Submitted:
29 April 2024
Posted:
29 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
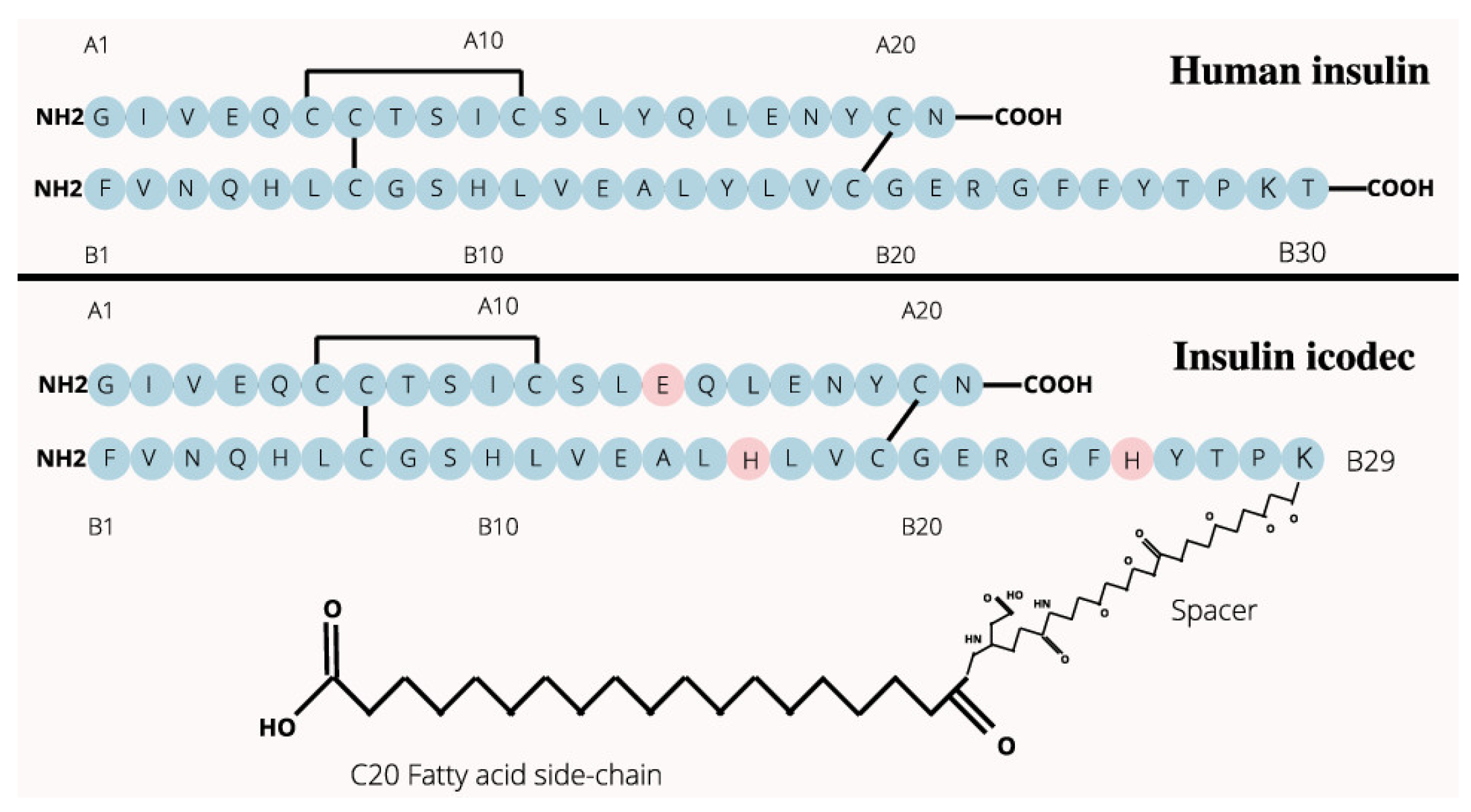
- insulin icodec is able to form aggregates or clusters at the subcutaneous injection site, gradually releasing into the bloodstream over an extended period.
- insulin icodec undergoes structural modifications that increases its stability and solubility and preventing enzyme-mediated degradation and rapid clearance [34].
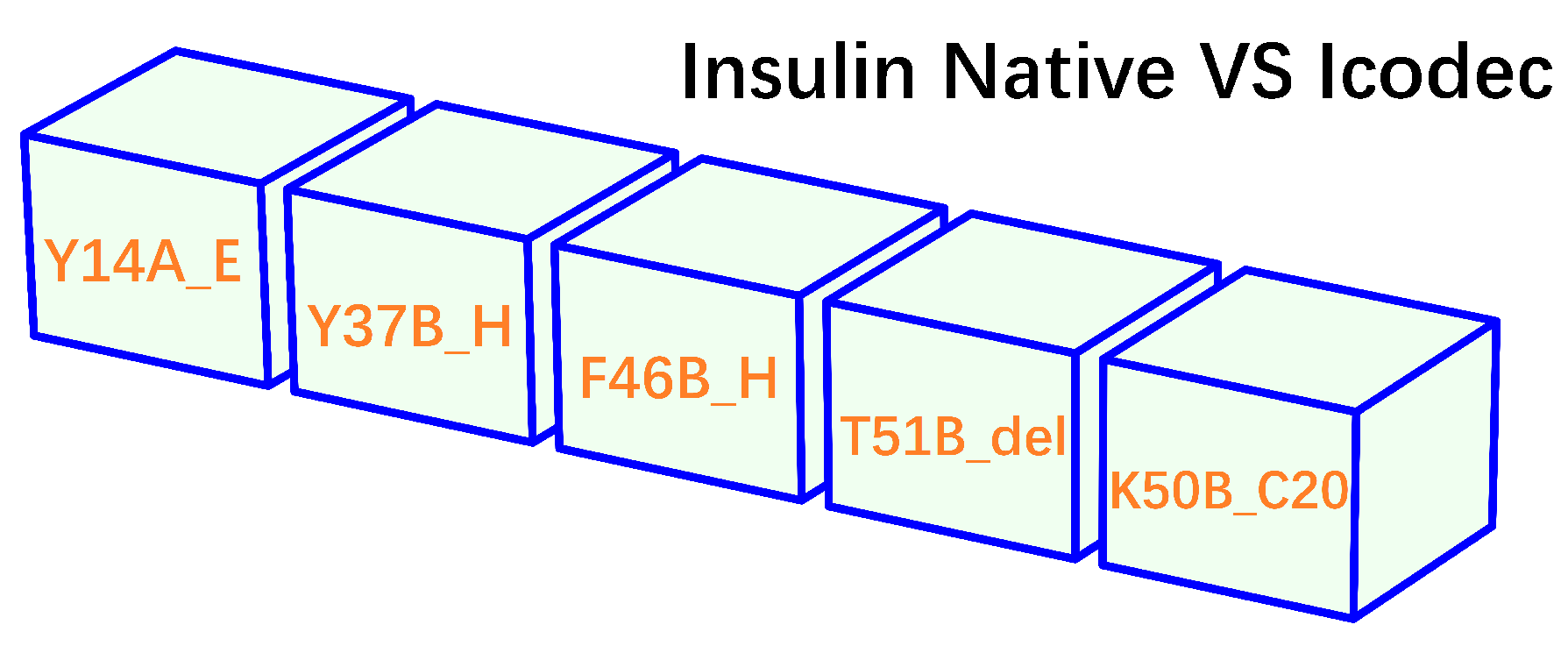
- insulin icodec is conjugated with a fatty acid at position B30 (K50B_C20, Figure 2). After injection, the fatty acid chain in insulin icodec interacts with albumin in the subcutaneous tissue, forming reversible albumin-insulin complexes. These complexes act as a reservoir, gradually releasing insulin icodec into the bloodstream, increasing its fat solubility and allowing it to bind to fatty acid-binding proteins, forming a depot of the insulin icodec reversibly bound to albumin.
- the incorporation of fatty acid chains facilitate the formation of stable hexameric structures, thereby delaying insulin absorption and promoting sustained release.
- two missense mutations of insulin icodec (Y37B_H and F46B_H) contributed to its prolonged duration of action through the induction of a modest decrease in the binding affinity of insulin icodec and its receptor (IR) [14]. Specifically, it is entirely due to the site-directed mutation Y37B_H that the salt bridge at the binding interface between insulin icodec and IR becomes weaker (from 3.204 Å to 3.669 Å), but is still not disrupted by the site-directed mutation Y37B_H, such that the binding affinity of ligand-receptor is lowered but not eliminated by the site-directed mutation Y37B_H [14], and thereby ensuring downstream signal transduction for the prolonged blood glucose-lowering effect of insulin icodec.
2. Motivation
3. Materials and Methods
4. Results
- the Prodigy-calculated Kd of the insulin analogue to its receptor (AE) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the first engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (AG) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the second engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (AI) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the third engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (AK) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the fourth engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (CE) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the first engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (CG) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the second engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (CI) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the third engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
- the Prodigy-calculated Kd of the insulin analogue to its receptor (CK) is larger than that of native insulin or insulin icodec or both, i.e., the binding affinity of the fourth engineered insulin analogue to its receptor is lower that that of native insulin or insulin icodec or both.
5. Conclusion and Discussion
6. Ethical Statement
7. Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
Author Contributions
Funding
Conflicts of Interest
References
- Bajaj, H.S.; Bergenstal, R.M.; Christoffersen, A.; Davies, M.J.; Gowda, A.; Isendahl, J.; Lingvay, I.; Senior, P.A.; Silver, R.J.; Trevisan, R.; Rosenstock, J. Switching to Once-Weekly Insulin Icodec Versus Once-Daily Insulin Glargine U100 in Type 2 Diabetes Inadequately Controlled on Daily Basal Insulin: A Phase 2 Randomized Controlled Trial. Diabetes Care 2021, 44, 1586–1594. [Google Scholar] [CrossRef] [PubMed]
- Wick, J.Y. Insulin: Almost a Century of Lifesaving. The Consultant Pharmacist 2017, 32, 190–198. [Google Scholar] [CrossRef]
- Griffin, T.P.; Dinneen, S.F. In T2DM, weekly insulin icodec did not differ from daily glargine for reducing HbA1c or significant/severe hypoglycemia. Annals of Internal Medicine 2021, 174, JC34. [Google Scholar] [CrossRef]
- Philis-Tsimikas, A.; Bajaj, H.S.; Begtrup, K.; Cailleteau, R.; Gowda, A.; Lingvay, I.; Mathieu, C.; Russell-Jones, D.; Rosenstock, J. Rationale and design of the phase 3a development programme (ONWARDS 1–6 trials) investigating once-weekly insulin icodec in diabetes. Diabetes, Obesity and Metabolism 2022, 25, 331–341. [Google Scholar] [CrossRef]
- Philis-Tsimikas, A.; Asong, M.; Franek, E.; Jia, T.; Rosenstock, J.; Stachlewska, K.; Watada, H.; Kellerer, M. Switching to once-weekly insulin icodec versus once-daily insulin degludec in individuals with basal insulin-treated type 2 diabetes (ONWARDS 2): a phase 3a, randomised, open label, multicentre, treat-to-target trial. The Lancet Diabetes & Endocrinology 2023, 11, 414–425. [Google Scholar]
- Mathieu, C.; Ásbjörnsdóttir, B.; Bajaj, H.S.; Lane, W.; Matos, A.L.S.A.; Murthy, S.; Stachlewska, K.; Rosenstock, J. Switching to once-weekly insulin icodec versus once-daily insulin glargine U100 in individuals with basal-bolus insulin-treated type 2 diabetes (ONWARDS 4): a phase 3a, randomised, open-label, multicentre, treat-to-target, non-inferiority trial. The Lancet 2023, 401, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Lingvay, I.; Asong, M.; Desouza, C.; Gourdy, P.; Kar, S.; Vianna, A.; Vilsbøll, T.; Vinther, S.; Mu, Y. Once-Weekly Insulin Icodec vs Once-Daily Insulin Degludec in Adults With Insulin-Naive Type 2 Diabetes. JAMA 2023, 330, 228. [Google Scholar] [CrossRef]
- Lingvay, I.; Buse, J.B.; Franek, E.; Hansen, M.V.; Koefoed, M.M.; Mathieu, C.; Pettus, J.; Stachlewska, K.; Rosenstock, J. A Randomized, Open-Label Comparison of Once-Weekly Insulin Icodec Titration Strategies Versus Once-Daily Insulin Glargine U100. Diabetes Care 2021, 44, 1595–1603. [Google Scholar] [CrossRef]
- Kjeldsen, T.B.; Hubálek, F.; Hjørringgaard, C.U.; Tagmose, T.M.; Nishimura, E.; Stidsen, C.E.; Porsgaard, T.; Fledelius, C.; Refsgaard, H.H.F.; Gram-Nielsen, S.; Naver, H.; Pridal, L.; Hoeg-Jensen, T.; Jeppesen, C.B.; Manfè, V.; Ludvigsen, S.; Lautrup-Larsen, I.; Madsen, P. Molecular Engineering of Insulin Icodec, the First Acylated Insulin Analog for Once-Weekly Administration in Humans. Journal of Medicinal Chemistry 2021, 64, 8942–8950. [Google Scholar] [CrossRef]
- Rosenstock, J.; Bajaj, H.S.; Janež, A.; Silver, R.; Begtrup, K.; Hansen, M.V.; Jia, T.; Goldenberg, R. Once-Weekly Insulin for Type 2 Diabetes without Previous Insulin Treatment. New England Journal of Medicine 2020, 383, 2107–2116. [Google Scholar] [CrossRef]
- Bajaj, H.S.; Goldenberg, R.M. Insulin Icodec Weekly: A Basal Insulin Analogue for Type 2 Diabetes. European Endocrinology 2023, 19, 4. [Google Scholar] [CrossRef] [PubMed]
- DiMarchi, R.D.; Mayer, J.P. Icodec Advances the Prospect of Once-Weekly Insulin Injection. Journal of Medicinal Chemistry 2021, 64, 8939–8941. [Google Scholar] [CrossRef]
- Rosenstock, J.; Prato, S.D. Basal weekly insulins: the way of the future! Metabolism 2022, 126, 154924. [Google Scholar] [CrossRef]
- Li, W. How Structural Modifications of Insulin Icodec Contributes to Its Prolonged Duration of Action: A Structural and Biophysical Perspective 2023. [CrossRef]
- Kalra, S.; Bhattacharya, S.; Kapoor, N. Contemporary Classification of Glucagon-Like Peptide 1 Receptor Agonists (GLP1RAs). Diabetes Therapy 2021, 12, 2133–2147. [Google Scholar] [CrossRef]
- Pratley, R.; Amod, A.; Hoff, S.T.; Kadowaki, T.; Lingvay, I.; Nauck, M.; Pedersen, K.B.; Saugstrup, T.; Meier, J.J. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomised, double-blind, phase 3a trial. The Lancet 2019, 394, 39–50. [Google Scholar] [CrossRef]
- Anderson, S.L.; Beutel, T.R.; Trujillo, J.M. Oral semaglutide in type 2 diabetes. Journal of Diabetes and its Complications 2020, 34, 107520. [Google Scholar] [CrossRef] [PubMed]
- Li, W. Strengthening Semaglutide-GLP-1R Binding Affinity via a Val27-Arg28 Exchange in the Peptide Backbone of Semaglutide: A Computational Structural Approach. Journal of Computational Biophysics and Chemistry 2021, 20, 495–499. [Google Scholar] [CrossRef]
- Nadkarni, P.; Chepurny, O.G.; Holz, G.G. Regulation of Glucose Homeostasis by GLP-1. In Progress in Molecular Biology and Translational Science; Elsevier, 2014; pp. 23–65.
- Lau, J.; Bloch, P.; Schäffer, L.; Pettersson, I.; Spetzler, J.; Kofoed, J.; Madsen, K.; Knudsen, L.B.; McGuire, J.; Steensgaard, D.B.; Strauss, H.M.; Gram, D.X.; Knudsen, S.M.; Nielsen, F.S.; Thygesen, P.; Reedtz-Runge, S.; Kruse, T. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry 2015, 58, 7370–7380. [Google Scholar] [CrossRef]
- Gabery, S.; Salinas, C.G.; Paulsen, S.J.; Ahnfelt-Rønne, J.; Alanentalo, T.; Baquero, A.F.; Buckley, S.T.; Farkas, E.; Fekete, C.; Frederiksen, K.S.; Helms, H.C.C.; Jeppesen, J.F.; John, L.M.; Pyke, C.; Nøhr, J.; Lu, T.T.; Polex-Wolf, J.; Prevot, V.; Raun, K.; Simonsen, L.; Sun, G.; Szilvásy-Szabó, A.; Willenbrock, H.; Secher, A.; Knudsen, L.B. Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight 2020, 5. [Google Scholar] [CrossRef]
- Li, W. High-Throughput Extraction of Interfacial Electrostatic Features from GLP-1-GLP-1R Complex Structures: A GLP-1-GLP-1R-Based Mini GIBAC Perspective 2024. [CrossRef]
- Pieber, T.R.; Arfelt, K.N.; Cailleteau, R.; Hart, M.; Kar, S.; Mursic, I.; Svehlikova, E.; Urschitz, M.; Haahr, H. Hypoglycaemia frequency and physiological response after double or triple doses of once-weekly insulin icodec vs once-daily insulin glargine U100 in type 2 diabetes: a randomised crossover trial. Diabetologia 2023, 66, 1413–1430. [Google Scholar] [CrossRef]
- Zerihun, K.; Mhanna, M.; Ayesh, H.; Ghazaleh, S.; Khader, Y.; Beran, A.; Aldhafeeri, A.; Sharma, S.; Iqbal, A.; Legesse, H.; Jaume, J. Efficacy and Safety of Insulin Icodec Versus Glargine U100: A Meta-Analysis of Randomized Controlled Trials. American Journal of Therapeutics 2022, 30, e480–e483. [Google Scholar] [CrossRef]
- Li, W. Structural and Functional Consequences of the SMA-Linked Missense Mutations of the Survival Motor Neuron Protein: A Brief Update. In Novel Aspects on Motor Neuron Disease; IntechOpen, 2019.
- Li, W. How do SMA-linked mutations of SMN1 lead to structural/functional deficiency of the SMA protein? PLOS ONE 2017, 12, e0178519. [Google Scholar] [CrossRef] [PubMed]
- Plum-Mörschel, L.; Andersen, L.R.; Hansen, S.; Hövelmann, U.; Krawietz, P.; Kristensen, N.R.; Lehrskov, L.L.; Haahr, H. Pharmacokinetic and Pharmacodynamic Characteristics of Insulin Icodec After Subcutaneous Administration in the Thigh, Abdomen or Upper Arm in Individuals with Type 2 Diabetes Mellitus. Clinical Drug Investigation 2023, 43, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Belal, H.; Gandhi, G.Y. In uncontrolled T2DM treated with a basal-bolus insulin regimen, weekly icodec was noninferior to daily glargine for HbA1c at 26 wk. Annals of Internal Medicine 2023, 176, JC94. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.L.; Bassetti, M.; Mangoni, A.A. Drugs in Context Editorial: Review of 2020 and what lies ahead in therapeutic interventions. Drugs in Context 2021, 10, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bellary, S.; Barnett, A.H. Insulin icodec: evolution or revolution in diabetes therapy? The Lancet Diabetes & Endocrinology 2023, 11, 379–380. [Google Scholar]
- e Silva, R.R.; de Miranda Gauza, M.; Guisso, M.E.S.; da Silva, J.O.N.; Kohara, S.K. Once-Weekly Insulin Icodec vs. Once-Daily Insulin Glargine U100 for type 2 diabetes: a systematic review and meta-analysis of phase 2 randomized controlled trials. Archives of Endocrinology and Metabolism 2023, 67. [Google Scholar] [CrossRef] [PubMed]
- Pieber, T.R.; Asong, M.; Fluhr, G.; Höller, V.; Kristensen, N.R.; Larsen, J.H.; Ribel-Madsen, R.; Svehlikova, E.; Vinther, S.; Voortman, M.; Haahr, H. Pharmacokinetic and pharmacodynamic properties of once-weekly insulin icodec in individuals with type 2 diabetes. Diabetes, Obesity and Metabolism, 2023. [Google Scholar]
- Russell-Jones, D.; Babazono, T.; Cailleteau, R.; Engberg, S.; Irace, C.; Kjaersgaard, M.I.S.; Mathieu, C.; Rosenstock, J.; Woo, V.; Klonoff, D.C. Once-weekly insulin icodec versus once-daily insulin degludec as part of a basal-bolus regimen in individuals with type 1 diabetes (ONWARDS 6): a phase 3a, randomised, open-label, treat-to-target trial. The Lancet 2023. [Google Scholar] [CrossRef] [PubMed]
- Feher, J. Digestion and Absorption of the Macronutrients. In Quantitative Human Physiology; Elsevier, 2017; pp. 821–833. [CrossRef]
- Ericsson, Å.; Fridhammar, A. Cost-effectiveness of once-weekly semaglutide versus dulaglutide and lixisenatide in patients with type 2 diabetes with inadequate glycemic control in Sweden. Journal of Medical Economics 2019, 22, 997–1005. [Google Scholar] [CrossRef]
- Han, J.; Fu, J.; Yang, Q.; Zhou, F.; Chen, X.; Li, C.; Yin, J. Rational design and biological evaluation of gemfibrozil modified Xenopus GLP-1 derivatives as long-acting hypoglycemic agents. European Journal of Medicinal Chemistry 2020, 198, 112389. [Google Scholar] [CrossRef]
- Li, W. Towards a General Intermolecular Binding Affinity Calculator 2022.
- Li, W.; Vottevor, G. Towards a Truly General Intermolecular Binding Affinity Calculator for Drug Discovery & Design 2023. [CrossRef]
- Pharmaceuticals, R. Recursion Bridges the Protein and Chemical Space with Massive Protein-Ligand Interaction Predictions Spanning 36 Billion Compounds, 2023. Accessed: (, 2023). 1 September.
- Reymond, J.L.; van Deursen, R.; Blum, L.C.; Ruddigkeit, L. Chemical space as a source for new drugs. MedChemComm 2010, 1, 30. [Google Scholar] [CrossRef]
- Berman, H.; Henrick, K.; Nakamura, H. Announcing the worldwide Protein Data Bank. Nature Structural & Molecular Biology 2003, 10, 980–980. [Google Scholar]
- Li, W. Visualising the Experimentally Uncharted Territories of Membrane Protein Structures inside Protein Data Bank 2020.
- Li, W. Half-a-century Burial of ρ, θ and φ in PDB 2021.
- Gutmann, T.; Schäfer, I.B.; Poojari, C.; Brankatschk, B.; Vattulainen, I.; Strauss, M.; Ünal Coskun. Cryo-EM structure of the complete and ligand-saturated insulin receptor ectodomain. Journal of Cell Biology 2019, 219. [Google Scholar] [CrossRef]
- DeLano, W.L. Pymol: An open-source molecular graphics tool. CCP4 Newsletter On Protein Crystallography 2002, 40, 82–92. [Google Scholar]
- Webb, B.; Sali, A. Protein Structure Modeling with MODELLER. In Methods in Molecular Biology; Springer US, 2020; pp. 239–255.
- Vangone, A.; Bonvin, A.M. Contacts-based prediction of binding affinity in protein-protein complexes. eLife 2015, 4. [Google Scholar] [CrossRef]
- Xue, L.C.; Rodrigues, J.P.; Kastritis, P.L.; Bonvin, A.M.; Vangone, A. PRODIGY: a web server for predicting the binding affinity of protein-protein complexes. Bioinformatics, 2016; btw514. [Google Scholar]
- Li, W. Gravity-driven pH adjustment for site-specific protein pKa measurement by solution-state NMR. Measurement Science and Technology 2017, 28, 127002. [Google Scholar] [CrossRef]
- Li, W. Calcium Channel Trafficking Blocker Gabapentin Bound to the -2–1 Subunit of Voltage-Gated Calcium Channel: A Computational Structural Investigation 2020.
- Li, W. Delving Deep into the Structural Aspects of the BPro28-BLys29 Exchange in Insulin Lispro: A Structural Biophysical Lesson 2020.
- Li, W. Extracting the Interfacial Electrostatic Features from Experimentally Determined Antigen and/or Antibody-Related Structures inside Protein Data Bank for Machine Learning-Based Antibody Design 2020.
| PDB ID | Structure Title (release date from newest to oldest) |
|---|---|
| 8DWN | Crystal structure of bis-phosphorylated insulin receptor kinase domain |
| 7YQ3 | human insulin receptor bound with A43 DNA aptamer and insulin |
| 7YQ4 | human insulin receptor bound with A62 DNA aptamer and insulin - locally refined |
| 7YQ5 | human insulin receptor bound with A62 DNA aptamer and insulin |
| 7YQ6 | human insulin receptor bound with A62 DNA aptamer |
| 8EYX | Cryo-EM structure of 4 insulins bound full-length mouse IR mutant with physically decoupled alpha CTs (C684S/C685S/C687S; denoted as IR-3CS) Asymmetric conformation 1 |
| 8EYY | Cryo-EM structure of 4 insulins bound full-length mouse IR mutant with physically decoupled alpha CTs (C684S/C685S/C687S, denoted as IR-3CS) Asymmetric conformation 2 |
| 8EZ0 | Cryo-EM structure of 4 insulins bound full-length mouse IR mutant with physically decoupled alpha CTs (C684S/C685S/C687S; denoted as IR-3CS) Symmetric conformation |
| 8GUY | human insulin receptor bound with two insulin molecules |
| 7U6D | Head region of insulin receptor ectodomain (A-isoform) bound to the non-insulin agonist IM459 |
| 7U6E | Head region of insulin receptor ectodomain (A-isoform) bound to the non-insulin agonist IM462 |
| 7PHT | Structure of Insulin receptor’s transmembrane domain |
| 8DTL | Cryo-EM structure of insulin receptor (IR) bound with S597 peptide |
| 8DTM | Cryo-EM structure of insulin receptor (IR) bound with S597 component 2 |
| 7S0Q | Head region of a complex of IGF-I with the ectodomain of a hybrid insulin receptor / type 1 insulin-like growth factor receptor |
| 7S8V | Leg region of a complex of IGF-I with the ectodomain of a hybrid insulin receptor / type 1 insulin-like growth factor receptor |
| 7SL1 | Full-length insulin receptor bound with site 1 binding deficient mutant insulin (A-V3E) |
| 7SL2 | Full-length insulin receptor bound with site 2 binding deficient mutant insulin (A-L13R) – asymmetric conformation |
| 7SL3 | Full-length insulin receptor bound with site 2 binding deficient mutant insulin (A-L13R) – symmetric conformation |
| 7SL4 | Full-length insulin receptor bound with site 2 binding deficient mutant insulin (B-L17R) – asymmetric conformation |
| 7SL6 | Full-length insulin receptor bound with site 2 binding deficient mutant insulin (B-L17R) – symmetric conformation |
| 7SL7 | Full-length insulin receptor bound with both site 1 binding deficient mutant insulin (A-V3E) and site 2 binding deficient mutant insulin (A-L13R) |
| 7STH | Full-length insulin receptor bound with unsaturated insulin WT (2 insulin bound) symmetric conformation |
| 7STI | Full-length insulin receptor bound with unsaturated insulin WT (1 insulin bound) asymmetric conformation |
| 7STJ | Full-length insulin receptor bound with unsaturated insulin WT (2 insulins bound) asymmetric conformation (Conformation 1) |
| 7STK | Full-length insulin receptor bound with unsaturated insulin WT (2 insulins bound) asymmetric conformation (Conformation 2) |
| 7MQO | The insulin receptor ectodomain in complex with a venom hybrid insulin analogue - "head" region |
| 7MQR | The insulin receptor ectodomain in complex with four venom hybrid insulins - symmetric conformation |
| 7MQS | The insulin receptor ectodomain in complex with three venom hybrid insulin molecules - asymmetric conformation |
| 7MD4 | Insulin receptor ectodomain dimer complexed with two IRPA-3 partial agonists |
| 7MD5 | Insulin receptor ectodomain dimer complexed with two IRPA-9 partial agonists |
| 7PG0 | Low resolution Cryo-EM structure of full-length insulin receptor bound to 3 insulin with visible ddm micelle, conf 1 |
| 7PG2 | Low resolution Cryo-EM structure of full-length insulin receptor bound to 3 insulin, conf 1 |
| 7PG3 | Low resolution Cryo-EM structure of the full-length insulin receptor bound to 3 insulin, conf 2 |
| 7PG4 | Low resolution Cryo-EM structure of the full-length insulin receptor bound to 2 insulin, conf 3 |
| 7QID | tentative model of the human insulin receptor ectodomain bound by three insulin |
| 7KD6 | Insulin Receptor L1-CR plus alphaCT fragment in co-complex with Fv 83-7 and single-chain insulin SCI-b |
| 7BW7 | Cryo-EM Structure for the Ectodomain of the Full-length Human Insulin Receptor in Complex with 1 Insulin. |
| 7BW8 | Cryo-EM Structure for the Insulin Binding Region in the Ectodomain of the Full-length Human Insulin Receptor in Complex with 1 Insulin |
| 7BWA | Cryo-EM Structure for the Ectodomain of the Full-length Human Insulin Receptor in Complex with 2 Insulin |
| 6VEP | Human insulin in complex with the human insulin microreceptor in turn in complex with Fv 83-7 |
| 6VEQ | Con-Ins G1 in complex with the human insulin microreceptor in turn in complex with Fv 83-7 |
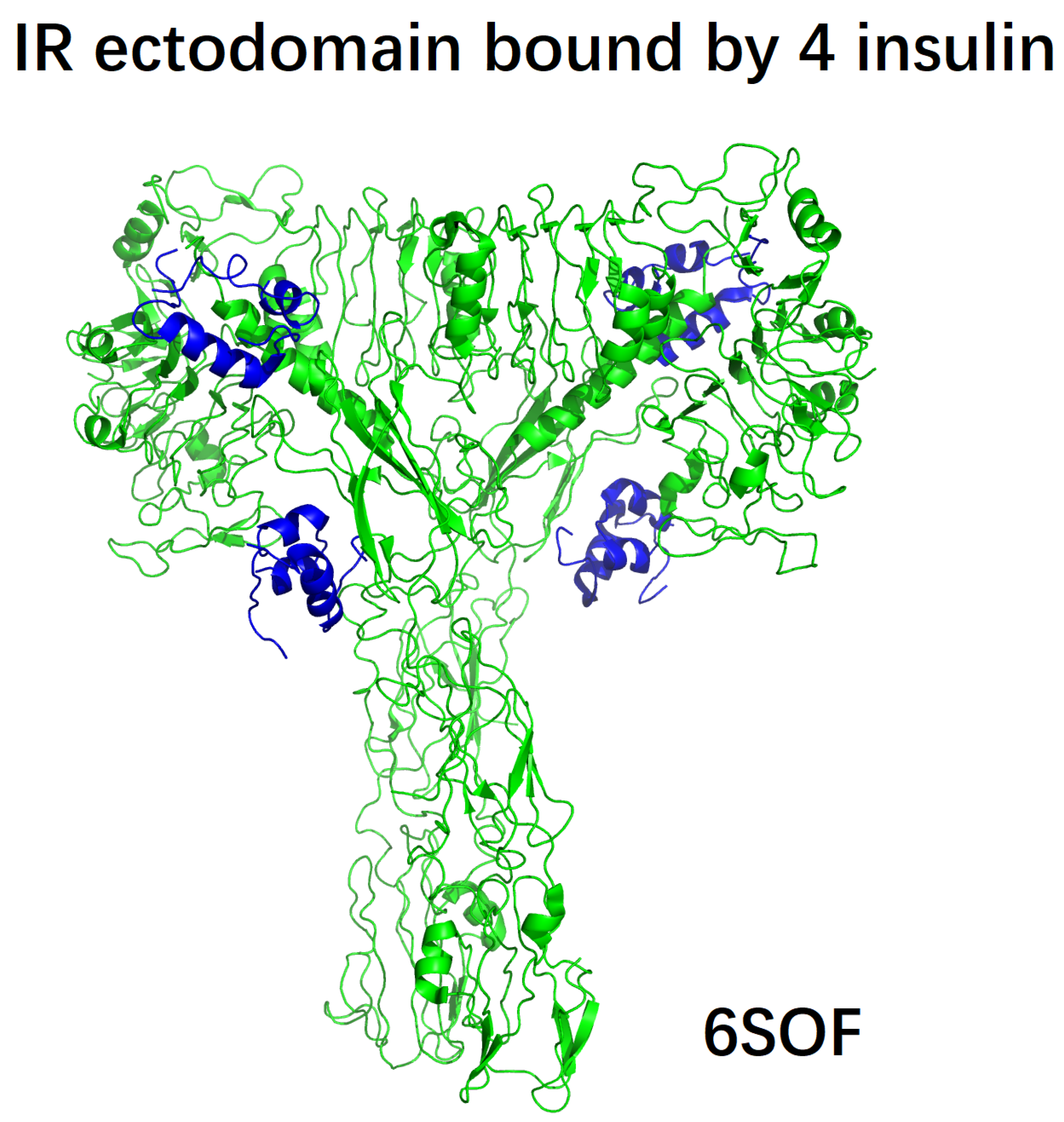
| 6SOF | human insulin receptor ectodomain bound by 4 insulin |
| 6PXV | Cryo-EM structure of full-length insulin receptor bound to 4 insulin. 3D refinement was focused on the extracellular region. |
| 6PXW | Cryo-EM structure of full-length insulin receptor bound to 4 insulin. 3D refinement was focused on the top part of the receptor complex. |
| 6HN4 | Leucine-zippered human insulin receptor ectodomain with single bound insulin - "lower" membrane-proximal part |
| 6HN5 | Leucine-zippered human insulin receptor ectodomain with single bound insulin - "upper" membrane-distal part |
| 6CE7 | Insulin Receptor ectodomain in complex with one insulin molecule |
| 6CE9 | Insulin Receptor ectodomain in complex with two insulin molecules |
| 6CEB | Insulin Receptor ectodomain in complex with two insulin molecules - C1 symmetry |
| 5U1M | Structure of the IRS-1 PTB Domain Bound to the Juxtamembrane Region of the Insulin Receptor |
| 5KQV | Insulin receptor ectodomain construct comprising domains L1,CR,L2, FnIII-1 and alphaCT peptide in complex with bovine insulin and FAB 83-14 (REVISED STRUCTURE) |
| 5TQ1 | Phospholipase C gamma-1 C-terminal SH2 domain bound to a phosphopeptide derived from the insulin receptor |
| 5J3H | Human insulin receptor domains L1-CR in complex with peptide S519C16 and 83-7 Fv |
| 5HHW | Crystal structure of insulin receptor kinase domain in complex with cis-(R)-7-(3-(azetidin-1-ylmethyl)cyclobutyl)-5-(3-((tetrahydro-2H-pyran-2-yl)methoxy)phenyl)-7H-pyrrolo[2,3-d]pyrimidin-4-amine |
| 4ZXB | Structure of the human insulin receptor ectodomain, IRDeltabeta construct, in complex with four Fab molecules |
| 5E1S | The Crystal structure of INSR Tyrosine Kinase in complex with the Inhibitor BI 885578 |
| 4XSS | Insulin-like growth factor I in complex with site 1 of a hybrid insulin receptor / Type 1 insulin-like growth factor receptor |
| 4XST | Structure of the endoglycosidase-H treated L1-CR domains of the human insulin receptor in complex with residues 697-719 of the human insulin receptor (A-isoform) |
| 4XLV | Crystal structure of the activated insulin receptor tyrosine kinase dimer |
| 4OGA | Insulin in complex with Site 1 of the human insulin receptor |
| 2MFR | Solution structure of the transmembrane domain of the insulin receptor in micelles |
| 4IBM | Crystal structure of insulin receptor kinase domain in complex with an inhibitor Irfin-1 |
| 3W11 | Insulin receptor ectodomain construct comprising domains L1-CR in complex with human insulin, Alpha-CT peptide(704-719) and FAB 83-7 |
| 3W12 | Insulin receptor ectodomain construct comprising domains L1-CR in complex with high-affinity insulin analogue [D-PRO-B26]-DTI-NH2, alpha-CT peptide(704-719) and FAB 83-7 |
| 3W13 | Insulin receptor ectodomain construct comprising domains L1-CR in complex with high-affinity insulin analogue [D-PRO-B26]-DTI-NH2, alphact peptide(693-719) and FAB 83-7 |
| 3ETA | Kinase domain of insulin receptor complexed with a pyrrolo pyridine inhibitor |
| 3EKN | Insulin receptor kinase complexed with an inhibitor |
| 3EKK | Insulin receptor kinase complexed with an inhibitor |
| 2Z8C | Phosphorylated insulin receptor tyrosine kinase in complex with (4-[5-carbamoyl-4-(3-methylanilino)pyrimidin-2-yl]aminophenyl)acetic acid |
| 3BU3 | Crystal structure of the insulin receptor kinase in complex with IRS2 KRLB peptide |
| 3BU5 | Crystal structure of the insulin receptor kinase in complex with IRS2 KRLB peptide and ATP |
| 3BU6 | Crystal structure of the insulin receptor kinase in complex with IRS2 KRLB phosphopeptide |
| 2HR7 | Insulin receptor (domains 1-3) |
| 2B4S | Crystal structure of a complex between PTP1B and the insulin receptor tyrosine kinase |
| 2AUH | Crystal structure of the Grb14 BPS region in complex with the insulin receptor tyrosine kinase |
| 1RQQ | Crystal Structure of the Insulin Receptor Kinase in Complex with the SH2 Domain of APS |
| 1LK2 | 1.35A crystal structure of H-2Kb complexed with the GNYSFYAL peptide |
| 1P14 | Crystal structure of a catalytic-loop mutant of the insulin receptor tyrosine kinase |
| 1I44 | CRYSTALLOGRAPHIC STUDIES OF AN ACTIVATION LOOP MUTANT OF THE INSULIN RECEPTOR TYROSINE KINASE |
| 1GAG | CRYSTAL STRUCTURE OF THE INSULIN RECEPTOR KINASE IN COMPLEX WITH A BISUBSTRATE INHIBITOR |
| 1IR3 | PHOSPHORYLATED INSULIN RECEPTOR TYROSINE KINASE IN COMPLEX WITH PEPTIDE SUBSTRATE AND ATP ANALOG |
| 1IRK | CRYSTAL STRUCTURE OF THE TYROSINE KINASE DOMAIN OF THE HUMAN INSULIN RECEPTOR |
| No. | Muta | AE | AG | AI | AK | CE | CG | CI | CK |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Y14A_E Y16B_H F25B_H | 2.404e-7 | 9.736e-6 | 5.827e-4 | 4.849e-6 | 7.038e-6 | 2.042e-8 | 7.222e-8 | 2.815e-4 |
| 1 | native insulin molecule | 3.279e-7 | 8.736e-6 | 5.946e-4 | 3.897e-6 | 7.431e-6 | 2.439e-8 | 6.379e-8 | 2.747e-4 |
| No. | Muta | AE | AG | AI | AK | CE | CG | CI | CK |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Y14A_T E34B_S H31B_S | 1.6e-6 | 2.2e-5 | 1.1e-3 | 1.4e-5 | 1.2e-5 | 5.9e-8 | 3.8e-7 | 3.0e-4 |
| 1 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 2 | H31B_Q Y14A_A H26B_Q | 4.4e-7 | 1.1e-5 | 4.3e-3 | 6.1e-6 | 9.7e-6 | 5.2e-8 | 5.0e-7 | 3.4e-4 |
| 2 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 5 | N18A_A Y47B_G H31B_S | 2.3e-6 | 1.7e-5 | 6.5e-4 | 9.2e-6 | 1.0e-5 | 9.2e-8 | 1.0e-7 | 3.6e-4 |
| 5 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 6 | E34B_A H26B_Q H31B_S | 5.4e-7 | 1.2e-5 | 1.4e-3 | 1.2e-5 | 9.6e-6 | 4.3e-8 | 5.3e-7 | 2.9e-4 |
| 6 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 7 | Y37B_P Y14A_A H31B_G | 5.7e-7 | 1.3e-5 | 8.9e-4 | 1.0e-5 | 1.1e-5 | 1.4e-7 | 1.6e-7 | 4.0e-4 |
| 7 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 15 | V23B_A H31B_G E34B_S | 9.1e-7 | 1.2e-5 | 7.1e-4 | 1.4e-5 | 1.2e-5 | 3.6e-8 | 2.2e-7 | 3.2e-4 |
| 15 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 17 | Y47B_G E34B_F E42B_T | 6.6e-7 | 2.2e-5 | 8.2e-4 | 5.6e-6 | 2.3e-5 | 5.8e-8 | 1.1e-7 | 3.2e-4 |
| 17 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 23 | N18A_A Y37B_P Y14A_M | 1.8e-6 | 1.4e-5 | 1.0e-3 | 7.1e-6 | 1.3e-5 | 3.7e-8 | 1.1e-7 | 2.9e-4 |
| 23 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 29 | Y14A_A K50B_A H31B_G | 4.2e-7 | 1.0e-5 | 1.1e-3 | 6.4e-6 | 8.0e-6 | 9.7e-8 | 2.7e-7 | 4.1e-4 |
| 29 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 30 | E42B_Q H26B_Q E34B_S | 4.5e-7 | 1.2e-5 | 1.0e-3 | 8.2e-6 | 1.8e-5 | 3.3e-8 | 3.4e-7 | 2.8e-4 |
| 30 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 31 | Y47B_G Y14A_A H31B_G | 4.1e-7 | 2.5e-5 | 7.4e-4 | 1.0e-5 | 1.3e-5 | 3.3e-8 | 2.6e-7 | 2.9e-4 |
| 31 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 33 | H31B_G E34B_S L32B_H | 6.3e-7 | 9.9e-6 | 8.7e-4 | 8.0e-6 | 1.1e-5 | 1.4e-7 | 1.1e-7 | 3.3e-4 |
| 33 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 35 | N18A_A Y37B_P Y14A_A | 6.6e-7 | 1.3e-5 | 8.8e-4 | 1.3e-5 | 9.2e-6 | 3.1e-8 | 2.4e-7 | 3.4e-4 |
| 35 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 38 | Y47B_G H31B_G Y14A_T | 3.6e-7 | 1.2e-5 | 6.6e-4 | 2.2e-5 | 8.5e-6 | 4.4e-8 | 2.7e-7 | 3.5e-4 |
| 38 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 41 | N18A_A E34B_K H31B_G | 1.5e-6 | 1.1e-5 | 6.8e-4 | 1.8e-5 | 1.0e-5 | 3.0e-8 | 1.0e-7 | 3.6e-4 |
| 41 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 45 | H31B_Q E4A_K E34B_A | 4.5e-7 | 2.1e-5 | 6.1e-4 | 7.8e-6 | 1.1e-5 | 6.3e-8 | 2.0e-7 | 3.2e-4 |
| 45 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 51 | K50B_N Y14A_T E34B_S | 6.2e-7 | 1.6e-5 | 1.1e-3 | 1.0e-5 | 1.1e-5 | 5.5e-8 | 9.6e-8 | 3.0e-4 |
| 51 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 54 | Y47B_G H26B_Q E34B_S | 5.1e-7 | 1.4e-5 | 1.0e-3 | 7.7e-6 | 1.4e-5 | 2.6e-8 | 2.9e-7 | 3.2e-4 |
| 54 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 59 | E42B_Q E34B_A H31B_G | 1.1e-6 | 1.8e-5 | 8.8e-4 | 5.1e-6 | 1.5e-5 | 2.8e-8 | 1.4e-7 | 3.4e-4 |
| 59 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 64 | H31B_Q Y37B_P E42B_Q | 9.4e-7 | 1.2e-5 | 8.5e-4 | 4.7e-6 | 2.1e-5 | 3.0e-8 | 1.8e-7 | 3.3e-4 |
| 64 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 65 | Y37B_P L38B_D H31B_G | 5.1e-7 | 1.6e-5 | 9.2e-4 | 9.5e-6 | 1.0e-5 | 2.8e-8 | 2.3e-7 | 3.6e-4 |
| 65 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 67 | Y47B_G G44B_E H31B_S | 9.5e-7 | 2.1e-5 | 6.3e-4 | 4.4e-6 | 1.2e-5 | 6.6e-8 | 1.1e-7 | 3.4e-4 |
| 67 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 70 | N18A_A E4A_K H26B_Q | 7.8e-7 | 1.4e-5 | 2.4e-3 | 5.5e-6 | 8.8e-6 | 4.2e-8 | 8.2e-8 | 3.6e-4 |
| 70 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 72 | F46B_Q L38B_D E34B_S | 3.3e-7 | 2.4e-5 | 7.4e-4 | 1.1e-5 | 1.1e-5 | 2.8e-8 | 2.8e-7 | 2.8e-4 |
| 72 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 79 | L38B_D E34B_K H31B_G | 5.6e-7 | 1.5e-5 | 6.0e-4 | 1.2e-5 | 9.1e-6 | 5.8e-8 | 1.4e-7 | 3.2e-4 |
| 79 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 85 | E34B_A H26B_Q T8A_K | 3.6e-7 | 1.1e-5 | 2.3e-3 | 6.2e-6 | 8.4e-6 | 4.7e-8 | 1.8e-7 | 3.5e-4 |
| 85 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 93 | K50B_N Y14A_M E34B_S | 6.4e-7 | 1.1e-5 | 1.1e-3 | 1.1e-5 | 1.3e-5 | 3.3e-8 | 1.2e-7 | 3.1e-4 |
| 93 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 99 | H31B_Q Y47B_G L38B_D | 7.9e-7 | 2.5e-5 | 6.1e-4 | 7.5e-6 | 1.1e-5 | 4.4e-8 | 8.8e-8 | 3.3e-4 |
| 99 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 107 | E42B_Q H31B_G T8A_K | 4.0e-7 | 1.3e-5 | 6.0e-4 | 4.8e-6 | 1.7e-5 | 8.0e-8 | 1.9e-7 | 3.2e-4 |
| 107 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 112 | H31B_Q I2A_D Y14A_T | 8.6e-7 | 1.2e-5 | 6.4e-4 | 1.1e-5 | 1.1e-5 | 2.7e-8 | 1.6e-7 | 3.4e-4 |
| 112 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 115 | F22B_D E34B_A Y14A_T | 3.9e-7 | 9.4e-6 | 7.8e-4 | 1.1e-5 | 1.0e-5 | 4.7e-8 | 2.6e-7 | 3.0e-4 |
| 115 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 117 | V23B_A L38B_D H31B_G | 4.9e-7 | 1.3e-5 | 7.3e-4 | 1.4e-5 | 9.3e-6 | 3.2e-8 | 1.8e-7 | 3.3e-4 |
| 117 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 118 | E34B_A H31B_S T8A_K | 5.8e-7 | 1.7e-5 | 6.2e-4 | 9.4e-6 | 7.6e-6 | 3.8e-8 | 2.1e-7 | 3.3e-4 |
| 118 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 127 | Y47B_G Y14A_A H31B_S | 5.8e-7 | 1.4e-5 | 8.5e-4 | 7.2e-6 | 1.3e-5 | 2.7e-8 | 1.9e-7 | 3.3e-4 |
| 127 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 128 | Y37B_P Y14A_A E42B_T | 3.5e-7 | 2.4e-5 | 6.2e-4 | 5.3e-6 | 1.3e-5 | 7.1e-8 | 1.3e-7 | 3.3e-4 |
| 128 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 132 | V23B_A Y14A_A E34B_A | 4.6e-7 | 1.3e-5 | 6.2e-4 | 1.8e-5 | 9.7e-6 | 3.0e-8 | 1.6e-7 | 3.5e-4 |
| 132 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 148 | N18A_A E4A_K E42B_T | 1.3e-6 | 1.0e-5 | 6.4e-4 | 4.4e-6 | 1.8e-5 | 4.6e-8 | 9.8e-8 | 3.4e-4 |
| 148 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 154 | Y47B_G E34B_A H31B_S | 3.5e-7 | 3.7e-5 | 6.7e-4 | 8.6e-6 | 1.0e-5 | 2.8e-8 | 1.3e-7 | 3.6e-4 |
| 154 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 162 | Y14A_M K50B_A H31B_G | 5.2e-7 | 2.4e-5 | 9.5e-4 | 9.4e-6 | 8.5e-6 | 3.0e-8 | 8.9e-8 | 3.7e-4 |
| 162 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 200 | N18A_A K50B_N H31B_G | 9.5e-7 | 1.4e-5 | 1.1e-3 | 5.6e-6 | 9.6e-6 | 3.2e-8 | 8.6e-8 | 3.6e-4 |
| 200 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 219 | V23B_A Y37B_P E34B_S | 7.2e-7 | 1.8e-5 | 7.8e-4 | 5.6e-6 | 1.2e-5 | 2.9e-8 | 1.2e-7 | 3.0e-4 |
| 219 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 222 | H31B_Q E42B_T E34B_K | 6.1e-7 | 1.1e-5 | 7.7e-4 | 9.2e-6 | 1.4e-5 | 3.6e-8 | 9.8e-8 | 3.0e-4 |
| 222 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 230 | Y37B_P Y47B_G H31B_S | 1.1e-6 | 1.7e-5 | 8.6e-4 | 5.0e-6 | 9.5e-6 | 2.8e-8 | 9.8e-8 | 3.3e-4 |
| 230 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 257 | H31B_Q Y37B_P E4A_K | 9.1e-7 | 1.4e-5 | 6.0e-4 | 5.9e-6 | 1.1e-5 | 3.1e-8 | 1.3e-7 | 3.2e-4 |
| 257 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 277 | E34B_F H31B_G Y14A_T | 3.8e-7 | 9.4e-6 | 9.6e-4 | 8.0e-6 | 7.6e-6 | 3.8e-8 | 2.0e-7 | 3.7e-4 |
| 277 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 337 | Y37B_P E34B_F H31B_S | 5.9e-7 | 1.4e-5 | 7.0e-4 | 4.5e-6 | 1.2e-5 | 2.9e-8 | 1.7e-7 | 3.2e-4 |
| 337 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 344 | Y47B_G L38B_D K50B_A | 5.8e-7 | 1.2e-5 | 8.6e-4 | 5.6e-6 | 7.6e-6 | 5.2e-8 | 1.1e-7 | 3.3e-4 |
| 344 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 354 | E34B_F H31B_S T8A_K | 4.2e-7 | 1.4e-5 | 6.3e-4 | 5.6e-6 | 9.4e-6 | 4.7e-8 | 1.5e-7 | 3.4e-4 |
| 354 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 383 | Y37B_P L27B_K E34B_S | 4.9e-7 | 1.2e-5 | 1.0e-3 | 9.9e-6 | 1.3e-5 | 2.6e-8 | 7.9e-8 | 2.8e-4 |
| 383 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 398 | V23B_A E42B_T H31B_S | 3.7e-7 | 1.1e-5 | 1.1e-3 | 5.4e-6 | 1.4e-5 | 3.0e-8 | 1.4e-7 | 3.0e-4 |
| 398 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 423 | E34B_F G44B_E L27B_K | 4.6e-7 | 1.2e-5 | 6.0e-4 | 8.2e-6 | 9.6e-6 | 4.4e-8 | 1.1e-7 | 3.2e-4 |
| 423 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 429 | V23B_A E4A_K E34B_K | 4.2e-7 | 1.3e-5 | 8.4e-4 | 9.8e-6 | 7.8e-6 | 4.9e-8 | 8.3e-8 | 2.8e-4 |
| 429 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 433 | H31B_Q E4A_K Y14A_M | 3.7e-7 | 1.0e-5 | 7.2e-4 | 5.4e-6 | 7.8e-6 | 3.7e-8 | 3.0e-7 | 3.2e-4 |
| 433 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 461 | Y47B_G K50B_A E34B_S | 3.9e-7 | 1.9e-5 | 6.1e-4 | 4.9e-6 | 1.4e-5 | 2.7e-8 | 1.4e-7 | 3.2e-4 |
| 461 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 479 | N18A_A F46B_Q Y14A_M | 4.7e-7 | 1.7e-5 | 6.9e-4 | 5.0e-6 | 1.2e-5 | 2.7e-8 | 1.3e-7 | 3.1e-4 |
| 479 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 530 | E4A_K H31B_G Y14A_T | 3.6e-7 | 9.4e-6 | 9.1e-4 | 1.2e-5 | 8.5e-6 | 3.4e-8 | 8.5e-8 | 3.5e-4 |
| 530 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 604 | N18A_A L38B_D H31B_G | 5.7e-7 | 1.1e-5 | 6.5e-4 | 6.7e-6 | 8.3e-6 | 3.0e-8 | 1.1e-7 | 3.5e-4 |
| 604 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 613 | V23B_A H31B_Q E42B_Q | 4.8e-7 | 1.3e-5 | 7.4e-4 | 5.9e-6 | 9.0e-6 | 3.3e-8 | 1.1e-7 | 2.9e-4 |
| 613 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 616 | H31B_Q Y37B_P F22B_D | 5.6e-7 | 9.9e-6 | 6.0e-4 | 4.3e-6 | 9.1e-6 | 2.8e-8 | 2.2e-7 | 3.2e-4 |
| 616 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 667 | H31B_Q E4A_K E34B_K | 4.0e-7 | 1.1e-5 | 6.3e-4 | 8.0e-6 | 8.0e-6 | 4.7e-8 | 1.0e-7 | 2.8e-4 |
| 667 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 680 | E34B_K I2A_D H31B_S | 6.0e-7 | 1.1e-5 | 8.4e-4 | 4.0e-6 | 7.9e-6 | 4.6e-8 | 8.8e-8 | 3.2e-4 |
| 680 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 745 | H31B_Q Y37B_P K50B_N | 3.7e-7 | 1.2e-5 | 8.3e-4 | 4.6e-6 | 8.8e-6 | 3.9e-8 | 1.1e-7 | 3.2e-4 |
| 745 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 965 | E34B_K H31B_G L32B_H | 3.4e-7 | 1.0e-5 | 8.4e-4 | 7.0e-6 | 9.2e-6 | 3.4e-8 | 7.5e-8 | 3.2e-4 |
| 965 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 1306 | E4A_K E34B_K T8A_K | 3.3e-7 | 1.1e-5 | 6.4e-4 | 7.5e-6 | 8.2e-6 | 2.9e-8 | 6.9e-8 | 3.4e-4 |
| 1306 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 1432 | E34B_K H31B_G T8A_K | 3.3e-7 | 1.3e-5 | 6.7e-4 | 4.5e-6 | 1.0e-5 | 2.7e-8 | 6.9e-8 | 3.5e-4 |
| 1432 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| No. | Muta | AE | AG | AI | AK | CE | CG | CI | CK |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Y14A_T E34B_S H31B_S | 1.6e-6 | 2.2e-5 | 1.1e-3 | 1.4e-5 | 1.2e-5 | 5.9e-8 | 3.8e-7 | 3.0e-4 |
| 1 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 2 | H31B_Q Y14A_A H26B_Q | 4.4e-7 | 1.1e-5 | 4.3e-3 | 6.1e-6 | 9.7e-6 | 5.2e-8 | 5.0e-7 | 3.4e-4 |
| 2 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 5 | N18A_A Y47B_G H31B_S | 2.3e-6 | 1.7e-5 | 6.5e-4 | 9.2e-6 | 1.0e-5 | 9.2e-8 | 1.0e-7 | 3.6e-4 |
| 5 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 6 | E34B_A H26B_Q H31B_S | 5.4e-7 | 1.2e-5 | 1.4e-3 | 1.2e-5 | 9.6e-6 | 4.3e-8 | 5.3e-7 | 2.9e-4 |
| 6 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 7 | Y37B_P Y14A_A H31B_G | 5.7e-7 | 1.3e-5 | 8.9e-4 | 1.0e-5 | 1.1e-5 | 1.4e-7 | 1.6e-7 | 4.0e-4 |
| 7 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 15 | V23B_A H31B_G E34B_S | 9.1e-7 | 1.2e-5 | 7.1e-4 | 1.4e-5 | 1.2e-5 | 3.6e-8 | 2.2e-7 | 3.2e-4 |
| 15 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 17 | Y47B_G E34B_F E42B_T | 6.6e-7 | 2.2e-5 | 8.2e-4 | 5.6e-6 | 2.3e-5 | 5.8e-8 | 1.1e-7 | 3.2e-4 |
| 17 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 21 | H26B_Q Y14A_T H31B_S | 6.9e-7 | 1.4e-5 | 1.4e-3 | 7.7e-6 | 7.3e-6 | 3.1e-8 | 4.1e-7 | 3.0e-4 |
| 21 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 23 | N18A_A Y37B_P Y14A_M | 1.8e-6 | 1.4e-5 | 1.0e-3 | 7.1e-6 | 1.3e-5 | 3.7e-8 | 1.1e-7 | 2.9e-4 |
| 23 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 24 | Y14A_A E42B_T E34B_A | 4.2e-7 | 2.0e-5 | 6.1e-4 | 2.4e-5 | 1.5e-5 | 2.3e-8 | 1.9e-7 | 3.3e-4 |
| 24 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 29 | Y14A_A K50B_A H31B_G | 4.2e-7 | 1.0e-5 | 1.1e-3 | 6.4e-6 | 8.0e-6 | 9.7e-8 | 2.7e-7 | 4.1e-4 |
| 29 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 31 | Y47B_G Y14A_A H31B_G | 4.1e-7 | 2.5e-5 | 7.4e-4 | 1.0e-5 | 1.3e-5 | 3.3e-8 | 2.6e-7 | 2.9e-4 |
| 31 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 32 | N18A_A L38B_D E34B_S | 1.1e-6 | 1.1e-5 | 5.9e-4 | 1.6e-5 | 1.0e-5 | 2.3e-8 | 3.0e-7 | 3.1e-4 |
| 32 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 33 | H31B_G E34B_S L32B_H | 6.3e-7 | 9.9e-6 | 8.7e-4 | 8.0e-6 | 1.1e-5 | 1.4e-7 | 1.1e-7 | 3.3e-4 |
| 33 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 35 | N18A_A Y37B_P Y14A_A | 6.6e-7 | 1.3e-5 | 8.8e-4 | 1.3e-5 | 9.2e-6 | 3.1e-8 | 2.4e-7 | 3.4e-4 |
| 35 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 38 | Y47B_G H31B_G Y14A_T | 3.6e-7 | 1.2e-5 | 6.6e-4 | 2.2e-5 | 8.5e-6 | 4.4e-8 | 2.7e-7 | 3.5e-4 |
| 38 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 41 | N18A_A E34B_K H31B_G | 1.5e-6 | 1.1e-5 | 6.8e-4 | 1.8e-5 | 1.0e-5 | 3.0e-8 | 1.0e-7 | 3.6e-4 |
| 41 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 45 | H31B_Q E4A_K E34B_A | 4.5e-7 | 2.1e-5 | 6.1e-4 | 7.8e-6 | 1.1e-5 | 6.3e-8 | 2.0e-7 | 3.2e-4 |
| 45 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 46 | E34B_A H26B_Q H31B_G | 3.0e-7 | 1.2e-5 | 8.3e-4 | 5.0e-6 | 9.4e-6 | 1.1e-7 | 3.9e-7 | 3.3e-4 |
| 46 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 51 | K50B_N Y14A_T E34B_S | 6.2e-7 | 1.6e-5 | 1.1e-3 | 1.0e-5 | 1.1e-5 | 5.5e-8 | 9.6e-8 | 3.0e-4 |
| 51 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 54 | Y47B_G H26B_Q E34B_S | 5.1e-7 | 1.4e-5 | 1.0e-3 | 7.7e-6 | 1.4e-5 | 2.6e-8 | 2.9e-7 | 3.2e-4 |
| 54 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 59 | E42B_Q E34B_A H31B_G | 1.1e-6 | 1.8e-5 | 8.8e-4 | 5.1e-6 | 1.5e-5 | 2.8e-8 | 1.4e-7 | 3.4e-4 |
| 59 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 61 | E42B_T H31B_G Y14A_T | 5.9e-7 | 1.3e-5 | 9.2e-4 | 1.5e-5 | 1.2e-5 | 2.1e-8 | 1.9e-7 | 3.5e-4 |
| 61 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 65 | Y37B_P L38B_D H31B_G | 5.1e-7 | 1.6e-5 | 9.2e-4 | 9.5e-6 | 1.0e-5 | 2.8e-8 | 2.3e-7 | 3.6e-4 |
| 65 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 70 | N18A_A E4A_K H26B_Q | 7.8e-7 | 1.4e-5 | 2.4e-3 | 5.5e-6 | 8.8e-6 | 4.2e-8 | 8.2e-8 | 3.6e-4 |
| 70 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 79 | L38B_D E34B_K H31B_G | 5.6e-7 | 1.5e-5 | 6.0e-4 | 1.2e-5 | 9.1e-6 | 5.8e-8 | 1.4e-7 | 3.2e-4 |
| 79 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 83 | E34B_F H26B_Q L32B_H | 3.2e-7 | 1.8e-5 | 9.9e-4 | 6.0e-6 | 8.1e-6 | 3.0e-8 | 5.0e-7 | 3.4e-4 |
| 83 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 85 | E34B_A H26B_Q T8A_K | 3.6e-7 | 1.1e-5 | 2.3e-3 | 6.2e-6 | 8.4e-6 | 4.7e-8 | 1.8e-7 | 3.5e-4 |
| 85 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 93 | K50B_N Y14A_M E34B_S | 6.4e-7 | 1.1e-5 | 1.1e-3 | 1.1e-5 | 1.3e-5 | 3.3e-8 | 1.2e-7 | 3.1e-4 |
| 93 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 94 | H31B_Q Y47B_G E34B_A | 5.5e-7 | 1.0e-5 | 5.9e-4 | 9.3e-6 | 7.5e-6 | 6.4e-8 | 2.8e-7 | 3.3e-4 |
| 94 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 99 | H31B_Q Y47B_G L38B_D | 7.9e-7 | 2.5e-5 | 6.1e-4 | 7.5e-6 | 1.1e-5 | 4.4e-8 | 8.8e-8 | 3.3e-4 |
| 99 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 112 | H31B_Q I2A_D Y14A_T | 8.6e-7 | 1.2e-5 | 6.4e-4 | 1.1e-5 | 1.1e-5 | 2.7e-8 | 1.6e-7 | 3.4e-4 |
| 112 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 117 | V23B_A L38B_D H31B_G | 4.9e-7 | 1.3e-5 | 7.3e-4 | 1.4e-5 | 9.3e-6 | 3.2e-8 | 1.8e-7 | 3.3e-4 |
| 117 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 118 | E34B_A H31B_S T8A_K | 5.8e-7 | 1.7e-5 | 6.2e-4 | 9.4e-6 | 7.6e-6 | 3.8e-8 | 2.1e-7 | 3.3e-4 |
| 118 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 127 | Y47B_G Y14A_A H31B_S | 5.8e-7 | 1.4e-5 | 8.5e-4 | 7.2e-6 | 1.3e-5 | 2.7e-8 | 1.9e-7 | 3.3e-4 |
| 127 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 128 | Y37B_P Y14A_A E42B_T | 3.5e-7 | 2.4e-5 | 6.2e-4 | 5.3e-6 | 1.3e-5 | 7.1e-8 | 1.3e-7 | 3.3e-4 |
| 128 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 132 | V23B_A Y14A_A E34B_A | 4.6e-7 | 1.3e-5 | 6.2e-4 | 1.8e-5 | 9.7e-6 | 3.0e-8 | 1.6e-7 | 3.5e-4 |
| 132 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 134 | Y14A_M I2A_D E34B_S | 6.9e-7 | 3.3e-5 | 5.9e-4 | 9.5e-6 | 8.9e-6 | 3.3e-8 | 9.3e-8 | 3.1e-4 |
| 134 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 154 | Y47B_G E34B_A H31B_S | 3.5e-7 | 3.7e-5 | 6.7e-4 | 8.6e-6 | 1.0e-5 | 2.8e-8 | 1.3e-7 | 3.6e-4 |
| 154 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 160 | Y14A_A G44B_E H31B_G | 2.7e-7 | 2.2e-5 | 6.6e-4 | 5.5e-6 | 1.2e-5 | 5.2e-8 | 2.0e-7 | 3.5e-4 |
| 160 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 162 | Y14A_M K50B_A H31B_G | 5.2e-7 | 2.4e-5 | 9.5e-4 | 9.4e-6 | 8.5e-6 | 3.0e-8 | 8.9e-8 | 3.7e-4 |
| 162 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 168 | N18A_A L38B_D I2A_D | 1.0e-6 | 1.2e-5 | 8.8e-4 | 9.3e-6 | 8.2e-6 | 2.2e-8 | 1.5e-7 | 3.4e-4 |
| 168 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 200 | N18A_A K50B_N H31B_G | 9.5e-7 | 1.4e-5 | 1.1e-3 | 5.6e-6 | 9.6e-6 | 3.2e-8 | 8.6e-8 | 3.6e-4 |
| 200 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 204 | Y14A_A E34B_A H31B_G | 2.9e-7 | 2.6e-5 | 6.5e-4 | 7.1e-6 | 9.7e-6 | 7.0e-8 | 9.6e-8 | 3.4e-4 |
| 204 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 219 | V23B_A Y37B_P E34B_S | 7.2e-7 | 1.8e-5 | 7.8e-4 | 5.6e-6 | 1.2e-5 | 2.9e-8 | 1.2e-7 | 3.0e-4 |
| 219 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 222 | H31B_Q E42B_T E34B_K | 6.1e-7 | 1.1e-5 | 7.7e-4 | 9.2e-6 | 1.4e-5 | 3.6e-8 | 9.8e-8 | 3.0e-4 |
| 222 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 228 | H31B_Q L38B_D K50B_A | 5.1e-7 | 2.0e-5 | 7.9e-4 | 9.2e-6 | 7.1e-6 | 4.4e-8 | 9.7e-8 | 3.1e-4 |
| 228 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 230 | Y37B_P Y47B_G H31B_S | 1.1e-6 | 1.7e-5 | 8.6e-4 | 5.0e-6 | 9.5e-6 | 2.8e-8 | 9.8e-8 | 3.3e-4 |
| 230 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 235 | H31B_Q Y37B_P Y14A_T | 2.9e-7 | 1.1e-5 | 9.0e-4 | 8.9e-6 | 1.2e-5 | 3.2e-8 | 2.0e-7 | 3.5e-4 |
| 235 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 249 | H31B_Q Y14A_A E42B_T | 2.5e-7 | 1.4e-5 | 8.0e-4 | 1.4e-5 | 1.2e-5 | 3.0e-8 | 1.5e-7 | 3.1e-4 |
| 249 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 253 | Y47B_G E42B_Q K50B_A | 4.1e-7 | 2.2e-5 | 5.9e-4 | 5.6e-6 | 1.6e-5 | 4.0e-8 | 1.1e-7 | 3.1e-4 |
| 253 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 254 | E42B_T H26B_Q E34B_A | 4.2e-7 | 1.6e-5 | 1.5e-3 | 5.5e-6 | 1.0e-5 | 2.1e-8 | 1.8e-7 | 3.1e-4 |
| 254 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 257 | H31B_Q Y37B_P E4A_K | 9.1e-7 | 1.4e-5 | 6.0e-4 | 5.9e-6 | 1.1e-5 | 3.1e-8 | 1.3e-7 | 3.2e-4 |
| 257 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 294 | K50B_N Y14A_A E34B_K | 2.5e-7 | 1.5e-5 | 1.2e-3 | 6.5e-6 | 1.0e-5 | 6.0e-8 | 9.5e-8 | 3.3e-4 |
| 294 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 321 | Y47B_G E34B_F Y14A_A | 2.7e-7 | 1.6e-5 | 6.7e-4 | 1.6e-5 | 1.1e-5 | 2.1e-8 | 1.3e-7 | 3.7e-4 |
| 321 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 322 | Y14A_M E34B_A T8A_K | 7.4e-7 | 1.5e-5 | 6.4e-4 | 7.2e-6 | 9.6e-6 | 2.2e-8 | 1.4e-7 | 3.4e-4 |
| 322 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 344 | Y47B_G L38B_D K50B_A | 5.8e-7 | 1.2e-5 | 8.6e-4 | 5.6e-6 | 7.6e-6 | 5.2e-8 | 1.1e-7 | 3.3e-4 |
| 344 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 351 | H31B_Q E34B_F Y14A_A | 2.9e-7 | 1.7e-5 | 8.2e-4 | 5.0e-6 | 1.0e-5 | 2.6e-8 | 2.8e-7 | 3.2e-4 |
| 351 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 354 | E34B_F H31B_S T8A_K | 4.2e-7 | 1.4e-5 | 6.3e-4 | 5.6e-6 | 9.4e-6 | 4.7e-8 | 1.5e-7 | 3.4e-4 |
| 354 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 365 | Y14A_M H31B_G R43B_F | 6.9e-7 | 1.8e-5 | 6.3e-4 | 5.0e-6 | 7.7e-6 | 2.4e-8 | 1.9e-7 | 3.3e-4 |
| 365 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 368 | Y47B_G E4A_K L38B_D | 4.4e-7 | 3.4e-5 | 6.1e-4 | 8.9e-6 | 1.1e-5 | 2.1e-8 | 7.5e-8 | 3.2e-4 |
| 368 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 380 | Y47B_G Y14A_A E42B_Q | 3.1e-7 | 1.3e-5 | 7.8e-4 | 9.6e-6 | 1.8e-5 | 3.0e-8 | 7.7e-8 | 3.5e-4 |
| 380 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 381 | L38B_D E34B_K I2A_D | 2.6e-7 | 1.4e-5 | 8.0e-4 | 8.0e-6 | 1.0e-5 | 3.2e-8 | 1.9e-7 | 3.1e-4 |
| 381 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 397 | N18A_A Y47B_G K50B_A | 1.1e-6 | 1.2e-5 | 6.8e-4 | 5.8e-6 | 7.4e-6 | 3.1e-8 | 9.4e-8 | 3.8e-4 |
| 397 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 398 | V23B_A E42B_T H31B_S | 3.7e-7 | 1.1e-5 | 1.1e-3 | 5.4e-6 | 1.4e-5 | 3.0e-8 | 1.4e-7 | 3.0e-4 |
| 398 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 423 | E34B_F G44B_E L27B_K | 4.6e-7 | 1.2e-5 | 6.0e-4 | 8.2e-6 | 9.6e-6 | 4.4e-8 | 1.1e-7 | 3.2e-4 |
| 423 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 433 | H31B_Q E4A_K Y14A_M | 3.7e-7 | 1.0e-5 | 7.2e-4 | 5.4e-6 | 7.8e-6 | 3.7e-8 | 3.0e-7 | 3.2e-4 |
| 433 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 439 | H31B_Q F46B_Q E34B_F | 3.1e-7 | 1.5e-5 | 7.6e-4 | 8.2e-6 | 9.7e-6 | 3.0e-8 | 1.6e-7 | 2.9e-4 |
| 439 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 461 | Y47B_G K50B_A E34B_S | 3.9e-7 | 1.9e-5 | 6.1e-4 | 4.9e-6 | 1.4e-5 | 2.7e-8 | 1.4e-7 | 3.2e-4 |
| 461 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 473 | Y14A_A E42B_T H31B_S | 3.2e-7 | 1.3e-5 | 6.8e-4 | 9.3e-6 | 1.4e-5 | 3.0e-8 | 1.1e-7 | 3.0e-4 |
| 473 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 479 | N18A_A F46B_Q Y14A_M | 4.7e-7 | 1.7e-5 | 6.9e-4 | 5.0e-6 | 1.2e-5 | 2.7e-8 | 1.3e-7 | 3.1e-4 |
| 479 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 495 | Y47B_G E34B_K H26B_Q | 3.9e-7 | 1.3e-5 | 8.3e-4 | 8.7e-6 | 8.2e-6 | 2.4e-8 | 1.4e-7 | 3.4e-4 |
| 495 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 504 | E42B_T Y14A_M E34B_K | 2.7e-7 | 1.1e-5 | 6.8e-4 | 7.9e-6 | 1.4e-5 | 2.7e-8 | 1.8e-7 | 3.1e-4 |
| 504 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 586 | H31B_Q Y47B_G T8A_K | 2.6e-7 | 1.4e-5 | 7.0e-4 | 5.0e-6 | 1.1e-5 | 7.2e-8 | 8.5e-8 | 3.2e-4 |
| 586 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 604 | N18A_A L38B_D H31B_G | 5.7e-7 | 1.1e-5 | 6.5e-4 | 6.7e-6 | 8.3e-6 | 3.0e-8 | 1.1e-7 | 3.5e-4 |
| 604 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 613 | V23B_A H31B_Q E42B_Q | 4.8e-7 | 1.3e-5 | 7.4e-4 | 5.9e-6 | 9.0e-6 | 3.3e-8 | 1.1e-7 | 2.9e-4 |
| 613 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 614 | S9A_D Y14A_K I2A_R | 2.9e-7 | 2.0e-5 | 6.8e-4 | 9.7e-6 | 7.4e-6 | 3.2e-8 | 7.9e-8 | 3.6e-4 |
| 614 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 632 | Y47B_G E34B_A H31B_G | 4.1e-7 | 1.3e-5 | 6.4e-4 | 5.9e-6 | 1.9e-5 | 2.4e-8 | 8.1e-8 | 3.4e-4 |
| 632 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 679 | Y37B_P E4A_K H26B_Q | 2.7e-7 | 1.4e-5 | 1.5e-3 | 4.9e-6 | 1.5e-5 | 2.3e-8 | 7.4e-8 | 3.2e-4 |
| 679 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 735 | N18A_A E42B_Q H26B_Q | 4.7e-7 | 1.6e-5 | 9.2e-4 | 5.7e-6 | 8.1e-6 | 2.2e-8 | 9.9e-8 | 3.0e-4 |
| 735 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 784 | Y47B_G E34B_K Y14A_T | 4.0e-7 | 1.4e-5 | 5.9e-4 | 8.9e-6 | 1.0e-5 | 2.3e-8 | 9.1e-8 | 3.1e-4 |
| 784 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 821 | L38B_D Y14A_M K50B_A | 4.1e-7 | 9.9e-6 | 8.2e-4 | 1.1e-5 | 7.3e-6 | 2.4e-8 | 8.7e-8 | 3.2e-4 |
| 821 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 837 | K50B_A H31B_G Y14A_T | 2.9e-7 | 1.1e-5 | 6.5e-4 | 8.3e-6 | 7.9e-6 | 2.9e-8 | 1.3e-7 | 3.4e-4 |
| 837 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 860 | H31B_Q E34B_A K50B_A | 3.1e-7 | 1.1e-5 | 8.1e-4 | 5.4e-6 | 8.4e-6 | 2.9e-8 | 1.5e-7 | 3.1e-4 |
| 860 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 965 | E34B_K H31B_G L32B_H | 3.4e-7 | 1.0e-5 | 8.4e-4 | 7.0e-6 | 9.2e-6 | 3.4e-8 | 7.5e-8 | 3.2e-4 |
| 965 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 994 | Y14A_A I2A_D E34B_A | 3.0e-7 | 1.1e-5 | 8.8e-4 | 7.4e-6 | 9.5e-6 | 2.1e-8 | 1.0e-7 | 3.4e-4 |
| 994 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 1000 | L38B_D I2A_D T8A_K | 7.5e-7 | 1.0e-5 | 6.1e-4 | 7.6e-6 | 7.8e-6 | 2.2e-8 | 7.6e-8 | 3.2e-4 |
| 1000 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 1310 | Y14A_A E34B_K K50B_A | 3.1e-7 | 1.2e-5 | 6.2e-4 | 5.3e-6 | 7.5e-6 | 2.9e-8 | 1.1e-7 | 3.3e-4 |
| 1310 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| No. | Muta | AE | AG | AI | AK | CE | CG | CI | CK |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Y14A_T E34B_S H31B_S | 1.6e-6 | 2.2e-5 | 1.1e-3 | 1.4e-5 | 1.2e-5 | 5.9e-8 | 3.8e-7 | 3.0e-4 |
| 1 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 2 | H31B_Q Y14A_A H26B_Q | 4.4e-7 | 1.1e-5 | 4.3e-3 | 6.1e-6 | 9.7e-6 | 5.2e-8 | 5.0e-7 | 3.4e-4 |
| 2 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 5 | N18A_A Y47B_G H31B_S | 2.3e-6 | 1.7e-5 | 6.5e-4 | 9.2e-6 | 1.0e-5 | 9.2e-8 | 1.0e-7 | 3.6e-4 |
| 5 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 6 | E34B_A H26B_Q H31B_S | 5.4e-7 | 1.2e-5 | 1.4e-3 | 1.2e-5 | 9.6e-6 | 4.3e-8 | 5.3e-7 | 2.9e-4 |
| 6 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 7 | Y37B_P Y14A_A H31B_G | 5.7e-7 | 1.3e-5 | 8.9e-4 | 1.0e-5 | 1.1e-5 | 1.4e-7 | 1.6e-7 | 4.0e-4 |
| 7 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 15 | V23B_A H31B_G E34B_S | 9.1e-7 | 1.2e-5 | 7.1e-4 | 1.4e-5 | 1.2e-5 | 3.6e-8 | 2.2e-7 | 3.2e-4 |
| 15 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 17 | Y47B_G E34B_F E42B_T | 6.6e-7 | 2.2e-5 | 8.2e-4 | 5.6e-6 | 2.3e-5 | 5.8e-8 | 1.1e-7 | 3.2e-4 |
| 17 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 23 | N18A_A Y37B_P Y14A_M | 1.8e-6 | 1.4e-5 | 1.0e-3 | 7.1e-6 | 1.3e-5 | 3.7e-8 | 1.1e-7 | 2.9e-4 |
| 23 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 29 | Y14A_A K50B_A H31B_G | 4.2e-7 | 1.0e-5 | 1.1e-3 | 6.4e-6 | 8.0e-6 | 9.7e-8 | 2.7e-7 | 4.1e-4 |
| 29 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 31 | Y47B_G Y14A_A H31B_G | 4.1e-7 | 2.5e-5 | 7.4e-4 | 1.0e-5 | 1.3e-5 | 3.3e-8 | 2.6e-7 | 2.9e-4 |
| 31 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 33 | H31B_G E34B_S L32B_H | 6.3e-7 | 9.9e-6 | 8.7e-4 | 8.0e-6 | 1.1e-5 | 1.4e-7 | 1.1e-7 | 3.3e-4 |
| 33 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 35 | N18A_A Y37B_P Y14A_A | 6.6e-7 | 1.3e-5 | 8.8e-4 | 1.3e-5 | 9.2e-6 | 3.1e-8 | 2.4e-7 | 3.4e-4 |
| 35 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 38 | Y47B_G H31B_G Y14A_T | 3.6e-7 | 1.2e-5 | 6.6e-4 | 2.2e-5 | 8.5e-6 | 4.4e-8 | 2.7e-7 | 3.5e-4 |
| 38 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 41 | N18A_A E34B_K H31B_G | 1.5e-6 | 1.1e-5 | 6.8e-4 | 1.8e-5 | 1.0e-5 | 3.0e-8 | 1.0e-7 | 3.6e-4 |
| 41 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 45 | H31B_Q E4A_K E34B_A | 4.5e-7 | 2.1e-5 | 6.1e-4 | 7.8e-6 | 1.1e-5 | 6.3e-8 | 2.0e-7 | 3.2e-4 |
| 45 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 51 | K50B_N Y14A_T E34B_S | 6.2e-7 | 1.6e-5 | 1.1e-3 | 1.0e-5 | 1.1e-5 | 5.5e-8 | 9.6e-8 | 3.0e-4 |
| 51 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 54 | Y47B_G H26B_Q E34B_S | 5.1e-7 | 1.4e-5 | 1.0e-3 | 7.7e-6 | 1.4e-5 | 2.6e-8 | 2.9e-7 | 3.2e-4 |
| 54 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 59 | E42B_Q E34B_A H31B_G | 1.1e-6 | 1.8e-5 | 8.8e-4 | 5.1e-6 | 1.5e-5 | 2.8e-8 | 1.4e-7 | 3.4e-4 |
| 59 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 65 | Y37B_P L38B_D H31B_G | 5.1e-7 | 1.6e-5 | 9.2e-4 | 9.5e-6 | 1.0e-5 | 2.8e-8 | 2.3e-7 | 3.6e-4 |
| 65 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 70 | N18A_A E4A_K H26B_Q | 7.8e-7 | 1.4e-5 | 2.4e-3 | 5.5e-6 | 8.8e-6 | 4.2e-8 | 8.2e-8 | 3.6e-4 |
| 70 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 79 | L38B_D E34B_K H31B_G | 5.6e-7 | 1.5e-5 | 6.0e-4 | 1.2e-5 | 9.1e-6 | 5.8e-8 | 1.4e-7 | 3.2e-4 |
| 79 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 85 | E34B_A H26B_Q T8A_K | 3.6e-7 | 1.1e-5 | 2.3e-3 | 6.2e-6 | 8.4e-6 | 4.7e-8 | 1.8e-7 | 3.5e-4 |
| 85 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 93 | K50B_N Y14A_M E34B_S | 6.4e-7 | 1.1e-5 | 1.1e-3 | 1.1e-5 | 1.3e-5 | 3.3e-8 | 1.2e-7 | 3.1e-4 |
| 93 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 99 | H31B_Q Y47B_G L38B_D | 7.9e-7 | 2.5e-5 | 6.1e-4 | 7.5e-6 | 1.1e-5 | 4.4e-8 | 8.8e-8 | 3.3e-4 |
| 99 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 112 | H31B_Q I2A_D Y14A_T | 8.6e-7 | 1.2e-5 | 6.4e-4 | 1.1e-5 | 1.1e-5 | 2.7e-8 | 1.6e-7 | 3.4e-4 |
| 112 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 117 | V23B_A L38B_D H31B_G | 4.9e-7 | 1.3e-5 | 7.3e-4 | 1.4e-5 | 9.3e-6 | 3.2e-8 | 1.8e-7 | 3.3e-4 |
| 117 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 118 | E34B_A H31B_S T8A_K | 5.8e-7 | 1.7e-5 | 6.2e-4 | 9.4e-6 | 7.6e-6 | 3.8e-8 | 2.1e-7 | 3.3e-4 |
| 118 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 127 | Y47B_G Y14A_A H31B_S | 5.8e-7 | 1.4e-5 | 8.5e-4 | 7.2e-6 | 1.3e-5 | 2.7e-8 | 1.9e-7 | 3.3e-4 |
| 127 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 128 | Y37B_P Y14A_A E42B_T | 3.5e-7 | 2.4e-5 | 6.2e-4 | 5.3e-6 | 1.3e-5 | 7.1e-8 | 1.3e-7 | 3.3e-4 |
| 128 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 132 | V23B_A Y14A_A E34B_A | 4.6e-7 | 1.3e-5 | 6.2e-4 | 1.8e-5 | 9.7e-6 | 3.0e-8 | 1.6e-7 | 3.5e-4 |
| 132 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 154 | Y47B_G E34B_A H31B_S | 3.5e-7 | 3.7e-5 | 6.7e-4 | 8.6e-6 | 1.0e-5 | 2.8e-8 | 1.3e-7 | 3.6e-4 |
| 154 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 162 | Y14A_M K50B_A H31B_G | 5.2e-7 | 2.4e-5 | 9.5e-4 | 9.4e-6 | 8.5e-6 | 3.0e-8 | 8.9e-8 | 3.7e-4 |
| 162 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 200 | N18A_A K50B_N H31B_G | 9.5e-7 | 1.4e-5 | 1.1e-3 | 5.6e-6 | 9.6e-6 | 3.2e-8 | 8.6e-8 | 3.6e-4 |
| 200 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 219 | V23B_A Y37B_P E34B_S | 7.2e-7 | 1.8e-5 | 7.8e-4 | 5.6e-6 | 1.2e-5 | 2.9e-8 | 1.2e-7 | 3.0e-4 |
| 219 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 222 | H31B_Q E42B_T E34B_K | 6.1e-7 | 1.1e-5 | 7.7e-4 | 9.2e-6 | 1.4e-5 | 3.6e-8 | 9.8e-8 | 3.0e-4 |
| 222 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 230 | Y37B_P Y47B_G H31B_S | 1.1e-6 | 1.7e-5 | 8.6e-4 | 5.0e-6 | 9.5e-6 | 2.8e-8 | 9.8e-8 | 3.3e-4 |
| 230 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 257 | H31B_Q Y37B_P E4A_K | 9.1e-7 | 1.4e-5 | 6.0e-4 | 5.9e-6 | 1.1e-5 | 3.1e-8 | 1.3e-7 | 3.2e-4 |
| 257 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 344 | Y47B_G L38B_D K50B_A | 5.8e-7 | 1.2e-5 | 8.6e-4 | 5.6e-6 | 7.6e-6 | 5.2e-8 | 1.1e-7 | 3.3e-4 |
| 344 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 354 | E34B_F H31B_S T8A_K | 4.2e-7 | 1.4e-5 | 6.3e-4 | 5.6e-6 | 9.4e-6 | 4.7e-8 | 1.5e-7 | 3.4e-4 |
| 354 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 398 | V23B_A E42B_T H31B_S | 3.7e-7 | 1.1e-5 | 1.1e-3 | 5.4e-6 | 1.4e-5 | 3.0e-8 | 1.4e-7 | 3.0e-4 |
| 398 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 423 | E34B_F G44B_E L27B_K | 4.6e-7 | 1.2e-5 | 6.0e-4 | 8.2e-6 | 9.6e-6 | 4.4e-8 | 1.1e-7 | 3.2e-4 |
| 423 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 433 | H31B_Q E4A_K Y14A_M | 3.7e-7 | 1.0e-5 | 7.2e-4 | 5.4e-6 | 7.8e-6 | 3.7e-8 | 3.0e-7 | 3.2e-4 |
| 433 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 461 | Y47B_G K50B_A E34B_S | 3.9e-7 | 1.9e-5 | 6.1e-4 | 4.9e-6 | 1.4e-5 | 2.7e-8 | 1.4e-7 | 3.2e-4 |
| 461 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 479 | N18A_A F46B_Q Y14A_M | 4.7e-7 | 1.7e-5 | 6.9e-4 | 5.0e-6 | 1.2e-5 | 2.7e-8 | 1.3e-7 | 3.1e-4 |
| 479 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 604 | N18A_A L38B_D H31B_G | 5.7e-7 | 1.1e-5 | 6.5e-4 | 6.7e-6 | 8.3e-6 | 3.0e-8 | 1.1e-7 | 3.5e-4 |
| 604 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 613 | V23B_A H31B_Q E42B_Q | 4.8e-7 | 1.3e-5 | 7.4e-4 | 5.9e-6 | 9.0e-6 | 3.3e-8 | 1.1e-7 | 2.9e-4 |
| 613 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
| 965 | E34B_K H31B_G L32B_H | 3.4e-7 | 1.0e-5 | 8.4e-4 | 7.0e-6 | 9.2e-6 | 3.4e-8 | 7.5e-8 | 3.2e-4 |
| 965 | native insulin molecule | 3.2e-7 | 8.7e-6 | 5.9e-4 | 3.8e-6 | 7.4e-6 | 2.4e-8 | 6.3e-8 | 2.7e-4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).





