1. Introduction
Platinum compounds (PC) are drugs used to treat a wide variety of colo-rectal, gynecological and pulmonary tumors. The incidence of drug hypersensitivity reactions (DHRs) with these drugs is directly related to the number of drug exposures (up to 40% with carboplatin after seven treatment cycles and 15% with oxaliplatin). The most frequent phenotype of DHRs to PC is Type 1 (IgE mediated). Cytokine release reactions, mixed reactions, and, in isolated cases, type IV reactions have also been described [
1,
2].
PC skin tests (STs) are available for diagnosis and risk assessment for severe reactions. The sensitivity of STs varies between studies (26-100% for oxaliplatin and around 80% for carboplatin) and they have a high specificity (91-100%, for both) [
2,
3,
4].
The gold standard of diagnosis for PC hypersensitivity is the drug provocation test (DPT). Few groups of authors have performed DPT to rule out or confirm hypersensitivity to PC. The largest published study of DPT with PC showed that 46% (43 of 93) of all DPT were negative and the number of unnecessary desensitizations was reduced [
3]. Nevertheless, DPT is a high-risk technique that requires close medical surveillance by personnel with expertise in drug allergy in adequate safety settings [
2,
3].
The basophil activation test by flow cytometry (BAT-FC) measures the expression of activation markers on the basophil surface [
5]. BAT-FC has been suggested as a promising predictor of severe DHR in platinum-treated patients and the risk of reactions during desensitization [
6,
7,
8]. However, the number of patients in these studies is small, and this tool has not yet been validated. On the other hand, specific IgE (sIgE) combined with STs seems to be a good predictor of platinum allergy, although it seems to have a reasonable specificity (75-100%) but low sensitivity (34-75%) [
3,
9].
Microarray technology allowed the simultaneous assessment of sIgE levels of hundreds of allergens, enabling the development of component-resolved diagnostics in an affordable way [
10,
11]. Humanized rat basophilic leukemia cell lines have been proposed as a suitable replacement for peripheral human basophils for detecting allergen- sIgE reactivity [
12,
13]. The combination of humanized rat basophil cell lines and microarray technology combines high-throughput screening and cellular activation rather than only sIgE binding [
14,
15]. BAT-FC has some limitations, including the need to process fresh samples, a small percentage of patients with non-responsive basophils, and it requires a larger amount of blood than other techniques, especially when patients are treated with cytostatics that reduce leukocyte counts, such as in oncological treatments.
The aim of this study is to explore the usefulness of in vitro techniques (BAT-FC, BAT-microarray, and sIgE-microarray) in a group of patients presenting hypersensitivity reactions to PC. We compared these assay results with the clinical diagnosis (STs and, if possible, DPT).
2. Results
2.1. Characteristics of the Patients
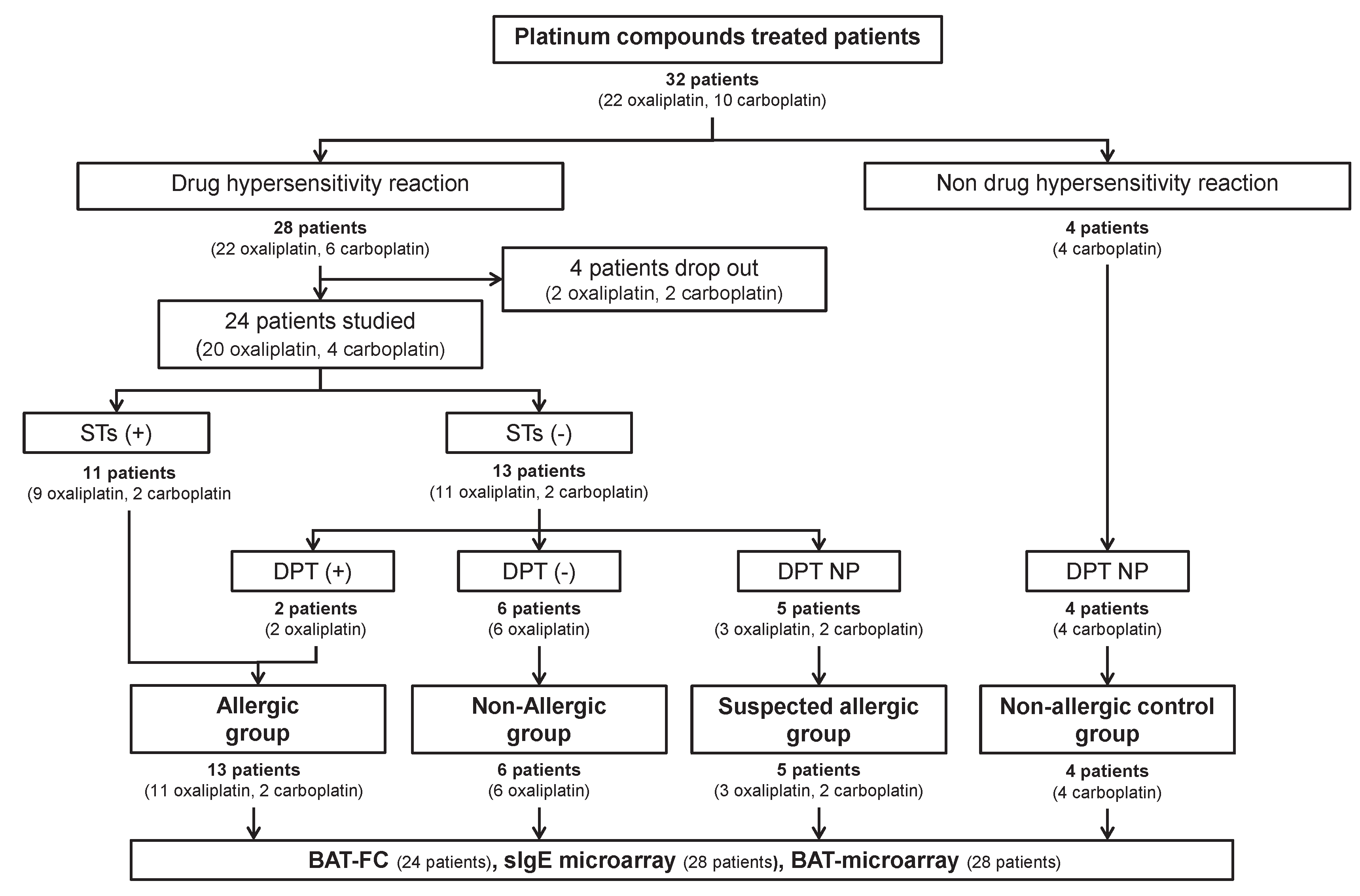
Twenty-eight patients (22 oxaliplatin and 6 carboplatin) gave their consent to participate in the study. Only 24 patients (20 oxaliplatin and 4 carboplatin) were finally evaluated as the remaining patients withdrew from the study (
Figure 1). We also included four patients treated with carboplatin who had not experienced a DHR (as non-allergic control). The baseline characteristics of the patients are presented in
Table 1. The severity of the initial reaction was moderate or severe (Brown's grade 2-3/ RCUH classification 2-4) in 62% (15/24) and 38% (9/24) of patients suffered a mild initial reaction (Brown's grade 1/ RCUH classification 1).
2.2. Allergological Work-Up Outcomes
The results of the allergological study are shown in
Table 2. Of the 24 patients, 11 had a positive STs (45.8%), and 13 were negative (54.2%) (
Figure 1). The negative patients underwent DPT; two had a positive DPT (25%), and 6 were negative (75%). DPT was not possible in five patients; one had a severe reaction (patient 5), and four patients refused to undergo a DPT (patients 13, 14, 22, and 24); all were classified as suspected allergic (
Figure 1). Of the two patients with a positive DPT, patient 3 suffered a mild reaction (local urticarial and palmar pruritus and needed intravenous antihistamines and corticosteroids). The other patient (#4) presented a moderate reaction (severe back pain, headache, and hypertension controlled by antihistamines, corticosteroids, and paracetamol administered intravenously). All of them were subjected to our "restart protocol" for positive patients and showed good tolerance to oxaliplatin.
2.3. Basophil Activation Test by Flow Cytometry (BAT-FC)
BAT-FC was performed on 23 of the 24 study patients and 2 of the 4 control samples. Only two patients in the allergic group had a positive BAT-FC. Patient 6, had CD63+ basophil percentages of 24.8%, 26.5%, and 35.4% at oxaliplatin concentrations of 50 µg/ml, 125 µg/ml, and 250 µg/ml, respectively (
Figure 2). In this case, the SI of CD203c was >2 for the two highest drug concentrations (125 µg/ml and 250 µg/ml). In addition, patient 21 showed an increase in CD63+ expression at carboplatin concentrations of 125 µg/ml (CD63+: 8.2%) and 250 µg/ml (CD63+: 9.2%); the SI of CD203c remained negative at all concentrations tested in this patient.
The remaining allergic patients had a negative BAT-FC. However, two were excluded from the analysis, one because of a higher basal activation of basophils and the other because of non-stimulation of the positive control. CD203c could not be stained in 4 patients. No positive BAT-FC was found in the control or non-allergic groups. The overall sensitivity and specificity of BAT-FC, including both drugs, were 15.4% and 100%, respectively.
2.4. sIgE Microarrays Immunoassay
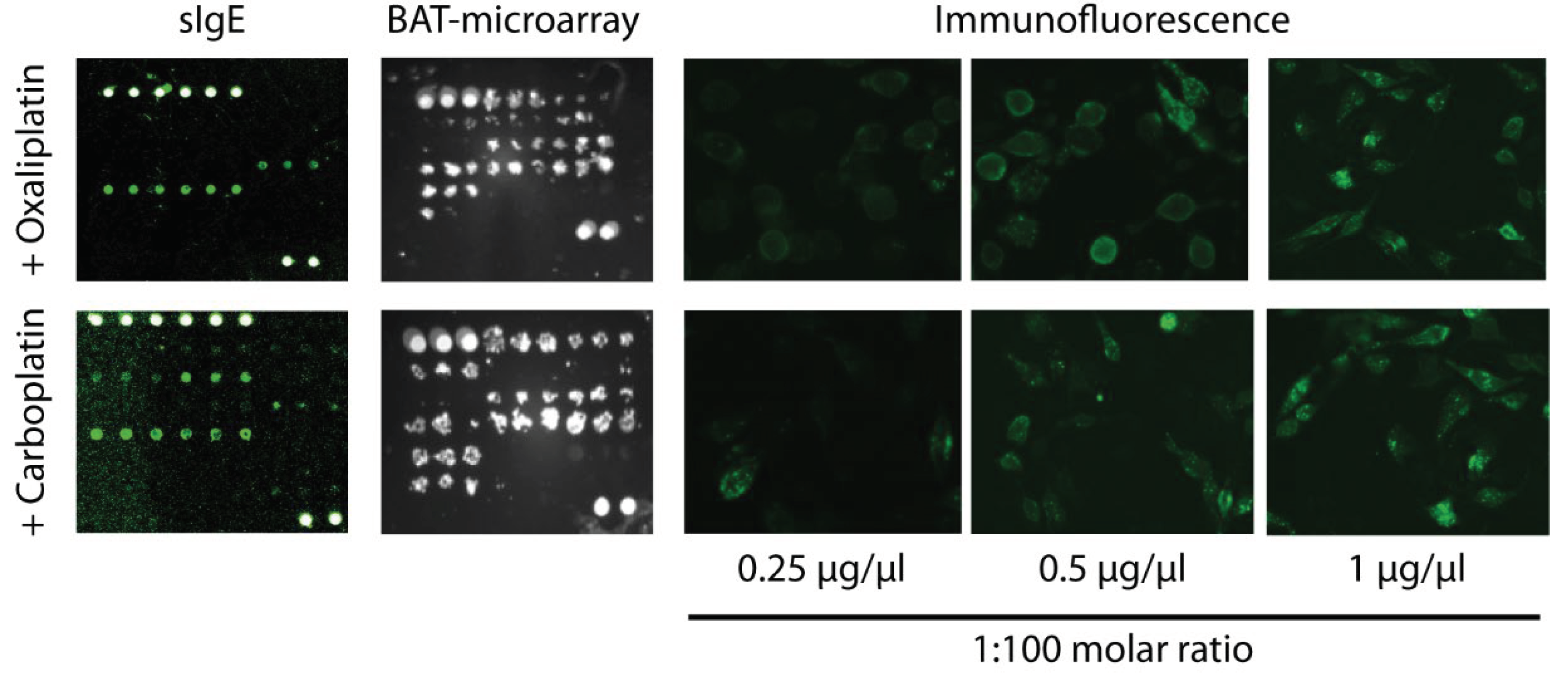
To study the oxaliplatin- and carboplatin- sIgE, we developed a microarray containing the drugs complexed with different amounts of albumin (
Figure 3). We tested three molar ratios of drug:albumin (1:10, 1:100, and 1:1000). The best-performing ratio for oxaliplatin was 1:100 (
Figure 4A and Supplementary Figures 1A and 2A). For carboplatin, the three-molar ratio had a good performance (
Figure 4B and Supplementary Figures 1B and 2B). In order to compare the data, we selected the higher values of the sIgE 1:100 molar ratio (Table II).
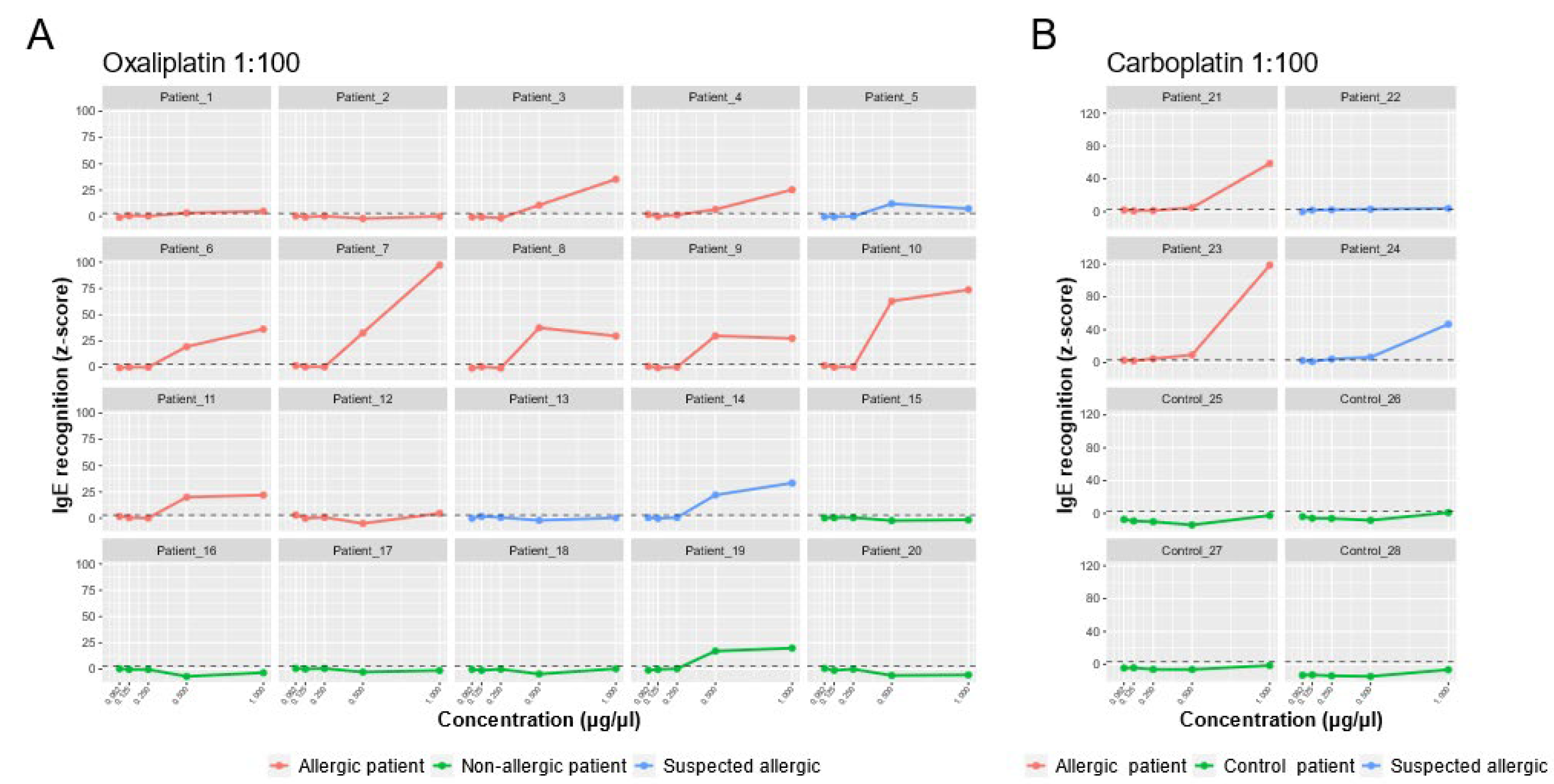
As shown in
Figure 4A, in the oxaliplatin-allergic group, 10 out of 11 patients (90.9%) had positive oxaliplatin sIgE (z-score > 3). In most cases, there was a dose effect, and the amount of IgE increased with increasing drug concentration. Of the patients with suspected oxaliplatin allergy, patient 13 was negative, patient 5 had moderate levels, and patient 14 had elevated IgE levels to the drug. All samples in the non-allergic group (except patient 19) were negative for sIgE.
Both patients with confirmed carboplatin allergy had elevated IgE levels (
Figure 4B). Of the two patients with suspected carboplatin allergy, 24 had high IgE levels, and 22 were slightly above the cut-off value (z-score > 3). The overall sensitivity and specificity of the sIgE-microarrays, including both drugs, were 92.3% and 90%, respectively.
2.5. Basophil Activation Test on Microarray Support (BAT-Microarray)
BAT-microarray was performed on all patients included in the study. A microarray containing the tested drugs complexed with different amounts of albumin (1:10, 1:100, and 1:1000 molar ratios) was incubated with RBL-30/25 cells previously sensitized with the patient's serum. Cell activation was assessed by surface expression of CD63 (
Figure 3). For both oxaliplatin and carboplatin, the best-performing molar ratio was 1:100 (
Figure 5 and Supplementary
Figure 3 and
Figure 3). As shown in
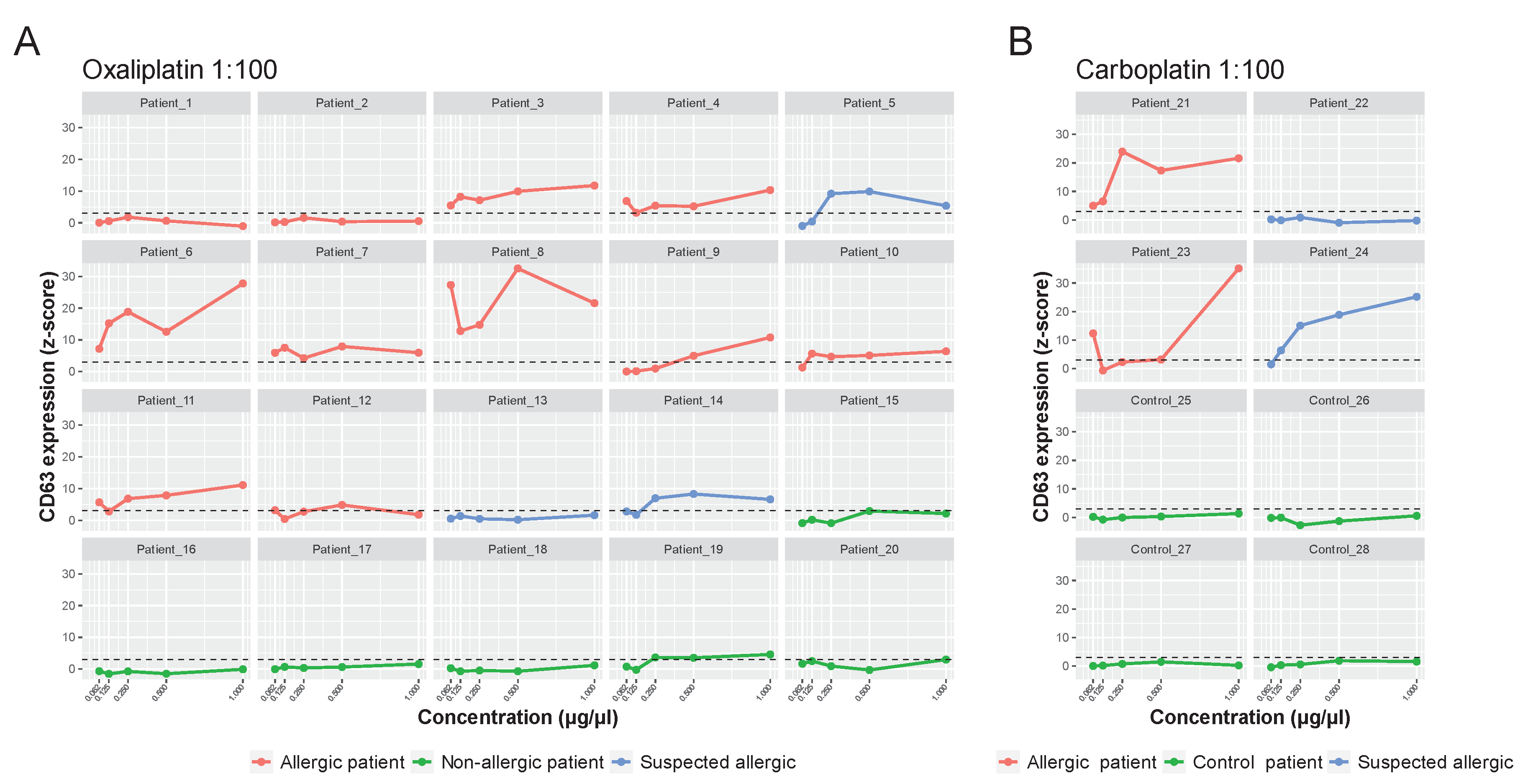
Figure 5A, in the oxaliplatin-allergic group, 9 out of 11 (81,8%) had a positive BAT-microarray at the 1:100 molar ratio. Of the patients with suspected oxaliplatin allergy, patients 5 and 14 had a positive BAT-microarray, and patient 13 was negative. All samples in the non-allergic group (except patient 19) had a negative BAT-microarray (Fig. 5A).
On the other hand, both patients with a confirmed allergy to carboplatin had a positive BAT-microarray in the 1:100 molar ratio (
Figure 5B). Of the two patients with suspected carboplatin allergy, 24 had a positive BAT-microarray, and 22 had a negative BAT-microarray. All samples in the control group had a negative BAT-microarray (
Figure 5B). The overall sensitivity and specificity of the BAT-microarray were 84.6% and 90%, respectively.
Figure 5: BAT-microarray immunoassay. The y-axis shows the average CD63 expression represented as average Z-scores. The x-axis shows the drug concentration: oxaliplatin (A) and carboplatin (B) 1:100 molar ratio. The standardized fluorescence intensity represented as the average Z-score was considered positive if it exceeded 3 (dotted line).
3. Discussion
DPT is considered the gold standard in the diagnosis of drug allergy, and its use in allergy clinical practice helps to establish or exclude hypersensitivity. However, DPT is a high-risk procedure, and the validation of other diagnostic tools, such as BAT-FC, is essential to reduce the risk of DPT. No studies have compared the results of BAT-FC with those of DPT in patients with DHR to platinum compounds. In the present study, we evaluated the utility of BAT-FC and, for the first time, the sIgE- and BAT-microarray as diagnostic tools for DHR to PC. Our results revealed that BAT-FC sensitivity was low at only 14.4%, compared to sIgE- and BAT-microarray, which were higher than 84%.
Interestingly, we found four patients with non-immediate reactions. In three of these patients, STs and DPT were negative (in patient 13, no challenge could be performed), and, as expected, IgE and BAT were negative. In the fourth patient, the sIgE- and the TAB-microarray were positive, which could indicate a mixed endotype.
In addition, one patient diagnosed with DHR did not exhibit a sIgE and yielded negative results on BAT-microarray, potentially indicating an IgE-independent mechanism such as Mas-related G protein–coupled receptor X2 (MRGPRX2) [
21], which could not be further investigated.
It is well known that BAT-FC sensitivity for diagnosing IgE-mediated hypersensitivity reactions differs according to the drug tested [
22]. Over the last years, the potential value of BAT-FC with different basophil activation markers (CD203c and CD63) in PC allergy has been investigated [
6,
7,
8]. Although BAT-FC is a good ex vivo technique in allergy assessment, it still lacks standardization. Each center employs different allergen preparations, dilutions, incubation times, and flow cytometry essential parameters, such as the data analysis, gating strategies, and positivity thresholds established in different laboratories, that could influence the BAT-FC results [
23,
24]. In this regard, in the present study, the positivity threshold has been chosen based on previous experience with other non-chemotherapeutic drugs [
25,
26]. However, compared with other studies, our cut-off would be more restrictive than those published by other groups [
6,
7,
8].
BAT-FC is a minimally invasive, safer, and less expensive tool than DPT in diagnosing DHR to platinum compounds. However, BAT-FC has some limitations, such as the completion time, as the sample must be investigated within 24 hours [
24], the test requires qualified personnel and needs validation in cohorts of adequate statistical power and in patients well-characterized (diagnosed as positive or negative after a protocol including STs and DPT). The time between the reaction and the test is an important aspect. In cancer patients, the interval to perform this diagnostic procedure is usually two weeks, a short interval that can interfere with BAT-FC results (basophils hyporeactivity response or anergy) [
27].
Due to the low frequency of the human peripheral blood basophils (less than 1%) [
14] and the difficulty of their purification [
28], several rat basophilic leukemia (RBL) cell lines transfected with human FcεRI have been developed to be used as a diagnostic tool for detection of allergen-sIgE and as surrogate for patient basophils [
12,
29]. Among them, RBL-30/25 and RBL SX-38 cell lines presented an optimal IgE-mediated degranulation [
19,
29]. In the present work, we have improved the protocol of BAT-microarray by using 96-well polystyrene plates instead of glass slides that allow a good deposition of allergens and provide an appropriate environment for cell cultures. Our results show that BAT-microarray, with a sensitivity of 84.6% and a specificity of 90%, has a better diagnostic performance than BAT-FC, with a sensitivity of 15.4% and a specificity of 100%.
The main limitation of RBL cells expressing the high-affinity IgE receptor is that, being rat cells, it is not known whether they can respond to the other molecules involved in the allergic response present in the patient's serum (cytokines, IgG4, IgG4 receptor, etc.) and thus resemble the activation of the patient's basophils or merely sensitization related to the presence of IgE. In the present work we found some differences between sIgE- and BAT-microarray. For instance, the serum of patients 7 and 10 have high levels of IgE but a limited ability to induce basophil activation. On the other hand, patient 6, 8, and 21 presented a stronger basophil activation than would be expected based on sIgE levels. This suggests that the two assays are not identical and could provide complementary information, as is the case with sIgE and basophil activation with the patient's cells. However, this aspect needs further study.
Oxaliplatin- and carboplatin- sIgE was also determined in all patients included in this study by microarray immunoassays. In previous studies, the determination of sIgE to platinum compound has also been investigated as a diagnostic method using ThermoFisher InmunoCAP technique [
9]. Our group, in a previous study, showed that sensitivity for oxaliplatin- sIgE was 34% and specificity 90%. However, Caiado J et al. reported lower values of specificity of oxaliplatin- sIgE (75%) and higher sensitivity values (75%). This work also showed sensitivity (59%) and specificity (100%) for carboplatin-sIgE. Our results show that sIgE to oxaliplatin by microarray immunoassay correlates with the results of ST and DPT with a 90.9% sensitivity and 83.3% specificity. Similar results, albeit with fewer samples, were observed with carboplatin. Therefore, sIgE by microarray and BAT-microarray may be complementary tools for diagnosing hypersensitivity reactions to chemotherapeutic agents such as oxaliplatin and carboplatin.
Implementation of DPT in diagnostic protocols helps to validate diagnostics tools. For the first time, we have evaluated the role of sIgE- and BAT-microarray in the most extensive series of patients with hypersensitivity to oxaliplatin reported to date, including DPT in the diagnosis. Our results show an improvement in sIgE determination and BAT-microarray, which can help identify endophenotypes to hypersensitivity reactions to platinum compounds. These in vitro diagnostic tools can clinically guide in identifying candidates for DPT and can be considered a safe alternative in case of severe reactions or high-risk patients. Further, more extensive multicenter studies are needed to evaluate BAT-microarrays in diagnosing platinum compound DHR.
4. Materials and Methods
4.1. Patient Population and Study Design
We carried out a prospective, observational, longitudinal study with consecutive patients who suffered DHR to PC and were referred to the Allergy Division's Desensitization Program of Ramón y Cajal University Hospital (RCUH) between January 2019 and June 2022. The study was conducted in accordance with the Declaration of Helsinki, approved by the RCUH Ethics Committee (institutional register number: 268/18), and a written informed consent was obtained from all participants.
The diagnostic protocol was based on a detailed clinical history, STs, basal serum tryptase determination, risk assessment, and DPT, when the appropriate criteria were met [
3].
Initial DHRs were classified as immediate and non-immediate and according to severity as in previous publications [
3,
16,
17].
We considered high-risk patients those who met any of the following criteria: previous severe (life-threatening) reaction, comorbidities such as uncontrolled lung disease with forced expiratory volume < 1 L, severe systemic illness, and unavoidable use of beta-blockers.
STs were performed following safe handling of antineoplastic requirements. Skin prick test (SPT) concentration for oxaliplatin was 5 mg/ml and the intradermal test (IDT) concentrations were 0.5 mg/ml and 5 mg/ml. Carboplatin STs were performed at 10 mg/ml for SPT and 1 mg/ml, and 1 mg/ml for IDT. Positive and negative controls were carried out with saline and histamine, respectively. A positive reaction was defined according to the European Network for Drug Allergy guideline [
18]. Standard STs protocols use stepwise SPT followed by IDT if the former is negative.
DPT was carried out in the medical intensive care unit, following the same detailed recommendations described in other articles by our group [
3]. It was considered positive when it reproduced the original symptoms or showed an objective DHR. Serum tryptase levels were determined during the reaction. The final diagnosis of DHR has been defined as allergic based on positive STs or positive drug provocation tests to PC. Patients with negative DPT results were considered non-allergic. DPT was not performed in high-risk patients or patients who did not consent; these patients were considered suspected allergic. Patients were on multi-drug regimens and were assessed separately if more than one drug was suspected to avoid misdiagnosis.
4.2. Basophil Activation Test by Flow Cytometry (BAT-FC)
200 µL of whole blood were preincubated for 10 minutes at 37ºC with 40 µL of stimulation buffer, containing fetal bovine serum and human IL-3 (9 ng/mL final concentration, Pharmingen). Subsequently, the samples were incubated at 37ºC for 30 minutes with four concentrations of oxaliplatin or carboplatin (5, 50, 125, and 250 µg/ml). Anti-IgE antibody (BD Biosciences) and PBS were positive and negative controls. Samples were incubated on ice for 5 minutes to stop the reaction and stained in the dark for 30 minutes with 20 µL of the antibody cocktail: anti-CD63-FITC, anti-CD123-PE, and anti-HLA-DR-PerCP (FastImmune™; BD Biosciences). In some experiments, 5 µL of CD203c-BV421 (BD Biosciences) was also added. Red cells were lysed with FACS™ Lysing Solution. Samples were analyzed immediately on a FACS Canto II flow cytometer (BD Biosciences) until each tube volume was exhausted or at least 500 basophil events were obtained. An example of the gating strategy is shown in
Figure 2A.
CD63 activation was measured as the percentage of CD63+ cells after subtracting the percentage of basally activated basophils in the negative control. The CD203c stimulation index (SI) was calculated as the ratio of the mean fluorescence intensity (MFI) of CD203c between stimulated and unstimulated basophils. A BAT-FC was considered positive when CD203c SI was > 2 and/or the percentage of CD63+ basophil was >5%. The BAT-FC results were considered invalid when the proportion of CD63+ basophils in the positive control was <5% or when the basal activation of basophils was >2%.
4.3. Microarray Printing
Oxaliplatin and carboplatin conjugates were prepared similarly to that described by Caiado [
9]. Oxaliplatin and carboplatin were mixed with different amounts of human albumin to obtain molar ratios of 1:10, 1:100, and 1:1000. The solutions were incubated for 18 hours at room temperature with gentle shaking. The unbound drug was removed using Slide-A-Lyzer® MINI Dialysis Device columns 10K MWCO (Thermo Scientific). The solutions were diluted with PBS and Protein Printing Buffer (Arrayit Corporation) to obtain the following concentrations: 0.062 µg/µl, 0.125 µg/µl, 0.25 µg/µl, 0.5 µg/µl, and 1 µg/µl, except for the 1:1000 molar ratio where only the concentration of 0.4 µg/µl could be achieved. Biotinylated anti-rat IgG antibody (0.1 µg/µl) (112-065-062, Jackson Immunoresearch) was printed as a positive reference control. Anti-human IgE (2 µg/µl) (DIA HE1, BioPorto Diagnostics) was printed as a positive control. PBS, Angiotensin II octa peptide (1 µg/µl) (Sigma-Aldrich), poly-DL-alanine (1 µg/µl) (Sigma-Aldrich), and human serum albumin (HSA) (1 µg/µl, 0.2 µg/µl, and 0.04 µg/µl) (Grifols) were printed as a negative controls. The microarray printing was performed as previously described using a sciFLEXARRAYER S3 piezoelectric spotter and a PDC 70 Type 2 (Scienion). Each feature was printed in triplicates on glass slides for IgE detection assay (sciCHIP Epoxy slides; Scienion) or 96-well microtiter plates for the BAT-microarray (sciPLEXPLATE Type 2; Scienion). After printing, the arrays were dried at RT overnight and stored in a desiccator. Before use, printed surfaces were washed twice with PBS and blocked with Protein Microarray Activation Buffer (PMAB, Scienion) for 1 hour at RT (sIgE-microarrays) or 3 mg/ml HSA diluted in PBS (HSA/PBS) for 1.5 h at RT (BAT-microarray).
4.4. sIgE Microarrays Immunoassay
After blocking, the microarray slides were washed twice with Protein Microarray Wash Buffer (PMWB, Scienion). Patient serum was diluted 1:1 with Protein Microarray Reaction Buffer (PMRB, Scienion), incubated with the microarray for 2.5 hours at 37ºC with gentle agitation, and washed 3 times with PMWB. The microarrays were then incubated with a biotinylated anti-human IgE antibody (clone G7-26, BD Pharmingen) diluted 1:50 in PMRB for 2 hours at 37ºC and washed 3 times with PMWB. Finally, the microarrays were incubated with Cy3-streptavidin (PA43001; cytiva) diluted 1:500 in PMRB for 30 minutes at RT, washed 6 times with PMWB, and scanned using the Scan Array Express (Perkin Elmer).
4.5. Basophil Activation Test on Microarray Support (BAT-Microarray)
The BAT-microarray was performed using rat basophil leukemia cells (RBL-30/25) transfected with the human FcεRIα receptor, produced at the Paul-Ehrlich-Institut (Langen, Germany; kindly donated by Stefan Vieths and Lothar Vogel) [
19]. Cells were cultured in MEM (GIBCO)-10% fetal bovine serum (Thermo Scientific) at 37ºC in a 5% CO2 atmosphere.
RBL-30/25 cells were seeded (7 x 105/300 µl) in 12-well plate. After 8 hours, cells were sensitized with 5% heat-inactivated serum of the patients for 16 hours, transferred to a 96-well microtiter plate containing the microarray and incubated for 2.5 hours under slow agitation. Afterward, cells were washed 3 times with 1 mg/ml HSA/PBS (WB) for 10 minutes under slow agitation, fixed with 4% formaldehyde for 1 hour, and washed 3 times with WB. After that, we added the biotinylated anti-rat CD63 antibody (Clon REA444, Miltenyi Biotech) diluted 1:50 with WB for 30 minutes and washed 3 times with WB. Finally, the cells were incubated for 30 minutes at RT with Cy3-Streptavidin (PA43001; cytiva), diluted 1:500 with WB, washed 6 times with WB, and scanned using the sciREADER FL2 (Scienion).
4.6. Data Analysis.
Microarray analysis was performed according to the method of Lin et al [
20]. An individual spot was considered positive if the standardized intensity of fluorescence (represented as the weighted average Z-score) exceed 3. Data analysis and presentation were performed using Microsoft Excel and R.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Figure S1: sIgE microarrays immunoassay.; Figure S2: sIgE microarrays immunoassay; Figure S3: BAT-microarray immunoassay; Figure S4: BAT-microarray immunoassay.
Author Contributions
Conceptualization B.d.l.H.C., E.R., C.P.L., J.M.-B., and M.P.B.-G; sample collection and data acquisition, C.F.-L., C.G.R., A.B.-C., and M.P.B.-G; data analysis, C.F.-L., C.G.R., A.B.-C., B.d.l.H.C., E.R., C.P.L., J.M.-B., and M.P.B.-G.; writing—original draft preparation, C.F.-L., C.G.R., A.B.-C., B.d.l.H.C., E.R., C.P.L., J.M.-B., and M.P.B.-G.; writing—review and editing, B.d.l.H.C., E.R., J.M.-B., and M.P.B.-G.; visualization, C.F.-L., C.G.R., J.M.-B., and M.P.B.-G.; supervision, B.d.l.H.C., E.R., J.M.-B., and M.P.B.-G.; funding acquisition, B.d.l.H.C., E.R., J.M.-B., and M.P.B.-G.. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by Instituto de Salud Carlos III (ISCIII), co-funded by European Regional Development Fund (ERDF) ("A way to make Europe”) [grant numbers PI18/00962] and Fundación Merck Salud, Spain [Ayudas Merck de investigación 2019].
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of the Hospital Universitario Ramón y Cajal (protocol code 268/18).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
We would like to thank the nurses, nurse auxiliaries, and allergists for their significant efforts to carry out the anti-neoplasic agent desensitization Program in the allergy department at the RCUH. J.M.-B. is a at FIBio-HRC researcher supported by the Consejería de Sanidad (Comunidad Autónoma de Madrid).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Silver, J.; Garcia-Neuer, M.; Lynch, D. M.; Pasaoglu, G.; Sloane, D. E.; Castells, M., Endophenotyping Oxaliplatin Hypersensitivity: Personalizing Desensitization to the Atypical Platin. The journal of allergy and clinical immunology. In practice 2020, 8, (5), 1668-1680 e2. [CrossRef]
- Pagani, M.; Bavbek, S.; Alvarez-Cuesta, E.; Berna Dursun, A.; Bonadonna, P.; Castells, M.; Cernadas, J.; Chiriac, A.; Sahar, H.; Madrigal-Burgaleta, R.; Sanchez Sanchez, S., Hypersensitivity reactions to chemotherapy: an EAACI Position Paper. Allergy 2022, 77, (2), 388-403. [CrossRef]
- Madrigal-Burgaleta, R.; Bernal-Rubio, L.; Berges-Gimeno, M. P.; Carpio-Escalona, L. V.; Gehlhaar, P.; Alvarez-Cuesta, E., A Large Single-Hospital Experience Using Drug Provocation Testing and Rapid Drug Desensitization in Hypersensitivity to Antineoplastic and Biological Agents. The journal of allergy and clinical immunology. In practice 2019, 7, (2), 618-632. [CrossRef]
- Caiado, J.; Picard, M., Diagnostic tools for hypersensitivity to platinum drugs and taxanes: skin testing, specific IgE, and mast cell/basophil mediators. Current allergy and asthma reports 2014, 14, (8), 451. [CrossRef]
- Alpan, O.; Wasserman, R. L.; Kim, T.; Darter, A.; Shah, A.; Jones, D.; McNeil, D.; Li, H.; Ispas, L.; Rathkopf, M.; Perez, E.; Siri, D.; O'Connor, M.; Plassmeyer, M.; Romito, K.; Pettibone, C.; O'Reilly, S.; Sonder, S. U.; Marti, G., Towards an FDA-cleared basophil activation test. Front Allergy 2022, 3, 1009437. [CrossRef]
- Iwamoto, T.; Yuta, A.; Tabata, T.; Sugimoto, H.; Gabazza, E. C.; Hirai, H.; Kojima, S.; Okuda, M., Evaluation of basophil CD203c as a predictor of carboplatin-related hypersensitivity reaction in patients with gynecologic cancer. Biol Pharm Bull 2012, 35, (9), 1487-95. [CrossRef]
- Giavina-Bianchi, P.; Galvao, V. R.; Picard, M.; Caiado, J.; Castells, M. C., Basophil Activation Test is a Relevant Biomarker of the Outcome of Rapid Desensitization in Platinum Compounds-Allergy. The journal of allergy and clinical immunology. In practice 2017, 5, (3), 728-736. [CrossRef]
- Ornelas, C.; Caiado, J.; Campos Melo, A.; Pereira Barbosa, M.; Castells, M. C.; Pereira Dos Santos, M. C., The Contribution of the Basophil Activation Test to the Diagnosis of Hypersensitivity Reactions to Oxaliplatin. Int Arch Allergy Immunol 2018, 177, (3), 274-280. [CrossRef]
- Caiado, J.; Venemalm, L.; Pereira-Santos, M. C.; Costa, L.; Barbosa, M. P.; Castells, M., Carboplatin-, oxaliplatin-, and cisplatin-specific IgE: cross-reactivity and value in the diagnosis of carboplatin and oxaliplatin allergy. The journal of allergy and clinical immunology. In practice 2013, 1, (5), 494-500. [CrossRef]
- Huang, H. J.; Campana, R.; Akinfenwa, O.; Curin, M.; Sarzsinszky, E.; Karsonova, A.; Riabova, K.; Karaulov, A.; Niespodziana, K.; Elisyutina, O.; Fedenko, E.; Litovkina, A.; Smolnikov, E.; Khaitov, M.; Vrtala, S.; Schlederer, T.; Valenta, R., Microarray-Based Allergy Diagnosis: Quo Vadis? Frontiers in immunology 2020, 11, 594978. [CrossRef]
- Sanchez-Ruano, L.; de la Hoz, B.; Martinez-Botas, J., Clinical utility of microarray B-cell epitope mapping in food allergies: A systematic review. Pediatr Allergy Immunol 2020, 31, (2), 175-185. [CrossRef]
- Falcone, F. H.; Alcocer, M. J.; Okamoto-Uchida, Y.; Nakamura, R., Use of humanized rat basophilic leukemia reporter cell lines as a diagnostic tool for detection of allergen-specific IgE in allergic patients: time for a reappraisal? Current allergy and asthma reports 2015, 15, (11), 67. [CrossRef]
- Blanc, F.; Adel-Patient, K.; Drumare, M. F.; Paty, E.; Wal, J. M.; Bernard, H., Capacity of purified peanut allergens to induce degranulation in a functional in vitro assay: Ara h 2 and Ara h 6 are the most efficient elicitors. Clin Exp Allergy 2009, 39, (8), 1277-85. [CrossRef]
- Lin, J.; Renault, N.; Haas, H.; Schramm, G.; Vieths, S.; Vogel, L.; Falcone, F. H.; Alcocer, M. J., A novel tool for the detection of allergic sensitization combining protein microarrays with human basophils. Clin Exp Allergy 2007, 37, (12), 1854-62. [CrossRef]
- Wang, X.; Cato, P.; Lin, H. C.; Li, T.; Wan, D.; Alcocer, M. J.; Falcone, F. H., Optimisation and use of humanised RBL NF-AT-GFP and NF-AT-DsRed reporter cell lines suitable for high-throughput scale detection of allergic sensitisation in array format and identification of the ECM-integrin interaction as critical factor. Molecular biotechnology 2014, 56, (2), 136-46. [CrossRef]
- Demoly, P.; Adkinson, N. F.; Brockow, K.; Castells, M.; Chiriac, A. M.; Greenberger, P. A.; Khan, D. A.; Lang, D. M.; Park, H. S.; Pichler, W.; Sanchez-Borges, M.; Shiohara, T.; Thong, B. Y., International Consensus on drug allergy. Allergy 2014, 69, (4), 420-37. [CrossRef]
- Brown, S. G., Clinical features and severity grading of anaphylaxis. J Allergy Clin Immunol 2004, 114, (2), 371-6. [CrossRef]
- Brockow, K.; Romano, A.; Blanca, M.; Ring, J.; Pichler, W.; Demoly, P., General considerations for skin test procedures in the diagnosis of drug hypersensitivity. Allergy 2002, 57, (1), 45-51.
- Vogel, L.; Luttkopf, D.; Hatahet, L.; Haustein, D.; Vieths, S., Development of a functional in vitro assay as a novel tool for the standardization of allergen extracts in the human system. Allergy 2005, 60, (8), 1021-8. [CrossRef]
- Lin, J.; Bardina, L.; Shreffler, W. G.; Andreae, D. A.; Ge, Y.; Wang, J.; Bruni, F. M.; Fu, Z.; Han, Y.; Sampson, H. A., Development of a novel peptide microarray for large-scale epitope mapping of food allergens. J Allergy Clin Immunol 2009, 124, (2), 315-22, 322 e1-3. [CrossRef]
- Kolkhir, P.; Ali, H.; Babina, M.; Ebo, D.; Sabato, V.; Elst, J.; Frischbutter, S.; Pyatilova, P.; Maurer, M., MRGPRX2 in drug allergy: What we know and what we do not know. J Allergy Clin Immunol 2023, 151, (2), 410-412. [CrossRef]
- Hoffmann, H. J.; Santos, A. F.; Mayorga, C.; Nopp, A.; Eberlein, B.; Ferrer, M.; Rouzaire, P.; Ebo, D. G.; Sabato, V.; Sanz, M. L.; Pecaric-Petkovic, T.; Patil, S. U.; Hausmann, O. V.; Shreffler, W. G.; Korosec, P.; Knol, E. F., The clinical utility of basophil activation testing in diagnosis and monitoring of allergic disease. Allergy 2015, 70, (11), 1393-405. [CrossRef]
- Depince-Berger, A. E.; Sidi-Yahya, K.; Jeraiby, M.; Lambert, C., Basophil activation test: Implementation and standardization between systems and between instruments. Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017, 91, (3), 261-269. [CrossRef]
- Santos, A. F.; Alpan, O.; Hoffmann, H. J., Basophil activation test: Mechanisms and considerations for use in clinical trials and clinical practice. Allergy 2021, 76, (8), 2420-2432. [CrossRef]
- Berges Gimeno, M. P.; Bernal Rubio, L.; Roldan, E.; Rita, C. G., Positive Basophil Activation Test Result in a Patient With Anaphylaxis to Cotrimoxazole. Journal of investigational allergology & clinical immunology 2022, 32, (5), 410-412. [CrossRef]
- Phillips-Angles, E.; Gonzalez-Munoz, M.; Dominguez-Ortega, J.; Cabanas, R.; Quirce, S., Usefulness of basophil activation test in diagnosis of hypersensitivity to etoricoxib. The journal of allergy and clinical immunology. In practice 2016, 4, (2), 350-1. [CrossRef]
- Puan, K. J.; Andiappan, A. K.; Lee, B.; Kumar, D.; Lai, T. S.; Yeo, G.; Bercin, D.; Starke, M.; Haase, D.; Lum, J.; Chew, F. T.; Connolly, J.; Wong, S. C.; Zolezzi, F.; Poidinger, M.; Wang, Y.; Rotzschke, O., Systematic characterization of basophil anergy. Allergy 2017, 72, (3), 373-384. [CrossRef]
- Falcone, F. H.; Gibbs, B. F., Purification of Basophils from Peripheral Human Blood. Methods Mol Biol 2020, 2163, 35-48.
- Ladics, G. S.; van Bilsen, J. H.; Brouwer, H. M.; Vogel, L.; Vieths, S.; Knippels, L. M., Assessment of three human FcepsilonRI-transfected RBL cell-lines for identifying IgE induced degranulation utilizing peanut-allergic patient sera and peanut protein extract. Regulatory toxicology and pharmacology : RTP 2008, 51, (3), 288-94.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).