Submitted:
26 July 2024
Posted:
30 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Protocol
2.3. Standard Echocardiography
2.4. Speckle-Tracking Echocardiography
2.5. Statistical Analysis
3. Results
3.1. Clinical Characteristics of Patients Included in The Study over Follow-Up
3.2. Echocardiography
4. Discussion
5. Study limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pop-Busui R, Januzzi JL, Bruemmer D, et al. Heart Failure: An Underappreciated Complication of Diabetes. A Consensus Report of the American Diabetes Association. Diabetes Care 2022;45:1670–1690. [CrossRef]
- Utina TG, Akasheva DU, Korsunsky DV, Drapkina OM. Significance of standard and speckle-tracking echocardiography for early diagnosis of asymptomatic left ventricular dysfunction in type 2 diabetes. Cardiovascular Therapy and Prevention 2023;22(1):47-57. [CrossRef]
- Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur J Echocardiogr 2016;17:1321–1360. [CrossRef]
- Nyberg J, Jakobsen EO, Østvik A, et al. Echocardiographic Reference Ranges of Global Longitudinal Strain for All Cardiac Chambers Using Guideline-Directed Dedicated Views. J Am Coll Cardiol Img 2023;16:1516–1531. [CrossRef]
- Cameli M. Echocardiography strain: why is it used more and more? European Heart Journal Supplements 2022;24(SupplementI):I38–I42. [CrossRef]
- Stanton AM, Vaduganathan M, Chang LS, et al. Asymptomatic Diabetic Cardiomyopathy: an Underrecognized Entity in Type 2 Diabetes. Current Diabetes Reports 2021;21:41. [CrossRef]
- Segar MW, Khan MS, Patel KV, et al. Prevalence and Prognostic Implications of Diabetes With Cardiomyopathy in Community-Dwelling Adults. J Am Coll Cardiol 2021;78:1587–1598. [CrossRef]
- Swiatkiewicz I, Patel NT, Villarreal-Gonzalez MA and Taub PR. Prevalence of diabetic cardiomyopathy in patients with type 2 diabetes in a large academic medical center. BMC Medicine 2024;22:195. [CrossRef]
- Marwick TH, Ritchie R, Shaw JE, Kaye D. Implications of Underlying Mechanisms for the Recognition and Management of Diabetic Cardiomyopathy. J Am Coll Cardiol 2018; 71:339. [CrossRef]
- Rubler S, Dlugash J, Yuceoglu YZ, et al. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol 1972; 30(6): 595-602.
- Lee SH, Park J-H. The Role of Echocardiography in Evaluating Cardiovascular Diseases in Patients with Diabetes Mellitus. Diabetes Metab J 2023;47:470-483. [CrossRef]
- Bouthoorn S, Valstar GB, Gohar A, et al. The prevalence of left ventricular diastolic dysfunction and heart failure with preserved ejection fraction in men and women with type 2 diabetes: A systematic review and meta-analysis. Diab Vasc Dis Res 2018;15:477-93. [CrossRef]
- Ponikowski P, Voors A, Anker S, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur J Heart Fail 2016;18:891-975. [CrossRef]
- Grigorescu ED, Lacatusu CM, Floria M, et al. Left Ventricular Diastolic Dysfunction in Type 2 Diabetes-Progress and Perspectives. Diagnostics 2019;9:121. [CrossRef]
- Kosmala W, Marwick TH. Asymptomatic Left Ventricular Diastolic Dysfunction. Predicting Progression to Symptomatic Heart Failure. J Am Coll Cardiol 2020;13:215-27. [CrossRef]
- Marwick TH, Shah SJ, Thomas JD. Myocardial Strain in the Assessment of Patients With Heart Failure. JAMA Cardiol 2019;4(3):287-94. [CrossRef]
- Silva TRW, Silva RL, Martins AF, Marques JLB. Role of Strain in the Early Diagnosis of Diabetic Cardiomyopathy. Arq Bras Cardiol: Imagem cardiovasc 2022;35(2):eabc293. [CrossRef]
- Tanaka H, Tatsumi K, Matsuzoe H, Matsumoto K and Hirata K. Impact of diabetes mellitus on left ventricular longitudinal function of patients with non-ischemic dilated cardiomyopathy. Cardiovasc Diabetol 2020;19:84. [CrossRef]
- Wang Y, Yang H, Huynh Q, et al. Diagnosis of Nonischemic Stage B Heart Failure in Type 2 Diabetes Mellitus: Optimal Parameters for Prediction of Heart Failure. JACC: Cardiovascular Imaging 2018;11:1390–400. [CrossRef]
- Holland DJ, Marwick TH, Haluska BA, et al. Subclinical LV dysfunction and 10-year outcomes in type 2 diabetes mellitus. Heart 2015;101(13):1061–6. [CrossRef]
- Athithan L, Gulsin GS, McCann GP, Levelt E. Diabetic cardiomyopathy: Pathophysiology, theories and evidence to date. World J Diabetes 2019, 10, 490–510. [CrossRef]
- Nakamura K, Miyoshi T, Yoshida M, et al. Pathophysiology and Treatment of Diabetic Cardiomyopathy and Heart Failure in Patients with Diabetes Mellitus. Int J Mol Sci 2022, 23, 3587. [CrossRef]
- Bozkurt B, Coats AJ, Tsutsui H, et al. Universal definition and classification of heart failure: a report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure. J Card Fail 2021 [Epub ahead of print]. [CrossRef]
- Meijers WC, Hoekstra T, Jaarsma T, et al. Patients with heart failure with preserved ejection fraction and low levels of natriuretic peptides. Neth Heart J 2016;24:287-95. [CrossRef]
- Verbrugge FH, Omote K, Reddy J, et al. Heart failure with preserved ejection fraction in patients with normal natriuretic peptide levels is associated with increased morbidity and mortality. European Heart Journal 2022; 43:ehab911. [CrossRef]
- Halabi A, Potter E, Yang H, et al. Association of biomarkers and risk scores with subclinical left ventricular dysfunction in patients with type 2 diabetes mellitus. Cardiovascular Diabetology 2022; 21:278. [CrossRef]
- Shah SJ. BNP: Biomarker Not Perfect in heart failure with preserved ejection fraction. European Heart Journal 2022; 43:1952. [CrossRef]
- Utina TG, Akasheva DU, Korsunsky DV, Dzhioeva ON, Drapkina OM. Biomarkers and subclinical left ventricular dysfunction in patients with type 2 diabetes without clinical manifestations of cardiovascular diseases. Cardiovascular Therapy and Prevention 2024;23(1):3914. [CrossRef]
- Dal Canto E, Scheffer M, Kortekaas K, et al. Natriuretic Peptide Levels and Stages of Left Ventricular Dysfunction in Heart Failure with Preserved Ejection Fraction. Biomedicines 2023, 11, 867. [CrossRef]

| Variable | Patients with T2D (n=29) | Patients without T2D (n=28) | p value |
| Sex м, n (%) | 13 (46,0) | 12 (45,0) | 0,14 |
| Age, years, Me [IQR] | 68 [58-74] | 60[55-63] | 0,81 |
| Body mass index, kg/m2, Me [IQR] | 28 [27-33] | 27 [21-28] | 0,05 |
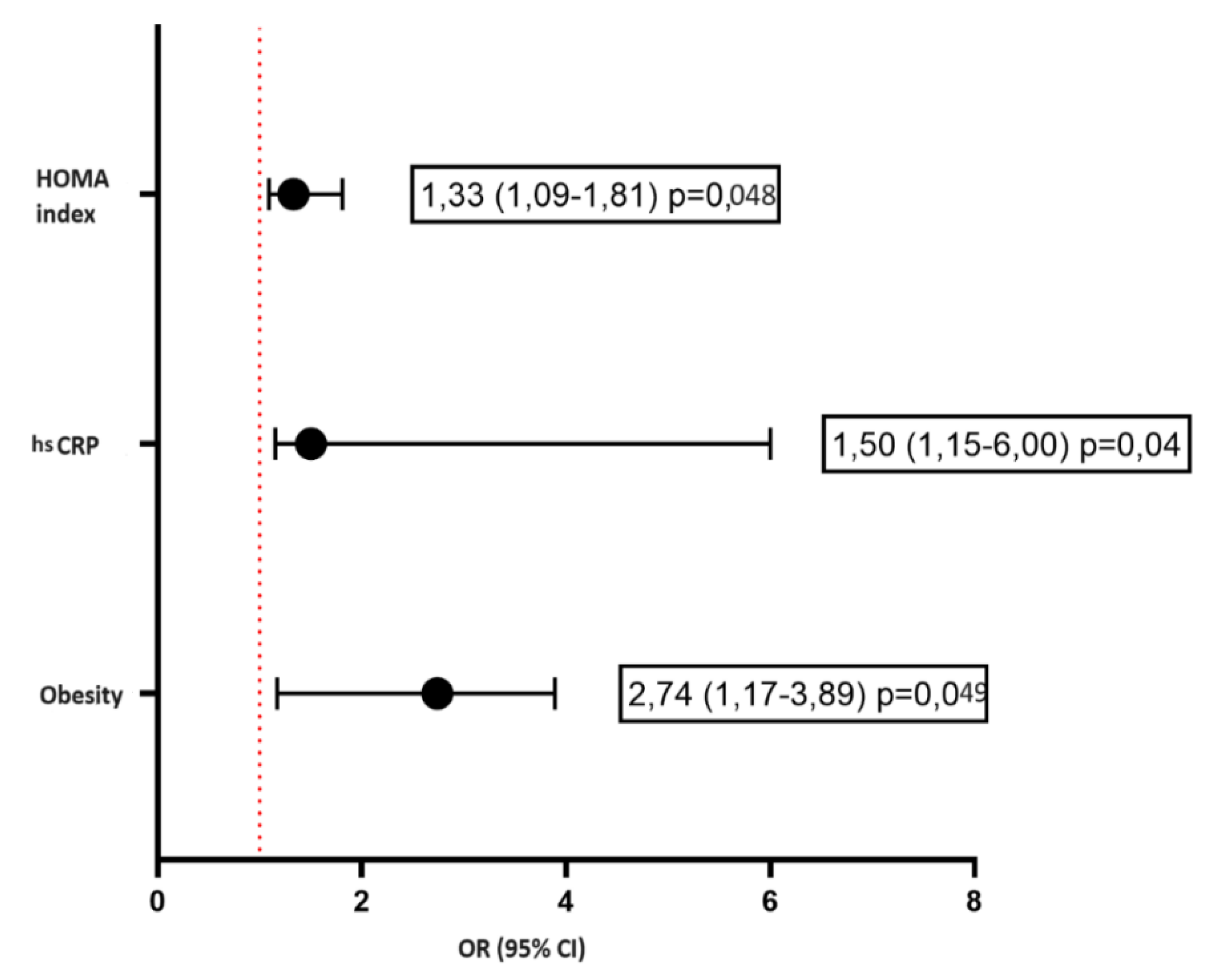
| Obesity 1-degree, n (%) | 13 (44,8) | 3 (11,0) | 0,08 |
| Exercise capacity (METs) | 6,0 ± 1,9 | 6,2 ± 2,0 | 0,59 |
| Current smoker, n (%) | 15 (52,0) | 16 (57,0) | 0,35 |
| Creatinine, µmol/l, Me [IQR] | 87 [70-91] | 74 [68-94] | 0,89 |
| Cholesterol, mmol/l, Me [IQR] | 4,4 [4,1-5,4] | 5,6 [5,0-6,1] | 0,003 |
| Low-density lipoprotein-cholesterol, mmol/l, Me [IQR] | 2,6 [1,6-3,3] | 3,6 [2,9-4,1] | 0,009 |
| High-density lipoprotein-cholesterol, mmol/l, Me [IQR] | 1,3 [1,2-1,4] | 1,5 [1,3-1,9] | 0,025 |
| Triglycerides, mmol/l, Me [IQR] | 1,7 [0,9-2,4] | 1,0 [0,7-1,3] | 0,085 |
| Increase in TG, n (%) | 10 (50,0) | 17 (89,5) | 0,017 |
| Presence of dyslipidemia, n (%) | 25 (68,8) | 16 (73,7) | 0,53 |
| C-reactive protein, ml/l, Me [IQR] | 1,7 [1,1-2,57] | 1,22 [0,66-3,01] | 0,75 |
| Increase in CRP, n (%) | 3 (11,0) | 0 | 0,63 |
| Fibrinogen, g/l, Me [IQR] | 4[3,1-5,8] | 3[2,6-3.9] | 0,12 |
| Increase in fibrinogen, n (%) | 5 (18,0) | 1 (4,0) | 0,034 |
| NT-proBNP, pg/ml, Me [IQR] | 104 [60-121] | 90 [52-116] | 0,8 |
| Increase in NT-proBNP, n (%) | 5 (18,0) | 6 (21,0) | 0,851 |
| Fasting plasma glucose, mmol/l, Me [IQR] | 8,23 [6,75-10,5] | 5,6 [4,7-6,3] | <0,001 |
| Glycated hemoglobin, %, Me [IQR] | 6,5 [6,0-7,2] | 5,3 [5,1-5,7] | <0,001 |
| Insulin, µU/ml, Me [IQR] | 13,6 [8-22,3] | 8,0 [5,7-11,0] | 0,007 |
| C-peptide, ng/ml, Me [IQR] | 2,74 [2,05-3,75] | 1,66 [1,5-2,3] | 0,013 |
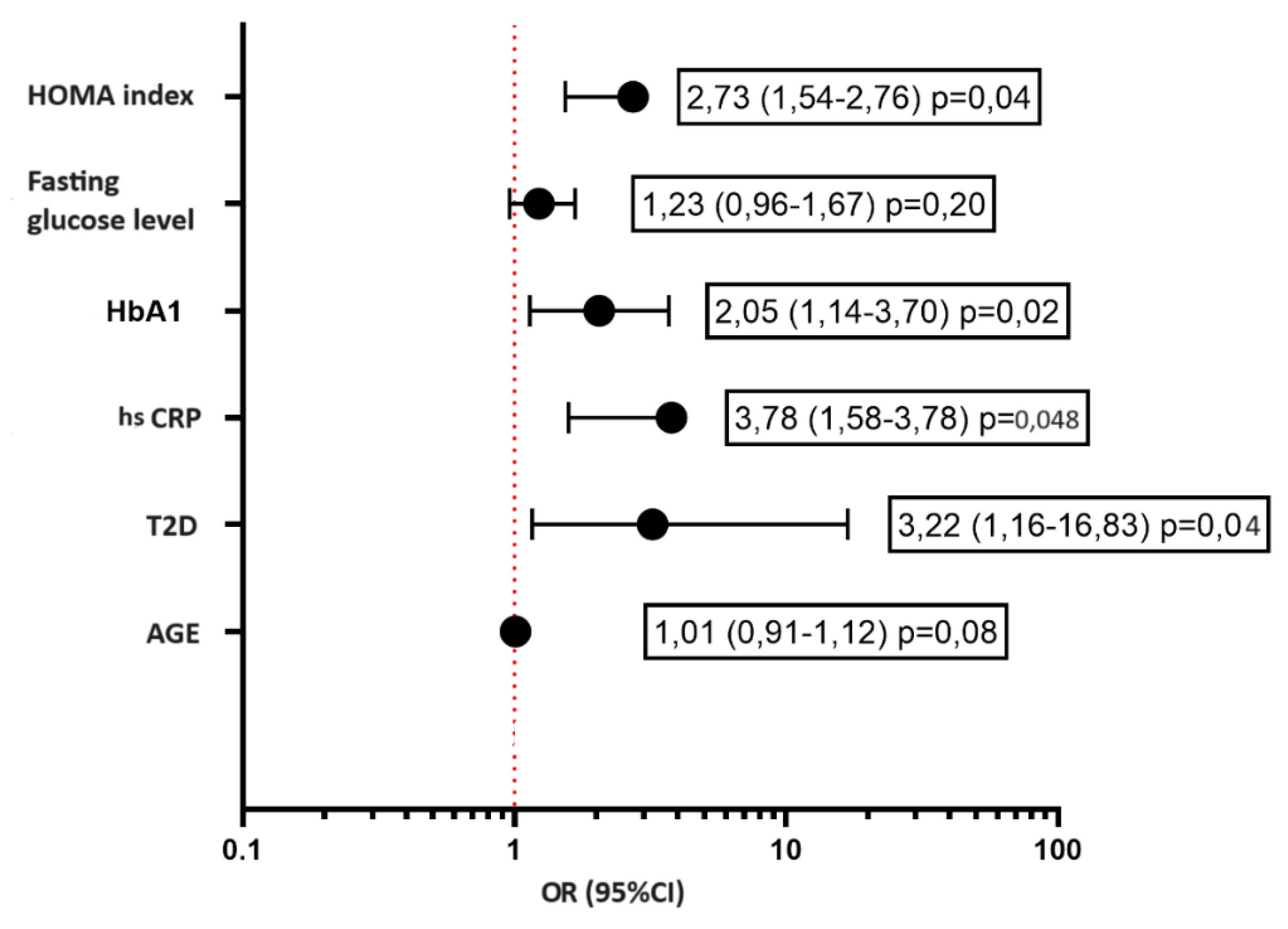
| NOMA Index, Me [IQR] | 6 [3-9] | 2 [1-3] | <0,001 |
| Availability of IR, n (%) | 25 (87,5) | 9 (32,0) | 0,004 |
| Variable | Patients with T2D | p value | Patients without T2D | p value | ||
| Visit 1 | Visit 2 | Visit 1 | Visit 2 | |||
| LVD, n (%) | 27 (53) | 18 (61) | 0,004* | 28 (39) | 9 (32) | 0,48 |
| LV mass index, g/m2 | 95 [84,1-110] | 90,5 [82,0-110,5] | 0,49 | 84,8 [73-98,3] | 88,0 [84,0-100,0] | 0,84 |
| LA volume index, ml/m2 | 27,0 [24,2-31,1] | 26,5 [23,2-33,0] | 0,63 | 26 [23,9-27,8] | 25,0 [21,0-33,0] | 0,66 |
| IVS in diastole, cm | 1,2 [1,1-1,2] | 1,25 [1,15-1,4] | 0,046 | 1,0 [1,0-1,2] | 1,2 [1,0-1,2] | 0,06 |
| Posterior Wall thickness, cm | 1,0 [1,0-1,0] | 1,1 [1,0-1,15] | 0,007 | 0,93 [0,9-1,0] | 0,9 [0,9-1,0] | 0,53 |
| Relative Wall Thickness | 0,44 [0,41-0,48] | 0,49 [0,45-0,53] | 0,06 | 0,43 [0,41-0,46] | 0,45 [0,43-0,49] | 0,58 |
| Сoncentric Hypertrophy, n (%) | 41 (22) | 15 (53) | 0,025 | 26 (12) | 5 (16) | 0,15 |
| Сoncentric Remodeling, n (%) | 28 (15) | 12 (42) | 0,063 | 33 (16) | 6 (20) | 0,65 |
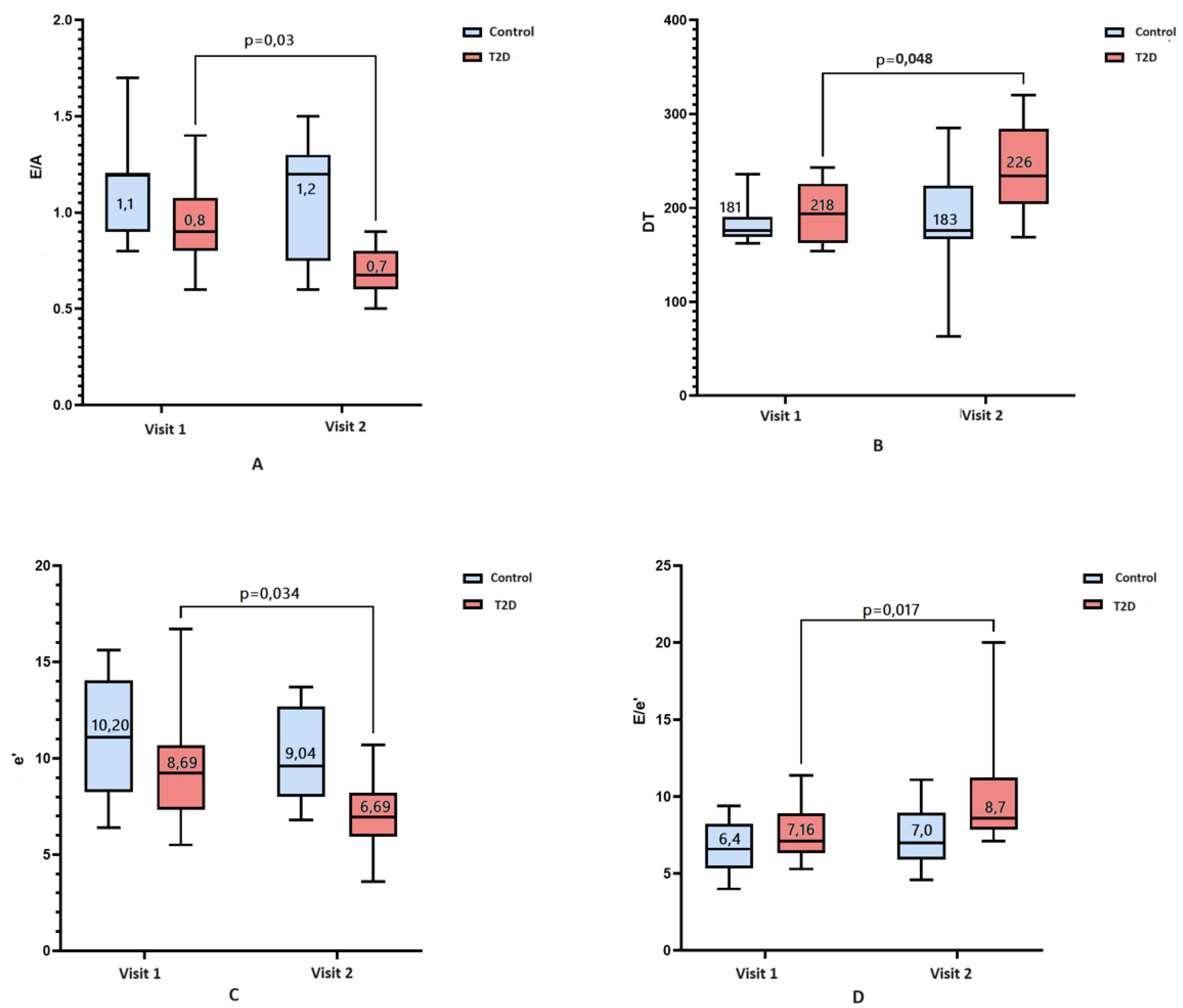
| Transmitral Е/А ratio | 0,8 [0,7-0,9] | 0,7 [0,6-0,9] | 0,03 * | 1,1 [0,9-1,2] | 1,2 [0,7-1,3] | 0,15 |
| Isovolumic relaxation time, ms | 91 [87-96] | 83 [72-99] | 0,49 | 77 [70-88] | 74[67-95] | 0,83 |
| Deceleraton time, ms | 218 [187-232] | 226 [191-277] | 0,05* | 181 [169-203] | 183 [169-229] | 0,53 |
| Average (med/lat) e', cm/s | 8,7 [7,1-9,6] | 6,7 [5,6-8,7] | 0,034 * | 10,2 [8,3-13,6] | 9,0 [8,0-12,6] | 0,059 |
| E/e' | 7,2 [6,4-8,5] | 8,7 [7,9-11,1] | 0,017* | 6,4 [5,4-7,6] | 7,0 [5,6-8,8] | 0,42 |
| Ejection Fraction, % | 60 [59-62] | 65 [58-79] | 0,088 | 64 [61-66] | 67 [60-71] | 0,48 |
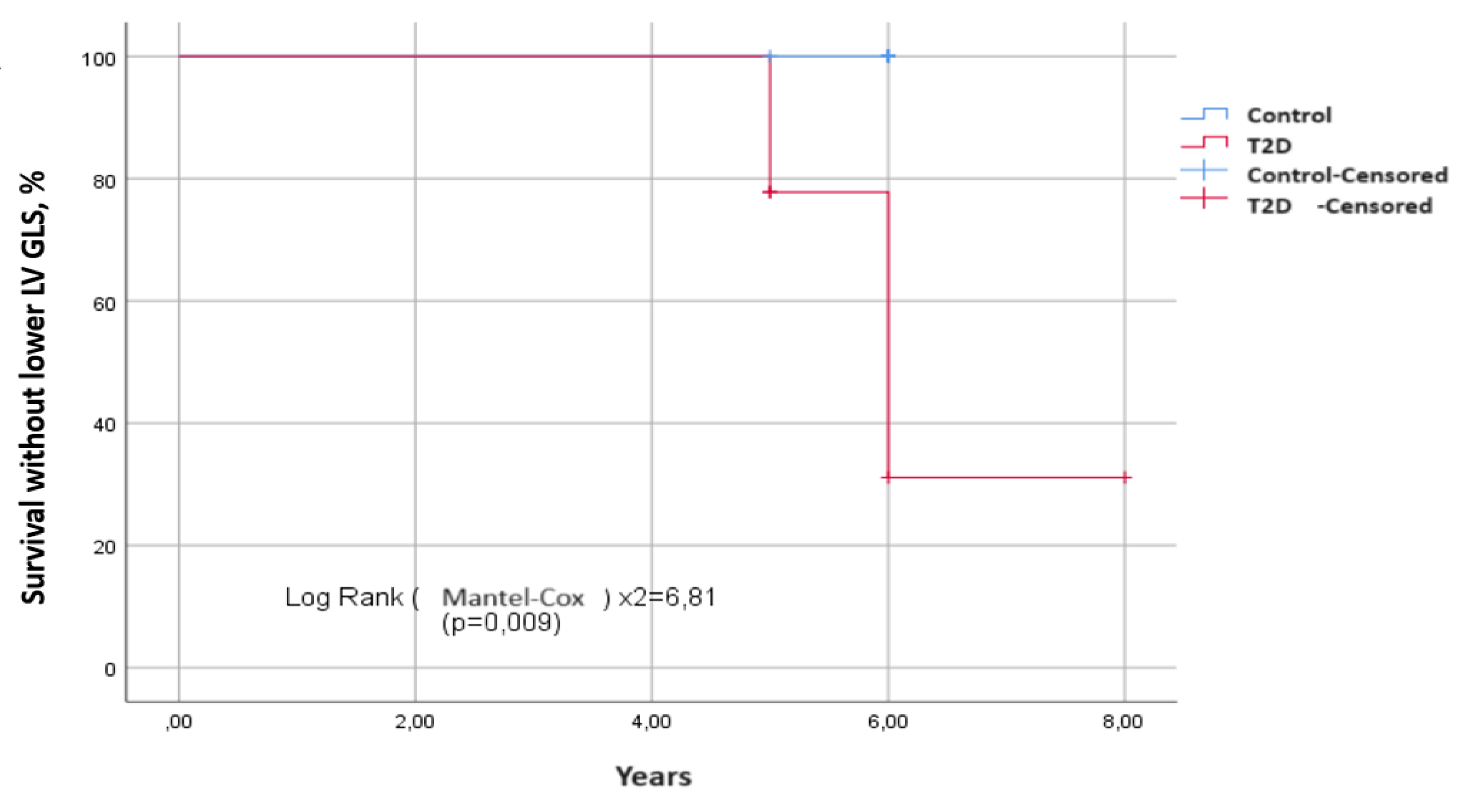
| GLS, % | -17,6 ±1,4 | -17,2 ±1,24 | 0,008* | -19,6 ±1,8 | -19,6 ±1,8 | 0,26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





