Submitted:
01 July 2024
Posted:
02 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
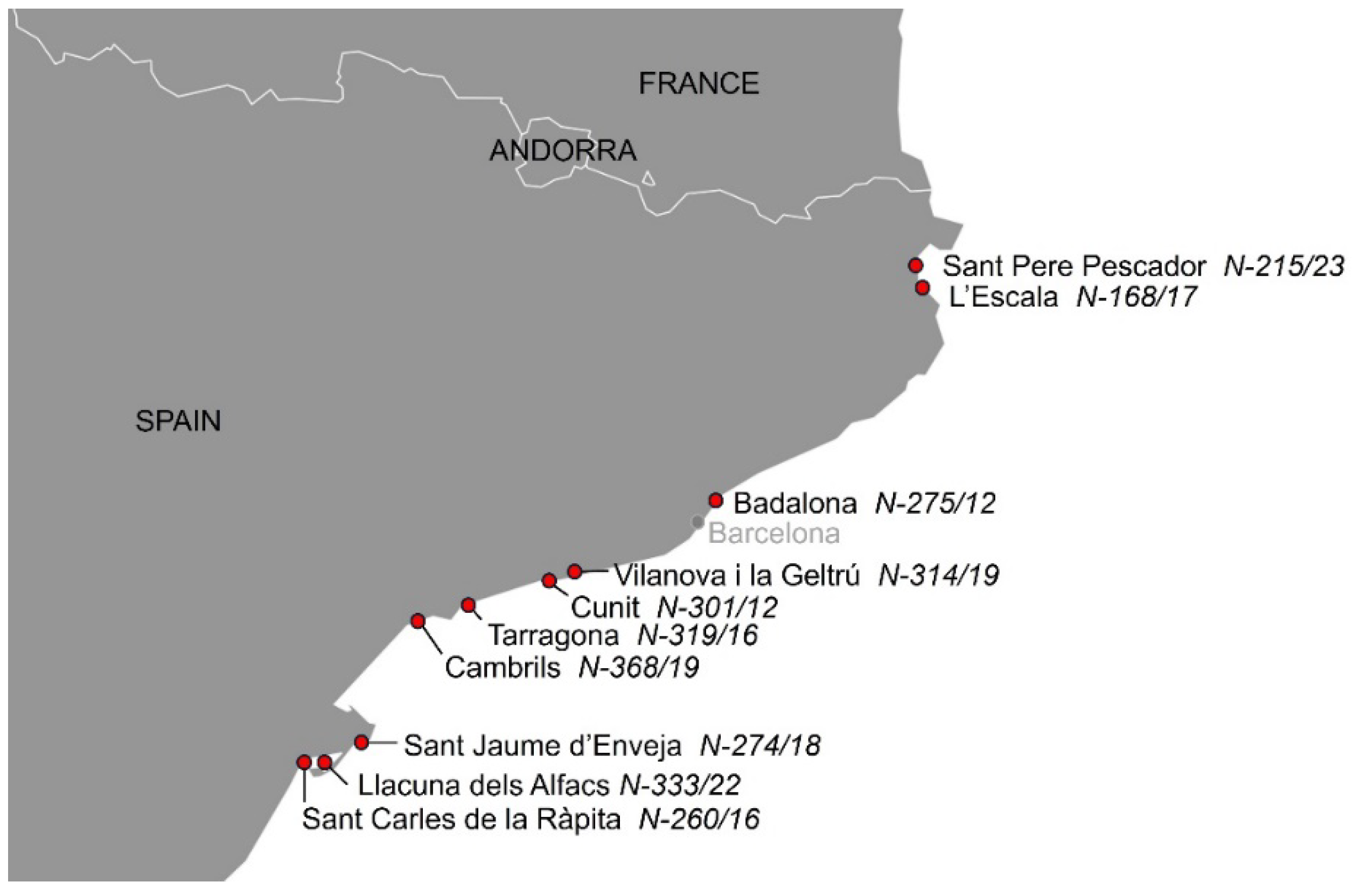
2.1. Cases Investigated and Necropsy Procedure
2.2. Serology for Brucella
2.3. Brucella Isolation
2.4. Age Determination
2.5. Data Analysis
3. Results
3.1. Pathological Investigation
3.2. Culture of Brucella
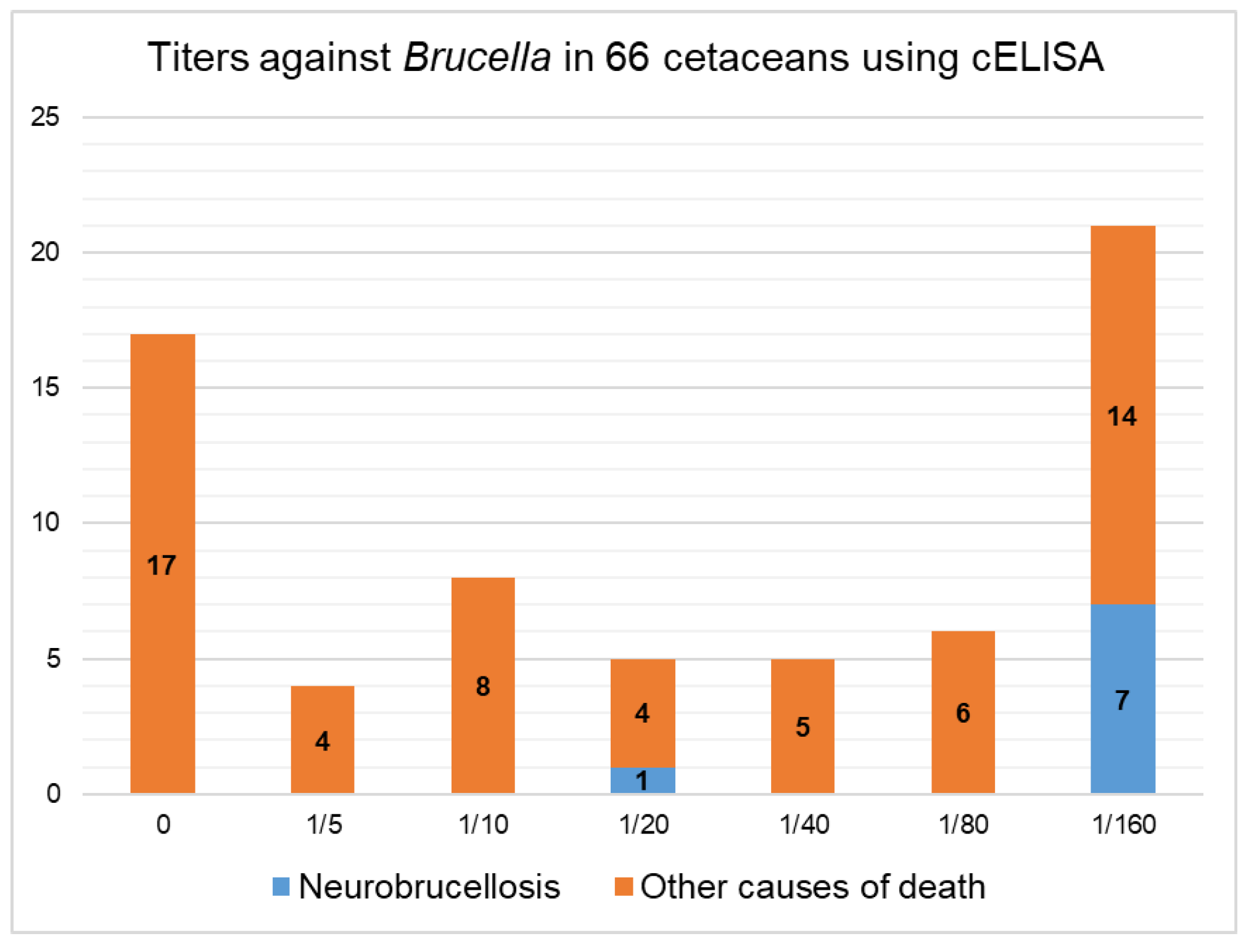
3.3. Serological Investigation
3.1.1. Serological Results Comparison by Cause of Death
3.1.1. Serology by Age Class
3.1.1. Brucella Serology in CeMV-Infected Cetaceans
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ewalt, D.R.; Payeur, J.B.; Martin, B.M.; Cummins, D.R.; Miller, W.G. Characteristics of a Brucella Species from a Bottlenose Dolphin (Tursiops truncatus). J. Vet. Diagn. Invest. 1994, 6, 448–452. [Google Scholar] [CrossRef]
- Foster, G.; Osterman, B.S.; Godfroid, J.; Jacques, I.; Cloeckaert, A. Brucella ceti Sp. Nov. and Brucella pinnipedialis Sp. Nov. for Brucella Strains with Cetaceans and Seals as Their Preferred Hosts. Int. J. Syst. Evol. Microbiol. 2007, 57, 2688–2693. [Google Scholar] [CrossRef]
- Nymo, I.H.; Tryland, M.; Godfroid, J. A Review of Brucella Infection in Marine Mammals, with Special Emphasis on Brucella pinnipedialis in the Hooded Seal (Cystophora cristata). Vet. Res. 2011, 42, 93. [Google Scholar] [CrossRef]
- Guzmán-Verri, C.; González-Barrientos, R.; Hernández-Mora, G.; Morales, J.-A.; Baquero-Calvo, E.; Chaves-Olarte, E.; Moreno, E. Brucella ceti and Brucellosis in Cetaceans. Front. Cell. Infect. Microbiol. 2012, 2. [Google Scholar] [CrossRef]
- Van Bressem, M.; Raga, A.; Di Guardo, G.; Jepson, P.; Duignan, P.; Siebert, U.; Barrett, T.; Santos, Mc.; Moreno, I.; Siciliano, S.; et al. Emerging Infectious Diseases in Cetaceans Worldwide and the Possible Role of Environmental Stressors. Dis. Aquat. Organ. 2009, 86, 143–157. [Google Scholar] [CrossRef]
- Alba, P.; Terracciano, G.; Franco, A.; Lorenzetti, S.; Cocumelli, C.; Fichi, G.; Eleni, C.; Zygmunt, M.S.; Cloeckaert, A.; Battisti, A. The Presence of Brucella ceti ST26 in a Striped Dolphin (Stenella coeruleoalba) with Meningoencephalitis from the Mediterranean Sea. Vet. Microbiol. 2013, 164, 158–163. [Google Scholar] [CrossRef]
- Garofolo, G.; Zilli, K.; Troiano, P.; Petrella, A.; Marotta, F.; Di Serafino, G.; Ancora, M.; Di Giannatale, E. Brucella ceti from Two Striped Dolphins Stranded on the Apulia Coastline, Italy. J. Med. Microbiol. 2014, 63, 325–329. [Google Scholar] [CrossRef]
- Isidoro-Ayza, M.; Ruiz-Villalobos, N.; Pérez, L.; Guzmán-Verri, C.; Muñoz, P.M.; Alegre, F.; Barberán, M.; Chacón-Díaz, C.; Chaves-Olarte, E.; González-Barrientos, R.; et al. Brucella ceti Infection in Dolphins from the Western Mediterranean Sea. BMC Vet. Res. 2014, 10, 206. [Google Scholar] [CrossRef]
- Cvetnić, Ž.; Duvnjak, S.; Đuras, M.; Gomerčić, T.; Reil, I.; Zdelar-Tuk, M.; Špičić, S. Evidence of Brucella Strain ST27 in Bottlenose Dolphin (Tursiops truncatus) in Europe. Vet. Microbiol. 2016, 196, 93–97. [Google Scholar] [CrossRef]
- Grattarola, C.; Petrella, A.; Lucifora, G.; Di Francesco, G.; Di Nocera, F.; Pintore, A.; Cocumelli, C.; Terracciano, G.; Battisti, A.; Di Renzo, L.; et al. Brucella ceti Infection in Striped Dolphins from Italian Seas: Associated Lesions and Epidemiological Data. Pathogens 2023, 12, 1034. [Google Scholar] [CrossRef] [PubMed]
- Van Bressem, M.; Van Waerebeek, K.; Raga, J.A.; Godfroid, J.; Brew, S.D.; MacMillan, A.P. Serological Evidence of Brucella Species Infection in Odontocetes from the South Pacific and the Mediterranean. Vet. Rec. 2001, 148, 657–661. [Google Scholar] [CrossRef]
- Pugliares, K.R.; Bogomolni, A.; Touhey, K.M.; Herzig, S.M.; Harry, C.T.; Moore, M.J. Marine Mammal Necropsy : An Introductory Guide for Stranding Responders and Field Biologists; Woods Hole Oceanographic Institution: Woods Hole, MA, 2007. [Google Scholar]
- Kuiken, Thijs; García-Hartmann, Manuel Proceedings of the First European Cetacean Society Workshop on Cetacean Pathology: Dissection Techniques and Tissue Sampling.; Leiden, The Netherlands, 1991; p. 39.
- Cuvertoret-Sanz, M.; López-Figueroa, C.; O’Byrne, A.; Canturri, A.; Martí-Garcia, B.; Pintado, E.; Pérez, L.; Ganges, L.; Cobos, A.; Abarca, M.; et al. Causes of Cetacean Stranding and Death on the Catalonian Coast (Western Mediterranean Sea), 2012-2019. Dis. Aquat. Organ. 2020, 142, 239–253. [Google Scholar] [CrossRef]
- López-Goñi, I.; García-Yoldi, D.; Marín, C.M.; De Miguel, M.J.; Barquero-Calvo, E.; Guzmán-Verri, C.; Albert, D.; Garin-Bastuji, B. New Bruce-Ladder Multiplex PCR Assay for the Biovar Typing of Brucella suis and the Discrimination of Brucella suis and Brucella canis. Vet. Microbiol. 2011, 154, 152–155. [Google Scholar] [CrossRef]
- Calzada, Nuria; Aguilar, Alex; Lockyer, Christina; Grau, Esteve Patterns of Growth and Physical Maturity in the Western Mediterranean Striped Dolphin, Stenella coeruleoalba (Cetacea: Odontoceti). Can. J. Zool. 1997, 75, 632–637. [CrossRef]
- Calzada, N.; Aguilar, A. Flipper Development in the Mediterranean Striped Dolphin (Stenella coeruleoalba). Anat. Rec. 1996, 245, 708–714. [Google Scholar] [CrossRef]
- Garofolo, G.; Petrella, A.; Lucifora, G.; Di Francesco, G.; Di Guardo, G.; Pautasso, A.; Iulini, B.; Varello, K.; Giorda, F.; Goria, M.; et al. Occurrence of Brucella ceti in Striped Dolphins from Italian Seas. PLOS ONE 2020, 15, e0240178. [Google Scholar] [CrossRef]
- Dorneles, E.M.S.; Teixeira-Carvalho, A.; Araújo, M.S.S.; Sriranganathan, N.; Lage, A.P. Immune Response Triggered by Brucella abortus Following Infection or Vaccination. Vaccine 2015, 33, 3659–3666. [Google Scholar] [CrossRef]
- De Figueiredo, P.; Ficht, T.A.; Rice-Ficht, A.; Rossetti, C.A.; Adams, L.G. Pathogenesis and Immunobiology of Brucellosis. Am. J. Pathol. 2015, 185, 1505–1517. [Google Scholar] [CrossRef]
- Dadelahi, A.S.; Abushahba, M.F.N.; Ponzilacqua-Silva, B.; Chambers, C.A.; Moley, C.R.; Lacey, C.A.; Dent, A.L.; Skyberg, J.A. Interactions between B Cells and T Follicular Regulatory Cells Enhance Susceptibility to Brucella Infection Independent of the Anti-Brucella Humoral Response. PLOS Pathog. 2023, 19, e1011672. [Google Scholar] [CrossRef]
- Cloeckaert, A.; Jacques, I.; Bosseray, N.; Limet, J.N.; Bowden, R.; Dubray, G.; Plommet, M. Protection Conferred on Mice by Monoclonal Antibodies Directed against Outer-Membrane-Protein Antigens of Brucella. J. Med. Microbiol. 1991, 34, 175–180. [Google Scholar] [CrossRef]
- Hernández-Mora, G.; Palacios-Alfaro, J.D.; González-Barrientos, R. Wildlife Reservoirs of Brucellosis: Brucella in Aquatic Environments.
- Meegan, J.; Field, C.; Sidor, I.; Romano, T.; Casinghino, S.; Smith, C.R.; Kashinsky, L.; Fair, P.A.; Bossart, G.; Wells, R.; et al. Development, Validation, and Utilization of a Competitive Enzyme-Linked Immunosorbent Assay for the Detection of Antibodies against Brucella Species in Marine Mammals. J. Vet. Diagn. Invest. 2010, 22, 856–862. [Google Scholar] [CrossRef]
- Prenger-Berninghoff, E.; Siebert, U.; Stede, M.; König, A.; Weiß, R.; Baljer, G. Incidence of Brucella Species in Marine Mammals of the German North Sea. Dis. Aquat. Organ. 2008, 81, 65–71. [Google Scholar] [CrossRef]
- Lynch, M.; Duignan, P.J.; Taylor, T.; Nielsen, O.; Kirkwood, R.; Gibbens, J.; Arnould, J.P.Y. Epizootiology of Brucella Infection in Australian Fur Seals. J. Wildl. Dis. 2011, 47, 352–363. [Google Scholar] [CrossRef]
- Norman, S.A.; Delaney, M.A.; Haman, K.H.; Thomas, A.C.; Godfroid, J.; Larsen, A.K.; Nymo, I.H.; Robbe-Austerman, S.; Quance, C.; Rhyan, J.C.; et al. Application of Real-Time Quantitative PCR Assays for Detecting Marine Brucella spp. in Fish. J. Vet. Diagn. Invest. 2018, 30, 150–154. [Google Scholar] [CrossRef]
- Hoover-Miller, A.; Dunn, J.; Field, C.; Blundell, G.; Atkinson, S. Seroprevalence of Brucella Antibodies in Harbor Seals in Alaska, USA, with Age, Regional, and Reproductive Comparisons. Dis. Aquat. Organ. 2017, 126, 1–12. [Google Scholar] [CrossRef]
- Kershaw, J.; Stubberfield, E.; Foster, G.; Brownlow, A.; Hall, A.; Perrett, L. Exposure of Harbour Seals Phoca vitulina to Brucella in Declining Populations across Scotland. Dis. Aquat. Organ. 2017, 126, 13–23. [Google Scholar] [CrossRef]
- Kroese, M.V.; Beckers, L.; Bisselink, Y.J.W.M.; Brasseur, S.; Van Tulden, P.W.; Koene, M.G.J.; Roest, H.I.J.; Ruuls, R.C.; Backer, J.A.; IJzer, J.; et al. Brucella pinnipedialis in Grey Seals (Halichoerus gryphus) and Harbor Seals (Phoca vitulina) in the Netherlands. J. Wildl. Dis. 2018, 54, 439. [Google Scholar] [CrossRef]
- Ruiz-Mesa, J.D.; Sánchez-Gonzalez, J.; Reguera, J.M.; Martín, L.; Lopez-Palmero, S.; Colmenero, J.D. Rose Bengal Test: Diagnostic Yield and Use for the Rapid Diagnosis of Human Brucellosis in Emergency Departments in Endemic Areas. Clin. Microbiol. Infect. 2005, 11, 221–225. [Google Scholar] [CrossRef]
- Casanova, A.; Ariza, J.; Rubio, M.; Masuet, C.; Díaz, R. BrucellaCapt versus Classical Tests in the Serological Diagnosis and Management of Human Brucellosis. Clin. Vaccine Immunol. 2009, 16, 844–851. [Google Scholar] [CrossRef]
- Zhao, S.; Cheng, Y.; Liao, Y.; Zhang, Z.; Yin, X.; Shi, S. Treatment Efficacy and Risk Factors of Neurobrucellosis. Med. Sci. Monit. 2016, 22, 1005–1012. [Google Scholar] [CrossRef]
- Soares, C.N.; Angelim, A.I.M.; Brandão, C.O.; Santos, R.Q.; Mehta, R.; Silva, M.T.T.D. Neurobrucellosis: The Great Mimicker. Rev. Soc. Bras. Med. Trop. 2022, 55, e0567–2021. [Google Scholar] [CrossRef]
- Solís García Del Pozo, J.; Lorente Ortuño, S.; Navarro, E.; Solera, J. Detection of IgM Antibrucella Antibody in the Absence of IgGs: A Challenge for the Clinical Interpretation of Brucella Serology. PLoS Negl. Trop. Dis. 2014, 8, e3390. [Google Scholar] [CrossRef]
- Grattarola, C.; Gallina, S.; Giorda, F.; Pautasso, A.; Ballardini, M.; Iulini, B.; Varello, K.; Goria, M.; Peletto, S.; Masoero, L.; et al. First Report of Salmonella 1,4,[5],12:I:- In Free-Ranging Striped Dolphins (Stenella coeruleoalba), Italy. Sci. Rep. 2019, 9, 6061. [Google Scholar] [CrossRef] [PubMed]
- Rebollada-Merino, A.; Giorda, F.; Pumarola, M.; Martino, L.; Gomez-Buendia, A.; Romani-Cremaschi, U.; Casalone, C.; Mattioda, V.; Di Nocera, F.; Lucifora, G.; et al. Neurobrucellosis (Brucella ceti) in Striped Dolphins (Stenella coeruleoalba): Immunohistochemical Studies on Immune Response and Neuroinflammation. Vet. Pathol. 2024, 03009858241250336. [Google Scholar] [CrossRef]
- Bravo, M.J.; Colmenero, J.D.; Martín, J.; Alonso, A.; Caballero, A. Polymorphism of the Transmembrane Region of the MICA Gene and Human Brucellosis. Tissue Antigens 2007, 69, 358–360. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; He, H.-Y.; Ojha, S.C.; Shi, H.; Sun, C.-F.; Deng, C.-L.; Sheng, Y.-J. Association of IL-6, IL-10 and TGF-Β1 Gene Polymorphisms with Brucellosis: A Systematic Review with Meta-Analysis. Microb. Pathog. 2019, 135, 103640. [Google Scholar] [CrossRef]
- Khatun, M.; Islam, A.; Baek, B.K. Comparative Analysis of Humoral Immune Response and Cognate Antigen Detection in Experimentally Infected Sprague Dawley Rats with Brucella abortus Biotype 1. Vector-Borne Zoonotic Dis. 2024, 24, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Goertz, C.E.C.; Frasca, S.; Bohach, G.A.; Cowan, D.F.; Buck, J.D.; French, R.A.; De Guise, S.; Maratea, J.; Hinckley, L.; Ewalt, D.; et al. Brucella Sp. Vertebral Osteomyelitis with Intercurrent Fatal Staphylococcus aureus Toxigenic Enteritis in a Bottlenose Dolphin (Tursiops truncatus). J. Vet. Diagn. Invest. 2011, 23, 845–851. [Google Scholar] [CrossRef]
- Islam, Md.A.; Khatun, Mst. M.; Werre, S.R.; Sriranganathan, N.; Boyle, S.M. A Review of Brucella Seroprevalence among Humans and Animals in Bangladesh with Special Emphasis on Epidemiology, Risk Factors and Control Opportunities. Vet. Microbiol. 2013, 166, 317–326. [Google Scholar] [CrossRef]
- Abo-Shehada, M.N.; Odeh, J.S.; Abu-Essud, M.; Abuharfeil, N. Seroprevalence of Brucellosis among High Risk People in Northern Jordan. Int. J. Epidemiol. 1996, 25, 450–454. [Google Scholar] [CrossRef]
| ID | Date | Place of stranding | Age class | Age estimation | Length (cm) | Weight (Kg) | Sex | A/D |
|---|---|---|---|---|---|---|---|---|
|
N-275/12 |
2012/05/23 |
Badalona | Ad | - | 300 | Unknown | M | D |
|
N-301/12 |
2012/06/03 |
Cunit | Ad | 6.25 | 184 | 54.5 | F | A |
| N-260/16 | 2016/06/19 |
Sant Carles de la Ràpita |
J | 3.88 | 170 | 39.5 | F | D |
|
N-319/16 |
2016/09/08 |
Tarragona | J | 5.35 | 180 | 47 | F | A |
| N-168/17 | 2017/04/18 |
L’Escala |
Ad | 10.39 | 193 | 79 | M | A |
|
N-274/18 |
2018/06/24 |
Sant Jaume d’Enveja | J | 2.43 | 152 | 38 | M |
A |
|
N-314/19 |
2019/09/01 |
Vilanova i la Geltrú |
J | 2.8 | 159 | 41 | F | A |
|
N-368/19 |
2019/10/12 |
Cambrils | J | 5.23 | 175 | 47 | M | A |
| N-333/22 | 2022/09/22 |
Llacuna dels Alfacs | Ad | 6.15 | 180 | 47.5 | M | A |
| N-215/23 | 2023/06/22 | Sant Pere Pescador | Ad | 13.79 | 197 | 59 | M | A |
| ID | Macroscopic lesions | Histopathology | Brucella culture | Others | Cause of death | Available serum |
|---|---|---|---|---|---|---|
| N-275/12* | Chronic suppurative diskospondylitis Multifocal brain malacia |
Mycotic pyogranulomatous-necrotizing meningoencephalomyelitis |
Positive (vertebral abscess) | Mycotic encephalitis | Mycotic encephalitis | No |
| N-301/12* | Not relevant | Non-suppurative meningoencephalitis, diffuse, more intense in cerebellum, brainstem and spinal cord; choroiditis; radiculoneuritis | Positive (brain) | - | Neurobrucellosis | Yes |
| N-260/16 | Emaciation; cerebral edema; absence of recent ingesta | Non-suppurative meningoencephalitis, diffuse, more intense in brainstem; choroiditis; radiculoneuritis, multifocal and milder | Positive (brain) | - | Neurobrucellosis | Yes |
| N-319/16 | Low body condition; absence of recent ingesta; meningeal congestion | Non-suppurative meningoencephalitis | Positive (brain, ventricle swab) | CeMV intensely positive (RT-PCR and IHC) | CeMV encephalitis | Yes |
| N-168/17 | Absence of recent ingesta; meningeal and CSF turbidity |
Non-suppurative meningitis, more intense in brainstem and cerebellum | Positive (lateral ventricle swab, spinal cord, mesenteric LN, brain, CSF) | - | Neurobrucellosis | Yes |
| N-274/18 | Loss of body condition; brain edema and meningeal hemorrhage |
Non-suppurative meningitis, diffuse, with multifocal encephalitis; choroiditis | Positive (lateral ventricle swab) | Aeromonas sobria and P. damselae in lung, liver, CNS | Neurobrucellosis | Yes |
| N-314/19 | CSF turbidity; brain edema and meningeal hemorrhage |
Non-suppurative meningoencephalitis, more intense in brainstem, cerebellum and thalamus; choroiditis; radiculoneuritis, multifocal and milder | Positive (CSF) | - | Neurobrucellosis | Yes |
| N-368/19 | Emaciation; absence of recent ingesta; CSF turbidity | Non-suppurative meningitis, localized in brainstem and, with less intensity, cerebellum |
Negative (contaminated sample) PCR intensely positive in CSF. |
- | Neurobrucellosis | Yes |
| N-333/22 | Slight loss of body condition; absence of regent ingesta; brain edema; meningeal and CSF turbidity | Non-suppurative meningitis, localized in brainstem and cerebellum, choroiditis | Positive (brain, CSF) | - | Neurobrucellosis | Yes |
| N-215/23 | Absence of regent ingesta; CSF turbidity; hydrocephalus |
Non-suppurative meningitis, especially in brainstem and, with lesser intensity, cerebellum; choroiditis; radiculoneuritis, multifocal and milder |
Positive (CSF, ventricular swab) | - | Neurobrucellosis | No |
| Species | Cause of death | n | Antibody titer | |||||||
| >1:5 | 1:5 | 1:10 | 1:20 | 1:40 | 1:80 | 1:160 | %Pos | |||
| Sc | Neurobrucellosis* | 8 | 1 | 7 | 87.5% | |||||
| Bycatch | 15 | 4 | 1 | 3 | 1 | 2 | 4 | 26.6% | ||
| CeMV | 12 | 5 | 1 | 1 | 1 | 4♦ | 33.3% | |||
| Infectious** | 6 | 1 | 1 | 2 | 2 | 0% | ||||
| PEM | 4 | 1 | 1 | 2 | 50% | |||||
| Mother-calf separation | 2 | 1 | 1 | 0% | ||||||
| Other*** | 7 | 1 | 2 | 2 | 2 | 28.6% | ||||
| Unknown | 3 | 1 | 2 | 0% | ||||||
| TOTAL | 57 | 13 | 4 | 7 | 5 | 4 | 5 | 19 | 33.3% | |
| Gg | Sinusitis Crassicauda | 3 | 2 | 1 | 0% | |||||
| Bycatch | 1 | 1 | 100% | |||||||
| Mother-calf separation | 1 | 1 | 100% | |||||||
| TOTAL | 5 | 2 | 1 | 2 | 40% | |||||
| Tt | Mother-calf separation | 1 | 1 | 0% | ||||||
| Erysipelas | 1 | 1 | 0% | |||||||
| TOTAL | 2 | 1 | 1 | 0% | ||||||
| Dd | Unknown | 1 | 1 | 0% | ||||||
| Gm | CeMV | 1 | 1 | 0% | ||||||
| TOTAL | 66 | 45 | 21 | 31.8% | ||||||
| Seropositives (%) | Neurobrucellosis (%) | |
| Adults | 12/41 (29.3%) | 4/64 (6.3%) |
| Juveniles | 7/13 (53.9%) | 5/24 (20.8%) |
| Calves | 0/3 (0%) | 0/14 (0%) |
| Fetus | 0 | 0/1 (0%) |
| Total | 19/57 (33.3%) | 9/88 (14%) |
| ID | Species | Age group | CeMV form | Titer |
| N-319/16 | Sc | J | Systemic* | 1/160 |
| N-044/17 | Sc | J | Systemic | >1:5 |
| N-045/17 | Sc | J | Systemic | >1:5 |
| N-077/17 | Sc | J | Systemic | >1:5 |
| N-454/17 | Sc | J | Systemic | >1:5 |
| N-488/17 | Sc | Ad | Systemic | 1/5 |
| N-497/17 | Sc | Ad | Systemic | 1/10 |
| N-604/17 | Sc | Ad | Chronic | >1:5 |
| N-001/18 | Sc | Ad | Chronic | 1/40 |
| N-232/18 | Sc | J | Chronic | 1/160 |
| N-293/18 | Sc | Ad | Chronic | 1/160 |
| N-362/18 | Sc | Ad | Chronic | 1/160 |
| N-361/19 | Sc | J | Chronic | 1/160 |
| N-334/21 | Sc | Ad | Chronic | 1/80 |
| N-023/21 | Gm | J | Chronic | >1:5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





