1. Introduction
Industrial development has brought great benefits to humanity, but many of its processes and products have also negatively impacted natural ecosystems, affecting human health and all other forms of life. One of the critical cases is the production and use of conventional plastics, because they not only represent one of the causes of the depletion of fossil material, a non-renewable resource, but they are also one of the main sources of environmental pollution, due to their resistance to degradation, which causes large accumulations of these contaminants in different ecosystems [
1].
As reported [
2], the production of plastics is increasing, from 1.5 million tons (mTon) in 1950, 250 mTon in 2010, to 400.3 mTon in 2022. With the emergence of circular plastics, which entails A second use of the existing ones has achieved a slight decrease in global production, close to 10% [
2]. Plastics represent a large fraction of the waste buried in the natural habitat and in the aquatic ecosystem, where microplastics and plastic islands have been found [
3,
4]. According to reports from 2019, plastic pollution generated 1.8 billion tons of greenhouse gas emissions, which is equivalent to 3.4% of the total generated worldwide [
5].
Faced with this environmental crisis, the production of bioplastics, or green polymers of the future, is becoming established as an alternative to replace petroplastics [
6]. These polyesters are classified into three types [
1]: polylactic acid (PLA), polybutylene succinate (SPB) and polyhydroxyalkanoates (PHA); The latter are the only ones that are synthesized and catabolized naturally by bacteria and, therefore, are natural chemical compounds, which confers greater added value to their industrial production, estimating that in 2023, their global market will reach 57.8 million Dollars. If government regulations and policies continue to reduce single-use plastics, it is possible that by 2028 the PHA market will have an increase of close to 50%, represented by 98.5 million dollars [
7].
PHAs are secondary metabolites of a wide variety of bacteria, fungi and some plants, which produce and accumulate them as an energy reserve when external conditions make their survival difficult [
8]. These biopolymers have a high potential to replace conventional plastics of petrochemical origin, given that they achieve similar physicochemical characteristics, but also have superior properties of thermal processability and biocompatibility, making them very appropriate in the manufacture of biomedical, dental and electronic devices; as well as, in the areas of construction, automotive, packaging and agriculture [
3,
8]. Due to its biodegradable and biocompatible properties, its applications extend to the production of medical and dental devices; With these, tests are being developed to stimulate tissue regeneration and microsphere production for drug administration [
9].
Although a variety of microorganisms (more than 300 species) have been used to obtain a wide range of biopolymers, only a few bacteria have been extensively investigated for the manufacture of bioplastics, due to their higher efficiency and production rates. These include
Bacillus megaterium, Bacillus cereus, Bacillus subtilis, Pseudomonas aeruginosa, P. putida, Halomonas sp., Pseudomonas fluorescens, P. oleovorans, R. eutropha, and Cupriavidus necator [
10]. Particularly, the bacterium
Bacillus megaterium is an excellent biological machine to produce PHA because it has a robust expression system and a cell wall deficient in lipopolysaccharides, which facilitates the release of the biopolymer synthesized by the bacteria intracellularly [
11,
12].
Structurally, PHAs are classified, according to the composition of their monomers, into: (a) short chain (SCL), made up of monomers with 3 to 5 carbons, such as Poly-3 hydroxyvalerate [P(3HV)] and Poly-3-hydroxybutyrate [P(3HB)]; (b) medium chain (MCL), with monomers of 6 to 14 carbons, such as the copolymer 3-Poly-(3-Hydroxybutyrate-Co-3-Hydroxyvalerate) [3 3(HB-co-HV] or [p( 3HB-co-3HV] and, (c) mixed chain (MCM), which combine the previous two and therefore consist of monomers between 3 and 14 carbons, as is the case of Poly (3HB co-3HV-co -3HHx). From the industrial point of view, PHB and the copolymer Poly 3 (HB-co-HV) [
9] stand out. The length of the carbon chain defines the fragile nature for those with a short, or flexible chain. for medium chain ones [
13].
To a large extent, the chain length is influenced by the type of substrate used to induce the production of PHA of microbial origin. Substrates with a high carbohydrate content mainly stimulate the production of PHASCL polymers, while a high fatty acid content stimulates those of PHAMCM. The most investigated PHA PHASCL are those formed by poly-3-hydroxybutyrate (P3HB); They are semi-crystalline bipolymers and therefore fragile. To improve the mechanical properties of P3HB and expand its applications, the production of heteropolymers such as PHBV has been investigated, which due to the presence of the 3HV monomer in its structure, presents better flexibility characteristics, improving the properties of PHB [
9].
To induce the production of copolymers in bacteria, carbon substrates that include mixtures of pure volatile fatty acids (VFAs), or materials or wastewater with high VFA content are used: Bacteria of the genus Bacillus synthesize copolymers of P(3HB- co-3HV), in crops supplemented with a mixture of VFA, from the controlled hydrolysis of pea peels, apple pomace, onion peels and potato peels. The authors found that Bacillus cereus EGU43 cultured on pea hull hydrolysates, accumulated the copolymer of P(3HB-co-3HV), with a 3HV content of 1% w/w.
The co-culture of Pseudomonas sp. ST2 and Bacillus sp. CS8 in culture media with acetic and propionic acids as carbon source, supplemented with glucose, produces up to 35% P(3HB-co-3HV) copolymer; when a mixture of glucose and propionic acid is used, individual strains produce more PHA than when glucose is used as the sole carbon source. With Alcaligenes eutrophus NCIMB 11599 grown in valeric acid or propionic acid, P(3HB co-3HV) is obtained with better yields in the first case. Ralstonia eutropha KCTC 2658 grown in a mixture of acetic, propionic and butyric acid in a ratio of 1:2:2 can achieve a P(3HB-co-3HV) production of 50% of the PSC. Cupriavidus necator grown in wastewater effluent rich in acetic, propionic and butyric acid, without adding any exogenous substrate, produces a substantial amount of P(3HB-co-3HV) copolymer (55% of the PSC). Recently, Ferre-Guell and Winterburn obtained high concentrations of P(3HB-co-3HV) copolymer by growing Haloferax mediterranei in a mixture of butyrate and valerate acids, with the addition of surfactants (Tween 80) to increase the bioavailability of the substrate, with a productivity of 10.2 mg/L*h, in batch fermentation. Such approaches can potentially reduce the production costs of PHAs and offer environmental benefits through waste reuse.
Now, although the global production capacity of bio-based PHA has expanded every year, industrial production of PHA has been limited due to its production costs (around USD 4,000–15,000/Tm), which significantly exceed the costs of polymers derived from fossil fuels (around USD 1000–1500/Tm) [
3]. The raw material (substrate) represents about 50% of the total production costs [
14], therefore, the profitability of PHA production is largely defined by the costs of raw materials, especially carbon source substrates. This is associated with the fact that the accumulation of PHA occurs under aerobic conditions, so a large part of the substrate is used in microbial intracellular respiration, with the formation of CO2 and water-soluble secondary metabolites; only part of the carbon source, much less than half, is directed to cellular biomass growth and PHA accumulation [
14].
In addition to the above, industrial PHA production processes are based on fermentation technology, in which they use expensive substrates (purified sugars, edible vegetable oils, among others) that, in addition, are part of the family basket; Therefore, its use puts the technology in competition with the food industry and makes the total production of PHA more expensive. Therefore, there is an urgent need to use carbon source substrates that do not affect food security, are economical and affordable and sustainable for the sustainable synthesis of PHAs [
3,
15].
Low-value substrates have been evaluated as carbon sources for the production of PHA, coming from different productive sectors: dairy industry (cheese/whey, dairy wastewater), oil and paper factories, agricultural crops (oil effluents palm/soybean/fruit,) and animal waste (chicken/cow manure). However, they are substrates that require prior treatments to be used as microbial culture media, which entails additional costs. For this reason, attention has been directed to using substrates that come from a first benefit such as the generation of biofuels, specifically volatile fatty acids (VFA), intermediate metabolites in the anaerobic digestion (AD) of residual biomass that are used for production biogas [
16]. In this sense, researchers have directed their attention to strengthening VFA production, as a collateral product of biogas production [
17]. Colombian researchers reported an interesting work in which they take advantage of fishing waste for this purpose, with which digestate rich in VFA is also obtained [
18].
Bacillus megaterium LVN01, native to Colombia, has shown the ability to produce PHAs from residual glycerol from the biodiesel industry [
19], carob fruits [
20], residues from fique processing (fique juice) [
12] and oil. of frying. In this research,
B. megaterium LVN1 showed the ability to synthesize PHBV from liquid digestate, a residual substrate from the anaerobic digestion of fish visors.
In this context, the PROBIOM (Production, structure and application of Biomolecules) research group of the Universidad Nacional de Colombia, contributed with its research experience, to give a second use to the waste from the fishing industry, through its participation in the royalty project "Strengthening of artisanal fishing activity in the Colombian Nariño Pacific towards a sustainable use of the resource. Tumaco". Within the project, a first alternative with fishing waste has been to produce biogas by the Environmental Prospective group of the National University of Colombia, Palmira headquarters; PROBIOM uses the digestate from this first phase as the main substrate of B. megaterium LVN01 for the production of PHA.
In Colombia, artisanal fishing supplies around 12,000 tons of fish/year to the national market, 8% of the total capture fishery in the country. It is estimated that 45% of the total catch by artisanal fishing becomes waste [
18,
21], which produces serious economic losses for this sector and environmental problems associated with the inadequate disposal of waste. In Colombia, waste fishing boats are thrown directly into the ocean or taken to land [
18]. The results of the research showed that the digestate has great potential as a sustainable substrate for the industrial production of PHA, while there is permanent availability of the starting material, giving added value to fishing waste.
2. Materials and Methods
2.1. Ethics Statement
The results of this research are articulated with the Agreement of Access to Genetic Resources and Derivatives No. 159, signed between the Ministry of Environment and Sustainable Development and the National University of Colombia, Resolution 0004, January 02, 2018 to the project called “Production and characterization of polyhydroxyalkaloates synthesized by native strains from organic waste.
2.2. Study Area and Bacterial Strain
B. megaterium LVN01 (Code GenBank: QJGY00000000.1) was isolated from soil samples collected in the municipality of Guarne (06°16′50"N and 75°26′37"W, department of Antioquia, Colombia) and initially characterized using molecular (16S rDNA sequence similarity), morphological, and biochemical techniques [
12]. Bacterial cultures were stored at -4 °C in LB broth with 20 % glycerol (v/v).
2.3. Carbon Source
A liquid residue from the biogas pilot plant, which operates with residual biomass from artisanal fishing waste, was utilized as a substrate for fermentations. The plant is situated in the city of San Andrés de Tumaco, on the premises of the National University of Colombia (at Tumaco, department of Nariño, Colombia). The samples were provided by the Environmental Prospective research group of the same University as part of the project "Strengthening Artisanal Fishing Activity in the Colombian Pacific of Nariño, Towards a Sustainable Use of the Resource".
This liquid residue, known as digestate, is a valuable source of Volatile Fatty Acids (VFA). For preservation, it was stored frozen at -4 °C, and before its use in the process, it underwent filtration through thick paper (pore size 20 μm) to remove sediments larger than 20 μm. This step facilitated microbial fermentation activity in the medium enriched with this substrate and prevented obstruction of the equipment sensors where the process was conducted.
The bromatological characterization of the digestate revealed the presence of several nutrients, including VFAs (59.04 ± 0.9 g/L), with significant concentrations of acetic acid (24.49 ± 1.82 g/L), butyric acid (11.81 ± 0.02 g/L), and isobutyric acid (9.36 ± 0.02 g/L). This composition represents a potential carbon source for
B. megaterium, which is of interest in this research (
Table S1).
2.4. Culture Media
Nutritive Agar (Merck, Microbiological Grade) composed of 5.0 x 10-3 g/L Pluripeptone, 3.0 x 10-3 g/L Meat Extract, 8.0 x 10-3 g/L Sodium Chloride, and 15.0 x 10-3 g/L Agar, with a final pH: 7.3 ± 0.2. Luria Bertani broth (LB broth) consisting of 10 g/L tryptone, 5 g/L yeast extract, and 10 g/L NaCl with the pH adjusted to 7.0 using approximately 0.2 mL of NaOH (5 N).
Minimum Medium Salts (MMS) supplemented with KH2PO4 (1.5 g/L), Na2HPO4 (3.6 g/L), (NH4)2SO4 (1.0 g/L), MgSO4 ⋅7H2O (0.2 g/L), yeast extract (0.1 g/L), residual glycerol (20 g/L) as a carbon source, 1.0 mL/L Trace Element (TE) solution consisting of FeSO4⋅7H2O (10 g/L), ZnSO4⋅7H2O (2.25 g/L), CuSO4⋅5H2O (1.0 g/L), MgSO4⋅5H2O (0.5 g/L), CaCl2⋅2H2O (2.0 g/L), NaB4O7⋅10H2O (0.23 g/L), (NH4)6Mo7O24 (0.1 g/L), and 10 mL HCl (35 %), with a final pH of 7.3 ± 0.2.
2.5. Activation of Bacterial Strain and Inoculum Preparation
2.5.1. Activation of B. Megaterium LVN01
10 µL of the previously preserved cells were cultured in LB broth with 20 % glycerol on Nutrient Agar plates at 37 °C for 24 h. The presence and purity of the microorganism were determined by Gram staining and spore staining with malachite green; PHA accumulation capacity was verified by Nile blue staining, described later in the paper.
2.5.2. Inoculum Preparation
For each fermentation, an aliquot of active B. megaterium LVN01 was resuspended in 10 mL of LB broth. The inoculated medium was incubated for 24 h in an orbital shaker (Heidolph Unimax 1010 with Inkubator 1000) at 37 °C and 200 rpm. Subsequently, 1 mL of the activated microorganism was diluted in 50 mL of LB culture medium in 250 mL flasks. The cultures were incubated in an orbital shaker (37 °C and 200 rpm) for 6 h or until the solution in each flask reached an optical density at 600 nm (OD600) greater than 1.0 unit. Finally, for the inoculum, a new fermentation was conducted taking 100 mL of the pre-inoculum and 200 mL of MMS medium. The culture was incubated in an orbital shaker (Heidolph Unimax 1010 with Inkubator 1000) at 37 °C and 200 rpm for approximately 24 h, until the estimated OD600 readings were between 0.1 and 0.3 units.
2.5.3. Gram Stain
The bacterial cell morphology of
B. megaterium LVN01 was approximately differentiated using the Gram staining protocol [
22]: after growing the culture on nutrient agar, a colony was taken and heat-fixed on a slide. Drops of crystal violet dye (Merck) were added consecutively for a minute and a half, followed by Lugol solution (Merck, 1%). The slide was then decolorized with alcohol-acetone (Merck, G.R) for 15 min. After washing with water and removing excess, a safranin solution (MERCK, G.R) was added for two minutes, followed by another wash with water. The stained slide was left to air dry and observed under a microscope (Leica DM500) (
Figure S1).
2.5.4. Spore Staining
The presence of spores was determined with the Shaeffer-Fulton methodology [
23,
24] from a culture of pure sporulated
B. megaterium. Sporulation was induced by incubating
B. megaterium LVN01 on nutrient agar at 44°C for 48 h. A smear of the culture was made, forming a homogeneous film to separate the microorganisms and fix them with heat. The smear was covered with malachite green aqueous solution (PANREAC AppliCHEM, for microbiology) for 1 minute at room temperature. The plate was then covered with filter paper and placed over a container with hot water for 10 minutes. As the dye evaporated due to the heat, drops were added to prevent the slide from drying out. After this step, the filter paper was removed and washed gently with plenty of water while the slide was left to cool at room temperature. Finally, the slide was covered with safranin for 2 minutes, washed, allowed to dry, and observed under the microscope (
Figure S2).
2.6. Visualization of PHA Accumulated by Bacillus Megaterium LVN01
2.5.1. Staining with Nile Red
100 µL of the strain were spread on Petri dishes with MMS, pH 7.0, supplemented with 20 g/L of glucose, 2 g/L of yeast extract, and 1 mL/L (0.1% in acetone) of Nile red dye (Sigma Aldrich, for microscopy) [
25]. The inoculated plates were incubated under aerobic conditions at room temperature for 72 h, and the resulting colonies were monitored at 24, 48, and 72 h under ultraviolet light at 340 nm to select those showing red-orange fluorescence, potentially classified as PHA producers (
Figure S3a).
2.5.2. Staining with Nile Blue
To confirm the accumulation of PHA in the bacteria selected with the Nile Red stain, they were stained with the Nile blue dye (Sigma Aldrich, for microscopy), following the methodology of Ostle and Holt, 1982 [
26]: A roast of the culture selected in the previous section was taken and transferred to nutrient agar medium and incubated at 37 °C for 24 h. A smear of each colony was made on a slide. Once fixed, the colony was covered with a 1% aqueous solution of Nile blue for 10 min at 55 °C; after this time, it was gently washed to remove excess dye. The plate was then covered with an 8 % (v/v) aqueous solution of acetic acid for 1 min at room temperature, excess was removed, and observed with a 100x lens in a fluorescence microscope at 450 nm to visualize the biopolymer granules (
Figure S3b).
2.6. Aerobic Fermentative Processes
The bioprocesses were carried out in a 7 L Bioreactor with a stirred tank (Applikon, equipped with two Rushton-type propellers and with EZ2 biocontroller, series 2310110012), operated in batch mode. The initial working volume was 3 L (diluted liquid digestate, MMS, and inoculum, in a ratio of 80:10:10) (
Figure S4). Temperature was set at 30.8 °C; stirring speed at 400 rpm and time at 60 h. To 300 mL of MMS, 300 mL of the
B. megaterium inoculum were added, followed by 2.4 L of the digestate or carbon source. Under these conditions, the initial OD
600 of the medium was recorded between 0.1 and 0.3 units. The fermentation medium was maintained at pH 7.0 ± 0.2 (adjusted using HCl or NaOH, 1 M, as required) and the dissolved oxygen was set at 100% saturation by injecting several pulses of extra dry industrial oxygen during fermentation (1 vvm), through a 0.22 μm vent filter, with an oxygen flow of 3 L/min to maintain aerobic conditions. Drops of a 10 % (v/v) antifoam solution of silicone (Antioqueña from chemicals, food grade) were added at the beginning of each batch and when necessary.
In this research, optimal operational parameters established by Gómez and collaborators [
19] were applied for PHA production using
B. megaterium LVN 1 and residual glycerol as a carbon source, in a 5 L bioreactor operated in batch mode, with an effective volume of 3 L. The proportion of digestate (carbon source) used was derived from a previous laboratory-scale study (unpublished data), which showed the highest values of PSC (27.1 mg) and PHA (14.8 mg) at 36 h, during batch fermentations in 500 mL flasks under orbital shaking (T: 30 °C; 200 rpm), pH: 7.0; and an MMS: Digestate ratio, 20:80.
Fermentation kinetics were monitored for 60 h, with samples (20 mL) taken in triplicate every two hours; bacterial growth was measured spectrophotometrically (OD600), followed by determination of dry cell weight (DCW), and PHA type biopolymer extraction.
2.6.1. Obtaining Biomass
Each sample was centrifuged at 10,000 g for 10 minutes using a centrifuge (Heraeus Megafuge 16R, Thermo Scientific). The supernatants were discarded, and the bacterial pellets were washed twice consecutively with 20 mL of deionized water followed by centrifugation at 10,000 g for 10 minutes at 4 °C. After the second wash, the granules were resuspended in 700 µL of deionized water and frozen at -4 °C until subsequent lyophilization (at -50 ºC, 0.01 mBar for 24 h).
2.6.2. Determination of DCW
Bacterial biomass was collected following the procedure described in item 2.6. The processed cells were dried in an oven at 60 °C (Binder class 2.0, Germany) and monitored until reaching a constant weight was reached. Dry cell weight (DCW) was reported as grams of cells per liter of culture medium.
2.7. Extraction and Purification of PHA
The biopolymer was isolated from the freeze-dried biomass by dispersion with organic solvents, following the methodology of Gómez-Cardozo and collaborators [
19]. The dry biomass was treated with a mixture of 10 % (v/v) sodium hypochlorite (NaClO) and chloroform (CHCl
3) (MERCK, 99 %) in a 1:1.5 ratio. The sample was homogenized by gentle and rapid shaking (vortex) and incubated at 40 °C and 200 rpm for 3 hours to break the microbial cell wall. Subsequently, it was centrifuged (10000 g, 10 min, 4 °C) to separate the phases. The organic phase containing the PHA was filtered through thick filter paper and collected in 2 mL Eppendorf tubes. The filtrate was dried at room temperature (27 °C) in a biosafety cabinet (BIOBASE, model BBS-DDC, series BBS11V1805175D, air speed 0.3 – 0.5 m/s), and its dry mass was measured using a Scale (Shimakzu, AUW22OD series, with working range from 220 g to 1 mg).
For the PHA purification of, the procedure of Gómez and collaborators (2020) was followed with some modifications: 500 µL of cold (0-4 ℃) 70 % v/v methanol (j.t. Baker, G.R) was added to each tube containing I the dry filtrate to precipitate the particles. The solution was refrigerated at 4 °C for 12 h and then centrifuged at 8000 g and 4 °C for 10 min. The solvent was evaporated at room temperature (27°C) in a gas extraction hood to recover the PHA.
Next, the biopolymer (PHA) was consecutively washed with n-hexane (MERCK, G.R), acetone (MERCK, G.R), and diethyl ether (Mallinckrodt, 99.9 %). Each solvent (250 μL) was added dropwise to the dry PHA sample, followed by centrifugation (8000 g, 4 °C, 10 min). The supernatant was discarded, and the precipitate was air-dried at room temperature (27 °C) for 12 h to remove any remaining solvent. This washing process was repeated 2 to 3 times until an almost white PHA was obtained to ensure its purity.
To calculate the PHA ratio based on the amount of accumulated dry weight, expressed as a percentage, the following equation was used:
2.8. PHA Characterization
2.8.1. Gas Chromatography-Mass Spectrometry (GC-MS/SIM)
The analysis of the samples was conducted according to ISO 12966-2:2017: “Preparation of methyl esters of fatty acids”, Second edition, 2017, part 2; with some modifications. Poly (3-hydroxybutyric acid) (Sigma-Aldrich, Lot: 09217BD-337) was used as a reference standard. The chromatographic analysis was performed on a GC AT 6890 Series Plus (AT, Palo Alto, California, USA), coupled to a mass selective detector (AT, MSD 5973), operated in SIM mode. The injection was performed in splitless mode (Viny = 2 μL). The column used in the analysis was 60 m x 0.25 mm x 0.25 μm DB-5MS 5%-Ph-PDMS.
2.8.2. Nuclear Magnetic Resonance Spectroscopy (1H-NMR and 13C-NMR)
A 600 MHz NMR spectrometer was used (Bruker, Avance III HD, nitrogen TCI cryoprobe, Topspin Software v3.6.5, 5mm tube). The sample was previously prepared by dissolving 30 mg of biopolymer in deuterated chloroform (CDCl3); 700 μL tetramethyl silane (TMS) was used as an internal control.
2.9. Thermal Properties of PHA
2.9.1. Differential Scanning Calorimetry (DSC)
A Q2000 TA INSTRUMENTS calorimeter (DSC) was used in this analysis. Exactly 5.72 mg of sample (PHA) was used for the analysis. The test was conducted with a flow of nitrogen as purge gas of 50 mL/min, then the equipment was parameterized for the material. There was a stabilization period at a temperature of 25°C, with heating up to 200°C at a speed of 10°C/min.
2.9.2. Thermogravimetric Analysis (TGA)
A METTLER TOLEDO TGA/SDTA851e thermogravimetric analyzer was used, operated under a Nitrogen atmosphere, with a flow rate of 40 mL/min. A 10 mg of polyhydroxybutyric acid (PHB) sample was subjected to a temperature range between 30 °C and 800 °C, at a rate of 10 °C/min.
2.10. Analysis and Presentation of Results
All tests conducted in this research were done in triplicate, the results are presented as median ± standard deviation in both tables and graphs. Sigma Plot 10.0 software was used to design the graphs, and the statistical analysis was developed in STATGRAPHICS Centurion XVI.II.
4. Discussion
Maximizing the production of PHA-type biopolymers through microbial biofermentation of different substrates has become a crucial field of research in the face of the growing demand for sustainable alternatives for the production of plastics. This bioindustry seeks environmentally friendly and economically viable processes that can compete with the petroleum-derived plastics industry [
39]. The main problem associated with the commercial disposal of bioplastics is the high production costs to which raw materials significantly contribute to [
15,
39,
40,
41]. Given this, researchers have turned their attention to the search for economic and sustainable substrates, especially residual biomass, that make an industrial process viable.
In this research, taking advantage of the ability of
B. megaterium LVN01 to metabolize different substrates through different pathways [
12,
19,
42,
43,
44,
45] was considered important to evaluate biogas digestate, a byproduct of anaerobic digestion of waste of artisanal fishing, not explored before, or at least not reported in the same sense of this research.
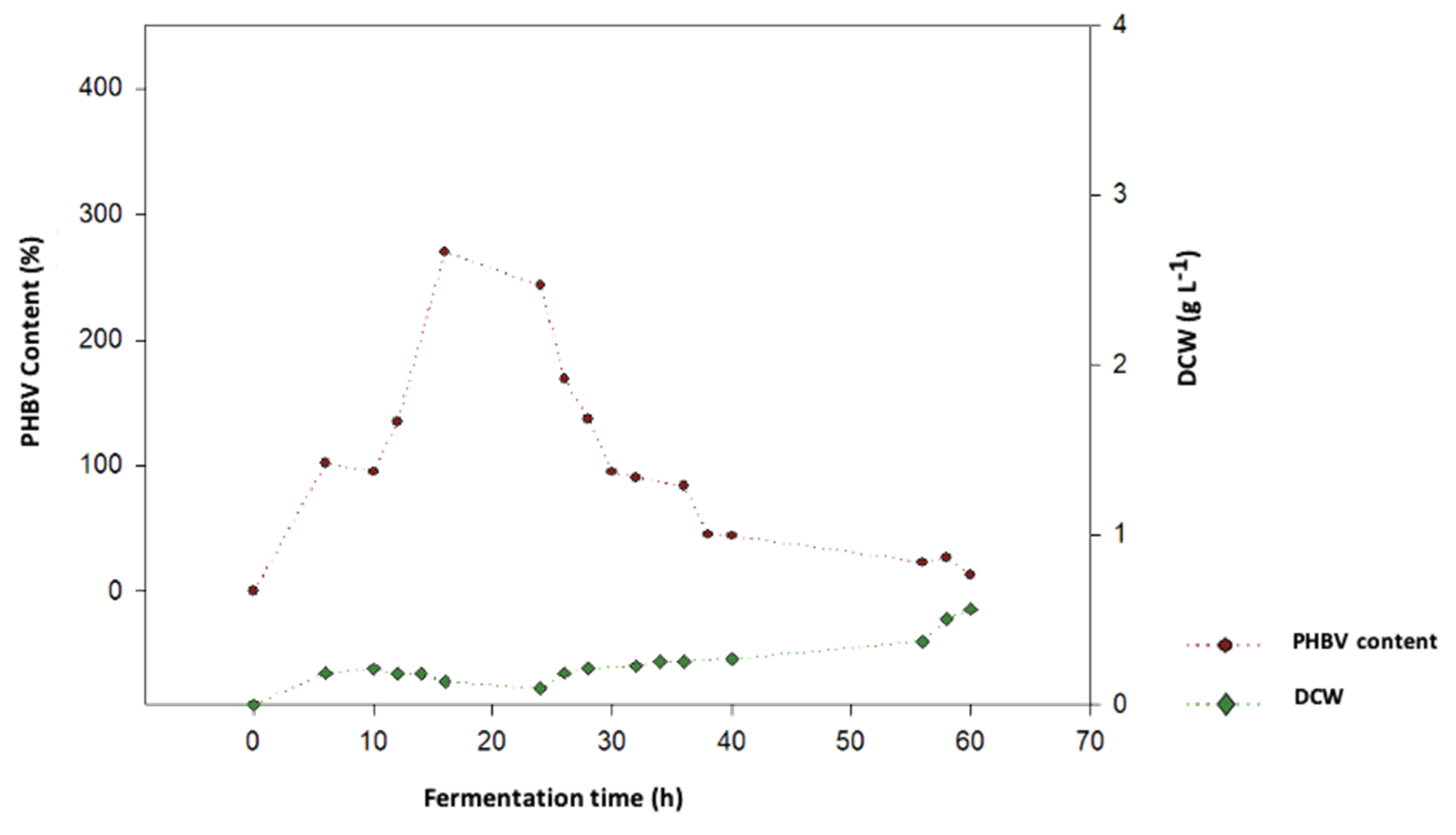
Our results demonstrate that biogas digestate, a substrate of residual origin rich in volatile fatty acids, is an excellent substrate for the growth of
B. megaterium and the production of PHA-type bioplastics. The dynamics of cell growth (
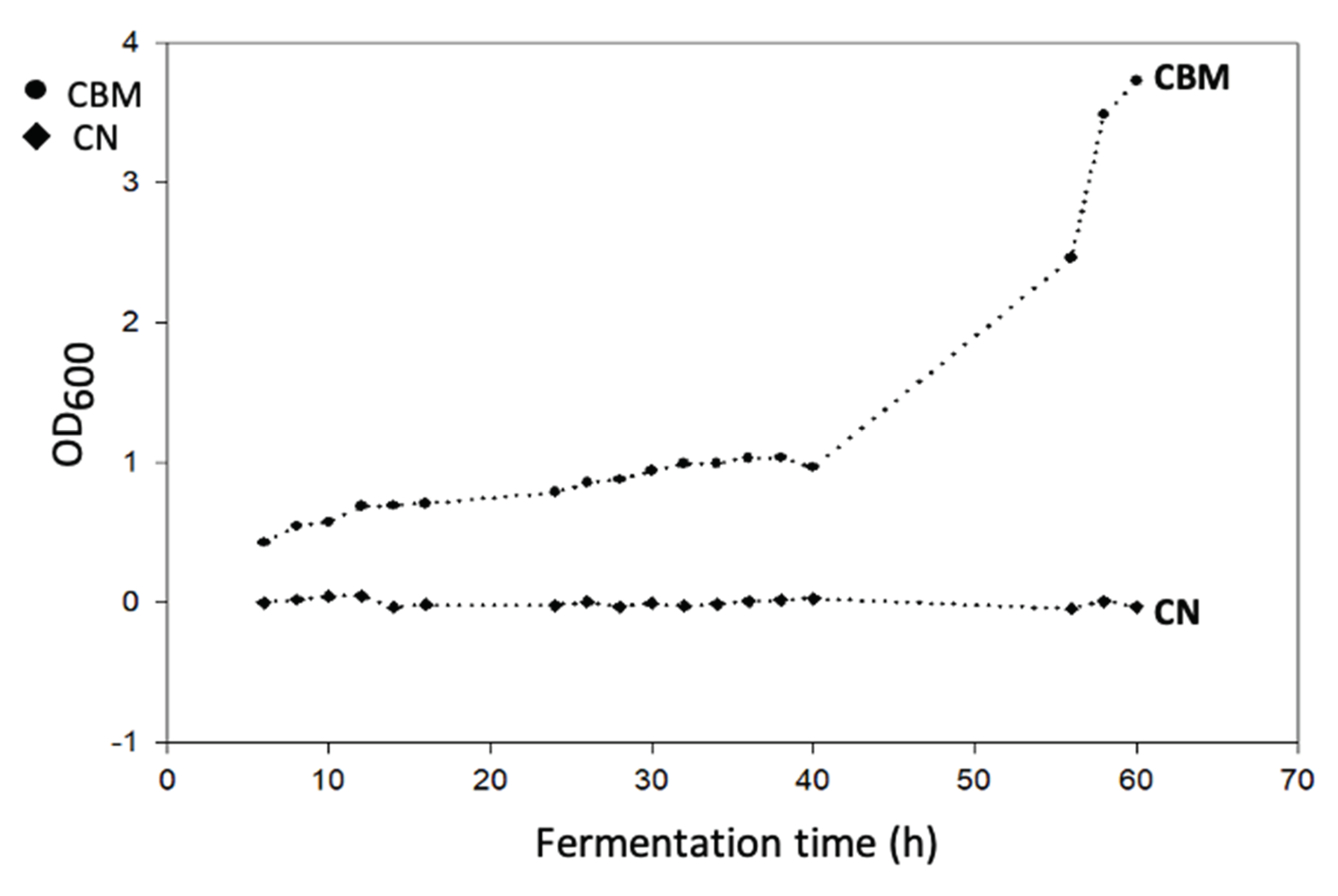
Figure 1), under the operational conditions of this work show the advantage of biogas digestate over residual glycerol, another substrate evaluated under similar conditions. As observed, the adaptive phase of
B. megaterium (LVN01) in the digestate lasts 40 h, almost double the time reported (24 h) for the same microbial strain in residual glycerol [
43]; but, once the exponential phase is reached, the microbial metabolism in digestate increases its efficiency (360 mg PHBV L-1 in 16 h). The optimal performance of
B. megaterium (LVN01) in digestate occurred at 16 h; in contrast, Gómez et al. showed that
B. megaterium (LVN01) in residual glycerol needs 48 h to reach a yield of 137.5 g PHB L-1 with a DCW of 58.1 mg PHB gcell-1, after this time these parameters decrease drastically [
42]
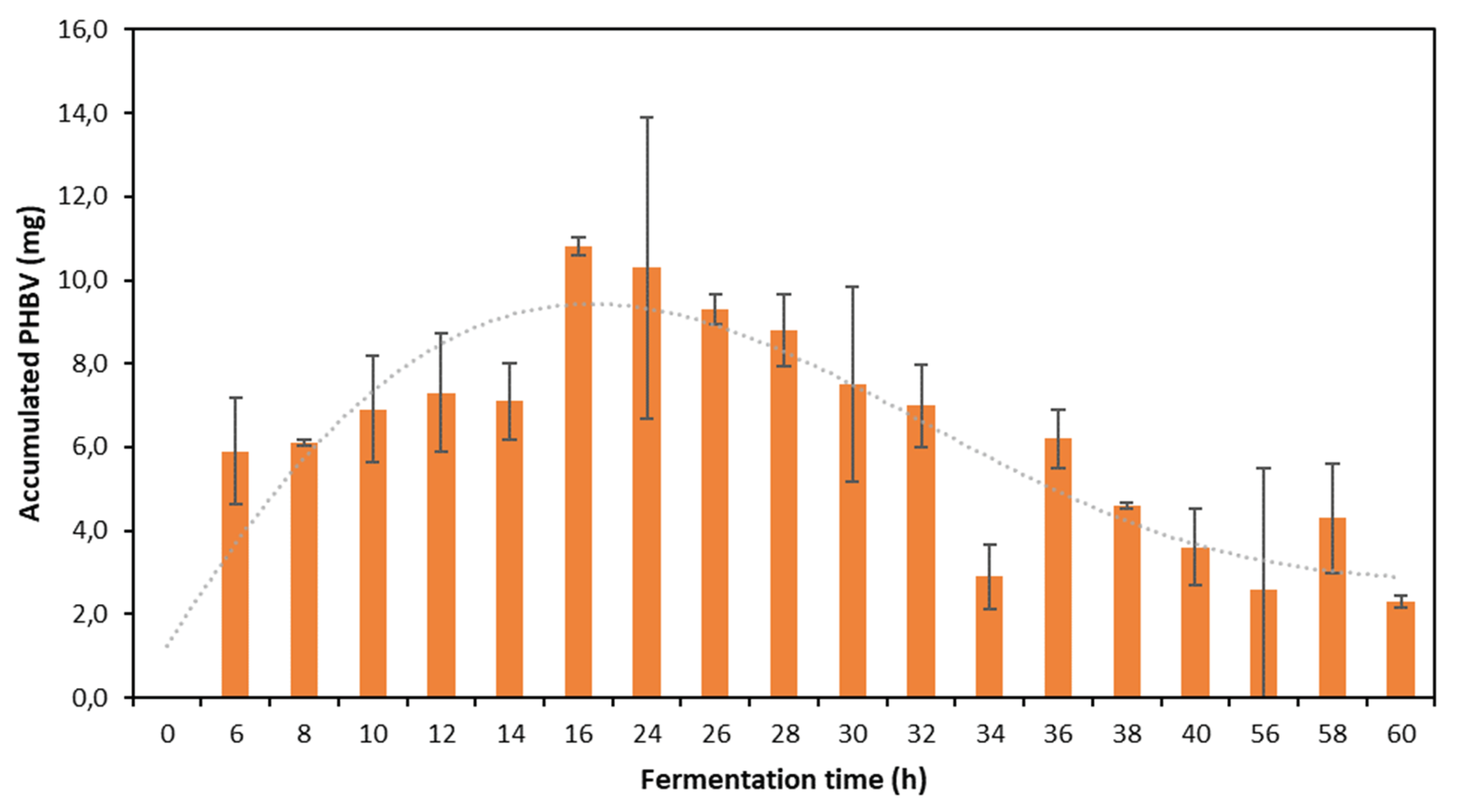
It is highlighted that, in the digestate, the DWC values are higher and the bacterial cells accumulate a greater amount of PHB per gram of biomass, in short periods of time. This excellent storage capacity of the biopolymer is particularly evident after the first 24 h of incubation and remains at relatively high levels until 32 h (
Figure 2). In agreement with what has been reported by other authors, it is inferred that the first stage of fermentation is a period of initial adaptation in which the bacteria multiply and increase their biomass before beginning to synthesize and accumulate PHA efficiently [
46,
47,
48,
49].
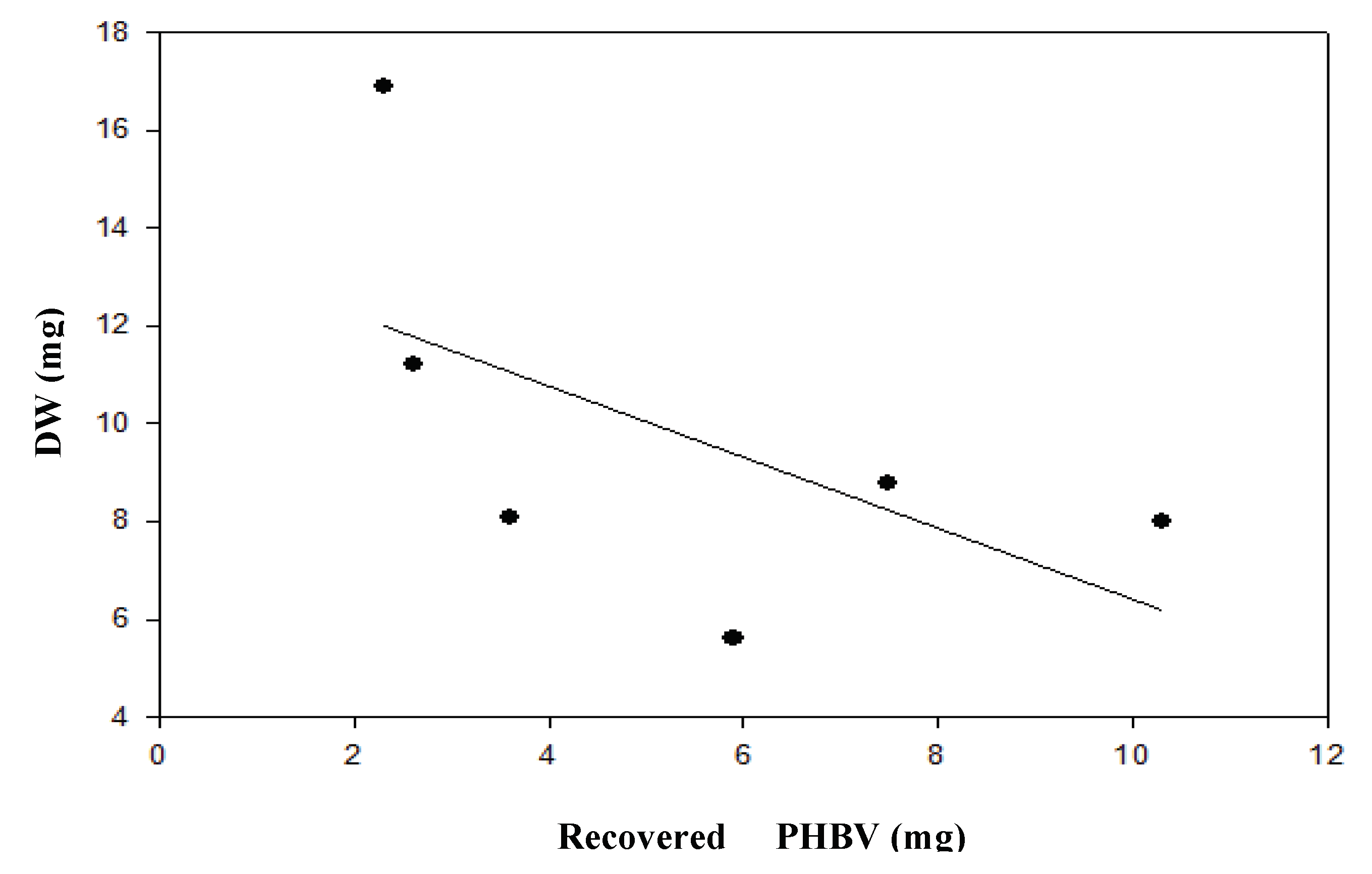
The results support the idea that microbial kinetics are strongly influenced by the chemical nature of the carbon source, unlike residual glycerol, the digestate is a complex mixture of organic compounds that includes fatty acids and other byproducts of anaerobic digestion of organic material. This complexity could require
B. megaterium (LVN01) a longer time to adapt to the medium and assimilate the available nutrients to grow efficiently, thus explaining its extended growth kinetics. Additionally, in the early stages of fermentation, rapid bacterial growth (higher DWC) is observed, before the bacteria prioritize PHB synthesis. This inverse relationship (
Figure 4) between biomass and PHB production follows the trend reported by other authors for
B. megaterium from other substrates [
19,
43,
50,
51].
In conclusion, the adaptive phase plays a crucial role in fermentation, being important for the microorganism to activate its metabolism and make the most of the nutrients in the culture medium. In complex substrates such as digestate,
B. megaterium requires long acclimatization times before it achieves optimal growth, while it can multiply rapidly in simple substrates. This information is essential to design and optimize fermentation processes, since the microbial adaptation time to the substrate can significantly affect the productivity and efficiency of the process [
52]. In this research,
B. megaterium was able to synthesize and accumulate PHBV during the first hours of the bioprocess (16h-32h), using biogas digestate as a carbon source, which is promising for efficient industrial production. Meanwhile, it could reduce dependence on oil, a non-renewable resource, and on expensive and less sustainable carbon sources such as agricultural crops [
53,
54,
55,
56,
57].
The chemical characterization of the biopolymer synthesized by B. megaterium from biogas digestate was conducted using gas chromatography with a mass selective detector (GC/MS) and Nuclear Magnetic Resonance Spectroscopy (1H-NMR and 13C NMR).
The results obtained in this research demonstrate the capacity of
B. megaterium to produce PHA from different substrates through fermentation processes. The chromatogram (GC/MS) of the polymeric extract (
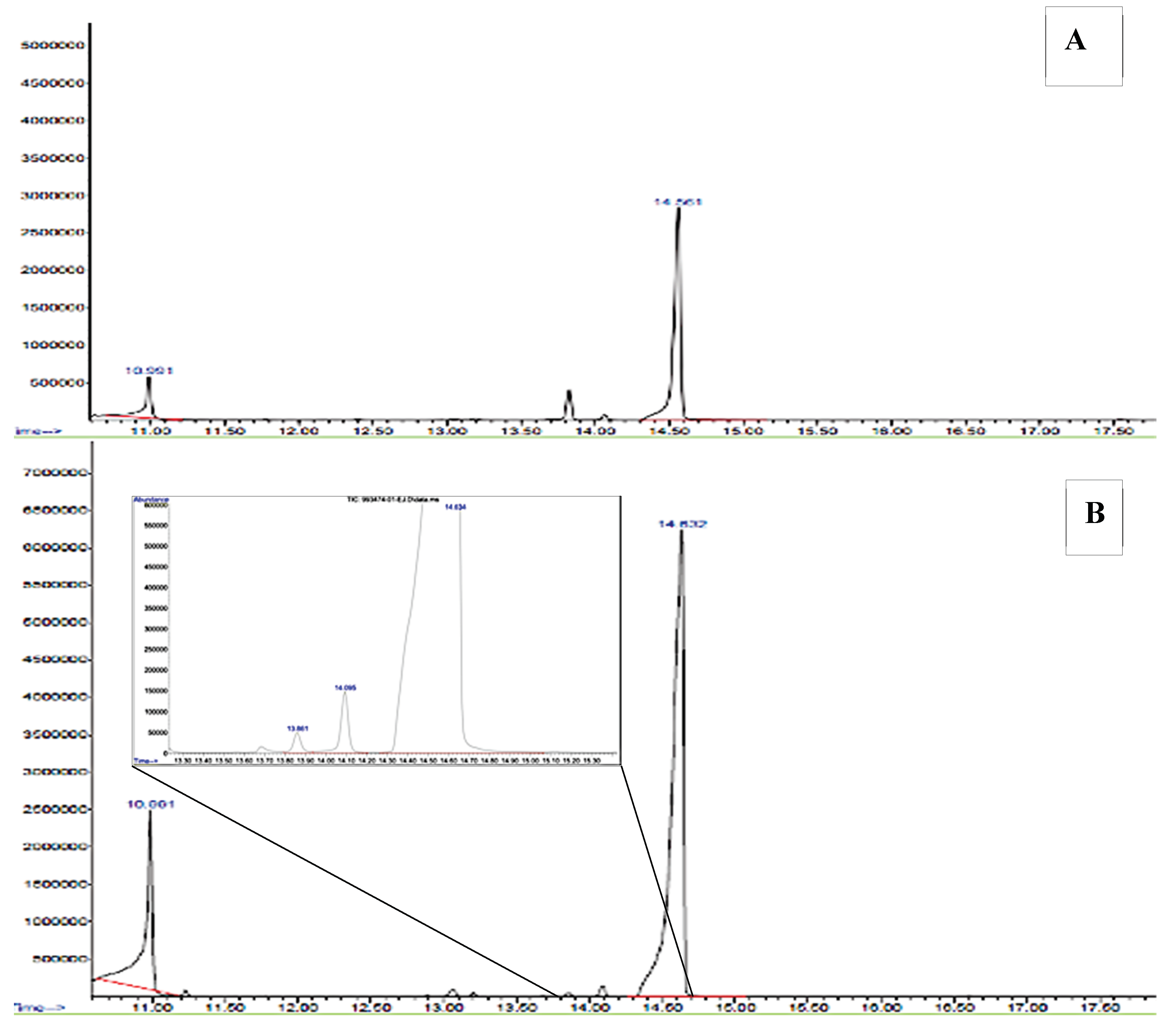
Figure 5) was compared with that of the commercial PHBV standard. In both cases, two main peaks were observed with retention times (TR) of 10.9 min, 14.6 min, identified by the GC/MS database as 2-Butenoic acid methyl ester and the methyl ether methyl ester of 3-hydroxybutyric acid, respectively, characteristic of the 3HB monomer. A peak of lower intensity appeared at 14.1 min, attributed to the methyl ester of 2-pentanoic acid, typical of the 3HV monomer. These are formed by methanolysis of 3HB, resulting in C4 esters, and 3HV, resulting in C4 and C5 esters (REF). 2-Butenoic, 3-hydroxybutyric, and 2-pentanoic acids are also byproducts of thermal degradation of PHBV [
58].
Nuclear magnetic resonance spectroscopy allowed us to elucidate the chemical structure of the biopolymer synthesized by
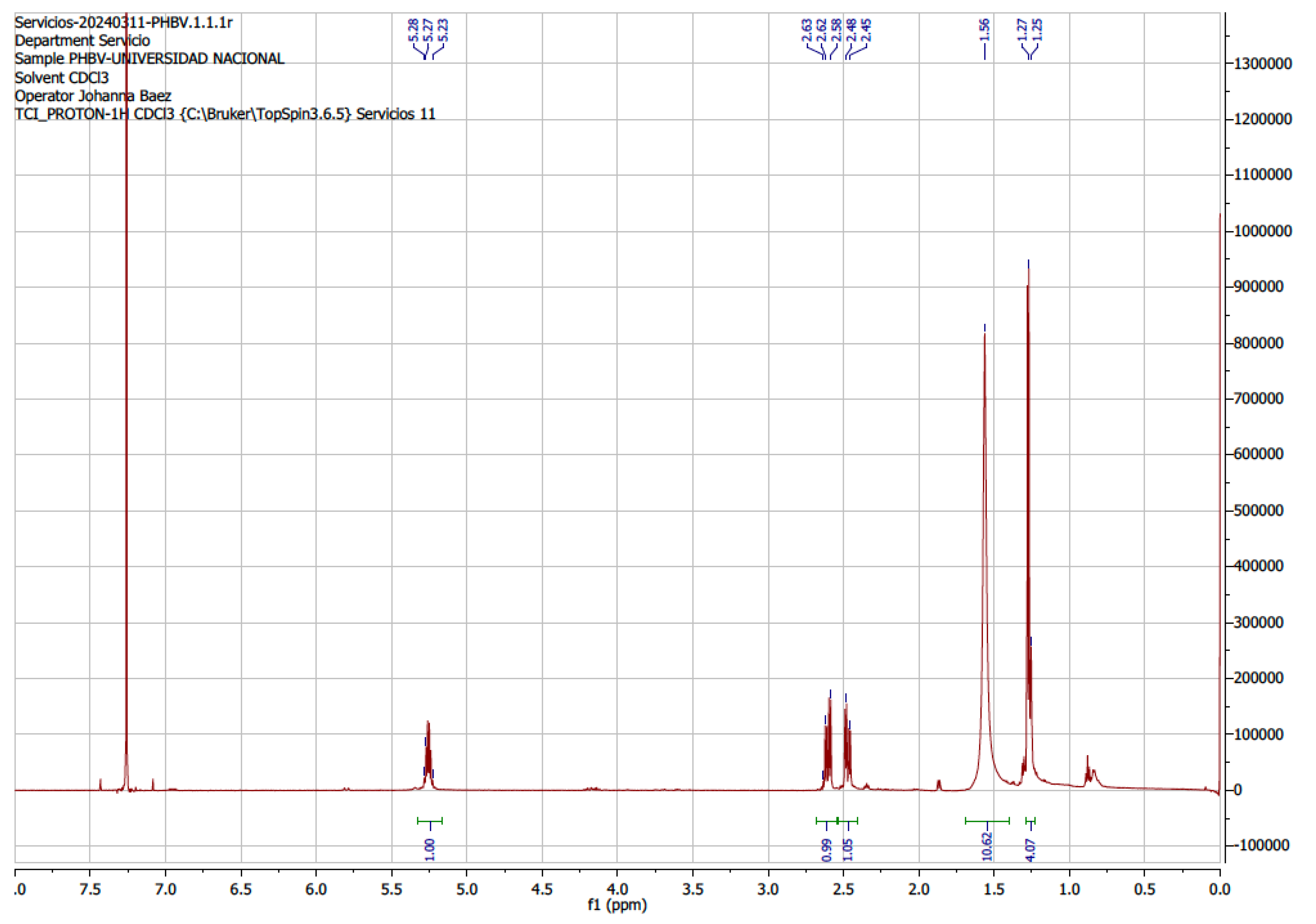
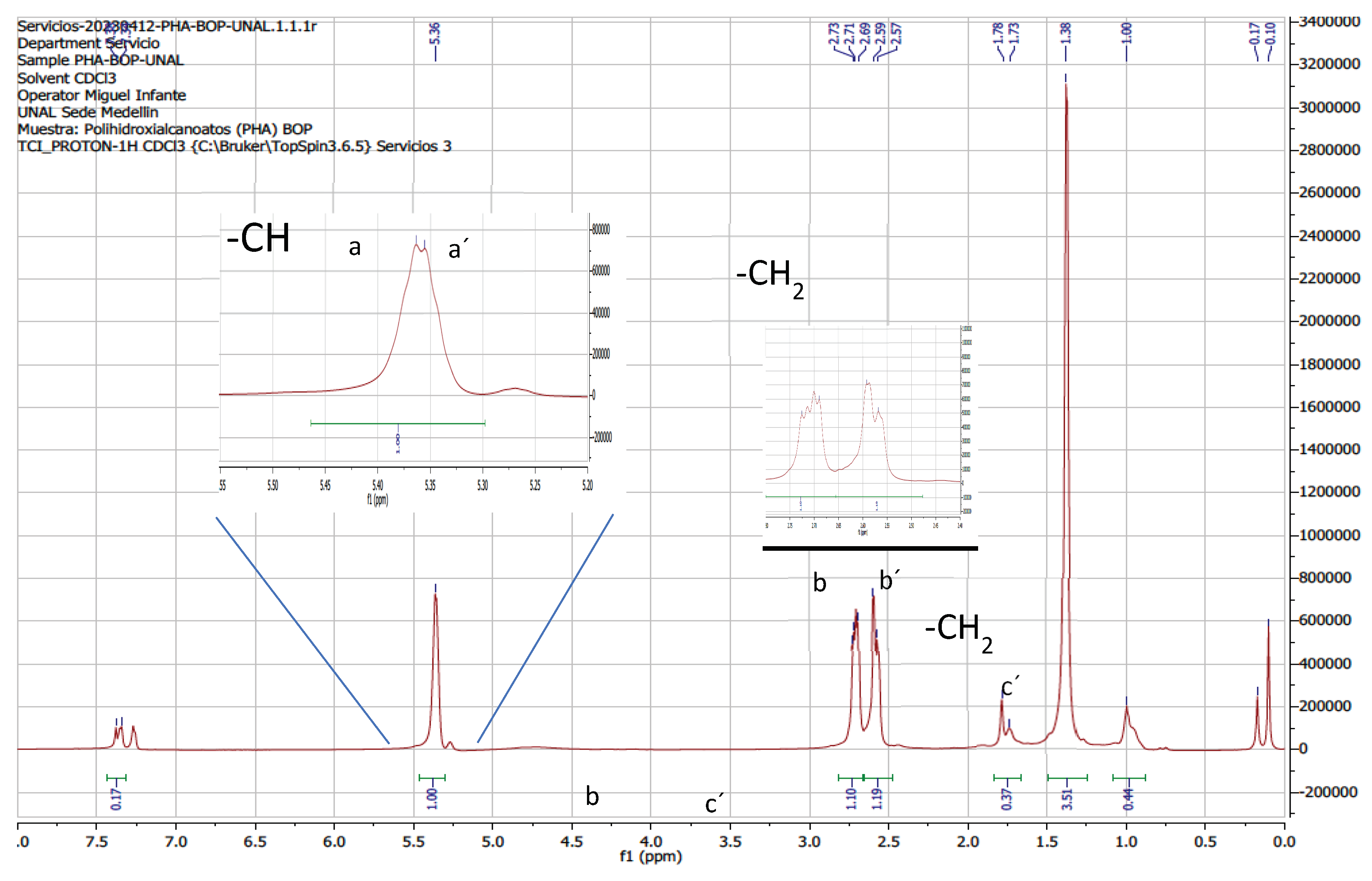
B. megaterium LVN01 under batch-type aerobic fermentation with carbon source biogas digestate. The 1H-NMR (
Figure 7) and 13C-NMR (
Figure 8) spectra of the purified PHA sample and the pure standard were similar, showing seven typical signals of the Poly(3-hydroxybutyrate-co-3 hydroxyvalerate) copolymer (P (3-HB-co-3-HV) between 0.75 ppm and 5.50 ppm, which are associated with the chemical shifts (δ) of the 3HB (3-hydroxybutyrate) and 3HV (3-hydroxyvalrate) subunits of the copolymer PHBV.
The H-NMR signals detect the presence of characteristic functional groups of both monomeric fractions (3HB and 3HV), i.e., δ= 5.356 – 5.363 ppm (a and a’), concerning the protons of the methine group (-CH) and δ= 2.57 – 2.59 ppm and δ= 2.69 – 2.73 ppm (b and b’), corresponding to the protons of the methylene group (CH2). Particularly, the 3HB subunit exhibits a δ=1.25 – 1.26 ppm, belonging to the methyl group (CH3) (c). On the other hand, 3HV gives rise to shifts at δ=1.73 – 1.78 ppm (d) and δ=1.0 ppm (e), which allow us to infer the resonance of the methylene (d) and methyl protons (e), side chain (ethyl). These last displacements mark the difference between the monomers (HV and HB) and confirm the presence of PHBV. The findings of this research coincide with those reported by other authors for the PHBV copolymer [
32,
59,
60,
61,
62,
63,
64].
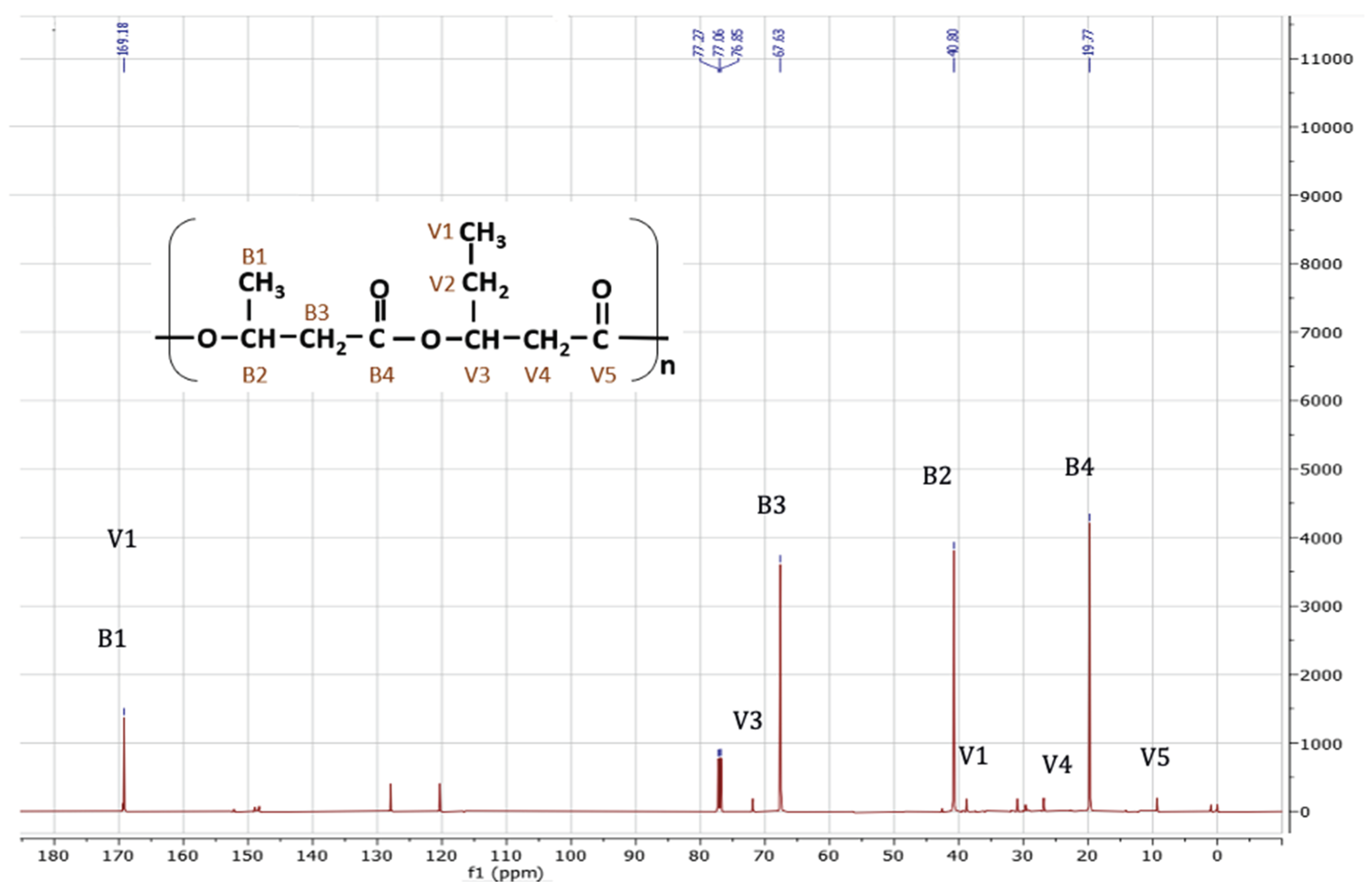
The results of 13C-NMR analysis are consistent with those of 1H NMR (
Figure 7). The 13C-NMR spectrum presents signals identifying the non-equivalent carbon atoms in each monomer. For HB, carbon shifts were observed at 19.65 ppm, 40.68 ppm, and 67.52 ppm, corresponding to the methyl (–CH 3), methylene (–CH 2 –), and ester (– O – CH–) groups, respectively. Particularly for HV, the signals of the two methylene groups (–CH 2 –) were observed at 26.74 ppm and 30.82 ppm. In this monomer, the peak corresponding to the ester group (–O–CH–) shifted to 71.90 ppm. Finally, a resonance 169.14 ppm of the carbon atoms of the carbonyl group (–C–) is presented for both monomers (3-HB and 3-HV). These data coincide with those reported by other researchers [
31,
32,
47,
63] and allow us to conclude the presence of the PHB-co-PHV copolymer in the polymeric extract of
B. megaterium.
The chemical characterization of the
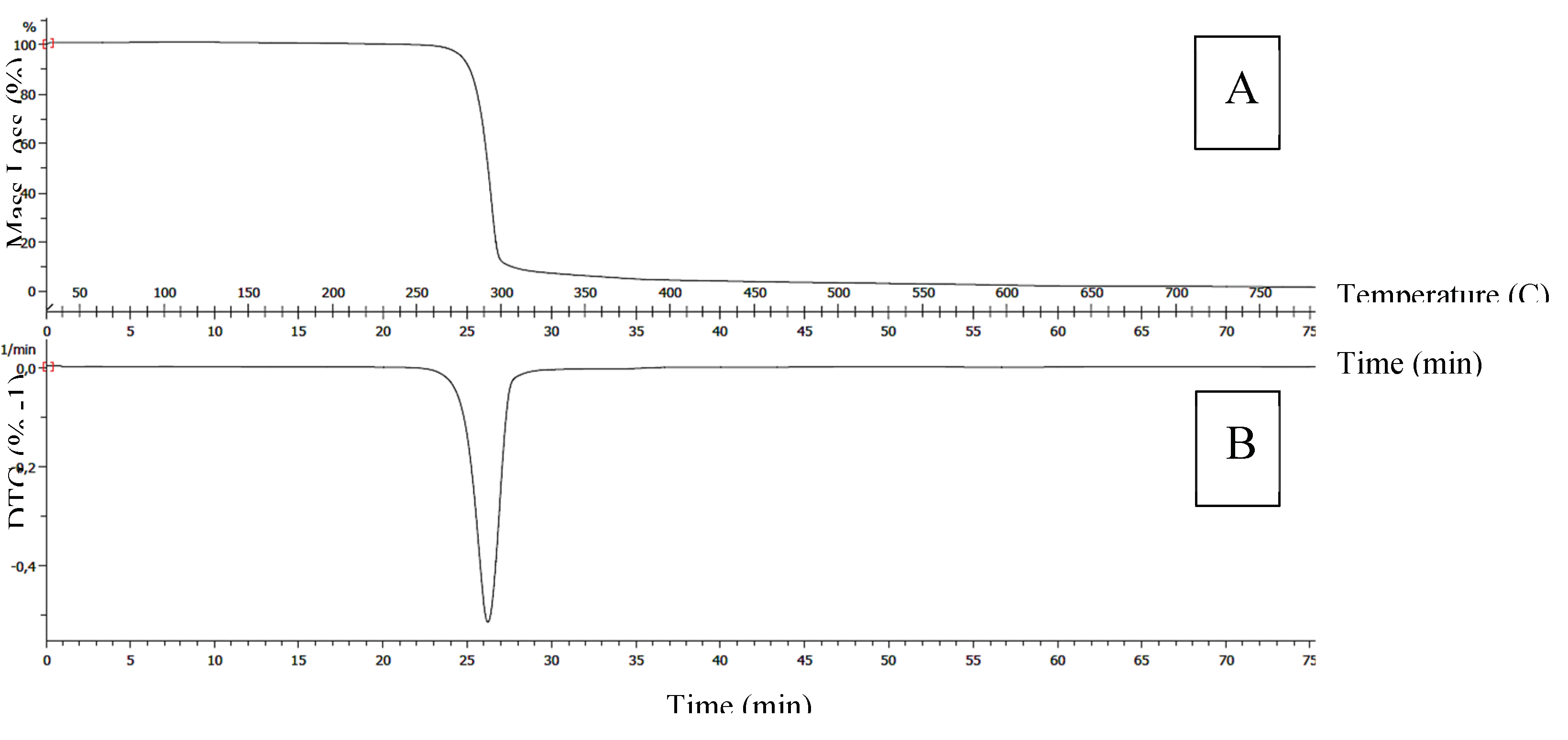
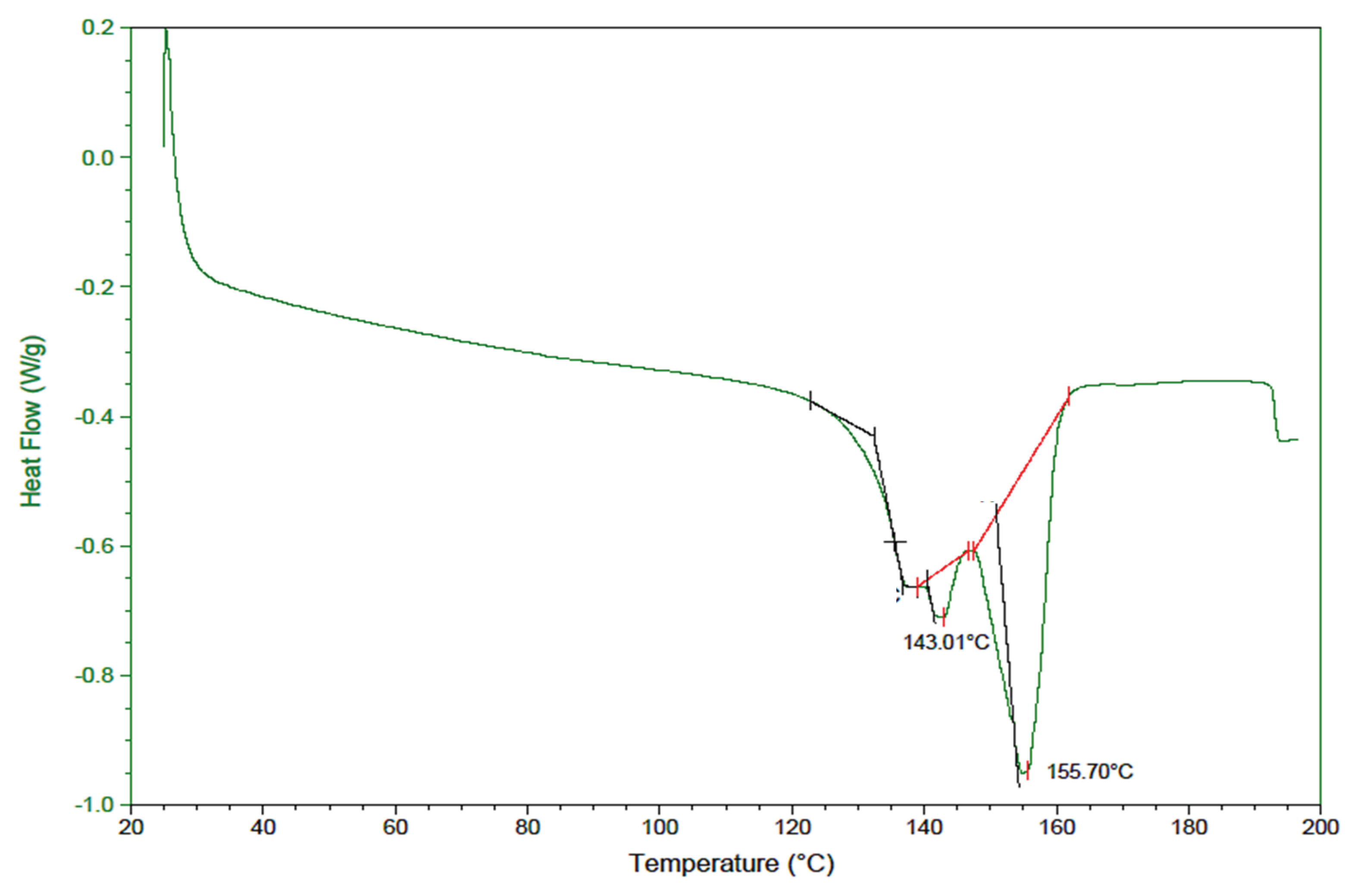
B. megaterium biopolymer is consistent with the characteristics derived from the analysis of the thermal properties of the biopolymer, using TGA (
Figure 9) and DSC (
Figure 10). The values of the thermal properties (Tm= 153 ºC and Td= 290.14ºC) of PHBV produced by
B. megaterium from biogas digestate are within the range of the values measured by Yuanpeng Wang et al., who analyzed three PHA synthesized by
Ralstonia eutropha from Levulinic Acid. The researchers found melting temperatures (Tm) of 101.93ºC, 150.18ºC, and 172.05ºC, and thermal decomposition temperatures (Td) of 284.4ºC, 298.3ºC and 263.4ºC, for PHB with 0%, 0.16% and 53% HV, respectively [
65]. However, the percentage of crystallinity (% Xc) = 18 of the PHBV under study differed markedly from those reported for PHB with 0%. (% Xc= 61.44%), 0.16% (% Xc= 50.34%) and HV 53% (% Xc= 51.92%). Nonetheless, regarding this parameter, the results of this research are also comparable with those of Abbasi et. al., who, for a series of PHBV produced by fermentation, reported Xc by DSC values between 16.6% and 29% [
32]. In their research, the PHBV with the lowest 3HV content (0.16) recorded the highest Xc (29%). From their study (Abbasi et al., 2022) and other researchers [
65], it is concluded that the degree of crystallinity and the melting point of the biopolymer decrease with the HV content in the polymer, potentially resulting in an improvement in the ductility and flexibility of the polymer.
The biopolymer produced in this research presents thermodynamic properties that differentiate it from the PHB structure and bring it closer to the PHBV line. In this context, it is relevant to note that, according to the literature, when a copolymer such as poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] is produced, it has a lower melting point and greater flexibility compared to the PHB homopolymer [
66]. The lower crystallinity and lower melting point of PHBV from
B. megaterum suggest an appreciable percentage of HV with a more flexible structural order. This aligns with the characterization of short-chain biopolymers, which are known to have lower crystallinity and lower melting points, classifying them as elastomers and providing them with an elongation capacity at break greater than 100% [
67,
68].
In this study, B. megaterium LVN01 demonstrated its efficient ability to produce PHBV from biogas digestate, a substrate rich in volatile fatty acids, in just a 16 to 24-hour fermentation period. This is promising for the industrial production of biopolymers since PHBV not only has appropriate physical characteristics for use in different industrial fields but also has a high degree of biodegradability, which makes it very attractive in the pharmaceutical industry of biopolymers.
The physical characteristics of PHBV from B. megaterium are intrinsically related to the biodegradation properties and ease of decomposition of PHAs. These degradation processes are influenced by the specific chemical composition of the polymer and the surrounding environmental conditions. The degradation of objects made with PHA has been reported to vary widely, from a few months in soils with stable temperatures to a couple of years in marine environments. It should be noted that certain types of PHAs, such as poly(3-hydroxybutyrate-co-3-hydroxyvalerate), are notably affected in their crystallinity, which modulates their degradation rates [69].
The potential of the aerobic fermentation process with the B. megaterium system using biogas digestate is strengthened by the use of an economical substrate for the production of PHA-type biopolymers, specifically PHBV. The main advantages of using digestate lie in its availability and low cost, considering that it is a co-product of the biogas production process from residual biomass from the artisanal fishing industry in Tumaco, Colombia. In addition to the economic advantage, there are the positive environmental and social impacts. First, alternatives to petroleum-derived plastics, which generate significant pollution during their manufacture and consume a non-renewable resource. Second, fishing waste (fish viscera) is utilized to produce biogas, and then, from the residual effluent (digestate rich in volatile fatty acids) PHA-type biopolymers are produced. With this approach, pollution from fishing waste, often discarded into the sea, is avoided. All this is done in a biorefinery environment, contributing to increasing the added value of the artisanal fishing production chain in communities living in vulnerable conditions, as well as the circular bioeconomy of biogas production.
Regarding the sustainability of the substrate, it is important to mention that artisanal fishing in the Colombian Pacific generates an abundant amount of waste, representing a valuable source of renewable organic matter for the efficient production of PHBV through fermentation with B. megaterium. Thus, the cost of raw materials for large-scale PHBV production could be significantly reduced, with important effects in terms of economics and environmental sustainability.
Author Contributions
Conceptualization, A.L.M-M., M.Y-P., and K.A.C-C.; data curation, A.L.M-M., M.Y-P., and K.A.C-C.; lab, A.L.M-M., M.Y-P., and K.A.C-C.; methodology, A.L.M-M., M.Y-P., and K.A.C-C.; project administration, A.L.M-M., M.Y-P., and K.A.C-C.; resources, A.L.M-M., M.Y-P., and K.A.C-C.; supervision, A.L.M-M., M.Y-P., and K.A.C-C.; validation, A.L.M-M., M.Y-P., and K.A.C-C.; visualization, A.L.M-M., M.Y-P., and K.A.C-C.; writing—original draft, A.L.M-M., M.Y-P., and K.A.C-C.; writing—review and editing, A.L.M-M., M.Y-P., and K.A.C-C.; All authors have read and agreed to the published version of the manuscript.
Figure 1.
Growth curve of B. megaterium LVN01 in Biogas digestate (circles); negative control: medium with digestate (diamonds); operational conditions: 3L, 400 rpm, 30.8 °C, pH 7.
Figure 1.
Growth curve of B. megaterium LVN01 in Biogas digestate (circles); negative control: medium with digestate (diamonds); operational conditions: 3L, 400 rpm, 30.8 °C, pH 7.
Figure 2.
PHB accumulated by B. megaterium LVN01 during fermentation with biogas digestate. (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0)
Figure 2.
PHB accumulated by B. megaterium LVN01 during fermentation with biogas digestate. (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0)
Figure 3.
Cell growth in terms of DCW of B. megaterium LVN-1 cultivated in biogas digestate vs PHA production (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0).
Figure 3.
Cell growth in terms of DCW of B. megaterium LVN-1 cultivated in biogas digestate vs PHA production (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0).
Figure 4.
Dry weight (DW) of B. megaterium LVN01 in digestate from the Biogas plant, San Andrés de Tumaco (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0).
Figure 4.
Dry weight (DW) of B. megaterium LVN01 in digestate from the Biogas plant, San Andrés de Tumaco (V: 3 L, t: 60 h, 400 rpm, T: 30.8°C, pH 7.0).
Figure 5.
Gas mass chromatography. A. Chromatogram of the commercial polymer Poly (3-hydroxybutyric acid-co-3-hydroxyvaleric acid) (Part N° 403121, Sigma–Aldrich, Lot: MKBL7384V). B. Chromatogram of the copolymer biosynthesized by B. megaterium LVN01 from biogas digestate.
Figure 5.
Gas mass chromatography. A. Chromatogram of the commercial polymer Poly (3-hydroxybutyric acid-co-3-hydroxyvaleric acid) (Part N° 403121, Sigma–Aldrich, Lot: MKBL7384V). B. Chromatogram of the copolymer biosynthesized by B. megaterium LVN01 from biogas digestate.
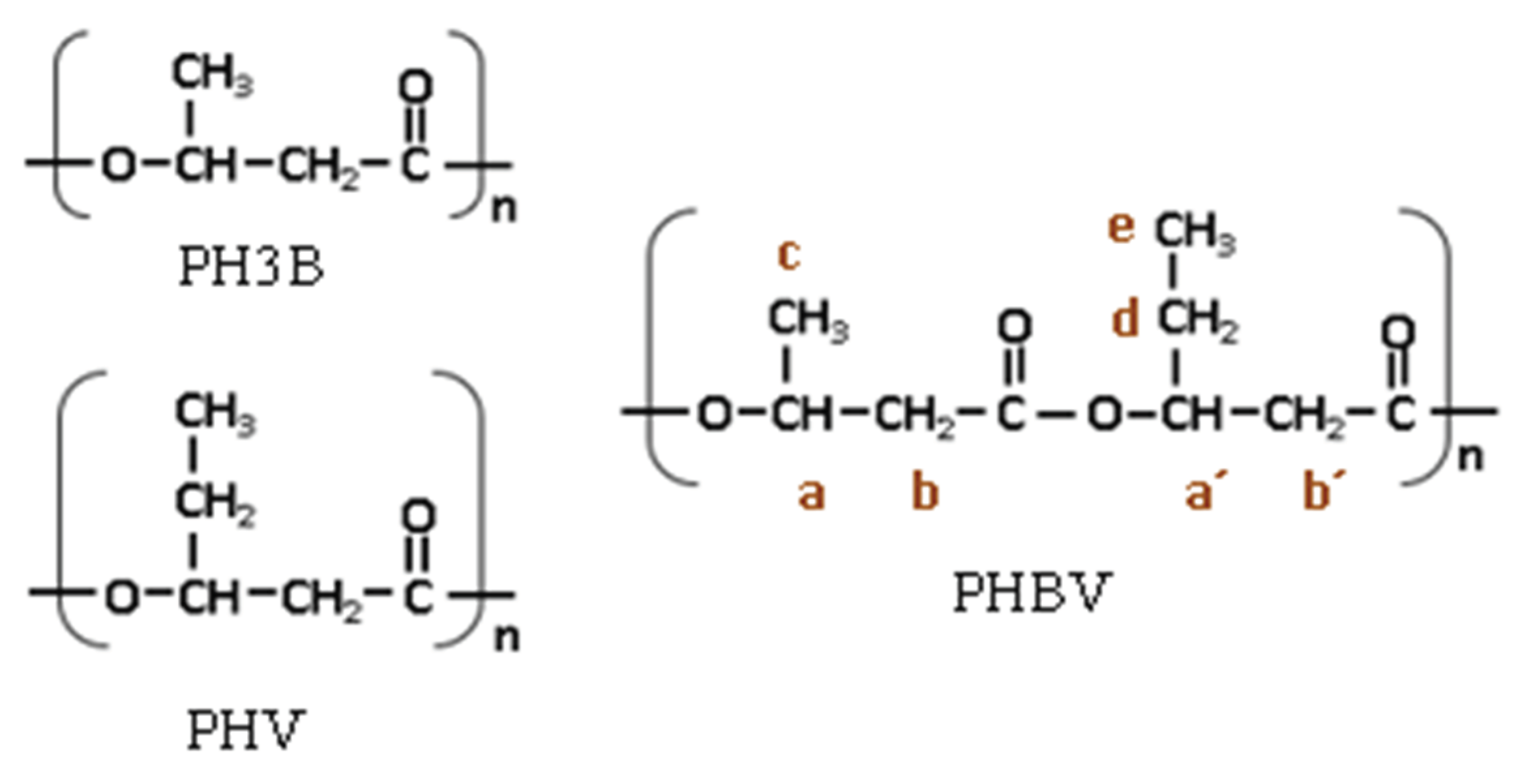
Figure 6.
Chemical structure of PHBV copolymer.
Figure 6.
Chemical structure of PHBV copolymer.
Figure 7.
1H-NMR spectrum of PHVB synthesized by B. megaterium from biogas digestate (batch fermentation), showing the chemical shift of the groups characteristic functional. A. Commercial standard (Figures S7A1, S7A2, S7A3). B. Synthesized by Bacillus megaterium (Figures S7B1, S7B2).
Figure 7.
1H-NMR spectrum of PHVB synthesized by B. megaterium from biogas digestate (batch fermentation), showing the chemical shift of the groups characteristic functional. A. Commercial standard (Figures S7A1, S7A2, S7A3). B. Synthesized by Bacillus megaterium (Figures S7B1, S7B2).
Figure 8.
13C-NMR spectrum, corresponding of the PHBV sample synthesized by B. megaterium LVN01 in liquid digestate.
Figure 8.
13C-NMR spectrum, corresponding of the PHBV sample synthesized by B. megaterium LVN01 in liquid digestate.
Figure 9.
Curves of the Thermogram-TG (A) and its Derivative-DTG (B) of the PHBV sample synthesized by B. megaterium LVN01 from biogas digestate.
Figure 9.
Curves of the Thermogram-TG (A) and its Derivative-DTG (B) of the PHBV sample synthesized by B. megaterium LVN01 from biogas digestate.
Figure 10.
DSC thermogram of the PHBV sample synthesized by B. megaterium LVN01 from biogas digestate.
Figure 10.
DSC thermogram of the PHBV sample synthesized by B. megaterium LVN01 from biogas digestate.
Table 1.
Characterization of protons 1H-NMR.
Table 1.
Characterization of protons 1H-NMR.
| Type of proton |
Chemical shift δ (ppm) |
| a and a’ |
Methin (- CH-) in PHB and PHV |
5.356 and 5.363 |
| b and b’ |
Methylene (-CH2) in PHB and PHV |
2.59-2.57 and 2,69 – 2,73 |
| c |
Methylene (-CH2) in PHV |
1.73-1.78 |
| d |
Methyl (-CH3) in PHB |
1.38 |
| e |
Methyl (-CH3) in PHV |
1.00 |