1. Introduction
Wilms tumor (WT), also known as nephroblastoma, is the most prevalent renal tumor in children, accounting for more than 90% of cases. It is also the second most frequent extracranial tumor after neuroblastoma. A global study found that the age-adjusted incidence rate of renal tumors in children was 8.3 per million, and that this rate showed an upward trend, particularly in Southeast Asia [
1]. In the United States, approximately 600 new cases are diagnosed annually, and the incidence appears to be relatively stable [
2]. The treatment of WT generally has a favorable prognosis, with an overall survival rate of over 90%. However, 10% of WT cases are resistant to chemotherapy and have poor outcomes [
3]. Moreover, disease recurrence is another key factor that influences patient survival. The survival rate for recurrent WT after treatment is only 25%, even with intensive chemotherapy [
4]. WT at advanced stages (COG stage III-V) has a higher risk of recurrence than WT at lower stages (COG stage < II). The 4-year event free survival for COG stage III and IV WT was 80.9% and 41.7%, respectively, while it was 86% for COG stage II WT [
3]. Current research on WT focuses on identifying molecular biomarkers and investigate molecular-targeted therapies as possible strategies to overcome chemotherapy resistance [
5].
WT exhibits molecular pathogenesis involving the activation of Wnt signaling pathways. This is supported by the detection of somatic variants in
CTNNB1 (10–15%), coupled with background germline variants in
WT1. These two genes normally involve in renal development process [
6,
7]. Additionally, somatic variant in the tumor suppressor gene
TP53 can be identified in 47.5% of WT patients with diffuse anaplastic histology. Other somatic variants found in WT include
DROSHA,
AMER1 (
WTX), and
DGCR8, which occur in around 5-10% of WT cases [
8]. Furthermore, somatic variant in genes associated with microRNA biogenesis process (
DGCR8,
DICER1,
XPO5 and
TARBP2) were found in up to 12 % of WTs [
9,
10]. Interestingly,
AMER1 variants have a high frequency (10.1%), but it does not appear to have any discernible clinical impact. However,
WT1 could be a potential target in anti-angiogenesis therapy, such as bevacizumab and AZD2171 [
11]. Although targeted therapy for
TP53 has not been specifically mentioned in WT, drugs targeting this gene with FDA approval and in clinical trials are commonly available [
12]. Unfortunately, few drugs have been developed specifically for childhood tumors due to a small market for a rare childhood disease. Current research on WT treatment mainly focused on repurposing pediatric tumor-specific targeted therapy and immunotherapy. These two types of drugs were believed to become the two major adjuvant treatment for postoperative WT [
11]. While most of the other variants are associated with clinical outcomes and treatment [
13], none of these variants have been used as markers for chemotherapy resistance or as molecular targets for therapy. The identification of somatic variants in chemotherapy resistant WT implies the possibility of finding mutation markers for predicting resistance in WT cases and discovering new molecular biomarkers and therapeutic targets to overcome chemotherapy resistance in WT [
14,
15]. This study aimed to investigate the somatic mutational landscape in WT using exome sequencing, identify potential mutation markers for predicting chemotherapy resistance events in advanced stage WT, and suggest the targeted chemotherapy based on the profile of driver mutations that might serve as novel therapeutic strategies against chemotherapy resistance.
2. Materials and Methods
2.1. Biological Samples and Sequencing Library Preparation
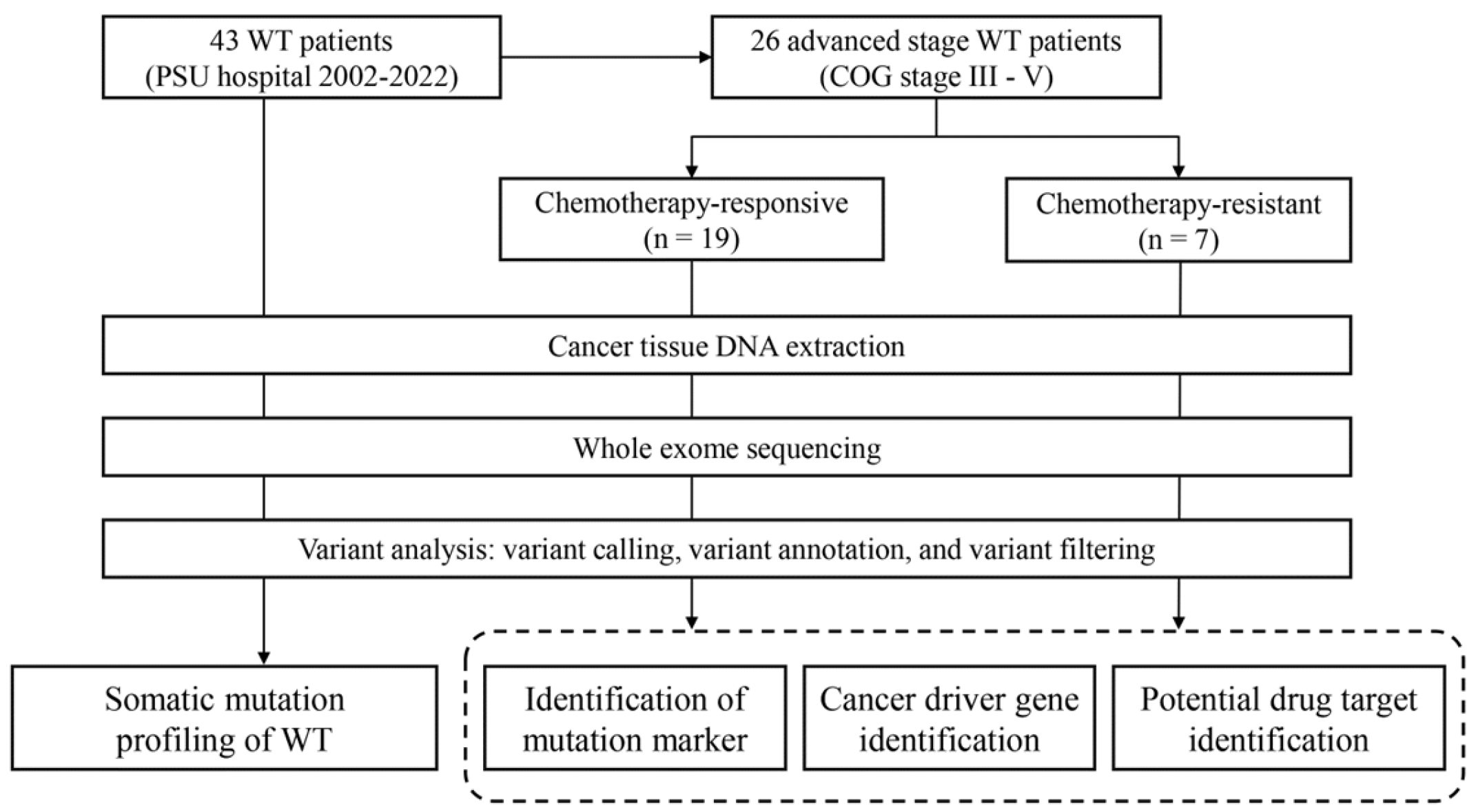
We obtained 43 fresh frozen tissue samples of WT from the biological repository of the Translational Medicine Research Center, Faculty of Medicine, Prince of Songkla University (Songkhla, Thailand). Unfortunately, their corresponding normal adjacent tissues or blood samples were unavailable among our cases. These samples originated from the surgical specimens of 43 WT patients who were younger than 15 years and had had been diagnosed with primary renal tumors. these patients received nephrectomy at Songklanagarind Hospital between January 10th, 2003 and December 26th, 2020. DNA extraction was done using DNeasy Blood & Tissue Kit (Qiagen Inc.). Subsequently, the quantity and quality of the extracted DNA were assessed using Nanodrop (Thermo Fisher Scientific, Inc.) and TapeStation (Agilent Technologies, Inc.). The clinical information of all the patients was retrieved from the electronic medical records (EMR) of the hospital. The EMR review process was conducted from April 1st, 2022, to May 31st, 2022. All patients and their legal guardians provided written informed consent before recruitment. The study was approved by the ethical committee of Faculty of Medicine, Prince of Songkla University (REC 64-195-10-1) (
Figure 1).
2.2. Whole Exome Sequencing
Our study employed whole exome sequencing as we primarily focused on comprehensive analysis of protein-affecting somatic variant. Whole exome sequencing was conducted using the library preparation system of Agilent SureSelect XT Human All Exon v6 (Agilent Technologies, Inc.) according to manufacturer’s specifications. The library was quantified using a Qubit dsDNA High Sense Assay Kit (Invitrogen, Carlsbad, CA, USA), and size measurements were performed using the Agilent D1000 ScreenTape assay. Sequencing was performed using an Illumina NovaSeq-6000 platform (Illumina, San Diego, California, United States) with paired-end reads of 150-bp. The average targeted coverage depth achieved was 200x.
The paired-end sequence files were assessed for quality using FastQC (version 0.11.9) and trimmed using Trimmomatic (version 0.39) [
16]. The optimally prepared FASTQ files were aligned with the human reference genome (version GRCh38.13) using the BWA program (version 0.7.17) [
17]. The resulting Sequence Alignment Map (SAM) files were converted to a Binary Alignment Map (BAM) format and sorted using the SAMtools (version 1.17) [
18]. Subsequently, the sorted BAM files were regrouped, and identical sequences were marked using Picard (version 3.0.0). To adjust the base quality score, unduplicated BAM files were processed using Genomic analysis toolkit (GATK, version 4.4.0) base quality score recalibration [
19]. The GATK tool was subsequently employed for somatic variant discovery, adhering to standard practices in genomic variant calling for both research and clinical contexts [
20]. Variant calling was performed using Mutect2 in tumor-only mode. A public Panel of Normals (PON) was downloaded from the public repository of GATK at
https://storage.googleapis.com/gatk-best-practices/somatic-hg38/1000g_pon.hg38.vcf.gz. We filtered the variants generated by Mutect2 using GATK4 GetPileupSummaries, CalculateContamination and FilterMutectCalls with a default value of argument. The resultant variants were then annotated using Funcotator. The annotated mutational data were stored in MAF files. It was subsequently summarized and visualized using maftools package in R [
21].
2.3. Identification of Mutation Marker for Chemotherapy-Resistant WT
Patients with advanced stage WT was divided into two group according to their response to standard postoperative chemotherapy following nephrectomy, as per the Thai Pediatric Oncology Group (ThaiPOG) protocol [
22]. Chemotherapy resistance was defined as either no change or increase in tumor size after chemotherapy, or recurrence during chemotherapy. Both primary and metastatic tumors were considered for chemotherapy resistance assessment. In our study, 19 patients with advanced stage WT responded to chemotherapy, while 7 were resistant. The proportion of affected patients with somatic mutations in each gene across two groups was compared using Fisher’s exact test. A p-value < 0.05 was considered as statistically significant. We also performed association analysis between somatic mutation profile and other clinical parameters, including histology, syndromic features, and lesion laterality, using the same method.
2.4. Identification of Cancer Driver Mutation and Their Potential Targeted Therapy
Driver mutation defined as the mutation that confer a growth advantage to cancer cells, enabling cancer initiation and progression [
23,
24]. We prioritized the annotated somatic mutations from chemotherapy-resistant group to identify potential driver mutations using two web-based machine learning tools, BoostDM and OncodriveMUT, accessible from the Cancer Genome Interpreter (CGI,
https://www.cancergenomeinterpreter.org) [
25]. These tools have accuracy above 0.8 in detect driver variant. Especially, BoostDM which outperformed when compare to other tools [
26]. CGI database would also provide the potential target therapy effective against the identified driver mutation CGI also provided potential target therapies effective against the identified driver mutations. Additionally, we searched the chemotherapy data, clinical trial studies, and level of evidence from MSK's Precision Oncology Knowledge Base (OncoKB,
https://www.oncokb.org), and Clinical Interpretation of Variant in Cancer (CIVIC,
https://civicdb.org).
2.5. Statistical Analysis
Statistical analyses were conducted using RStudio, based on the statistical language R version 4.3.0. We utilized Fisher’s exact test to assess the association between genes with somatic mutations and patients’ clinical phenotypes. Specifically, we compared the proportion of patients with somatic mutations and wildtype genes to the proportion of patients exhibiting the phenotype of interest. Survival analysis was performed using the Kaplan-Meier survival probability for survival function and the log-rank test for survival comparison. The Mantel-Haenszel method was used to estimate the hazard ratio for each identified marker. The level of statistical significance was set at p-value < 0.05.
3. Results
3.1. Clinical Characteristics of the Patients
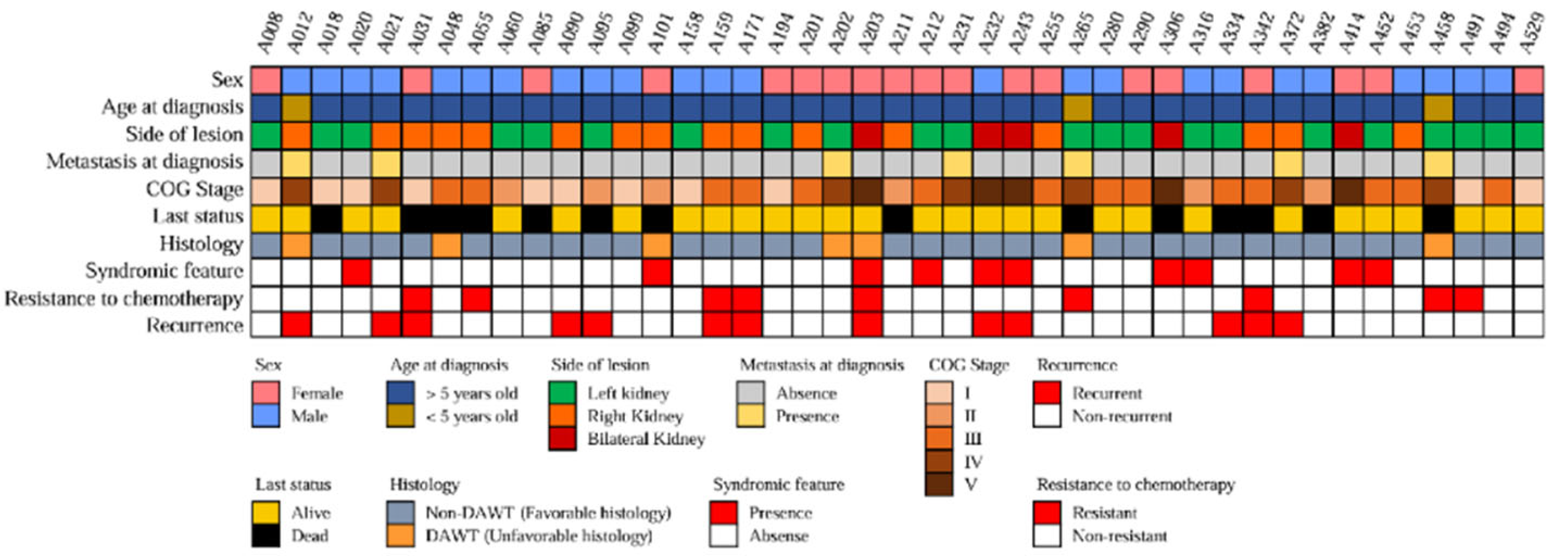
The analysis of 43 pediatric WT tissue samples was performed in this study.
Figure 2 shows the clinical features of all the patients. The diagnosis was made at a median age of 20.2 months (IQR: 10.9–39.5 months). The primary tumor was located on the left side in 24 cases (48.9%), on the right side in 20 cases (40.8%), and bilaterally in five cases (10.2%). The COG staging system classified 11 WT cases (25.6%) as stage I, six (14.0%) as stage II, 14 (32.6%) as stage III, seven (16.3%) as stage IV, and five (11.6%) as stage V. Thus, advanced stage disease (stage III - V) accounted for 60.47% (n=26) of all cases. The ThaiPOG protocol recommended a combination of Vincristine and actinomycin D as the postoperative chemotherapy regimen for localized WT, and doxorubicin, cyclophosphamide, and etoposide may be added for patients with stage III-IV. The study revealed that nine WT cases (20.93%) were resistant to the aforementioned chemotherapy regimen, of which seven cases (16.28%) had advanced stage disease (the chemotherapy regimen for these patients can be found in
supplementary data). Furthermore, the study detected recurrence disease in 13 cases (30.23%), despite standard surgery and chemotherapy. Additionally, ten patients displayed clinical phenotypes that could be linked to cancer syndromes, such as genitourinary tract anomalies, aniridia, albinism, and congenital spine anomalies.
3.2. Somatic Mutation Profiling of WT and Clinical Relevance
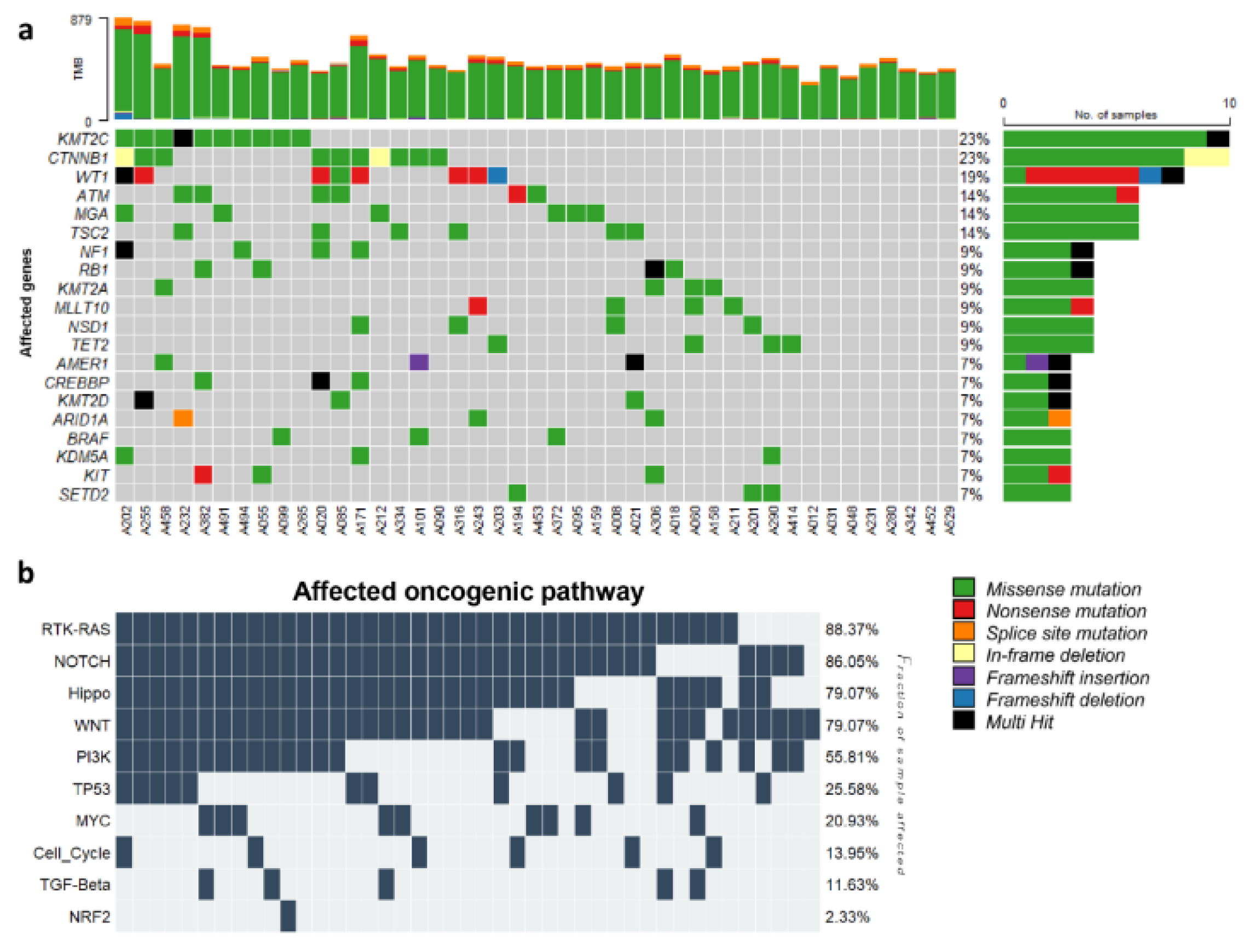
The bioinformatics analysis of the exome sequences of 43 WT samples identified 15,492 single nucleotide variants (SNVs) and 295 insertions or deletions (indels). These variants were exclusively exonic non-synonymous variants with a high or moderate impact, and a coverage of at least 20x. The analysis detected mutations in several cancer-related genes previously reported to be implicated in pediatric cancer, such as CTNNB1, WT1, AMER1, and NF1. These genes also listed among the 15 most frequently mutated genes in this analysis, which include KMT2C, CTNNB1, WT1, ATM, MGA, TSC2, NF1, RB1, KMT2A, MLLT10, NSD1, TET2, AMER1, CREBBP, and KMT2D (
Figure 3a). Among them, CTNNB1, WT1, and AMER1 have been identified in large cohorts of WT cases. Additionally, NF1 and CREBBP were previously reported in a single WT case. Although the remaining ten genes have not been previously reported to be mutated in WT, they have been associated with other pediatric malignancies. TSC2 and RB1 genes have been found to be mutated in acute T cell lymphoid leukemia and osteosarcoma. Mutations in ATM, MGA, KMT2A, MLLT10, NSD1, and TET2 have been commonly identified in several hematological neoplasms, including both acute lymphoid and myeloid leukemia [
27,
28]. KMT2C and KMT2D were frequently mutated in multiple types of cancer [
29,
30].
Furthermore, we analyzed the KEGG pathway of 43 patients with WT and identified the top 10 biological pathways affected by genetic alterations (
Figure 3b). Six pathways were affected by at least one mutated gene in all 43 WTs. All were signaling pathways, including the RTK-RAS, WNT, NOTCH, Hippo, PI3K, and MYC signaling pathways. The activation of the Wnt/β-catenin pathway is commonly associated with WTs [
31]. Aberrant activation of the PI3K-Akt signaling pathway, which plays a crucial role in cell proliferation, differentiation, and growth, [
32], has been identified in different types of leukemia and solid tumors, including WT [
33,
34]. Mutation of genes in the MYC signaling pathway, namely MYCN and MAX, has been found to be strongly associated with an increased risk of WT relapse [
35]. However, the remaining pathways were cancer-related signaling pathways that are commonly found in several types of human cancer [
36,
37].
We conducted a study to investigate the association between somatic variants and clinical parameters in our cohort. We used Fisher’s exact test to evaluate the association and found significant associations (
Supplementary Table S1). Specifically, we found that syndromic feature was associated with variant detection in 38 genes, and tumor laterality was linked to 10 genes. We also identified 7 genes, including
PRSS2, PRSS1, AKR7A3, ARID1A, METTL14, BRD8, and
CHD8, that were associated with both syndromic feature and bilaterality of tumor.
Furthermore, we found that 23 genes were associated with unfavorable histology, among which
MMP17, OR6C70, and
MPDZ variants were only present in unfavorable histology with the lowest p-value. In addition, we focused on the therapeutic response of WT in our cohort. We found that, from all advanced stage WT (n=26), 7 cases were resistant to postoperative chemotherapy. The median time of resistance was 190 days (ranging from 72 to 320 days) after surgery. Resistance to chemotherapeutic drug is a significant problem in cancer therapy, as it causes most cases of cancer recurrence and reduces the chances of survival for patients [
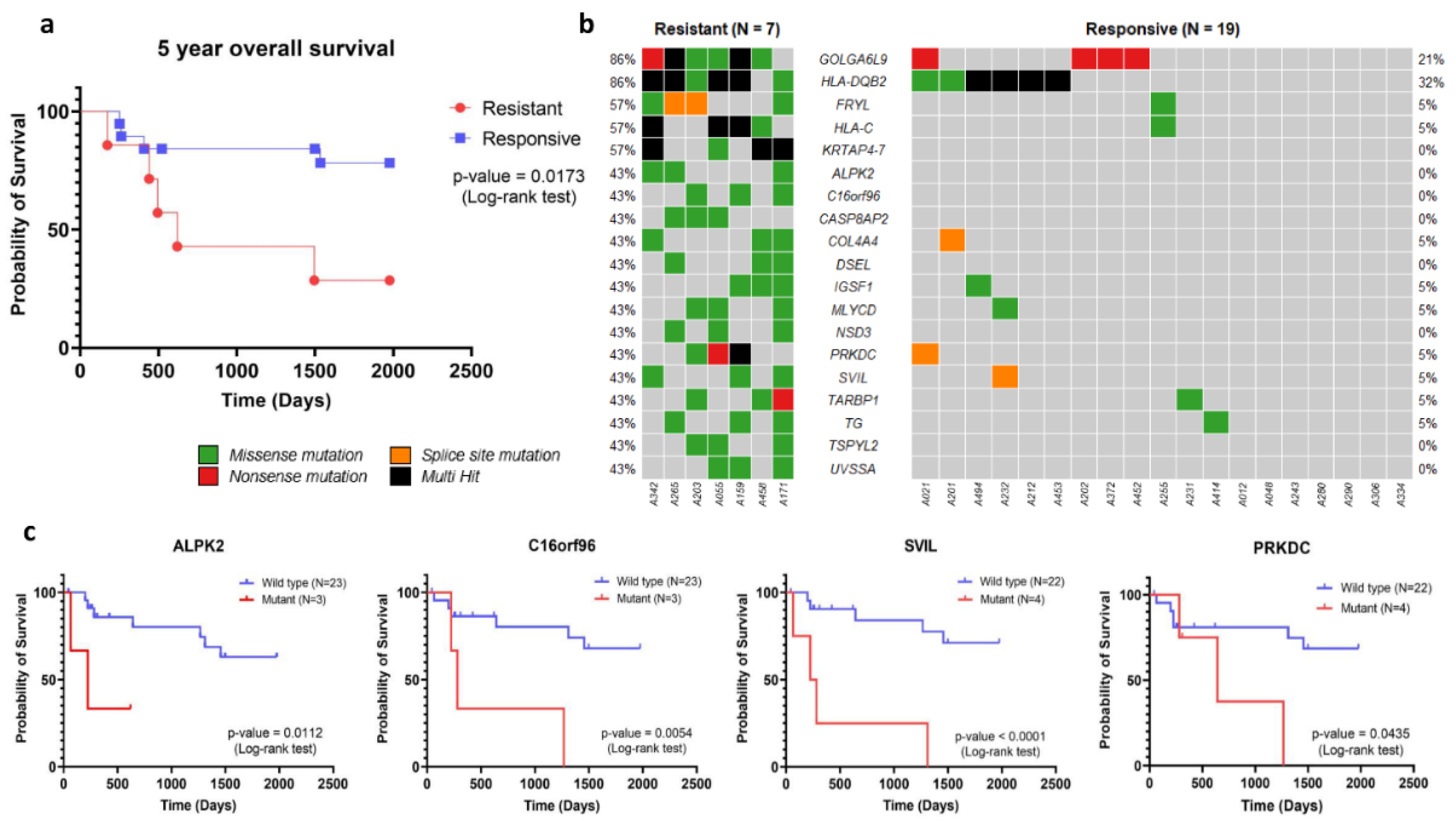
38]. The 5-year overall survival of advanced stage WT patients with chemotherapy resistance (n=7) indicated that the median time of survival in this group was 619 days, with a corresponding hazard ratio (HR) calculated using the Mantel-Haenszel method at 6.75 (95% confidence interval (CI): 1.40 - 32.53). These results demonstrated significantly poorer survival in the chemotherapy resistance group compared to that of chemotherapy responsive patients (n=19) (
Figure 4a, p-value 0.0173). The poorer 5-year overall survival of advanced stage WT patient with chemotherapy resistance had emphasized its impact to patient survival probability. To identify genetic marker for chemotherapy resistance, an association analysis using Fisher’s exact test was carried out to determine the relationship between genetic variants and chemotherapy resistance. Significant associations were found for variants in 19 genes that were associated with chemotherapy resistance (p-value < 0.05,
Figure 4b). When we used more stringent criteria at p-value < 0.01, we could identify 4 genes, including
KRTAP4-7, GOLGA6L9, FRYL, and
HLA-C, that were associated with chemotherapy resistance (
Table 1).
In the analysis of disease-free survival, we identified variants in four genes, namely
ALPK2,
C16orf96,
PRKDC, and
SVIL, that were significantly associated with lower disease-free survival (
Figure 4c). We estimated the hazard ratio of these genes using the Mantel-Haenszel method. The results showed that
ALPK2 (HR, 60.04 [95% CI: 2.534 - 1423]),
C16orf96 (HR, 31.20 [95% CI: 2.765-351.9]),
PRKDC (HR, 8.438 [95% CI: 1.064-66.94]), and
SVIL (HR, 63.82 [95% CI: 6.679-609.8]) were found to independently predict survival outcomes. Notably, these four genes might serve as putative candidate markers for predicting chemotherapy resistance among advanced stage WT patients.
3.3. Cancer Driver Gene and Potential Drug Target for Chemotherapy Resistant WT
The analysis could identify driver mutation in 55 genes (
Supplementary Figure S1). Among those, 8 driver genes were the most commonly recurrent driver, including MUC4, MUC16, NSD3, HLA-A, HERC2, CSMD3, HLF, and LRP1B, which could be identified in at least two WT cases each (
Figure 5). Driver mutation in MUC4 and MUC16 were the two most commonly identified driver. These two genes belong to the membrane-bound mucins family. Studies have reported a regulatory relationship between these two mucins and the canonical Wnt/β-catenin signaling pathway, which is a common pathway found activated in WT [
39,
40,
41]. Although the association between these two mucins and β-catenin has been reported in many cancers including pancreatic, lung, colorectal and ovarian cancer, this relationship in WT is less known [
42]. Moreover, the identification of Wnt/β-catenin as a pathway associated with WT and the mutation of MUC4 and MUC16 among WT cases might hint at a relationship between β-catenin and these two mucins.
To discover potential drug target of our identified driver mutation, the annotated drivers were examined for their potential association with molecular-targeting drugs, as determined by the Cancer Genome Interpreter. The analysis could identify druggable mutation on 14 cancer driver genes (
Supplementary Table S2). Notably, platinum-based chemotherapeutic agents, namely Cisplatin and Carboplatin, has favorable response in cancer with the presence of three driver mutation: PALB2 (p.M723X), BRCA1 (p.Q262H), and ERCC6 (p.M867V). In our cohort of resistant patients, 43% (3 out of 7) had mutations in these genes. Although the effect of platinum-based agents on WT with mutations in these genes has not been evaluated, previous clinical trials have investigated the response of mentioned agents in other cancers, such as breast, ovarian, and pancreatic cancer, that harbor the same driver mutations as our WT cases [
43,
44,
45,
46]. Therefore, these agents may be potential candidates for overcoming chemotherapy resistance in WT. In addition to platinum-based agents, PARP inhibitors are effective treatments for cancer patients with mutations in PALB2 (p.M723X) or BRCA1 (p.Q262H) genes. Several studies have demonstrated the clinical benefits of PARP inhibitors for these patients, and the US FDA has approved their use for breast [
47,
48], ovarian [
49], and prostate cancer [
50,
51,
52] patient with PALB2 or BRCA1 mutations.
Antibiotic/antineoplastic agents, such as Daunorubicin, Doxorubicin, and Mytomycin C, are promising chemotherapeutic agents that could be the preferred therapy for chemotherapy-resistant WT. These agents interfere with DNA synthesis as their mechanism of action. Clinical trials have confirmed the effectiveness of Daunorubicin in treating acute myeloid leukemia with DNMT3A (p.V687F) mutation, and professional guidelines have endorsed its use [
53]. Early trial studies have also demonstrated the responsiveness of Mytomycin C in pancreatic cancer with PALB2 (p.M723X) [54]. Furthermore, the CGI database has documented the efficacy of Doxorubicin against breast adenocarcinoma with TP53 (p.R273C) was reported in CGI database. However, liposomal Doxorubicin was found to be ineffective in ovarian cancer with LRP1B (p.S1148P and p.W3333L) [55]. These findings indicate the potential of antibiotic/antineoplastic agents in treating WT cancer with the driver mutations that we have also detected in 43% (3 out of 7) of our patients. Another druggable driver mutations that were identified in chemotherapy-resistant WT are PBRM1 (p.G989C) and CDH1 (p.D433G), which were responsive to EZH2 inhibitors and AR inhibitor (Bicalutamide), respectively [56,57]. Each variant was detected in a different patient out of 7 in our cohort. However, the effectiveness of these drugs has only been reported in pre-clinical studies.
The findings from our investigation of variant markers and alternative drugs for chemotherapy-resistant WT offer a novel perspective on the treatment of this condition. However, it is important to note that these results were derived from a small patient cohort. To enhance the validity of our study, further validation in a larger clinical cohort would be beneficial.
4. Discussion
Studies on somatic mutations in various cancers have led to the identification of molecular targets that might suggest novel therapies for the diseases, either through the development of new drugs or by repurposing existing ones. Molecular targeted therapy is an essential part of modern cancer treatment, but the options for WT are limited in terms of molecular drugs. This is due to the low incidence and distinct pathogenesis of these tumors. The analysis detected many genetic variants and mutations in genes related to cancer, including
CTNNB1 and
WT1, which have been previously reported to be associated with WT [
6,
7]. Consistent with the analysis result,
CTNNB1 and
WT1 were found in 23% and 19% of our cohort, respectively. The genetic alteration profile revealed 15 genes that were most frequently mutated in our WT patients. Among these genes, 5 genes have been previously identified in WT tumor. The other ten genes have not been reported to be mutated in WT before, but they have been associated with various pediatric cancers [
27,
28,
29,
30]. The analysis also showed the top 10 biological pathways that were affected by the genetic alterations, most of which were signaling pathways that regulate cell growth and survival, corroborating the pathogenesis process of this malignancy.
Previous studies have reported that about 10% of WT patients exhibit resistance to standard chemotherapy, leading to poor prognosis [
3]. Furthermore, disease recurrence is another major factor affecting patient survival. It has been shown that the survival rate for recurrent WT was only 25% despite aggressive chemotherapy [
4]. These findings indicate the importance of chemotherapy resistance and disease recurrence as predictors of poor disease outcome. In our study, we found that advanced stage WT patients with chemotherapy resistance (n=7) had significantly lower 5-year overall survival than advanced stage WT who responded to chemotherapy (n=19). The prediction of drug resistance in patients before chemotherapy administration could facilitate the selection of alternative chemotherapy regimens and improve patient prognosis. Our association analysis identified variants in 19 genes that were associated with chemotherapy resistance in advanced stage WT. Among them, variants in four genes,
ALPK2,
C16orf96,
PRKDC, and
SVIL, were significantly associated with lower disease-free survival. However, additional validation studies are necessary to recruit more clinical samples and assess their predictive power. In this validation study, we recommend using a simple sequencing method, such as capillary DNA sequencing, to enhance the clinical utility of the marker.
To find a therapeutic agent that could overcome chemotherapy resistance in WT, we first identified driver mutations in advanced stage WT with chemotherapy resistance. We found recurrent driver mutations in 55 genes, of which 14 were cancer driver genes with available drugs. Notably, 43% (3 out of 7) of the resistant WT cases had at least one driver mutation in
PALB2 (p.M723X),
BRCA1 (p.Q262H), or
ERCC6 (p.M867V). These mutations have been shown to be predictive of sensitivity to platinum-based chemotherapeutic agents in several clinical trials. Furthermore, cancers with
PALB2 (p.M723X) and
BRCA1 (p.Q262H) mutations can also benefit from PARP inhibitors, which are recommended in professional guidelines for treating breast, ovarian, and prostate cancer [
47,
48,
49,
50,
51,
52].
Besides the aforementioned drugs, antibiotic/antineoplastic agents, such as Daunorubicin, Doxorubicin, and Mytomycin C, can also target cancers with
DNMT3A (V687F),
PALB2 (M723X), and
TP53 (R273C) mutation [
53,54,55]. These mutations were also present in 43% (3 out of 7) of the resistant WT cases. Other druggable driver mutations that we detected in resistant WT cases were
PBRM1 (p.Gly989Cys) and
CDH1 (p.Asp433Gly), which were responsive to EZH2 inhibitors and AR inhibitor (Bicalutamide), respectively [56,57]. Although the efficacy of these chemotherapeutic agents on WT with the same mutations as ours has not been assessed, previous clinical trials have demonstrated their activity in other cancers with the same driver mutations. Moreover, this lack of assessment also affects our understanding of drug safety, as there is limited information available for WT patients. Consequently, further research is needed to evaluate the therapeutic potential of targeting the driver mutations that we identified in our advanced stage WT with chemotherapy resistance.
In summary, we performed exome sequencing to discover somatic variants in 43 WT cases. Our findings reveal the possible mutation markers for chemotherapy resistance in advanced stage WT, and suggest the potential therapeutic options that could be effective for this group of patients.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org, Supplementary Figure S1: The distribution of driver mutations in chemotherapy-resistant and chemotherapy-responsive WT patients; Supplementary Table S1: The mutation rate in the genes associated with clinicopathological features; Supplementary Table S2: The potential targeted drugs for the driver mutations in chemotherapy-resistant WT. Supplementary data: The dosage of chemotherapy administered to the patients in the resistant group. Supplementary Material: Collaborators/Membership of the TPCA Consortium.
Author Contributions
Conceptualization, P.C. and S.S.; methodology, P.C., N.K., N.N. and S.S.; Software, P.C., N.K., N.N. and K.S.; Validation P.C. and S.S.; Investigation, P.C., W.L., T.C., T.S. and S.S.; Formal Analysis and interpretation, P.C., N.K., N.N., and S.S.; Resources, P.C., N.K., N.N., K.S., T.S. and S.S.; Data Curation, P.C., W.L., and S.S.; writing-original draft preparation, P.C. and S.S.; writing-review and editing, P.C., K.S., and S.S.; Visualization, P.C.; Supervision, K.S. and S.S.; Project Administration S.S.; funding acquisition, S.S. All authors contributed significantly to the article and approved the final version for publication.
Funding
The study is supported by the Fundamental Research Fund through Prince of Songkla University (NRIIS172177 and MED6505138S) and the Genomic Thailand Initiative through Health Systems Research Institute (NRIIS178921 and HSRI 65-093).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by The Human Research Ethics Committee at the Faculty of Medicine, Prince of Songkla University, Thailand (REC 64-195-10-1).
Informed Consent Statement
Prior to their participation in the study, written informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author due to ethical concerns regarding patient privacy.
Acknowledgments
The authors would like to gratefully acknowledge Translational Medicine Research Center, Faculty of Medicine, Prince of Songkla University, for providing laboratory equipment support. Our grat-itude also extends to the doctors, nurses, and genetic counselors who facilitated the enrollment of patients into this study.
Conflicts of Interest
The authors declare that the research was carried out without any existing commercial or financial affiliations that might be interpreted as potential conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Nakata, K.; Colombet, M.; Stiller, C.A.; Pritchard-Jones, K.; Steliarova-Foucher, E.; Contributors, I.-. Incidence of childhood renal tumours: An international population-based study. Int J Cancer. 2020, 147, 3313–3327. [Google Scholar] [CrossRef] [PubMed]
- Doganis, D.; Karalexi, M.A.; group N.-S.c.; Panagopoulou, P.; Bouka, P.; Bouka, E.; et al. Incidence patterns of childhood non-Wilms renal tumors: Comparing data of the Nationwide Registry of Childhood Hematological Malignancies and Solid Tumors (NARECHEM-ST), Greece, and the Surveillance, Epidemiology, and End Results Program (SEER), USA. Cancer Epidemiol. 2022, 78, 102153. [Google Scholar] [CrossRef] [PubMed]
- Groenendijk, A.; Spreafico, F.; de Krijger, R.R.; Drost, J.; Brok, J.; Perotti, D.; et al. Prognostic Factors for Wilms Tumor Recurrence: A Review of the Literature. Cancers (Basel). 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Dome, J.S.; Graf, N.; Geller, J.I.; Fernandez, C.V.; Mullen, E.A.; Spreafico, F.; et al. Advances in Wilms Tumor Treatment and Biology: Progress Through International Collaboration. J Clin Oncol. 2015, 33, 2999–3007. [Google Scholar] [CrossRef] [PubMed]
- Hontecillas-Prieto, L.; Garcia-Dominguez, D.J.; Vaca, D.P.; Garcia-Mejias, R.; Marcilla, D.; Ramirez-Villar, G.L.; et al. Multidrug resistance transporter profile reveals MDR3 as a marker for stratification of blastemal Wilms tumour patients. Oncotarget. 2017, 8, 11173–11186. [Google Scholar] [CrossRef]
- Sangkhathat, S.; Kanngurn, S.; Chaiyapan, W.; Gridist, P.; Maneechay, W. Wilms' tumor 1 gene (WT1) is overexpressed and provides an oncogenic function in pediatric nephroblastomas harboring the wild-type WT1. Oncol Lett. 2010, 1, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Brok, J.; Treger, T.D.; Gooskens, S.L.; van den Heuvel-Eibrink, M.M.; Pritchard-Jones, K. Biology and treatment of renal tumours in childhood. Eur J Cancer. 2016, 68, 179–195. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, L.C.; De Souza, K.R.; De, O.R.A.H.; Andrade, R.C.; Britto, A.C., Jr.; De Lima, M.A.; et al. WT1, WTX and CTNNB1 mutation analysis in 43 patients with sporadic Wilms' tumor. Oncol Rep. 2013, 29, 315–320. [Google Scholar] [CrossRef]
- Rakheja, D.; Chen, K.S.; Liu, Y.; Shukla, A.A.; Schmid, V.; Chang, T.C.; et al. Somatic mutations in DROSHA and DICER1 impair microRNA biogenesis through distinct mechanisms in Wilms tumours. Nat Commun. 2014, 2, 4802. [Google Scholar] [CrossRef]
- Perlman, E.J.; Gadd, S.; Arold, S.T.; Radhakrishnan, A.; Gerhard, D.S.; Jennings, L.; et al. MLLT1 YEATS domain mutations in clinically distinctive Favourable Histology Wilms tumours. Nat Commun. 2015, 6, 10013. [Google Scholar] [CrossRef]
- Walz, A.L.; Ooms, A.; Gadd, S.; Gerhard, D.S.; Smith, M.A.; Guidry Auvil, J.M.; et al. Recurrent DGCR8, DROSHA, and SIX homeodomain mutations in favorable histology Wilms tumors. Cancer Cell. 2015, 27, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Aungkawattanapong, N.; Techavichit, P.; Lauhasurayotin, S.; Chiengthong, K.; Sosothikul, D.; Monsereenusorn, C.; et al. A study of the TP53 Germline Mutation and Clinicopathologic Features in Thai Children with Adrenocortical Carcinoma. J Health Sci Med Res. 2021, 39, 491–502. [Google Scholar] [CrossRef]
- Mahamdallie, S.; Yost, S.; Poyastro-Pearson, E.; Holt, E.; Zachariou, A.; Seal, S.; et al. Identification of new Wilms tumour predisposition genes: an exome sequencing study. Lancet Child Adolesc Health. 2019, 3, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics. 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed]
- Van der Auwera, G.A.; Carneiro, M.O.; Hartl, C.; Poplin, R.; Del Angel, G.; Levy-Moonshine, A.; et al. From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr Protoc Bioinformatics. 2013, 43, 11 0 1- 0 33. [Google Scholar] [CrossRef]
- Mayakonda, A.; Lin, D.C.; Assenov, Y.; Plass, C.; Koeffler, H.P. Maftools: efficient and comprehensive analysis of somatic variants in cancer. Genome Res. 2018, 28, 1747–1756. [Google Scholar] [CrossRef]
- Group, T.T.P.O. National protocol for the treatment of childhood cancers, 1 ed.; M Print Corporation co.,ltd.: Bangkok, January 2016. [Google Scholar]
- Petrov, I.; Alexeyenko, A. Individualized discovery of rare cancer drivers in global network context. Elife 2022, 11. [Google Scholar] [CrossRef]
- Ostroverkhova, D.; Przytycka, T.M.; Panchenko, A.R. Cancer driver mutations: predictions and reality. Trends Mol Med. 2023, 29, 554–566. [Google Scholar] [CrossRef]
- Tamborero, D.; Rubio-Perez, C.; Deu-Pons, J.; Schroeder, M.P.; Vivancos, A.; Rovira, A.; et al. Cancer Genome Interpreter annotates the biological and clinical relevance of tumor alterations. Genome Med. 2018, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Liu, Y.; Liu, Y.; Alexandrov, L.B.; Edmonson, M.N.; Gawad, C.; et al. Pan-cancer genome and transcriptome analyses of 1,699 paediatric leukaemias and solid tumours. Nature. 2018, 555, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Klco, J.; Thomas, M., 3rd; Qi, W.; Walsh, M.; Ma, J.; Westover, T.; et al. Functional Characterization of Cooperating MGA Mutations in RUNX1::RUNX1T1 Acute Myeloid Leukemia. Res Sq. 2023. [Google Scholar]
- Chen, X.; Zhang, G.; Chen, B.; Wang, Y.; Guo, L.; Cao, L.; et al. Association between histone lysine methyltransferase KMT2C mutation and clinicopathological factors in breast cancer. Biomed Pharmacother. 2019, 116, 108997. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhong, A.; Zhang, S.; Chen, M.; Zhang, L.; Hang, X.; et al. KMT2D Deficiency Promotes Myeloid Leukemias which Is Vulnerable to Ribosome Biogenesis Inhibition. Adv Sci (Weinh). 2023, 10, e2206098. [Google Scholar] [CrossRef] [PubMed]
- Schweigert, A.; Fischer, C.; Mayr, D.; von Schweinitz, D.; Kappler, R.; Hubertus, J. Activation of the Wnt/beta-catenin pathway is common in wilms tumor, but rarely through beta-catenin mutation and APC promoter methylation. Pediatr Surg Int. 2016, 32, 1141–1146. [Google Scholar] [CrossRef]
- Fleming-de-Moraes, C.D.; Rocha, M.R.; Tessmann, J.W.; de Araujo, W.M.; Morgado-Diaz, J.A. Crosstalk between PI3K/Akt and Wnt/beta-catenin pathways promote colorectal cancer progression regardless of mutational status. Cancer Biol Ther. 2022, 23, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Langhammer, T.-S.; Roolf, C.; Krohn, S.; Kretzschmar, C.; Huebner, R.; Rolfs, A.; et al. PI3K/Akt Signaling Interacts With Wnt/β-Catenin Signaling But Does Not Induce An Accumulation Of β-Catenin In The Nucleus Of Acute Lymphoblastic Leukemia Cell Lines. Blood. 2013, 122, 4886. [Google Scholar] [CrossRef]
- Bitaraf, M.; Mahmanzar, M.; Zafari, N.; Mohammadpour, H.; Vasei, M.; Moradi Matin, L.; et al. The potential key genes and pathways associated with Wilms tumor in quest of proper candidates for diagnostic and therapeutic purposes. Sci Rep. 2022, 12, 17906. [Google Scholar] [CrossRef]
- Jimenez Martin, O.; Schlosser, A.; Furtwangler, R.; Wegert, J.; Gessler, M. MYCN and MAX alterations in Wilms tumor and identification of novel N-MYC interaction partners as biomarker candidates. Cancer Cell Int. 2021, 21, 555. [Google Scholar] [CrossRef]
- Zhou, B.; Lin, W.; Long, Y.; Yang, Y.; Zhang, H.; Wu, K.; et al. Notch signaling pathway: architecture, disease, and therapeutics. Signal Transduct Target Ther. 2022, 7, 95. [Google Scholar] [CrossRef]
- Ney, G.M.; McKay, L.; Koschmann, C.; Mody, R.; Li, Q. The Emerging Role of Ras Pathway Signaling in Pediatric Cancer. Cancer Res. 2020, 80, 5155–5163. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.; Sadeghi, S.; Tabatabaeian, H. Battling Chemoresistance in Cancer: Root Causes and Strategies to Uproot Them. Int J Mol Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Zhi, X.; Tao, J.; Xie, K.; Zhu, Y.; Li, Z.; Tang, J.; et al. MUC4-induced nuclear translocation of beta-catenin: a novel mechanism for growth, metastasis and angiogenesis in pancreatic cancer. Cancer Lett. 2014, 346, 104–113. [Google Scholar] [CrossRef]
- Gao, L.; Liu, J.; Zhang, B.; Zhang, H.; Wang, D.; Zhang, T.; et al. Functional MUC4 suppress epithelial-mesenchymal transition in lung adenocarcinoma metastasis. Tumour Biol. 2014, 35, 1335–1341. [Google Scholar] [CrossRef]
- Giannakouros, P.; Comamala, M.; Matte, I.; Rancourt, C.; Piche, A. MUC16 mucin (CA125) regulates the formation of multicellular aggregates by altering beta-catenin signaling. Am J Cancer Res. 2015, 5, 219–230. [Google Scholar] [PubMed]
- Pai, P.; Rachagani, S.; Dhawan, P.; Sheinin, Y.M.; Macha, M.A.; Qazi, A.K.; et al. MUC4 is negatively regulated through the Wnt/beta-catenin pathway via the Notch effector Hath1 in colorectal cancer. Genes Cancer. 2016, 7, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Waddell, N.; Pajic, M.; Patch, A.M.; Chang, D.K.; Kassahn, K.S.; Bailey, P.; et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature. 2015, 518, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Alsop, K.; Fereday, S.; Meldrum, C.; deFazio, A.; Emmanuel, C.; George, J.; et al. BRCA mutation frequency and patterns of treatment response in BRCA mutation-positive women with ovarian cancer: a report from the Australian Ovarian Cancer Study Group. J Clin Oncol. 2012, 30, 2654–2663. [Google Scholar] [CrossRef]
- Dann, R.B.; DeLoia, J.A.; Timms, K.M.; Zorn, K.K.; Potter, J.; Flake D.D., 2nd; et al. BRCA1/2 mutations and expression: response to platinum chemotherapy in patients with advanced stage epithelial ovarian cancer. Gynecol Oncol. 2012, 125, 677–682. [Google Scholar] [CrossRef]
- Ceccaldi, R.; O'Connor, K.W.; Mouw, K.W.; Li, A.Y.; Matulonis, U.A.; D'Andrea, A.D.; et al. A unique subset of epithelial ovarian cancers with platinum sensitivity and PARP inhibitor resistance. Cancer Res. 2015, 75, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Litton, J.K.; Rugo, H.S.; Ettl, J.; Hurvitz, S.A.; Goncalves, A.; Lee, K.H.; et al. Talazoparib in Patients with Advanced Breast Cancer and a Germline BRCA Mutation. N Engl J Med. 2018, 379, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Gradishar, W.J.; Moran, M.S.; Abraham, J.; Aft, R.; Agnese, D.; Allison, K.H.; et al. Breast Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022, 20, 691–722. [Google Scholar]
- Moore, K.N.; Secord, A.A.; Geller, M.A.; Miller, D.S.; Cloven, N.; Fleming, G.F.; et al. Niraparib monotherapy for late-line treatment of ovarian cancer (QUADRA): a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019, 20, 636–648. [Google Scholar] [CrossRef] [PubMed]
- Mateo, J.; Carreira, S.; Sandhu, S.; Miranda, S.; Mossop, H.; Perez-Lopez, R.; et al. DNA-Repair Defects and Olaparib in Metastatic Prostate Cancer. N Engl J Med. 2015, 373, 1697–1708. [Google Scholar] [CrossRef] [PubMed]
- Abida, W.; Patnaik, A.; Campbell, D.; Shapiro, J.; Bryce, A.H.; McDermott, R.; et al. Rucaparib in Men With Metastatic Castration-Resistant Prostate Cancer Harboring a BRCA1 or BRCA2 Gene Alteration. J Clin Oncol. 2020, 38, 3763–3772. [Google Scholar] [CrossRef]
- de Bono, J.; Mateo, J.; Fizazi, K.; Saad, F.; Shore, N.; Sandhu, S.; et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2020, 382, 2091–2102. [Google Scholar] [CrossRef]
- Patel, J.P.; Gonen, M.; Figueroa, M.E.; Fernandez, H.; Sun, Z.; Racevskis, J.; et al. Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N Engl J Med. 2012, 366, 1079–1089. [Google Scholar] [CrossRef]
- Villarroel, M.C.; Rajeshkumar, N.V.; Garrido-Laguna, I.; De Jesus-Acosta, A.; Jones, S.; Maitra, A.; et al. Personalizing cancer treatment in the age of global genomic analyses: PALB2 gene mutations and the response to DNA damaging agents in pancreatic cancer. Mol Cancer Ther. 2011, 10, 3–8. [Google Scholar] [CrossRef]
- Cowin, P.A.; George, J.; Fereday, S.; Loehrer, E.; Van Loo, P.; Cullinane, C.; et al. LRP1B deletion in high-grade serous ovarian cancers is associated with acquired chemotherapy resistance to liposomal doxorubicin. Cancer Res. 2012, 72, 4060–4073. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, W.; Howard, T.P.; Vazquez, F.; Tsherniak, A.; Wu, J.N.; et al. SWI/SNF-mutant cancers depend on catalytic and non-catalytic activity of EZH2. Nat Med. 2015, 21, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Iorio, F.; Knijnenburg, T.A.; Vis, D.J.; Bignell, G.R.; Menden, M.P.; Schubert, M.; et al. A Landscape of Pharmacogenomic Interactions in Cancer. Cell. 2016, 166, 740–754. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).