Submitted:
27 June 2024
Posted:
28 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Presentation
2.1. Case 1
2.2. Case 2
3. Discussion
3.1. Ultrasound-Guided Greater Occipital Nerve (GON) Hydrodissection with 5% Dextrose in Water in the Lateral Decubitus Position
3.1.1. Technical Notes for the Hydrodissection of the GON with 5% Dextrose in Water in a Lateral Decubitus Position
- Patient selection: Patients fulfill the diagnostic criteria as described in the introduction for occipital neuralgia, with other differential diagnoses excluded. They have failed other conservative treatments. The local tenderness is felt mainly over the OCI (usually slightly lateral than the GON entrapped within the SCC and UT groups), and compression may reproduce their occipital neuralgia-type pain between the SCC and OCI but not within the SCC and UT. There is no swelling or tenderness in the SCC or UT. Ultrasound shows swelling of the GON compared to the normal, non-painful side between the SCC and OCI, or a cross-sectional area exceeding the upper limit of 3 mm2 [28]. If they have swelling and tenderness of the SCC and UT (more medial than the entrapment between the SCC and OCI group), it means the GON may also be entrapped within the SCC and UT, indicating that the GON may also need to be hydrodissected using the cranial-caudal technique described below.
- Patient’s Position: The patient is typically positioned in a lateral decubitus position with the GON under treatment facing upward. This positioning allows for an alternative treatment option if the patient is unable to lie prone. From the patient’s perspective, it is generally more comfortable if the needle comes from behind, especially when they are fully awake, as it prevents them from seeing the needle.
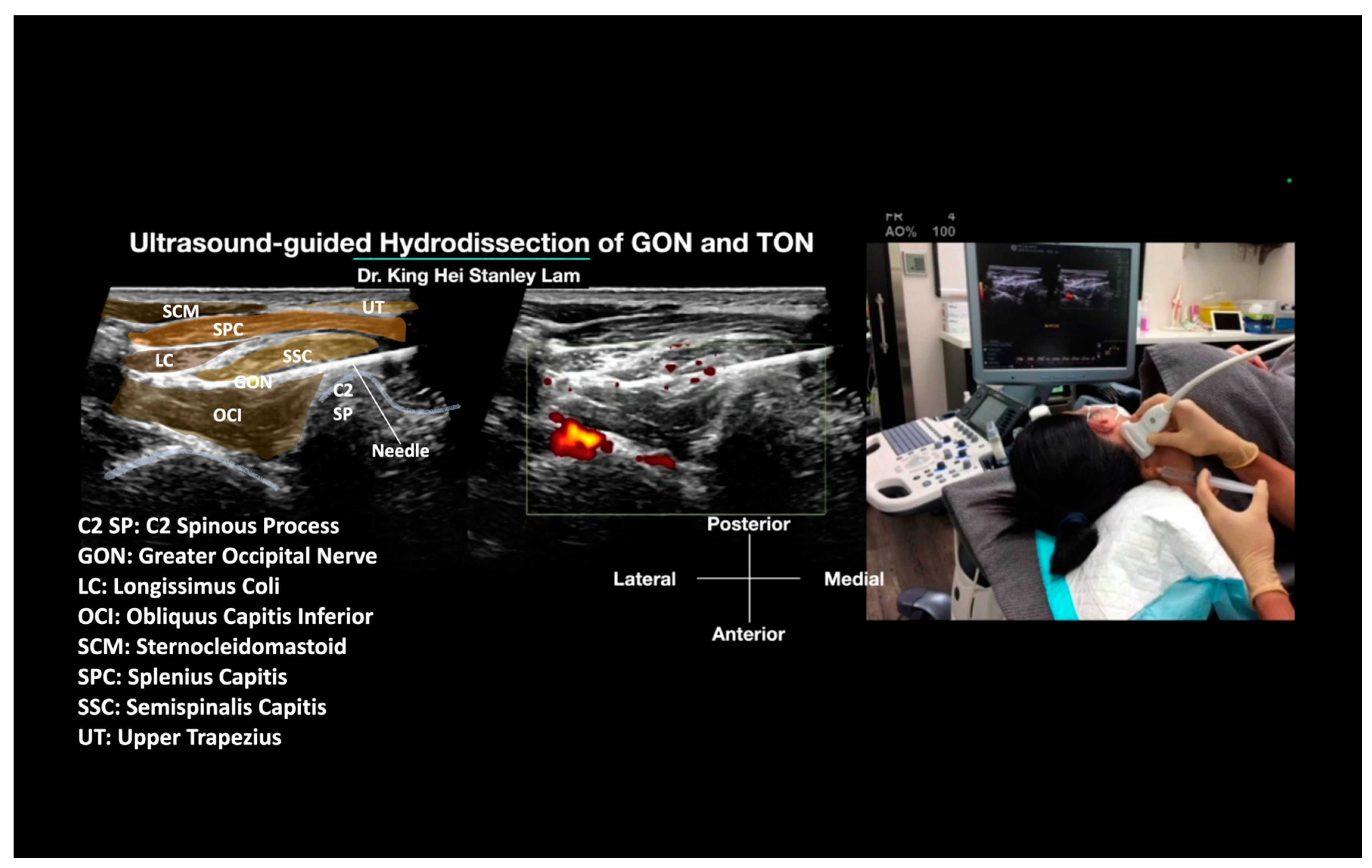
- The Ultrasound Machine and Probe: The ultrasound machine is placed in front of the patient, facing the treating physician, to optimize ergonomics. The ultrasound used in this manuscript was the Logiq S7 (General Electric, Boston, MA, USA). A linear transducer with a broad frequency range (GE L3-12 D, General Electric, Boston, MA, USA) is used, which provides sufficient penetration and resolution. It is positioned on the transverse plane, aligned with the long axis of the OCI, and emits ultrasound waves to visualize the underlying anatomy.
- Identifications of Important Sonoanatomy: Obliquus Capitis Inferior Muscle: This is a striated muscle that originates from the spinous process of the axis (C2) and inserts into the posterior aspect of the transverse process of the atlas (C1) (Figure 1). Semispinalis Capitis Muscle: The SSC muscle is a deep neck muscle visualized in the short-axis view. It is located superficial to the OCI and underneath the splenius capitis and UT. The UT and sternocleidomastoid muscles are the most superficial muscles in that region (Figure 1). Greater Occipital Nerve: The GON is identified as a hypoechoic oval structure situated between the SSC and OCI (Figure 1). In cases where there is swelling or inflammation, the GON may appear enlarged compared to the normal side.
- Inject: D5W without local anesthetic.
- Method: Appropriate disinfection preparation uses 2% chlorhexidine in 75% isopropyl alcohol. It is also advisable to use the same disinfectant solution as the contact media for the ultrasound transducer [29]. The needle entry point is anesthetized with 0.5% lidocaine to create a skin wheel. With ultrasound guidance, a 25 G or 27 G 2-inch hypodermic needle is inserted using an in-plane technique. The needle tip is advanced to the site of the GON using a hydrodissection technique. Injection of D5W during needle advancement pushes away any soft tissue in front of the needle tip and creates a halo, allowing the needle tip to follow from the subcutaneous fat to the site and the whole tract of the GON to be hydrodissected. The needle is guided to hydrodissect the GON between the SSC and OCI, and the soft tissue is completely hydrodissected above and below the GON, with the GON completely encircled with anechoic fluid. Typically, 20 to 30 mL of D5W without local anesthetic will be used for one side of GON hydrodissection, from skin entry to complete hydrodissection of the GON, resulting in the patient’s pain being relieved.
3.1.2. Advantages of the Ultrasound-Guided Greater Occipital Nerve (GON) Hydrodissection Procedure in the Lateral Decubitus Position
- Patient Comfort:
- -
- The lateral decubitus position is generally more comfortable for the patient compared to lying prone, especially if they have difficulty lying prone.
- -
- Patients may feel more at ease when the needle comes from behind, as they cannot directly see the needle approach.
- -
- Some patients reported feeling very nervous when lying prone and not knowing what to expect during the procedure.
- Improved Visualization:
- -
- With the patient in the lateral decubitus position and the ultrasound probe placed in the long axis of the OCI, the transducer can be optimally positioned parallel to the OCI muscle fibers to provide clear visualization of the target anatomy, including the OCI, GON, the relevant arteries, and the C2 nerve root.
- -
- The utilization of the in-plane technique, with the needle entering from a posterior to anterior direction, offers a distinct advantage in terms of visualization. This technique allows for a highly clear and detailed visualization of the needle when hydrodissecting above and below the GON, between the OCI and SSC. This is because the transducer aligns more parallelly with the needle, enhancing the precision and accuracy of the procedure.
- -
- This approach can provide a clearer needle trajectory compared to the traditional prone position, even when injecting the C2 nerve root over the pedicle.
- -
- Even with the traditional prone position, the needle entry point utilized in the lateral decubitus approach, at the C2 spinous process, may offer better needle visualization and trajectory. This is because the needle would be more parallel to the transducer, enhancing the precision of the procedure.
- Learning Curve:
- -
- The improved visualization of the anatomical structures and needle trajectory may provide clinicians with a shorter learning curve to properly visualize the target anatomy and guide the needle accurately.
- Procedural Safety:
- -
- The lateral decubitus position can provide better visualization of the suboccipital structures while also relaxing the patient, which may significantly reduce the risk of inadvertent vascular puncture or injection compared to the prone position.
- -
- The improved visualization of the target anatomy and needle trajectory can help the clinician accurately guide the needle to the desired location, minimizing the risk of nerve injury.
3.1.3. Disadvantages of the Ultrasound-Guided GON Hydrodissection Procedure in the Lateral Decubitus Position
- Patient Positioning:
- -
- While the lateral decubitus position may be more comfortable for some patients, it may still be difficult for patients with limited neck mobility or other physical limitations. Patients with pain over the contralateral shoulder and hip may not be able to tolerate the lateral decubitus position.
3.2. Cranial to Caudal Approach of Ultrasound-Guided Hydrodissection with 5% Dextrose of the Greater Occipital Nerve inside the Semispinalis Capitis and Upper Trapezius Muscle
3.2.1. Technical Notes of the Cranial Approach of Ultrasound-Guided Hydrodissection with 5% Dextrose in Water of the Greater Occipital Nerve inside the Semispinalis Capitis and Upper Trapezius Muscle
- Patient selection: Patients fulfill the diagnostic criteria as described in the introduction for occipital neuralgia, with other differential diagnoses excluded. They have failed other conservative treatments. Local tenderness and swelling are felt mainly over the UT and SCC (usually more medial than the GON entrapped between the SCC and OCI groups). Compression of the SCC and UT may reproduce their occipital neuralgia-type pain, but not compression between the SCC and OCI, and there is no swelling and tenderness between the SCC and OCI. Ultrasound may show swelling of the GON as a hypoechoic structure passing through the SCC and UT, compared to the normal, non-painful side, or a cross-sectional area exceeding the upper limit of 3 mm2. However, the cross-sectional area of the GON within the SCC and UT is very difficult to measure accurately due to the similarity of echogenicity with the surrounding muscles. If they have swelling and tenderness of the SCC and OCI (more lateral than the entrapment within the SCC and UT group), it suggests an additional entrapment site of the GON between the SCC and OCI, indicating a need to add hydrodissection using the posteromedial to anterolateral approach technique described above (in Section 3.1).
- Patient’s Position: The patient is typically positioned in a prone position with the head and neck fully flexed to allow for optimal ultrasound scanning and guided injection. This position also helps to minimize the curvature in the suboccipital region, which can otherwise make the procedure challenging to perform.
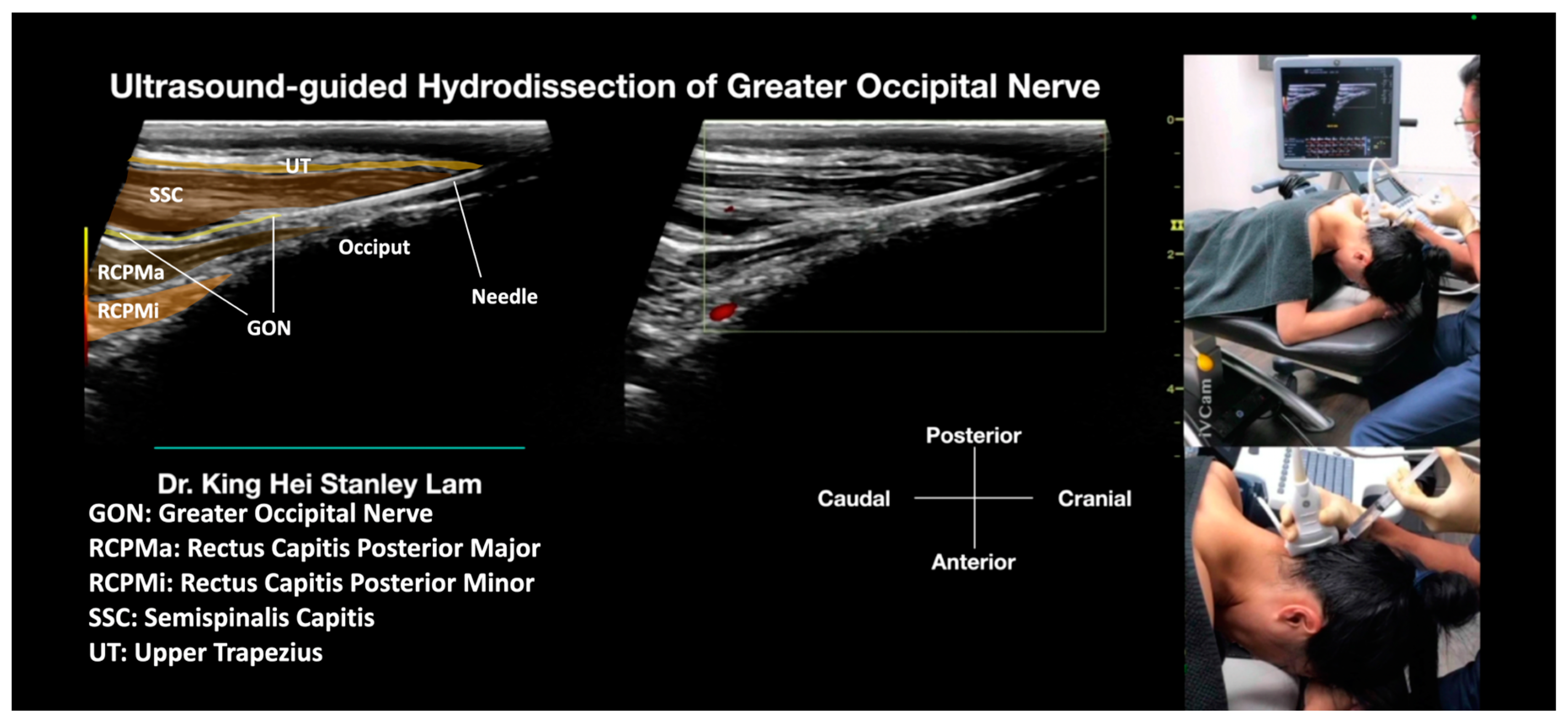
- The Ultrasound Machine and Probe: The ultrasound machine is placed by the side of the treatment table with the monitor in front of the treating physician for optimal ergonomics. The ultrasound used in this manuscript was the Logiq S7 (General Electric, Boston, MA, USA). A linear transducer with a broad frequency range (GE L3–12 D, General Electric, Boston, MA, USA) was used, which provided sufficient penetration and resolution. The transducer was positioned in the sagittal plane, aligned with the long axis of the SSC and UT muscles.
- Identifications of Important Sonoanatomy: Upper Trapezius Muscle: The UT muscle is located superficially in this region, beneath the adipose tissue and fascia. It is a thin layer of striated muscle. Semispinalis Capitis Muscle: The SSC muscle is a deep neck muscle that is visualized in a long-axis view. It is located superficially to the rectus capitis superior muscle and underneath the UT and splenius capitis muscles. Greater Occipital Nerve: The GON is identified as a hypoechoic fascicular structure that passes through the SSC and UT muscles. In cases where there is swelling or inflammation, the GON may appear enlarged compared to the normal side.
- Needle: A 25 G or 27 G 2-inch hypodermic needle is inserted under ultrasound guidance to access the GON for hydrodissection above and below the nerve.
- Inject: D5W without local anesthetic.
- Method: Due to the presence of hair in this region, a larger amount of 2% chlorhexidine in isopropyl alcohol is advised to be used as the disinfection agent. The disinfectant solution may need to be applied several times to ensure proper disinfection. It is also advised to use the same disinfectant solution as the contact media for the ultrasound transducer. The needle entry point is anesthetized with 0.5% lidocaine to create a skin wheel. With ultrasound guidance, a 25 G or 27 G 2-inch hypodermic needle is inserted. The physician keeps injecting with D5W with needle advancement to push away any soft tissue in front of the needle tip and create a halo, allowing the needle tip to follow from the subcutaneous fat to the site. This allows for the whole tract of the GON to be hydrodissected, within the UT and SCC, and between the SCC and RCPMa, as the GON passes through these anatomical structures. The soft tissue is completely hydrodissected above and below, lateral, and medial to the GON, with the GON completely encircled with anechoic fluid. During the procedure, in-plane and out-of-plane techniques will be used interchangeably to validate the needle position. Typically, 20 to 30 mL of D5W without local anesthetic will be used for one side of GON hydrodissection, from skin entry to complete hydrodissection of the GON, resulting in the patient’s pain being relieved.
3.2.2. Advantages of the Cranial to Caudal Approach for Ultrasound-Guided Hydrodissection of the Greater Occipital Nerve (GON)
- True Access to Nerve Entrapment Sites: This approach is the first ultrasound-guided hydrodissection approach to truly target the GON as it passes through the SSC and UT muscles, which are other common sites of nerve entrapment [25]. Currently, no other proposed non-surgical treatments are effective [30].
- Potential for More Comprehensive Treatment: By addressing the GON within the SSC and UT muscles and between the SCC and RCPMa, this technique can complement the previously described lateral decubitus approach to hydrodissect the GON between the SSC and OCI muscles, as described in Section 1.1. This combination of techniques may offer a more comprehensive management strategy and treat a longer length of the affected portion of the GON in patients with occipital neuralgia.
- Improved Visualization: The prone position with neck flexion helps minimize the curvature of the suboccipital region, allowing for better visualization of the underlying anatomy using the high-frequency linear ultrasound transducer.
3.2.3. Disadvantages of the Cranial Approach for Ultrasound-Guided Hydrodissection of the Greater Occipital Nerve (GON)
- Patient Positioning Challenges: The prone position with neck flexion may be less comfortable for some patients, particularly those with pre-existing neck or back problems. Maintaining this position throughout the procedure can be challenging.
- Increased Technical Difficulty: The curved anatomy of the suboccipital region and the depth of the GON within the SSC and UT muscles may make the ultrasound-guided needle placement more technically demanding and increase the potential risks, e.g., nerve injury, compared to the posteromedial-to-anterolateral approach described in Section 1.1.
3.3. Selection of Injectate for Ultrasound-Guided Hydrodissection of the Greater Occipital Nerve (GON)
3.4. Mechanism of Action of Glucose
3.5. Potential Limitations of Ultrasound-Guided Greater Occipital Nerve (GON) Hydrodissection
- Technical Challenges:
- -
- Anatomical variation in the course and branching pattern of the GON can make it difficult to precisely locate and target the nerve during the hydrodissection procedure.
- -
- Identification of the exact fascial planes and muscle layers surrounding the GON may be challenging, especially in patients with previous injuries and surgery involving the musculature or adipose tissue in the suboccipital region.
- -
- While hydrodissection of the GON shows promising results, long-term studies with larger sample sizes are needed to confirm its efficacy and safety.
- -
- The success of the procedure relies heavily on accurate needle placement, which requires expertise in ultrasound-guided injections.
- -
- The presence of excessive hair in the suboccipital region and the curvature of the suboccipital region may make the ultrasound-guided procedure in this area challenging, especially with the use of prone positioning.
- Patient Factors:
- -
- Patients with limited neck mobility or difficulty lying in the required position (e.g., intolerance to the prone position) may have difficulty tolerating the procedure.
- -
- Patients with significant cervical spine pathology, such as severe degenerative changes or instability, may not be suitable candidates for the hydrodissection procedure.
- -
- It is important to consider the potential underlying causes of occipital neuralgia when assessing the effectiveness of hydrodissection for long-term pain relief. In cases where the neuralgia is secondary to conditions such as arthritis or spinal stenosis, hydrodissection may not provide sustained relief. Additionally, if the impingement of other occipital nerves, such as the lesser and/or third occipital nerves, is contributing to the neuralgia, targeting only the GON through hydrodissection may not be sufficient for effective pain management.
- -
- Empirical observations suggest that the response to hydrodissection can vary significantly between individuals, and large-scale studies are needed to standardize this technique, its protocol, and indications for this procedure.
- Incomplete or Transient Pain Relief:
- -
- While the procedure has shown promising results, in some cases, the pain relief may be incomplete or temporary, and require repeat hydrodissection or other interventions.
- -
- The long-term durability of pain relief with GON hydrodissection is still being evaluated, and the optimal frequency of repeat procedures is not yet well-established.
- Possible Side Effects of Ultrasound-Guided GON Hydrodissection Using D5W Without Local Anesthetic:
- -
- The potential side effects of the ultrasound-guided GON hydrodissection described in this manuscript, according to the authors’ experiences, are primarily related to the needling procedure itself. Systemic effects or side effects from the unintentional flow of fluid to surrounding structures are not expected using D5W as the injectate rather than a local anesthetic.
- -
- Its side effects profile is similar to, but considerably less severe than, the ultrasound-guided GON block. This is because a hydrodissecting technique is used, which theoretically pushes the nerves and vascular structures away from the needle tip, further reducing the chance of inadvertent vascular puncture and nerve irritation.
- -
- Temporary discomfort may be possible due to the pressure effect of the solution.
4. Conclusions
Institutional Review Board Statement
Conflicts of Interest
References
- Urits, I.; Schwartz, R.H.; Patel, P.; Zeien, J.; Connor, D.; Hasoon, J.; Berger, A.A.; Kassem, H.; Manchikanti, L.; Kaye, A.D.; et al. A Review of the Recent Findings in Minimally Invasive Treatment Options for the Management of Occipital Neuralgia. Neurol. Ther. 2020, 9, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Gobel, H. 13.4 Occipital Neuralgia [Internet] ICHD-3. Available online: https://ichd-3.org/13-painful-cranial-neuropathies-and-other-facial-pains/13-4-occipital-neuralgia/ (accessed on 14 June 2024).
- Dougherty, C. Occipital neuralgia. Curr. Pain Headache Rep. 2014, 18, 411. [Google Scholar] [CrossRef] [PubMed]
- Finiels, P.J.; Batifol, D. The treatment of occipital neuralgia: Review of 111 cases. Neurochirurgie 2016, 62, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Mathew, P.G.; Najib, U.; Khaled, S.; Krel, R. Prevalence of Occipital Neuralgia at a Community Hospital-based Headache Clinic. Neurol. Clin. Pract. 2021, 11, 6–12. [Google Scholar] [CrossRef]
- Djavaherian, D.M.; Guthmiller, K.B. Occipital Neuralgia. [Updated 2023 Mar 6]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024; Available online: https://www.ncbi.nlm.nih.gov/books/NBK538281/ (accessed on 14 June 2024).
- Swanson, D.; Guedry, R.; Boudreaux, M.; Muhlenhaupt, E.; Kaye, A.D.; Viswanath, O.; Urits, I. An Update on the Diagnosis, Treatment, and Management of Occipital Neuralgia. J. Craniofac. Surg. 2022, 33, 779–783. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.G.; Joseph, K.S.; Samouil, M.M.; Hill, D.S.; Ibrahim, M.M.; Assi, P.E.; Joseph, J.T.; Kassis, S.A. Nerve blocks for occipital headaches: A systematic review and meta-analysis. J. Anaesthesiol. Clin. Pharmacol. 2023, 39, 170–180. [Google Scholar] [CrossRef]
- Kastler, A.; Attye, A.; Maindet, C.; Nicot, B.; Gay, E.; Kastler, B.; Krainik, A. Greater occipital nerve cryoneurolysis in the management of intractable occipital neuralgia. J. Neuroradiol. 2018, 45, 386–390. [Google Scholar] [CrossRef]
- Eskilsson, A.; Ageberg, E.; Ericson, H.; Marklund, N.; Anderberg, L. Decompression of the greater occipital nerve improves outcome in patients with chronic headache and neck pain—A retrospective cohort study. Acta Neurochir. 2021, 163, 2425–2433. [Google Scholar] [CrossRef] [PubMed]
- Pietramaggiori, G.; Scherer, S. Minimally Invasive Nerve- and Muscle-Sparing Surgical Decompression for Occipital Neuralgia. Plast. Reconstr. Surg. 2023, 151, 169–177. [Google Scholar] [CrossRef]
- Choi, I.; Jeon, S.R. Neuralgias of the Head: Occipital Neuralgia. J. Korean Med. Sci. 2016, 31, 479–488. [Google Scholar] [CrossRef]
- Hammond, S.R.; Danta, G. Occipital neuralgia. Clin. Exp. Neurol. 1978, 15, 258–270. [Google Scholar]
- Guvencer, M.; Akyer, P.; Sayhan, S.; Tetik, S. The importance of the greater occipital nerve in the occipital and the suboccipital region for nerve blockade and surgical approaches--an anatomic study on cadavers. Clin. Neurol. Neurosurg. 2011, 113, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Greher, M.; Moriggl, B.; Curatolo, M.; Kirchmair, L.; Eichenberger, U. Sonographic visualization and ultrasound-guided blockade of the greater occipital nerve: A comparison of two selective techniques confirmed by anatomical dissection. Br. J. Anaesth. 2010, 104, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Lauretti, G.R.; Correa, S.W.; Mattos, A.L. Efficacy of the Greater Occipital Nerve Block for Cervicogenic Headache: Comparing Classical and Subcompartmental Techniques. Pain Pract. 2015, 15, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Yang, H.; Li, X.; Lin, C.; Ke, S.; Wu, S.; Ma, C. Ultrasound-Guided versus Fluoroscopy-Guided Deep Cervical Plexus Block for the Treatment of Cervicogenic Headache. BioMed Res. Int. 2017, 2017, 4654803. [Google Scholar] [CrossRef] [PubMed]
- Rose, G. Treatment of post-traumatic occipital neuralgia with ultrasound-guided greater occipital nerve hydrodissection in the emergency department. JEM Rep. 2024, 3, 100078. [Google Scholar] [CrossRef]
- Ryan, P.J.; Harmon, D.C. Ultrasound-Guided Greater Occipital Nerve Hydrodissection for Treatment of Cervicogenic Headache: A Case Report. Pain Stud. Treat. 2023, 11, 1–8. [Google Scholar] [CrossRef]
- Kaga, M. First Case of Occipital Neuralgia Treated by Fascial Hydrodissection. Am. J. Case Rep. 2022, 23, e936475. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.T.; Ho, T.Y.; Chou, Y.C.; Ke, M.J.; Li, T.Y.; Tsai, C.K.; Chen, L.C. Six-month efficacy of perineural dextrose for carpal tunnel syndrome: A Prospective, randomized, double-blind, controlled trial. Mayo Clin. Proc. 2017, 92, 1179–1189. [Google Scholar] [CrossRef]
- Huang, C.Y.; Lai, C.Y.; Reeves, K.D.; Lam, K.H.S.; Li, T.Y.; Cheng, C.I.; Wu, Y.T. Volume Effect of Nerve Hydrodissection for Carpal Tunnel Syndrome: A Prospective, Randomized, and Single-Blind Study. J. Ultrasound Med. 2024, 43, 161–169. [Google Scholar] [CrossRef]
- Vernon, H. The Neck Disability Index: State-of-the-art, 1991–2008. J. Manip. Physiol. Ther. 2008, 31, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Macken, B. Questioning short-term memory and its measurement: Why digit span measures long-term associative learning. Cognition 2015, 144, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.H.S. Subject: Letter to the Editor—Suboccipital Sonoanatomy—Sonoanatomy Labelling Errors and Effective Ultrasound-Guided Hydrodissection of the Greater Occipital Nerve. JEM Rep. 2024, 100095. [Google Scholar] [CrossRef]
- Wright, A.; Hannon, J.; Hegedus, E.J.; Kavchak, A.E. Clinimetrics corner: A closer look at the minimal clinically important difference (MCID). J. Man. Manip. Ther. 2012, 20, 160–166. [Google Scholar] [CrossRef]
- Young, I.A.; Dunning, J.; Butts, R.; Mourad, F.; Cleland, J.A. Reliability, construct validity, and responsiveness of the neck disability index and numeric pain rating scale in patients with mechanical neck pain without upper extremity symptoms. Physiother. Theory Pract. 2019, 35, 1328–1335. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.C.; Haun, D.W.; Kettner, N.W.; Scali, F.; Clark, T.B. Sonography of the normal greater occipital nerve and obliquus capitis inferior muscle. J. Clin. Ultrasound 2010, 38, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.H.S.; Lai, W.W.; Ngai, H.Y.; Wu, W.K.R.; Wu, Y.-T. Comment on the safety of the ultrasound-guided hydrodissection technique for carpal tunnel syndrome. J. Ultrasound 2022, 26, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Son, B.C.; Kim, D.R.; Lee, S.W. Intractable occipital neuralgia caused by an entrapment in the semispinalis capitis. J. Korean Neurosurg. Soc. 2013, 54, 268–271. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.K.H.; Reeves, K.D.; Cheng, A.L. Transition from deep regional blocks toward deep nerve hydrodissection in the upper body and torso: Method description and results from a retrospective chart review of the analgesic effect of 5% dextrose water as the primary hydrodissection injectate to enhance safety. BioMed Res. Int. 2017, 2017, 7920438. [Google Scholar] [CrossRef]
- Cherng, J.H.; Chang, S.J.; Tsai, H.D.; Chun, C.F.; Fan, G.Y.; Reeves, K.D.; Lam, K.H.S.; Wu, Y.T. The Potential of Glucose Treatment to Reduce Reactive Oxygen Species Production and Apoptosis of Inflamed Neural Cells In Vitro. Biomedicines 2023, 11, 1837. [Google Scholar] [CrossRef]
- Wu, Y.T.; Wu, C.H.; Lin, J.A.; Su, D.C.; Hung, C.Y.; Lam, S.K.H. Efficacy of 5% Dextrose Water Injection for Peripheral Entrapment Neuropathy: A Narrative Review. Int. J. Mol. Sci. 2021, 22, 12358. [Google Scholar] [CrossRef]
- Wu, Y.T.; Chen, Y.P.; Lam, K.H.S.; Reeves, K.D.; Lin, J.A.; Kuo, C.Y. Mechanism of Glucose Water as a Neural Injection: A Perspective on Neuroinflammation. Life 2022, 12, 832. [Google Scholar] [CrossRef]
- Lam, K.H.S.; Wu, Y.T.; Reeves, K.D.; Galluccio, F.; Allam, A.E.; Peng, P.W.H. Ultrasound-Guided Interventions for Carpal Tunnel Syndrome: A Systematic Review and Meta-Analyses. Diagnostics 2023, 13, 1138. [Google Scholar] [CrossRef] [PubMed]
- Buntragulpoontawee, M.; Chang, K.V.; Vitoonpong, T.; Pornjaksawan, S.; Kitisak, K.; Saokaew, S.; Kanchanasurakit, S. The Effectiveness and Safety of Commonly Used Injectates for Ultrasound-Guided Hydrodissection Treatment of Peripheral Nerve Entrapment Syndromes: A Systematic Review. Front. Pharmacol. 2020, 11, 621150. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.T.; Ke, M.J.; Ho, T.Y.; Li, T.Y.; Shen, Y.P.; Chen, L.C. Randomized double-blinded clinical trial of 5% dextrose versus triamcinolone injection for carpal tunnel syndrome patients. Ann. Neurol. 2018, 84, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Young, K.M.; Moo, N.Y.; Ho, M.J. Comparison on Treatment Effects of Dextrose Water, Saline, and Lidocaine for Trigger Point Injection. Ann. Rehabil. Med. 1997, 21, 967–973. [Google Scholar]
- MacIver, M.B.; Tanelian, D.L. Activation of C fibers by metabolic perturbations associated with tourniquet ischemia. Anesthesiology 1992, 76, 617–623. [Google Scholar] [CrossRef]
- Maniquis-Smigel, L.; Reeves, K.D.; Rosen, H.J.; Lyftogt, J.; Graham-Coleman, C.; Cheng, A.L.; Rabago, D. Analgesic Effect and Potential Cumulative Benefit from Caudal Epidural D5W in Consecutive Participants with Chronic Low-Back and Buttock/Leg Pain. J. Altern. Complement. Med. 2018, 24, 1189–1196. [Google Scholar] [CrossRef]
- Bonora, M.; Patergnani, S.; Rimessi, A.; De Marchi, E.; Suski, J.M.; Bononi, A.; Giorgi, C.; Marchi, S.; Missiroli, S.; Poletti, F.; et al. ATP synthesis and storage. Purinergic Signal. 2012, 8, 343–357. [Google Scholar] [CrossRef]
- Rangaraju, V.; Calloway, N.; Ryan, T.A. Activity-driven local ATP synthesis is required for synaptic function. Cell 2014, 156, 825–835. [Google Scholar] [CrossRef]
- Nakayama, T.; Naono, R.; Ikeda, T.; Nishimori, T. NMDA and AMPA receptors contribute to the maintenance of substance P-induced thermal hyperalgesia. Neurosci. Res. 2010, 67, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Holden, J.E.; Pizzi, J.A.; Jeong, Y. An NK1 receptor antagonist microinjected into the periaqueductal gray blocks lateral hypothalamic-induced antinociception in rats. Neurosci. Lett. 2009, 453, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Parenti, C.; Arico, G.; Ronsisvalle, G.; Scoto, G.M. Supraspinal injection of Substance P attenuates allodynia and hyperalgesia in a rat model of inflammatory pain. Peptides 2012, 34, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Chen, W.N.; Chen, C.J.; Lin, Y.W.; Zimmer, A.; Chen, C.C. An antinociceptive role for substance P in acid-induced chronic muscle pain. Proc. Natl. Acad. Sci. USA 2012, 109, E76–E83. [Google Scholar] [CrossRef]
- Chung, E.; Yoon, T.G.; Kim, S.; Kang, M.; Kim, H.J.; Son, Y. Intravenous Administration of Substance P Attenuates Mechanical Allodynia Following Nerve Injury by Regulating Neuropathic Pain-Related Factors. Biomol. Ther. 2017, 25, 259–265. [Google Scholar] [CrossRef]
- Komatsu, T.; Sasaki, M.; Sanai, K.; Kuwahata, H.; Sakurada, C.; Tsuzuki, M.; Iwata, Y.; Sakurada, S.; Sakurada, T. Intrathecal substance P augments morphine-induced antinociception: Possible relevance in the production of substance P N-terminal fragments. Peptides 2009, 30, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Han, D.S.; Lee, C.H.; Shieh, Y.D.; Chang, C.T.; Li, M.H.; Chu, Y.C.; Wang, J.L.; Chang, K.V.; Lin, S.H.; Chen, C.C. A role for substance P and acid-sensing ion channel 1a in prolotherapy with dextrose-mediated analgesia in a mouse model of chronic muscle pain. Pain 2022, 163, e622–e633. [Google Scholar] [CrossRef]
- Topol, G.A.; Pestalardo, I.G.; Reeves, K.D.; Elias, F.; Steinmetz, N.J.; Cheng, A.L.; Rabago, D. Dextrose Prolotherapy for Symptomatic Grade IV Knee Osteoarthritis: A Pilot Study of Early and Longer-Term Analgesia and Pain-Specific Cytokine Concentrations. Clin. Pract. 2022, 12, 926–938. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




