Submitted:
07 March 2024
Posted:
08 March 2024
Read the latest preprint version here
Abstract
Keywords:
Introduction
Materials and Methods
Statistical Analysis
Results
Patients
Seizure Outcome
QOL and Postural-Motor Outcome
Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Waaler, P.E.; Blom, B.H.; Skeidsvoll, H.; Mykletun, A. Prevalence, classification, and severity of epilepsy in children in western Norway. Epilepsia. 2000, 41, 802–810. [Google Scholar] [CrossRef]
- Berg, A.T.; Berkovic, S.F.; Brodie, M.J.; Buchhalter, J.; Cross, J.H.; van Emde Boas, W.; et al. Revised terminology and concepts for organization of seizures and epilepsies: report of the ILAE Commission on Classification and Terminology, 2005-2009. Epilepsia. 2010, 51, 676–685. [Google Scholar] [CrossRef]
- Russo, A.; Gobbi, G. A reflection on the role of genetics in the concept of "epileptic encephalopathy", as emerged from the most recent ILEA classification of epilepsy. Ital. J. Pediatr. 2020, 46, 2. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Brodie, M.J.; Liew, D.; Kwan, P. Treatment outcomes in patients with newly diagnosed epilepsy treated with established and new antiepileptic drugs: a 30-year longitudinal cohort study. JAMA Neurol. 2018, 75, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Parisi, P.; Spalice, A.; Nicita, F.; et al. “Epileptic encephalopathy” of infancy and childhood: electro-clinical pictures and recent understandings. Curr. Neuropharmacol. 2010, 8, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Raspall-Chaure, M.; Neville, B.G.; Scott, R.C. The medical management of the epilepsies in children: conceptual and practical considerations. Lancet Neurol. 2008, 7, 57–69. [Google Scholar] [CrossRef]
- Russo, A.; Hyslop, A.; Gentile, V.; Boni, A.; Miller, I.; Chiarello, D.; Pellino, G.; Zenesini, C.; Martinoni, M.; Lima, M.; Ragheb, J.; Cordelli, D.M.; Pini, A.; Jayakar, P.; Duchowny, M. Early vagus nerve stimulator implantation as a main predictor of positive outcome in pediatric patients with epileptic encephalopathy. Epileptic Disord. 2021, 23, 563–571. [Google Scholar] [CrossRef] [PubMed]
- van der Louw, E.; van den, Hurk D.; Neal, E.; Leiendecker, B.; Fitzsimmon, G.; Dority, L.; et al. Ketogenic diet guidelines for infants refractory epilepsy. JH. Eur.J Paediatr. Neurol. 2016, 20, 798–809. [Google Scholar] [CrossRef]
- Gupta, R.; Appleton, R. Corticosteroids in the management of the paediatric epilepsies. Arch. Dis. Child 2005, 90, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Verhelst, H.; Boon, P.; Buyse, G.; Ceulemans, B.; D’Hooghe, M.; Meirleir, L.D.; et al. Steroids in intractable childhood epilepsy: clinical experience and review of the literature. Seizure 2005, 14, 412–421. [Google Scholar] [CrossRef]
- Kramer, U.; Watemberg, N.; Zelnik, N.; et al. Efficacy of corticosteroid therapy in treating epileptic encephalopathies and refractory epilepsies other than West syndrome. J. Pediatr. Neurol. 2006, 4, 147–153. [Google Scholar]
- Kotagal, P. Current status of treatments for children with electrical status in slow-wave sleep (ESES/CSWS). Epilepsy Curr. 2017, 17, 214–216. [Google Scholar] [CrossRef] [PubMed]
- van den Munckhof, B.; Arzimanoglou, A.; Perucca, E.; van Teeseling, H.C.; Leijten, F.S.S.; Braun, K.P.J.; Jansen, F.E. «Corticosteroids versus Clobazam in 14. Epileptic Encephalopathy with ESES: A European Multicentre Randomised Controlled Clinical Trial (RESCUE ESES*)». Trials 2020, 21, 957. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.; Livingston, S. The effect of adrenocorticotropic hormone in epilepsy. J. Pediatr. 1950, 37, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Sorel, L.; Dusaucy-Bauloye, A. Findings in 21 cases of Gibbs' hypsarrhythmia; spectacular effectiveness of ACTH. Acta Neurol. Psychiatr. Belg. 1958, 58, 130–141. [Google Scholar] [PubMed]
- Okumura, A.; Tsuji, T.; Kato, T.; Natsume, J.; Negoro, T.; Watanabe, K. ACTH therapy for generalized seizures other than spasms. Seizure 2006, 15, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, D.B. Prednisone therapy in pediatric epilepsy. Pediatr Neurol 2003, 28, 194–198. [Google Scholar] [CrossRef]
- Wanigasinghe, J.; Arambepola, C.; Ranganathan, S.S.; Sumanasena, S. Randomized, single-blind, parallel clinical trial on efficacy of oral prednisolone versus intramuscular corticotropin: a 12-month assessment of spasm control in West syndrome. Pediatr. Neurol. 2017, 76, 14–19. [Google Scholar] [CrossRef]
- Sevilla-Castillo, R.A.; Palacios, G.C.; Ramirez-Campos, J.; Mora-Puga, M.; Diaz-Bustos, R. Methylprednisolone for the treatment of children with refractory epilepsy. Neuropediatrics 2009, 40, 265–268. [Google Scholar] [CrossRef]
- Mytinger, J.R.; Quigg, M.; Taft, W.C.; Buck, M.L.; Rust, R.S. Outcomes in treatment of infantile spasms with pulse methylprednisolone. J. Child Neurol. 2010, 25, 948–953. [Google Scholar] [CrossRef]
- Bast, T.; Richter, S.; Ebinger, F.; Rating, D.; Wiemer-Kruel, A.; Schubert-Bast, S. Efficacy and tolerability of methylprednisolone pulse therapy in childhood epilepsies other than infantile spasms. Neuropediatrics 2014, 45, 378–385. [Google Scholar] [PubMed]
- Almaabdi, K.H.; Alshehri, R.O.; Althubiti, A.A..; et al. Intravenous methylprednisolone for intractable childhood epilepsy. Pediatr. Neurol. 2014, 50, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Pera, M.C.; Randazzo, G.; Masnada, S.; Dontin, S.D.; De Giorgis, V.; Balottin, U.; et al. Intravenous methylprednisolone pulse therapy for children with epileptic encephalopathy. Funct. Neurol. 2015, 30, 173–179. [Google Scholar] [CrossRef]
- Yeh, H.R.; Kim, M.J.; Ko, T.S.; Yum, M.S.; You, S.J. Short-Term Outcome of Intravenous Methylprednisolone Pulse Therapy in Patients With Infantile Spasms. Pediatr Neurol. 2017, 7, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Kimizu, T.; Takahashi, Y.; Oboshi, T.; Horino, A.; Omatsu, H.; Koike, T.; Yoshitomi, S.; et al. Methylprednisolone Pulse Therapy in 31 Patients with Refractory Epilepsy: A Single-Center Retrospective Analysis. Epilepsy & Behavior 2020, 109, 107116. [Google Scholar]
- Chatterjee, A.; Mundlamuri, R.C.; Kenchaiah, R.; Asranna, A.; Nagappa, M.; Bindu, P.S.; et al. Role of pulse methylprednisolone in epileptic encephalopathy: A retrospective observational analysis. Epilepsy Res. 2021, 173, 10661. [Google Scholar] [CrossRef]
- Rajpurohit, M.; Gupta, A.; Madaan, P.; Sahu, J.K.; Singhi, P. Safety, Feasibility and Effectiveness of Pulse Methylprednisolone Therapy in Comparison with Intramuscular Adrenocorticotropic Hormone in Children with West Syndrom. Indian J. Pediatr. 2021, 88, 663–667. [Google Scholar] [CrossRef]
- Rangarajan, A.; Mundlamuri, R.C.; Kenchaiah, R.; Prathyusha, P.V.; Viswanathan, L.G.; Asranna, A.; Chatterjee, A.; Nagappa, M.; Seshagiri, D.V.; Kulanthaivelu, K.; Bharath, R.D.; Jitender, S.; Thennarasu, K.; Sinha, S. Efficacy of pulse intravenous methylprednisolone in epileptic encephalopathy: a randomised controlled trial. J. Neurol. Neurosurg. Psychiatry. 2022, 93, 1299–1305. [Google Scholar] [CrossRef]
- Miller, A.L.; Chaptal, C.; McEwen, B.S.; Peck Jr, E.J. Modulation of high affinity GABA uptake into hippocampal synaptosomes by glucocorticoids. Psychoneuroendocrinology 1978, 3, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, L.; Sapolsky, R. The role of the hippocampus in feedback regulation of the hypothalamic pituitary adrenocortical axis. Endocr. Rev. 1991, 12, 118–134. [Google Scholar] [CrossRef]
- Baram, T.Z. Pathophysiology of massive infantile spasms: perspective on the putative role of the brain adrenal axis. Ann. Neurol. 1993, 33, 231–236. [Google Scholar] [CrossRef]
- Vidal, C.; Jordan, W.; Zieglgansberger, W. Corticosterone reduces the excitability of hippocampal pyramidal cells in vitro. Brain Res. 1986, 383, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Joels, M. Steroid hormones and excitability in the mammalian brain. Front Neuroendocrinol. 1997, 18, 2–48. [Google Scholar] [CrossRef] [PubMed]
- Baram, T.Z.; Hatalski, C.G. Neuropeptide-mediated excitability: a key triggering mechanism for seizure generation in the developing brain. Trends Neurosci. 1998, 21, 471–476. [Google Scholar] [CrossRef]
- Watzka, M.; Bidlingmaier, F.; Beyenburg, S.; et al. Corticosteroid receptor mRNA expression in the brains of patients with epilepsy. Steroids 2000, 65, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Joels, M. Corticosteroid actions in the hippocampus. J. Neuroendocrinol. 2001, 13, 657–669. [Google Scholar] [CrossRef]
- Vezzani, A.; Granata, T. Brain inflammation in epilepsy: experimental and clinical evidence. Epilepsia 2005, 46, 724–743. [Google Scholar] [CrossRef]
- Marchi, N.; Granata, T.; Freri, E.; Ciusani, E.; Ragona, F.; Puvenna, V.; et al. Efficacy of anti- inflammatory therapy in a model of acute seizures and in a population of pediatric drug resistant epileptics. PLoS One 2011, 6, e18200. [Google Scholar] [CrossRef]
- Reddy, D.S. Role of anticonvulsant and antiepileptogenic neurosteroids in the pathophysiology and treatment of epilepsy. Front. Endocrinol. 2011, 2, 38. [Google Scholar] [CrossRef]
- Xu, D.; Miller, S.D.; Koh, S. Immune mechanisms in epileptogenesis. Front. Cell. Neurosci. 2013, 7, 195. [Google Scholar] [CrossRef] [PubMed]
- Frauman, A.G. An overview of the adverse reactions to adrenal corticosteroids. Adverse Drug React Toxicol Rev. 1996, 15, 203–206. [Google Scholar] [PubMed]
- Wanigasinghe, J.; Arambepola, C.; Ranganathan, S.S.; Jayasundara, K.; Weerasinghe, A.; Wickramarachchi, P. Epilepsy outcome at four years in a randomized clinical trial comparing oral prednisolone and intramuscular ACTH in west syndrome. Pediatr. Neurol. 2021, 119, 22–26. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, F.J.; Edwards, S.W.; Alber, F.D.; Hancock, E.; Johnson, A.L.; Kennedy, C.R.; et al. Safety and effectiveness of hormonal treatment versus hormonal treatment with vigabatrin for infantile spasms (ICISS): a randomised, multicentre, open-label trial. Lancet Neurol. 2017, 16, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Gowda, V.K.; Narayanaswamy, V.; Shivappa, S.K.; Benakappa, N.; Benakappa, A. Corticotrophin-ACTH in comparison to prednisolone in west syndrome - a randomized study. Indian J. Pediatr. 2019, 86, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Lux, A.L.; Edwards, S.W.; Hancock, E.; Johnson, A.L.; Kennedy, C.R.; Newton, R.W.; et al. The United Kingdom Infantile Spasms Study comparing vigabatrin with prednisolone or tetracosactide at 14 days: a multicentre, randomised controlled trial. Lancet. 2004, 364, 1773–1778. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, F.J.K.; Edwards, S.W.; Alber, F.D.; Cortina Borja, M.; Hancock, E.; Johnson, A.L.; et al. Vigabatrin with hormonal treatment versus hormonal treatment alone (ICISS) for infantile spasms: 18-month outcomes of an open- label, randomised controlled trial. Lancet Child Adolesc. Health 2018, 2, 715–725. [Google Scholar] [CrossRef]
- Darke, K.; Edwards, S.W.; Hancock, E.; ohnson, A.L.; Kennedy, C.R.; Lux, A.L.; et al. Developmental and epilepsy outcomes at age 4 years in the UKISS trial comparing hormonal treatments to vigabatrin for infantile spasms: a multi-centre randomised trial. Arch Dis Child 2010, 95, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Buzatu, M.; Bulteau, C.; Altuzarra, C.; Dulac, O.; Van Bogaert, P. Corticosteroids as treatment of epileptic syndromes with continuous spike-waves during slow-wave sleep. Epilepsia 2009, 50, 68–72. [Google Scholar] [CrossRef]
- Sinclair, D.B.; Snyder, T.J. Corticosteroids for the treatment of Landau-kleffner syndrome and continuous spike-wave discharge during sleep. Pediatr. Neurol. 2005, 32, 300–306. [Google Scholar] [CrossRef]
- Chen, J.; Cai, F.; Jiang, L.; Hu, Y.; Feng, C. A prospective study of dexamethasone therapy in refractory epileptic encephalopathy with continuous spike-and-wave during sleep. Epilepsy Behav. 2016, 55, 1–5. [Google Scholar] [CrossRef]
- Fatema, K.; Rahman, M.M.; Begum, S. Characteristics and management of children with continuous spikes and waves during slow sleep. Mymensingh Med. J. 2015, 24, 806–812. [Google Scholar] [PubMed]
- van den Munckhof, B.; van Dee, V.; Sagi, L.; Caraballo, R.H.; Veggiotti, P.; Liukkonen, E.; et al. Treatment of electrical status epilepticus in sleep: a pooled analysis of 575 cases. Epilepsia 2015, 56, 1738–1746. [Google Scholar] [CrossRef]
- Yamatogi, Y.; Ohtsuka, Y.; Ishida, T.; Ichiba, N.; Ishida, S.; Miyake, S.; et al. Treatment of the Lennox syndrome with ACTH: a clinical and electroencephalographic study. Brain Dev. 1979, 1, 267–276. [Google Scholar] [CrossRef]
- Gofshteyn, J.S.; Kevin Gurcharran, K.; Marquis, B.O.; Jacqueline Lamothe, J.; Gourley, D.; Grinspan, Z.; et al. Measurable outcomes for pediatric epileptic encephalopathy: a single-center experience with corticosteroid therapy. Epileptic Disord. 2021, 23, 111–122. [Google Scholar] [CrossRef]
- Chatterjee, A.; Mundlamuri, R.C.; Kenchaiah, R.; Asranna, A.; Nagappa, M.; Bindu, P.S.; et al. Role of pulse methylprednisolone in epileptic encephalopathy: a retrospective observational analysis. Epilepsy Res. 2021, 173, 106611. [Google Scholar] [CrossRef] [PubMed]
- 56. Chatterjee, A.; Mundlamuri, R.C.; Kenchaiah, R.; Asranna, A.; Nagappa, M.; Bindu, P.S.; et al. ACTH. treatment in intractable seizures of childhood. Brain Dev. 1992, 14, 102–106. [Google Scholar]
- Nasiri, J.; Sarajan, A.; Salari, M.; Sedghi, M. Therapeutic effects of adrenocorticotropic hormone ACTH in children with severely intractable seizure. Iran J. Child Neurol. 2017, 11, 19–26. [Google Scholar] [PubMed]
- Bakker, D.P.; Catsman-Berrevoets, C.E.; Neuteboom, R.F. Effectiveness of a hybrid corticosteroid treatment regimen on refractory childhood seizures and a review of other corticosteroid treatments. Eur. J. Paediatr. Neurol. 2015, 19, 553–560. [Google Scholar] [CrossRef]
- Kalra, V.; Sharma, S. ; Arya RACTH therapy in refractory generalized epilepsy Indian. J. Pediatr. 2009, 76, 91–93. [Google Scholar]
- Inui, T.; Kobayashi, T.; Kobayashi, S.; Sato, R.; Endo, W.; Kikuchi, A.; et al. Efficacy of long term weekly ACTH therapy for intractable epilepsy. Brain Dev. 2015, 37, 449–54. [Google Scholar] [CrossRef]
- Vossler, D.G.; Bainbridge, J.L.; Boggs, J.G.; Novotny, E.J.; Loddenkemper, T.; Faught, E.; et al. Treatment of refractory convulsive status epilepticus: a comprehensive review by the american epilepsy society treatments committee. Epilepsy Curr. 2020, 20, 245–264. [Google Scholar] [CrossRef]
- Hirsch, L.J.; Gaspard, N.; van Baalen, A.; Nabbout, R.; Demeret, S.; Loddenkemper, T.; et al. Proposed consensus definitions for new-onset refractory status epilepticus (NORSE), febrile infection-related epilepsy syndrome (FIRES), and related conditions. Epilepsia 2018, 59, 739–744. [Google Scholar] [CrossRef]
- Vezzani, A.; Ruegg, S. The pivotal role of immunity and inflammatory processes in epilepsy is increasingly recognized: introduction. Epilepsia 2011, 52, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Shorvon, S.; Ferlisi, M. The treatment of super-refractory status epilepticus: a critical review of available therapies and a clinical treatment protocol. Brain. 2011, 134, 2802–2818. [Google Scholar] [CrossRef]
- Vezzani, A.; Balosso, S.; Aronica, E.; Ravizza, T. Basic mechanisms of status epilepticus due to infection and inflammation. Epilepsia 2009, 50, 56–57. [Google Scholar] [CrossRef] [PubMed]
- Vezzani, A. Epilepsy and inflammation in the brain: overview and pathophysiology. Epilepsy Curr. 2014, 14, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, R.; et al. Stressful life events and with seizure frequency in patients epilepsy. Epilepsia 1994, 35, 336–343. [Google Scholar] [CrossRef]
- HRMcKee, M.D. Privitera, Stress as a seizure precipitant: identification, associated factors, and treatment options. Seizure 2017, 44, 21–26. [Google Scholar]
- Privitera, M.; et al. Characteristics of people with self-reported stress-precipitated seizures. Epilepsy Behav. 2014, 41, 74–77. [Google Scholar] [CrossRef]
- Sawyer, N.T.; Escayg, A. Stress and epilepsy: multiple models, multiple outcomes. J. Clin. Neurophysiol. 2010, 27, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Maguire, J.; Salpekar, J.A. Stress, seizures, and hypothalamic-pituitary-adrenal axis targets for the treatment of epilepsy. Epilepsy Behav 2013, 26, 352–362. [Google Scholar] [CrossRef]
- Mackenzie, G.; Maguire, J. Chronic stress compromises GABAergic inhibition in the hippocampus and increases seizure susceptibility. Epilepsy Res. 2015, 109, 13–27. [Google Scholar] [CrossRef]
- Castro, O.W.; Santos, V.R.; Pun, R.Y.K.; McKlveen, J.M.; Batie, M.; Holland, K.D.; et al. Impact of Corticosterone Treatment on Spontaneous Seizure Frequency and Epileptiform Activity in Mice with Chronic Epilepsy. PLoS ONE 2012, 7, e46044. [Google Scholar] [CrossRef]
- Culebras, A.; Miller, M.; Bertram, L.; Koch, J. Differential response of growth hormone, cortisol, and prolactin to seizures and to stress. Epilepsia 1987, 28, 564–570. [Google Scholar] [CrossRef]
- Abbott, R.J.; Browning, M.C.; Davidson, D.L. Serum prolactin and cortisol concentrations after grand mal seizures. Journal of Neurology, Neurosurgery & Psychiatry 1980, 43, 163–167. [Google Scholar]
- Pritchard, P.B.; Wannamaker, B.B.; Sagel, J.; Daniel, C.M. Serum prolactin and cortisol levels in evaluation of pseudoepileptic seizures. Ann. Neurol. 1985, 18, 87–89. [Google Scholar] [CrossRef]
- Wulsin, A.C.; Solomon, M.B.; Privitera, M.D.; Danzer, S.C.; Herman, J.P. Hypothalamic-pituitary- adrenocortical axis dysfunction in epilepsy. Physiology & Behavior 2016, 166, 22–31. [Google Scholar]
- Yao, T.C.; Huang, Y.W.; Chang, S.M.; Tsai, S.Y.; Wu, A.C.; Tsai, H.J. Association between oral corticosteroid bursts and severe adverse events: a nationwide population-based cohort study. Ann Intern Med. 2020, 173, 325–330. [Google Scholar] [CrossRef]
- Stuart, F.A.; Segal, T.Y.; Keady, S. Adverse psychological effects of corticosteroids in children and adolescents. Arch. Dis. Child. 2005, 90, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Yasir, M.; Goyal, A.; Sonthalia, S. Corticosteroid Adverse Effects; StatPearls: Tampa, FL, 2022. [Google Scholar]
- Rice, J.B.; White, A.G.; Scarpati, L.M.; Wan, G.; Nelson, W.W. Long-term systemic corticosteroid exposure: a systematic literature review. Clin. Ther. 2017, 39, 2216–2229. [Google Scholar] [CrossRef]
- Aljebab, F.; Choonara, I.; Conroy, S. Systematic review of the toxicity of short-course oral corticosteroids in children. Arch. Dis. Child. 2016, 101, 365–370. [Google Scholar] [CrossRef]
- Bassett, D.S.; Bullmore, E. Small-world brain networks. Neuroscientist. 2006, 12, 512–523. [Google Scholar] [CrossRef]
- Meunier, D.; Lambiotte, R.; Bullmore, E.T. Modular and hierarchically modular organization of brain network. Front. Neurosci. 2010, 4, 200. [Google Scholar] [CrossRef]
- Stam, C.J.; Reijneveld, J.C. Graph theoretical analysis of complex networks in the brain. Nonlinear Biomed. Phys. 2007, 1, 3. [Google Scholar] [CrossRef] [PubMed]
- Clemens, B.; Puskás, S.; Bessenyei, M.; Emri, M.; Spisák, T.; Koselák, M.; et al. EEG functional connectivity of the intrahemispheric cortico-cortical network of idiopathic generalized epilepsy. Epilepsy Res. 2011, 96, 11–23. [Google Scholar] [CrossRef]
- Pittau, F.; Grova, C.; Moeller, F.; Dubeau, F.; Gotman, J. Patterns of altered functional connectivity in mesial temporal lobe epilepsy. Epilepsia 2012, 53, 1013–1023. [Google Scholar] [CrossRef] [PubMed]
- Otte, W.M.; Dijkhuizen, R.M.; van Meer, M.P.A.; van der Hel, W.S.; Verlinde, S.A.M.W.; van Nieuwenhuizen, O.; et al. Characterization of functional and structural integrity in experimental focal epilepsy: reduced network efficiency coincides with white matter changes. PLoS One 2012, 7, 39078. [Google Scholar] [CrossRef]
- Vollmar, C.; O’Muircheartaigh, J.; Symms, M.R.; GJBarker, G.J.; Thompson, P.; Kumari, V.; et al. Altered microstructural connectivity in juvenile myoclonic epilepsy: the missing link. Neurology 2012, 78, 1555–1559. [Google Scholar] [CrossRef] [PubMed]
- Chavez, M.; Valencia, M.; Navarro, V.; Latora, V.; Martinerie, J. Functional modularity of background activities in normal and epileptic brain networks. Phys. Rev. Lett. 2010, 104, 118701. [Google Scholar] [CrossRef]
- Douw, L.; van Dellen, E.; de Groot, M.; Heimans, J.J.; Klein, M.; Stam, C.J.; et al. Epilepsy is related to theta band brain connectivity and network topology in brain tumor patients. BMC Neurosci. 2010, 11, 103. [Google Scholar] [CrossRef]
- Ponten, S.C.; Bartolomei, F.; Stam, C.J. Small-world networks and epilepsy: graph theoretical analysis of intracerebrally recorded mesial temporal lobe seizures, Clin. Neurophysiol. 2007, 118, 918–927. [Google Scholar] [CrossRef]
- Terry, J.R.; Benjamin, O.; Richardson, M.P. Seizure generation: the role of nodes and networks. Epilepsia 2012, 53, e166–e169. [Google Scholar] [CrossRef] [PubMed]
- Bartolomei, F.; Bettus, G.; Stam, C.J.; Guye, M. Interictal network proper- ties in mesial temporal lobe epilepsy: a graph theoretical study from intracerebral recordings. Clin. Neurophysiol. 2013, 124, 2345–2353. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, B.C.; Chen, Z.; He, Y.; Evans, A.C.; Bernasconi, N. Graph-theoretical analysis reveals disrupted small-world organization of cortical thickness correlation networks in temporal lobe epilepsy. Cereb. Cortex. 2011, 21, 2147–2157. [Google Scholar] [CrossRef] [PubMed]
- Horstmann, M.T.; Bialonski, S.; Noennig, N.; Mai, H.; Prusseit, J.; Wellmer, J.; et al. State dependent properties of epileptic brain networks: comparative graph-theoretical analyses of simultaneously recorded EEG and MEG. Clin. Neurophysiol. 2010, 121, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Engel, J.; Thompson, P.M.; Stern, J.M.; Staba, R.J.; Bragin, A.; Mody, I. Connectomics and epilepsy. Curr. Opin. Neurol. 2013, 26, 186–194. [Google Scholar] [CrossRef]
- Kramer, M.A.; Kolaczyk, E.D.; Kirsch, H.E. Emergent network topology at seizure onset in humans. Epilepsy Res. 2008, 79, 173–186. [Google Scholar] [CrossRef]
- Van Diessen, E.; Diederen, S.J.H.; Braun, K.P.J.; Jansen, F.E.; Stam, C.J. Functional and structural brain networks in epilepsy: what have we learned? Epilepsia 2013, 54, 1855–1865. [Google Scholar] [CrossRef]
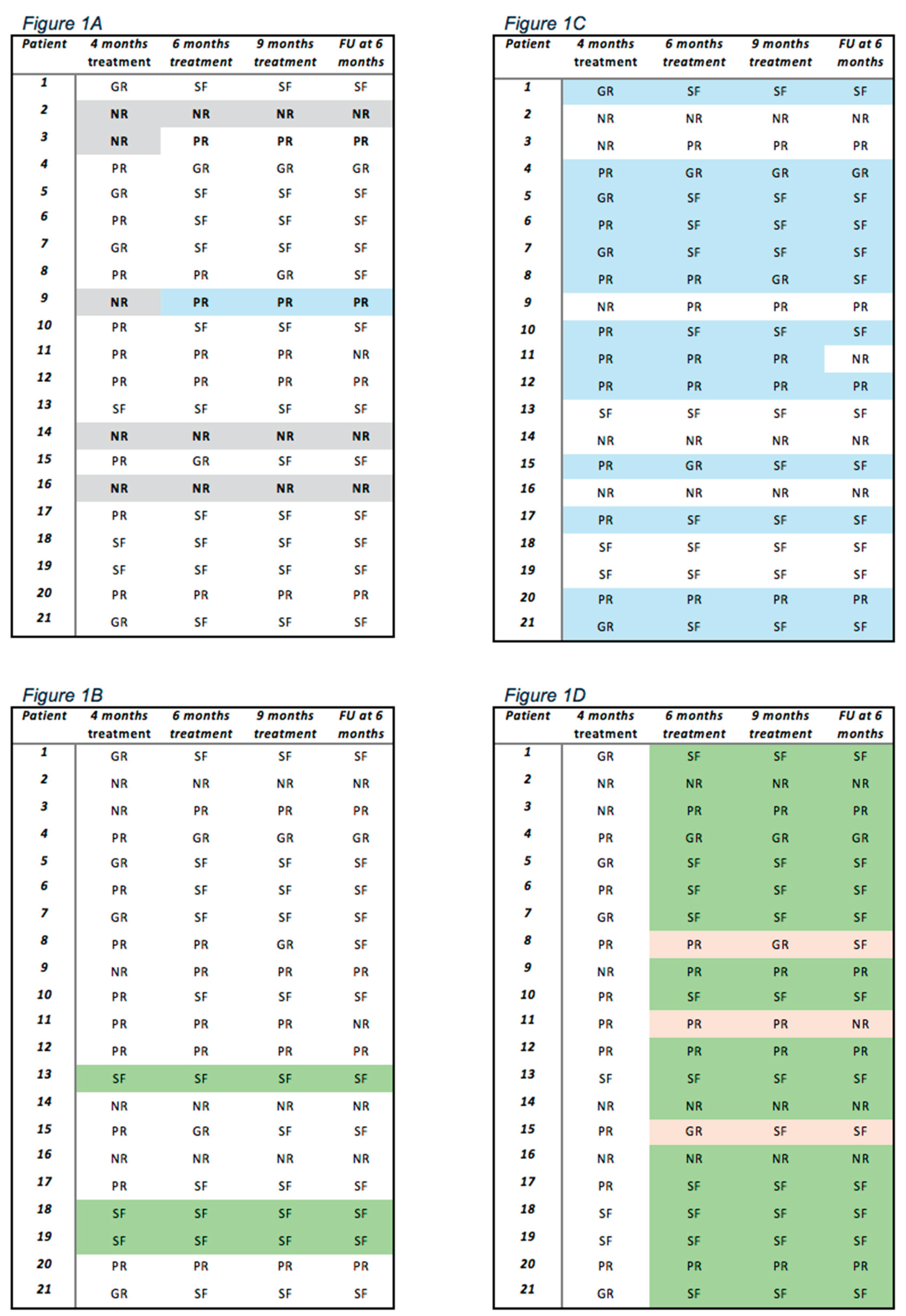
| OUTCOME | 4 MONTHS TREATMENT | 6-9 MONTHS TREATMENT | 6 MONTHS POST-TREATMENT | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Responders N=16 % 76.2 |
Non-Responders N=5 % 23.8 |
p-value | Responders N=17 % 81 |
Non-Responders N=4 % 19 |
p-value | Responders N=16 % 76.2 |
Non-Responders N=5 % 23.8 |
p-value | |
| Male gender N (%) | 9 (56.3) | 3 (60) | 0.9999 | 10 (58.8) | 2 (50) | 0.9999 | 10 (62.5) | 2 (40) | 0.6108 |
|
Age at epilepsy onset (mo) Mean (SD) Median [range] |
28.8 (25.9) 17.5 [6.5] |
25 (42.7) 4 [4–13] |
0.3412 |
27.9 (25.3) 13 [7–43] |
28 (48.7) 4 [3.552.5] |
0.2618 |
26.9 (25.9) 12.5 [6.5-51] |
31 (42.7) 4 [4–43] |
0.5079 |
|
Age at treatment beginning (mo) Mean (SD) Median [range] |
94.2 (39.1) 85.5 [71.5-121.5] |
133.4 (76.3) 107 [76–156] |
0.3855 |
93.1 (38.2) 82 [72–117] |
147.8 (80) 131.5 [89.5-206] |
0.2094 |
95.4 (38.2) 85.5 [74-121.5] |
129.6 (80.3) 107 [72–156] |
0.6794 |
|
Epilepsy duration pre-treatment (mo) Mean (SD) Median (range) |
65.4 (34.6) 69.5 [39.5-82] |
108.4 (44.1) 104 [68-152] |
0.1265 |
65.2 (33.5) 69 [42-79] |
119.8 (41.7) 128 [86-153.5] |
0.0438 |
68.4 (31.8) 69.5 [49-82] |
98.6 (59.5) 104 [68-152] |
0.3018 |
|
Epilepsy etiology N (%) Structural Genetic |
4 (25) 12 (75) |
4 (80) 1 (20) |
0.0475 |
4 (23.5) 13 (76.5) |
4 (100) 0 (0) |
0.0117 |
4 (25) 12 (75) |
4 (80) 1 (20) |
0.0475 |
|
Epilepsy syndrome N (%) ESES Other early onset EE |
13 (81.3) 3 (18.7) |
0 (0) 5 (100) |
0.0028 |
13 (76.5) 4 (23.5) |
0 (0) 4 (100) |
0.0117 |
12 (75) 4 (25) |
1 (20) 4 (80) |
0.0475 |
|
Seizure frequency pre-treatment N (%) Multi-daily Multi-weekly Multi-monthly Multi-yearly |
7 (43.8) 1 (6.2) 6 (37.5) 2 (12.5) |
4 (80) 1 (20) 0 (0) 0 (0) |
0.3054 |
8 (47.1) 1 (5.9) 6 (35.3) 2 (11.7) |
3 (75) 1 (25) 0 (0) 0 (0) |
0.4762 |
7 (43.8) 1 (6.3) 6 (37.5) 2 (12.5) |
4 (80) 1 (20) 0 (0) 0 (0) |
0.3054 |
| OUTCOME |
4 MONTHS TREATMENT N (%) |
6 MONTHS TREATMENT N (%) |
9 MONTHS TREATMENT N (%) |
6 MONTHS POST-TREATMENT N (%) |
|---|---|---|---|---|
| Seizures free | 3 (14.3) | 10 (47.6) | 11 (52.4) | 12 (57.1) |
| Good responder | 4 (19) | 2 (9.5) | 2 (9.5) | 1 (4.8) |
| Partial responder | 9 (42.7) | 6 (28.6) | 5 (23.8) | 4 (19) |
| Non responders | 5 (24) | 3 (14.3) | 3 (14.3) | 4 (19.1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





