directly interfered with the attachment or entry of the influenza virus into host cells, the virus and the extract were treated simultaneously with the cells. The viral inoculum (at an MOI of 0.1) was mixed with the extract and absorbed into the MDCK cell. After 48 h, the cell pellet and supernatant were analyzed with the experiments above (
Figure S1D). A serial concentration (from 0.01 to 100 µg/mL) of M20A4R8 extract did not show antiviral activity on CPE and viral genome replication by A/PR8 infection (
Figure S1E,F), suggesting that A/PR8 infections were not prevented by the pre- or co-treatment.
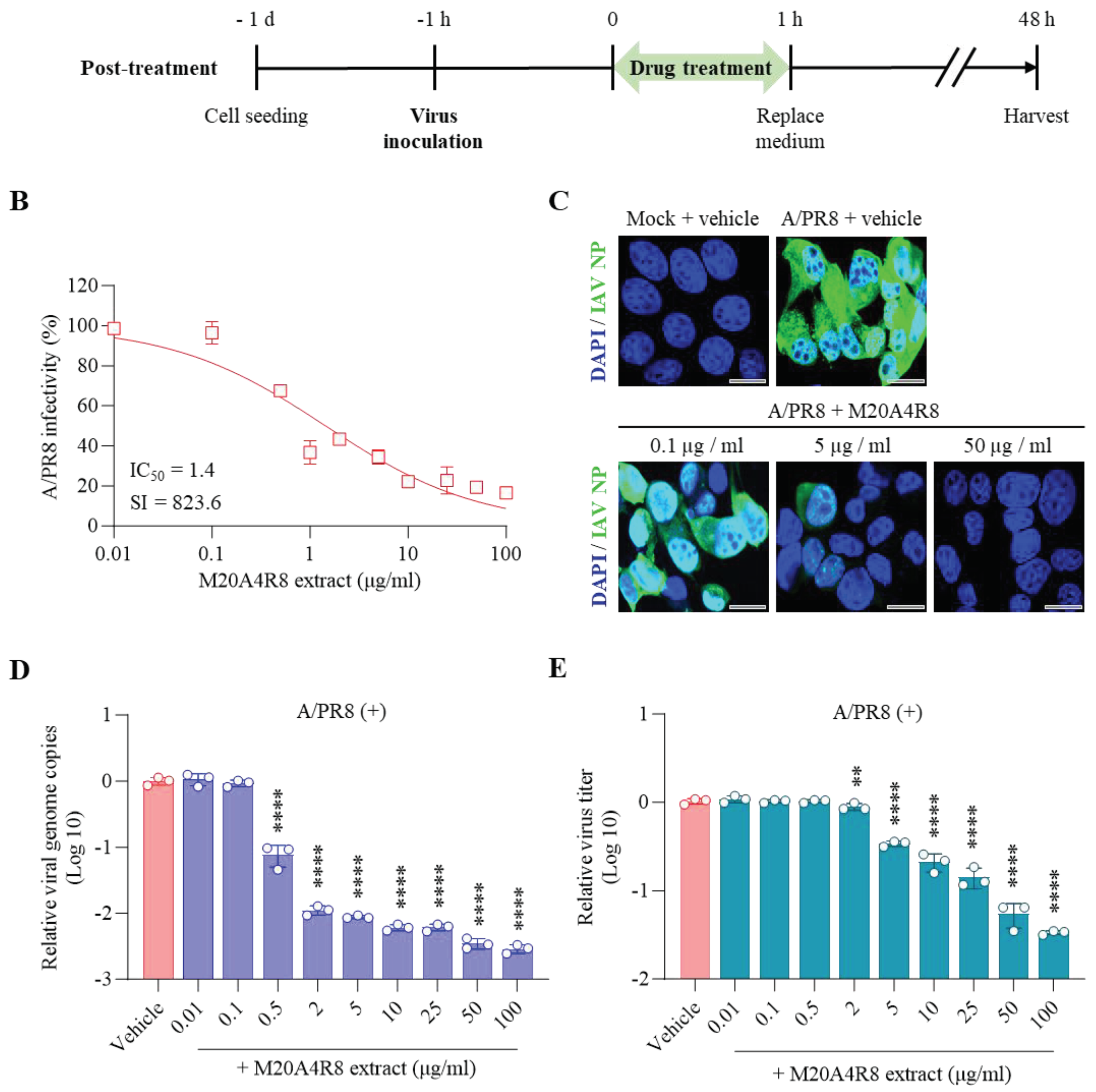
Finally, the extract was post-treated 1 hr after virus infection to determine whether the extract affected later stages, such as the virus replication or synthesis (
Figure 3A). Briefly, MDCK cells infected with the A/PR8 strain (0.1 or 1 MOI) received the extract at various concentrations as above. As a result, the extract showed remarkable inhibition of CPE induced by IAV in a dose-dependent manner, producing a great therapeutic index (IC
50 = 1.4 µg/mL, SI = 823.6) in comparison with chloroquine (IC
50 = 1.5 µg/mL, SI = 20.9, a commercial anti-IAV drug) (
Figure 3B). In parallel, the IFA results showed gradual inhibition of viral protein synthesis toward higher concentrations of M20A4R8 extract (
Figure 3C). Moreover, viral genome replication and progeny production were significantly suppressed by the post-treatment of M20A4R8 extract (
Figure 3D-E).
3.4. Protection of Host through M20A4R8 Extract from IAV-Induced Cell Death
To specify the initiation of antiviral response mediated by M20A4R8 extract, virus attachment and penetration assays were performed in MDCK cells infected with the A/PR8 strain (1 MOI) (
Figure S2). As a result, MDCK cells pre-incubated with M20A4R8 did not suppress virus binding to a cellular receptor, in comparison with the group applied with an inoculum mixture of IAV and an antibody against IAV HA before inoculation on the host cells (
Figure S2A). Interestingly, the cells post-treated with the extract after the virus binding to the host significantly reduced viral genomes (
Figure S2B). However, it was considered that the antiviral action would only last for a short time, which was insufficient to curb severe infection, given that the pre-and co-treatment resulted in no inhibition of viral infection during long-time incubation at 48 h. In other words, the potent antiviral activity in
Figure 3 was highly expected to be linked to the late stage of viral replication or post-entry stages.
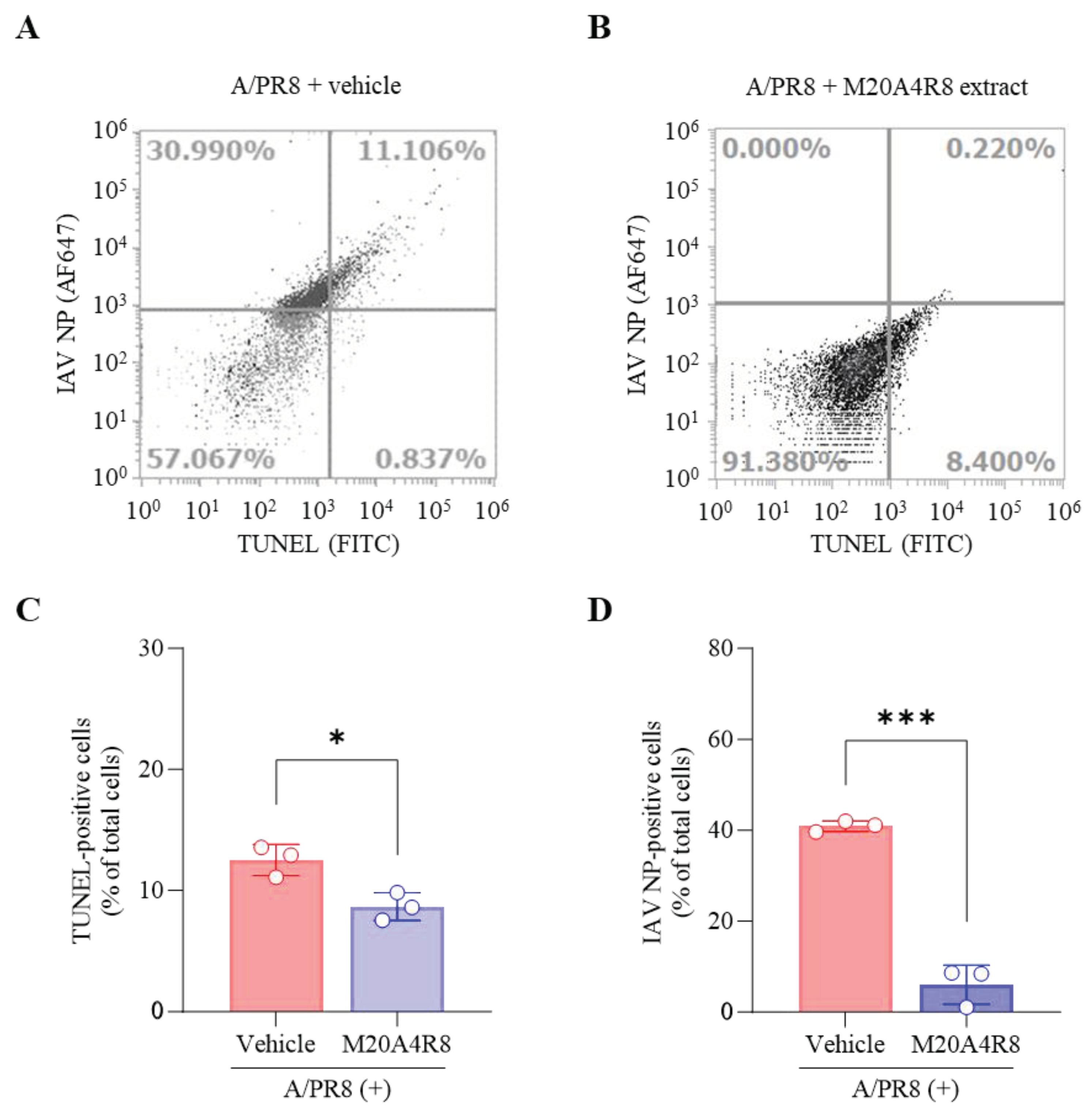
IAV promoted host cell death by induction of the mitochondrial apoptosis pathway. Many studies have found that IAV-induced apoptosis facilitates viral replication by disseminating and killing the host immune effectors [
36,
37]. Apoptotic cell death was analyzed by flow cytometry to evaluate host cell protection from IAV infection (
Figure 4). Apoptotic DNA fragmentations were detected by TUNEL labeling with FITC, and IAV-infected cells were recognized by monoclonal antibody against IAV NP conjugated with Alexa Fluor (AF) 647. The A/PR8-infected, vehicle-treated group showed apoptotic cell death (approximately 12%) by severe viral replication (about 42%) in the host (
Figure 4A). However, post-treatment of M20A4R8 extract could counteract virus-induced cell death, dramatically reducing viral replication (approximately 0.2%) (
Figure 4B). The antiviral response was evidenced by a significant reduction of apoptosis (
Figure 4C) and viral proteins (
Figure 4D). Collectively, our data strongly indicated that the bacterial extract efficiently protects the host in parallel with the attenuation of apoptotic cell death.
4. Discussion
The emergence and re-emergence of infectious RNA viruses pose a constant threat to public health and global economies. Existing vaccines and antiviral drugs are often rendered ineffective due to the evasion of the host immune response or genetic mutations, leading to the accumulation of pandemic risks. Developing new antiviral lead agents to combat problematic RNA viruses has become an urgent necessity.
In this study, in vitro antiviral screening has resulted in the identification of a bacterial extract exhibiting significant antiviral activity against the PR8 strain of influenza A virus, with an IC50 of 1.3 µg/mL and an SI of 919.4. The level of activity displayed by this extract is comparable to that of chloroquine, an FDA-approved antiviral drug, and is an encouraging indication of its safety and efficacy, with the potential to be a potent weapon against IAV infection. The bacterial extract, identified as Pseudomonas sp. M20A4R8 is a new strain of Pseudomonas initially reported in seawater worldwide.
Notably,
Pseudomonas aeruginosa, a well-known strain of the
Pseudomonas genus, is an opportunistic pathogen that causes cystic fibrosis, burns wounds, immunodeficiency, and chronic obstructive pulmonary disorder [
39,
40]. However, antiviral peptides derived from
Pseudomonas chlororaphis O6 or
Pseudomonas sp. can effectively inhibit viral replication against Tobacco Mosaic Virus, Oncorhynchus masou virus, and infectious hematopoietic necrosis virus [
41]. Therefore, further metabolic and chemical investigations are imperative to identify the specific component(s) or metabolite(s) responsible for the antiviral activity of
Pseudomonas sp. M20A4R8.
Many antiviral therapeutics have been found to disrupt particular stages of viral replication, such as chloroquine, a drug preventing endosomal acidification, and remdesivir, a drug suppressing RNA-dependent RNA polymerase of SARS-CoV-2 [
40,
42,
43,
44]. Previous studies have suggested plausible explanations for the antiviral mechanisms of
Pseudomonas sp. [
45,
46]. For instance,
Pseudomonas aeruginosa (strain PM1) been reported to exhibit inhibition of viral attachment against tobacco mosaic virus and Sunn-hemp rosette virus [
45]. In addition,
Pseudomonas aeruginosa has been found to enhance acute inflammation in the airway, promoting neutrophil response and antiviral activity against rhinovirus [
46]. Time-of-addition experiments have been carried out to investigate the related mechanisms of the antiviral action of
Pseudomonas sp. M20A4R8. The inhibitory effects were not involved in viral attachment or direct interaction with virus particles according to pre- and co-treatment and attachment and penetration assays. As a result, post-treatment has been found to be the most effective approach, inhibiting genome replication and protein synthesis with a remarkable therapeutic index against influenza virus A/PR8, with an IC
50 of 1.4 μg/mL and an SI of 823.6. This approach is more adaptable in clinical uses and a more feasible strategy for controlling epidemic diseases than prophylactic therapy.
Influenza virus infections have been found to induce apoptosis mediated by a multifactorial process. The mitochondrial apoptosis pathway is mainly promoted by IAV NP, resulting from the counteraction of cellular anti-apoptotic protein Clusterin [
37]. The influenza virus also stimulates host autophagy, activating Bax/caspase-dependent apoptosis [
47]. Modulation of virus-induced apoptosis is essential in host protection, preventing the death of immune effectors and secondary bacterial infection to respiratory organs.
Pseudomonas sp. M20A4R8 extract efficiently suppresses IAV-induced apoptosis and dramatically reduces viral replication. In another way, it is due to a reduced amount of viral protein that could not stimulate pattern recognition receptors (PRRs)-mediated apoptosis. In cases of pulmonary infection caused by pathogenic microorganisms, ARDS is a significant complication with a high percentage of fatality, especially those caused by the influenza virus. This is primarily due to apoptotic cell death and impaired efferocytosis [
48,
49,
50]. The interaction of viral components as a pathogen-associated molecular pattern with PRRs can induce a cytokine storm, resulting in severe lung injury [
23].
Moreover, the extract of Pseudomonas sp. M20A4R8 has demonstrated remarkable antiviral potential, exhibiting broad-spectrum antiviral activity against different strains of the influenza virus with outstanding therapeutic indexes. It has been found to be effective against influenza virus A/Wisconsin (IC50 = 8.1 µg/mL, SI = 146.2) and B/Florida (IC50 = 3.5 µg/mL, SI = 343.8) and has been shown to be effective in a lung epithelial cell line (A549) against A/PR8 infection (IC50 = 1.9 µg/mL, SI = 403.6), which is significantly higher than that of chloroquine. Although further investigation in primary human respiratory epithelial cells is required in future studies to see the therapeutic feasibility for clinical use, M20A4R8 extract showed great antiviral potential.
Toxicity represents a limiting factor in the therapeutic application of many drugs with known antiviral activity [
51,
52]. However, the bacterial extract has potently blocked different strains of influenza virus infection at low concentrations and has shown a relatively high selectivity index compared to FDA-approved chemicals [
53], suggesting its safety over effectiveness as a therapeutic agent. Nevertheless, further research is necessary to identify the specific compound in the M20A4R8 extract that modulates apoptosis through additional studies using liquid chromatography-tandem mass spectrometry. Future studies on individual or combined compounds from the bacterial extract will provide further insights into its antiviral properties.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Figure S1: Antiviral effect of M20A4R8 extract by pre-treatment (A-C) and co-treatment (D-F) against A/PR8 strain. (A) Schematic diagram of virus inoculation and extract treatment. (B) IC50 and SI of M20A4R8 extract measured by CPE-inhibition assay. (C) Viral genome copies detected by RT-qPCR. (D) Schematic diagram of virus inoculation and extract treatment. (E) IC50 and SI of M20A4R8 extract measured by CPE-inhibition assay. (F) Viral genome copies detected by RT-qPCR. All data in the graphs are presented as arithmetic means ± S.D. from 3 independent experiments; Figure S2: Viral genome copies measured at 20 h of incubation after virus attachment and penetration assays along with the treatment of M20A4R8 extract (50 µg/mL). (A) The extract was pre-treated before binding of the virus to the MDCK cells at 4°C for 1 h, followed by infection with A/PR8 strain (1 MOI). (B) After binding of A/PR8 strain (1 MOI) to the host cells, the extract was absorbed at 37°C for 1 h. Viral genome copies was measure by RT-qPCR using a pair of primer detecting IAV PB1. All data in the graphs are presented as arithmetic means ± S.D. from 3 independent experiments. HA, hemagglutinin. **, p < 0.01; ****, p < 0.0001.
Author Contributions
Conceptualization, S.-B. J. and G. C.; methodology, K.-S. M.; software, H.-J. K.; validation, Y. M. K. and J. M.; formal analysis, H. C.; investigation, S.-B. J., G. L., and K.-H N.; resources, G. C., M. Y. S., and J.-Y. Y.; data curation, K.-S. M.; writing—original draft preparation, S.-B. J.; writing—review and editing, G. C.; visualization, H.-J. K.; supervision, Y.-B. B.; project administration, J.-G. P.; funding acquisition, S.-I. P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (No. NRF-2022R1A2C1011742). This research was funded by an in-house grant from the National Marine Biodiversity Institute of Korea (grant number MABIK 2024M00600). This research was partially funded by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (Host-directed Antiviral Research Center, grant RS-2023-00219517).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data supporting the conclusions of this article are included within the article. Raw data are available from the corresponding author upon reasonable request.
Acknowledgments
We express our deepest gratitude to the National Marine Biodiversity Institute of Korea (MABIK) for providing the bacterial isolate which are deposited at the Microbial Marine Bio Bank (MMBB) under the number MI00006289.
Conflicts of Interest
The authors declare they have no conflict of interest.
References
- Bhadoria, P.; Gupta, G.; Agarwal, A. Viral Pandemics in the Past Two Decades: An Overview. J. Family Med. Prim. Care. 2021, 10, 2745–2750. [Google Scholar] [CrossRef]
- Paixao, E.S.; Teixeira, M.G.; Rodrigues, L.C. Zika, chikungunya and dengue: the causes and threats of new and re-emerging arboviral diseases. BMJ Glob. Health 2018, 3, e000530. [Google Scholar] [CrossRef]
- Pierson, T.C.; Diamond, M.S. The continued threat of emerging flaviviruses. Nat. Microbiol 2020, 5, 796–812. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.; Bhola, S.; Thakur, P.; Patel, S.K.S.; Kulshrestha, S.; Ratho, R.K.; Kumar, P. Waves and variants of SARS-CoV-2: understanding the causes and effect of the COVID-19 catastrophe. Infection 2022, 50, 309–325. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wu, T.; Liu, Q.; Yang, Z. The SARS-CoV-2 outbreak: What we know. Int. J. Infect. Dis. 2020, 94, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Munoz, S.; Upegui-Porras, N.; Gomez, A.P.; Ramirez-Nieto, G. Key Factors That Enable the Pandemic Potential of RNA Viruses and Inter-Species Transmission: A Systematic Review. Viruses 2021, 13. [Google Scholar] [CrossRef]
- Barzkar, N.; Tamadoni Jahromi, S.; Poorsaheli, H.B.; Vianello, F. Metabolites from Marine Microorganisms, Micro, and Macroalgae: Immense Scope for Pharmacology. Mar. Drugs 2019, 17. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Park, J.G.; Moon, K.S.; Jung, S.B.; Kwon, Y.M.; Kang, N.S.; Kim, J.H.; Nam, S.J.; Choi, G.; Baek, Y.B.; et al. Identification and characterization of a marine bacterium extract from Mameliella sp. M20D2D8 with antiviral effects against influenza A and B viruses. Arch. Virol. 2024, 169, 41. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2018, 35, 8–53. [Google Scholar] [CrossRef] [PubMed]
- Cheung, R.C.; Wong, J.H.; Pan, W.L.; Chan, Y.S.; Yin, C.M.; Dan, X.L.; Wang, H.X.; Fang, E.F.; Lam, S.K.; Ngai, P.H.; et al. Antifungal and antiviral products of marine organisms. Appl. Microbiol. Biotechnol. 2014, 98, 3475–3494. [Google Scholar] [CrossRef]
- Uzair, B.; Mahmood, Z.; Tabassum, S. Antiviral activity of natural products extracted from marine organisms. Bioimpacts 2011, 1, 203–211. [Google Scholar]
- Yasuhara-Bell, J.; Lu, Y. Marine compounds and their antiviral activities. Antiviral Res. 2010, 86, 231–240. [Google Scholar] [CrossRef]
- Yi, M.; Lin, S.; Zhang, B.; Jin, H.; Ding, L. Antiviral potential of natural products from marine microbes. Eur. J. Med. Chem. 2020, 207, 112790. [Google Scholar] [CrossRef]
- Yasuhara-Bell, J.; Yang, Y.; Barlow, R.; Trapido-Rosenthal, H.; Lu, Y. In vitro evaluation of marine-microorganism extracts for anti-viral activity. Virol. J. 2010, 7, 182. [Google Scholar] [CrossRef] [PubMed]
- Lane, D. 16S/23S rRNA sequencing. In Nucleic acid techniques in bacterial systematics; Stackebrandt., E., Goodfellow., M., Eds.; John Wiley & Sons: New York, USA, 1991. [Google Scholar]
- Lee, I.; Ouk Kim, Y.; Park, S.C.; Chun, J. OrthoANI: An improved algorithm and software for calculating average nucleotide identity. Int. J. Syst. Evol. Microbiol. 2016, 66, 1100–1103. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Evolutionary trees from DNA sequences: a maximum likelihood approach. J. Mol. Evol. 1981, 17, 368–376. [Google Scholar] [CrossRef]
- Fitch, W.M. Toward defining the course of evolution: minimum change for a specific tree topology. Systematic Biology 1971, 20, 406–416. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Mega, X. molecular evolutionary genetics analysis across computing platforms; S Kumar, G Stecher, M Li, C Knyaz, K Tamura. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar]
- Nawrocki, E.P.; Eddy, S.R. Query-dependent banding (QDB) for faster RNA similarity searches. PLoS Comput. Biol. 2007, 3, e56. [Google Scholar] [CrossRef]
- Choi, E.J.; Nam, S.J.; Paul, L.; Beatty, D.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Previously Uncultured Marine Bacteria Linked to Novel Alkaloid Production. Chem. Biol. 2015, 22, 1270–1279. [Google Scholar] [CrossRef]
- Baek, Y.B.; Kwon, H.J.; Sharif, M.; Lim, J.; Lee, I.C.; Ryu, Y.B.; Lee, J.I.; Kim, J.S.; Lee, Y.S.; Kim, D.H.; et al. Therapeutic strategy targeting host lipolysis limits infection by SARS-CoV-2 and influenza A virus. Signal Transduct. Target Ther. 2022, 7, 367. [Google Scholar] [CrossRef]
- Derksen, A.; Kuhn, J.; Hafezi, W.; Sendker, J.; Ehrhardt, C.; Ludwig, S.; Hensel, A. Antiviral activity of hydroalcoholic extract from Eupatorium perfoliatum L. against the attachment of influenza A virus. J. Ethnopharmacol 2016, 188, 144–152. [Google Scholar] [CrossRef]
- Li, Y.H.; Lai, C.Y.; Su, M.C.; Cheng, J.C.; Chang, Y.S. Antiviral activity of Portulaca oleracea L. against influenza A viruses. J. Ethnopharmacol 2019, 241, 112013. [Google Scholar] [CrossRef]
- Hsu, J.T.; Yeh, J.Y.; Lin, T.J.; Li, M.L.; Wu, M.S.; Hsieh, C.F.; Chou, Y.C.; Tang, W.F.; Lau, K.S.; Hung, H.C.; et al. Identification of BPR3P0128 as an inhibitor of cap-snatching activities of influenza virus. Antimicrob. Agents. Chemother. 2012, 56, 647–657. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, R.; Balzarini, J.; Baba, M.; Snoeck, R.; Schols, D.; Herdewijn, P.; Desmyter, J.; De Clercq, E. Rapid and automated tetrazolium-based colorimetric assay for the detection of anti-HIV compounds. J. Virol. Methods 1988, 20, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Dohme, A.; Knoblauch, M.; Egorova, A.; Makarov, V.; Bogner, E. Broad-spectrum antiviral diazadispiroalkane core molecules block attachment and cell-to-cell spread of herpesviruses. Antiviral Res. 2022, 206, 105402. [Google Scholar] [CrossRef] [PubMed]
- Buehler, J.; Navi, D.; Lorusso, A.; Vincent, A.; Lager, K.; Miller, C.L. Influenza A virus PB1-F2 protein expression is regulated in a strain-specific manner by sequences located downstream of the PB1-F2 initiation codon. J. Virol. 2013, 87, 10687–10699. [Google Scholar] [CrossRef]
- Courtney, D.G.; Kennedy, E.M.; Dumm, R.E.; Bogerd, H.P.; Tsai, K.; Heaton, N.S.; Cullen, B.R. Epitranscriptomic enhancement of influenza A virus gene expression and replication. Cell host microbe 2017, 22, 377–386. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Loo, D.T. In situ detection of apoptosis by the TUNEL assay: an overview of techniques. Methods Mol. Biol. 2011, 682, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Vermes, I.; Haanen, C.; Reutelingsperger, C. Flow cytometry of apoptotic cell death. J. Immunol. Methods 2000, 243, 167–190. [Google Scholar] [CrossRef] [PubMed]
- Betancur-Galvis, L.A.; Morales, G.E.; Forero, J.E.; Roldan, J. Cytotoxic and antiviral activities of Colombian medicinal plant extracts of the Euphorbia genus. Mem. Inst. Oswaldo. Cruz. 2002, 97, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.J.; Nightingale, K.; Smith, H.; Sweet, C. Influenza A virus-induced apoptosis is a multifactorial process: exploiting reverse genetics to elucidate the role of influenza A virus proteins in virus-induced apoptosis. Virology 2005, 335, 198–211. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, S.; Batra, J.; Cao, W.; Sharma, K.; Patel, J.R.; Ranjan, P.; Kumar, A.; Katz, J.M.; Cox, N.J.; Lal, R.B.; et al. Influenza A virus nucleoprotein induces apoptosis in human airway epithelial cells: implications of a novel interaction between nucleoprotein and host protein Clusterin. Cell Death Dis. 2013, 4, e562. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Cootes, T.; McLean, A. From influenza-induced acute lung injury to multiorgan failure. Ann. Update in Intensive Care and Emerg. Med. 2019 2019, 449–458. [Google Scholar]
- Qin, S.; Xiao, W.; Zhou, C.; Pu, Q.; Deng, X.; Lan, L.; Liang, H.; Song, X.; Wu, M. Pseudomonas aeruginosa: pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduct. Target Ther. 2022, 7, 199. [Google Scholar] [CrossRef]
- Kimura, T.; Yoshimizu, M.; Ezura, Y.; Kamei, Y. An antiviral agent (46NW-04A) produced by Pseudomonas sp. and its activity against fish viruses. J. Aquat. Anim. Health 1990, 2, 12–20. [Google Scholar] [CrossRef]
- Park, J.Y.; Yang, S.Y.; Kim, Y.C.; Kim, J.-C.; Dang, Q.L.; Kim, J.J.; Kim, I.S. Antiviral peptide from Pseudomonas chlororaphis O6 against tobacco mosaic virus (TMV). J. Kor. Soc. Appl. Biol. Chem. 2012, 55, 89–94. [Google Scholar] [CrossRef]
- Chiang, G.; Sassaroli, M.; Louie, M.; Chen, H.; Stecher, V.J.; Sperber, K. Inhibition of HIV-1 replication by hydroxychloroquine: mechanism of action and comparison with zidovudine. Clin. Ther. 1996, 18, 1080–1092. [Google Scholar] [CrossRef] [PubMed]
- Savarino, A.; Boelaert, J.R.; Cassone, A.; Majori, G.; Cauda, R. Effects of chloroquine on viral infections: an old drug against today's diseases? Lancet. Infect. Dis. 2003, 3, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Mao, C.; Luan, X.; Shen, D.D.; Shen, Q.; Su, H.; Wang, X.; Zhou, F.; Zhao, W.; Gao, M.; et al. Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science 2020, 368, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Verma, J.; Srivastava, A.; Srivastava, S.; Prasad, V. Pseudomonas aeruginosa isolate PM1 effectively controls virus infection and promotes growth in plants. Arch. Microbiol 2022, 204, 494. [Google Scholar] [CrossRef] [PubMed]
- Laucirica, D.R.; Schofield, C.J.; McLean, S.A.; Margaroli, C.; Agudelo-Romero, P.; Stick, S.M.; Tirouvanziam, R.; Kicic, A.; Garratt, L.W.; Western Australian Epithelial Research, P.; et al. Pseudomonas aeruginosa modulates neutrophil granule exocytosis in an in vitro model of airway infection. Immunol. Cell Biol. 2022, 100, 352–370. [Google Scholar] [CrossRef]
- Yeganeh, B.; Ghavami, S.; Rahim, M.N.; Klonisch, T.; Halayko, A.J.; Coombs, K.M. Autophagy activation is required for influenza A virus-induced apoptosis and replication. Biochim. Biophys. Acta. Mol. Cell Res. 2018, 1865, 364–378. [Google Scholar] [CrossRef]
- Liu, C.; Xiao, K.; Xie, L. Progress in preclinical studies of macrophage autophagy in the regulation of ALI/ARDS. Front. Immunol. 2022, 13, 922702. [Google Scholar] [CrossRef]
- Noone, P.M.; Reddy, S.P. Recent advances in dead cell clearance during acute lung injury and repair. Fac. Rev. 2021, 10. [Google Scholar] [CrossRef]
- Oladunni, F.S.; Park, J.G.; Pino, P.A.; Gonzalez, O.; Akhter, A.; Allue-Guardia, A.; Olmo-Fontanez, A.; Gautam, S.; Garcia-Vilanova, A.; Ye, C.; et al. Lethality of SARS-CoV-2 infection in K18 human angiotensin-converting enzyme 2 transgenic mice. Nat Commun 2020, 11, 6122. [Google Scholar] [CrossRef] [PubMed]
- Lewis, W.; Dalakas, M.C. Mitochondrial toxicity of antiviral drugs. Nat. Med. 1995, 1, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Robertson, W.C., Jr. Carbamazepine toxicity after influenza vaccination. Pediatr. Neurol. 2002, 26, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Touret, F.; Gilles, M.; Barral, K.; Nougairede, A.; van Helden, J.; Decroly, E.; de Lamballerie, X.; Coutard, B. In vitro screening of a FDA approved chemical library reveals potential inhibitors of SARS-CoV-2 replication. Sci. Rep. 2020, 10, 13093. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).