Submitted:
20 June 2024
Posted:
21 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Methods
3.2. Photophysical Methods
3.3. Cyclic Voltammetry
3.4. Synthetic Procedures and Product Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nicewicz, D.A.; MacMillan, D.W.C. Merging photoredox catalysis with organocatalysis. Science 2008, 322, 77–80. [Google Scholar] [CrossRef]
- Ischay, M.A.; Anzovino, M.E.; Du, J.; Yoon, T.P. Efficient Visible Light Photocatalysis of [2+2] Enone Cycloadditions. J. Am. Chem. Soc. 2008, 130, 12886–12887. [Google Scholar] [CrossRef]
- Narayanam, J.M.R.; Tucker, J.W.; Stephenson, C.R. Electron-Transfer Photoredox Catalysis: Development of a Tin-Free Reductive Dehalogenation Reaction. J. Am. Chem. Soc. 2009, 131, 8756–8757. [Google Scholar] [CrossRef]
- a) Narayanam, J.W.; Stephenson, C.R. Visible light photoredox catalysis: applications in organic synthesis. Chem. Soc. Rev. 2011, 40, 102–113. https://doi.org/10.1039/b913880n. b) Prier, C.K.; Rankic, D.A.; MacMillan, D.W.C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 2013, 113, 5322–5363. https://doi.org/10.1021/cr300503r. c) Tucker, J.W.; Stephenson, C.R.J. Shining Light on Photoredox Catalysis: Theory and Synthetic Applications. J. Org. Chem. 2012, 77, 1617–1622. https://doi.org/10.1021/jo202538x. d) Striet-Kalthoff, F.; James, M.J.; Teders, M.; Pitzer, L.; Glorius, F. Energy transfer catalysis mediated by visible-light: principles, applications, directions. Chem. Soc. Rev. 2018, 47, 7190–7202. https://doi.org/10.1039/c8cs00054a. e) Rigotti, Th.; Alemán, J. Visible light photocatalysis – from racemic to asymmetric activation strategies. Chem. Commun. 2020, 56, 11169–11190. https://doi.org/10.1039/d0cc03738a. f) Cauwenbergh, R.; Das, S. Photocatalysis. A Green Tool for Redox Reactions. Synlett 2022, 33, 129–149. https://doi.org/10.1055/s-0040-1706042.
- Wu, Y.; Kim, D.; Teets, T.S. Photophysical Properties and Redox Potentials of Photosensitizers for Organic Photoredox Transformations. Synlett 2022, 33, 1154–1179. [Google Scholar] [CrossRef]
- a) Campestrini, S.; Tonellato, U. Photoinitiated Olefin Epoxidation with Molecular Oxygen, Sensitized by Free Base Porphyrins and Promoted by Hexacarbonylmolybdenum in Homogeneous Solution. Eur. J. Org. Chem. 2002, 3827–3832. https://doi.org/10.1002/1099-0690. b) Clennan, E. L.; Pace, A. Advances in singlet oxygen chemistry. Tetrahedron 2005, 61, 6665–6691. https://doi.org/10.1016/j.tet.2005.04.017. c) Chen, J.-J.; Hong, G.; Gao, L.-J.; Liu, T.-J.; Cao, W.-J. In vitro and in vivo antitumor activity of a novel porphyrin-based photosensitizer for photodynamic therapy, J. Cancer Res. Clin. Oncol. 2015, 141, 1553–1561. https://doi.org/10.1007/s00432-015-1918-1. d) Rostami, M.; Rafiee, L.; Hassanzadeh, F.; Dadrass, A.R.; Khodarahmi, G.A. Synthesis of some new porphyrins and their metalloderivatives as potential sensitizers in photo-dynamic therapy. Res. Pharm. Sci. 2015, 10, 504–513. PMID: 26779270; PMCID: PMC4698861.
- Rybicka-Jasinska, K.; Shan, W.; Zawada, K.; Kadish, K.M.; Gryko, D. Porphyrins as Photoredox Catalysts: Experimental and Theoretical Studies. J. Am. Chem. Soc. 2016, 138, 15451−15458. [Google Scholar] [CrossRef]
- Costa e Silva, R.; da Silva, L.O.; Bartolomeu, A. A.; Brocksom, T. J.; de Oliveira, K. T. Recent applications of porphyrins as photocatalysts in organic synthesis: batch and continuous flow approaches. Beilstein J. Org. Chem. 2020, 16, 917–955. [Google Scholar] [CrossRef]
- a) Arlegui, A.; El-Hachemi, Z.; Crusats, J.; Moyano, A. 5-Phenyl-10,15,20-Tris(4-sulfonatophenyl)porphyrin: Synthesis, Catalysis, and Structural Studies. Molecules 2018, 23, 3363. https://doi.org/10.3390/molecules23123363. b) Arlegui, A.; Soler, B.; Galindo, A.; Orteaga, O.; Canillas, A.; Ribó, J.M.; El-Hachemi, Z.; Crusats, J.; Moyano, A. Spontaneous mirror-symmetry breaking coupled to top-bottom chirality transfer: From porphyrin self-assembly to scalemic Diels–Alder adducts. Chem. Commun. 2019, 55, 12219–12222. https://doi.org/10.1039/c9cc05946f.
- a) Arlegui, A.; Torres, P.; Cuesta, V.; Crusats, J.; Moyano, A. A pH-Switchable Aqueous Organocatalysis with Amphiphilic Secondary Amine–Porphyrin Hybrids. Eur. J. Org. Chem. 2020, 4399-4407. https://doi.org/10.1002/ejoc.202000648. b) Arlegui, A.; Torres, P.; Cuesta, V.; Crusats, J.; Moyano, A. Chiral Amphiphilic Secondary Amine-Porphyrin Hybrids for Aqueous Organocatalysis. Molecules 2020, 25, 3420. https://doi.org/10.3390/molecules25153420.
- Torres, P.; Guillén, M.; Escribà, M.; Crusats, J.; Moyano, A. Synthesis of New Amino-Functionalized Porphyrins: Preliminary Study of Their Organophotocatalytic Activity. Molecules 2023, 28, 1997. [Google Scholar] [CrossRef]
- Kadish, K.M.; Cornillon, J.-L.; Yao, C.-L.; Malinski, T.; Gritzner, G. Solvent effects on electrode potentials of metalloporphyrins. Reduction of 5,10,15,20-tetraphenylporphinato complexes in non-aqueous media. J. Electroanal. Chem. 1987, 235, 189–207. [Google Scholar] [CrossRef]
- Mandal, T.; Das, S.; De Sarkar, S. Nickel(II) Tetraphenylporphyrin as an Efficient Photocatalyst Featuring Visible Light Promoted Dual Redox Reactions. Adv. Synth. Catal. 2019, 361, 3200–3209. [Google Scholar] [CrossRef]
- Bhyrappa, P.; Sankar, M.; Varghese, B. Mixed Substituted Porphyrins: Structural and Electrochemical Redox Properties. Inorg. Chem. 2006, 45, 4136–4149. [Google Scholar] [CrossRef]
- Fang, Y.; Jiang, X.; Ou, Zh.; Michelin, C.; Desbois, N.; Gros, C.P.; Kadish, K.M. Redox properties of nitrophenylporphyrins and electrosynthesis of nitrophenyl-linked Zn porphyrin dimers or arrays. J. Porphyr. Phthalocyanines 2014, 18, 832–841. [Google Scholar] [CrossRef]
- a) Adler, A.D.; Longo, F.R.; Finarelli, J.D.; Goldmacher, J.; Assour, J.; Korsakoff, L. A simplified synthesis for meso-tetraphenylporphine. J. Org. Chem. 1967, 32, 476–476. https://doi.org/10.1021/jo01288a053. b) Wagner, R.W.; Lawrence, D.S.; Lindsey, J.S. An improved synthesis of tetramesitylporphyrin. Tetrahedron Lett. 1987, 28, 3069–3070. https://doi.org/10.1016/S0040-4039(00)96287-7. c) Lindsey, J.S.; Prathapan, S.; Johnson, T.E.; Wagner, R. W. Porphyrin Building Blocks for Modular Construction of Bioorganic Model Systems. Tetrahedron 1994, 50, 8941–8968. doi: 10.106/S0040-40200185364-3.
- Gadde, K.; De Vos, D.; Maes, B.U.W. Basic Concepts and Activation Modes in Visible-Light Photocatalyzed Organic Synthesis. Synthesis 2023, 55, 164–192. [Google Scholar] [CrossRef]
- a) Seely, G.R. The energetics of electron-transfer reactions of chlorophyll and other compounds. Photochem. Photobiol. 1978, 27, 639–654. https://doi.org/10.1111/j.1751-1097.1978.tb07658.x. b) Rillema, D.P.; Nagle, J.K.; Barringer, L.F., Jr.; Meyer, T.J. Redox Properties of Metalloporphyrin Excited States, Lifetimes, and Related Properties of a Series of Para-Substituted Tetraphenylporphine Carbonyl Complexes of Rutenium(II). J. Am. Chem. Soc. 1981, 103, 56–62. https://doi.org/10.1021/ja00391a013.
- Capaldo, C.; Ravelli, D.; Fagnoni, M. Direct Photocatalyzed Hydrogen Atom Transfer (HAT) for Aliphatic C–H Bonds Elaboration. Chem. Rev. 2022, 122, 1875–1924. [Google Scholar] [CrossRef]
- Bonfantini, E.E.; Burrell, A.K.; Campbell, W.M.; Crossley, M.J.; Gosper, J.J.; Harding, M.M.; Officer, D.L.; Reid, D.C.W. Efficient synthesis of free-base 2-formyl-5,10,15,20-tetraarylporphyrins, their reduction and conversion to [(porphyrin-2-yl)methyl]phosphonium salts. J. Porphyr. Phthalocyanines 2002, 6, 708–719. [Google Scholar] [CrossRef]
- Richeter, S.; Jeandon, C.; Gisselbrecht, J.-P.; Ruppert, R.; Callot, H. Synthesis and Optical and Electrochemical Properties of Porphyrin Dimers Linked by Metal Ions. J. Am. Chem. Soc. 2002, 124, 6168–6179. [Google Scholar] [CrossRef]
- Lower, S.K.; El-Sayed, M.A. The triplet state and molecular electronic processes in organic molecules, Chem. Rev. 1966, 66, 199–241. [Google Scholar] [CrossRef]
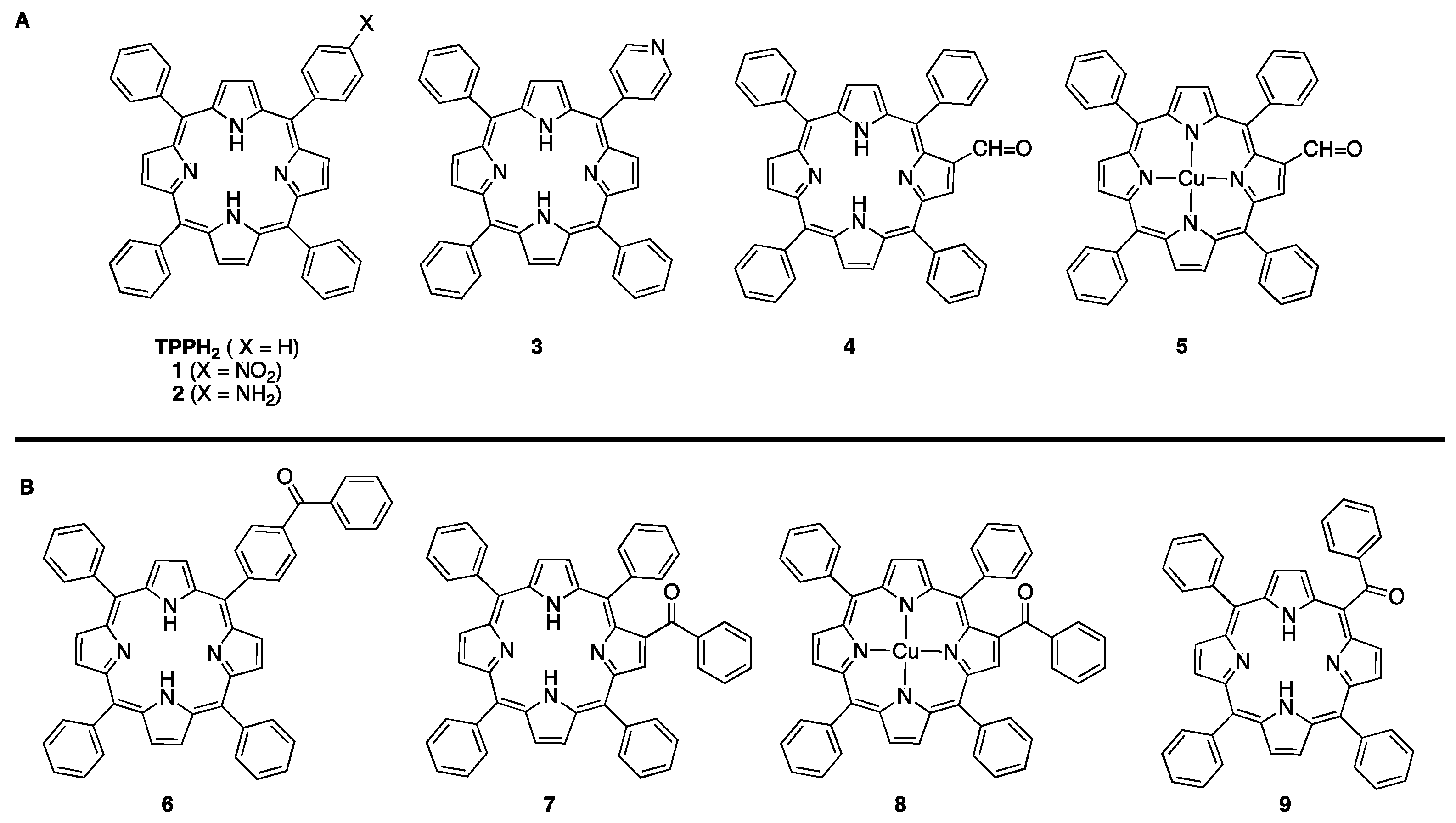
| Porphyrin | Experimental ground-state redox half-wave potentials (V vs. SCE) | |||
| [PC.–/PC2–] | [PC/PC.–] | [PC.+/PC] | [PC2+/ PC.+] | |
| TPPH2a | -1.63 | -1.21 | +1.09 | +1.28 |
| 1b | -1.66c | -1.21d/-1.06e | +1.10f | +1.20f |
| 2 | -1.56 | -1.22 | +1.00f,g/+1.10f | +1.70f |
| 3 | -1.49 | -1.14 | +1.05f,h/+1.15 | +1.60f |
| 4 | -1.23 | -0.95 | +1.11 | +1.45 |
| 5 | -1.43 | -1.07 | +1.11 | +1.48 |
| Porphyrin | Ground-state redox potentials (V vs. SCE) | (Singlet) excited-state redox potentials (V vs. SCE) | ||
| [PC/PC.–] | [PC.+/PC] | [*PC/PC.–] | [PC.+/*PC] | |
| TPPH2a | -1.21 | +1.09 | +0.70 | -0.82 |
| 1 | -1.06 | +1.10 | +0.85 | -0.81 |
| 2 | -1.22 | +1.00 | +0.69 | -0.91 |
| 3 | -1.14 | +1.05 | +0.77 | -0.86 |
| 4 | -0.95 | +1.11 | +0.96 | -0.80 |
| 5 | -1.07 | +1.11 | +0.84 | -0.80 |
| Porphyrin | Experimental ground-state redox half-wave potentials (V vs. SCE) | |||
| [PC.–/PC2–] | [PC/PC.–] | [PC.+/PC] | [PC2+/ PC.+] | |
| TPPH2 | -1.63 | -1.21 | +1.09 | +1.28 |
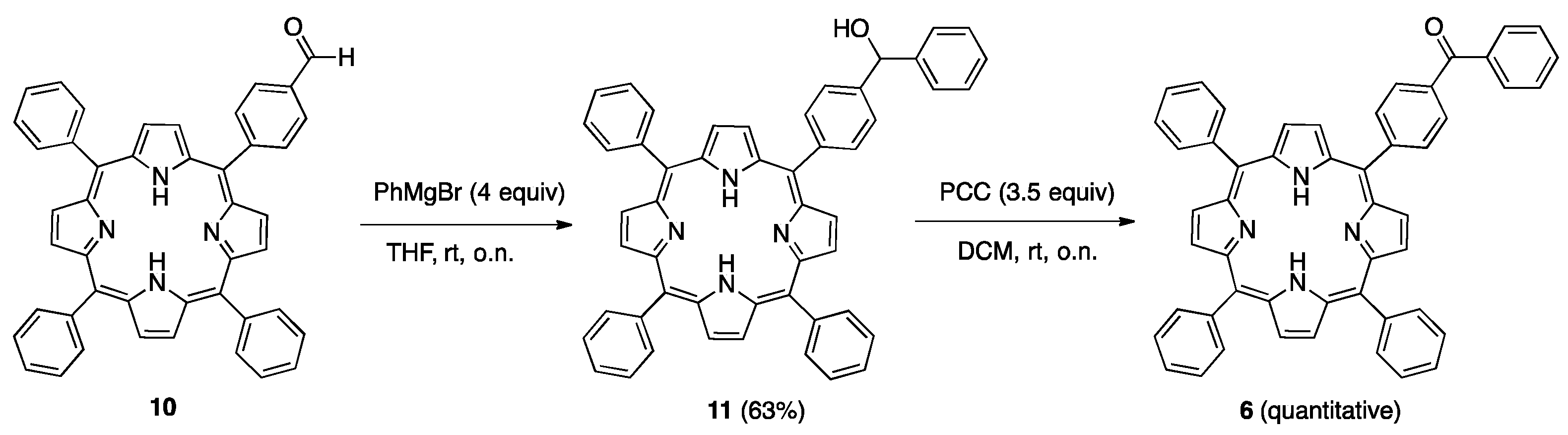
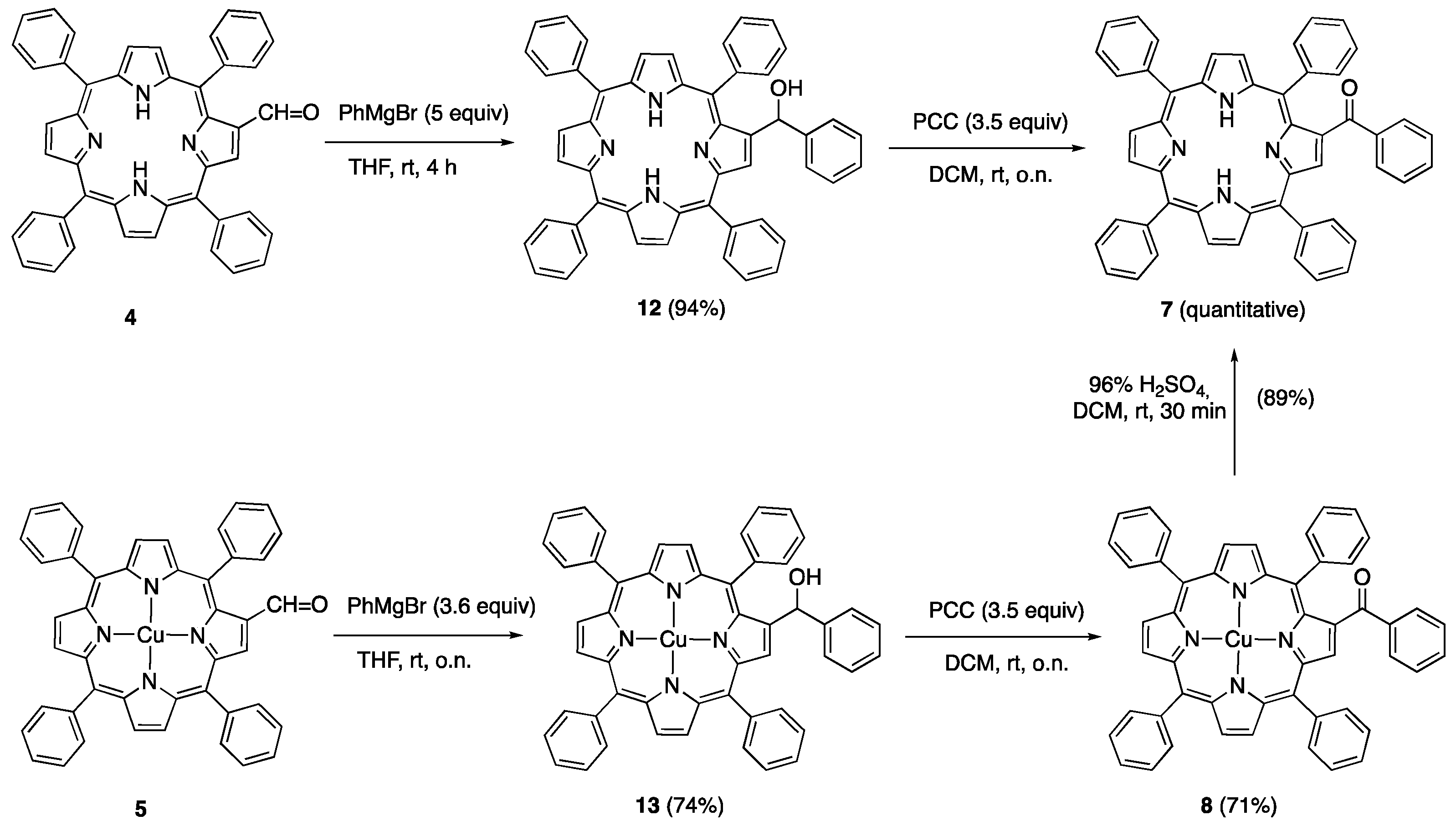
| 6 | -1.37 | -1.10 | +1.24 | +1.56 |
| 7 | -1.28 | -1.00 | +1.22 | +1.43 |
| 8 | -1.31 | -1.09 | +1.23 | +1.60 |
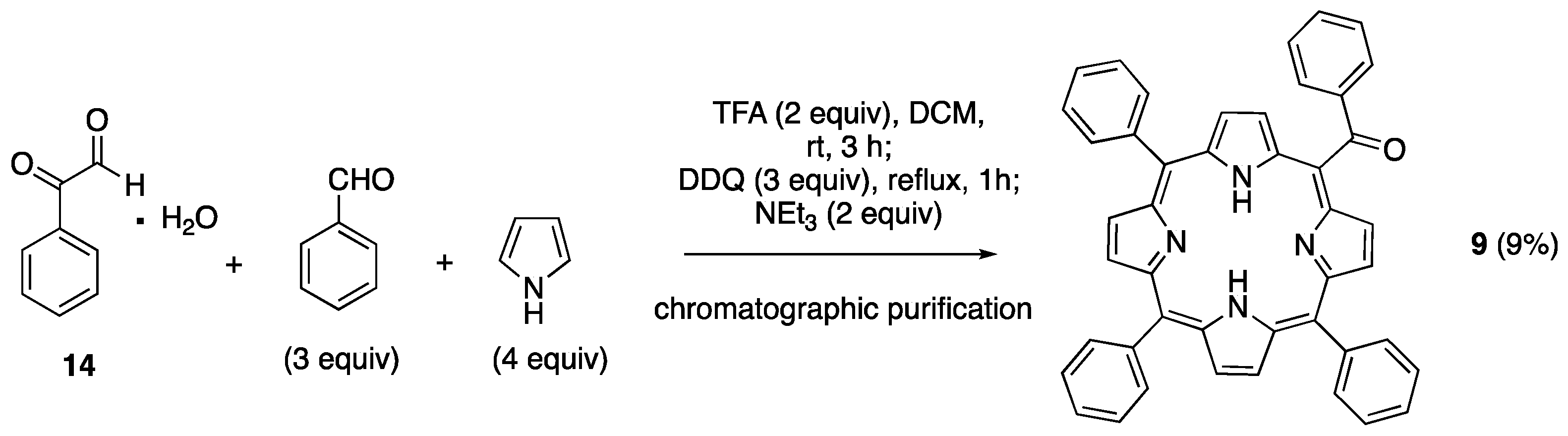
| 9 | -1.17 | -0.97 | +1.32 | +1.62 |
| Porphyrin | Less energetic absorption Q band (nm) | More energetic emission Q(0,0) band (nm) | Relative fluorescence quantum yield (φf)a | Singlet excited state energies (ES10,0), V |
| 6 | 645 | 651 | 0.09 | 1.91 |
| 7 | 651 | 662 | 0.09 | 1.89 |
| 8 | 587 | 650 | 0.002 | 2.00 |
| 9 | 647 | 650 | 0.08 | 1.91 |
| Porphyrin | Ground-state redox potentials (V vs. SCE) | (Singlet) excited-state redox potentials (V vs. SCE) | ||
| [PC/PC.–] | [PC.+/PC] | [*PC/PC.–] | [PC.+/*PC] | |
| TPPH2a | -1.21 | +1.09 | +0.70 | -0.82 |
| 6 | -1.10 | +1.24 | +0.81 | -0.71 |
| 7 | -1.00 | +1.22 | +0.89 | -0.67 |
| 8 | -1.09 | +1.23 | +0.91 | -0.77 |
| 9 | -0.97 | +1.32 | +0.94 | -0.59 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





