1. Introduction
The standard treatment protocol for patients facing advanced ovarian cancer typically involves debulking surgery and platinum-based chemotherapy, administered in varying sequences, followed by a phase of maintenance therapy. Despite the acknowledged significance of residual disease following cytoreduction as a crucial prognostic factor [
1,
2], the absence of a dependable indicator for tumor chemosensitivity poses a challenge in assessing the efficacy of first-line treatments [
3]. Compounding this challenge is the fact that nearly 75% of ovarian cancer patients are diagnosed with an advanced stage of the disease, often challenging to accurately evaluate using conventional imaging methods [
4]. Even with the application of the Response Evaluation Criteria in Solid Tumors (RECIST) [
5], limitations persist. Consequently, there is a pressing need for precise outcome predictors to inform treatment decisions in the first-line setting, a concern recognized by both ESGO and ESMO [
6].
The monitoring of CA-125 decline during chemotherapy has emerged as a valuable tool for predicting treatment response [
7], particularly in circumventing the limitations of imaging techniques. This has been extensively studied in ovarian cancer patients [
8,
9]. Ongoing research in ovarian cancer patients has explored parameters such as the CA-125 nadir level, half-life value, and time to nadir as potential indicators [
10]. Additionally, the Gynecologic Cancer InterGroup (GCIG) has defined CA125-based response as a 50% reduction in CA-125 levels sustained for at least 28 days [
5]. However, neither approach has conclusively proven to accurately predict chemosensitivity, and their predictive value has come under scrutiny [
11]. As the quest for more reliable indicators persists, refining these methods remains crucial for enhancing the precision of treatment strategies.
Recently, the ELIMination rate constant K (KELIM), a modeled kinetic parameter derived from CA-125 measurements over the initial 100 days of systemic therapy, whether adjuvant or neoadjuvant chemotherapy, has demonstrated considerable promise as a valuable predictive tool [
12]. This innovative approach involves a mathematical modeling method that does not rely on the absolute values of the biomarker but instead focuses on the longitudinal kinetics, specifically CA-125 elimination, throughout the course of treatment. Remarkably, this method is independent of renal function. Numerous studies have substantiated its credibility as a reliable and independent prognostic factor, particularly in the first-line setting for assessing chemosensitivity [
13,
14,
15,
16]. KELIM's association with radiological response during neoadjuvant chemotherapy, the likelihood of achieving complete cytoreduction during interval debulking surgery, the probability of platinum-resistant relapse, and its impact on patient progression-free survival (PFS) and overall survival (OS) further strengthens its clinical relevance. It is noteworthy that a higher KELIM corresponds to a more rapid CA-125 elimination, indicative of heightened chemosensitivity and a more favorable prognosis [
17]. This provides clinicians with a valuable tool for predicting treatment outcomes and tailoring therapeutic strategies based on the individual dynamics of CA-125 elimination during the critical early stages of systemic therapy.
On the other hand, the landscape of maintenance therapy for ovarian cancer has undergone recent transformations with the introduction of Poly (ADP-ribose) polymerase inhibitors (PARPi). However, the question of which patients stand to derive genuine benefits from PARPi maintenance therapy remains a subject of contention. The well-established correlation between PARPi efficacy and platinum-sensitivity in both the first-line and recurrent settings has added complexity to this discussion [
18,
19]. While PARPi activity is undeniably linked to BRCA mutations and homologous recombination deficiency (HRD) [
20,
21,
22,
23], the complete efficacy of these drugs remains not entirely explained by these two biomarkers, leaving substantial knowledge gaps. Additionally, HRD plays a crucial role in DNA double-strand break repair defects and serves as a highly predictive marker for primary platinum-sensitivity, as tumor cells become incapable of repairing double-strand breaks induced by platinum [
24,
25]. Consequently, exploring a potential relationship between PARPi efficacy and KELIM, a robust predictor of chemosensitivity, becomes an intriguing avenue of investigation.
This study's primary objective is to conduct a comparative analysis between KELIM and HRD as potential alternative tools in identifying patients who could potentially benefit from PARPi maintenance therapy. By delving into this comparison, the aim is to shed light on novel avenues for patient stratification and personalized treatment approaches in the realm of ovarian cancer maintenance therapy.
2. Materials and Methods
2.1. Study Characteristics
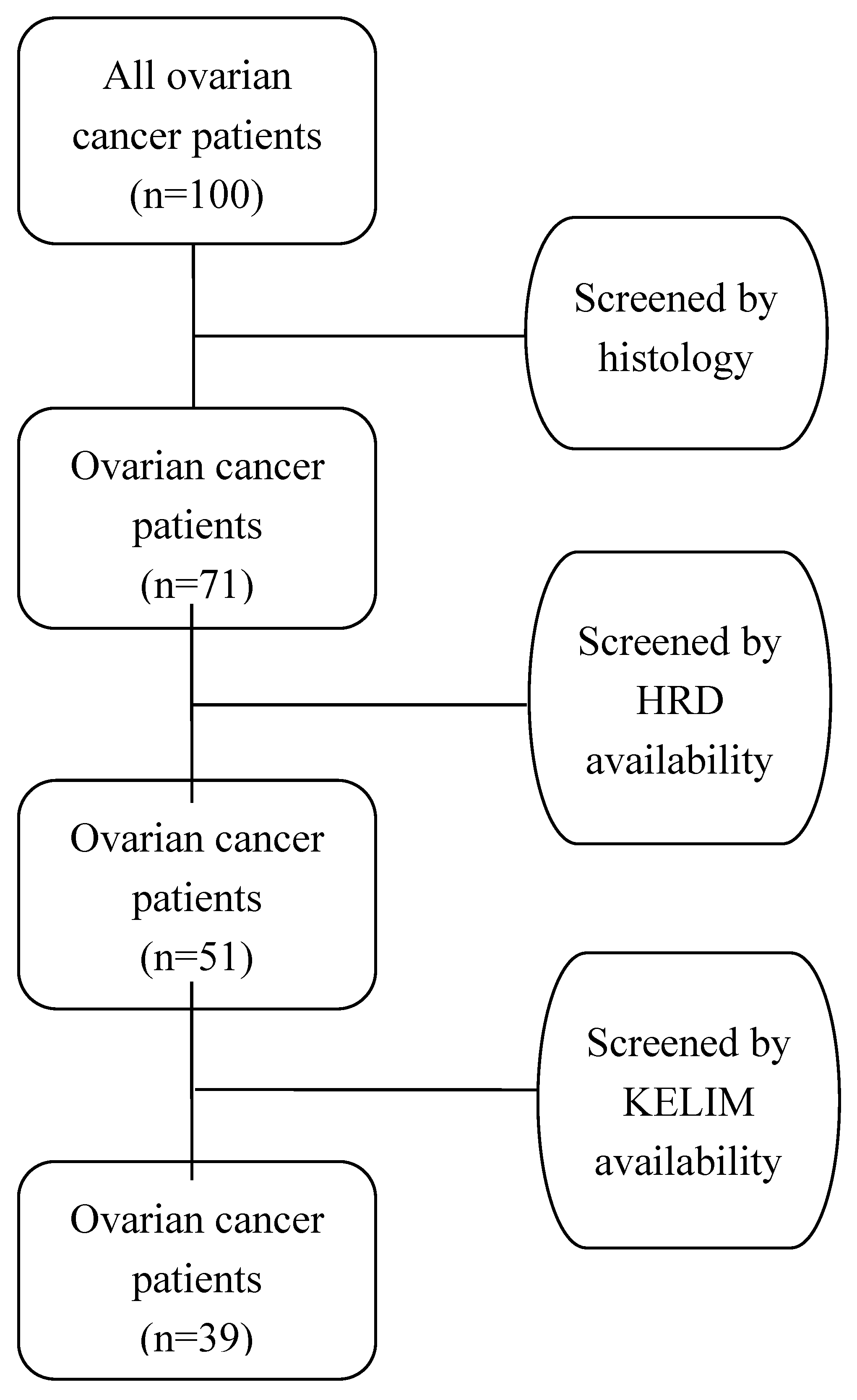
In conducting a comprehensive retrospective analysis, we meticulously examined the sequential medical records of all female patients diagnosed with ovarian cancer and treated within the 1st Department of Obstetrics & Gynecology at AUTh, "Papageorgiou" General Hospital. The retrospective review encompassed a timeframe extending from January 1, 2019, to December 31, 2022. Specifically, our focus was on identifying individuals who underwent primary or interval debulking surgery within this specified duration. Remarkably, a cohort of 100 patients received a diagnosis of ovarian cancer during the aforementioned period. In adherence to ethical protocols, we sought and obtained written approval from the Institutional Review Board of the hospital, ensuring that our retrospective analysis aligns with the highest standards of research integrity and patient confidentiality.
2.2. Patients
Inclusion criteria:
Exclusion criteria:
Any other histological type except high grade serous.
Patients not tested for HRD.
Missing important registry data of CA-125 values to calculate KELIM.
The application of the aforementioned stringent criteria resulted in the exclusion of 29 out of the initial 100 women diagnosed with ovarian cancer, primarily due to histological types other than high-grade serous. Additionally, 20 women were omitted from the study as they did not undergo testing for homologous recombination deficiency (HRD). Another 12 women were excluded due to the absence of crucial registry data related to CA-125 values, which are essential for calculating KELIM. Consequently, after thorough screening and exclusion based on these criteria, a refined cohort of 39 women diagnosed with high-grade serous ovarian cancer emerged as eligible for further analysis. It is crucial to note that this final cohort exhibits a careful elimination of duplicate data and addresses the issue of missing values that could potentially impact the integrity of the analysis. The patient selection process (flowchart) is visually represented in
Figure 1, providing a transparent overview of the refined cohort and the exclusion criteria applied to ensure the robustness of the subsequent analysis.
2.3. Data Collection
The data collection process spanned a period of one month, during which we scrupulously gathered information. To streamline and standardize the data collection process, we leveraged the online registry of our Gynecological – Oncology Unit containing all pertinent details from the patients' medical records. In an effort to mitigate potential inconsistencies stemming from variations in data collection dates, a systematic approach was adopted. A uniform data collection sheet, formatted as an Excel file, was employed for the retrospective compilation of patients' medical records. This standardized sheet served as a systematic tool to ensure consistency and accuracy in the recorded information. The data collection sheet encompassed a range of essential information, contributing to a holistic understanding of each patient's medical history. This included the following information:
-
Patient’s identifiers:
- o
Name
- o
Hospital identification number
Patient’s age
Tumor marker CA-125 serial values during chemotherapy
KELIM Score
Homologous Recombination Deficiency (HRD) status
Residual Disease after debulking surgery
-
Chemotherapy:
The determination of HRD status was tested either by myChoice HRD CDx assay [
26] or by RediScore assay [
27]. Both assays assess the genomic instability score (GIS score) and the cut-off point for HRD positive status was 42 or greater (≥ 42). This dual-testing approach ensures a robust evaluation of homologous recombination deficiency. On the other hand, KELIM score was measured in both neoadjuvant and adjuvant settings with the online tool biomarker-kinetics.org/CA-125-neo or biomarker-kinetics.org/CA-125, respectively. The dates of every cycle of chemotherapy were entered and the relevant values of CA-125 within the first 100 days from the start of neoadjuvant or adjuvant chemotherapy. Preferably the CA-125 values before cycles 2, 3 and 4 were used to calculate KELIM score, but if one was missing, the CA-125 value prior to the 1st cycle of chemotherapy (within 7 days from neoadjuvant chemotherapy start) was considered. The KELIM score was analyzed as a continuous and as a binary index test with the cut-off point of 1 or greater (≥ 1) for favorable result.
2.4. Statistical Analysis
In the statistical analysis the baseline characteristics of the patients who participated in the study were calculated. There was no case of missing data. Continuous variables are demonstrated as means with standard deviation (SD) while categorical variables with frequencies and precents n (%). A receiver operating characteristic (ROC) curve analysis and the Youden index (J) [
28,
29] of KELIM score were performed to identify the most appropriate cut-off value for the ovarian cancer’s diagnosis. The sensitivity, specificity, positive predictive value, negative predictive value and area under the curve (AUC) with 95% confidence intervals (CI) were calculated. Test of normality was conducted using Shapiro-Wilk test. All reported P-values were two-tailed at a 5% significance level. We analyzed data using R statistical software (R Project for Statistical Computing) , version 4.3.0 using the package called pROC [
30].
3. Results
This retrospective diagnostic accuracy cross-sectional study included 100 women, who were treated during the period of the study for histologically proven high grade ovarian cancer in the Gynecological – Oncology Unit, 1st Department of Obstetrics & Gynecology, Aristotle University of Thessaloniki, “Papageorgiou” Hospital. After screening the patients’ records based on the inclusion and exclusion criteria, 39 patients were eligible for further analysis in this study.
Patients’ data are outlined in
Table 1. Thirty-nine patients had all necessary data for both HRD status and CA-125 values, at the correct time frame in relation to cycles of chemotherapy during the first 100 days of treatment, in order to calculate the KELIM score through the online tool. The interplay between KELIM score and HRD status revealed notable findings, with approximately half (54%) of the patients exhibiting a favorable KELIM score (≥ 1). Similarly, an equivalent proportion (54%) demonstrated a positive HRD status. The mean age of the patients was 60 years old with a standard deviation of 10. Furthermore, concerning the timing of systemic treatment, our patients were equally distributed between the 2 groups: 22 (56%) women were offered neoadjuvant, while 17 (44%) adjuvant chemotherapy. Importantly, all patients underwent debulking surgery, with a significant majority (87%) achieving either complete or optimal cytoreduction.
The main endpoint of our study was to compare KELIM to HRD as a possible alternative tool to identify patients that could benefit from PARPi maintenance therapy. Our reference standard was the HRD test (either myChoice HRD CDx assay or by RediScore assay, which are both validated) and our index test was the KELIM score, through the online tool biomarker-kinetics.org/CA-125-neo or biomarker-kinetics.org/CA-125, based on the neoadjuvant or adjuvant setting. Firstly, when assuming KELIM score as a binary index test with the value 1, as the cut-off point, which is also proposed by the creators of the model, the sensitivity was 0.86, 95% CI (0.64-0.97), and the specificity 0.83, 95% CI (0.59-0.96). The relevant data are presented in
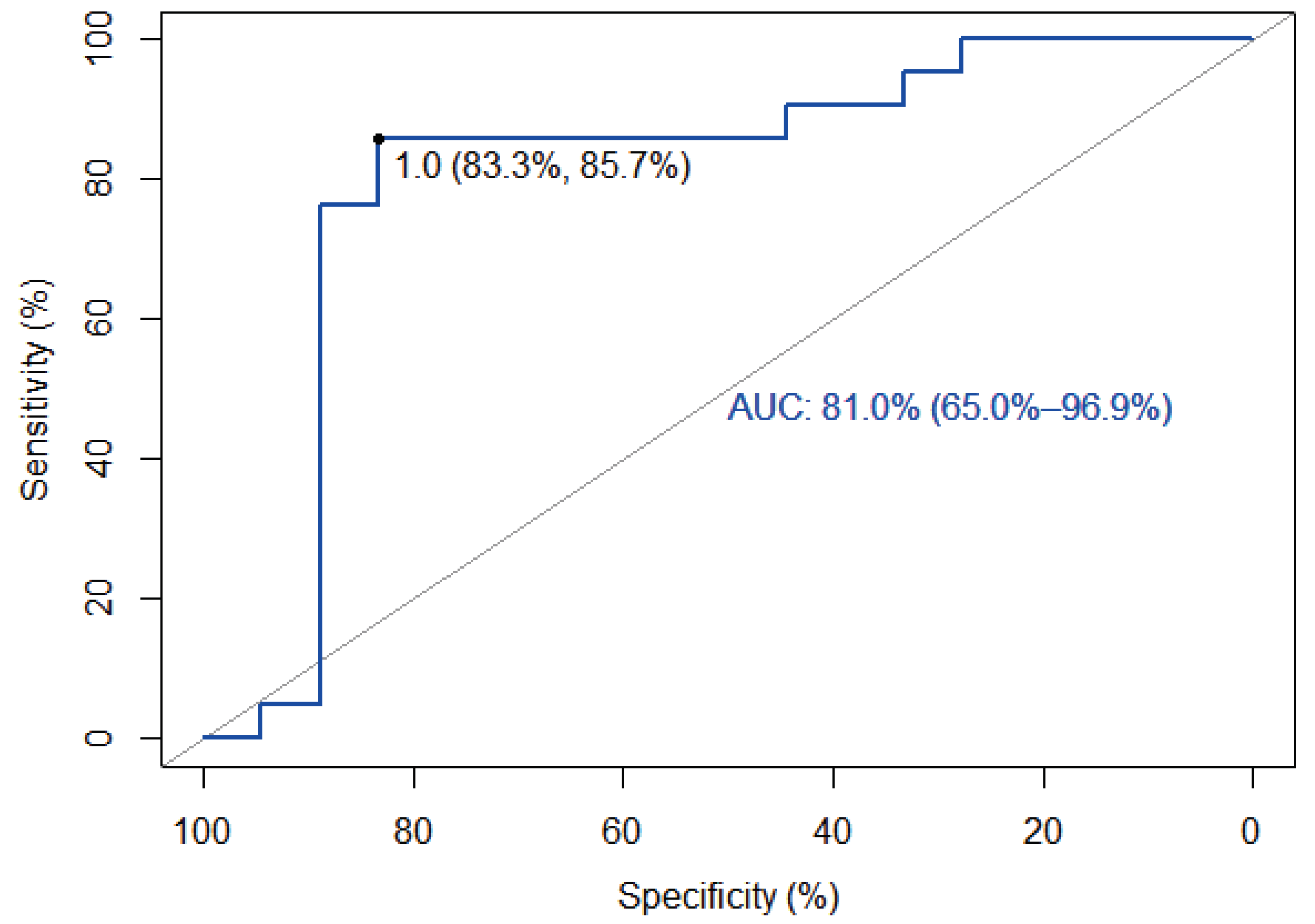
Table 2. On the other hand, when assuming KELIM as a continuous index test and constructing a ROC curve, the AUC was 81% and the optimal threshold, using the Youden index, was identified as 1.03, which is in accordance with proposal of the creators of KELIM, with a sensitivity of 85.7% and a specificity of 83.3%.
Figure 2 shows the aforementioned data.
4. Discussion
The primary objective of our study was to investigate the potential utility of the KELIM score as an additional tool for identifying patients who may benefit from PARPi therapy. To achieve this, we conducted a comparative analysis with HRD status, currently regarded as the gold standard test, along with BRCA mutation, for the administration of maintenance PARPi therapy. We designed a retrospective diagnostic accuracy cross-sectional study, structured according to the PICO model: The population was high grade serous advanced ovarian cancer patients, the index test was KELIM score, the control was HRD test and outcomes were the sensitivity – specificity and the AUC of the new diagnostic test.
Thirty-nine patients were included in the study and they almost equally underwent neoadjuvant (56%) and adjuvant (44%) chemotherapy, while the percentage of complete of optimal debulking was high (84%). Furthermore, almost half (54%) of the patients had a positive HRD status and a favorable (≥ 1) KELIM score. When assuming KELIM score as a binary index test with a cut-off value of 1 the sensitivity was 86% and the specificity 83%, results that classify the diagnostic accuracy of the KELIM score as very good. On the other hand, when assuming KELIM score as a continuous index test and constructing a ROC curve the AUC was 81%, which is considered as an excellent score. The optimal threshold value of the diagnostic test was calculated at 1.03, with a sensitivity of 85.7% and a specificity of 83.3%.
To our best knowledge this is the only diagnostic accuracy study in the literature for KELIM score, as a predictor for PARPi response. Two recent meta-analyses [
7,
17] showed that KELIM is an independent prognostic biomarker for survival outcomes and that can predict chemosensitivity. There are only a few studied in the literature that investigate the role of KELIM as a potential indicator of which patients might benefit from PARPi maintenance therapy.
The most relevant one is a post hoc study [
31], which used the data of 854 patients that were enrolled in the VELIA trial and explored the prognostic value of KELIM with the benefit from veliparib. The study included patients that underwent both adjuvant and neoadjuvant chemotherapy, but had as a “control” test both HRD and BRCA status. The results agree with our findings and suggest that KELIM score might be another complementary determinant of PARPi efficacy. Moreover, a retrospective study from Canada [
32], that was published only as an abstract from the ASCO 2022 meeting, included 70 patients and stated that KELIM can be used to aid clinical decision-making when HRD testing is unfunded. Another post hoc study [
33], that used the datasets of ARIEL2 and STUDY 10, concluded that KELIM is a pragmatic indicator of rucaparib efficacy, but in the recurrent not in the first-line setting.
Last but not least, a recent narrative review of the literature [
3] from the same authors that created the KELIM score , in the chapter about the potential utilities of KELIM, that it may be a useful tool for identifying patients who might benefit from PARPi maintenance therapy. However, they underline that further studies are needed to better clarify this possible association between KELIM and PARPi efficacy, which is exactly the knowledge gap that our study tries to fill in.
This is the first diagnostic accuracy study that investigates the role of KELIM score, compared to HRD status, in identifying patients that could benefit from PARPi maintenance therapy. All the required parameters were collected from an online system, therefore minimizing the percentage of missing important data. It is of high importance to state that the dataset, on which this study was conducted, comes from an ESGO certified center for Advanced Ovarian Cancer Surgery for over a decade, thus ensuring the high-quality of the data. Nevertheless, the main limitation of our study is the low number of the population included in the final analysis and its retrospective nature.
The results of our study could have a huge impact in every-day clinical practice, because KELIM score seems to be as good as HRD status in the selection of patients that will benefit from PARPi maintenance therapy, while being an easy, cheap and reproduceable tool to use in clinical practice. The value of KELIM score seems to be an alternative triage tool in world areas where HRD testing is unavailable. Further prospective studies are imperative to validate these promising results, before KELIM score is established as a diagnostic test for PARPi efficacy.
5. Conclusions
The KELIM score stands as a novel, cost-effective, and easily reproducible biomarker, offering a streamlined approach to identify patients poised to benefit from PARPi maintenance therapy, especially when compared to the conventional assessment of HRD/BRCA status. This emerging biomarker presents a promising avenue for refining patient stratification, enhancing treatment efficacy, and optimizing resource utilization in the context of PARPi maintenance therapy. Therefore, it is recommended that future investigations in the field of ovarian cancer therapy embrace the inclusion of the KELIM score as a fundamental component in their study designs. This will contribute to the ongoing evolution of precision medicine, fostering a deeper comprehension of the dynamics between KELIM scores and treatment outcomes in the era of PARPi.
Author Contributions
Conceptualization, D.Z. and D.T.; methodology, K.C.; software, V.T.; validation, D.T., G.G. and E.T.; formal analysis, D.Z. and P.T.; investigation, P.T. and I.S.; resources, G.G. and E.T.; data curation, P.T.; writing—original draft preparation, D.Z.; writing—review and editing, D.T.; visualization, I.S.; supervision, E.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board) of PAPAGEORGIOU General Hospital (No. 7848/08/05/2023).
Informed Consent Statement
Patient consent was waived due to fact that this was retrospective study and no change in the treatment of the patients was made.
Data Availability Statement
In accordance with the journal’s guidelines, the data presented in this study are available on request from the corresponding author for the reproducibility of this study if such is requested.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bristow, R.E.; Tomacruz, R.S.; Armstrong, D.K.; Trimble, E.L.; Montz, F.J. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J. Clin. Oncol. 2002, 20, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, N.S.; Miller, A.; Rungruang, B.; Richard, S.D.; Rodriguez, N.; Bookman, M.A.; Hamilton, C.A.; Krivak, T.C.; Maxwell, G.L. Does aggressive surgery improve outcomes? Interaction between preoperative disease burden and complex surgery in patients with advanced-stage ovarian cancer: an analysis of GOG 182. J. Clin. Oncol. 2015, 33, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Lauby, A.; Colomban, O.; Corbaux, P.; Peron, J.; Van Wagensveld, L.; Gertych, W.; Bakrin, N.; Descargues, P.; Lopez, J.; Kepenekian, V.; et al. The Increasing Prognostic and Predictive Roles of the Tumor Primary Chemosensitivity Assessed by CA-125 Elimination Rate Constant K (KELIM) in Ovarian Cancer: A Narrative Review. Cancers (Basel). 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Rutten, M.J.; van de Vrie, R.; Bruining, A.; Spijkerboer, A.M.; Mol, B.W.; Kenter, G.G.; Buist, M.R. Predicting surgical outcome in patients with International Federation of Gynecology and Obstetrics stage III or IV ovarian cancer using computed tomography: a systematic review of prediction models. Int. J. Gynecol. Cancer 2015, 25, 407–415. [Google Scholar] [CrossRef]
- Rustin, G.J.S.; Quinn, M.; Thigpen, T.; du Bois, A.; Pujade-Lauraine, E.; Jakobsen, A.; Eisenhauer, E.; Sagae, S.; Greven, K.; Vergote, I.; et al. Re: New guidelines to evaluate the response to treatment in solid tumors (ovarian cancer). J. Natl. Cancer Inst. 2004, 96, 487–488. [Google Scholar] [CrossRef] [PubMed]
- Colombo, N.; Sessa, C.; Bois, A. du; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Int. J. Gynecol. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Cho, H.-W.; Park, E.Y.; Han, K.-H.; Kim, E.T.; Lee, J.-K.; Park, S.-Y.; Armbrust, R.; Fotopoulou, C.; Lim, M.C. Prognostic value of CA125 kinetics, half-life, and nadir in the treatment of epithelial ovarian cancer: a systematic review and meta-analysis. Int. J. Gynecol. Cancer 2023, 33, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- JL, X.; J, C.; E, P.; TL, R.; PC, P.; CC, J.; SS, B. Longitudinal evaluation of CA-125 velocity and prediction of ovarian cancer. Gynecol. Oncol. 2012, 125. [Google Scholar] [CrossRef]
- Almufti, R.; Wilbaux, M.; Oza, A.; Henin, E.; Freyer, G.; Tod, M.; Colomban, O.; You, B. A critical review of the analytical approaches for circulating tumor biomarker kinetics during treatment. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2014, 25, 41–56. [Google Scholar] [CrossRef]
- Riedinger, J.M.; Wafflart, J.; Ricolleau, G.; Eche, N.; Larbre, H.; Basuyau, J.P.; Dalifard, I.; Hacene, K.; Pichon, M.F. CA 125 half-life and CA 125 nadir during induction chemotherapy are independent predictors of epithelial ovarian cancer outcome: results of a French multicentric study. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2006, 17, 1234–1238. [Google Scholar] [CrossRef]
- Lee, C.K.; Friedlander, M.; Brown, C.; Gebski, V.J.; Georgoulopoulos, A.; Vergote, I.; Pignata, S.; Donadello, N.; Schmalfeldt, B.; Delva, R.; et al. Early decline in cancer antigen 125 as a surrogate for progression-free survival in recurrent ovarian cancer. J. Natl. Cancer Inst. 2011, 103, 1338–1342. [Google Scholar] [CrossRef]
- You, B.; Colomban, O.; Heywood, M.; Lee, C.; Davy, M.; Reed, N.; Pignata, S.; Varsellona, N.; Emons, G.; Rehman, K.; et al. The strong prognostic value of KELIM, a model-based parameter from CA 125 kinetics in ovarian cancer: data from CALYPSO trial (a GINECO-GCIG study). Gynecol. Oncol. 2013, 130, 289–294. [Google Scholar] [CrossRef]
- Colomban, O.; Tod, M.; Leary, A.; Ray-Coquard, I.; Lortholary, A.; Hardy-Bessard, A.C.; Pfisterer, J.; Du Bois, A.; Kurzeder, C.; Burges, A.; et al. Early Modeled Longitudinal CA-125 Kinetics and Survival of Ovarian Cancer Patients: A GINECO AGO MRC CTU Study. Clin. Cancer Res. 2019, 25, 5342–5350. [Google Scholar] [CrossRef]
- Ducoulombier, S.; Golfier, F.; Colomban, O.; Benayoun, D.; Bolze, P.-A.; Tod, M.; You, B. Modeling CA-125 During Neoadjuvant Chemotherapy for Predicting Optimal Cytoreduction and Relapse Risk in Ovarian Cancer. Anticancer Res. 2017, 37, 6879–6886. [Google Scholar] [CrossRef]
- You, B.; Purdy, C.; Copeland, L.J.; Swisher, E.M.; Bookman, M.A.; Fleming, G.; Coleman, R.; Randall, L.M.; Tewari, K.S.; Monk, B.J.; et al. Identification of Patients With Ovarian Cancer Experiencing the Highest Benefit From Bevacizumab in the First-Line Setting on the Basis of Their Tumor-Intrinsic Chemosensitivity (KELIM): The GOG-0218 Validation Study. J. Clin. Oncol. 2022, 40, 3965–3974. [Google Scholar] [CrossRef]
- You, B.; Robelin, P.; Tod, M.; Louvet, C.; Lotz, J.-P.; Abadie-Lacourtoisie, S.; Fabbro, M.; Desauw, C.; Bonichon-Lamichhane, N.; Kurtz, J.-E.; et al. CA-125 ELIMination Rate Constant K (KELIM) Is a Marker of Chemosensitivity in Patients with Ovarian Cancer: Results from the Phase II CHIVA Trial. Clin. Cancer Res. 2020, 26, 4625–4632. [Google Scholar] [CrossRef]
- Corbaux, P.; You, B.; Glasspool, R.M.; Yanaihara, N.; Tinker, A. V; Lindemann, K.; Ray-Coquard, I.L.; Mirza, M.R.; Subtil, F.; Colomban, O.; et al. Survival and modelled cancer antigen-125 ELIMination rate constant K score in ovarian cancer patients in first-line before poly(ADP-ribose) polymerase inhibitor era: A Gynaecologic Cancer Intergroup meta-analysis. Eur. J. Cancer 2023, 191, 112966. [Google Scholar] [CrossRef]
- Labidi-Galy, S.I.; de La Motte Rouge, T.; Derbel, O.; Wolfer, A.; Kalbacher, E.; Olivier, T.; Combes, J.-D.; Heimgartner-Hu, K.; Tredan, O.; Guevara, H.; et al. Clinical factors associated with prolonged response and survival under olaparib as maintenance therapy in BRCA mutated ovarian cancers. Gynecol. Oncol. 2019, 155, 262–269. [Google Scholar] [CrossRef]
- Fong, P.C.; Yap, T.A.; Boss, D.S.; Carden, C.P.; Mergui-Roelvink, M.; Gourley, C.; De Greve, J.; Lubinski, J.; Shanley, S.; Messiou, C.; et al. Poly(ADP)-ribose polymerase inhibition: frequent durable responses in BRCA carrier ovarian cancer correlating with platinum-free interval. J. Clin. Oncol. 2010, 28, 2512–2519. [Google Scholar] [CrossRef]
- Monk, B.J.; Parkinson, C.; Lim, M.C.; O’Malley, D.M.; Oaknin, A.; Wilson, M.K.; Coleman, R.L.; Lorusso, D.; Bessette, P.; Ghamande, S.; et al. A Randomized, Phase III Trial to Evaluate Rucaparib Monotherapy as Maintenance Treatment in Patients With Newly Diagnosed Ovarian Cancer (ATHENA-MONO/GOG-3020/ENGOT-ov45). J. Clin. Oncol. 2022, 40, 3952–3964. [Google Scholar] [CrossRef] [PubMed]
- Ray-Coquard, I.; Pautier, P.; Pignata, S.; Pérol, D.; González-Martín, A.; Berger, R.; Fujiwara, K.; Vergote, I.; Colombo, N.; Mäenpää, J.; et al. Olaparib plus Bevacizumab as First-Line Maintenance in Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2416–2428. [Google Scholar] [CrossRef]
- González-Martín, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2391–2402. [Google Scholar] [CrossRef]
- Coleman, R.L.; Fleming, G.F.; Brady, M.F.; Swisher, E.M.; Steffensen, K.D.; Friedlander, M.; Okamoto, A.; Moore, K.N.; Efrat Ben-Baruch, N.; Werner, T.L.; et al. Veliparib with First-Line Chemotherapy and as Maintenance Therapy in Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2403–2415. [Google Scholar] [CrossRef]
- Stoppa-Lyonnet, D. The biological effects and clinical implications of BRCA mutations: where do we go from here? Eur. J. Hum. Genet. 2016, 24 Suppl 1, S3–9. [Google Scholar] [CrossRef]
- Pennington, K.P.; Walsh, T.; Harrell, M.I.; Lee, M.K.; Pennil, C.C.; Rendi, M.H.; Thornton, A.; Norquist, B.M.; Casadei, S.; Nord, A.S.; et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin. Cancer Res. 2014, 20, 764–775. [Google Scholar] [CrossRef]
- Arora, S.; Balasubramaniam, S.; Zhang, H.; Berman, T.; Narayan, P.; Suzman, D.; Bloomquist, E.; Tang, S.; Gong, Y.; Sridhara, R.; et al. FDA Approval Summary: Olaparib Monotherapy or in Combination with Bevacizumab for the Maintenance Treatment of Patients with Advanced Ovarian Cancer. Oncologist 2021, 26, e164–e172. [Google Scholar] [CrossRef]
- Tsantikidi, A.; Papazisis, K.; Floros, T.; Gazouli, M.; Papadopoulou, E.; Tsaousis, G.; Nasioulas, G.; Mester, A.; Milan, K.P.; Gozman, B.; et al. RediScore: Prospective validation of a pipeline for homologous recombination deficiency analysis. Oncol. Lett. 2023, 26, 480. [Google Scholar] [CrossRef]
- Liu, X. Classification accuracy and cut point selection. Stat. Med. 2012, 31, 2676–2686. [Google Scholar] [CrossRef]
- Fluss, R.; Faraggi, D.; Reiser, B. Estimation of the Youden Index and its associated cutoff point. Biom. J. 2005, 47, 458–472. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.-C.; Müller, M. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics 2011, 12, 77. [Google Scholar] [CrossRef]
- You, B.; Sehgal, V.; Hosmane, B.; Huang, X.; Ansell, P.J.; Dinh, M.H.; Bell-McGuinn, K.; Luo, X.; Fleming, G.F.; Friedlander, M.; et al. CA-125 KELIM as a Potential Complementary Tool for Predicting Veliparib Benefit: An Exploratory Analysis From the VELIA/GOG-3005 Study. J. Clin. Oncol. 2023, 41, 107–116. [Google Scholar] [CrossRef]
- Hannaway, N.; Kassaris, S.; Davies, J.M.; Smrke, A.; Tinker, A.; Drew, Y. Using chemotherapy response by KELIM score to predict response to first line maintenance PARP inhibitor therapy in non-BRCA mutant/homologous recombination deficiency (HRD) unknown high grade serous ovarian cancer (HGSOC). J. Clin. Oncol. 2023, 41, e17547–e17547. [Google Scholar] [CrossRef]
- Colomban, O.; Swisher, E.M.; Kristeleit, R.; McNeish, I.; Shapira-Frommer, R.; Goble, S.; Lin, K.K.; Maloney, L.; Freyer, G.; You, B. Mathematical modeling of the early modeled CA-125 longitudinal kinetics (KELIM-PARP) as a pragmatic indicator of rucaparib efficacy in patients with recurrent ovarian carcinoma in ARIEL2 & STUDY 10. EBioMedicine 2023, 89, 104477. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).