Submitted:
25 May 2024
Posted:
27 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
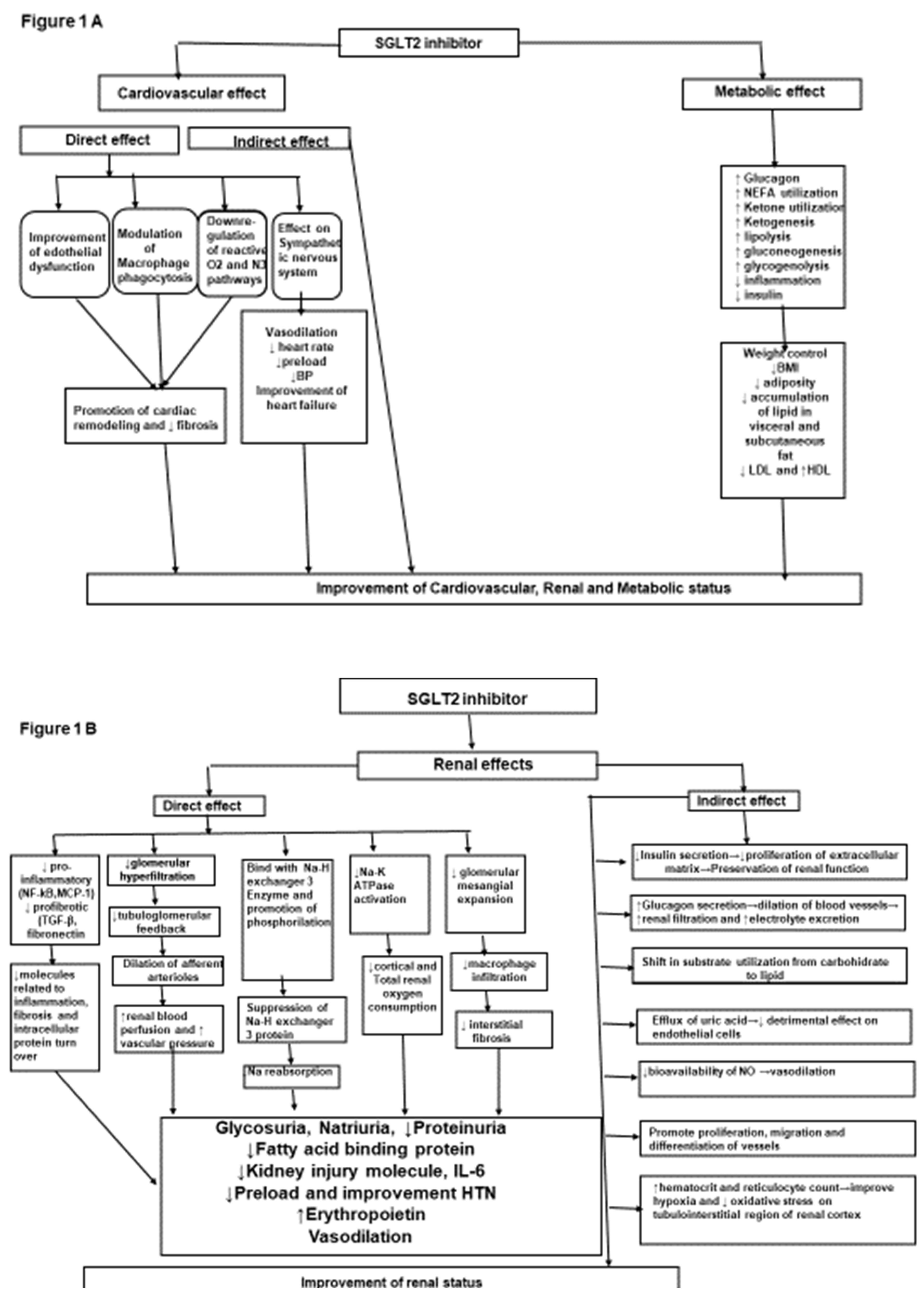
Mechanism of action
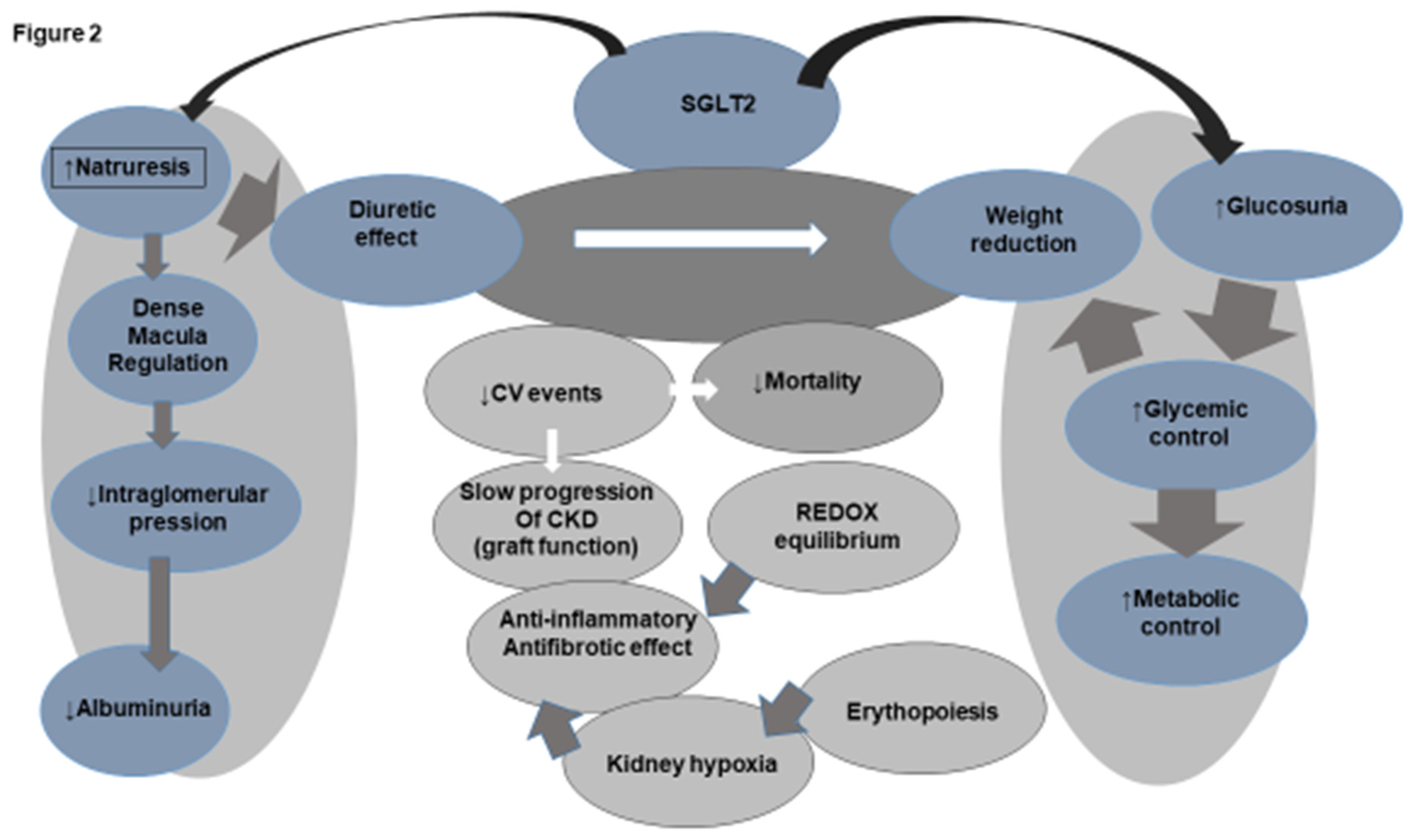
SGLT2i Action on Heart, Metabolism and Kidney in Patients without Diabetes
SGLT2 Inhibitors in Kidney Transplant Patients
Conclusions and Recommendation
- a)
- Start treatment with SGLT” inhibitors at least 6 months after transplantation
- b)
- Start treatment if no previous rejection happened
- c)
- Start treatment in patients with no history of UTI 6 months before starting therapy
- d)
- No history of vascular disease
Authors Contributions
Conflicts of Interest
References
- McGuire DK, Shih WJ, Cosentino F, Charbonnel B, Cherney DZI, Dagogo-Jack S, Pratley R, Greenberg M, Wang S, Huyck S, Gantz I, Terra SG, Masiukiewicz U, Cannon CP. Association of SGLT2 Inhibitors With Cardiovascular and Kidney Outcomes in Patients With Type 2 Diabetes: A Meta-analysis. JAMA Cardiol. 2021 ; 6:148-158.
- Hurren KM, Pinelli NR. Drug-drug interactions with glucagon-like peptide-1 receptor agonists. Ann Pharmacother. 2012; 46:710-717.
- Zinman B, Lachin JM, Inzucchi SE. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2016; 374, 1094.
- Neal B, Perkovic V, Matthews DR. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377: 2099.
- Lee TM, Chang NC, Lin SZ. Dapagliflozin, a selective SGLT2 Inhibitor, attenuated cardiac fibrosis by regulating the macrophage polarization via STAT3 signaling in infarcted rat hearts. Free Radic Biol Med. 2017; 104: 298-310.
- Hasan I, Rashid T, Jaikaransingh V, Heilig C, Abdel-Rahman EM, Awad AS. SGLT2 inhibitors: Beyond glycemic control. J Clin Transl Endocrinol. 2024 35:100335.
- List JF, Woo V, Morales E, Tang W, Fiedorek FT. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care. 2009; 32: 650-657.
- Dekkers CCJ, Sjöström CD, Greasley PJ, Cain V, Boulton DW, Heerspink HJL. Effects of the sodium-glucose co-transporter-2 inhibitor dapagliflozin on estimated plasma volume in patients with type 2 diabetes. Diabetes Obes Metab. 2019 ; 21: 2667-2673.
- Crawford PA. Refueling the Failing Heart: A Case for Sodium-Glucose Cotransporter 2 Inhibition in Cardiac Energy Homeostasis. JACC Basic Transl Sci. 2018; 3: 588-590.
- Packer M. SGLT2 Inhibitors Produce Cardiorenal Benefits by Promoting Adaptive Cellular Reprogramming to Induce a State of Fasting Mimicry: A Paradigm Shift in Understanding Their Mechanism of Action. Diabetes Care. 2020; 43: 508-511.
- Inoue MK, Matsunaga Y, Nakatsu Y, Yamamotoya T, Ueda K, Kushiyama A, Sakoda H, Fujishiro M, Ono H, Iwashita M, Sano T, Nishimura F, Morii K, Sasaki K, Masaki T, Asano T. Possible involvement of normalized Pin1 expression level and AMPK activation in the molecular mechanisms underlying renal protective effects of SGLT2 inhibitors in mice. Diabetol Metab Syndr. 2019; 11: 57.
- Chang YK, Choi H, Jeong JY, Na KR, Lee KW, Lim BJ, Choi DE. Dapagliflozin, SGLT2 Inhibitor, Attenuates Renal Ischemia-Reperfusion Injury. PLoS One. 2016 ; 11:e0158810.
- Aragón-Herrera A, Feijóo-Bandín S, Otero Santiago M, Barral L, Campos-Toimil M, Gil-Longo J, Costa Pereira TM, García-Caballero T, Rodríguez-Segade S, Rodríguez J, Tarazón E, Roselló-Lletí E, Portolés M, Gualillo O, González-Juanatey JR, Lago F. Empagliflozin reduces the levels of CD36 and cardiotoxic lipids while improving autophagy in the hearts of Zucker diabetic fatty rats. Biochem Pharmacol. 2019; 170: 113677.
- Mizuno M, Kuno A, Yano T, Miki T, Oshima H, Sato T, Nakata K, Kimura Y, Tanno M, Miura T. Empagliflozin normalizes the size and number of mitochondria and prevents reduction in mitochondrial size after myocardial infarction in diabetic hearts. Physiol Rep. 2018; 6: e13741.
- Ren FF, Xie ZY, Jiang YN, Guan X, Chen QY, Lai TF, Li L. Dapagliflozin attenuates pressure overload-induced myocardial remodeling in mice via activating SIRT1 and inhibiting endoplasmic reticulum stress. Acta Pharmacol Sin. 2022; 43:1721-1732.
- Wang CC, Li Y, Qian XQ, Zhao H, Wang D, Zuo GX, Wang K. Empagliflozin alleviates myocardial I/R injury and cardiomyocyte apoptosis via inhibiting ER stress-induced autophagy and the PERK/ATF4/Beclin1 pathway. J Drug Target. 2022; 30: 858-872.
- Heerspink HJL, Perco P, Mulder S, Leierer J, Hansen MK, Heinzel A, Mayer G. Canagliflozin reduces inflammation and fibrosis biomarkers: a potential mechanism of action for beneficial effects of SGLT2 inhibitors in diabetic kidney disease. Diabetologia. 2019; 62: 1154-1166.
- Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Ruff CT, Gause-Nilsson IAM, Fredriksson M, Johansson PA, Langkilde AM, Sabatine MS; DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2019; 380: 347-357.
- Scheen AJ. Drug-drug interactions with sodium-glucose cotransporters type 2 (SGLT2) inhibitors, new oral glucose-lowering agents for the management of type 2 diabetes mellitus. Clin Pharmacokinet. 2014; 53: 295-304.
- Kruger DF, Bode B, Spollett GR. Understanding GLP-1 analogs and enhancing patients success. Diabetes Educ. 2010 ;36 Suppl 3:44S-72S.
- Whaley JM, Tirmenstein M, Reilly TP, Poucher SM, Saye J, Parikh S, List JF. Targeting the kidney and glucose excretion with dapagliflozin: preclinical and clinical evidence for SGLT2 inhibition as a new option for treatment of type 2 diabetes mellitus. Diabetes Metab Syndr Obes. 2012; 5:135-148.
- Patel N, Hindi J, Farouk SS. Sodium-Glucose Cotransporter 2 Inhibitors and Kidney Transplantation: What Are We Waiting For? Kidney360. 2021; 2:1174-1178.
- Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, Cannon CP, Capuano G, Chu PL, de Zeeuw D, Greene T, Levin A, Pollock C, Wheeler DC, Yavin Y, Zhang H, Zinman B, Meininger G, Brenner BM, Mahaffey KW; CREDENCE Trial Investigators. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N Engl J Med. 2019; 380: 2295-2306.
- 24. Anker SD, Butler J, Filippatos G, Khan MS, Marx N, Lam CSP, Schnaidt S, Ofstad AP, Brueckmann M, Jamal W, Bocchi EA, Ponikowski P, Perrone SV, Januzzi JL, Verma S, Böhm M, Ferreira JP, Pocock SJ, Zannad F, Packer M. Effect of Empagliflozin on Cardiovascular and Renal Outcomes in Patients With Heart Failure by Baseline Diabetes Status: Results From the EMPEROR-Reduced Trial. Circulation. 2021 ;143: 337-349.
- Díaz-Cruz C, González-Ortiz M, Rosales-Rivera LY, Patiño-Laguna AJ, Ramírez-Rodríguez ZG, Díaz-Cruz K, Martínez-Abundis E. Effects of dapagliflozin on blood pressure variability in patients with prediabetes and prehypertension without pharmacological treatment: a randomized trial. Blood Press Monit. 2020 ; 25: 346-350.
- Petrie MC, Verma S, Docherty KF, Inzucchi SE, Anand I, Belohlávek J, Böhm M, Chiang CE, Chopra VK, de Boer RA, Desai AS, Diez M, Drozdz J, Dukát A, Ge J, Howlett J, Katova T, Kitakaze M, Ljungman CEA, Merkely B, Nicolau JC, O’Meara E, Vinh PN, Schou M, Tereshchenko S, Køber L, Kosiborod MN, Langkilde AM, Martinez FA, Ponikowski P, Sabatine MS, Sjöstrand M, Solomon SD, Johanson P, Greasley PJ, Boulton D, Bengtsson O, Jhund PS, McMurray JJV. Effect of Dapagliflozin on Worsening Heart Failure and Cardiovascular Death in Patients With Heart Failure With and Without Diabetes. JAMA. 2020; 323:1353-1368.
- Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, Brunner-La Rocca HP, Choi DJ, Chopra V, Chuquiure-Valenzuela E, Giannetti N, Gomez-Mesa JE, Janssens S, Januzzi JL, Gonzalez-Juanatey JR, Merkely B, Nicholls SJ, Perrone SV, Piña IL, Ponikowski P, Senni M, Sim D, Spinar J, Squire I, Taddei S, Tsutsui H, Verma S, Vinereanu D, Zhang J, Carson P, Lam CSP, Marx N, Zeller C, Sattar N, Jamal W, Schnaidt S, Schnee JM, Brueckmann M, Pocock SJ, Zannad F, Packer M; EMPEROR-Preserved Trial Investigators. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021; 385:1451-1461.
- Bays HE, Weinstein R, Law G, Canovatchel W. Canagliflozin: effects in overweight and obese subjects without diabetes mellitus. Obesity (Silver Spring). 2014 ; 22:1042-1049.
- Neeland IJ, de Albuquerque Rocha N, Hughes C, Ayers CR, Malloy CR, Jin ES. Effects of Empagliflozin Treatment on Glycerol-Derived Hepatic Gluconeogenesis in Adults with Obesity: A Randomized Clinical Trial. Obesity (Silver Spring). 2020 ; 28:1254-1262.
- Færch K, Blond MB, Bruhn L, Amadid H, Vistisen D, Clemmensen KKB, Vainø CTR, Pedersen C, Tvermosegaard M, Dejgaard TF, Karstoft K, Ried-Larsen M, Persson F, Jørgensen ME. The effects of dapagliflozin, metformin or exercise on glycaemic variability in overweight or obese individuals with prediabetes (the PRE-D Trial): a multi-arm, randomised, controlled trial. Diabetologia. 2021; 64: 42-55.
- Veelen A, Andriessen C, Op den Kamp Y, Erazo-Tapia E, de Ligt M, Mevenkamp J, Jörgensen JA, Moonen-Kornips E, Schaart G, Esterline R, Havekes B, Oscarsson J, Schrauwen-Hinderling VB, Phielix E, Schrauwen P. Effects of the sodium-glucose cotransporter 2 inhibitor dapagliflozin on substrate metabolism in prediabetic insulin resistant individuals: A randomized, double-blind crossover trial. Metabolism. 2023 ; 140:155396.
- Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, Mann JFE, McMurray JJV, Lindberg M, Rossing P, Sjöström CD, Toto RD, Langkilde AM, Wheeler DC; DAPA-CKD Trial Committees and Investigators. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2020 ; 383:1436-1446.
- The EMPA-KIDNEY Collaborative Group; Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, Emberson JR, Preiss D, Judge P, Mayne KJ, Ng SYA, Sammons E, Zhu D, Hill M, Stevens W, Wallendszus K, Brenner S, Cheung AK, Liu ZH, Li J, Hooi LS, Liu W, Kadowaki T, Nangaku M, Levin A, Cherney D, Maggioni AP, Pontremoli R, Deo R, Goto S, Rossello X, Tuttle KR, Steubl D, Petrini M, Massey D, Eilbracht J, Brueckmann M, Landray MJ, Baigent C, Haynes R. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2023 ; 388:117-127.
- Podestà MA, Sabiu G, Galassi A, Ciceri P, Cozzolino M. SGLT2 Inhibitors in Diabetic and Non-Diabetic Chronic Kidney Disease. Biomedicines. 2023; 11:279.
- Lim JH, Kwon S, Jeon Y, Kim YH, Kwon H, Kim YS, Lee H, Kim YL, Kim CD, Park SH, Lee JS, Yoo KD, Son HE, Jeong JC, Lee J, Lee JP, Cho JH. The Efficacy and Safety of SGLT2 Inhibitor in Diabetic Kidney Transplant Recipients. Transplantation. 2022;106: e404-e412.
- Sánchez Fructuoso AI, Bedia Raba A, Banegas Deras E, Vigara Sánchez LA, Valero San Cecilio R, Franco Esteve A, Cruzado Vega L, Gavela Martínez E, González Garcia ME, Saurdy Coronado P, Morales NDV, Zarraga Larrondo S, Ridao Cano N, Mazuecos Blanca A, Hernández Marrero D, Beneyto Castello I, Paul Ramos J, Sierra Ochoa A, Facundo Molas C, González Roncero F, Torres Ramírez A, Cigarrán Guldris S, Pérez Flores I. Sodium-glucose cotransporter-2 inhibitor therapy in kidney transplant patients with type 2 or post-transplant diabetes: an observational multicentre study. Clin Kidney J. 2023; 16:1022-1034.
- Rajasekeran H, Kim SJ, Cardella CJ, Schiff J, Cattral M, Cherney DZI, Singh SKS. Use of Canagliflozin in Kidney Transplant Recipients for the Treatment of Type 2 Diabetes: A Case Series. Diabetes Care. 2017;40:e75-e76.
- Shah M, Virani Z, Rajput P, Shah B. Efficacy and Safety of Canagliflozin in Kidney Transplant Patients. Indian J Nephrol. 2019; 29: 278-281.
- Schwaiger E, Burghart L, Signorini L, Ristl R, Kopecky C, Tura A, Pacini G, Wrba T, Antlanger M, Schmaldienst S, Werzowa J, Säemann MD, Hecking M. Empagliflozin in posttransplantation diabetes mellitus: A prospective, interventional pilot study on glucose metabolism, fluid volume, and patient safety. Am J Transplant. 2019 ; 19: 907-919.
- Halden TAS, Kvitne KE, Midtvedt K, Rajakumar L, Robertsen I, Brox J, Bollerslev J, Hartmann A, Åsberg A, Jenssen T. Efficacy and Safety of Empagliflozin in Renal Transplant Recipients With Posttransplant Diabetes Mellitus. Diabetes Care. 2019 ; 42:1067-1074.
- Mahling M, Schork A, Nadalin S, Fritsche A, Heyne N, Guthoff M. Sodium-Glucose Cotransporter 2 (SGLT2) Inhibition in Kidney Transplant Recipients with Diabetes Mellitus. Kidney Blood Press Res. 2019; 44: 984-992.
- Attallah N, Yassine L. Use of Empagliflozin in Recipients of Kidney Transplant: A Report of 8 Cases. Transplant Proc. 2019; 51: 3275-3280.
- Kong j, Joon J, Chul Y, Eun W, Hyuk K, Hyun SS Sodium/glucose cotransporter 2 inhibitor for the treatment of diabetes in kidney transplant patients Nephrol Dial Transplant 2019, 34.
- AlKindi F, Al-Omary HL, Hussain Q, Al Hakim M, Chaaban A, Boobes Y. Outcomes of SGLT2 Inhibitors Use in Diabetic Renal Transplant Patients. Transplant Proc. 2020; 52:175-178.
- Song CC, Brown A, Winstead R, Yakubu I, Demehin M, Kumar D, Gupta G. Early initiation of sodium-glucose linked transporter inhibitors (SGLT-2i) and associated metabolic and electrolyte outcomes in diabetic kidney transplant recipients. Endocrinol Diabetes Metab. 2020; 4:e00185.
- Lemke A, Brokmeier HM, Leung SB, Mara KC, Mour GK, Wadei HM, Hill JM, Stegall M, Kudva YC, Shah P, Kukla A Sodium-glucose cotransporter 2 inhibitors for treatment of diabetes mellitus after kidney transplantation. Clin Transplant. 2022 ; 36: e14718.
- Oikonomaki D, Dounousi E, Duni A, Roumeliotis S, Liakopoulos V. Incretin based therapies and SGLT-2 inhibitors in kidney transplant recipients with diabetes: A systematic review and meta-analysis. Diabetes Res Clin Pract. 2021;172:108604.
- Nissaisorakarn P, Pavlakis M, Aala A. Novel Glucose-Lowering Therapies in the Setting of Solid Organ Transplantation. Adv Chronic Kidney Dis. 2021; 28: 361-370.
- Kanbay M, Demiray A, Afsar B, Karakus KE, Ortiz A, Hornum M, Covic A, Sarafidis P, Rossing P. Sodium-glucose cotransporter 2 inhibitors for diabetes mellitus control after kidney transplantation: Review of the current evidence. Nephrology (Carlton). 2021; 26:1007-1017.
- Chewcharat A, Prasitlumkum N, Thongprayoon C, Bathini T, Medaura J, Vallabhajosyula S, Cheungpasitporn W. Efficacy and Safety of SGLT-2 Inhibitors for Treatment of Diabetes Mellitus among Kidney Transplant Patients: A Systematic Review and Meta-Analysis. Med Sci (Basel). 2020; 8: 47.
- Schwarzenbach M, Bernhard FE, Czerlau C, Sidler D. Chances and risks of sodium-glucose cotransporter 2 inhibitors in solid organ transplantation: A review of literatures. World J Transplant. 2021;11: 254-262.
- Kwon H, Son SH, Kim K Sodium-Glucose Cotransprter 2 inhibitors reduce microalbuminuria in diabetic renal transplant patients. Transplantation 104 (S3): p S430.
- Menne J, Dumann E, Haller H, Schmidt BMW. Acute kidney injury and adverse renal events in patients receiving SGLT2-inhibitors: A systematic review and meta-analysis. PLoS Med. 2019;16: e1002983.
| Author, Year, Study type, Follow-up | Basal eGFR (ml/min) |
Effect in Renal Function (eGFR mL/min/1.73) | Proteinuria (uPCR) g/d/(uACR mg/g | Adverse Events |
|---|---|---|---|---|
| Rajasekeran, (37) 2017, CS n6, 8mo | 78.6±18.2 | No differences | NA | Cellulitis |
| Shah, (38) 2019, PS n25, 8mo | 86±20 | No differences | NA | None |
| Schwaiger,(39) 2019, PS, n14, 12mo | 55.6±20.3 | Decrease and then stabilize | ΔuACR: -25 ΔuACR: -73 |
UTI 5 |
| Halden, (40) 2019, RCT, n44, 6mo | 66±10.5 | No differences | NA | UTI 3 |
| Mahling, (41) 2019, PS, n10, 6mo | 57±19.3 | No differences | NA | UTI 2 |
| Attallah, (42) 2019, CS, n25, 12mo | NA | Decrease and than stabilize | ΔuPCR -0.6 g/d | UTI 2 |
| Kong, (43) 2019, PS, n42, 12mo | 60.36±17 | No differences | ΔuACR No significant change | Acute cystitis 3 |
| Alkindi,(44) 2020, CS, n8, 12mo | 75.8±13.4 | No differences | NA | UTI 1 |
| Song, (45) 2021, RS, n50, 6mo | 66.7 | No differences | NA | UTI 7 |
| Lemke,(46) 2021, RS, n39, 12mo | NA | No differences | NA | UTI 6, Ketoacidosis 1 |
| S-Fructuoso,(36) 2022, MCO, n339, 12mo | 58.4 (56.2-60.6) | No differences | ΔuPCR: -230 at 6 mo | UTI 14%, AKI 1.8% |
| Lim, (35) 2022, OR, PSM, n2083, 63mo | S: 66.9±17.7 C: 68.4±20.1 |
Decrease, stabilization and amelioration | ΔuPCR: the urine PCR significantly decreased after SGTLi p=0.005 | NA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





