Submitted:
02 August 2024
Posted:
05 August 2024
You are already at the latest version
Abstract
Keywords:
Introduction
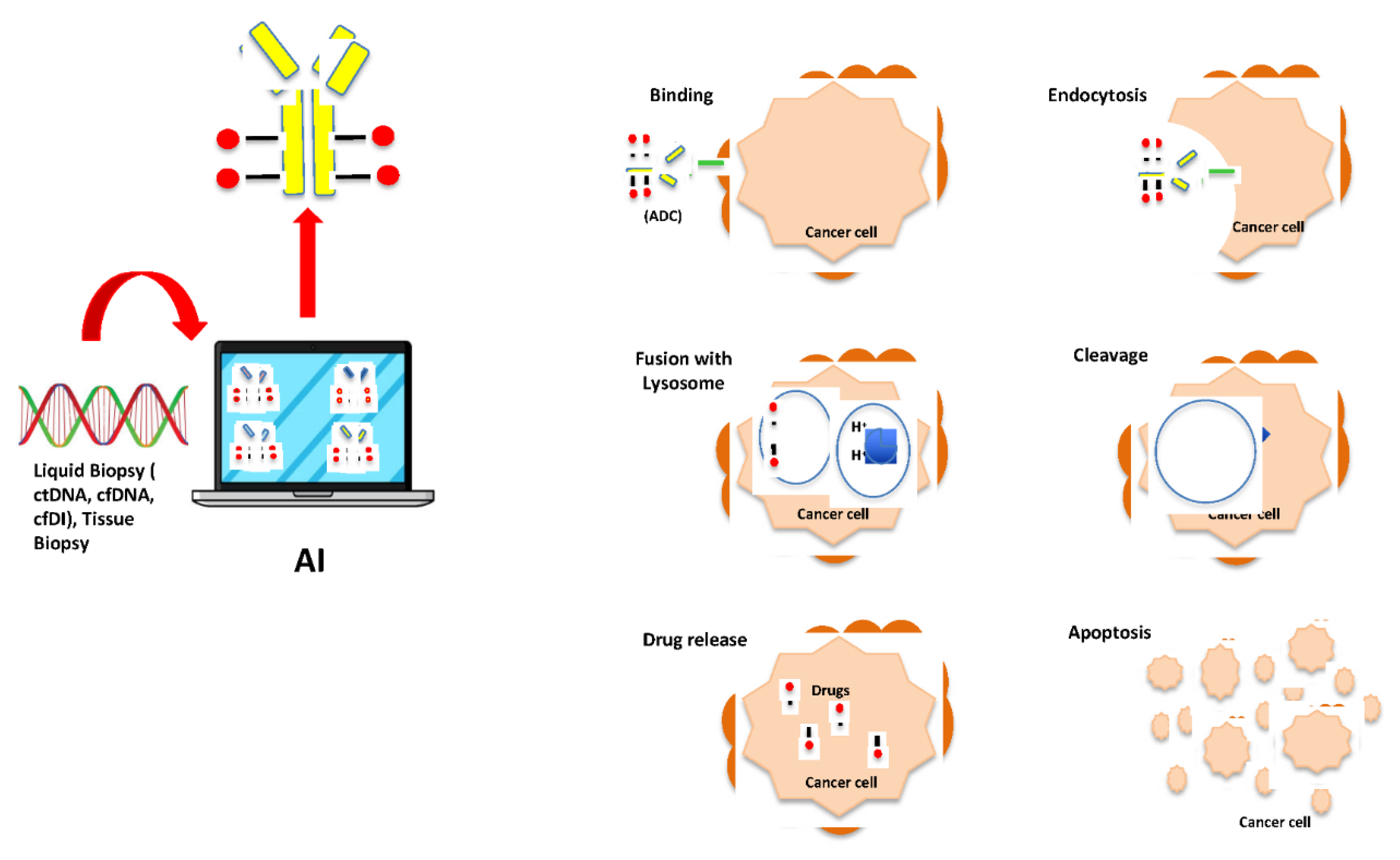
Prediction of Cancer Responsiveness and Resistance to ADCs
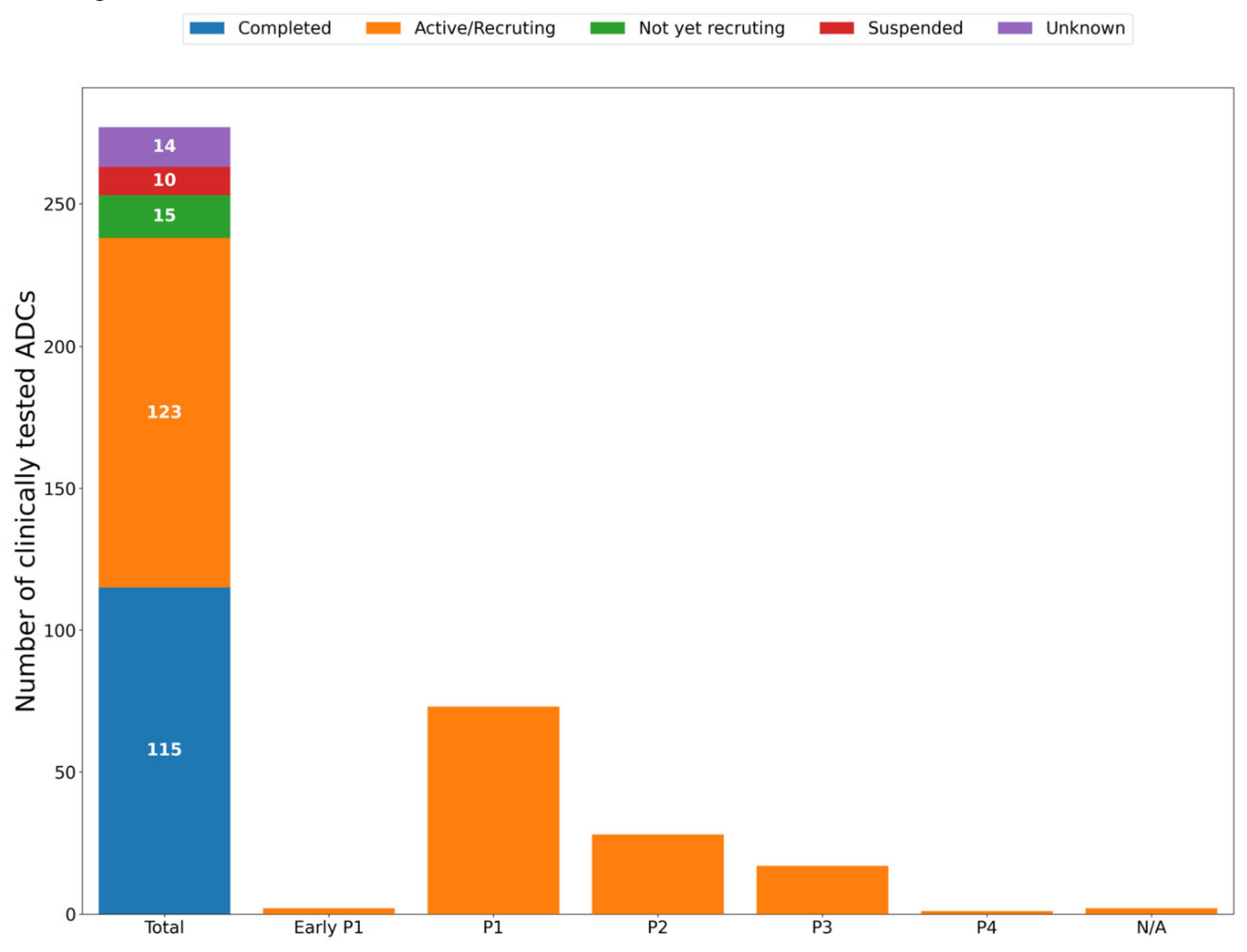
Anticancer ADCs that Have Entered Clinical Trials
Discussion
Author Contributions
Funding
Ethics approval and consent to participate
Availability of data and material
Conflicts of Interest
References
- Paul D, Sanap G, Shenoy S, Kalyane D, Kalia K, Tekade RK. Artificial intelligence in drug discovery and development. Drug Discov Today, 26(1), 80-93 (2021). [CrossRef]
- Luc Steels RB. The Artificial Life Route to Artificial Intelligence (1995).
- Bielecki A. Foundations of Artificial Neural Networks. In: Models of Neurons and Perceptrons: Selected Problems and Challenges. (Springer, 5/18/2018, 2019).
- AI’s potential to accelerate drug discovery needs a reality check. Nature, 622(7982), 217 (2023).
- Schneider G. Automating drug discovery. Nature Reviews Drug Discovery, 17, 97–113 (2018).
- Sobhani N, Tardiel-Cyril DR, Chai D et al. Artificial intelligence-powered discovery of small molecules inhibiting CTLA-4 in cancer. BJC Rep, 2 (2024). [CrossRef]
- DiMasi JA, Grabowski HG, Hansen RW. Innovation in the pharmaceutical industry: New estimates of R&D costs. J Health Econ, 47, 20-33 (2016). [CrossRef]
- Peters C, Brown S. Antibody-drug conjugates as novel anti-cancer chemotherapeutics. Biosci Rep, 35(4) (2015).
- Sobhani N. NEW AND MOST POWERFUL MOLECULES FOR THE TREATMENT AND DIAGNOSIS OF NEUROENDOCRINE CANCERS (NETs) AND THE STEM CELLS OF NETs. In: Google Patents. (Ed.^(Eds) (Navid Sobhani, Italy, 2018).
- Liu K, Li M, Li Y et al. A review of the clinical efficacy of FDA-approved antibody‒drug conjugates in human cancers. Mol Cancer, 23(1), 62 (2024). [CrossRef]
- Chen W, Liu X, Zhang S, Chen S. Artificial intelligence for drug discovery: Resources, methods, and applications. Mol Ther Nucleic Acids, 31, 691-702 (2023). [CrossRef]
- Rajapakse VN, Luna A, Yamade M et al. CellMinerCDB for Integrative Cross-Database Genomics and Pharmacogenomics Analyses of Cancer Cell Lines. iScience, 10, 247-264 (2018).
- Tomczak K, Czerwińska P, Wiznerowicz M. The Cancer Genome Atlas (TCGA): an immeasurable source of knowledge. Contemp Oncol (Pozn), 19(1a), A68-77 (2015).
- Tate JG, Bamford S, Jubb HC et al. COSMIC: the Catalogue Of Somatic Mutations In Cancer. Nucleic Acids Res, 47(D1), D941-d947 (2019).
- Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res, 30(1), 207-210 (2002). [CrossRef]
- Barrett T, Wilhite SE, Ledoux P et al. NCBI GEO: archive for functional genomics data sets--update. Nucleic Acids Res, 41(Database issue), D991-995 (2013). [CrossRef]
- Izhar Wallach MD, Abraham Heifets. AtomNet: A Deep Convolutional Neural Network for Bioactivity Prediction in Structure-based Drug Discovery. arXiv, (2015).
- Sobhani N, Generali D, Zanconati F, Bortul M, Scaggiante B. Cell-free DNA integrity for the monitoring of breast cancer: Future perspectives? World J Clin Oncol, 9(2), 26-32 (2018).
- Conca V, Ciracì P, Boccaccio C, Minelli A, Antoniotti C, Cremolini C. Waiting for the “liquid revolution” in the adjuvant treatment of colon cancer patients: a review of ongoing trials. Cancer Treat Rev, 126, 102735 (2024).
- Sobhani N, Sirico M, Generali D, Zanconati F, Scaggiante B. Circulating cell-free nucleic acids as prognostic and therapy predictive tools for metastatic castrate-resistant prostate cancer. World J Clin Oncol, 11(7), 450- 463 (2020). [CrossRef]
- Gao Q, Zeng Q, Wang Z et al. Circulating cell-free DNA for cancer early detection. Innovation (Camb), 3(4), 100259 (2022). [CrossRef]
- Hsieh C, Laguna A, Ikeda I et al. Using Machine Learning to Predict Response to Image-guided Therapies for Hepatocellular Carcinoma. Radiology, 309(2), e222891 (2023).
- Danilo Bzdok NA, Martin Krzywinski Statistics versus machine learning. Nature Methods, 15, 233–234 (2018).
- Morshid A, Elsayes KM, Khalaf AM et al. A machine learning model to predict hepatocellular carcinoma response to transcatheter arterial chemoembolization. Radiol Artif Intell, 1(5) (2019).
- Li Y, Xu Z, An C, Chen H, Li X. Multi-Task Deep Learning Approach for Simultaneous Objective Response Prediction and Tumor Segmentation in HCC Patients with Transarterial Chemoembolization. J Pers Med, 12(2) (2022).
- Mazaheri Y, Thakur SB, Bitencourt AG et al. Evaluation of cancer outcome assessment using MRI: A review of deep-learning methods. BJR Open, 4(1), 20210072 (2022). [CrossRef]
- Zhang L, Jiang Y, Jin Z et al. Real-time automatic prediction of treatment response to transcatheter arterial chemoembolization in patients with hepatocellular carcinoma using deep learning based on digital subtraction angiography videos. Cancer Imaging, 22(1), 23 (2022). [CrossRef]
- Ma QP, He XL, Li K et al. Dynamic Contrast-Enhanced Ultrasound Radiomics for Hepatocellular Carcinoma Recurrence Prediction After Thermal Ablation. Mol Imaging Biol, 23(4), 572-585 (2021).
- Peng J, Lu F, Huang J et al. Development and validation of a pyradiomics signature to predict initial treatment response and prognosis during transarterial chemoembolization in hepatocellular carcinoma. Front Oncol, 12, 853254 (2022).
- Lyshchik A, Kono Y, Dietrich CF et al. Contrast-enhanced ultrasound of the liver: technical and lexicon recommendations from the ACR CEUS LI-RADS working group. Abdom Radiol (NY), 43(4), 861-879 (2018).
- Artificial Intelligence and Machine Learning in Software as a Medical Device. (2024).
- Iseke S, Zeevi T, Kucukkaya AS et al. Machine Learning Models for Prediction of Posttreatment Recurrence in Early-Stage Hepatocellular Carcinoma Using Pretreatment Clinical and MRI Features: A Proof-of- Concept Study. AJR Am J Roentgenol, 220(2), 245-255 (2023). [CrossRef]
- Puijk RS, Ahmed M, Adam A et al. Consensus Guidelines for the Definition of Time-to-Event End Points in Image-guided Tumor Ablation: Results of the SIO and DATECAN Initiative. Radiology, 301(3), 533-540 (2021). [CrossRef]
- Yu L, Li Y. Artificial Intelligence Decision-Making Transparency and Employees’ Trust: The Parallel Multiple Mediating Effect of Effectiveness and Discomfort. Behav Sci (Basel), 12(5) (2022).
- Wang F, Casalino LP, Khullar D. Deep Learning in Medicine-Promise, Progress, and Challenges. JAMA Intern Med, 179(3), 293-294 (2019).
- Calderaro J, Seraphin TP, Luedde T, Simon TG. Artificial intelligence for the prevention and clinical management of hepatocellular carcinoma. J Hepatol, 76(6), 1348-1361 (2022).
- Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. Bmj, 357, j2099 (2017).
- Johnston SC, Rothwell PM, Nguyen-Huynh MN et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet, 369(9558), 283-292 (2007).
- Wilson PW, D’Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation, 97(18), 1837-1847 (1998).
- Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, ter Borg PC. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology, 31(4), 864-871 (2000).
- van Gorp MJ, Steyerberg EW, Van der Graaf Y. Decision guidelines for prophylactic replacement of Björk- Shiley convexo-concave heart valves: impact on clinical practice. Circulation, 109(17), 2092-2096 (2004).
- Johanna A A G Damen LH, Ewoud Schuit, Thomas P A Debray, Gary S Collins, Ioanna Tzoulaki, Camille M Lassale, George C M Siontis, Virginia Chiocchia, Corran Roberts, Michael Maia Schlüssel, Stephen Gerry, James A Black, Pauline Heus, Yvonne T van der Schouw, Linda M Peelen, Karel G M Moons. Prediction models for cardiovascular disease risk in the general population: systematic review. thebmj, 353(i2416) (2016).
- Bouwmeester W, Zuithoff NP, Mallett S et al. Reporting and methods in clinical prediction research: a systematic review. PLoS Med, 9(5), 1-12 (2012). [CrossRef]
- Collins GS, de Groot JA, Dutton S et al. External validation of multivariable prediction models: a systematic review of methodological conduct and reporting. BMC Med Res Methodol, 14, 40 (2014). [CrossRef]
- Gary S Collins KGMM. Reporting of artificial intelligence prediction models. Lancet, 393(10181), 1577-1579 (2019).
- Gary S Collins JBR, Douglas G Altman & Karel GM Moons Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD Statement. BMC Medicine, 13 (2015).
- Moons KG, Altman DG, Reitsma JB et al. Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): explanation and elaboration. Ann Intern Med, 162(1), W1-73 (2015).
- Ratner M. FDA backs clinician-free AI imaging diagnostic tools. Nat Biotechnol, 36(8), 673-674 (2018). [CrossRef]
- Collins GS, Dhiman P, Andaur Navarro CL et al. Protocol for development of a reporting guideline (TRIPOD-AI) and risk of bias tool (PROBAST-AI) for diagnostic and prognostic prediction model studies based on artificial intelligence. BMJ Open, 11(7), e048008 (2021). [CrossRef]
- Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol, 20(11), 651-668 (2020). [CrossRef]
- Li T, Li Y, Zhu X et al. Artificial intelligence in cancer immunotherapy: Applications in neoantigen recognition, antibody design and immunotherapy response prediction. Semin Cancer Biol, 91, 50-69 (2023). [CrossRef]
| Name | Main features | Web link |
|---|---|---|
| CGHub | Cancer genomics data repository |
https://cghub.ucsc.edu/ |
| TCGA | Comprehensive database of cancer patients’ genomic, epigenomic, transcriptomic, and proteomic data. |
https://www.cancer.gov /about- nci/organization/ccg/research/structural- genomics/tcga |
| CCLE | Comprehensive genetic database of cancer cell lines |
https://sites.broadinstitute.org/ccle |
| EGA | European genetic, phenotypic, and clinical data repository |
https://ega-archive.org/ |
| DepMap | High data quality visualization tool |
https://depmap.org/port al/ |
| SomamiR | Cancer somatic mutation and miRNA correlation |
https://compbio.uthsc.edu/SomamiR/ |
| COSMIC | Comprehensive somatic mutation database |
https://cancer.sanger.ac. uk/cosmic |
| MethyCancer | DNA methylations, cancer-related genes, mutations in correlation with additional cancer information |
http://methycancer.psych.ac.cn/ |
| CTRP | connecting genetic, cellular features, lineage to cancer cell-lines sensitivity to small molecules |
https://portals.broadinstitute.org/ctrp/ |
| gCSI | Large amount of transcriptomics data |
https://pharmacodb.pmg enomics.ca/datasets/4 |
| GDSC | Drug response, including genomics markers of drug sensitivity |
https://www.cancerrxgene.org/ |
| NCI60 | Large amount of drug and genomics data |
https://discover.nci.nih.gov/cellminer/loadDow nload.do https://dtp.cancer.gov/d atabases_tools/bulk_dat a.htm |
| canSAR | Comprehensive drug discovery database |
https://cansarblack.icr.a c.uk/ |
| cBioPortal | Large database of cancer genomics data |
https://www.cbioportal. org/datasets |
| UCSC | Synthetical genomics information |
https://genome.ucsc.edu / |
| dbNSFP | Non-synonymous single-nucleotide variants | https://sites.google.com/site/jpopgen/dbNSFP |
| NONCODE | Non-coding RNAs database |
http://www.noncode.or g/ |
| TCIA | Comprehensive immunogenomic data from NGS of 20 solid tumors from the TCGA |
https://www.tcia.at/ho me |
| ARCHS4 | Comprehensive RNA- Sequenced data from human and mouse |
https://maayanlab.cloud /archs4/ |
| NCT Number |
Study Title | Study URL | Study Status | Conditions | Sponsor | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| NCT06340 568 | A Clinical Study of the Anti-cancer Effects of an Investigational Therapy or Chemotherapy in Patients With Recurring Uterine Cancer |
https://clinicaltrials.gov/study/NCT0 6340568 | Not yet recruiting | Endometrial Cancer | DRUG: BNT323/DB- 1303|DRUG: Doxorubicin|DRUG: Paclitaxel |
BioNTech SE | |||||
| NCT05609 | Study of | https://clinicaltrials.gov/study/NCT0 | Recruiting | Carcinoma, Non- | BIOLOGICAL: | Merck | |||||
| 968 | Pembrolizumab | 5609968 | Small-Cell Lung | Sacituzumab | Sharp & | ||||||
| (MK-3475) | Govitecan|BIOLOGIC | Dohme LLC | |||||||||
| Monotherapy | AL: Pembrolizumab | ||||||||||
| Versus | |||||||||||
| Sacituzumab | |||||||||||
| Govitecan in | |||||||||||
| Combination With | |||||||||||
| Pembrolizumab for | |||||||||||
| Participants With | |||||||||||
| Metastatic Non- | |||||||||||
| small Cell Lung | |||||||||||
| Cancer (NSCLC) | |||||||||||
| With Programmed | |||||||||||
| Cell Death Ligand | |||||||||||
| 1 (PD-L1) Tumor | |||||||||||
| Proportion Score | |||||||||||
| (TPS) ‚â•50% (MK- | |||||||||||
| 3475-D46) | |||||||||||
| NCT03529 | DS-8201a Versus | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Breast Cancer | DRUG: Trastuzumab | Daiichi | |||||
| 110 | T-DM1 for Human | 3529110 | recruiting | deruxtecan (T- | Sankyo | ||||||
| Epidermal Growth | DXd)|DRUG: Ado- | ||||||||||
| Factor Receptor 2 | trastuzumab | ||||||||||
| (HER2)-Positive, | emtansine (T-DM1) | ||||||||||
| Unresectable | |||||||||||
| and/or Metastatic Breast Cancer Previously Treated With Trastuzumab and Taxane [DESTINY- Breast03] |
|||||||||||
| NCT06203 210 | A Study of Ifinatamab Deruxtecan Versus Treatment of Physician's Choice in Subjects With Relapsed Small Cell Lung Cancer |
https://clinicaltrials.gov/study/NCT0 6203210 | Not yet recruiting | Small Cell Lung Cancer | DRUG: Ifinatamab deruxtecan|DRUG: Topotecan|DRUG: Amrubicin|DRUG: Lurbinectedin | Daiichi Sankyo | |||||
| NCT02631 | A Study of | https://clinicaltrials.gov/study/NCT0 | Completed | Epithelial Ovarian | DRUG: Mirvetuximab | ImmunoGen | |||||
| 876 | Mirvetuximab | 2631876 | Cancer|Primary | soravtansine|DRUG: | , Inc. | ||||||
| Soravtansine vs. | Peritoneal | Paclitaxel|DRUG: | |||||||||
| Investigator's | Carcinoma|Fallopian | Pegylated liposomal | |||||||||
| Choice of | Tube | doxorubicin|DRUG: | |||||||||
| Chemotherapy in | Cancer|Ovarian | Topotecan | |||||||||
| Women With | Cancer | ||||||||||
| Folate Receptor | |||||||||||
| (FR) Alpha Positive | |||||||||||
| Advanced | |||||||||||
| Epithelial Ovarian | |||||||||||
| Cancer (EOC), | |||||||||||
| Primary Peritoneal | |||||||||||
| or Fallopian Tube | |||||||||||
| Cancer | |||||||||||
| NCT03734 | Trastuzumab | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Breast Cancer | DRUG: Trastuzumab | Daiichi | |||||
| 029 | Deruxtecan (DS- | 3734029 | recruiting | deruxtecan (DS- | Sankyo | ||||||
| 8201a) Versus | 8201a)|DRUG: | ||||||||||
| Investigator's | Capecitabine|DRUG: | ||||||||||
| Choice for HER2- | Eribulin|DRUG: | ||||||||||
| low Breast Cancer | Gemcitabine|DRUG: | ||||||||||
| That Has Spread or | Paclitaxel|DRUG: | ||||||||||
| Cannot be | Nab-paclitaxel | ||||||||||
| Surgically | |||||||||||
| Removed [DESTINY- Breast04] |
|||||||||||
| NCT04494 | Study of | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Advanced or | DRUG: Trastuzumab | AstraZenec | |||||
| 425 | Trastuzumab | 4494425 | recruiting | Metastatic Breast | deruxtecan|DRUG: | a | |||||
| Deruxtecan (T- | Cancer | Capecitabine|DRUG: | |||||||||
| DXd) vs | Paclitaxel|DRUG: | ||||||||||
| Investigator's | Nab-Paclitaxel | ||||||||||
| Choice | |||||||||||
| Chemotherapy in | |||||||||||
| HER2-low, | |||||||||||
| Hormone Receptor | |||||||||||
| Positive, Metastatic | |||||||||||
| Breast Cancer | |||||||||||
| NCT04595 | Sacituzumab | https://clinicaltrials.gov/study/NCT0 | Recruiting | HER2-negative | DRUG: | German | |||||
| 565 | Govitecan in | 4595565 | Breast Cancer|Triple | Capecitabine|DRUG: | Breast | ||||||
| Primary HER2- | Negative Breast | Carboplatin|DRUG: | Group | ||||||||
| negative Breast | Cancer | Cisplatin|DRUG: | |||||||||
| Cancer | Sacituzumab | ||||||||||
| govitecan | |||||||||||
| NCT05687 | Phase III, Open- | https://clinicaltrials.gov/study/NCT0 | Recruting | NSCLC | DRUG: Datopotamab | AstraZenec | |||||
| 266 | label, First-line | 5687266 | deruxtecan|DRUG: | a | |||||||
| Study of Dato-DXd | Durvalumab|DRUG: | ||||||||||
| in Combination | Carboplatin|DRUG: | ||||||||||
| With Durvalumab | Pembrolizumab|DRU | ||||||||||
| and Carboplatin for | G: Cisplatin|DRUG: | ||||||||||
| Advanced NSCLC | Pemetrexed|DRUG: | ||||||||||
| Without Actionable | Paclitaxel | ||||||||||
| Genomic | |||||||||||
| Alterations | |||||||||||
| NCT05104 | A Phase-3, Open- | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Breast Cancer | DRUG: Dato- | AstraZenec | |||||
| 866 | Label, Randomized | 5104866 | recruiting | DXd|DRUG: | a | ||||||
| Study of Dato-DXd | Capecitabine|DRUG: | ||||||||||
| Versus | Gemcitabine|DRUG: | ||||||||||
| Investigator's | Eribulin|DRUG: | ||||||||||
| Choice of | Vinorelbine | ||||||||||
| Chemotherapy | |||||||||||
| (ICC) in | |||||||||||
| Participants With Inoperable or Metastatic HR- Positive, HER2- Negative Breast Cancer Who Have Been Treated With One or Two Prior Lines of Systemic Chemotherapy (TROPION- Breast01) |
|||||||||||
| NCT06161 | A Study of | https://clinicaltrials.gov/study/NCT0 | Recruiting | Solid Cancer | DRUG: R- | Daiichi | |||||
| 025 | Raludotatug | 6161025 | DXd|DRUG: | Sankyo | |||||||
| Deruxtecan (R- | Gemcitabine|DRUG: | ||||||||||
| DXd) in Subjects | Paclitaxel|DRUG: | ||||||||||
| With Platinum- | Topotecan|DRUG: | ||||||||||
| resistant, High- | PLD | ||||||||||
| grade Ovarian, | |||||||||||
| Primary Peritoneal, | |||||||||||
| or Fallopian Tube | |||||||||||
| Cancer | |||||||||||
| NCT04639 | Asian Study of | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Metastatic Breast | DRUG: Sacituzumab | Gilead | |||||
| 986 | Sacituzumab | 4639986 | recruiting | Cancer | Govitecan- | Sciences | |||||
| Govitecan (IMMU- | hziy|DRUG: Eribulin | ||||||||||
| 132) in HR+/HER2- | Mesylate | ||||||||||
| Metastatic Breast | Injection|DRUG: | ||||||||||
| Cancer (MBC) | Capecitabine Oral | ||||||||||
| Product|DRUG: | |||||||||||
| Gemcitabine | |||||||||||
| Injection|DRUG: | |||||||||||
| Vinorelbine injection | |||||||||||
| NCT04296 | A Study of | https://clinicaltrials.gov/study/NCT0 | Completed | Epithelial Ovarian | DRUG: Mirvetuximab | ImmunoGen | |||||
| 890 | Mirvetuximab | 4296890 | Cancer|Peritoneal | Soravtansine | , Inc. | ||||||
| Soravtansine in | Cancer|Fallopian | ||||||||||
| Platinum-Resistant, | Tube Cancer | ||||||||||
| Advanced High- | |||||||||||
| Grade Epithelial | |||||||||||
| Ovarian, Primary | |||||||||||
| Peritoneal, or Fallopian Tube Cancers With High Folate Receptor- Alpha Expression |
|||||||||||
| NCT01100 | A Phase 3 Study of | https://clinicaltrials.gov/study/NCT0 | Completed | Disease, Hodgkin | DRUG: brentuximab | Seagen Inc. | |||||
| 502 | Brentuximab | 1100502 | vedotin|DRUG: | ||||||||
| Vedotin (SGN-35) | placebo | ||||||||||
| in Patients at High | |||||||||||
| Risk of Residual | |||||||||||
| Hodgkin | |||||||||||
| Lymphoma | |||||||||||
| Following Stem | |||||||||||
| Cell Transplant | |||||||||||
| (The AETHERA | |||||||||||
| Trial) | |||||||||||
| NCT06103 | A Phase III Study | https://clinicaltrials.gov/study/NCT0 | Recruiting | Breast Cancer | DRUG: Dato- | AstraZenec | |||||
| 864 | of Dato-DXd With | 6103864 | DXd|DRUG: | a | |||||||
| or Without | Durvalumab|DRUG: | ||||||||||
| Durvalumab | Paclitaxel|DRUG: | ||||||||||
| Compared With | Nab- | ||||||||||
| Investigator's | paclitaxel|DRUG: | ||||||||||
| Choice of | Gemcitabine|DRUG: | ||||||||||
| Chemotherapy in | Carboplatin|DRUG: | ||||||||||
| Combination With | Pembrolizumab | ||||||||||
| Pembrolizumab in | |||||||||||
| Patients With PD- | |||||||||||
| L1 Positive Locally | |||||||||||
| Recurrent | |||||||||||
| Inoperable or | |||||||||||
| Metastatic Triple- | |||||||||||
| negative Breast | |||||||||||
| Cancer | |||||||||||
| NCT01712 | A Frontline | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Hodgkin Lymphoma | DRUG: brentuximab | Takeda | |||||
| 490 | Therapy Trial in | 1712490 | recruiting | vedotin|DRUG: | |||||||
| Participants With | doxorubicin|DRUG: | ||||||||||
| Advanced Classical | bleomycin|DRUG: | ||||||||||
| Hodgkin | vinblastine|DRUG: | ||||||||||
| Lymphoma | dacarbazine | ||||||||||
| NCT05622 890 | A Single-arm Clinical Trial of IMGN853 in Chinese Adult Patients With Platinum-resistant, Epithelial Ovarian Cancer |
https://clinicaltrials.gov/study/NCT0 5622890 | Recruiting | Epithelial Ovarian Cancer|Peritoneal Cancer|Fallopian Tube Cancer | DRUG: Mirvetuximab Soravtansine | Hangzhou Zhongmei Huadong Pharmaceut ical Co., Ltd. | |||||
| NCT06112 379 | A Phase III Randomised Study to Evaluate Dato- DXd and Durvalumab for Neoadjuvant/Adjuv ant Treatment of Triple-Negative or Hormone Receptor- low/HER2-negative Breast Cancer |
https://clinicaltrials.gov/study/NCT0 6112379 | Recruiting | Breast Cancer | DRUG: Dato- DXd|DRUG: Durvalumab|DRUG: Pembrolizumab|DRU G: Doxorubicin|DRUG: Epirubicin|DRUG: Cyclophosphamide|D RUG: Paclitaxel|DRUG: Carboplatin|DRUG: Capecitabine|DRUG: Olaparib |
AstraZenec a | |||||
| NCT04209 855 | A Study of Mirvetuximab Soravtansine vs. Investigator's Choice of Chemotherapy in Platinum-Resistant, Advanced High- Grade Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancers With High Folate Receptor- Alpha Expression |
https://clinicaltrials.gov/study/NCT0 4209855 | Active – Not yet recruiting | Epithelial Ovarian Cancer|Peritoneal Cancer|Fallopian Tube Cancer | DRUG: Mirvetuximab Soravtansine|DRUG: Paclitaxel|DRUG: Topotecan|DRUG: Pegylated liposomal doxorubicin | ImmunoGen , Inc. |
|||||
| NCT05751 512 | A Study to Evaluate MRG003 vs | https://clinicaltrials.gov/study/NCT0 5751512 | Not yet recruiting | Squamous Cell Carcinoma of the Head and Neck | DRUG: MRG003|DRUG: Cetuximab |
Shanghai Miracogen Inc. | |||||
| Cetuximab/Methotr exate in in the Treatment of Patients With RM- SCCHN |
injection|DRUG: Methotrexate Injection | ||||||||||
| NCT05374 | A Study of Dato- | https://clinicaltrials.gov/study/NCT0 | Recruiting | Breast Cancer | DRUG: Dato- | AstraZenec | |||||
| 512 | DXd Versus | 5374512 | DXd|DRUG: | a | |||||||
| Investigator's | Paclitaxel|DRUG: | ||||||||||
| Choice | Nab- | ||||||||||
| Chemotherapy in | paclitaxel|DRUG: | ||||||||||
| Patients With | Carboplatin|DRUG: | ||||||||||
| Locally Recurrent | Capecitabine|DRUG: | ||||||||||
| Inoperable or | Eribulin mesylate | ||||||||||
| Metastatic Triple- | |||||||||||
| negative Breast | |||||||||||
| Cancer, Who Are | |||||||||||
| Not Candidates for | |||||||||||
| PD-1/PD-L1 | |||||||||||
| Inhibitor Therapy | |||||||||||
| (TROPION- | |||||||||||
| Breast02) | |||||||||||
| NCT05629 | A Study of Dato- | https://clinicaltrials.gov/study/NCT0 | Recruiting | Breast Cancer | DRUG: Dato- | AstraZenec | |||||
| 585 | DXd With or | 5629585 | DXd|DRUG: | a | |||||||
| Without | Durvalumab|DRUG: | ||||||||||
| Durvalumab | Capecitabine|DRUG: | ||||||||||
| Versus | Pembrolizumab | ||||||||||
| Investigator's | |||||||||||
| Choice of Therapy | |||||||||||
| in Patients With | |||||||||||
| Stage I-III Triple- | |||||||||||
| negative Breast | |||||||||||
| Cancer Without | |||||||||||
| Pathological | |||||||||||
| Complete | |||||||||||
| Response | |||||||||||
| Following | |||||||||||
| Neoadjuvant | |||||||||||
| Therapy | |||||||||||
| (TROPION- Breast03) |
|||||||||||
| NCT03523 | DS-8201a in Pre- | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Breast Cancer | DRUG: Trastuzumab | Daiichi | |||||
| 585 | treated HER2 | 3523585 | recruiting | deruxtecan|DRUG: | Sankyo | ||||||
| Breast Cancer That | Capecitabine|DRUG: | ||||||||||
| Cannot be | Lapatinib|DRUG: | ||||||||||
| Surgically | Trastuzumab | ||||||||||
| Removed or Has | |||||||||||
| Spread [DESTINY- | |||||||||||
| Breast02] | |||||||||||
| NCT01777 | ECHELON-2: A | https://clinicaltrials.gov/study/NCT0 | Completed | Anaplastic Large- | DRUG: brentuximab | Seagen Inc. | |||||
| 152 | Comparison of | 1777152 | Cell Lymphoma|Non- | vedotin|DRUG: | |||||||
| Brentuximab | Hodgkin | doxorubicin|DRUG: | |||||||||
| Vedotin and CHP | Lymphoma|T-Cell | prednisone|DRUG: | |||||||||
| With Standard-of- | Lymphoma | vincristine|DRUG: | |||||||||
| care CHOP in the | cyclophosphamide | ||||||||||
| Treatment of | |||||||||||
| Patients With | |||||||||||
| CD30-positive | |||||||||||
| Mature T-cell | |||||||||||
| Lymphomas | |||||||||||
| NCT06074 | MK-2870 Versus | https://clinicaltrials.gov/study/NCT0 | Recruiting | Non-small Cell Lung | BIOLOGICAL: MK- | Merck | |||||
| 588 | Chemotherapy in | 6074588 | Cancer (NSCLC) | 2870|DRUG: | Sharp & | ||||||
| Previously Treated | Docetaxel|DRUG: | Dohme LLC | |||||||||
| Advanced or | Pemetrexed | ||||||||||
| Metastatic | |||||||||||
| Nonsquamous | |||||||||||
| Non-small Cell | |||||||||||
| Lung Cancer | |||||||||||
| (NSCLC) With | |||||||||||
| EGFR Mutations or | |||||||||||
| Other Genomic | |||||||||||
| Alterations (MK- | |||||||||||
| 2870-004) | |||||||||||
| NCT03474 | A Study to | https://clinicaltrials.gov/study/NCT0 | Active – Not yet | Ureteral | DRUG: Enfortumab | Astellas | |||||
| 107 | Evaluate | 3474107 | recruiting | Cancer|Urothelial | Vedotin|DRUG: | Pharma | |||||
| Enfortumab | Cancer|Bladder | Docetaxel|DRUG: | Global | ||||||||
| Vedotin Versus (vs) | Cancer | ||||||||||
| Chemotherapy in Subjects With Previously Treated Locally Advanced or Metastatic Urothelial Cancer (EV-301) |
Vinflunine|DRUG: Paclitaxel | Developme nt, Inc. | |||||||||
| NCT05754 | A Study of | https://clinicaltrials.gov/study/NCT0 | Recruiting | Advanced or | DRUG: | Shanghai | |||||
| 853 | MRG002 Versus | 5754853 | Metastatic | MRG002|DRUG: | Miracogen | ||||||
| Investigator's | Urothelium Cancer | Docetaxel | Inc. | ||||||||
| Choice of | Injection|DRUG: | ||||||||||
| Chemotherapy in | Paclitaxel | ||||||||||
| the Treatment of | Injection|DRUG: | ||||||||||
| Patients With | Gemcitabine | ||||||||||
| HER2-positive | Hydrochloride for | ||||||||||
| Unresectable | Injection|DRUG: | ||||||||||
| Advanced or | Pemetrexed | ||||||||||
| Metastatic | Disodium Injection | ||||||||||
| Urothelial Cancer | |||||||||||
| NCT05445 | Mirvetuximab | https://clinicaltrials.gov/study/NCT0 | Recruiting | Ovarian | DRUG: Mirvetuximab | ImmunoGen | |||||
| 778 | Soravtansine With | 5445778 | Cancer|Peritoneal | soravtansine plus | , Inc. | ||||||
| Bevacizumab | Cancer|Fallopian | Bevacizumab|DRUG: | |||||||||
| Versus | Tube Cancer | Bevacizumab | |||||||||
| Bevacizumab as | |||||||||||
| Maintenance in | |||||||||||
| Platinum-sensitive | |||||||||||
| Ovarian, Fallopian | |||||||||||
| Tube, or Peritoneal | |||||||||||
| Cancer | |||||||||||
| (GLORIOSA) | |||||||||||
| NCT02785 | Vadastuximab | https://clinicaltrials.gov/study/NCT0 | Terminated | Acute Myeloid | DRUG: 33A|DRUG: | Seagen Inc. | |||||
| 900 | Talirine (SGN- | 2785900 | Leukemia | placebo|DRUG: | |||||||
| CD33A; 33A) | azacitidine|DRUG: | ||||||||||
| Combined With | decitabine | ||||||||||
| Azacitidine or | |||||||||||
| Decitabine in Older | |||||||||||
| Patients With | |||||||||||
| Newly Diagnosed | |||||||||||
| Acute Myeloid Leukemia | |||||||||||
| NCT06132 | MK-2870 in Post | https://clinicaltrials.gov/study/NCT0 | Recruiting | Endometrial Cancer | BIOLOGICAL: MK- | Merck | |||||
| 958 | Platinum and Post | 6132958 | 2870|DRUG: | Sharp & | |||||||
| Immunotherapy | Doxorubicin|DRUG: | Dohme LLC | |||||||||
| Endometrial | Paclitaxel | ||||||||||
| Cancer (MK-2870- | |||||||||||
| 005) | |||||||||||
| NCT02573 | A Study of ABT- | https://clinicaltrials.gov/study/NCT0 | Completed | Glioblastoma|Gliosar | DRUG: | AbbVie | |||||
| 324 | 414 in Participants | 2573324 | coma | Temozolomide|DRU | |||||||
| With Newly | G: Depatuxizumab | ||||||||||
| Diagnosed | mafodotin|RADIATIO | ||||||||||
| Glioblastoma | N: Radiation|DRUG: | ||||||||||
| (GBM) With | Placebo for ABT-414 | ||||||||||
| Epidermal Growth | |||||||||||
| Factor Receptor | |||||||||||
| (EGFR) | |||||||||||
| Amplification | |||||||||||
| NCT03262 | SYD985 vs. | https://clinicaltrials.gov/study/NCT0 | Completed | Metastatic Breast | DRUG: | Byondis | |||||
| 935 | Physician's Choice | 3262935 | Cancer | (vic-)trastuzumab | B.V. | ||||||
| in Participants With | duocarmazine|DRUG | ||||||||||
| HER2-positive | : Physician's choice | ||||||||||
| Locally Advanced | |||||||||||
| or Metastatic | |||||||||||
| Breast Cancer | |||||||||||
| NCT04924 | A Study of | https://clinicaltrials.gov/study/NCT0 | Recruiting | Advanced Breast | DRUG: | Shanghai | |||||
| 699 | MRG002 in the | 4924699 | Cancer|Metastatic | MRG002|DRUG: | Miracogen | ||||||
| Treatment of | Breast Cancer | Trastuzumab | Inc. | ||||||||
| Patients With | Emtansine for | ||||||||||
| HER2-positive | Injection | ||||||||||
| Unresectable | |||||||||||
| Locally Advanced | |||||||||||
| or Metastatic | |||||||||||
| Breast Cancer | |||||||||||
| NCT05950 | Trastuzumab | https://clinicaltrials.gov/study/NCT0 | Recruiting | Breast Cancer | DRUG: Trastuzumab | Daiichi | |||||
| 945 | Deruxtecan (T- | 5950945 | Deruxtecan | Sankyo | |||||||
| DXd) in Patients | |||||||||||
| Who Have | |||||||||||
| Hormone Receptor-negative and Hormone Receptor-positive HER2-low or HER2 IHC 0 Metastatic Breast Cancer |
|||||||||||
| NCT05329 | Upifitamab | https://clinicaltrials.gov/study/NCT0 | Terminated | High Grade Serous | DRUG: Upifitimab | Mersana | |||||
| 545 | Rilsodotin | 5329545 | Ovarian | rilsodotin|OTHER: | Therapeutic | ||||||
| Maintenance in | Cancer|Fallopian | Placebo | s | ||||||||
| Platinum-Sensitive | Tube | ||||||||||
| Recurrent Ovarian | Cancer|Primary | ||||||||||
| Cancer (UP-NEXT) | Peritoneal Cancer | ||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





