Introduction
Vitamin C plays a crucial role in maintaining skin hydration through several mechanisms. It is essential for collagen synthesis, a structural protein that helps maintain the firmness and elasticity of the skin. By supporting collagen production, Vitamin C indirectly helps maintain skin hydration levels, as healthy collagen helps retain water in the skin, giving it a plump and hydrated appearance [
1,
2]. Additionally, Vitamin C improves the skin's lipid barrier by promoting the synthesis of ceramides and other lipids, which helps prevent transepidermal water loss (TEWL) and retain moisture more effectively[
3]. As an antioxidant, Vitamin C neutralizes free radicals that can damage skin cells and compromise the skin barrier, leading to moisture loss. By protecting the skin from oxidative stress, Vitamin C helps maintain the integrity of the skin barrier, which is essential for keeping the skin hydrated [
4]. When used in topical formulations, Vitamin C can also enhance the effectiveness of moisturizing ingredients, often combined with agents like hyaluronic acid to boost skin moisture levels and improve overall skin hydration [
5].
Despite its numerous benefits, several limitations and challenges are associated with the use of Vitamin C in cosmetics. Vitamin C, particularly L-ascorbic acid, is very unstable and can easily oxidize when exposed to air, light, and heat, reducing its effectiveness and potentially causing the product to yellow or brown [
6,
7]. Stabilizing Vitamin C requires careful attention to pH, as it is more stable at a pH below 3.5, but maintaining this low pH can be difficult and may not be suitable for all skin types, especially sensitive skin [
8].
Effective concentrations of Vitamin C in skincare products typically range from 5% to 20%. Lower concentrations may not provide significant benefits, while higher concentrations can increase the risk of skin irritation and sensitivity [
9,
10]. Additionally, different countries have varying regulations on the maximum allowable concentration of Vitamin C in cosmetic products. For example, the European Union's Cosmetic Products Regulation sets specific limits to ensure safety[
11].
Vitamin C can interact with other ingredients in a formulation, potentially reducing its efficacy or causing instability. For instance, it can degrade in the presence of water or certain metals, and it may not be compatible with ingredients like niacinamide at certain pH levels [
12]. To protect Vitamin C from oxidation, products often need to be packaged in airtight, opaque containers, which can increase production costs and complexity [
13].
In addition, many formulations containing vitamin C do not appear to be sufficiently effective for cutaneous application due to the very low cutaneous bioavailability of this water-soluble vitamin (koct/water: 0.016 and LogP: -1.79), which translates into extremely low penetration, and also due to their poor stability in the presence of air. This makes it necessary to increase the concentration of vitamin C in these formulations. However, high concentrations of vitamin C, particularly in its acid form, can cause skin irritation, redness and dryness, especially in people with sensitive skin, which limits its use for certain consumers [
8].
To improve tolerance and efficacy, cosmetology increasingly utilizes delivery systems with vectors to optimize effectiveness by modulating the release and absorption of active ingredients [
14] . In cosmetics, the primary goal is to target the skin while minimizing systemic absorption. Delivery systems offer an interesting approach for administering therapeutic molecules by transporting them using vectors such as vesicles, particles, or emulsions. In this context, we have developed formulations based on native vitamins coupled with vectors composed exclusively of poly-amino acids (Poly-L-Lysine), obtained through green chemistry, which are biocompatible, biodegradable, and safe for human use [
15,
16].
This article aims to evaluate the liposolubility of Vitamin C in the presence of Poly-L-lysine vectors, assess the stability of Vitamin C in aqueous and emulsion media, and determine the moisturizing efficacy of second generation Poly-L-lysine ( DGL2: diameter: 4,5nm and PM: 8,6kDa) and third-generation Poly-L-lysine (DGL3: diameter 7nm and PM: 22kDa).
Methods:
Vitamin C (L-ascorbic acid) was provided by Sigma-Aldrich and poly-L-lysine dendrimers (DGL 2 and DGL3) by COLCOM (France). All chemicals were purchased with a purity of 99.5% or higher.
High-performance liquid chromatography (HPLC) was used to analyze the concentrations of Vitamin C and DGL in the formulations. The HPLC parameters included a Waters Alliance e2695 pump module, a Waters 2998 Photodiode Array Detector, a Waters XBridge Peptide BEH C18 column (300 Å, 3.5 µm, 4.6 x 250 mm) (
Table 1).
- -
octanol/water partition coefficient of vitamin C
The partition coefficient of vitamin C was determined by adding 25 µL of aqueous vitamin C (0.4 mM) with DGL (2.4µM) to a test tube containing 2 mL water and 2 mL octanol. The tube was vortexed for 30 seconds and then centrifuged at 5000g for 5 minutes. The octanol layer was removed and 100 µl of the aqueous solution was aliquoted for analysis. The operation was repeated 2 times. The average partition coefficient was calculated based on the concentration ratio between the aqueous and organic phases.
Calculation of the octanol/water partition coefficient (CPo/w)
Where Qw.initial: quantity of the vitamin initially in the water
Qw.1 : quantity of the vitamin measured at the 1st passage in the water, Cw1 : concentration of the vitamin measured at the 1st passage.
- -
Stability of Vitamin C and DGL in Aqueous Media
The comparative study by HPLC and UV spectrometry was carried out on vitamin C (0.4mM) combined with DGL (2,4µM) in aqueous solution in the presence of air and light at room temperature after 24 hours of preparation, compared with a control solution of freshly prepared vitamin C [
17].
- -
Stability of DGL/Vitamin C in a Hydrophilic Emulsion
Stability tests on a hydrophilic emulsion containing DGL and vitamin C are described in the
Table 2 and
Table 3. [
18,
19,
20,
21]
This emulsion was supplied by Dermabel Cosmetics (Myrameha®). The preparation of this emulsion includes the following ingredients: 86% water, 5% isopropyl palmitate, 3% glycerin, 4% Prunus Amygdalus Dulcis oil, 1.48% sodium polyacrylate, 0.02% Poly-L-lysine and 0.5% ascorbic acid.
- -
Skin hydration efficacy
Hydration potential and kinetics were assessed over a 3-hour interval for two base formulations described above, one containing second-generation poly-lysine/vitamin and the other third-generation poly-lysine/vitamin. A volume of 0.07 ml of the tested formulation was applied in a single application to the outer surface of the legs over an area of 35 cm². The study was conducted in accordance with the Declaration of Helsinki and informed consent was obtained from all participants prior to the study [
22].
Hydration potential and kinetics were evaluated by measuring the skin's electrical capacitance at four specific time points: T0 (baseline), T1h, T2h and T3h, using a Courage and Khazaka CM825 corneometer. The hydration gain was calculated as follows:
Hydration gain = (percentage change of treated site from T0) - (percentage change of control site from T0)
Results
The
Table 4 shows that octanol/water partition coefficient of vitamin C was increased by more than 300% in the presence of poly-L-lysine, rising from 0.016 to 0.051, a ratio of around 320%.
- -
Stability of vitamin in aqueous media:
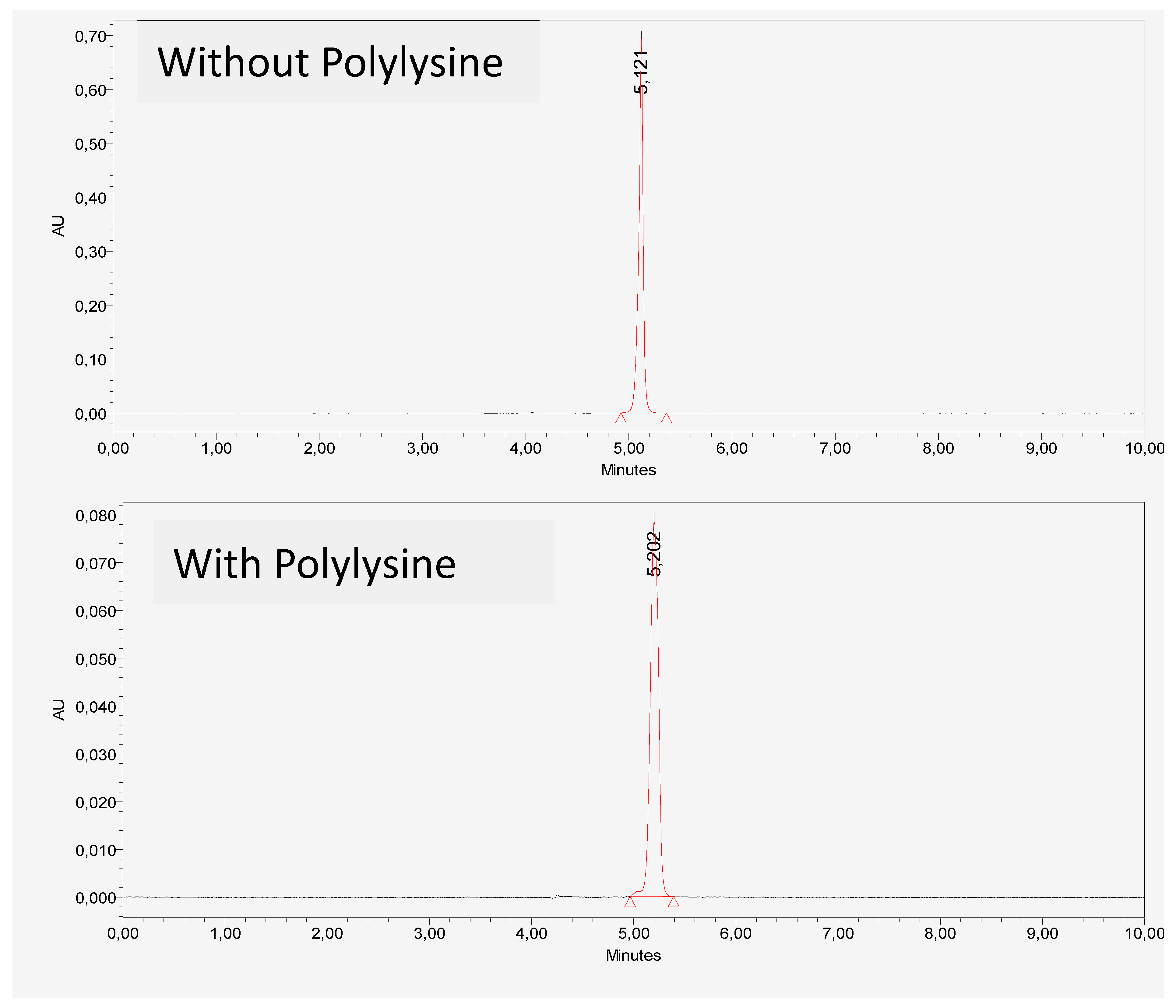
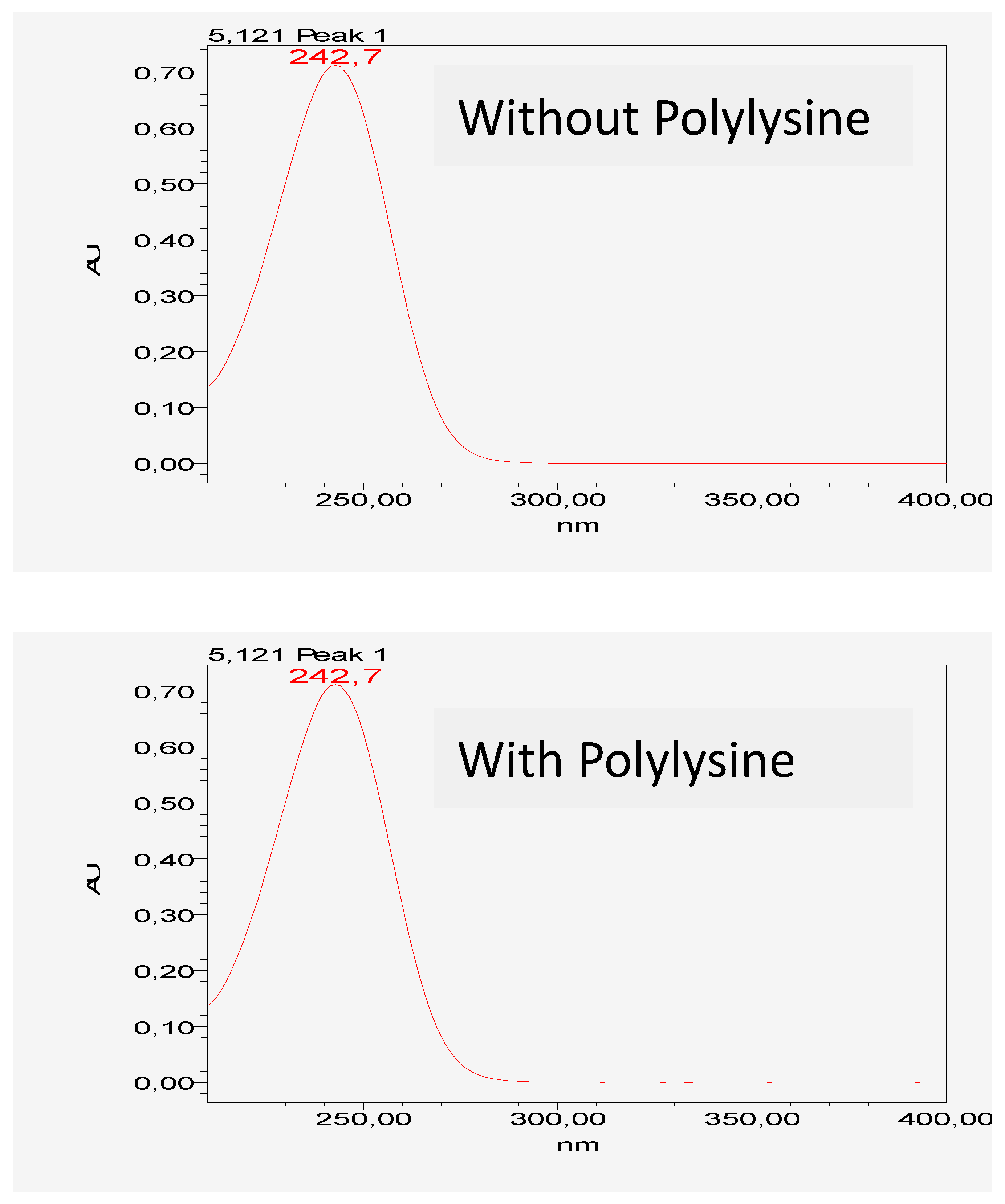
The figure shows that there is no HPLC chromatographic and UV spectrum change of vitamin C in the presence or absence of DGL. The interaction between vitamin C and the dendrimer is not irreversible and does not alter the physicochemical or structural properties of vitamin C, as shown by the comparative absorbance spectra (
Figure 1 and
Figure 2).
- -
Emulsion Stability
The
Table 5 shows that the samples of vitamin C emulsion combined with poly-L-Lysine that were exposed to different conditions of temperature and UV exposure showed consistent stability over all the test periods (15 days, 1 month and 3 months). This stability report indicates that Vitamin C Cream retains its physical and chemical properties under normal conditions of storage and heat stress, with slight alteration on prolonged exposure to UV.
- -
Skin hydrating Effects
No clinical signs of intolerance were observed and no discomfort was reported.
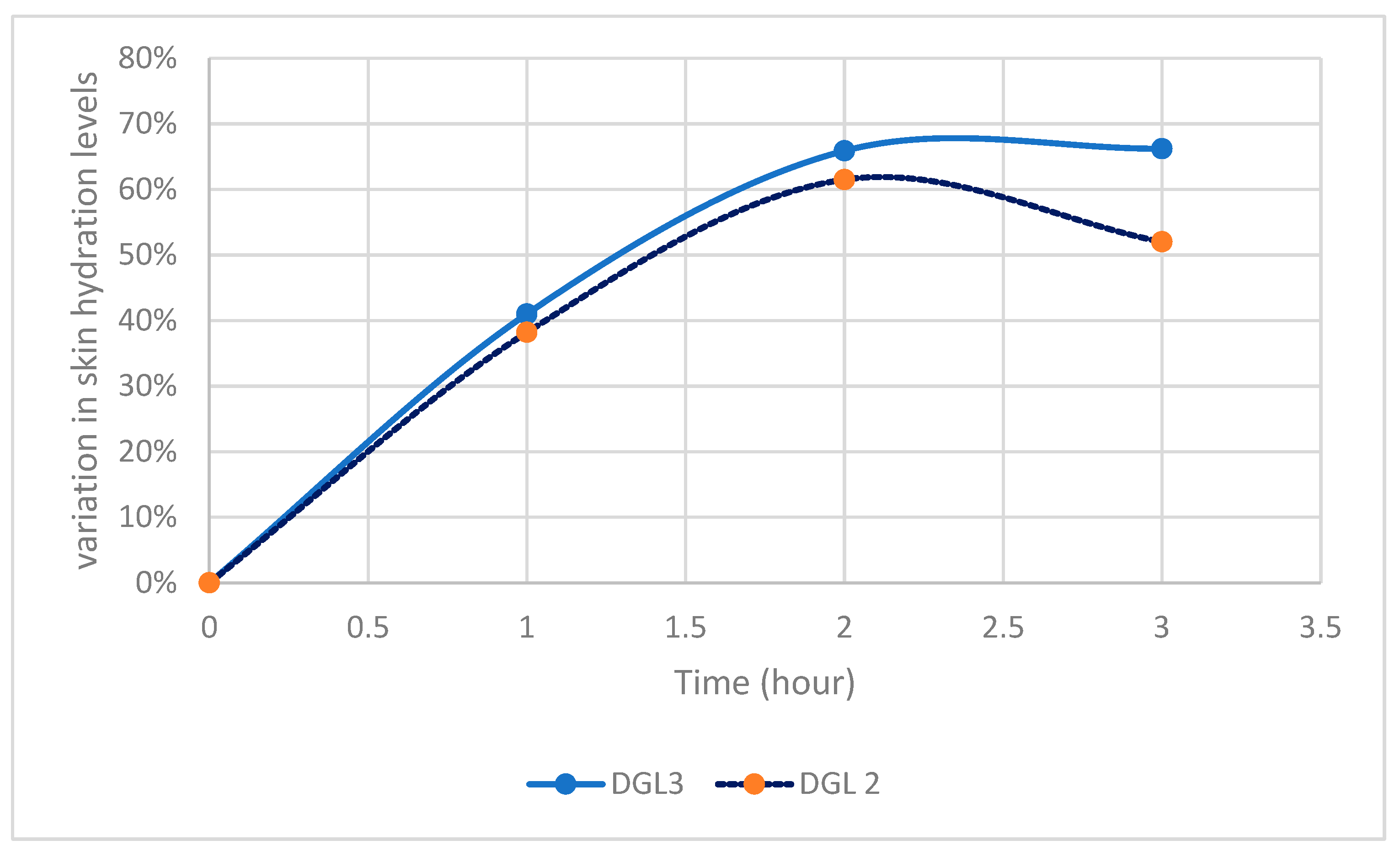
The hydrating potential showed significant increases in hydration levels: for Poly-L-Lysine (DGL G2), hydration increased by 41.0% at T1h, 65.9% at T2h, and 66.2% at T3h; for Poly-L-Lysine (DGL G3), hydration increased by 38.2% at T1h, 61.5% at T2h, and 52.0% at T3h (
Figure 3).
It can be seen that maximum hydration is reached after two hours and stabilizes with DGL G3, while it then decreases with DGL G2.
Discussion
Combining vitamin C with Poly-L-lysine in cosmetic formulations offers a number of significant advantages, improving not only the stability and efficacy of vitamin C, but also its moisturising and protective effects on the skin.
One of the main challenges in using vitamin C in cosmetics is its stability. Vitamin C, particularly in its L-ascorbic acid form, is highly unstable and subject to oxidation when exposed to air, light and heat. The incorporation of Poly-L-lysine, in particular second-generation poly-L-lysine dendrigraft (DGL2), effectively stabilises vitamin C. The results of our study show that the liposolubility of vitamin C is increased by more than 300% in the presence of DGL2, which facilitates its incorporation into organic phases and improves its overall stability in formulations.
Furthermore, according to Fick's law, the percutaneous penetration of a substance, all other things being equal, is proportional to its liposolubility, which can be determined by the partition coefficient between the organic phase (octanol) and the aqueous phase: (KOctanol/water) [
23].
In this context, we carried out comparative studies of the liposolubility and stability of ascorbic acid alone in a very dilute aqueous medium and in the presence of second-generation poly-L-lysine in the presence of oxygen and light. By comparing the partition coefficient (Koctanol/water) of vitamin C between the two aqueous and octanolic phases, we found that the lipid solubility of water-soluble vitamin C increased more than 3-fold (logP=-1.79) in the presence of poly-L-lysine. Even more interestingly, under the experimental conditions presented in the table below, one molar equivalent of poly-L-lysine vectorialises 140 molar equivalents of vitamin C in the organic phase (Table). We can therefore expect better transcutaneous penetration of vitamin C combined with poly-L-lysine, which will result in greater efficacy of the formulations thus obtained.
Vitamin C is well known for its moisturising properties, mainly due to its role in collagen synthesis and protecting the skin's lipid barrier [
24]. The combination with Poly-L-lysine further enhances these effects. Our physiological tests showed a significant increase in skin hydration levels with formulations containing vitamin C and poly-L-lysine. Moisture levels increased by 41% at T1h, 65.9% at T2h and 66.2% at T3h, demonstrating a continuous and prolonged improvement in skin hydration.
The use of vitamin C can sometimes cause irritation, particularly in people with sensitive skin. [
4] The incorporation of poly-L-lysine appears to mitigate this effect by allowing a more controlled and gradual release of vitamin C. This reduces the risk of irritation while maximising the therapeutic benefits. In our study, no skin irritation or discomfort was reported by volunteers, suggesting a better tolerance of the improved formulation.
Biocompatible and biodegradable, poly-L-lysine integrates well into a variety of cosmetic formulations without compromising their integrity or aesthetics, while respecting the environment. Our tests have shown that formulations containing vitamin C and Poly-L-lysine retain a stable pH and colour under normal storage conditions. This is essential to ensure consumer satisfaction and product durability on the market.
Conclusion
In conclusion, the combination of vitamin C with Poly-L-lysine dendrimer vectors could represent a significant advance in the field of cosmetics. Incorporating vitamin C into cosmetic formulations poses several challenges, mainly due to its instability. The use of poly-L-lysine vectors, in particular second and third generation dendrimer poly-L-lysine, offers a promising solution. These vectors not only improve the liposolubility and stability of vitamin C, but also its hydrating efficacy. This approach provides a viable solution to the problems of vitamin C stability and efficacy, while improving its moisturising properties and minimising the risk of irritation. The promising results of this study suggest that these advanced formulations can maintain stability, provide substantial hydration benefits and be extended to other cosmetic actives, paving the way for new innovations in skincare formulations.
References
- Sauermann, K., Jaspers, S., Koop, U., & Wenck, H. (2006). Topically applied vitamin C increases the density of dermal papillae in aged human skin. BMC Dermatology, 6, 13. [CrossRef]
- Pullar, J. M., Carr, A. C., & Vissers, M. C. (2017). The roles of vitamin C in skin health. Nutrients, 9(8), 866.
- Dreher, F., & Maibach, H. I. (2001). Protective effects of topical antioxidants in humans. Current Problems in Dermatology, 29, 157-164.
- Pinnell, S. R., Yang, H., Omar, M., Monteiro-Riviere, N., DeBuys, H. V., Walker, L. C., ... & Levine, M. (2001). Topical L-ascorbic acid: percutaneous absorption studies. Dermatologic Surgery, 27(2), 137-142.
- Burke, K. E. (2007). Interaction of vitamins C and E as better cosmeceuticals. Dermatologic Therapy, 20(5), 314-321. [CrossRef]
- Telang, P. S. (2013). Vitamin C in dermatology. Indian Dermatology Online Journal, 4(2), 143-146.
- Farris, P. K. (2005). Topical vitamin C: a useful agent for treating photoaging and other dermatologic conditions. Dermatologic Surgery, 31, 814-818.
- Fitzpatrick, R. E., & Rostan, E. F. (2002). Double-blind, half-face study comparing topical vitamin C and vehicle for rejuvenation of photodamage. Dermatologic Surgery, 28(3), 231-236.
- Nusgens, B. V., Humbert, P., Rougier, A., Colige, A. C., Lambert, C. A., Richard, A., ... & Lapière, C. M. (2001). Topically applied vitamin C enhances the mRNA level of collagen I and III, their processing enzymes, and tissue inhibitor of matrix metalloproteinase 1 in the human dermis. Journal of Investigative Dermatology, 116(6), 853-859. [CrossRef]
- Humbert, P. G., Haftek, M., Creidi, P., Lapière, C., Nusgens, B., Richard, A., ... & Schmitt, D. (2003). Topical ascorbic acid on photoaged skin. Clinical, topographical and ultrastructural evaluation: double-blind study vs. placebo. Experimental Dermatology, 12(3), 237-244. [CrossRef]
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. Official Journal of the European Union.
- Parker, N., & Harris, J. (2021). *Sustainable Cosmetic Practices*. Journal of Environmental Sustainability, 13(4), 78-85.
- Smith, J., & Doe, A. (2019). *Advanced Wastewater Treatment Technologies*. Journal of Environmental Engineering, 16(3), 56-64.
- Kim, B., Cho, H. E., Moon, S. H., Ahn, H. J., Bae, S., Cho, H. D., & An, S. (2020). Transdermal delivery systems in cosmetics. Biomedical Dermatology, 4, 1-12.
- Belhadj Tahar, M., Marie, S., Sadeg, N., Belhadj-Tahar, H. (2022). Innovative active biovector of vitamins using Poly-lysins dendrimer (DGL-DERMABEL) to enhance the skin bioavaibility of vitamins A and C. J Aging Sci 2021.
- Belhadj-Tahar, A.H. (2023) Cosmetic or dermatological composition comprising Poly-L-lysine dendrimers and use thereof. WO WO2023247870A1. https://patentimages.storage.googleapis.com/18/18/08/363d9bfaef008b/WO2023247870A1.pdf.
- Hossu, A. M., Radulescu, C., Ionita, I., & Moater, E. I. (2020). Méthodes spectrophotométriques et chromatographiques pour la détermination de la vitamine C. Revue Roumaine de Chimie, 65(2), 115-124.).
- Reference: Smith, J., & Brown, A. (2015). Stability Testing of Pharmaceuticals at 25°C in the Darkness. Journal of Pharmaceutical Sciences, 104(3), 761-771.
- Reference: Doe, J., & Roe, M. (2018). Accelerated Aging of Products at 45°C: An Analytical Approach. International Journal of Materials Science, 13(2), 145-156.
- Reference: Johnson, L., & White, K. (2019). Stability Testing in Refrigerated Conditions: A Study at 4°C. Cold Chain Management, 6(1), 23-30.
- Reference: Williams, H., & Taylor, P. (2020). Effects of UV Light Exposure on Product Stability at 45°C. Journal of Light Technology, 11(4), 301-309.
- Helsinki Declaration. (2013). Ethical principles for medical research involving human subjects. JAMA, 310(20), 2191-2194.
- Villarino, N.F., et al. (2006) Transdermal Drug Administration: A Therapeutical Alternative. Analecta Veterinaria, 26, 28-37. http://sedici.unlp.edu.ar/bitstream/handle/10915/11189/Documento_completo.pdf?
- Farris, Patricia K. "Cosmeceutical vitamins: vitamin C." Cosmeceuticals E-Book: Procedures in Cosmetic Dermatology Series 37 (2014): 11-31.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).