1. Introduction
Stress, whether in form of short- or long-term exposure, poses major health threats for society [
3,
4,
5,
6]. Short-term, or acute stress, can lead to instantaneous health hazards such as accidents caused by poor driving decisions [
7], whereas long-term, or chronic stress, can cause serious mental conditions affecting people’s physical and psychological health [
4].
Wearable-based physiological data collection has become a main line of research with applications in the fields of medicine, psychology, bioinformatics and geoinformatics. Due to the advancement of sensor technologies, several biosignals have been used to derive and understand the complex psychophysiological response of the human body in accordance with a particular environmental or psychological stressor [
8]. Before individual sensor recordings can be leveraged by an algorithm, appropriate signal filtering and processing procedures need to be performed to remove noise and other unwanted artifacts from the signal. After preprocessing the raw data, biometric features can be derived from the time- and frequency-domain representation of the signal. These features are then used as input to expert-driven rule-based [
9,
10] or machine learning (ML) [
11] based classification systems for the detection of stress.
While machine learning approaches have shown to be effective approaches for identifying a (non-) linear mapping function between a set of input features and a particular output, the individual algorithms still require manually crafted features as inputs. Time-series biosignals however, can be considered unstructured information, data without an inherent data-model or a predetermined organization, where features have to be derived from aggregations of their time- or frequency-domain representations. This is where deep learning (DL) approaches have come to shine within recent years. Deep learning algorithms are known to be highly non-linear systems which are powerful tools to learn structure from data [
12]. End-to-end neural network (NN) architectures can deal with unstructured, raw data inputs and allow for skipping the manual feature engineering step in the ML pipeline. A prominent example is the transformer architecture, which recent large language models (LLMs), i.e., BERT [
13], are based on.
A major requirement for all these approaches, however, is the amount of available data that the model can be trained on. Depending on the type of input data, different NN architectures are advantageous with respect to their ability to process respective data sources. For text and image data, there exists a vast number of open data sources which can be used to (pre-) train individual architectures. Within the domain of wearable sensor data, there is only a small number of open-source datasets, i.e., the WESAD dataset [
14], which has frequently been used to detect stress and other health conditions based on the various psychological stress inducement scenarios that are used to simulate a particular stress condition, i.e., the Trier Social Stress Test (TSST) [
15]. In addition to this lack of publicly available benchmark datasets for the evaluation of physiological responses, existing methodologies have focused on the long-term effects of stress, using physiological biosignal data over time periods of several minutes, hours, or even days [
8,
16,
17], limiting the comparability of the proposed methodologies’ results.
Considering there is sufficient training data available, another downside of the increased predictive capabilities of complex DL models is the lack of explainability. Feature extraction mechanisms in the form of multiple non-linearities make interpretation of the decision process of the algorithm non-trivial, constituting a major drawback of several NN architectures. This lack of explainability in artificial intelligence (AI) methods has increasingly drawn attention to a new field of research, explainable AI (XAI). The main goal of XAI methods is to unfold the hidden processes involved in feature learning and decision making, aiming to better understand algorithmic decisions and making results more interpretable. Central motivations of explainable AI approaches are the explainability of a model’s decision process, the interpretability of the model’s predictions, and the transferability of the resulting information to other problem settings. A popular class of XAI techniques is feature attribution, which aims to find the most relevant features used by the model to arrive at a particular prediction [
18]. Within the context of stress detection based on psychophysiological reactions, the main objective is to automatically derive meaningful and explainable features from subsequences of a time series, ideally relating subjective human stress responses to existing knowledge stated in current literature.
Taking into consideration these challenges, we propose a deep learning framework to detect acute stress in a time window of 16 seconds, focusing on the explainable derivation of physiological signal features that indicate an immediate response to a stress stimulus. Potential applications of the proposed methodology range from traffic and urban planning use-cases, where city planners can gain a better understanding of people’s subjective perceptions in urban areas, to medical research, where the effectiveness of anesthesia can be evaluated based on people’s perceived stress levels. The algorithm is evaluated based on a dataset that was recorded in a strictly controlled laboratory environment where audio stress stimuli were used to induce states of emotional arousal.
The main contributions of our work are as follows:
An ensemble of LSTM networks, enriched through a deep generative ensemble of conditional GANs, outperforms current state-of-the-art rule-based stress detection systems proposed by [
10] and [
9].
Integrated Gradients offers an XAI approach to highlight significant features used by the DL model to predict stress. For electrodermal activity, these features are in line with existing literature and expert knowledge.
Skin temperature does not lead to significant contributions in the classification of acute stress, neither in the rule-based system, nor in the DL approach.
DL methodologies enable the automatic derivation of meaningful features from raw physiological biosignals in the time- and frequency domain.
2. Related Work
Researchers in the field of physiological stress detection have proposed several methods to automatically classify stress related events. Within this process, various use-cases, distinct physiological indicators and different experimental protocols were tested to classify stress in human subjects [
8,
19]. Some of the most promising physiological signals related to stress are electrodermal activity (EDA), skin temperature (ST), and cardiovascular activity measurements such as heart rate (HR), heart rate variability (HRV) and inter-beat interval (IBI) [
8,
20].
Current literature shows that especially EDA and derived features serve as reliable indicators for the detection of instant and non-subjective states of emotional arousal [
8,
17,
21,
22]. Based on a bandpass frequency filter, the raw EDA signal can be split up into the Skin Conductance Level (SCL), also known as the tonic part, and the Skin Conductance Response (SCR), which describes the phasic part of the signal, where non-specific SCR can be mainly attributed to noise [
8].
The SCR component of the EDA signal correlates with immediate stress, showing an immediate increase of the signal component as a response to a stressor [
23]. It has been shown that EDA, in combination with the cvxEDA [
24] preprocessing methodology and a support vector machine, is sufficient to accurately classify stress [
22]. A study conducted by [
17] shows a strong correlation of the EDA signal with stress during driving tasks, highlighting the suitability of skin conductance response measurements for stress assessments in non-stationary settings.
Another biosignal marker which has frequently been attributed to stress is ST [
8,
25]. Findings of these studies suggest that during a stress response of the human body, ST either decreases or increases, depending on the different body part the sensor is applied to. [
26] states that ST significantly decreases at the hand-palm and fingertips during TSST, but increases when measured at the upper arm. Another study [
25] suggests filtering the ST signal and using the information contained in the slope, instead of the mean value of the ST signal.
While previous studies propose different ST features in relation to stress, mainly long-term effects of ST changes in accordance with a stress response are considered. [
27] on the other hand, looks at the short-term response of ST and derived features, where oscillations caused by a stressor were found in the low frequency spectrum of the signal due to blood flow changes. Hence, appropriate frequency filtering enables the measurement of ST changes as a response to acute stressors.
To automatically detect stress from psychophysiological data, the majority of research leverages machine learning methods [
19].
Before individual signals can be fed as input to a ML algorithm, feature engineering needs to be performed, where various features are manually extracted via summary statistics over a given sliding window [
14,
28,
29]. The main issue here is the cost of feature engineering, where expert knowledge or extensive feature selection heuristics need to be evaluated to find valuable features that serve as indicators for stress.
Recent deep learning techniques have shown to be successful feature extractors when being exposed to raw input data, making them valuable tools for end-to-end machine learning pipelines. In these approaches, features are extracted automatically by the neural network. [
30] has shown that a Convolutional Neural Network (CNN) outperforms traditional machine learning approaches on various emotion and stress classification tasks, leveraging the WESAD [
14] dataset. In [
28], the authors compare handcrafted features as input to ML algorithms with deep learning methods and conclude that modern DL architectures show better performance in the task of classifying stress.
While previously mentioned DL approaches proved suitable for handling complex, unstructured input data, considering there exists a sufficient amount of training data [
31], their main drawback is the lack of explainability and interpretability.
Rule-based systems, crafted based on expert knowledge, offer an attractive alternative in this case, where the decision process of the algorithm follows a logical structure and results can be communicated to stakeholders more transparently. However, to avoid issues of generalization to new test subjects, physiological differences among individual participants are important to consider [
9].
Considering the strengths and weaknesses of the aforementioned approaches, we identified research gaps in terms of explainability of deep learning methods, and the suitability of these methodologies to work as automatic feature extractors in the context of time-series physiological sensor data. To close this research gap, the main goal of this paper is to compare rule-based expert systems such as [
9,
10] with an explainable deep learning algorithm to evaluate the model’s ability to automatically extract highly non-linear features from physiological data sources, serving as short-term time features for the task of classifying acute stress. Explainability is added to the approach by using Integrated Gradients to uncover the most relevant time-dependent features used by the DL algorithm for generating predictions.
3. Methodology
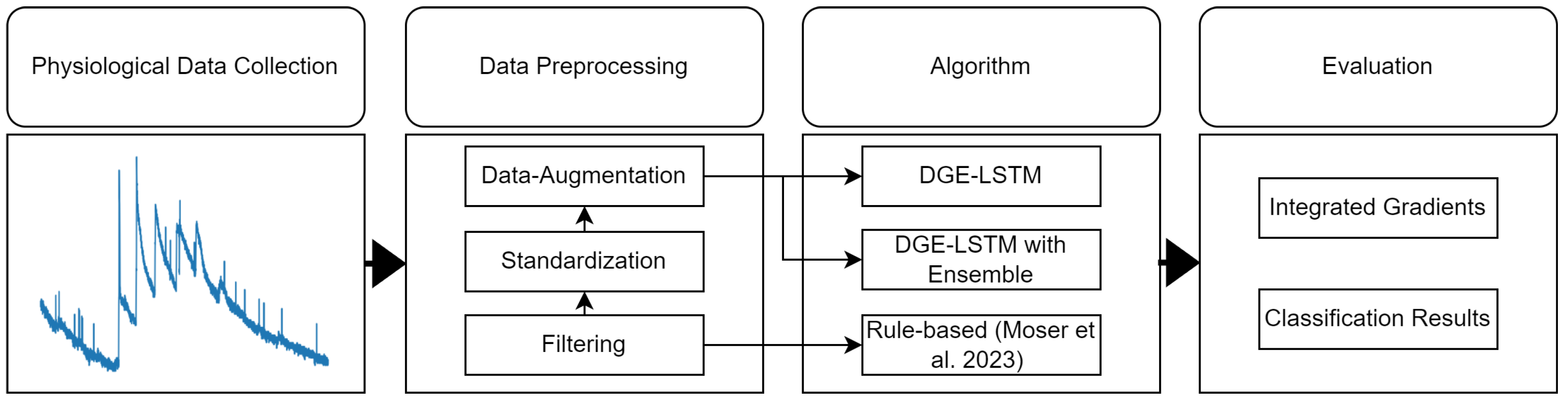
We propose a deep learning based approach for stress detection in wearable sensor data considering a short-term time-window of 16 seconds. Results of the DL methodology are compared to state-of-the-art rule-based expert systems, where the physiological dataset used for training and evaluation was collected in a strictly controlled laboratory test setting at the University of Salzburg. To assign appropriate labels to the physiological signals at given stress times, a specifically designed experimental protocol was followed. The data collection process is further described in
Section 3.1.
For comparison of the individual approaches, we developed a method which collects, processes and then automatically classifies individual stress moments with the Rule-based and the Deep learning-based algorithm. Results of the individual methodologies are used to evaluate the algorithms’ performance in terms of recall, precision, and accuracy, with a particular emphasis on Explainable AI (XAI), concretely focusing on the interpretability of the approach. A complete description of the Rule-based algorithm and the implementational details can be found in [
9].
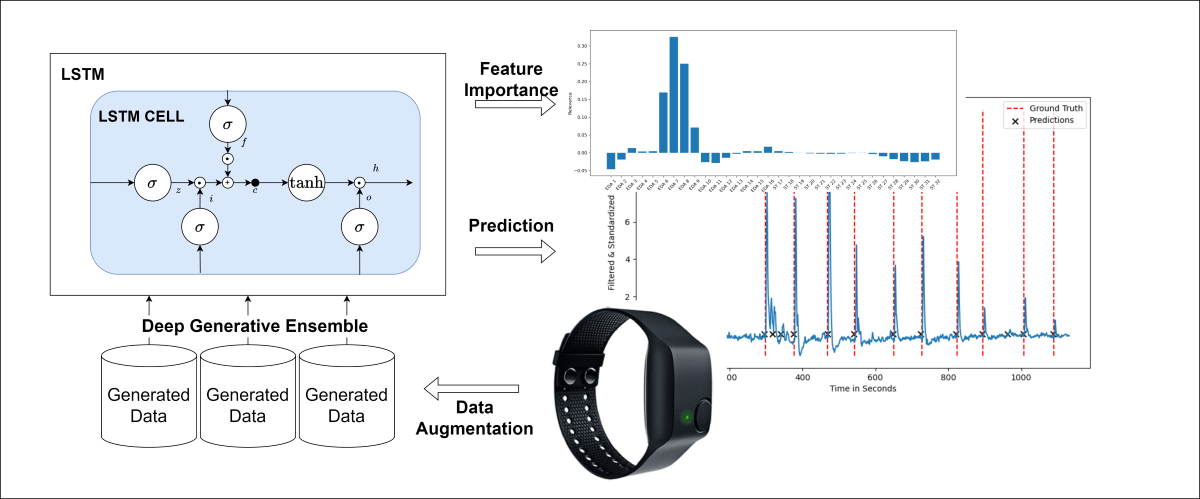
Figure 1 gives an overview of the methodology.
3.1. Physiological Data Collection
To the best of our knowledge, there is no publicly available dataset at the time of this writing where stress labels are given on the granularity level of individual seconds, which would be suitable for the task of short-term stress classification. To close this gap, we collect our own physiological stress datasets, where we follow the laboratory experimental protocol introduced in [
9,
10,
32]. High quality physiological sensor data was collected over a period of 4 years between 2018 and 2022, resulting in a dataset consisting of 28 subjects. All participants were asked not to consume any stimulants prior to the experiment. Each subject was equipped with the Empatica E4 wristband, which was then connected to an eDiary app [
33] to save the data into individual SQLite database files. More information about the sensor’s technical specifications is available at [
34]. Participants were divided into groups of 5, 4, or 3 persons per session and each session lasted between 15 to 22 minutes. After a short initiation phase to determine a baseline during a person’s state of relaxation, 10 stress causing interventions were performed via audio stimuli. These occurred in random intervals ranging from 52 seconds up to 125 seconds, with an average distance of 86 seconds between two consecutive stimuli. More details on the data collection procedure can be found in [
9,
10,
32].
3.2. Signal Processing
To prepare the data for the different stress detection methodologies, the following preprocessing steps were performed. First, individual signals obtained from the laboratory test participants were excluded when the sensor did not record any data, or in case of highly noisy recordings caused by improper sensor attachment. The resulting data therefore presents a homogeneous physiological dataset of high quality that will be made available to the public to foster research in the domain of physiological stress detection. EDA and ST signals were filtered using a bandpass filter, implemented in the SciPy python package [
35]. Prior to applying a first order high-pass filter with a cut off frequency of 0.05 Hz to extract the phasic component of the EDA signal, the raw signal was processed with a low-pass filter to remove noise. For the frequency filtering of the skin temperature signal, we use the same approach as proposed in [
32] and [
9].
After filtering both signals based on their frequency representation, each signal was downsampled from the original E4 sampling frequency of 4 Hz to 1 Hz in order to further clean the signals from artifacts that can be attributed to movement. In a next step, the signals were standardized with
to have a mean of 0 and a standard deviation of 1. The standardization of the individual signals has the effect of making the training procedure of the deep learning algorithm more efficient and removing bias related to differing value ranges among the attributes from the data, due to individual inputs being converted to the same scale. To finalize the preprocessing pipeline, the signals were split into sequences using a sliding window of 16 seconds, in which common EDA features such as the variation in latency between stressor and onset of the EDA, the rising time from the onset to the peak, and the recovery of the signal to the usual state are included. The individual characteristics of the EDA response, their duration, and the time window of 16 seconds are chosen based on current literature [
8,
36]
3.3. Deep Learning for Physiological Stress Detection
This section explains the proposed deep learning methodology to classify acute stress in more detail. Since we use physiological time series data, a recurrent neural network architecture with a sigmoid layer at the end provides a suitable choice to extract features from the sequential input signal and perform the classification. Since we focus on the derivation of explainable features through IG, we stick to a classic Recurrent Neural Network (RNN) architecture variant, the LSTM, rather than identifying the best possible combination of network architecture and hyperparameters for our classification setting. We decide against transformer architecture variants as we have a relatively short sequence length and our learning task does not require the identification of long-term dependencies. We opt for the LSTM network architecture as it provides a sufficiently complex recurrent neural network variant to capture the short-term time-dependent dependencies in the signal. The LSTM [
37] deals with vanishing gradient problem by using a memory cell and different gating mechanisms. As shown in Equation (5) the recurrent connections in the network are set to the identity, resulting in a constant error carousel, having the effect that the error is distributed evenly among the time steps when weights get updated during backpropagation. The gating mechanism controls how much information gets into and out of the memory cell, which can be seen in Equations (1), (2), (3) and (4). The forget gate [
38] was introduced to learn the removal of information from the memory cell, when the learning process gets saturated. Introducing the forget gate comes at the cost of re-introducing the problem of vanishing gradients, which can be mitigated by initializing the forget gate bias with 1 [
37,
38,
39]. In Equation (6), the updated hidden state is then computed. The recurrent network connection is initialized orthogonally, the input weights with xavier, and biases are initialized with 0. To find the best hyperparameters for the network, a grid search with Cross-validation (CV) on the training dataset is performed. The defined grid-search space can be seen in the
Appendix A, where results of the 5-Fold CV are displayed in bold
Table A1
While having a relatively large sample of test subjects compared to other studies [
14], our dataset can still be considered small for training a NN. To deal with the relatively small sample size and the imbalance with regard to stress and non-stress sequences, we used the conditional GAN proposed in [
32] to augment the dataset with synthetic data. To improve generalization while preventing overfitting on the training data, we added random gaussian noise to the stress moments and scaled the amplitude randomly, ultimately arriving at new samples for the training dataset [
40]. To increase generalization, we leverage a Deep Generative Ensemble (DGE) approach proposed in [
1]. Within this ensemble, synthetic data is generated from different random seeds, where predictions of the classifiers are then averaged for each of the seeds. This mitigates the effect of overfitting to the generative model’s samples, resulting in better predictions within a low data regime. To reduce variations in the predictions caused by a small test dataset, a homogenous ensemble [
41] of LSTMs, is used as our final classification model. The LSTM architecture is implemented with PyTorch [
42].
On top of the DL-based classification model, we apply Integrated Gradients (IG) [
2] to identify important features derived by the model with regard to classifying sequences as stress. In this time-series setting, we compute a straight-line path integral from a baseline, i.e., zero vector, to the original input over which the gradients at each point are calculated. As IG is a local feature attribution method, we take the average over all the stress samples in the test set to derive global information concerning the features used for predicting stress.
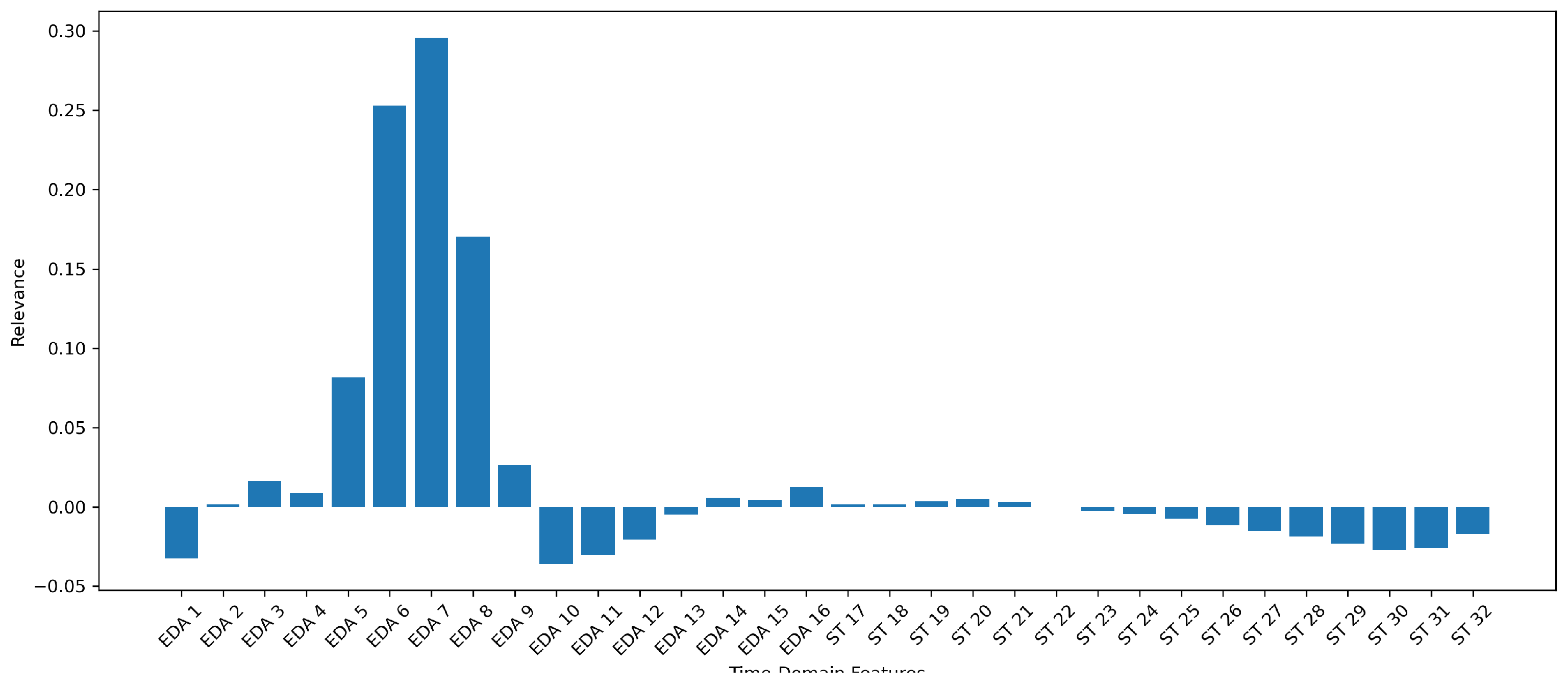
Figure 2 displays the relevance of the derived time-domain features in terms of stress predictions for one selected seed and ensemble model.
4. Experiments and Results
To compare the rule-based system proposed in [
9] with the deep learning algorithm described in the previous section, a number of different experiments were carried out. The first experiment focuses on the classification performance of the algorithms on our test dataset, using different seeds in the train-test split. In the second experiment, contributions of the individual features with regard to the overall classification performance are tested. The third and final experiment uses integrated gradients to enhance the interpretability and explainability of the results produced by the DL algorithm. The test dataset for each of the experiments consists of 10 randomly selected participants from the study setup described in
Section 3.1.
4.1. Stress Detection Results
The first experiment shows the ability of the LSTM and the Rule-Based algorithm to classify stress in a short-term time window of 16 seconds. Among the 28 laboratory test subjects, we perform a train-test split on the participant level, randomly selecting 10 participants per seed for evaluation, and the remaining 18 participants for training. The LSTM network is reinitialized in each of the 3 seeds and the training procedure is terminated once the validation loss stops improving. As mentioned in
Section 3.3, the best hyperparameter setting is determined based on a training dataset selected through 5-fold cross-validation. The selection is based on F1 score, with a special focus on recall, to avoid any False Negative (FN) predictions, corresponding to missed reference stress moments in our classification task. The hyperparameter setting with the maximum F1 score is chosen as the final model. For the Rule-Based algorithm, we implement and use the same rules and parameters as [
9]. Each algorithm is evaluated based on a sliding window approach, where a specified sliding window is moved over all participants within the evaluation set.
A True Positive (TP), a detected stress moment which coincides with a reference stress moment, is considered when the algorithm’s prediction is within a time window of 2 seconds before, and 6 seconds after a reference stressor. This evaluation window was chosen to account for all the variations within a stress situation considering current EDA literature [
8,
36]. A False Positive (FP) is a moment of stress (MOS) predicted by the algorithm, where within an 8 second time window no reference stressor occurred. On the contrary, if there is a reference stress moment and no prediction from the algorithm within 8 seconds, then the prediction is considered a False Negative (FN). Lastly, if the algorithm does not predict a stress situation and within 8 seconds of this prediction there is also no stressor, then the prediction is considered a True Negative (TN). To avoid an artificially high number of TPs and TNs, predictions which occur within a series of 8 seconds are summarized into one stress prediction, as can be seen in
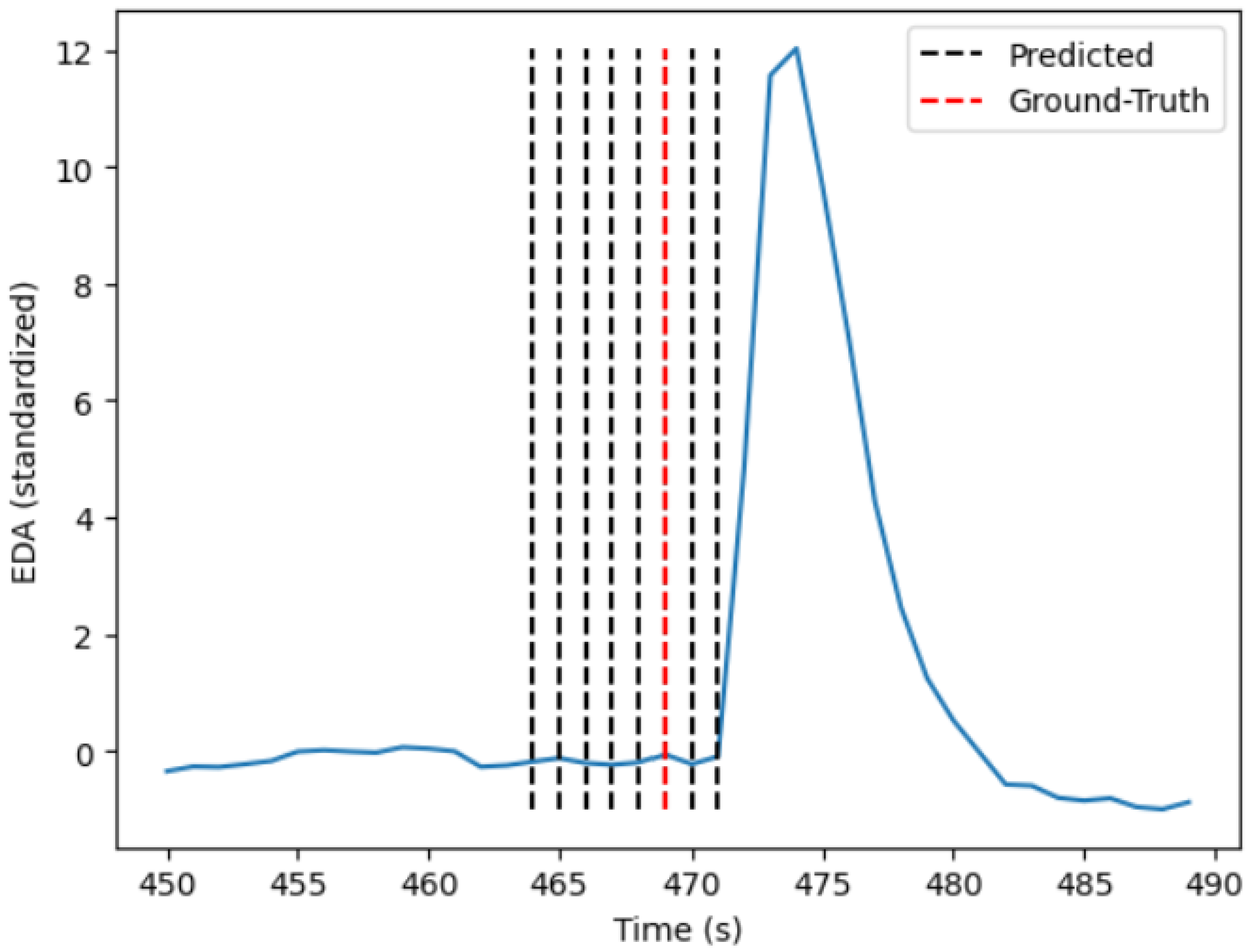
Figure 3.
Table 1 shows the results of the stress detection experiment. The best average recall is achieved by the LSTM (DGE) with an average score of 0.7633, followed by the ensemble of LSTMs (DGE) with an average recall score of 0.7367, and the rule-based algorithm with an average recall score of 0.7333. Precision values are higher for the deep learning approaches with an average precision score of 0.384 for the LSTM ensemble (DGE), and an average score of 0.359 for the LSTM (DGE). The rule-based algorithm reaches an average precision of 0.323, which is significantly lower than the ones achieved by the proposed DL models. In terms of accuracy, the best average score stems from the ensemble LSTM (DGE) with an accuracy of 0.9816, followed by the LSTM (DGE) with an accuracy of 0.9809, and the rule-based algorithm with an accuracy score of 0.9806. The best recall on a single seed, however, is from the rule-based algorithm, with a maximum score of 0.82.
4.2. Results with Regard to ST Contribution
Results of the second experiment show the contributions of the EDA and the ST signal to the overall classification score of the LSTM, considering the test-dataset. The LSTM model is trained without data-augmentation, but with the same hyperparameters determined by the previous experiment
Section 4.1, which can be seen in the appendix. Similarly, the same test-dataset seeds are used for evaluation.
Table 2 displays the results of the experiment on three different train-test split seeds. The first column shows the results of training with the EDA signal only, whereas the second shows the results of a combination of both signals during the training process. The average recall value with the combination of EDA and ST exceeds the one from EDA by an average of 2.66 %. While this suggests that adding the ST signal to the model enhances the algorithm’s stress prediction capability, this result should be interpreted with care, as the improvement mainly comes from one seed, hinting towards significant differences within the test individuals’ stress reaction, expressed in form of skin temperature variations. In terms of precision, there is only a minor average difference of 0.12 %, between the EDA and the combination of the two.
4.3. Interpretability of the Deep Learning Approach
As stated in the literature [
8,
10,
36] and used in the rule-based system [
9,
10], the relevant features of the phasic part of the EDA signal are the rise time from the onset to the peak, the latency from the stressor to the onset, and the recovery time of the signal back to the usual state. This is also present in the features used by the DL model.
Figure 2 highlights that the most relevant features are the rise of the peak and the recovery of the signal after the peak. Concerning the ST signal, where immediate patterns are not as clear as in the phasic component of the EDA signal, the model only found little relevance in the initial time frame of the signal and even some negative contributions towards the end of the signal. This can be interpreted as the likelihood of stress predictions being reduced if given features are present and used by the model.
5. Discussion
5.1. Discussion of Methodology
The paper proposes a methodology to classify short-term stress-related events with an ensemble of LSTMs, enriched through a Deep Generative Conditional GAN, and compares the results to a state-of-the-art Rule-Based System [
9].
As already mentioned, a deep learning approach tends to require plenty of training data to generalize well on unseen data [
31]. Since the collection of a physiological dataset with ground truth labels is costly, our dataset containing 180 MOS for training and 100 MOS for testing ends up being relatively small for a DL setting. To mitigate this problem, data-augmentation and a deep ensemble are used to reduce the chance of overfitting and minimize the variance within the predictions. We would recommend a similar procedure for researchers, who also work in the low data regime, i.e., [
14].
As generative models such as the conditional GAN [
32] used in this work, suffer from problems such as memorization of the dataset, mode collapse, or noisy data, we decided to use the Deep Generative Ensemble (DEG) approach proposed by [
1]. It has been shown that especially in the low data regime there is an improvement with the DGE approach. While this approach helps with typical problems that generative models face, it comes at the cost of increased computational complexity, as the generation process needs to run multiple times. Since we are dealing with a relatively small dataset, DGE provides a feasible approach for our use-case.
We decided to use a simple LSTM network since we wanted to demonstrate the capabilities of a deep learning algorithm to automatically extract meaningful and interpretable features from sequential physiological signals. To further improve the classification performance of the model, a more powerful deep ensemble method, e.g., stacking of a more heterogeneous ensemble of different model classes, or hyperparameters [
41], can be tested. In future studies, we suggest trialing other backbone architectures, i.e., a CNN or transformer, in combination with feature attribution methods such as integrated gradients, and comparing results to our presented algorithm.
To test the generalization capabilities of the presented algorithm, additional data collection studies in a laboratory test setting or non-stationary, real-world environments should be considered. One could use uncertainty estimation as proposed in [
43], to perform out of distribution classification and test the generalization capabilities of the model on different datasets.
5.2. Discussion of Results
The LSTM with DGE proposed in this paper outperforms the rule-based algorithm in terms of average recall and average precision.
The rule-based algorithm achieves an average value of 0.7333 and 0.2872 in terms of recall and precision. The LSTM with DGE results in an average recall of 0.7633 and an average precision of 0.359, demonstrating the capability of the DL methodology to derive meaningful features from the raw signal data.
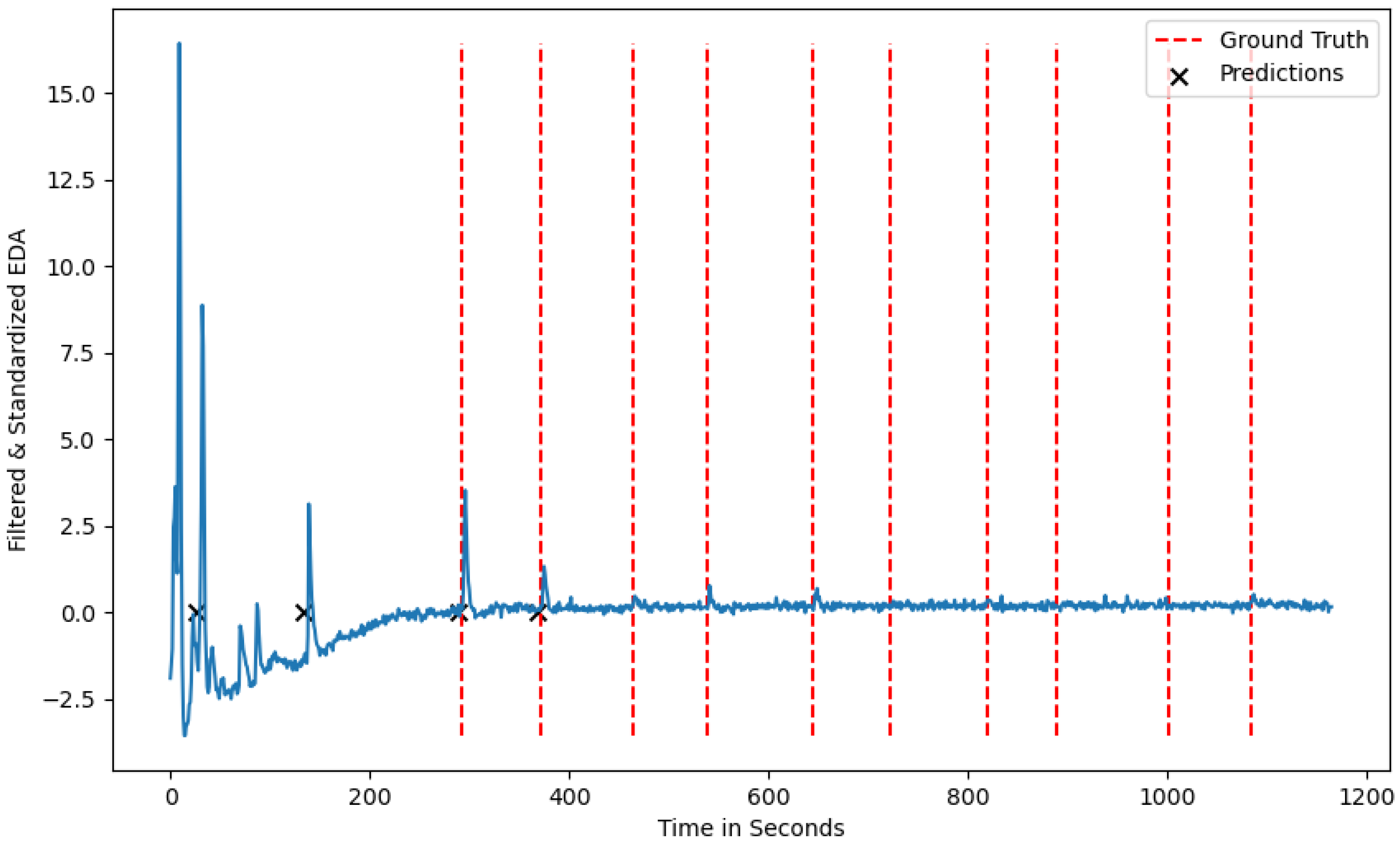
Both algorithms show some variations in terms of performance metrics for the different seeds. This variation can be attributed to the fact that certain participants show a less intense physiological reaction due to familiarity with the induced stressor, and extreme cases where there was no physiological reaction at all. The decreased stress response pattern of one participant can be seen in Figure 5, where only 4 out of 10 ground truth stressors are visually present in the signal. Considering this observation, no algorithm would be able to detect more than the number of stress responses inherent in the phasic component of the EDA signal, leading to a decreased recall score for certain seeds.
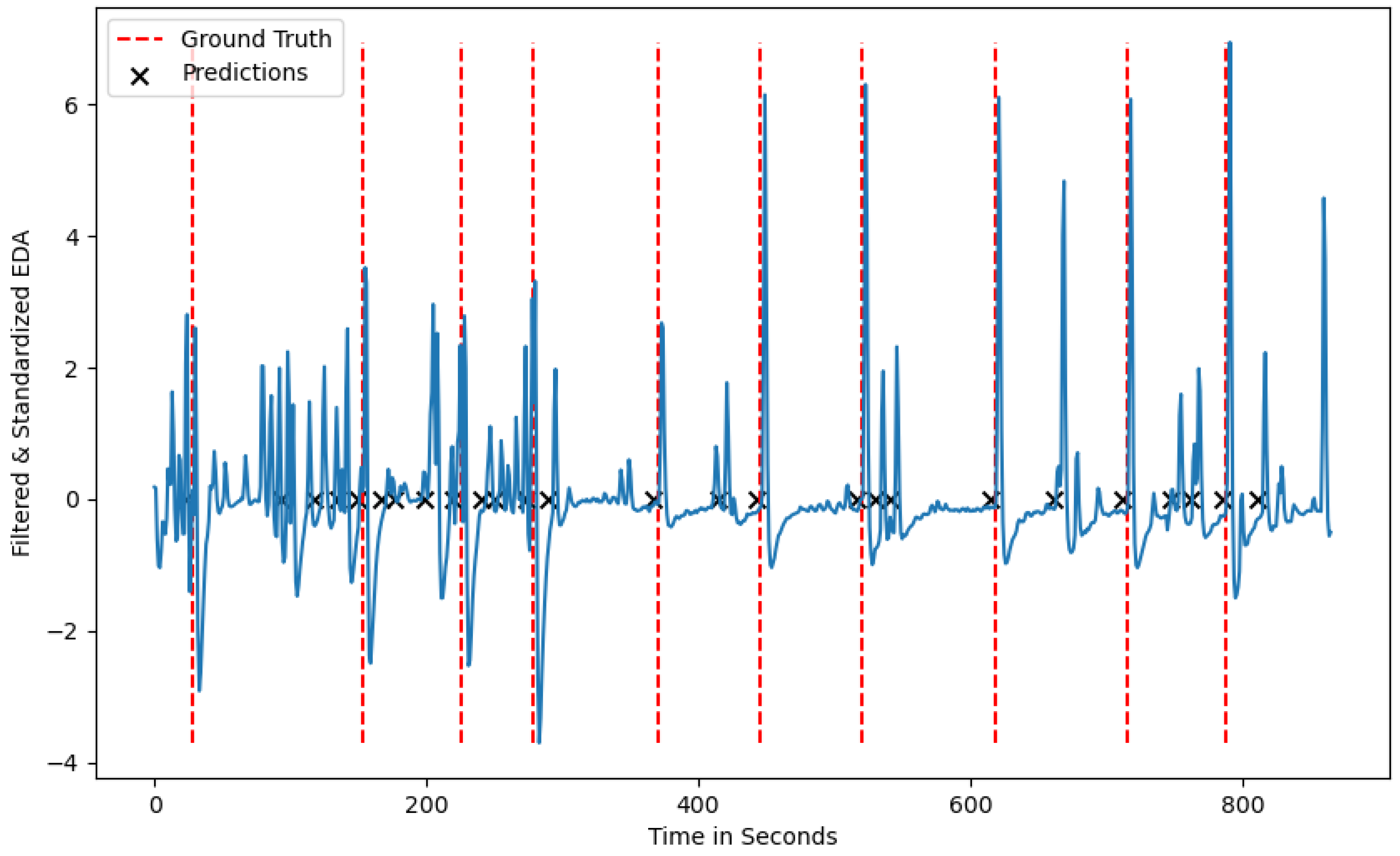
Low average precision values can be attributed to participants who show phasic EDA peaks in between the ground truth stressors, which the algorithm considers as stress situations. This is visualized in
Figure 4, where multiple peaks occur in between the induced stress times and can be attributed to participants being stressed by other sources, or the sensor being loosely attached to a participant’s wrist. Depending on the use-case the algorithm is applied to, the trade-off between TP and FP needs to be considered. As we plan on using the algorithm in the context of urban planning to detect spatio-temporal stress clusters, we aim for a high recall value which minimizes the number of FN predictions, i.e., stress situations the algorithm fails to detect [
44]. To further reduce the number of FN, we aim to incorporate other biometric information such as cardiovascular activity and eye-tracking data. Adding such information could lead to a more comprehensive understanding of the situation and potential stress-causing factors.
Figure 4.
A test participant from the test-dataset with the ground-truth stressors and the predicted stressors showing noise in between the ground-truth labels.
Figure 4.
A test participant from the test-dataset with the ground-truth stressors and the predicted stressors showing noise in between the ground-truth labels.
Figure 5.
A test participant from the test-dataset with the ground-truth stressors and the predicted stressors without a reaction at the ground-truth labels.
Figure 5.
A test participant from the test-dataset with the ground-truth stressors and the predicted stressors without a reaction at the ground-truth labels.
6. Conclusions
We propose an explainable LSTM approach to acute stress classification in a controlled laboratory environment based on wearable sensor data, where we compare the results to a state-of-the-art rule-based system which implements domain knowledge from experts. The proposed Deep LSTM Ensemble method for detecting stress improves the current state-of-the-art by an average recall of up to 3 % and an average precision of up to 3.6 %. The problem of a small and unbalanced data set is mitigated by a combination of data augmentation, synthetic data generation based on a conditional GAN, and a Deep Generative Ensemble. To address explainability aspects of the proposed LSTM ensemble, we use Integrated Gradients (IG) to explore the prediction behavior of the model. By using IG, we show that the LSTM learns and uses similar signal patterns as the rule-based algorithm to predict stress-related events, which is in line with current literature on physiological stress detection. In a separate experiment, we show that the addition of the filtered short-term skin temperature signal does not significantly improve the predictive score in terms of recall and precision, implying that more research is needed to find consistent patterns in the ST signal which can be related to acute stress.
In order to better understand and algorithmically capture the complex stress reaction of the human body, we recommend that other biomarkers which can be measured through non-invasive wearable devices, i.e., BVP, HR or HRV, should be considered and integrated in future research.
Author Contributions
Conceptualization, Martin Moser, Maximilian Ehrhart and Bernd Resch; Data curation, Martin Moser; Methodology, Martin Moser, Maximilian Ehrhart and Bernd Resch; Supervision, Bernd Resch; Writing – original draft, Martin Moser and Maximilian Ehrhart; Writing – review & editing, Martin Moser and Bernd Resch.
Funding
This work was submitted for review on 04 July 2024. The research was funded by the Austrian Science Fund (FWF) through the project “Urban Emotions” (reference number I-3022) and by the German Federal Ministry for Digital and Transport (BMDV) through the project ESSEM (reference no. 19F2195A).
Institutional Review Board Statement
This work involved human subjects in its research. Approval of the research activities including ethical and experimental considerations was granted by the Austrian Science Fund (FWF) through the Urban Emotions project (reference number I-3022), and performed in line with the 2013 WMA Declaration of Helsinki.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Abbreviations
The following abbreviations are used in this manuscript:
| AC |
Alternating Current |
| ANS |
Autonomic Nervous System |
| DC |
Direct Current |
| DL |
Deep Learning |
| ECG |
Electrocardiography |
| EDA |
Electrodermal Activity |
| EDL |
Electrodermal Level |
| EDR |
Electrodermal Response |
| FN |
False Negative |
| FP |
False Positive |
| FPR |
False Positive Rate |
| GAN |
Generative Adversarial Network |
| GNSS |
Global Navigation Satellite System |
| GSR |
Galvanic Skin Response |
| HR |
Heart Rate |
| HRV |
Heart Rate Variability |
| Hz |
Hertz |
| IBI |
Inter-Beat Interval |
| IG |
Integrated Gradients |
| LDA |
Linear Discriminant Analysis |
| LUCCK |
Learning Using Concave and Convex Kernels |
| LSTM |
Long-Short Term Memory Network |
| ML |
Machine Learning |
| MOS |
Moment of Stress |
| NN |
Neural Network |
| PPG |
Photoplethysmography |
| SC |
Skin Conductance |
| SCL |
Skin Conductance Level |
| SCR |
Skin Conductance Response |
| ST |
Skin Temperature |
| SVM |
Support Vector Machine |
| TN |
True Negative |
| TP |
True Positive |
| TPR |
True Positive Rate |
| VR |
Virtual Reality |
Appendix A
Appendix A.1. Hyperparameters
Table A1.
Hyperparameters tested in the Grid-Search (values in bold highlight the best hyperparameters found).
Table A1.
Hyperparameters tested in the Grid-Search (values in bold highlight the best hyperparameters found).
| Hyperparameter |
Values |
| Number of Layers |
[1, 2] |
| Number of MOS-Augmented |
[400, 800, 1200] |
| Number of non-MOS-Augmented |
[400, 800, 1200] |
| Units |
[32, 64] |
| Inital Learning Rate |
[0.01, 0.001, 0.0001] |
| Learning Rate Schedular |
Cosine Scheduler |
| Optimizer |
Adam with Weight Decay |
References
- Van Breugel, B.; Qian, Z.; van der Schaar, M. Synthetic data, real errors: how (not) to publish and use synthetic data. arXiv preprint arXiv:2305.09235 2023.
- Sundararajan, M.; Taly, A.; Yan, Q. Axiomatic attribution for deep networks. In Proceedings of the International conference on machine learning. PMLR, 2017, pp. 3319–3328.
- Hefez, A.; Metz, L.; Lavie, P. Long-term effects of extreme situational stress on sleep and dreaming. Am J Psychiatry 1987, 144, 344–347. [Google Scholar] [PubMed]
- McGonagle, K.A.; Kessler, R.C. Chronic stress, acute stress, and depressive symptoms. American journal of community psychology 1990, 18, 681–706. [Google Scholar] [CrossRef] [PubMed]
- Schubert, C.; Lambertz, M.; Nelesen, R.; Bardwell, W.; Choi, J.B.; Dimsdale, J. Effects of stress on heart rate complexity—a comparison between short-term and chronic stress. Biological psychology 2009, 80, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Dhabhar, F.S. Effects of stress on immune function: the good, the bad, and the beautiful. Immunologic research 2014, 58, 193–210. [Google Scholar] [CrossRef] [PubMed]
- McMurray, L. Emotional stress and driving performance: The effect of divorce. Behavioral Research in Highway Safety 1970. [Google Scholar]
- Giannakakis, G.; Grigoriadis, D.; Giannakaki, K.; Simantiraki, O.; Roniotis, A.; Tsiknakis, M. Review on psychological stress detection using biosignals. IEEE Transactions on Affective Computing 2019, 13, 440–460. [Google Scholar] [CrossRef]
- Moser, M.K.; Resch, B.; Ehrhart, M. An Individual-oriented Algorithm for Stress Detection in Wearable Sensor Measurements. IEEE Sensors Journal 2023. [Google Scholar] [CrossRef]
- Kyriakou, K.; Resch, B.; Sagl, G.; Petutschnig, A.; Werner, C.; Niederseer, D.; Liedlgruber, M.; Wilhelm, F.; Osborne, T.; Pykett, J. Detecting moments of stress from measurements of wearable physiological sensors. Sensors 2019, 19, 3805. [Google Scholar] [CrossRef] [PubMed]
- Gedam, S.; Paul, S. A review on mental stress detection using wearable sensors and machine learning techniques. IEEE Access 2021, 9, 84045–84066. [Google Scholar] [CrossRef]
- Goodfellow, I.; Bengio, Y.; Courville, A. Deep learning; MIT press, 2016.
- Devlin, J.; Chang, M.W.; Lee, K.; Toutanova, K. Bert: Pre-training of deep bidirectional transformers for language understanding. arXiv preprint arXiv:1810.04805 2018.
- Schmidt, P.; Reiss, A.; Duerichen, R.; Marberger, C.; Van Laerhoven, K. Introducing wesad, a multimodal dataset for wearable stress and affect detection. In Proceedings of the Proceedings of the 20th ACM international conference on multimodal interaction, 2018, pp. 400–408.
- Kirschbaum, C.; Pirke, K.M.; Hellhammer, D.H. The ‘Trier Social Stress Test’–a tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 1993, 28, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Setz, C.; Arnrich, B.; Schumm, J.; La Marca, R.; Tröster, G.; Ehlert, U. Discriminating stress from cognitive load using a wearable EDA device. IEEE Transactions on information technology in biomedicine 2009, 14, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Healey, J.A.; Picard, R.W. Detecting stress during real-world driving tasks using physiological sensors. IEEE Transactions on intelligent transportation systems 2005, 6, 156–166. [Google Scholar] [CrossRef]
- La Rosa, B.; Blasilli, G.; Bourqui, R.; Auber, D.; Santucci, G.; Capobianco, R.; Bertini, E.; Giot, R.; Angelini, M. State of the art of visual analytics for explainable deep learning. In Proceedings of the Computer Graphics Forum. Wiley Online Library, 2023, Vol. 42, pp. 319–355.
- Vos, G.; Trinh, K.; Sarnyai, Z.; Azghadi, M.R. Generalizable machine learning for stress monitoring from wearable devices: a systematic literature review. International Journal of Medical Informatics 2023, 105026. [Google Scholar] [CrossRef] [PubMed]
- Kreibig, S.D. Autonomic nervous system activity in emotion: A review. Biological psychology 2010, 84, 394–421. [Google Scholar] [CrossRef] [PubMed]
- Farrow, T.F.; Johnson, N.K.; Hunter, M.D.; Barker, A.T.; Wilkinson, I.D.; Woodruff, P.W. Neural correlates of the behavioral-autonomic interaction response to potentially threatening stimuli. Frontiers in human neuroscience 2013, 6, 349. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Valenza, G.; Lazaro, J.; Garzon-Rey, J.M.; Aguilo, J.; De-la Camara, C.; Bailon, R.; Scilingo, E.P. Acute stress state classification based on electrodermal activity modeling. IEEE Transactions on Affective Computing 2021. [Google Scholar] [CrossRef]
- Dawson, M.E.; Schell, A.M.; Filion, D.L. The electrodermal system. Handbook of psychophysiology 2007, 2, 200–223. [Google Scholar]
- Greco, A.; Valenza, G.; Lanata, A.; Scilingo, E.P.; Citi, L. cvxEDA: A convex optimization approach to electrodermal activity processing. IEEE transactions on biomedical engineering 2015, 63, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Barreto, A. Stress detection in computer users based on digital signal processing of noninvasive physiological variables. In Proceedings of the 2006 international conference of the IEEE engineering in medicine and biology society. IEEE, 2006, pp. 1355–1358.
- Vinkers, C.H.; Penning, R.; Hellhammer, J.; Verster, J.C.; Klaessens, J.H.; Olivier, B.; Kalkman, C.J. The effect of stress on core and peripheral body temperature in humans. Stress 2013, 16, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Shusterman, V.; Anderson, K.P.; Barnea, O. Spontaneous skin temperature oscillations in normal human subjects. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 1997, 273, R1173–R1181. [Google Scholar] [CrossRef] [PubMed]
- Bobade, P.; Vani, M. Stress detection with machine learning and deep learning using multimodal physiological data. In Proceedings of the 2020 Second International Conference on Inventive Research in Computing Applications (ICIRCA). IEEE, 2020, pp. 51–57.
- Smets, E.; Casale, P.; Großekathöfer, U.; Lamichhane, B.; De Raedt, W.; Bogaerts, K.; Van Diest, I.; Van Hoof, C. Comparison of machine learning techniques for psychophysiological stress detection. In Proceedings of the Pervasive Computing Paradigms for Mental Health: 5th International Conference, MindCare 2015, Milan, Italy, September 24-25, 2015, Revised Selected Papers 5. Springer, 2016, pp. 13–22.
- Li, R.; Liu, Z. Stress detection using deep neural networks. BMC Medical Informatics and Decision Making 2020, 20, 1–10. [Google Scholar] [CrossRef] [PubMed]
- LeCun, Y.; Bengio, Y.; Hinton, G. Deep learning. nature 2015, 521, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Ehrhart, M.; Resch, B.; Havas, C.; Niederseer, D. A Conditional GAN for Generating Time Series Data for Stress Detection in Wearable Physiological Sensor Data. Sensors 2022, 22, 5969. [Google Scholar] [CrossRef] [PubMed]
- Petutschnig, A.; Reichel, S.; Měchurová, K.; Resch, B. An eDiary App Approach for collecting physiological Sensor Data from Wearables together with subjective observations and emotions. Sensors 2022, 22, 6120. [Google Scholar] [CrossRef] [PubMed]
- E4 wristband | Real-time physiological signals | Wearable PPG, EDA, Temperature, Motion sensors.
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental Algorithms for Scientific Computing in Python. Nature Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Boucsein, W. Electrodermal activity; Springer Science & Business Media, 2012.
- Hochreiter, S.; Schmidhuber, J. Long short-term memory. Neural computation 1997, 9, 1735–1780. [Google Scholar] [CrossRef] [PubMed]
- Gers, F.A.; Schmidhuber, J.; Cummins, F. Learning to forget: Continual prediction with LSTM. Neural computation 2000, 12, 2451–2471. [Google Scholar] [CrossRef] [PubMed]
- Jozefowicz, R.; Zaremba, W.; Sutskever, I. An empirical exploration of recurrent network architectures. In Proceedings of the International conference on machine learning. PMLR, 2015, pp. 2342–2350.
- Um, T.T.; Pfister, F.M.J.; Pichler, D.; Endo, S.; Lang, M.; Hirche, S.; Fietzek, U.; Kulić, D. Data augmentation of wearable sensor data for parkinson’s disease monitoring using convolutional neural networks. In Proceedings of the Proceedings of the 19th ACM International Conference on Multimodal Interaction. ACM, 2017, ICMI ’17. [CrossRef]
- Ganaie, M.; Hu, M.; Malik, A.; Tanveer, M.; Suganthan, P. Ensemble deep learning: A review. Engineering Applications of Artificial Intelligence 2022, 115, 105151. [Google Scholar] [CrossRef]
- Paszke, A.; Gross, S.; Massa, F.; Lerer, A.; Bradbury, J.; Chanan, G.; Killeen, T.; Lin, Z.; Gimelshein, N.; Antiga, L.; et al. PyTorch: An Imperative Style, High-Performance Deep Learning Library. In Advances in Neural Information Processing Systems 32; Curran Associates, Inc., 2019; pp. 8024–8035.
- Lakshminarayanan, B.; Pritzel, A.; Blundell, C. Simple and scalable predictive uncertainty estimation using deep ensembles. Advances in neural information processing systems 2017, 30. [Google Scholar]
- Kyriakou, K.; Resch, B. Spatial analysis of moments of stress derived from wearable sensor data. Advances in Cartography and GIScience of the ICA 2019, 2, 1–8. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).