1. Introduction
Biofuels are currently produced through the fermentation of food crops such as corn, sugarcane, sorghum, and others, potentially impacting food crop prices due to their dual use as food and fuel. As a result, the use of lignocellulosic biomass (including energy crops, agricultural residues, farm waste, etc.) as fermentation substrates for biofuel production is gaining traction as a viable alternative to using food crops (Palmqvist and Hahn-Hagerdal, 2000; Isar and Rangaswamy, 2011; Zhang and Ezeji, 2013; Okonkwo et al., 2016). However, the use of lignocellulosic biomass (LB) as fermentation substrate has a major limitation due to the presence of lignocellulosic-derived microbial inhibitory compounds (LDMICs) formed during the pretreatment and hydrolysis of LB to monomeric sugars (Ezeji et al., 2007). The LDMICs such as furan aldehydes (e.g., furfural and hydroxymethylfurfural_HMF) and phenolics (hydroxybenzaldehyde, ferulic acid, syringic acid, etc.) affect the growth and sugar utilization capacity of fermenting microorganisms such as Clostridium species, resulting in reduced growth and product yields (Baral and Shah, 2014; Ujor et al., 2016). To overcome these limitations, considerable research endeavors have investigated both direct and indirect approaches targeting various factors within the ABE production network of molecules (Agu et al., 2019; Okonkwo et al., 2019; Patil, 2019). The aim of these efforts is to enable the development of solvent-producing Clostridium strains that can be used economically and industrially.
The genus Clostridium is a diverse group of gram-positive, motile, rod-shaped anaerobes with tremendous diversity in habitat/ecological niche, substrate utilization, primary and secondary metabolite secretions, and morphological structures. These variations are regulated mostly by a network of complex but precisely molecular factors and mechanisms induced by environmental and physiological signals. Species in the genus Clostridium include several toxin-producing human pathogens (e.g., C. difficile, C. botulinum, C. tetani, C. perfringens) and many non-pathogenic species (e.g., C. acetobutylicum, C. beijerinckii, C. butylicum, C. carboxydivorans) which produce a range of products by fermenting a variety of carbohydrates (Liou et al., 2005; Ezeji et al., 2010). A common feature of Clostridium genus (over 200 species) is the production of oval sub-terminal endospores.
Clostridium species occur in a wide variety of habitats. Although most of them are found in the soil, some are also found in the intestines of animals and humans and are therefore excreted through feces (Maczulak, 2011). Others can be found in habitats like waterbodies and food/industrial waste dumps (Maczulak, 2011). Clostridium species can be utilized to produce cosmetics, medicine and/or for industrial biotechnological purposes. For example, C. botulinum, which is known to produce a neurotoxin that attacks the nerves in the body, causing muscle paralysis, difficult breathing, and potentially fatal consequences, is currently being used in the cosmetic and beauty industry. The toxin produced by C. botulinum has been modified and used to make Botox, a substance widely used in the cosmetic industry to remove or mask wrinkles (Small, 2014). Some Clostridium strains can selectively target cancer cells (e.g., C. noyvi-NT, C. sporogenes, and C. sordellii), with the rare capacity to penetrate and/or replicate within solid tumors (Staedtke et al., 2016). Hence, these Clostridium strains are potential candidates for delivering therapeutic proteins and tumor-killing agents into active tumors. The potential of using Clostridium species in cancer therapy related purposes has been established in various preclinical studies (Dang et al., 2001; Patyar et al., 2010).
In addition to the use of
Clostridium species in cosmetics and medicine, they can convert a range of substrates into fuels and bulk chemicals (
Table 1). In the early 19th century, solventogenic
Clostridium species were used for ABE production. Unfortunately, the discovery of low-cost fossil fuel source led to the cessation of bacterial methods of butanol production. Fortunately, the urgent demand for a clean environment and the finite nature of fossil fuels have rekindled the need to exploit biotechnological applications of
Clostridium species in the production of fuels and important industrial chemicals (
Table 1). Genome wise, solventogenic
Clostridium species have a wide range of genome sizes. For example,
C. acetobutylicum ATCC 824 has a 3.94-Mb chromosome with 11 ribosomal operons and contains a large plasmid, pSOL1 (Nölling et al., 2001). The pSOL1 is about 192 kb in size and harbors the ABE production genes. Conversely,
C. beijerinckii NCIMB 8052 possesses the largest chromosome of the
Clostridium genus with a 6.1 Mbp genome. In addition,
C. beijerinckii contains the largest number of ribosomal operons (13 plus) of the
Clostridium species. A considerable portion of genes in
C. beijerinckii have unknown functions, making it a promising candidate for exploratory research.
Most solvent-producing Clostridium species exhibit a biphasic fermentation profile regardless of the type of substrate used. An initial acidogenic phase characterized by the production of hydrogen, carbon dioxide, acetate, and butyrate, leads to a decrease in the pH of the fermentation medium. The solventogenic phase of the fermentation process is marked by a metabolic shift from producing hydrogen, carbon dioxide, and acids to generating solvents or ABE (stationary phase) as the primary products. During solventogenesis, substrate uptake takes place concurrently with acid re-assimilation, resulting in a rise in the culture pH. Depending on the Clostridium species and the carbon source, a wide range of metabolic pathways are activated for solvent formation. For instance, in C. carboxidivorans and C. ljungdahl, the wood-ljungdahl pathway is triggered to produce ethanol from synthesis gas during solventogenesis (Younesi et al., 2005; Han et al., 2020). In C. tetanomorphum, the methylaspartate pathway for glutamate conversion to acetate, butyrate, and carbon dioxide is activated (Buckel and Barker, 1974). In C. acetobutylicum and C. beijerinckii, both glycolytic (Embden-Meyerhof pathway) and acid production pathways are active for concurrent uptake of glucose and acids production during the exponential growth phase (Aristilde et al., 2015). Subsequently, the solventogenic pathway is activated for the reassimilation and conversion of acids to ABE during the late exponential and stationary phases.
Previous research efforts focused on random mutagenesis and mutant selection (Gottumukkala et al., 2017), modulation of acid and ABE production (adh, pta, ptb, buk, ack, bdh, ald, etc.), stress response (groES, groEL, DnaK, DnaJ), and LDMIC detoxification genes (Okonkwo et al., 2019). Some other indirect approaches focused on improving redox balance through NAD(P)H regeneration, calcium and acetate supplementations, and process and media optimizations (Han et al., 2013; Ujor et al., 2016, Xue et al., 2017; Patil, 2019). The use of metabolic engineering techniques to increase butanol production by solventogenic Clostridium species has yielded mixed results. It is therefore imperative to focus on the regulatory factors that play roles in butanol production in the future. Understanding the functions of these factors will be helpful in identifying specific targets for metabolic engineering of these microorganisms (Yang et al., 2018) considering that Clostridium species contain common sets of genes that encode proteins that modulate basic processes such as ABE production, stress response and sporulation. Most Clostridium species share common regulatory protein networks that play crucial roles in ABE production. These networks are responsible for the regulation of various processes, including sporulation, sugar uptake/utilization, carbon catabolite repression (CCR), and the transition from acidogenesis to solventogenesis. The key proteins involved in these regulations are Spo0A, the phosphoenolpyruvate:carbohydrate phosphotransferase system (PTS), carbon catabolite protein A (CcpA), and the transition phase regulator (AbrB; Smith, 1993; Tangney et al., 2003; Scotcher et al., 2005; Ren et al., 2010; Noguchi et al., 2013). The redox potential regulator (Rex) and the carbon storage regulator A (CsrA) proteins also play a role in solvent production in Clostridium species (Wietzke and Bahl, 2012; Tan et al., 2015).
A greater understanding of sporulation, sugar uptake/utilization, ABE production, and response to stressors such as LDMICs and butanol in solventogenic Clostridium species is crucial for deciphering the regulatory networks involving various molecular factors. This review specifically addresses the role of sigma factors, small non-coding RNAs (sRNAs) and secondary metabolites in the regulation of ABE production. It also examines their responses to solvent toxicity and LDMICs, as well as possible interactions between these regulatory elements. Furthermore, the review highlights the importance of molecular markers within these networks that may guide strategic decisions in metabolic engineering aimed at increasing butanol production by Clostridium species.
2. Sigma Factor 54 Directed Regulation
The transition from exponential to stationary growth phase in solventogenic Clostridium species results in a wave of altered gene expression patterns (Zhang and Ezeji, 2013). Sigma factors play a central role in regulating gene transcription in bacteria as they bind to RNA polymerase to form a catalytically active RNA polymerase holoenzyme (Davis et al., 2017; Buck and Cannon, 1992). The sigma factor in the RNA polymerase holoenzyme controls the tight binding of the core RNA polymerase to the promoter to initiate for transcription (Shen, 2019). The vegetative sigma factor family (σ70 or σ A) is well established for its role in promoting exponential growth in Clostridium species, while sporulation-specific sigma factors (σF, σE, σG, and σK) are responsible for initiating sporulation in these microorganisms. A review of the regulatory roles of sigma factors σA, σF, σE, σG, and σK in sporulation in Clostridium species has been published elsewhere (Sauer et al., 1995; Davis et al., 2017). Although often overlooked, sigma factor σ54 plays a critical role in regulatory functions such as sugar uptake/utilization and solventogenesis in solventogenic Clostridium species (Nie et al., 2016, Hocq et al., 2019; Yang et al., 2020).
Sigma factor 54 (σ
54), encoded by the
SigL gene, is a transcription factor that occurs as a single copy in solventogenic
Clostridium species such as
C. acetobutylicum, C. beijerinckii C. ljungdahlii, C. carboxidivorans, and C. autoethanogenum (Nie et al., 2019). While σ
54 was initially determined to be involved in nitrogen metabolism in
E. coli, this transcription factor also regulates solvent production in solventogenic
Clostridium species (Nie et al., 2016, Hocq et al., 2019; Yang et al., 2020). Unlike σ
A, σ
54 binds strongly to conserved consensus sequence at the GG −24 and TGC −12 promoter elements (Morett and Buck, 1989). Data from recent studies suggest that σ
54 promoter elements are located upstream of most phosphotransferase system (PTS) operons (
cel, lev, man, atl, and
gfr operons) involved in sugar transport in
C. acetobutylicum and
C. beijerinckii (Nie et al., 2016). Similarly, a putative σ
54 promoter element is located upstream of genes involved in ABE production (
adh, crt-hbd-thl-maoC-bcd-etfAB operon) in solventogenic
Clostridium species (Nie et al., 2019, Yang et al., 2020). The strategic location of the σ
54 promoter element is an indication that σ
54 may have important functions in the regulation of sugar metabolism and ABE production. Unlike most sigma factors that bind to RNA polymerase to form an open complex that initiates transcription, σ
54 binds to RNA polymerase and forms a stable closed RNA polymerase-σ
54 (RNAP-σ
54) complex which requires an activator to initiate transcription. The enhancer binding protein (EBP) serves as a vital activator for the conversion of the closed RNAP-σ
54 complex to its open form (
Table 2). EBP is widespread in
Clostridium species, and it is a modular protein consisting of an N-terminal regulatory domain, a central ATPase domain and a C-terminal DNA binding domain. The central ATPase domain utilizes the energy released from ATP hydrolysis to initiate isomerization of the closed RNAP-σ
54 complex to form an open complex (Schumacher et al., 2004; Jones, 2009). The EBP binds to upstream activator sequences (UASs) and forms higher-order oligomers that leads to formation of a stable complex with the DNA, and consequently, induces transcription activation. The EBP requirement for transcriptional activation is a unique feature of σ
54-dependent promoters. A review of functions of EBPs in σ
54 mediated transcription activation in bacteria has been published elsewhere (Bush and Dixon, 2012). The EBPs are grouped based on regulatory domains involved in sensing environmental or cellular signals (
Table 2).
The σ
54-dependent promoters and associated EBPs have been identified for various operons involved in the uptake of sugars (CelR, LevR, ManR, AtlR, and Gfr) and ABE production (AdhR, CrbR, AhcR, AorR) in
C. acetobutylicum,
C. beijerinckii, C. saccharoperbutylicum, C. carboxydivorans, and
C. ljungdahlii. This suggests that σ
54 plays important role in the regulation of sugar metabolism and ABE production in solventogenic
Clostridium species (Nie
et al., 2016, 2019). The EBPs associated with the PTS operons contain the PTS regulatory domain (PRD) at the N-terminals which enables the regulation of Hpr, EI and cognate EIIA/Bs, which are PTS proteins, via phosphorylation/dephosphorylation (Deutscher et al., 2014). Disruption of the
sigL (which codes for σ
54) gene led to impaired cell growth and ABE production in
C. beijerinckii, whereas the overexpression of this gene resulted in a significant increase in acetone production with no apparent change in butanol production (Hocq et al., 2019; Yang et al., 2020). In contrast to
sigL overexpression,
AdhR overexpression resulted in poor cell growth and ABE production (Yang et al., 2020). These data suggest that EBP mediated σ
54 regulation may be present, effective, and concentration dependent in solventogenic
Clostridium species. It is also plausible that a second protein/molecule may be required for transcriptional activation. The overabundance of AdhR may lead to binding exclusion of the second regulatory protein/molecule that may be required for transcriptional activation. Another possibility is the requirement of optimal AdhR concentration for UASs binding because increased abundance of AdhR may distort the concentration balance. Indeed, many proteins compete for the same UAS sites, and this competition can result in a partial or total inhibition of binding. For instance, alcohol dehydrogenase (Adh) is an important enzyme for ABE production in
Clostridium species. This is because Adh catalyzes the conversion of precursor aldehydes (acetaldehyde and butyraldehyde) to their respective alcohols (ethanol and butanol). A precise understanding of regulatory factors involved in the expression of
Adh gene is important to improve ABE production. Delineating the regulatory domain and activation signal within AdhR is a crucial aspect in comprehending the regulatory mechanism of AdhR and exploring possible strategies for enhancing butanol production. The presence of EBPs and the diverse regulatory domains suggest some important functions of EBPs in signal transduction and regulation of sugar metabolism and ABE production (
Figure 1).
2.1. Interaction between σ54 Mediated Regulation and Carbon Catabolite Repression/Activation
Carbon catabolite repression (CCR) is a major barrier to simultaneous utilization of hexose and pentose sugars in bacteria (
Figure 2). Solventogenic
Clostridium species are no exception. With ongoing efforts to utilize lignocellulose derived sugars as feedstocks for ABE production, it is important to understand and engineer solventogenic
Clostridium species to improve simultaneous utilization of hexose and pentose sugars because these sugars are significant component of the lignocellulosic biomass (LB) hydrolysates (LBH). CCR occurs when the presence of a preferred carbon source (usually glucose) in the growth medium induces transcriptional repression of genes encoding proteins required for the uptake and metabolism of non-preferred sugars (e.g., xylose, arabinose, galactose, cellobiose, sucrose etc.; Ren et al., 2010). This transcriptional repression process is modulated by the HPr protein of the PTS which in turn is mediated by the catabolite control protein A (CcpA), an allosteric transcriptional regulator. The CcpA binds to the catabolite repression elements (
cre) in the promoter or coding region of the target gene and represses transcription. Interestingly, recent research studies have shown that the CcpA of
C. acetobutylicum and
C. beijerinckii also activates transcription of
sol operon,
adhA1,
adhA2
, crt, thl, bcd, etfAB, hbd which are genes involved in ABE formation (Ren et al., 2012; Yang et al., 2017). In
C. acetobutylicum and C. beijerinckii, several sugar uptake/utilization operons that contain
cre sites (e.g.,
Cel operon,
man operon) for CcpA mediated activation/repression possess σ
54 dependent promoters with associated EBPs (CelR and ManR; Nie et al., 2016). While no conventional
cre site has been identified in genes regulating ABE production (e.g.,
sol operon,
adhA1,
adhA2
, crt, thl, bcd, etfAB, hbd), their expression is downregulated in CcpA null mutants (Ren et al., 2012, Yang et al., 2018). This finding indicates that
cre is regulated by CcpA. However, sugar uptake and genes involved in ABE production, however, are regulated to some extent by the σ
54 regulon with EBPs in
C. beijerinckii (Nie et al., 2016, 2019). This indicates possible interactions between σ
54 mediated and CcpA-dependent regulation processes (ccr) that modulate gene expression. Hence, the question arises: which regulatory mechanism prevails, and is there a possibility of collaborative regulation during sugar uptake and ABE production?
The genes of the Cel operon and most sugar uptake/utilization operons contain typical cre site (WTGWAAACGWTWWCAW: W is A or T) in the coding region. The CcpA dependent CCR of genes encoding proteins that modulate uptake/utilization of non-preferred sugars (cel, xyl, ara operons) is regulated by the HPr-Ser-46. The CcpA-HPr-Ser-46 complex binds to cre sites on the promoter or protein coding sequence of the target operon/gene to repress gene expression (Deutscher et al., 2006, 2014). This mechanism of regulation is clearly CcpA/Hpr mediated, indicating that there is no interaction with the σ54 promoter. The CcpA regulation of genes modulating ABE production in C. beijerinckii (adhA1, adhA2, crt, thl, bcd, etfAB, hbd), however, is not straightforward. The absence of typical cre sites in the promoter or coding region, even though the genes for ABE production are being regulated, is indicative that the mechanism for this type of regulation is yet to be elucidated. Data from a recent study indicate that CcpA binds to three atypical crevar sites (1. AACTGCTAAATGTAAA-TTATACG-TTTACATTTAGCAGTTT, 2: TGTAAAAGTTGCTATTTACA. 3: TATTGTAAACCTTGTTTTGTTTTGCAGTTTACAA TA) upstream (within -350 to -1 base pairs proximal to transcription start site) of the sol operon promoter in C. acetobutylicum in a regulatory manner (Yang et al., 2017). These atypical binding sites involving CcpA are predominantly present in genes associated with sporulation and ABE formation, and the binding of CcpA to crevar was thought to be associated with gene activation. Additionally, transcriptomic analysis show that CcpA is constitutively expressed in C. acetobutylicum and C. beijerinckii during ABE fermentation (Alsaker et al., 2004; Wang et al., 2012). This suggests that at least one gene or protein molecule may be required to mediate regulation. Because EBPs consist of a N-terminal regulatory domain through which various cellular signals and proteins are regulated, EBPs could possibly serve as a direct control molecule of the CcpA. The EBPs possibly interact with CcpA to induce a greater activation signal for the σ54-dependent transcription in the presence of activating cellular signals. The crevar binding motif has been identified to consist of a core palindromic sequence TGTAAA/TTTACA with intervening sequence of variable length (Yang et al., 2017). Similarly, EBPs bind to three UASs (-80 to -150 bp upstream of the promoter sequence) in a cooperative manner to mediate σ54-dependent transcription (Nie et al., 2016; Yang et al., 2020). Specifically, UASs for EBPs genes (AdhR, AhcR, CrbR) modulating ABE production genes (adh, atoE-hbd-cotX, crt-hbd-thl-maoC-bcd-etfAB) are of variable length with flanking TGT/ACA palindromic sequences (Nie et al., 2019). The matching flanking palindromic sequences and similarly flexible binding sequence lengths recognized by both EBPs and CcpA at locations upstream of the promoter indicate the occurrence of either competitive or cooperative binding to induce repression or activation of gene transcription. Binding of EBPs to the three UASs may be required for activation and modulation of target gene transcription (Nie et al., 2016, Yang et al., 2020). Competitive binding of CcpA to one or more of these sites could either distort or enhance (possibly depending on position) EBP oligomeric structure stability required for ATPase activity necessary for the formation of the open RNA polymerase- σ54 complex. Another potential scenario involves the activation or repression facilitated through direct interaction of the CcpA and EBP regulatory domains, specifically through protein-protein interaction. EBPs can be activated or repressed through interaction with other proteins.
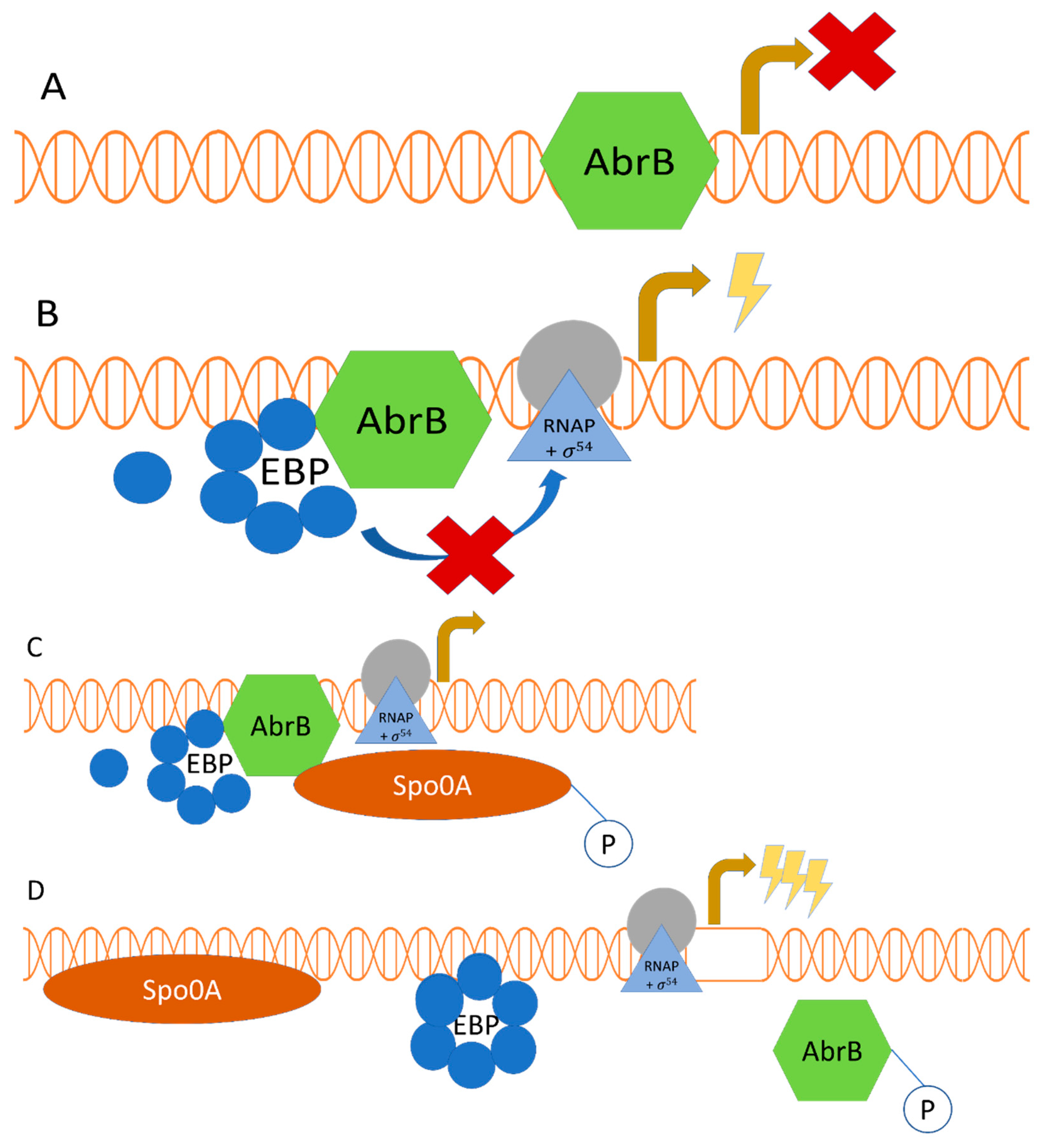
2.2. Regulatory Interplay between σ54-Mediated Regulation and the Transition Phase Regulator AbrB, along with the Sporulation Master Regulator Spo0A
The metabolic shift from acid to solvent production may be regulated by the transition phase regulator AbrB (Scotcher et al., 2005). AbrB was identified to play a role in activating transcription for the phosphotransferase acetylase (pta), acetate kinase (ack), phosphotransferase butyrylase (ptb), and butyrate kinase (buk) genes. The proteins encoded by these genes are accountable for acid production in C. acetobutylicum. Proteins associated with ABE production (sol operon: adhE1-ctfa-ctfb, adc) are negatively regulated by the AbrB. Three homologues of the AbrB gene (AbrB1941, AbrB0310 and AbrB3647) are active in regulating different genes in C. acetobutylicum (Scotcher et al., 2005; Xue et al., 2016). Data from an AbrB3647 gene knockout experiment in which transcriptomic analysis was conducted in C. acetobutylicum mutants indicated a marked upregulation of the sol operon and other genes that encode proteins involved in ABE production (bdhA, bdhB, edh, and adc; Xue et al., 2016). Although there are σ54 dependent promoters and associated EBPs that modulate genes of C. acetobutylicum involved in ABE production, orthologs of these genes (adhA1, adhA2, crt, thl, bcd, etfAB, hbd) have been predicted in C. beijerinckii, C. carboxidivorans and C. ljungdahlii to encode the σ54 dependent promoters of associated EBPs (AdhR, AhcR, CrbR; Nie et al., 2019). Albeit the function of AbrBs has not been fully elucidated in solventogenic Clostridium species, orthologs of these genes are present in their genomes, suggesting there may be AbrB mediated regulation (Yang et al., 2018). Data from enzyme mobility shift assays in C. acetobutylicum show that AbrB binds upstream of solvent production genes (sol operon, adhE2, edh, bdhA, bdhB) promoters (Xue et al., 2016) in the same region as the putative UAS for EBP binding. This suggests that AbrBs can repress the expression of genes that modulate ABE production by either inhibiting EBPs or preventing interaction of EBP with the RNA polymerase holoenzyme. The AbrB possibly competes for one or more UASs thereby preventing the formation of the higher order oligomer of EBPs required for stabilization in the DNA and induction of the ATPase activity of the central domain.
Activated sporulation transcriptional regulator protein, Spo0A, inactivates AbrBs via phosphorylation. The Spo0A protein regulates sporulation and solventogenesis by activating genes (
ctfA-ctfB,
adc, ptb, adhE) that modulate acid and ABE production in
Clostridium species (
Figure 1 and
Figure 2; Ravagnani et al., 2000; Thormann et al., 2002; Dürre and Hollergschwandner, 2004; Hollergschwandner and Dürre, 2004). The activation is facilitated by Spo0A binding to the 0A boxes upstream of the promoters of genes involved in ABE production. Data from functional analysis of Spo0A provided insights that Spo0A is a negative regulator of AbrB (Smith, 1993). Because the onset of ABE production coincides with the onset of sporulation in solventogenic
Clostridium species, deletion of
Sp0A in
C. beijerinckii resulted in a strain with lesser capacity to produce butanol and ethanol with no ability for acetone production relative to the wildtype (Harris et al., 2002; Ravagnani et al., 2000). This indicates that in the absence of the Spo0A protein, the genes involved in ABE production may not be optimally expressed in solventogenic
Clostridium species. It is also plausible that the production of lesser quantities of butanol and ethanol may be due to the continued AbrB repression of
ctfAB (CoA-transferase) transcription. The CoA-transferase mediates acid re-assimilation via Coenzyme A transfer to acetic and butyric acids, thus activating these acids for subsequent conversion to solvents during solventogenesis. The
C. beijerinckii Spo0A null mutant has impaired ability to produce acetone since the acetoacetate decarboxylase (
adc) gene expression is regulated by Spo0A (Sullivan and Bennett, 2006). The Adc protein is recognized for facilitating the transformation of acetoacetate into acetone and CO
2. Nevertheless, research has revealed that inactivation of
adc gene in
C. beijerinckii and
C. acetobutylicum do not completely abolish acetone production, owing to an operational non-enzymatic acetoacetate decarboxylation route for acetone production in these organisms (Jiang et al., 2009; Han et al., 2011). The analysis of the σ
54 regulon in
C. beijerinckii demonstrated the presence of σ
54-dependent promoters in nearly all genes involved in regulating ABE production, except for the aldehyde dehydrogenase (
ald), ctfAB, and
adc genes (Nie et al., 2019). The production of butanol and ethanol (although in low concentration) when there was Spo0A inactivation could be due to EBP-σ
54 mediated transcriptional activation of other genes such as
adhE that modulate ABE production. The dependence of the
adc and
ctfAB genes on Spo0A expression (AbrB inhibition and binding to the 0A box to allow a conformation favorable for transcription activation) could be attributed to the suppression of acetone production in the
C. beijerinckii Spo0A null mutant. The results of DNAse I foot-printing assays validated the precise binding of Spo0A to the 0A boxes at the promoter region of the
abrB gene (Ravagnani et al., 2002). The binding of Spo0A, however, did not protect 0A boxes from restriction digest, which suggests that Spo0A does not bind tightly to 0A boxes. It is possible that Spo0A binds transiently to the 0A boxes of genes and modulate ABE production, while simultaneously regulating transcription via AbrB inactivation. A plausible mechanism for Spo0A regulation of ABE production is that Spo0A phosphorylates and inactivates AbrB which dissociates from the 0A boxes on the DNA allowing the binding of Spo0A. The change in DNA conformation following Spo0A binding possibly favors the binding of EBP and formation of the higher order oligomeric structure, which promotes transcription activation (
Figure 1). The transient binding of Spo0A may also result in modulation of the conformation of DNA leading to stabilization of the EBP, improved contact of EBP with σ
54 for ATPase activity, and ultimately, transcriptional activation.
3. Regulation by Small Non-Coding RNAs
Bacterial small non-coding RNAs, also referred to as small RNAs (sRNAs; 50 to 400 nucleotides), regulate expression of diverse genes post-transcriptionally. The sRNAs modulate the expression of genes that control important cellular functions such as virulence, quorum sensing, iron homeostasis and stress responses (Vogel and Sharma, 2005). The sRNAs are involved in a wide range of biological functions such as translation (transfer messenger RNA) and RNA processing by RNase P (ribozyme moiety and protein secretion, 4.5S RNA;
Table 3; Brosse and Guillier, 2018). The sRNAs are usually located at intergenic regions which are independently transcribed or are nucleolytically cleaved from the 3’ untranslated region of mRNAs (Chao et al., 2012; Argaman et al., 2001). Generally, sRNA synthesis is induced in response to environmental and cellular signals, and many regulate gene expression post transcriptionally. The sRNAs imperfectly base pair with target mRNAs to inhibit protein synthesis or serve as markers for mRNAs degradation. Furthermore, sRNAs can bind to proteins to form ribonucleoproteins (RNPs). These complexes can either stabilize and facilitate specific functions or catalysis for the associated proteins or inhibit their normal functions (Richards and Vanderpool, 2011; Storz et al., 2011). Conventionally, sRNAs are encoded by the sense strand of DNA. Nevertheless, recent research findings indicate an increasing number of sRNAs are encoded on the anti-sense strand of coding nucleotide sequences. These are known as antisense RNAs (asRNAs) and they possess the ability to influence the expression of the corresponding coding gene (Thomason and Storz, 2010; Georg and Hess, 2011). The sRNA-mediated regulation is often closely associated with transcriptional regulatory networks that modulate growth, virulence, biofilm formation, etc., in different bacteria. While Brosse and Guillier (2018) published a review on the regulatory roles of sRNAs in diverse bacterial species, the review did not delve into the regulatory functions of sRNAs in solventogenic
Clostridium species or elucidate how these functions impact ABE production and stress response. The need to understand the regulatory functions of sRNAs in solventogenic
Clostridium species is beginning to emerge. Data from predictive comparative genome analysis suggest that seven solventogenic
Clostridium species harbor sRNA genes (Chen et al., 2011;
Table 3). The data also highlight the prevalence of metabolism-dependent sRNA genes in solventogenic
Clostridium species, revealing that saccharolytic
Clostridium species possess a higher number of sRNAs genes compared to their cellulolytic counterparts (
Table 3). Furthermore, the presence of several predicted sRNAs has been validated using RT-qPCR and Northern analysis of
C. acetobutylicum of mRNA. An abundance of sRNAs that respond to cellular stressors through either transcriptional regulation (6S, S-box and solB) or translational (tmRNA and SRP-RNA) processes have also been predicted (Venkataramanan
et al., 2013). It is worth noting that only a small proportion of the validated sRNAs have been subjected to experimental characterization (Keiler, 2008, Venkataramanan
et al., 2013; Craig
et al., 2014, Wach
et al., 2014; Wassarman, 2018, Yang
et al., 2020).
3.1. SolB Mediated Regulation
SolB is an sRNA that was identified by Peter Durre. This sRNA is associated with response to butanol stress in C. acetobutylicum (Wach et al., 2014). The SolB coding region lies upstream of the adhE2 gene on the sol operon as a 196 bp sequence (Zimmerman, 2013). Data from transcriptomic analysis indicated that SolB was upregulated specifically during butanol stress (Wang et al., 2013). Overexpression of SolB in solventogenic Clostridium species induces downregulation at the sol-locus leading to modulation in rate of gene transcription (Jones et al., 2018). While the repression of sol operon by SolB results in a longer cell viability, it also leads to reduced ABE production (Jones et al., 2018). Interestingly, coenzyme A transferase (CoAT) activity was abolished in solB knockout mutants, resulting in complete loss of acetone production with a marked decrease in butanol production (Jones et al.,2018). This suggests that the regulatory activity of solB may be concentration dependent. In addition, a small abundance of SolB transcripts is probably necessary for the stabilization of CoAT for optimal activity. The lack of acetone production in solB knockout mutant could be primarily due to the lack of ctfAB transcription. Transcriptomic analysis and ABE production pattern in the SolB overexpressing mutant show that the plausible mechanism of regulation of ABE production is via SolB in solventogenic Clostridium species. Northern blotting, q-RT-PCR and transcriptomics analysis have showed that solB transcripts are present in low quantities in C. acetobutylicum wildtype during the acidogenic phase followed by remarkable increase in expression as fermentation progresses (Wang et al., 2013; Jones et al., 2018). Overexpression of SolB in C. acetobutylicum led to a notable decrease in transcript levels of solvent producing sol operon and adc gene in the mutant (Jones et al., 2018). Conversely, deletion of solB impacted the expression of ctfAB and adhE1, but did not affect adc expression (Jones et al., 2018). Therefore, a plausible regulatory mechanism for solvent production in C. acetobutylicum arises when considering the upregulation of SolB during butanol stress and its correlation with the progression of fermentation, leading to the accumulation of primarily butanol. This is further influenced by the impact of ctfAB and adhE1 deletion on coenzyme A expression and solvent production. It suggests that initial low levels of solB are necessary for the expression of sol operon genes. However, as butanol builds up in the fermentation medium during the process, the transcription of SolB intensifies, targeting mRNAs produced from the sol operon for degradation. This, in turn, leads to the regulation of ABE production. This could help elucidate the decrease in ABE production rate once a certain butanol concentration threshold is reached in the fermentation medium. Given these observations, it is conceivable that SolB acts as a regulatory inhibitor of butanol biosynthesis, potentially exerting its influence even before butanol titer reaches a toxic threshold. Wang et al. (2013) noted that solB expression increased following supplementation of the fermentation medium with 2 g/L butanol. Nevertheless, the precise threshold and mechanism by which butanol triggers the accumulation of SolB transcripts remain unclear. Given that the coding region of SolB is located upstream of the adhE2 gene, it is conceivable that the solB sRNA transcript acts in a cis-like manner and promptly triggers the degradation of mRNA transcripts of adjacent genes.
Furthermore, riboswitches can mediate gene control by modulating ligand binding and influencing transcriptional regulation. For example, an S-box riboswitch is involved in the regulation of sulfur metabolism in C. acetobutylicum (André et al., 2008). In response to sulfur concentration in the cell, activation of the S-box riboswitch induces the production of antisense RNA to regulate the transcription of sulfur utilization genes (André et al., 2008). The abundance of synthesized antisense RNAs corresponds to changes in sulfur concentrations in the cells, leading to adjustments in gene transcript levels and the activities of proteins involved in sulfur utilization (André et al., 2008). It is possible that SolB is transcribed because of activation of a riboswitch located at the 5′ untranslated region of the sol operon. While a direct interaction between butanol and a regulatory element on DNA is unlikely, there is a possibility that one or more intermediate regulatory protein(s) bind to the riboswitch or directly to the operator sequence on the solB promoter. Moreover, the regulatory protein(s) may either detach or bind in the presence of butanol, facilitating transcription of the solB gene. It is also plausible that the riboswitch/solB gene senses butanol concentration through the intermediary regulatory protein, leading to the production of SolB transcripts in response to increasing butanol levels, providing an explanation for the observed phenotype. Notably, Spo0A null mutants did not produce acetone, but rather butanol and ethanol in relatively small amounts relative to the C. beijerinckii wildtype (Ravagnani et al., 2002, Harris et al., 2002). These findings are consistent with the ABE production pattern in SolB null mutants indicating an association between SolB and Spo0A functions. Although, Spo0A is the master regulator of ABE production, SolB may serve as the regulation effector for ABE production to maintain cellular homeostasis. A comprehensive investigation of the regulation of solvent production by solB requires in-depth studies on the impact of butanol on the upregulation of solB expression. Understanding the interaction between SolB and sol locus genes during transcription and translation processes and their role in modulating CoAT activity, is vital. It may be crucial to uncover the relationships between the accumulation of butanol in the fermentation medium and the expression of SolB and Spo0A. This knowledge may be instrumental in applying metabolic engineering techniques to enhance the ABE production capacity of solventogenic Clostridium species. Additionally, insight into the influence of SolB on genes regulating ABE production (hbd, bcd, bdhB, bdhA, thl) would deepen our understanding of the regulatory mechanisms of SolB.
3.2. The 6S RNA and tmRNA Mediated Regulation
The 6S RNA in E. coli and Bacillus subtilis activates the transcription of stationary phase genes via inhibition of exponential phase RNA polymerase holoenzyme, while enabling the accumulation and activity of the stationary phase RNA polymerase holoenzyme (Barrick et al., 2005; Gildehaus et al., 2007; Neusser et al., 2010). Specifically, 6S RNA forms a binding interaction with σA (σ70) RNA polymerase, thereby impeding DNA binding and repressing transcription initiated by σA-dependent promoters during the stationary phase. Sigma A (σA), commonly known as vegetative sigma factor, initiates transcription of genes associated with exponential growth phase in Clostridium species. Interestingly, 6S RNA is associated with stress response and survival in E. coli and B. subtilis (Trotochaud and Wassarman, 2006). While the regulatory functions of 6S RNA in E. coli and B. subtilis have been extensively studied (Wassarman, 2018), there are limited information on its role in solventogenic Clostridium species. Given that ABE production in solventogenic Clostridium species occurs predominantly during the stationary growth phase, it is crucial to investigate the regulatory functions of 6S RNA in this group of microorganisms at this phase. Likewise, further studies are needed to understand the contribution of transfer-messenger RNA (tmRNA) in restoring suppressed ribosomal functions, which play a role in cell survival under stress conditions (Shin and Price, 2007). In cases where protein synthesis is suppressed due to damaged mRNA, the tmRNA complex, consisting of tmRNA together with small protein B, ribosomal protein S1, and elongation factor EF-Tu, triggers the activation of complex factors. This activation leads to an improvement in ribosomal functions and the recycling of ribosomes, allowing ongoing protein synthesis to continue. At the same time, tmRNA serves as a marker for damaged proteins that are destined for intracellular degradation (Withey and Friedman, 2003). The multifunctional nature of tmRNA, encompassing both tRNA and mRNA functions, facilitates the recycling of ribosomes for continuous repair and protein synthesis, thereby promoting cell survival (Keiler, 2008; Craig et al., 2014).
Meanwhile, there are three homologues of the 6S RNA gene in the genome of C. acetobutylicum contains. Over-expression of a homolog (6S RNA) in C. acetobutylicum resulted in increased ABE production, growth, and cell survival under butanol stress (Jones et al., 2016). Other homologs are probably inactive in the cells. Although 6S RNA is constitutively expressed in cells, it is present in small abundances during the exponential growth phase and its accumulation is characteristic of the transition to stationary phase. The relatively larger abundance of 6S RNA in the stationary phase relative to exponential growth phase results in the repression of σA dependent gene transcription and other genes activated by the alternative sigma factor B (also called σ38 in E. coli). The σ38 is a stationary phase sigma factor that is associated with the regulation of stress response genes, which are activated during conditions such as osmotic shock, oxidative stress, and nutrient starvation of Gram-negative microbes such as E. coli (Sharma and Chatterji, 2010). Similarly, in Gram positive bacteria such as B. subtillis and L. monocytogenes, transcription of stress-related genes is regulated by sigma factor B (σB), which is more abundant when there are stressors such as heat, acids, and solvent accumulations. The σB protein has been detected in C. acetobutylicum and it is known to modulate the stress response machinery (e.g., GrpE, DnaK DnaJ) in the microorganisms (Venkataramanan et al., 2015; Jones et al., 2016). Additionally, the vegetative sigma factor (σA) in Clostridium species regulates initiation of sporulation during the stationary phase (see Durré, 2016 for a review on Clostridia sporulation). The sporulation specific σE and SpoIIGA genes required for initiation of sporulation contain the σA promoter-specific sequence along with 0A boxes consistent with regulation of transcriptional activation by both σA and Spo0A (Santangelo et al., 1998). Constitutive overexpression of 6S RNA, which inhibits σA-dependent promoters, could delay or inhibit onset of sporulation. Meanwhile, constitutive overexpression of tmRNA only results in increased butanol tolerance and cell survival of C. acetobutylicum without increasing ABE production (Jones et al., 2016). Excess tmRNAs likely trigger a stress response and subsequent protein degradation driven by the action of the σE (stress and extracellular protein sigma factor) proteins which lead to the activation of genes encoding proteases. These proteases target repressors of membrane proteins and ultimately cause the bacterial membrane to thicken (Craig et al., 2014; Rhodius et al., 2005; Konovalova et al., 2018). Additionally, tmRNAs might play a specific role in degradation of mRNAs that encode proteins required for substrate uptake and acid production prior to the onset of solventogenesis (Craig et al., 2014). Taken together, while elevated levels of tmRNAs in C. acetobutylicum improved microbial tolerance to ABE, it did not result in enhanced ABE production. The use of inductive or regulatable promoters to control tmRNA abundance may address this limitation and potentially increase ABE production.
3.3. Sr8384 Mediated Regulation of Growth and ABE Production
A recently identified type of small non-coding RNA has shown to control both growth and ABE production in C. acetobutylicum. Phenotypic screening of a transposon-mediated mutant library of C. acetobutylicum revealed the existence of a coding sequence for a novel sncRNA (sr8384) located in the intergenic region between CAC2383 and CAC2384. (Yang et al., 2020). Knockdown of sr8384 in C. acetobutylicum resulted in a strain with impaired growth and poor ABE production. In fact, a 50% knockdown of sr8384 mRNA transcripts resulted in ~1.6-fold reduction in cell population and ~1.4-fold decrease in butanol titer relative to the wildtype strain with intact sr8384 (Yang et al., 2020). Furthermore, when sr8384 was overexpressed in C. acetobutylicum, the growth and ABE production capacity of the generated strain improved ~1.38- and ~1.35-fold, respectively, relative to the wildtype strain. Consequently, sr8384 was predicted to exert a regulatory influence directly or indirectly on both growth and ABE production in C. acetobutylicum. In addition, a homolog of sr8384 was discovered in the genome of C. beijerinckii (sr8889) in the intergenic region between Cbei_1788 and Cbei_1789. Interestingly, the genes Cbei_1788 and Cbei_1789 in C. beijerinckii show homology with the CAC2385 and CAC2383 genes, respectively, in C. acetobutylicum. The genes code for a hypothetical protein and a polysaccharide/xylanase/chitin deacetylase, respectively. Besides, sr8889 share a 51% nucleotide homology with sr8384 and is predicted to be a critical regulator of growth and ABE production in C. beijerinckii (Yang et al., 2020). For a comprehensive understanding of the regulatory functions of sr8384 in solventogenic Clostridium species, it is imperative to identify sr8384 proteins/genes, target markers, transcription inducers, and their expression pattern throughout the fermentation period.
4. Bacteria Secondary Metabolites and Their Potential Role in Regulation
Secondary metabolites, often referred to as biologically active compounds, are generated by microorganisms. While these secondary metabolites typically do not have a direct impact on the growth or survival of microorganisms, they can provide protection and competitive advantage against competitive microbes, and occasionally play a role in cell-to-cell signaling (Gokulan et al., 2014). Secondary metabolites are mostly species specific, and their production can occur in response to environmental stress such as extreme pH, temperature, nutrient deficiency, etc. Secondary metabolites in bacteria are synthesized via the β-lactam, oligosaccharide, shikimate, polyketide, and non-ribosomal pathways (Gokulan et al., 2014). Polyketides (PKs) and non-ribosomal peptides (NRPs) are the most studied chemically diverse families of secondary metabolites in bacteria. While polyketides are synthesized by polyketide synthases (PKSs), NRPs are produced when non-ribosomal peptide synthetases (NRPSs) are activated. Antimicrobial agents such as endo- and exo-toxins are examples of secondary metabolites, and some bacteria including the genus, Clostridium, produce secondary metabolites. Most prominent is the botulinum toxin synthesized by Clostridium botulinum, which is of clinical and cosmetic importance. The production of bioactive compounds, including antibiotics and lantibiotics, by Clostridium species has been reviewed by Pahalagedara et al. (2020). Solventogenic Clostridium species produce secondary metabolites in addition to ABE. For example, C. beijerinckii produce antibiotic clostrubin, which is active against methicillin- and vancomycin-resistant bacteria (Yang et al., 2015).
Previous research suggests the existence of secondary metabolites that serve as signal transduction molecules that may trigger important processes such as sporulation and ABE formation in solventogenic Clostridium species (Li et al., 2019 Alsaker et al., 2004; Herman et al., 2017). Genome mining efforts have revealed that Clostridium species possess unique potential for polyketide synthesis (Li et al., 2019). Furthermore, transcriptome analysis of C. acetobutylicum revealed the presence and expression of the type I modular polyketide synthase (pks) gene. There was a notable surge in the transcription of type I modular polyketide synthase (pks) gene during the early stationary growth phase (Alsaker et al., 2004). In vitro studies revealed that pks is responsible for the production of three polyketide compounds (clostrienoic acid, unidentified polyketide and clostrienose) from the condensation of malonyl coenzyme A (Herman et al., 2017). The C. acetobutylicum pks null mutant had a significantly delayed sporulation pattern, reduced granulose accumulation, and produced more butanol than the wildtype strain, although both strains had similar growth profiles (Herman et al., 2017). Remarkably, the addition of clostrinose to the medium resulted in a resurgence of granulose accumulation, overcoming its previous suppression (Herman et al., 2017). Polyketide compounds may have a broader presence and play more important role in regulating processes within solventogenic Clostridium species than previously thought. Hence, they represent promising targets for metabolic engineering aimed at delaying sporulation and increasing the duration of ABE production in these species.
4.1. Non-Ribosomal Peptides (NRPs) Mediated Regulation
The genome analysis of C. saccharoperbutylacetonicum N1-4 revealed its capacity to produce polyketides and non-ribosomal peptide secondary metabolites (Li et al., 2020). Additionally, bioinformatics investigation revealed the presence of seven biosynthesis gene clusters (nrps1-4 and hyb1-3; Li et al., 2020). In addition, transcriptomics analysis confirmed the expression of five of these gene clusters in C. saccharoperbutylacetonicum N1-4 during fermentation (Li et al., 2020). The nrps3 gene has been reported to be widely expressed and conserved in other solventogenic Clostridium species (Li et al., 2020). This gene codes for an N-acylated dipeptidyl alcohol thought to be involved in microbial tolerance to ABE (Li et al., 2020). Indeed, the C. saccharoperbutylacetonicum N1-4 nrps3 null mutant had a defective exponential growth pattern in medium supplemented with butanol. Transcriptomics analysis of the nrps3 null mutant revealed a downregulation of the glycerol metabolism operon. Despite the inability of C. saccharoperbutylacetonicum N1-4 to use glycerol as sole carbon source, it has been suggested that the glycerol metabolism operon contributes to ABE tolerance (Li et al., 2020).
Typically, bacteria synthesize glycerol to facilitate the production of glycerophospholipids, which are essential for cell membrane formation. Consequently, gene activation of glycerol metabolism is essential during the bacterial growth process. The glycerol metabolism operon DhaKLM in C. beijerinckii is predicted to be regulated by a σ54 dependent promoter activated by the dhaQ EBP (Nie et al., 2019). Thus, nrp3 could serve as an EBP activator signal for transcriptional activation of genes that regulate glycerol metabolism.
5. Potential Target Points for Metabolic Engineering
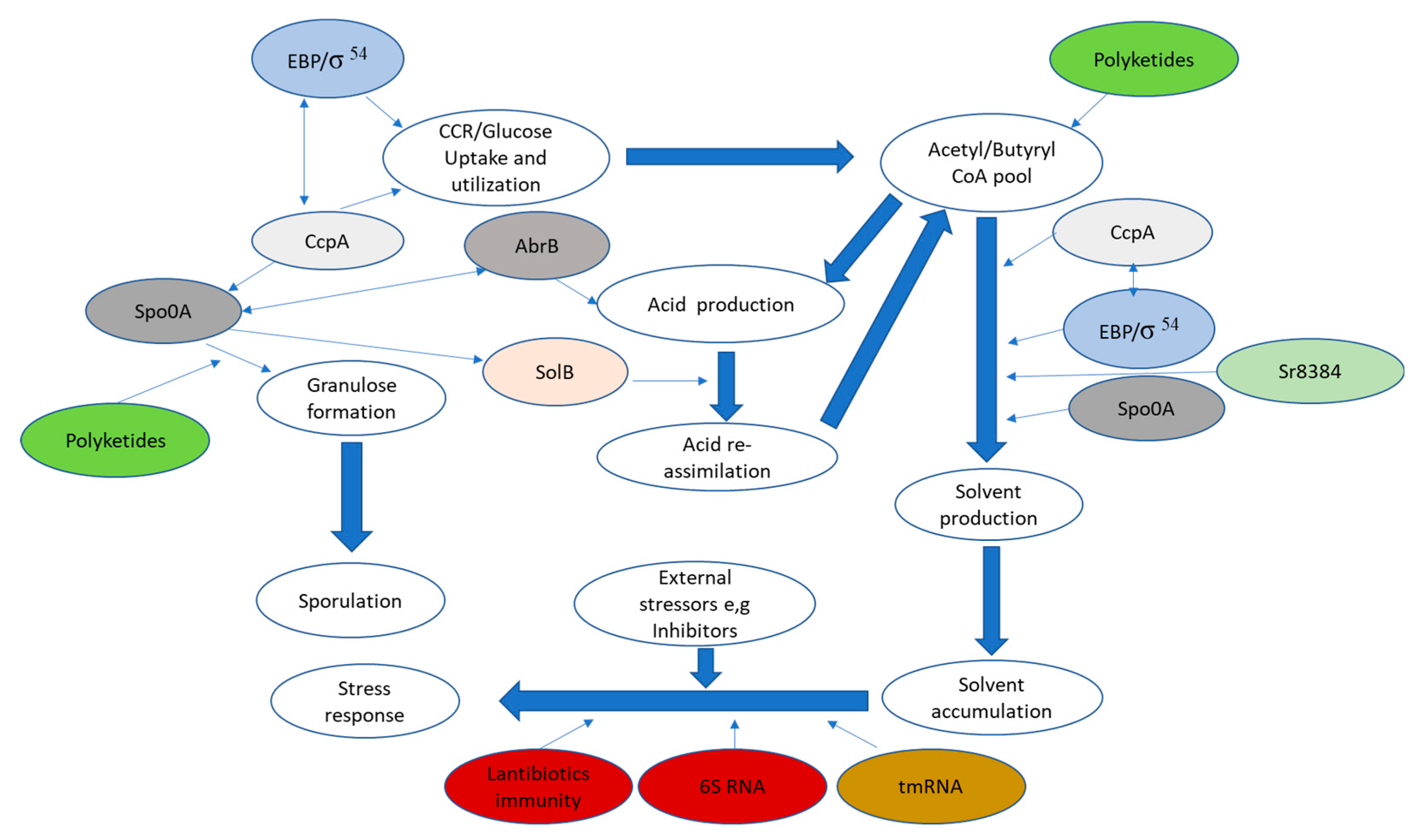
Metabolic network proteins in solventogenic
Clostridium species are tightly regulated as each regulatory molecule is associated with a different process and ultimately influences ABE production directly or indirectly (
Figure 2). Throughout the diverse stages of growth in solventogenic
Clostridium species and ABE production, there appear to be regulatory controls and equilibria in place. These mechanisms likely evolved to counteract potential adverse effects in the event of regulatory perturbations during different phases of ABE fermentation and ultimately ensure cell survival. The toxicity of butanol to solventogenic
Clostridium species is probably the main reason for the tight regulation of ABE production. Hence, the intricately interactive regulatory proteins and RNA networks involved in ABE production make metabolic engineering of solventogenic
Clostridium species challenging.
Research efforts are currently underway to identify new regulatory molecules and to gain an in-depth understanding of the functions of existing regulatory molecules in solventogenic Clostridium species during ABE fermentation. Findings from overexpression and knockout experiments involving genes responsible for regulatory molecules such as Spo0A, AbrB, CcpA, Rex, and CsrA suggest a trade-off scenario. This trade-off entails a balance between production-oriented phenotypes (e.g., increased population growth and ABE production) and survival-oriented phenotypes (e.g., ABE tolerance, stress response adaptation). Indeed, investigations into protein/RNA regulatory networks are starting to offer insights into important molecular markers and crucial molecular interactions that influence ABE production. These discoveries are significant for identifying promising targets for metabolic engineering, particularly when integrated into a comprehensive approach. A thorough understanding of the regulatory networks involving EBPs/sigma factor 54, sRNAs and secondary metabolites can be used to metabolically manipulate solventogenic Clostridium species to generate industrial strains with desirable phenotypes. Some potentially beneficial strategies are described in this review.
5.1. EBP/Sigma Factor 54 as Potential Targets for Metabolic Engineering
Sigma factors play a crucial and precise role in the regulation of gene transcription due to their ability to influence a series of cascading processes that can lead to either desirable or undesirable microbial phenotypes. The functional integrity of sigma factors depends on their sequence integrity. Direct alteration of the
sigL gene that encodes sigma factor 54 (σ
54), could therefore be counterproductive. Thus, altering enhancer binding proteins that control σ
54-dependent transcription could be an effective target for modulation. The EBP regulatory domains can be modified so that they are non-responsive to gene repressors. For example, EBPs of non-preferred sugar PTS operons are regulated by proteins of the PTS (Hpr, EI or EIIA/B) at the PTS regulatory domains (PRD). According to Nie et al. (2016), activated CelR and σ
54 directly regulate the transcription of the
Cel operon which is involved in cellobiose utilization. The P~His-HPr (Hpr protein phosphorylated on histidine residue) phosphorylation of His-551 activates CelR, while P~EIIB
Cel (phosphorylated cellobiose-specific EIIB protein) phosphorylation on His-829 in PRD2 inactivates CelR. In addition to this regulation, binding of CcpA-Hpr-Ser46 to the
Cre site within the coding region results in repression of target genes. This results in non-preferred sugars either remaining unused, being utilized only after preferred sugars are exhausted, or utilized to a limited degree—a phenomenon known as Carbon Catabolite Repression (CCR). Two possible approaches are necessary to counteract the repression of genes responsible for encoding proteins/enzymes involved in non-preferred sugar utilization and abolish CCR without interfering with other processes regulated by CcpA (
Figure 2). To alleviate the repression of genes responsible for non-preferred sugar utilization and eliminate CCR without impacting other CcpA-regulated processes, two potential mechanisms are considered. First, the interaction of CcpA with the typical
Cre site hinges on its interaction with Hpr-ser46. This interaction can be disrupted by either suppressing phosphorylation of Hpr’s ser46 or targeting its CcpA interaction domain, thereby preserving the structural integrity of CcpA for regulation of processes that are not connected to CCR (
Figure 1). Second, the need for P~His-HPr phosphorylation on His-551 that trigger CelR activation can be bypassed by modifying CelR’s His-551, rendering it constitutively active. Even in the presence of glucose, CelR remains active and ensures the transcription of the
Cel operon genes. A process akin to desensitization of CelR to P~His-HPr phosphorylation can be extended to other PTS/non-PTS sugars or non-sugars. This will help in the simultaneous utilization of diverse sugars (e.g., hydrolysates from lignocellulosic biomass) to increase ABE production. Moreover, identifying activators/repressors of EBPs is crucial for understanding how they can be modified to resist repression, thereby promoting a well-regulated expression of sugar transports and utilization genes and ABE production. Delineating the relationship between EBPs and CcpA and how this association affects σ
54-dependent transcription can be a useful strategy in developing effective metabolic engineering approaches for enhanced ABE production (
Figure 1,
Table 2).
5.2. sRNAs as Potential Targets for Metabolic Engineering
Given that knockout and overexpression of SolB in solventogenic Clostridium species typically result in the production of strains that produce relatively reduced amounts of ABE, metabolic manipulation of SolB through interference with butanol sensors will be likely more feasible. If SolB is an sRNA produced as part of a riboswitch-mediated regulation, then the intermediary regulatory protein(s) can be engineered to reduce sensitivity to butanol. Decreased sensitivity leads to a decrease in SolB RNA transcripts, resulting in maintenance of expression of ABE formation genes. Metabolic engineering of SolB can be performed in conjunction with overexpression of tmRNA. Accumulated ABE can influence mRNA structures, leading to inhibited ribosomal functions and misfolding of proteins, and ultimately impacting the viability of solventogenic Clostridium species. Overexpression of tmRNA could potentially boost the ability of solventogenic Clostridium species to revive ribosomal functionality and improve cellular responses to disrupted functions. This metabolic engineering strategy can lead to the development of strains with enhanced ABE production. Understanding the scope of SolB regulation necessitates a thorough investigation into its effects on non-sol operon ABE formation genes. Furthermore, the recently identified sr8384 represents a promising target marker molecule that warrants further investigation. Examining the direct or indirect target(s) of sr8384 and elucidating its induction mechanism would offer valuable insights into the role of sr8384 in regulating ABE production in solventogenic Clostridium species.
5.3. Secondary Metabolites as Potential Targets for Metabolic Engineering
Recent evidence has shown that solventogenic Clostridium species produce metabolites that are of medicinal importance (Li et al., 2020; Pahalagedara et al., 2020; Pahalagedara et al., 2021; Lincke et al., 2010). The understanding that solventogenic Clostridium species possess the capability to produce secondary metabolites with potential medical significance is emerging. The potential role of these secondary metabolites or compounds in regulating ABE production could spark a new avenue of exploration in the study of solventogenic Clostridium species and ABE production. Apart from metabolic engineering for enhanced ABE production, conferring cellular resistance to LDMICs present in LBH may be of great importance for the development of industrially applicable strains. For example, the synthesis of lantibiotics and immunity proteins from solventogenic Clostridium species can be exploited for metabolic engineering to generate ABE- and LDMICs-tolerant strains. Polyketide synthesis factors in solventogenic Clostridium species can also be engineered to delay sporulation and prolong the duration of ABE production.
6. Conclusions
Solventogenic Clostridium species are gaining increasing interest in biotechnology due to their inherent ability to metabolize a wide variety of substrates to produce fuels and chemicals. To ensure sustainability in the energy, transportation, and chemical sectors, it is essential to develop sustainable raw materials with environmentally compatible technologies to produce fuels and chemicals. To achieve this goal, regulation of processes in fermenting microorganisms such as solventogenic Clostridium species during growth and ABE fermentation is essential. Although much has been achieved to gain a better understanding of the complex regulatory networks in this group of microorganisms, there are still knowledge gaps that need to be filled to piece together the physiological solventogenic functions of Clostridium species. Targeted metabolic engineering and development of solventogenic Clostridium species, therefore, needs to be a priority if there is to be efficacious production of fuels and chemicals, which may not be possible without a comprehensive understanding of the physiology of this group of microorganisms. Clearly, understanding the regulatory molecules and networks highlighted in this review can lead to the development of effective metabolic engineering approaches in solventogenic Clostridium species to sustainably produce fuels and chemicals.
Acknowledgments
This work was funded by the National Science Foundation Cellular & Biochemical Engineering program (Award Number: 1803022), USDA NIFA Hatch grant (Project No. OHO01333), and U.S. Department of Agriculture under the award number 2014-38502-22598 through South Central Sun Grant Program USDA-NIFA Project number OKL03163. The authors express their gratitude to Dr. James Kinder from the Department of Animal Sciences at The Ohio State University for reviewing the manuscript and offering invaluable feedback.
References
- Agu, C.V.; Ujor, V.; Ezeji, T.C. Metabolic engineering of Clostridium beijerinckii to improve glycerol metabolism and furfural tolerance. Biotechnol. Biofuels 2019, 12, 50. [Google Scholar] [CrossRef]
- Alsaker, K.V.; Spitzer, T.R.; Papoutsakis, E.T. Transcriptional Analysis of spo0A Overexpression in Clostridium acetobutylicum and Its Effect on the Cell's Response to Butanol Stress. J. Bacteriol. 2004, 186, 2006–2018. [Google Scholar] [CrossRef]
- André, G.; Even, S.; Putzer, H.; Burguière, P.; Croux, C.; Danchin, A.; Martin-Verstraete, I.; Soutourina, O. S-box and T-box riboswitches and antisense RNA control a sulfur metabolic operon of Clostridium acetobutylicum. Nucleic Acids Res. 2008, 36, 5955–5969. [Google Scholar] [CrossRef]
- Argaman, L.; Hershberg, R.; Vogel, J.; Bejerano, G.; Wagner, E.H.; Margalit, H.; Altuvia, S. Novel small RNA-encoding genes in the intergenic regions of Escherichia coli. Curr. Biol. 2001, 11, 941–950. [Google Scholar] [CrossRef]
- Aristilde, L.; Lewis, I.A.; Park, J.O.; Rabinowitz, J.D. Hierarchy in Pentose Sugar Metabolism in Clostridium acetobutylicum. Appl. Environ. Microbiol. 2015, 81, 1452–1462. [Google Scholar] [CrossRef]
- Baral, N.R.; Shah, A. Microbial inhibitors: formation and effects on acetone-butanol-ethanol fermentation of lignocellulosic biomass. Appl. Microbiol. Biotechnol. 2014, 98, 9151–9172. [Google Scholar] [CrossRef] [PubMed]
- Barrick, J.E.; Sudarsan, N.; Weinberg, Z.; Ruzzo, W.L.; Breaker, R.R. 6S RNA is a widespread regulator of eubacterial RNA polymerase that resembles an open promoter. RNA 2005, 11, 774–784. [Google Scholar] [CrossRef] [PubMed]
- Biebl, H. Fermentation of glycerol by Clostridium pasteurianum -- batch and continuous culture studies. J. Ind. Microbiol. Biotechnol. 2001, 27, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Brosse, A.; Guillier, M. Bacterial Small RNAs in Mixed Regulatory Networks. In Regulating with RNA in Bacteria and Archaea; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018; pp. 453–469. [Google Scholar] [CrossRef]
- Buck, M.; Cannon, W. Activator-independent formation of a closed complex between ?54-holoenzyme and nifH and nifU promoters of Klebsiella pneumoniae. Mol. Microbiol. 1992, 6, 1625–1630. [Google Scholar] [CrossRef] [PubMed]
- Buckel, W.; Barker, H.A. Two Pathways of Glutamate Fermentation by Anaerobic Bacteria. J. Bacteriol. 1974, 117, 1248–1260. [Google Scholar] [CrossRef] [PubMed]
- Bush, M.; Dixon, R. The Role of Bacterial Enhancer Binding Proteins as Specialized Activators of σ 54 -Dependent Transcription. Microbiol. Mol. Biol. Rev. 2012, 76, 497–529. [Google Scholar] [CrossRef]
- Chen, Y.; Indurthi, D.C.; Jones, S.W.; Papoutsakis, E.T. Small RNAs in the Genus Clostridium. mBio 2011, 2, e00340-10. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Bao, T.; Yang, S.-T. Engineering Clostridium for improved solvent production: recent progress and perspective. Appl. Microbiol. Biotechnol. 2019, 103, 5549–5566. [Google Scholar] [CrossRef] [PubMed]
- Craig, N.L.; Cohen-Fix, O.; Storz, G. Molecular Biology: Principles of Genome Function; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Dang, L.H.; Bettegowda, C.; Huso, D.L.; Kinzler, K.W.; Vogelstein, B. Combination bacteriolytic therapy for the treatment of experimental tumors. Proc. Natl. Acad. Sci. USA 2001, 98, 15155–15160. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.C.; Kesthely, C.A.; Franklin, E.A.; MacLellan, S.R. The essential activities of the bacterial sigma factor. Can. J. Microbiol. 2017, 63, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Deutscher, J.; Aké, F.M.D.; Derkaoui, M.; Zébré, A.C.; Cao, T.N.; Bouraoui, H.; Kentache, T.; Mokhtari, A.; Milohanic, E.; Joyet, P. The Bacterial Phosphoenolpyruvate:Carbohydrate Phosphotransferase System: Regulation by Protein Phosphorylation and Phosphorylation-Dependent Protein-Protein Interactions. Microbiol. Mol. Biol. Rev. 2014, 78, 231–256. [Google Scholar] [CrossRef]
- Deutscher, J.; Francke, C.; Postma, P.W. How Phosphotransferase System-Related Protein Phosphorylation Regulates Carbohydrate Metabolism in Bacteria. Microbiol. Mol. Biol. Rev. 2006, 70, 939–1031. [Google Scholar] [CrossRef] [PubMed]
- Dror, T.W.; Morag, E.; Rolider, A.; Bayer, E.A.; Lamed, R.; Shoham, Y. Regulation of the Cellulosomal celS ( cel48A ) Gene of Clostridium thermocellum Is Growth Rate Dependent. J. Bacteriol. 2003, 185, 3042–3048. [Google Scholar] [CrossRef] [PubMed]
- Dror, T.W.; Rolider, A.; Bayer, E.A.; Lamed, R.; Shoham, Y. Regulation of Expression of Scaffoldin-Related Genes in Clostridium thermocellum. J. Bacteriol. 2003, 185, 5109–5116. [Google Scholar] [CrossRef]
- Dürre, P. Physiology and Sporulation in Clostridium. In The Bacterial Spore; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2016; pp. 313–329. [Google Scholar] [CrossRef]
- Dürre, P.; Hollergschwandner, C. Initiation of endospore formation in Clostridium acetobutylicum. Anaerobe 2004, 10, 69–74. [Google Scholar] [CrossRef]
- Ezeji, T.C.; Milne, C.; Price, N.D.; Blaschek, H.P. Achievements and perspectives to overcome the poor solvent resistance in acetone and butanol-producing microorganisms. Appl. Microbiol. Biotechnol. 2010, 85, 1697–1712. [Google Scholar] [CrossRef]
- Ezeji, T.; Qureshi, N.; Blaschek, H.P. Butanol production from agricultural residues: Impact of degradation products on Clostridium beijerinckii growth and butanol fermentation. Biotechnol. Bioeng. 2007, 97, 1460–1469. [Google Scholar] [CrossRef]
- Georg, J.; Hess, W.R. cis -Antisense RNA, Another Level of Gene Regulation in Bacteria. Microbiol. Mol. Biol. Rev. 2011, 75, 286–300. [Google Scholar] [CrossRef]
- George, H.A.; Johnson, J.L.; Moore, W.E.C.; Holdeman, L.V.; Chen, J.S. Acetone, Isopropanol, and Butanol Production by Clostridium beijerinckii (syn. Clostridium butylicum ) and Clostridium aurantibutyricum. Appl. Environ. Microbiol. 1983, 45, 1160–1163. [Google Scholar] [CrossRef] [PubMed]
- Gildehaus, N.; Neußer, T.; Wurm, R.; Wagner, R. Studies on the function of the riboregulator 6S RNA from E. coli: RNA polymerase binding, inhibition of in vitro transcription and synthesis of RNA-directed de novo transcripts. Nucleic Acids Res. 2007, 35, 1885–1896. [Google Scholar] [CrossRef]
- Gokulan, K.; Khare, S.; Cerniglia, C. Metabolic Pathways: Production of Secondary Metabolites of Bacteria. In Encyclopedia of Food Microbiology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 561–569. [Google Scholar] [CrossRef]
- Gottumukkala, L.D.; Haigh, K.; Görgens, J. Trends and advances in conversion of lignocellulosic biomass to biobutanol: Microbes, bioprocesses and industrial viability. Renew. Sustain. Energy Rev. 2017, 76, 963–973. [Google Scholar] [CrossRef]
- Han, B.; Ujor, V.; Lai, L.B.; Gopalan, V.; Ezeji, T.C. Use of proteomic analysis to elucidate the role of calcium on acetone-butanol-ethanol (ABE) fermentation in Clostridium beijerinckii NCIMB 8052. Appl. Environ. Microbiol. 2013, 79, 282–293. [Google Scholar] [CrossRef]
- Han, B.; Gopalan, V.; Ezeji, T.C. Acetone production in solventogenic Clostridium species: new insights from non-enzymatic decarboxylation of acetoacetate. Appl. Microbiol. Biotechnol. 2011, 91, 565–576. [Google Scholar] [CrossRef]
- Han, Y.-F.; Xie, B.-T.; Wu, G.-X.; Guo, Y.-Q.; Li, D.-M.; Huang, Z.-Y. Combination of Trace Metal to Improve Solventogenesis of Clostridium carboxidivorans P7 in Syngas Fermentation. Front. Microbiol. 2020, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.M.; Welker, N.E.; Papoutsakis, E.T. Northern, Morphological, and Fermentation Analysis of spo0A Inactivation and Overexpression in Clostridium acetobutylicum ATCC 824. J. Bacteriol. 2002, 184, 3586–3597. [Google Scholar] [CrossRef]
- Herman, N.A.; Kim, S.J.; Li, J.S.; Cai, W.; Koshino, H.; Zhang, W. The industrial anaerobe Clostridium acetobutylicum uses polyketides to regulate cellular differentiation. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- Hocq, R.; Bouilloux-Lafont, M.; Ferreira, N.L.; Wasels, F. σ54 (σL) plays a central role in carbon metabolism in the industrially relevant Clostridium beijerinckii. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Isar, J.; Rangaswamy, V. Improved n-butanol production by solvent tolerant Clostridium beijerinckii. Biomass- Bioenergy 2012, 37, 9–15. [Google Scholar] [CrossRef]
- Jiang, Y.; Xu, C.; Dong, F.; Yang, Y.; Jiang, W.; Yang, S. Disruption of the acetoacetate decarboxylase gene in solvent-producing Clostridium acetobutylicum increases the butanol ratio. Metab. Eng. 2009, 11, 284–291. [Google Scholar] [CrossRef]
- Jones, A.J.; Fast, A.G.; Clupper, M.; Papoutsakis, E.T. Small and Low but Potent: the Complex Regulatory Role of the Small RNA SolB in Solventogenesis in Clostridium acetobutylicum. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.J.; Venkataramanan, K.P.; Papoutsakis, T. Overexpression of two stress-responsive, small, non-coding RNAs, 6S and tmRNA, imparts butanol tolerance inClostridium acetobutylicum. FEMS Microbiol. Lett. 2016, 363, fnw063. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.T.; Woods, D.R. Acetone-butanol fermentation revisited. Microbiol. Rev. 1986, 50, 484–524. [Google Scholar] [CrossRef] [PubMed]
- Jones, S. Sigma 54 minds the gap. Nat. Rev. Microbiol. 2009, 7, 3. [Google Scholar] [CrossRef]
- Keiler, K.C. Biology of trans-Translation. Annu. Rev. Microbiol. 2008, 62, 133–151. [Google Scholar] [CrossRef]
- Konovalova, A.; Grabowicz, M.; Balibar, C.J.; Malinverni, J.C.; Painter, R.E.; Riley, D.; Mann, P.A.; Wang, H.; Garlisi, C.G.; Sherborne, B.; et al. Inhibitor of intramembrane protease RseP blocks the σ E response causing lethal accumulation of unfolded outer membrane proteins. Proc. Natl. Acad. Sci. USA 2018, 115, 201806107–E6621. [Google Scholar] [CrossRef]
- Li, J.S.; Barber, C.C.; Zhang, W. Natural products from anaerobes. J. Ind. Microbiol. Biotechnol. 2018, 46, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Li, J.S.; Barber, C.C.; A Herman, N.; Cai, W.; Zafrir, E.; Du, Y.; Zhu, X.; Skyrud, W.; Zhang, W. Investigation of secondary metabolism in the industrial butanol hyper-producer Clostridium saccharoperbutylacetonicum N1-4. J. Ind. Microbiol. Biotechnol. 2020, 47, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Lincke, T.; Behnken, S.; Ishida, K.; Roth, M.; Hertweck, C. Closthioamide: An Unprecedented Polythioamide Antibiotic from the Strictly Anaerobic Bacterium Clostridium cellulolyticum. Angew. Chem. 2010, 122, 2055–2057. [Google Scholar] [CrossRef]
- Liou, J.S.-C.; Balkwill, D.L.; Drake, G.R.; Tanner, R.S. Clostridium carboxidivorans sp. nov., a solvent-producing clostridium isolated from an agricultural settling lagoon, and reclassification of the acetogen Clostridium scatologenes strain SL1 as Clostridium drakei sp. nov. Int. J. Syst. Evol. Microbiol. 2005, 55, 2085–2091. [Google Scholar] [CrossRef] [PubMed]
- Maczulak, A. Clostridium. Encycl. Microbiol. 2011, 168–173. [Google Scholar]
- Morett, E.; Buck, M. In Viva Studies on the Interaction of RNA Polymerase-a54 with the Klebsiella pneumoniae and Rhizobium meliloti nzfw Promoters. 13.
- Neusser, T.; Polen, T.; Geissen, R.; Wagner, R. Depletion of the non-coding regulatory 6S RNA in E. coli causes a surprising reduction in the expression of the translation machinery. BMC Genom. 2010, 11, 1–14. [Google Scholar] [CrossRef]
- Nie, X.; Dong, W.; Yang, C. Genomic reconstruction of σ54 regulons in Clostridiales. BMC Genom. 2019, 20, 1–14. [Google Scholar] [CrossRef]
- Nie, X.; Yang, B.; Zhang, L.; Gu, Y.; Yang, S.; Jiang, W.; Yang, C. PTS regulation domain-containing transcriptional activator CelR and sigma factor σ54 control cellobiose utilization in Clostridium acetobutylicum. Mol. Microbiol. 2016, 100, 289–302. [Google Scholar] [CrossRef]
- Noguchi, T.; Tashiro, Y.; Yoshida, T.; Zheng, J.; Sakai, K.; Sonomoto, K. Efficient butanol production without carbon catabolite repression from mixed sugars with Clostridium saccharoperbutylacetonicum N1-4. J. Biosci. Bioeng. 2013, 116, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Nölling, J.; Breton, G.; Omelchenko, M.V.; Makarova, K.S.; Zeng, Q.; Gibson, R.; Lee, H.M.; Dubois, J.; Qiu, D.; Hitti, J.; et al. Genome Sequence and Comparative Analysis of the Solvent-Producing BacteriumClostridium acetobutylicum. J. Bacteriol. 2001, 183, 4823–4838. [Google Scholar] [CrossRef] [PubMed]
- Okonkwo, C.C.; Azam, M.M.; Ezeji, T.C.; Qureshi, N. Enhancing ethanol production from cellulosic sugars using Scheffersomyces (Pichia) stipitis. Bioprocess Biosyst. Eng. 2016, 39, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Okonkwo, C.C.; Ujor, V.; Ezeji, T.C. Chromosomal integration of aldo-keto-reductase and short-chain dehydrogenase/reductase genes in Clostridium beijerinckii NCIMB 8052 enhanced tolerance to lignocellulose-derived microbial inhibitory compounds. Sci. Rep. 2019, 9, 7634. [Google Scholar] [CrossRef] [PubMed]
- Pahalagedara, A.S.N.W.; Flint, S.; Palmer, J.; Brightwell, G.; Gupta, T.B. Antimicrobial production by strictly anaerobic Clostridium spp. Int. J. Antimicrob. Agents 2020, 55, 105910. [Google Scholar] [CrossRef] [PubMed]
- Pahalagedara, A.S.N.W.; Jauregui, R.; Maclean, P.; Altermann, E.; Flint, S.; Palmer, J.; Brightwell, G.; Gupta, T.B. Culture and genome-based analysis of four soil Clostridium isolates reveal their potential for antimicrobial production. BMC Genom. 2021, 22, 1–14. [Google Scholar] [CrossRef]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates. II: inhibitors and mechanisms of inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Patil, R.C.; Suryawanshi, P.G.; Kataki, R.; Goud, V.V. Current challenges and advances in butanol production. In Sustainable Bioenergy; Elsevier: Amsterdam, The Netherlands, 2019; pp. 225–256. [Google Scholar] [CrossRef]
- Patyar, S.; Joshi, R.; Byrav, D.P.; Prakash, A.; Medhi, B.; Das, B. Beaviecwteria in cancer therapy: A novel experimental strategy. J. Biomed. Sci. 2010, 17, 21. [Google Scholar] [CrossRef]
- Ravagnani, A.; Jennert, K.C.B.; Steiner, E.; Grünberg, R.; Jefferies, J.R.; Wilkinson, S.R.; Young, D.I.; Tidswell, E.C.; Brown, D.P.; Youngman, P.; et al. Spo0A directly controls the switch from acid to solvent production in solvent-forming clostridia. Mol. Microbiol. 2000, 37, 1172–1185. [Google Scholar] [CrossRef]
- Ren, C.; Gu, Y.; Hu, S.; Wu, Y.; Wang, P.; Yang, Y.; Yang, C.; Yang, S.; Jiang, W. Identification and inactivation of pleiotropic regulator CcpA to eliminate glucose repression of xylose utilization in Clostridium acetobutylicum. Metab. Eng. 2010, 12, 446–454. [Google Scholar] [CrossRef]
- Ren, C.; Gu, Y.; Wu, Y.; Zhang, W.; Yang, C.; Yang, S.; Jiang, W. Pleiotropic functions of catabolite control protein CcpA in Butanol-producing Clostridium acetobutylicum. BMC Genom. 2012, 13, 349. [Google Scholar] [CrossRef] [PubMed]
- A Rhodius, V.; Suh, W.C.; Nonaka, G.; West, J.; A Gross, C. Conserved and Variable Functions of the σE Stress Response in Related Genomes. PLOS Biol. 2005, 4, e2. [Google Scholar] [CrossRef] [PubMed]
- Richards, G.R.; Vanderpool, C.K. Molecular call and response: The physiology of bacterial small RNAs. Biochim. et Biophys. Acta (BBA) - Gene Regul. Mech. 2011, 1809, 525–531. [Google Scholar] [CrossRef]
- Saint-Amans, S.; Perlot, P.; Goma, G.; Soucaille, P. High production of 1,3-propanediol from glycerol by Clostridium butyricum VPI 3266 in a simply controlled fed-batch system. Biotechnol. Lett. 1994, 16, 831–836. [Google Scholar] [CrossRef]
- Santangelo, J.D.; Kuhn, A.; Treuner-Lange, A.; Dã¼Rre, P. Sporulation and time course expression of sigma-factor homologous genes inClostridium acetobutylicum. FEMS Microbiol. Lett. 1998, 161, 157–164. [Google Scholar] [CrossRef]
- Sauer, U.; Santangelo, J.D.; Treuner, A.; Buchholz, M.; Dürre, P. Sigma factor and sporulation genes inClostridium. FEMS Microbiol. Rev. 1995, 17, 331–340. [Google Scholar] [CrossRef]
- Scotcher, M.C.; Rudolph, F.B.; Bennett, G.N. Expression of abrB310 and sinR, and Effects of Decreased abrB310 Expression on the Transition from Acidogenesis to Solventogenesis, in Clostridium acetobutylicum ATCC 824. Appl. Environ. Microbiol. 2005, 71, 1987–1995. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.K.; Chatterji, D. Transcriptional switching inEscherichia coliduring stress and starvation by modulation of σ70activity. FEMS Microbiol. Rev. 2010, 34, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.-H. Gene expression: transcription of the genetic code. In Diagnostic Molecular Biology; Academic Press: Cambridge, MA, USA, 2019; pp. 59–86. [Google Scholar] [CrossRef]
- Shin, J.-H.; Price, C.W. The SsrA-SmpB Ribosome Rescue System Is Important for Growth of Bacillus subtilisat Low and High Temperatures. J. Bacteriol. 2007, 189, 3729–3737. [Google Scholar] [CrossRef] [PubMed]
- Small, R. Botulinum toxin injection for facial wrinkles. American Family Physician 2014, 90, 168–75.
- Smith, I. Regulatory Proteins That Control Late-Growth Development. In Bacillus subtilis and Other Gram-Positive Bacteria; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 1993; pp. 785–800. [Google Scholar] [CrossRef]
- Staedtke, V.; Roberts, N.J.; Bai, R.-Y.; Zhou, S. Clostridium novyi-NT in cancer therapy. Genes Dis. 2016, 3, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Storz, G.; Vogel, J.; Wassarman, K.M. Regulation by Small RNAs in Bacteria: Expanding Frontiers. Mol. Cell 2011, 43, 880–891. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, L.; Bennett, G.N. Proteome analysis and comparison of lostridium acetobutylicum ATCC 824 and Spo0A strain variants. J. Ind. Microbiol. Biotechnol. 2006, 33, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Liu, Z.-Y.; Liu, Z.; Zheng, H.-J.; Li, F.-L. Comparative transcriptome analysis between csrA-disruption Clostridium acetobutylicum and its parent strain. Mol. Biosyst. 2015, 11, 1434–1442. [Google Scholar] [CrossRef]
- Tangney, M.; Galinier, A.; Deutscher, J.; Mitchell, W.J. Analysis of the Elements of Catabolite Repression in Clostridium acetobutylicum ATCC 824. J. Mol. Microbiol. Biotechnol. 2003, 6, 6–11. [Google Scholar] [CrossRef]
- Thomason, M.K.; Storz, G. Bacterial Antisense RNAs: How Many Are There, and What Are They Doing? Annu. Rev. Genet. 2010, 44, 167–188. [Google Scholar] [CrossRef] [PubMed]
- Thormann, K.; Feustel, L.; Lorenz, K.; Nakotte, S.; Dürre, P. Control of Butanol Formation in Clostridium acetobutylicum by Transcriptional Activation. J. Bacteriol. 2002, 184, 1966–1973. [Google Scholar] [CrossRef] [PubMed]
- Trotochaud, A.E.; Wassarman, K.M. 6S RNA Regulation of pspF Transcription Leads to Altered Cell Survival at High pH. J. Bacteriol. 2006, 188, 3936–3943. [Google Scholar] [CrossRef]
- Ujor, V.; Okonkwo, C.; Ezeji, T.C. Unorthodox methods for enhancing solvent production in solventogenic Clostridium species. Appl. Microbiol. Biotechnol. 2015, 100, 1089–1099. [Google Scholar] [CrossRef]
- Venkataramanan, K.P.; Min, L.; Hou, S.; Jones, S.W.; Ralston, M.T.; Lee, K.H.; Papoutsakis, E.T. Complex and extensive post-transcriptional regulation revealed by integrative proteomic and transcriptomic analysis of metabolite stress response in Clostridium acetobutylicum. Biotechnol. Biofuels 2015, 8, 1–29. [Google Scholar] [CrossRef]
- Venkataramanan, K.P.; Jones, S.W.; McCormick, K.P.; Kunjeti, S.G.; Ralston, M.T.; Meyers, B.C.; Papoutsakis, E.T. The Clostridium small RNome that responds to stress: the paradigm and importance of toxic metabolite stress in C. acetobutylicum. BMC Genom. 2013, 14, 849. [Google Scholar] [CrossRef]
- Vogel, J.; Sharma, C.M. How to find small non-coding RNAs in bacteria. Biol. Chem. 2005, 386, 1219–1238. [Google Scholar] [CrossRef] [PubMed]
- Wach, W.; Harms, K.; Klingeberg, M.; Duerre, P.; Nold, N.; Schiel, B. Production of Acid and Solvent in Microorganisms. United. States Patent No. US8679799B2, 2014. [Google Scholar]
- Wang, Q.; Venkataramanan, K.P.; Huang, H.; Papoutsakis, E.T.; Wu, C.H. Transcription factors and genetic circuits orchestrating the complex, multilayered response of Clostridium acetobutylicum to butanol and butyrate stress. BMC Syst. Biol. 2013, 7, 120. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Mao, Y.; Blaschek, H.P. Genome-wide dynamic transcriptional profiling in clostridium beijerinckii NCIMB 8052 using single-nucleotide resolution RNA-Seq. BMC Genom. 2012, 13, 102. [Google Scholar] [CrossRef]
- Wassarman, K.M. 6S RNA: a regulator of transcription. Mol. Microbiol. 2007, 65, 1425–1431. [Google Scholar] [CrossRef]
- Wietzke, M.; Bahl, H. The redox-sensing protein Rex, a transcriptional regulator of solventogenesis in Clostridium acetobutylicum. Appl. Microbiol. Biotechnol. 2012, 96, 749–761. [Google Scholar] [CrossRef]
- Withey, J.H.; Friedman, D.I. A Salvage Pathway for Protein Synthesis: tmRNA and Trans-Translation. Annu. Rev. Microbiol. 2003, 57, 101–123. [Google Scholar] [CrossRef]
- Wilde, E.; Hippe, H.; Tosunoglu, N.; Schallehn, G.; Herwig, K.; Gottschalk, G. Clostridium tetanomorphum sp. nov., nom. rev. Int. J. Syst. Evol. Microbiol. 1989, 39, 127–134. [Google Scholar] [CrossRef]
- Wilkinson, S.R.; Young, D.I.; Morris, J.G.; Young, M. Molecular genetics and the initiation of solventogenesis inClostridium beijerinckii(formerlyClostridium acetobutylicum) NCIMB 8052. FEMS Microbiol. Rev. 1995, 17, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Zhao, J.; Chen, L.; Yang, S.-T.; Bai, F. Recent advances and state-of-the-art strategies in strain and process engineering for biobutanol production by Clostridium acetobutylicum. Biotechnol. Adv. 2017, 35, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.; Yang, Y.; Chen, J.; Chen, L.; Yang, S.; Jiang, W.; Gu, Y. Roles of three AbrBs in regulating two-phase Clostridium acetobutylicum fermentation. Appl. Microbiol. Biotechnol. 2016, 100, 9081–9089. [Google Scholar] [CrossRef]
- Yang, B.; Nie, X.; Gu, Y.; Jiang, W.; Yang, C. Control of solvent production by sigma-54 factor and the transcriptional activator AdhR in Clostridium beijerinckii. Microb. Biotechnol. 2019, 13, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Li, J.; Li, A. Total synthesis of clostrubin. Nat. Commun. 2015, 6, 6445. [Google Scholar] [CrossRef]
- Yang, Y.; Lang, N.; Zhang, H.; Zhang, L.; Chai, C.; Jiang, W.; Gu, Y. The small noncoding RNA sr8384 determines solvent synthesis and cell growth in industrial solventogenic Clostridia [Preprint]. Microbiology 2019. [CrossRef]
- Yang, Y.; Nie, X.; Jiang, Y.; Yang, C.; Gu, Y.; Jiang, W. Metabolic regulation in solventogenic clostridia: regulators, mechanisms and engineering. Biotechnol. Adv. 2018, 36, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, H.; Lang, N.; Zhang, L.; Chai, C.; He, H.; Jiang, W.; Gu, Y. The Small RNA sr8384 Is a Crucial Regulator of Cell Growth in Solventogenic Clostridia. Appl. Environ. Microbiol. 2020, 86. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, L.; Huang, H.; Yang, C.; Yang, S.; Gu, Y.; Jiang, W. A Flexible Binding Site Architecture Provides New Insights into CcpA Global Regulation in Gram-Positive Bacteria. mBio 2017, 8. [Google Scholar] [CrossRef]
- Younesi, H.; Najafpour, G.; Mohamed, A.R. Ethanol and acetate production from synthesis gas via fermentation processes using anaerobic bacterium, Clostridium ljungdahlii. Biochem. Eng. J. 2005, 27, 110–119. [Google Scholar] [CrossRef]
- Zhang, Y.; Ezeji, T.C. Transcriptional analysis of Clostridium beijerinckii NCIMB 8052 to elucidate role of furfural stress during acetone butanol ethanol fermentation. Biotechnol. Biofuels 2013, 6, 66. [Google Scholar] [CrossRef]
- Zimmermann, T. Untersuchungen zur Butanolbildung von Hyperthermus butylicus und Clostridium acetobutylicum [Dissertation, Universität Ulm].2013. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).