1. Introduction
Antibiotics are biologically active substances with an extensive use in human and veterinary medicine as therapeutics or growth promoters [
1]. A large amount of antibiotic (50 to 90%) are excreted from the body as unchanged form through urine, feces and manure, eventually reaching the water environment through sewage treatment plants, soil amendment or organic fertilizers used in the agricultural fields [
2,
3]. In addition, antibiotics may directly reach the water environment through the wastes of pharmaceutical plants and hospitals [
4]. Thus, antibiotic contamination have commonly been detected in waste water effluents, surface waters, ground waters, sea water and living organism [
5,
6,
7,
8,
9]. This causes the development of antibiotic resistance genotypes of bacteria [
10] and the fatal risk to the indigenous microorganisms [
11] and threatened to the ecosystem and human life. In addition, their sustained release to the various environmental compartments produce complex toxicities after mixing with other chemical substances in the environment. Thus there is a great concern in monitoring the overall antibiotic toxicities in the water environment by using appropriate analytical methods. Chemical analysis methods are used to determine the absolute concentration of known single pollutant. However, these methods are not suitable for evaluating the overall toxicities in the water environment.
Consequently, bioassay have been developed to determine the overall toxicities in the environment using fish [
12], animal cell, luminescent bacteria [
13], algae [
14,
15] and microorganisms [
16,
17,
18,
19,
20,
21]. Microorganisms have been extensively used in bioassay because of their high sensitivity towards toxin, easy manipulation [
22,
23,
24,
25,
26,
27,
28,
29]. Furthermore, microorganism are a primary producer of the ecosystem and exhibits similar physiological response as higher organisms [
30], thus the toxic impact analysis on microorganism help us to know the influence on higher organism by illuminating any small changes in water quality [
31,
32].
There are many benefits of electrochemical detection techniques in bioassay, such as relatively simple, sensitive, rapid and inexpensive method. In addition, it is applicable to evaluate the toxicities in color sample with suspended solid containing analytes. In several recent works, the electrochemical bioassay using redox mediators have been developed to evaluate the toxicity [
33,
34,
35,
36,
37]. The mediators participated in the microorganism respiration [
38] and accept the electrons from the respiratory chains. Thus, the currents of reduced mediators reflects the microbial activity. In response to a toxicant, the microbial activity is disturbed which can be easily evaluated as a deviation away from the currents produced by healthy cells. This approach enables a rapid and reliable screening for the evaluation of toxicity [
34,
39].
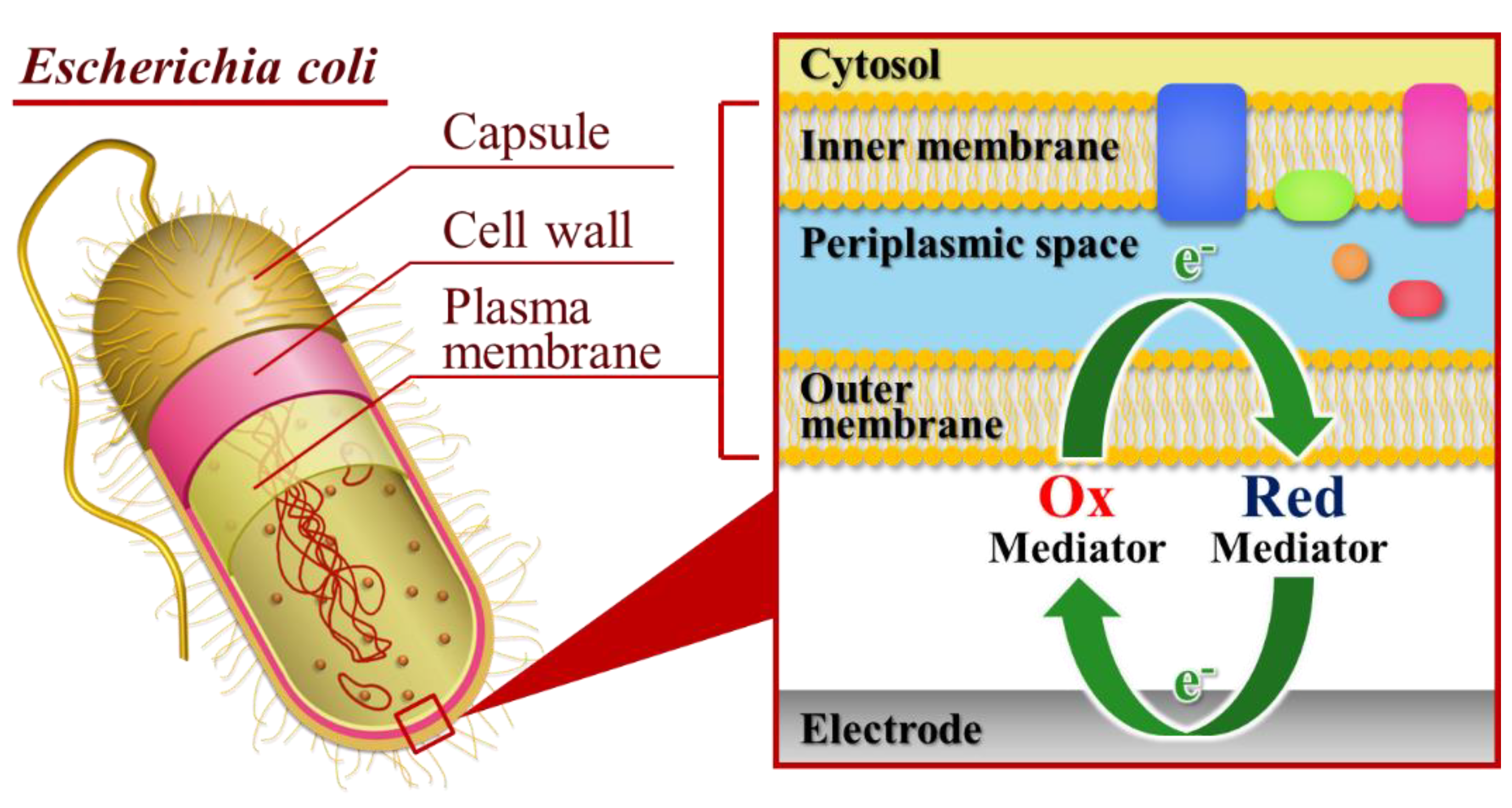
The aim of this study is to develop a new electrochemical bioassay based on the rotating droplet electrochemistry using an electron-transfer mediator. Ferricyanide was used as electron transfer. The mediator participates in the respiration chain of
Escherichia coli by accepting the electron in periplasmic space and oxidized at the electrode surface as shown in
Figure 1. Thus, the oxidation current of mediator can reflect the microbial activity. In the presence of pollutants, the capacity of the central function of cellular metabolism is disturbed, caused a change of current flow compared to the healthy cells. In this technique which enables the hydrodynamic amperometric analysis in a microliter volume of sample solution, the rotating disk electrode (RDE) exhibits dual functions, such as, it can be used to efficiently blend the sample solution and concurrently acts as an electrochemical detection device. The RDE promotes the convective mass transport to the electrode surface because of the fast rotation speed. As a result, it causes effective mixing that reduces the incubation time for the biochemical reaction. The advantages acquired from RDE detection in a microdroplet (40 μL) reduced the diffusional distance, which results in shorter assay time, rapid detection, and, therefore, faster assay times. In addition, another benefit of using microdroplet is the decrease in dilution of sample, which causes lower detection limits [
40,
41]. Thus using RDE for detecting the electrochemical reaction in microliter sample solution is of great interest.
2. Materials and Methods
2.1. Reagent and Solution Preparation
Potassium hexacyanoferrate (Ш) was purchased from Sigma-Aldrich (St. Louis, MO, USA) and dissolved in phosphate buffer solution. Ampicillin, tetracycline, sulfamonomethoxine, chloroteracycline, oxytetracycline were obtained from Sigma-Aldrich (St. Louis, MO, USA). The phosphate buffer solution (PBS, pH 7.4) contained 0.044 M NaH2PO4, 0.056 M NaH2PO4. All reagents used were of analytical grade, and the water was sterile deionized water. Bacto tryptone was purchased from Difco Company (Detroit, MI, USA). The Luria–Bertani (LB: 10.0 g/L bacto tryptone, 5.0 g/L yeast extract, and 10.0 g/L NaCl) broth was sterilized in high-pressure steam at 120˚C for 20 min.
2.2. Culture, Maintenance and Harvesting the E. coli
Escherichia coli K-12 strain was used as a bacterial stain. E.coli K-12 was cultured on nutrient agar plates at 4˚C, replicated regularly to confirm its viability. A 200 ml solution of autoclaved broth was inoculated with a colony of E.coli, then incubated anaerobically at 37˚C for 5 hours to reach the stationary phase. Later on, the microorganisms were harvested by centrifuging at 3000 rpm for 30 min and resuspended in PBS. The solution was diluted 10-fold with PBS buffer and checked the absorbance value at 600 nm (OD600) by using UV-2450 UV/Vis detector (Japan spectroscopic company Co., Tokyo Japan). The bacterial suspension was used for the experiments on the day of harvesting.
2.3. Instrumentation and Selection of Electrodes
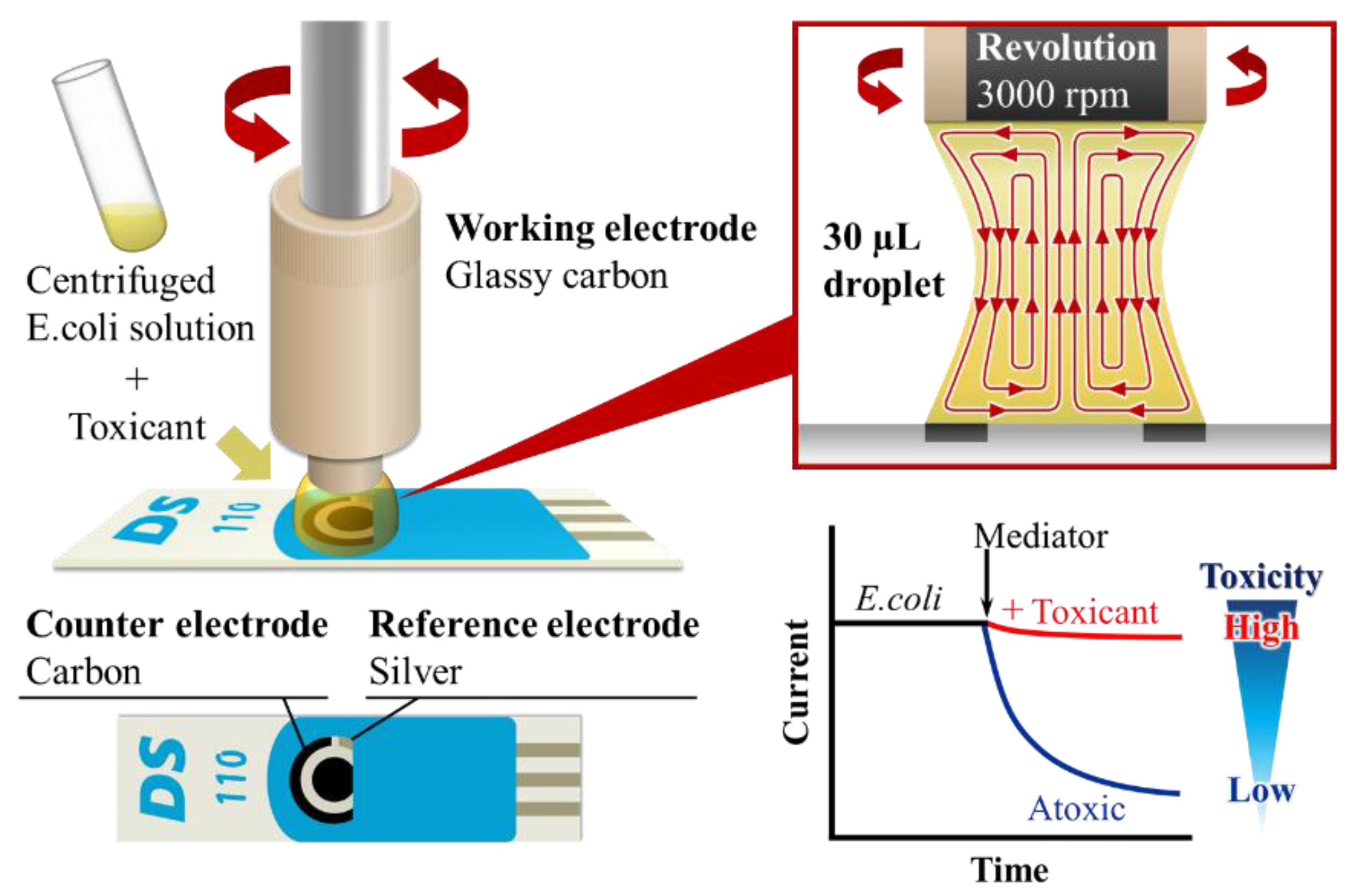
Hydrodynamic amperometry with a rotating disk electrode (RDE) were carried out using an electrochemical analyzer (ALS-1200, Bioanalytical systems Inc. (BAS) (West Lafayette, IN, USA). The thick-film screen printed electrochemical micro-device printed onto a PVC pad with a size of 3.4×1.0×0.05 cm (DRP-110, Dropsens) and embedded with solvent resistant three electrodes system, like reference, working and counter electrode. The glassy carbon, carbon and Ag/AgCl of screen-printed electrode were used as working, counter and reference electrode, respectively. The box connector (DSC, Dropsens) was used to obtain the electrochemical interface between the RDE, SPCE and the potentiostat. Before starting the measurement, the surface area of the glassy carbon disk electrode was polished, consecutively by adding 0.3 and 0.05 µm alumina paste and later washed well with water.
2.4. Procedures of Experimental Equipment Settings and Electrochemical Detection
The RDE was placed perpendicular to the screen print electrode and contacted at a distance of 2.5 mm.
E.coli solution (40 µL) was kept in center position between SPCE and RDE. The optimum potential for the ferricyanide (300 mV) was applied to the electrode and the rotation rate was fixed at 3,000 rpm. The assay for antibiotic toxicity on
E.coli was demonstrated by pre-incubation test method. The measurement was carried out with 40 µL of
E.coli and toxicant mixture and 30 s later 10 µL mediator solution was added. In this test method, the antibiotic was firstly added during the incubation of
E.coli. The hydrodynamic chronoamperograms were recorded for 300 s (
Figure 2).
3. Results and Discussion
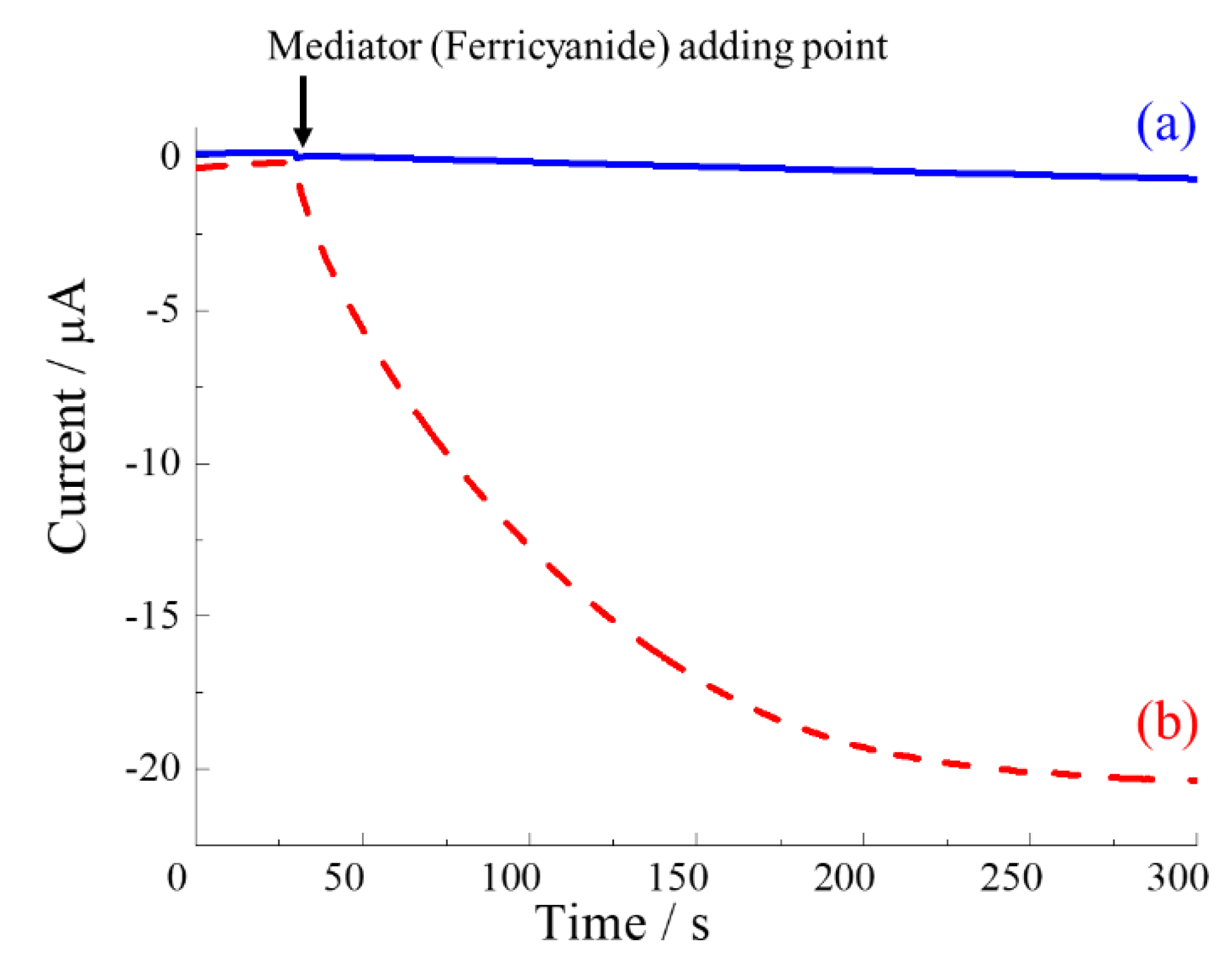
3.1. Investigation of Catalytic Response of E.coli
Figure 3 demonstrates the catalytic response of
E.coli in presence of ferricyanide mediator by using hydrodynamic chronoamperometry. In comparing with baseline current, a sharp increase of response current after adding the ferricyanide (1 mM) with the
E.coli was observed at 300 s. The obtained current response was increased to 20 µA. This result indicated that ferricyanide were mediated by the catalytic response of
E.coli. The current response was measured at higher (µA level) even at higher concentration of ferricyanide (45 mM; nA current) reported in other study [
42]. This might be due to the high rotation rate of RDE which allows the rapid mass transfer [
43] to the electrode surface.
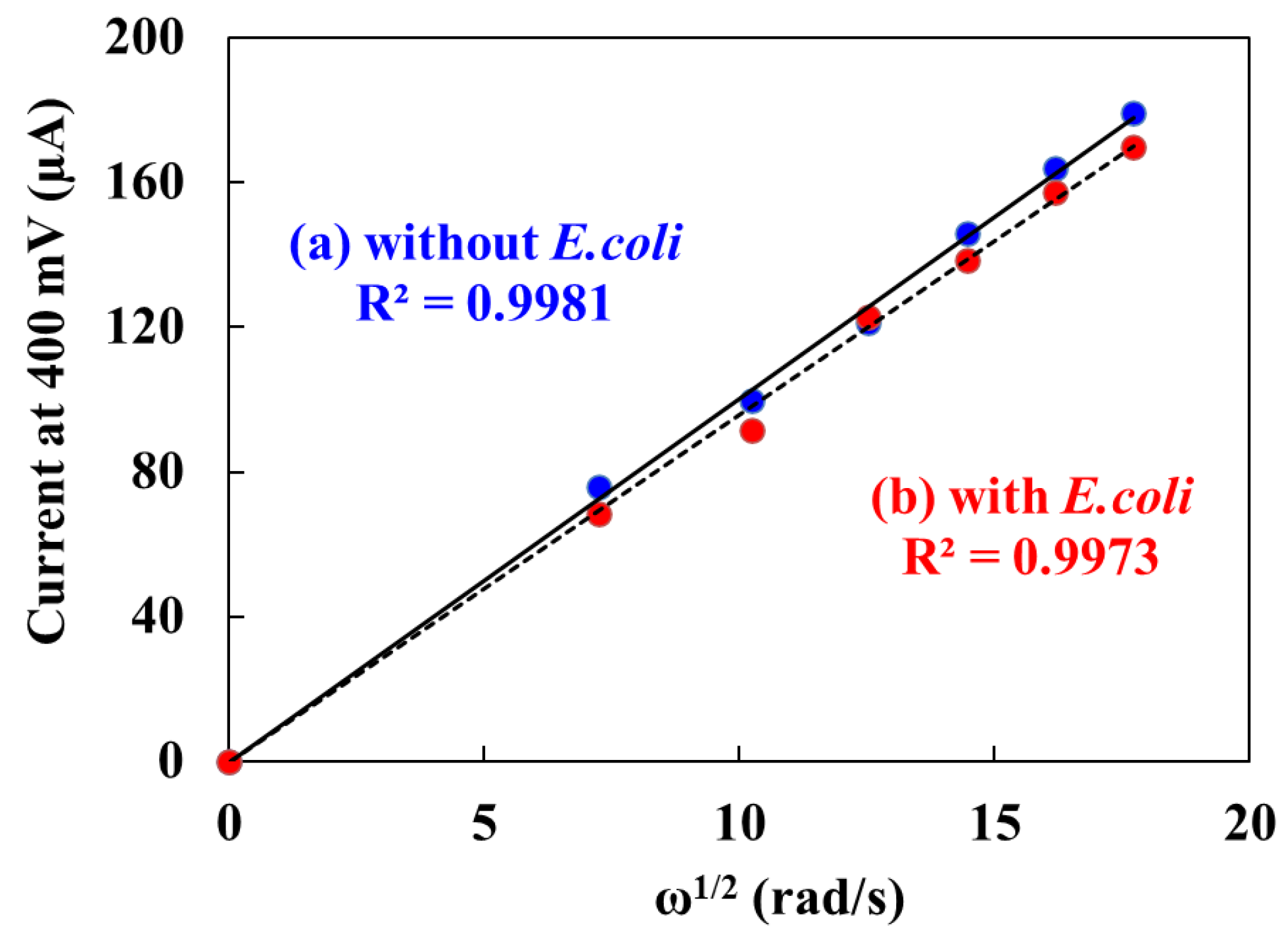
3.2. Effect of E. coli on the Electrochemical Detection
The effect of
E.coli on the electrochemical detection were evaluated by the hydrodynamic linear sweep voltammetry (
Figure 4). Fifty μL of droplet including 10 μL of ferrocyanide with
E.coli or PBS buffer were measured with different rotation rates of 500, 1,000, 1,500, 2,000, 2,500 and 3,000 rpm at a scan rate of 100 mV·s
-1. The Levich’s equation [
40] was used to find the relationship between mass–transfer limited current for oxidation of ferrocyanide and rotation of angular velocity. The equation is as follows,
where i
l is the mass-transfer limited current,
n is the number of electrons involved in the reaction, C
0 is the analyte concentration, ν is the kinematic viscosity of the fluid, and ω is the angular velocity of the disk (2π × rpm). The limiting currents showed a positive linear relationship with square root of ω in presence of
E.coli. This indicates that the electrochemical response of
E.coli depends on the convective mass transport of mediator resulting from the hydrodynamic flow caused by RDE. The limiting currents also showed a similar positive linear relationship with square root of ω in absence of
E.coli. This indicates that the electrochemical reaction of mediator is not influenced by the high concentration of
E.coli. Therefore,
E. coli did not affect the electrochemical reaction of mediator.
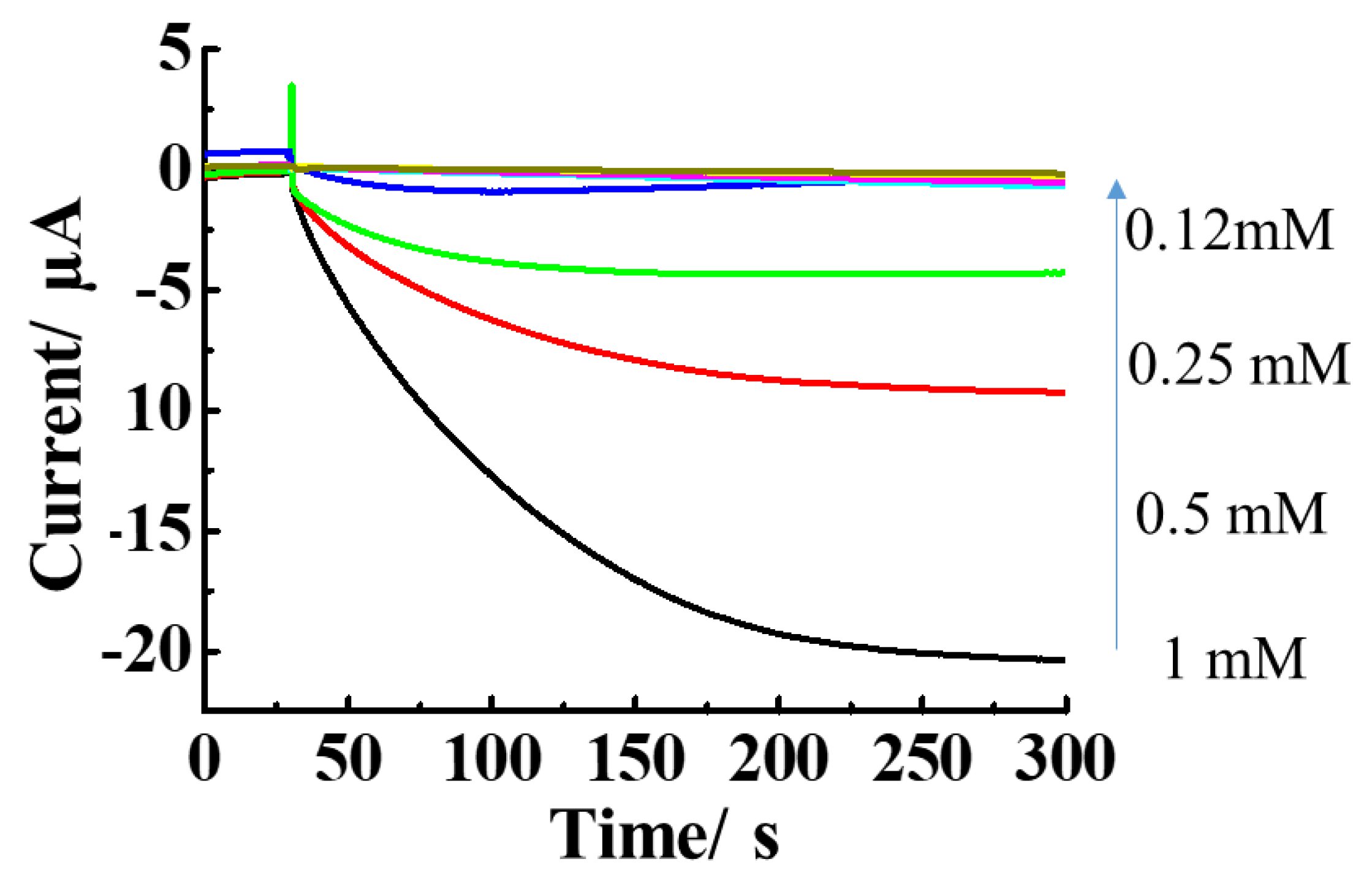
3.3. Effects of Mediator Concentration on E.coli Catalytic Response
Figure 5 shows the results for demonstration of the hydrodynamic chronoamperograms obtained from different concentrations of ferricyanide. Each concentration of ferricyanide (10 µL) was injected to 40 µL of
E.coli solution. There was an increment of anodic current with increasing the concentration of ferricyanide added into the droplet solution containing
E.coli. This result was found within only 300 s. The increase of catalytic response of
E.coli was observed with the increment of concentration of ferricyanide. When the concentration of ferricyanide was increased to 20 mM, there will be more ferricyanide transforming into ferrocyanide and oxidized at the electrode surface. As a result the greatest response current was observed at high concentration of ferricyanide. On the other hand, at minimum concentration of 0.25 mM of ferricyanide, the smallest response current was detected, indicated that the catalytic response of
E.coli depends on the concentration of ferricyanide which was consistent with a previous report [
42].
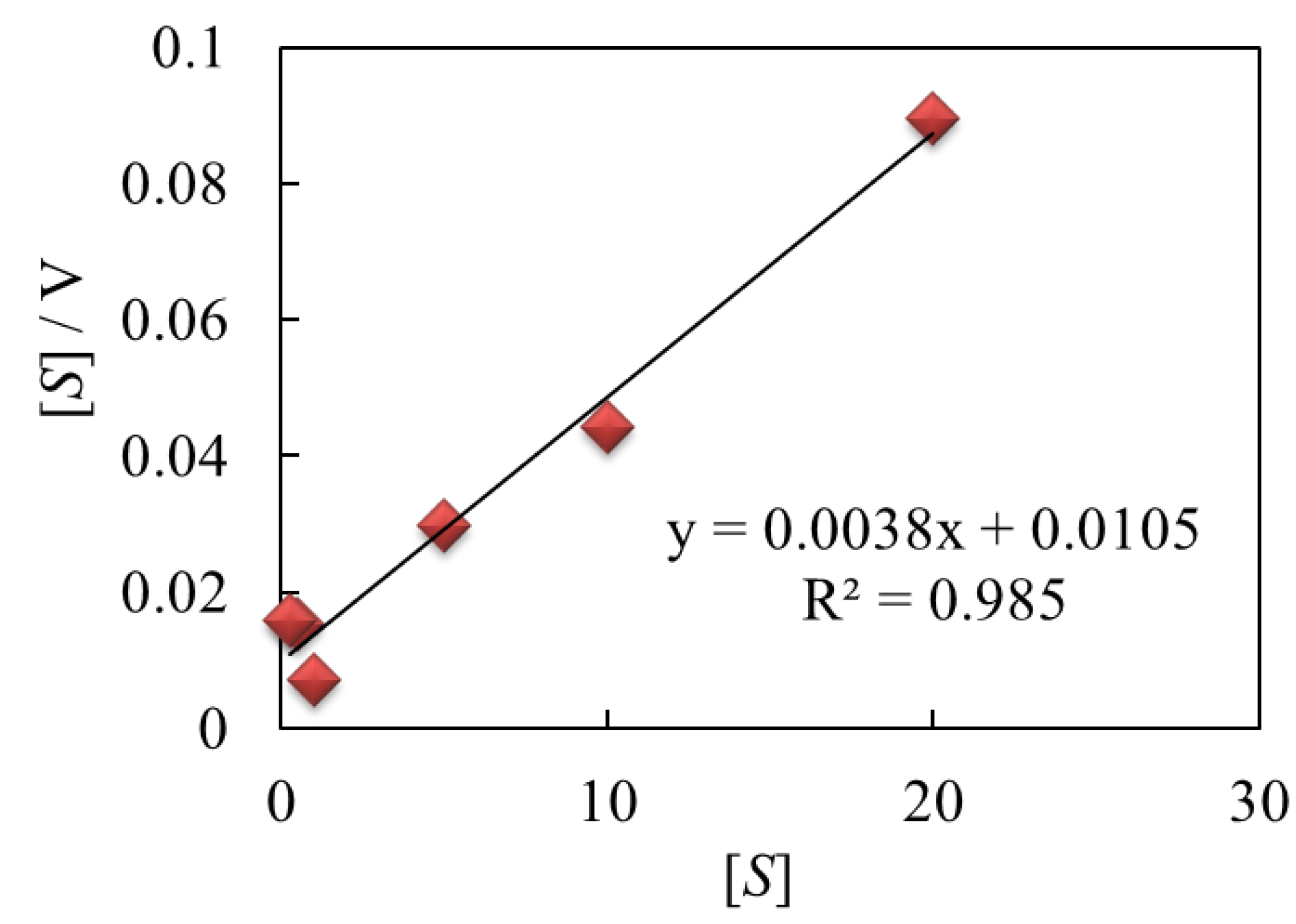
The hydrodynamic amperometric responses of
E.coli with different concentrations of ferricyanide were superbly fitted by using Michaelis-Menten equation:
The Michaelis-Menten constant (KM) and the maximum velocity (vmax) acquired from the Hanes-Woolf plots was 0.27 mM at a rotation rate of 3,000 rpm. This clearly indicated that the hydrodynamic amperometry with RDE system is applicable in investigating the catalytic activity of E.coli for performing the toxicity test. In addition, the catalytic response of E.coli was even observed at a low concentration of ferricyanide.
Figure 6.
Hanes-Woolf plots of ferricyanide in E.coli solution obtained by the RDE system at 300 mV applied potential with a rotation rate of 3,000 rpm.
Figure 6.
Hanes-Woolf plots of ferricyanide in E.coli solution obtained by the RDE system at 300 mV applied potential with a rotation rate of 3,000 rpm.
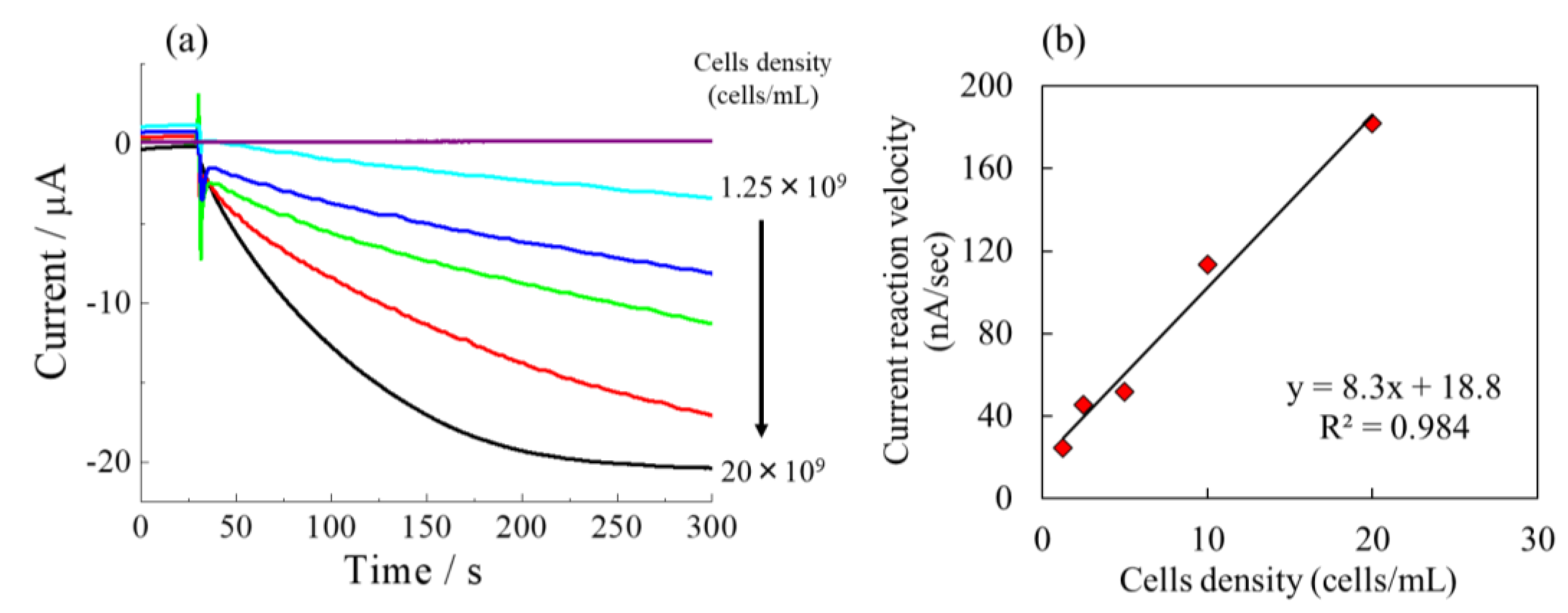
3.4. Effects of E.coli Concentration on Catalytic Response
The relationship between catalytic response and the concentrations of
E.coli was investigated as shown in
Figure 7. The increase of current mediated by the ferricyanide was observed with the increment of cell densities of
E.coli. When the cell density of
E.coli increased to 20×10
9 cells/mL, there will be more
E.coli to show the catalytic response. As a result, the greatest response current was observed at high call densities of
E.coli. On the other hand, at minimum cell density of 1.25×10
9 cells/mL of
E.coli, the smallest response current was detected, indicated that the catalytic current depends on the
E.coli cell densities. In addition, the proposed method can detected the catalytic response at a low concentration of
E.coli solution.
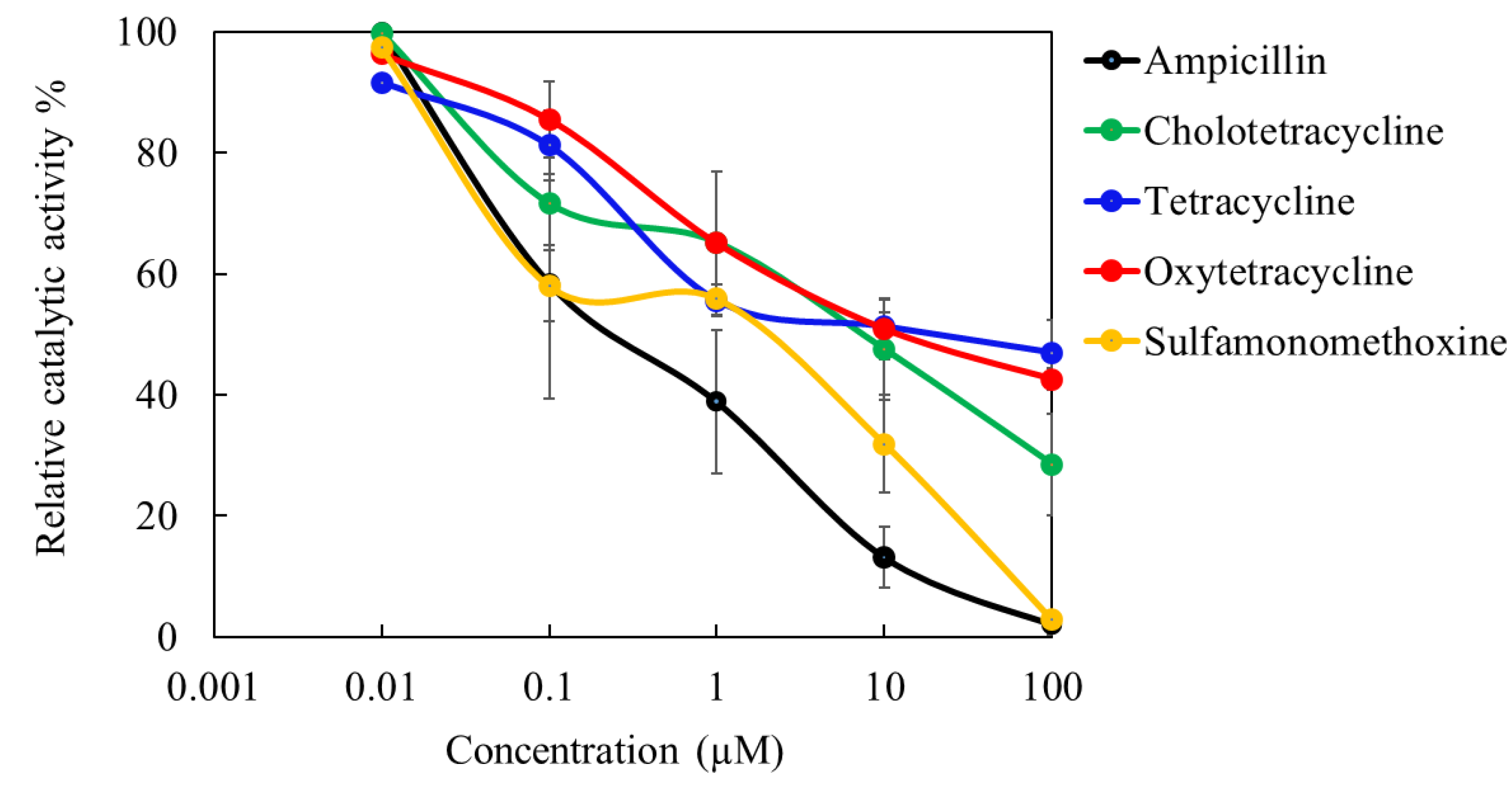
3.5. Evaluation of Toxicities of Antibiotics on E.coli
The dose-response curves of total 5 antibiotics obtained by using the proposed pre-incubation method are shown in
Figure 8. Electrochemical response of
E.coli is decreased resulting a deviation of current flow compared to the unaffected cells. This change of current is considered as the bacterial inhibition to those pollutants. The EC
50 value for ampicillin, sulfamonomethoxine, chlorotetracycline, oxytetracycline, and tetracycline were calculated as 0.26, 0.77, 5.25, 19.0, and 18.5 µM and the toxicity order was ampicillin > sulfamonomethoxine > chlorotetracycline > tetracycline > oxytetracyclien.
3.6. Mean Recovery Test
In order to evaluate the possible analytical applications of the proposed toxicity test, various concentrations (1 and 10 µM) of tetracycline was evaluated in different matrixes, such as powder milk, pond water and raw milk, as shown in
Table 1. The levels of antibiotics in powder milk, pond water and raw milk were almost similar with the levels of experimental sample, indicated that the proposed method can be usable for the determination of toxicity in real sample. In addition, recovery studies were done for testing the accuracy of the proposed method. In all cases, mean recovery values ranged between 90 and 99%, as shown in
Table 1. The value of relative standard deviation was small. These observations showed that the proposed method is precise and reproducible.
Table 1.
Recovery study of tetracycline in different matrixes.
Table 1.
Recovery study of tetracycline in different matrixes.
| Matrix |
Additional Concentration of Tetracycline (µM) |
Found Concentration
(µM) |
Recovery (%) |
RSD (%) |
| Pond water |
1 |
0.97 |
97 |
2.1 |
| 10 |
9.61 |
96 |
3.8 |
| Milk |
1 |
0.90 |
90 |
10 |
| 10 |
9.02 |
90 |
10 |
| Powder milk |
1 |
0.99 |
99 |
0.18 |
| 10 |
9.27 |
92 |
7.7 |
4. Conclusions
The result presented in this study confirm that the electrochemical bioassay based on the rotating droplet electrochemistry successfully evaluate the catalytic response of E.coli in presence of ferricyanide. The pre-incubation test method can evaluate the toxicities of antibiotics in wide variety of real sample with a good accuracy. Because the proposed method did not require additional incubation time, it can evaluate the water toxicity within short period of time.
Author Contributions
Conceptualization, H.K; methodology, K.S. and H.K.; investigation, K.S., A.Y. and H.K.; resources, K.S., K.S. (Kazuharu Sugawara), and H.K.; writing—original draft preparation, A.Y.; writing—review and editing, K.S., K.S. (Kazuharu Sugawara) and H.K.; visualization, K.S., A.Y., and H.K.; supervision, K.S. and H.K.; project administration, H.K.; funding acquisition, H.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Japan Society for the Promotion of Science (JSPS) via a Grant-in-Aid for Scientific Research (Grant JP16H02976 and 23K22894).
References
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B.A. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Schlüsener, M.P.; Bester, K. Persistence of antibiotics such as macrolides, tiamulin and salinomycin in soil. Environ. Pollut. 2006, 143, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Elmund, G.K.; Morrison, S.M.; Grant, D.W.; Nevins, M.P. Role of excreted chlortetracycline in modifying the decomposition process in feedlot waste. Bull. Environ. Contam. Toxicol. 1971, 6, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, M.E.; Meyer, M.; Thurman, E.M. Analysis of trace levels of sulfonamide and tetracycline antimicrobials in groundwater and surface water using solid-phase extraction and liquid chromatography/mass spectrometry. Anal. Chem. 2001, 73, 4640–4646. [Google Scholar] [CrossRef] [PubMed]
- Kolpin, D.W.; Furlong, E.T.; Meyer, M.T.; Thurman, E.M.; Zaugg, S.D.; Barber, L.B.; Buxton, H.T. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999−2000: A national reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Weinberg, H.S.; Meyer, M.T. Trace analysis of trimethoprim and sulfonamide, macrolide, quinolone, and tetracycline antibiotics in chlorinated drinking water using liquid chromatography electrospray tandem mass spectrometry. Anal. Chem. 2007, 79, 1135–1144. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.Y.-C.; Tsai, Y.-T. Occurrence of pharmaceuticals in Taiwan’s surface waters: Impact of waste streams from hospitals and pharmaceutical production facilities. Sci. Total Environ. 2009, 407, 3793–3802. [Google Scholar] [CrossRef] [PubMed]
- Dolliver, H.; Kumar, K.; Gupta, S. Sulfamethazine uptake by plants from manure-amended soil. J. Environ. Qual. 2007, 36, 1224–1230. [Google Scholar] [CrossRef]
- Kong, W.D.; Zhu, Y.G.; Liang, Y.C.; Zhang, J.; Smith, F.A.; Yang, M. Uptake of oxytetracycline and its phytotoxicity to alfalfa (Medicago sativa L.). Environ. Pollut. 2007, 147, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Aga, D.S. Potential ecological and human health impacts of antibiotics and antibiotic-resistant bacteria from wastewater treatment plants. J. Toxicol. Environ. Heal. Part B 2007, 10, 559–573. [Google Scholar] [CrossRef]
- Flaherty, C.M.; Dodson, S.I. Effects of pharmaceuticals on aphnia survival, growth, and reproduction. Chemosphere 2005, 61, 200–207. [Google Scholar] [CrossRef] [PubMed]
- van der Schalie, W.H.; Shedd, T.R.; Knechtges, P.L.; Widder, M.W. Using higher organisms in biological early warning systems for real-time toxicity detection. Biosens. Bioelectron. 2001, 16, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Thomulka, K.; McGee, D.; Lange, J. Use of the bioluminescent bacterium Photobacterium phosphoreum to detect potentially biohazardous materials in water. Bull. Environ. Contam. Toxicol. 1993, 51, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Xu, L.; Wang, S.; Zheng, R.; Jin, S.; Huang, S.; Huang, Y. Toxicity of 40 herbicides to the green alga Chlorella vulgaris. Ecotoxicol. Environ. Saf. 2002, 51, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Islam, M.S.; Sazawa, K.; Hata, N.; Taguchi, S.; Nakamura, S.; Sugawara, K.; Kuramitz, H. Development of an electrochemical bioassay based on the alkaline phosphatase activity of to assess the toxicity of heavy metals. Int. J. Electrochem. Sci. 2016, 11, 5090–5102. [Google Scholar] [CrossRef]
- Zhu, B.; Wu, Z.-F.; Li, J.; Wang, G.-X. Single and joint action toxicity of heavy metals on early developmental stages of chinese rare minnow (Gobiocypris rarus). Ecotoxicol. Environ. Saf. 2011, 74, 2193–2202. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Ngoc, H.; Durrieu, C.; Tran-Minh, C. Synchronous-scan fluorescence of algal cells for toxicity assessment of heavy metals and herbicides. Ecotoxicol. Environ. Saf. 2009, 72, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Fu, G.; Tao, L.; Zhu, C. Roles of nitric oxide in alleviating heavy metal toxicity in plants. Arch. Biochem. Biophys. 2010, 497, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Cai, H.; Xu, Y.; Xiao, L.; Yang, M.; Wang, P. Detection of heavy metal toxicity using cardiac cell-based biosensor. Biosens. Bioelectron. 2007, 22, 3224–3229. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yao, J.; Si, Y.; Chen, H.; Russel, M.; Chen, K.; Qian, Y.; Zaray, G.; Bramanti, E. Short-time effect of heavy metals upon microbial community activity. J. Hazard. Mater. 2010, 173, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.H.A.; Van Ginkel, S.W.; Kim, S.-M.; Yoon, S.-H.; Joo, J.-H.; Shin, B.-S.; Jeon, B.-H.; Bae, W.; Oh, S.-E. Isolation and characterization of acidithiobacillus caldus from a sulfur-oxidizing bacterial biosensor and its role in detection of toxic chemicals. J. Microbiol. Methods 2010, 82, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Tizzard, A.; Webber, J.; Gooneratne, R.; John, R.; Hay, J.; Pasco, N. MICREDOX: Application for rapid biotoxicity assessment. Anal. Chim. Acta 2004, 522, 197–205. [Google Scholar] [CrossRef]
- Fulladosa, E.; Debord, J.; Villaescusa, I.; Bollinger, J.C.; Murat, J.C. Effect of arsenic compounds on Vibrio fischeri light emission and butyrylcholinesterase activity. Environ. Chem. Lett. 2007, 5, 115–119. [Google Scholar] [CrossRef]
- Hernando, M.; Malato, O.; Farré, M.; Fernandez-Alba, A.; Barceló, D. Application of ring study: Water toxicity determinations by bioluminescence assay with Vibrio fischeri. Talanta 2006, 69, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Lee, P.Y.; Ho, B.; Ding, J.L.; Lim, C.T. Atomic force microscopy study of the antimicrobial action of Sushi peptides on Gram negative bacteria. Biochim. Biophys. Acta - Biomembr. 2007, 1768, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Pasco, N.; Joanne, H.; Webber, J. Biosensors: MICREDOX - a new biosensor technique for rapid measurement of BOD and toxicity. Biomarkers 2001, 6, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.R.; Jordinson, G.M.; Rawson, D.M.; Rogerson, J.G. Biosensors for the measurement of toxicity of wastewaters to activated sludge. Pestic. Sci. 1998, 54, 447–452. [Google Scholar] [CrossRef]
- Farré, M.; Pasini, O.; Carmen Alonso, M.; Castillo, M.; Barceló, D. Toxicity assessment of organic pollution in wastewaters using a bacterial biosensor. Anal. Chim. Acta 2001, 426, 155–165. [Google Scholar] [CrossRef]
- Hsieh, C.-Y.; Tsai, M.-H.; Ryan, D.K.; Pancorbo, O.C. Toxicity of the 13 priority pollutant metals to Vibrio fisheri in the Microtox® chronic toxicity test. Sci. Total Environ. 2004, 320, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, M.; Wang, X.; Wu, Z.; Yang, L.; Xia, S.; Chen, L.; Zhao, J. P-benzoquinone-mediated amperometric biosensor developed with Psychrobacter sp. for toxicity testing of heavy metals. Biosens. Bioelectron. 2013, 41, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Stauber, J.L.; Davies, C.M. Use and limitations of microbial bioassays for assessing copper bioavailability in the aquatic environment. Environ. Rev. 2000, 8, 255–301. [Google Scholar] [CrossRef]
- Walsh, G.E.; Merrill, R.G. Algal bioassays of industrial and energy process effluents. In Algae as Ecological Indicators, Schubert, L.E., Eds.; Academic Press, London, UK, 1984; pp. 329–360.
- Liu, C.; Yong, D.; Yu, D.; Dong, S. Cell-based biosensor for measurement of phenol and nitrophenols toxicity. Talanta 2011, 84, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sun, T.; Xu, X.; Dong, S. Direct toxicity assessment of toxic chemicals with electrochemical method. Anal. Chim. Acta 2009, 641, 59–63. [Google Scholar] [CrossRef]
- Yong, D.; Liu, C.; Yu, D.; Dong, S. A sensitive, rapid and inexpensive way to assay pesticide toxicity based on electrochemical biosensor. Talanta 2011, 84, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Yong, D.; Liu, L.; Yu, D.; Dong, S. Development of a simple method for biotoxicity measurement using ultramicroelectrode array under non-deaerated condition. Anal. Chim. Acta 2011, 701, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Catterall, K.; Robertson, D.; Hudson, S.; Teasdale, P.R.; Welsh, D.T.; John, R. A sensitive, rapid ferricyanide-mediated toxicity bioassay developed using Escherichia coli. Talanta 2010, 82, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, G.; Turner, A.P.F. Development of an electrochemical method for the rapid determination of microbial concentration and evidence for the reaction mechanism. Anal. Chim. Acta 1988, 215, 61–69. [Google Scholar] [CrossRef]
- Yu, D.; Zhai, J.; Yong, D.; Dong, S. A rapid and sensitive p-benzoquinone-mediated bioassay for determination of heavy metal toxicity in water. Analyst 2013, 138, 3297. [Google Scholar] [CrossRef] [PubMed]
- Kuramitz, H.; Dziewatkoski, M.; Barnett, B.; Halsall, H.B.; Heineman, W.R. Application of an automated fluidic system using electrochemical bead-based immunoassay to detect the bacteriophage MS2 and ovalbumin. Anal. Chim. Acta 2006, 561, 69–77. [Google Scholar] [CrossRef]
- Kuramitz, H.; Halsall, H.B.; Heineman, W.R. Magnetic microbead-based enzyme immunoassay for ovalbumin using hydrodynamic voltammetry and fluorometric detection. Anal. Methods 2012, 4, 1783. [Google Scholar] [CrossRef]
- Yong, D.; Liu, C.; Zhu, C.; Yu, D.; Liu, L.; Zhai, J.; Dong, S. Detecting total toxicity in water using a mediated biosensor system with flow injection. Chemosphere 2015, 139, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Kuramitz, H.; Sazawa, K.; Nanayama, Y.; Hata, N.; Taguchi, S.; Sugawara, K.; Fukushima, M. Electrochemical genotoxicity assay based on a SOS/umu test using hydrodynamic voltammetry in a droplet. Sensors 2012, 12, 17414–17432. [Google Scholar] [CrossRef] [PubMed]
- Halling-Sørensen, B. Inhibition of aerobic growth and nitrification of bacteria in sewage sludge by antibacterial agents. Arch. Environ. Contam. Toxicol. 2001, 40, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Halling-Sørensen, B.; Sengeløv, G.; Ingerslev, F.; Jensen, L.B. Reduced antimicrobial potencies of oxytetracycline, tylosin, sulfadiazin, streptomycin, ciprofloxacin, and olaquindox due to environmental processes. Arch. Environ. Contam. Toxicol. 2003, 44, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Brooks, B.W.; Maul, J.D.; Belden, J.B. Antibiotics in aquatic and terrestrial ecosystems. In Encyclopedia of Ecology; Elsevier, 2008; pp. 210–217.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).