1. Introduction
The performance of materials is significantly influenced by various factors such as grain size, grain size distribution, subgrain boundaries, phase composition, and the characteristics of precipitates [
1,
2,
3]. Thermal treatment and controlled rolling and cooling processes can enhance the properties of materials by refining grain size, optimizing microstructure, and managing phase transformations [
4,
5,
6,
7]. The evolution of microstructure, the formation of new phases, and grain growth behavior during these processes play crucial roles in determining the mechanical properties of materials. However, due to the limitations of experimental equipment and conditions, it is often challenging to directly observe the microstructural evolution during hot processing [
8,
9]. This limitation hinders the comprehensive understanding of microstructural changes and high-temperature phase transformations.
Confocal Laser Scanning Microscopy (CLSM), developed in the 1980s, offers a solution for in-situ observation of microstructural changes at high temperatures [
10,
11,
12]. CLSM provides high resolution and can generate clear three-dimensional images through simultaneous multi-fluorescence observation, advantages not available with traditional optical microscopy. Lasertec Corporation in Japan has advanced CLSM technology by integrating confocal laser scanning, infrared heating, and tensile testing into the High Temperature Confocal Laser Scanning Microscopy (HTCLSM) [
13,
14]. This innovation has become an essential tool for visually studying processes such as melting, solidification, and high-temperature deformation of materials. The Lasertec confocal laser scanning microscope uses a He-Ne laser source and precise confocal spatial filtering to form a conjugate image. The laser creates a point light source at the focal plane of the objective lens, scanning the sample. Spatial filtering at the detection pinhole in the focal plane of the measurement lens effectively suppresses stray fluorescence and interference from out-of-focus planes. This results in high signal-to-noise ratio optical tomographic images. The laser's monochromatic nature and small depth of focus yield high longitudinal resolution, enabling non-destructive layer scanning and fluorescence intensity measurements at different sample depths. The optical sections from various focal planes are reconstructed into a three-dimensional structure of the sample. The development of HTCLSM has significantly advanced the in-situ observation of material microstructure transformations, such as high-temperature phase changes in steel, high-temperature precipitation processes, ferrite-austenite transformations, and austenite decomposition [
15,
16,
17]
HTCLSM's capability to observe in real-time the microstructural changes under high temperatures makes it indispensable for materials research and development. It is particularly useful in studying phase transformations in steels, where rapid heating and cooling rates can be simulated to mimic industrial processes. This allows for the investigation of phenomena such as austenitization, martensitic transformation, and carbide precipitation, providing valuable insights into the kinetics and thermodynamics of these transformations [
18,
19,
20]. These advancements have deepened the understanding of microstructural transformations in materials. Microstructural evolution during thermal processing is a complex interplay of nucleation, growth, and coarsening of different phases [
21,
22,
23]. The precise control of these processes is vital for achieving the desired mechanical properties in advanced materials. For instance, in steels, the formation and dissolution of carbides, nitrides, and other intermetallic compounds during heating and cooling significantly affect hardness, strength, and toughness [
24,
25,26]. Understanding these transformations at the microscopic level allows for the development of heat treatment schedules that optimize performance characteristics for specific applications.
To optimize the hot working process, particularly the heat treatment of high-nickel shipbuilding steel, and improve its mechanical properties, this study employs HTCLSM for in-situ observation of the microstructural evolution during heating and holding. Combining these observations with scanning electron microscopy (SEM) and energy-dispersive spectroscopy (EDS) analysis, the study discusses the precipitation of carbides in high-nickel shipbuilding steel. By providing a deeper understanding of microstructural evolution in high-nickel steels, the research can contribute to the development of more robust and reliable materials for applications. Additionally, the methodologies employed in this study can be applied to other alloy systems, facilitating advancements in various high-performance materials used in aerospace, automotive, and energy sectors.
2. Experimental Materials and Methods
2.1. Experimental Materials
The primary chemical composition of the steels used in this study is detailed in
Table 1. The steel ingots were produced using a 50 kg vacuum induction furnace and subsequently forged into blocks with dimensions of 120×120×260 mm. These blocks were then soaked at 1200°C for a minimum of one hour to ensure uniform temperature distribution and homogenization. Following the soaking process, the blocks were hot-rolled into plates with a cross-sectional dimension of 20 mm × 150 mm.
2.2. High-Temperature In-Situ Equipment
The in-situ observation of the material heating process was conducted using High Temperature Confocal Laser Scanning Microscopy (HTCLSM). This sophisticated equipment integrates multiple advanced systems, including a high-temperature furnace, an imaging system, a temperature control system, and a computer system. Additionally, it features a tensile furnace, a protective gas system, a cooling system, a workbench control system, a tensile control system, and an image detection system, as illustrated in
Figure 1.
HTCLSM is particularly suited for observing the microstructural evolution of materials at elevated temperatures. The high-temperature furnace provides precise thermal conditions required for studying phase transformations, grain growth, and other high-temperature phenomena. The imaging system, equipped with a He-Ne laser source, ensures high-resolution and high-contrast images by utilizing confocal spatial filtering. This allows for the suppression of out-of-focus light and stray fluorescence, resulting in clear, three-dimensional reconstructions of the sample's microstructure. The temperature control system ensures accurate and stable thermal conditions during the experiments, which is critical for reproducibility and reliability of the observations. The integration with a computer system allows for real-time data acquisition and analysis, facilitating the monitoring and recording of microstructural changes as they occur. The protective gas system, usually involving an inert gas like argon, is employed to prevent oxidation and contamination of the sample during high-temperature exposure. The cooling system is used to control the sample's cooling rate, which is essential for studying the effects of different cooling regimes on the microstructure and properties of the material.
The HTCLSM setup provides a comprehensive platform for in-situ high-temperature studies, combining precise thermal and mechanical control with advanced imaging capabilities to enable detailed analysis of microstructural evolution and its impact on material properties.
2.3. In-Situ Observation Samples
The samples used for in-situ microstructural observation were cylindrical with dimensions of φ7.5×3.5 mm, as shown in
Figure 2. Prior to observation, the sample surfaces were meticulously ground, polished, and cleaned. Due to the self-imaging capabilities of HTCLSM, etching of the polished samples was not necessary.
Once prepared, the samples were placed into an Al2O3 crucible, which was then inserted into the heating furnace. The furnace was evacuated to a vacuum pressure of 1×10⁻⁶ MPa, followed by the introduction of a protective gas to prevent oxidation and contamination during heating. Various parameters of the HTCLSM were set to ensure optimal conditions for in-situ observation.
Figure 2.
In-Situ Observation Sample.
Figure 2.
In-Situ Observation Sample.
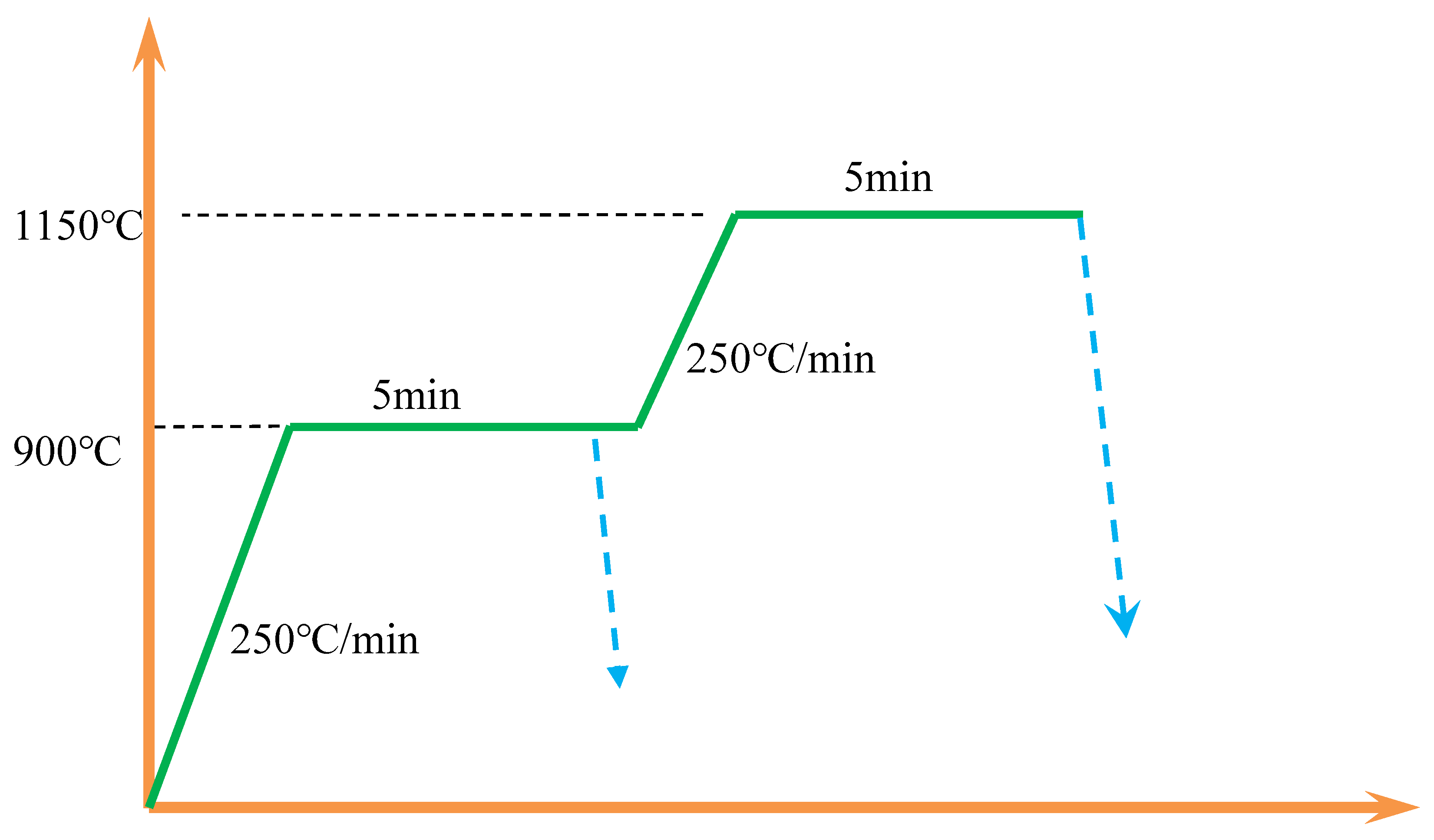
The heating and holding procedures for the in-situ observation samples are depicted in
Figure 3. The samples were initially heated at a rate of 250°C/min to 900°C and held at this temperature for 5 minutes. Subsequently, one group of samples was further heated to 1150°C at the same rate and held for an additional 5 minutes before being rapidly cooled to room temperature. Another group was directly cooled to room temperature after the initial hold at 900°C.
After the completion of the in-situ observation experiments, the samples were subjected to SEM and EDS analysis to investigate their surface composition and microstructural characteristics. This meticulous process ensures that the observed changes in microstructure are accurately captured and analyzed, providing valuable insights into the behavior of the material under controlled thermal conditions. The combination of HTCLSM's advanced imaging capabilities with precise thermal control allows for a comprehensive study of microstructural evolution, critical for optimizing material properties and processing techniques.
3. Results and Discussion
3.1. In-Situ Observation of Microstructural Transformation
The in-situ observation of the microstructural transformation during the heating process was conducted using High Temperature Confocal Laser Scanning Microscopy (HTCLSM). The changes in the material's microstructure during heating are shown in
Figure 4.
In
Figure 4, the numbers in the top left corner indicate the time (seconds) and the real-time temperature (°C) of the sample. Initially, at temperatures below 580°C, no significant microstructural changes are observed, indicating that the material remains stable within this temperature range. However, as the temperature reaches 700°C, the material begins to enter the austenitization phase. During this phase, austenite nuclei first form at the grain boundaries, as indicated by the arrows in
Figure 4c. These nuclei appear as fine, discrete particles along the grain boundaries, highlighting the initial stages of austenite formation. As the temperature increases further, these austenite nuclei grow and coalesce, forming larger austenite grains, as shown in
Figure 4e. By approximately 740°C, the microstructure is predominantly composed of austenite. The transformation to austenite within the temperature range of 700°C to 740°C is relatively rapid, occurring over approximately 12 seconds. This rapid transformation can be attributed to the high mobility of grain boundaries at elevated temperatures, facilitating the quick formation and growth of austenite grains.
When the temperature reaches 900°C, precipitates start to form within the microstructure. The changes in microstructure and the formation of precipitates during the holding period at 900°C are depicted in
Figure 5.
Figure 5 illustrates that the sample reaches the set temperature of 900°C after 240 seconds, at which point black precipitates begin to appear (
Figure 5a). Comparing Figures 5a and 5b, it is evident that the number of precipitates increases with holding time. After 50 seconds at 900°C, the quantity of precipitates reaches its maximum, as shown in
Figure 5c. These precipitates are likely to be carbides or other intermetallic compounds, which form at specific nucleation sites such as grain boundaries and dislocations. As holding continues, the size of the precipitates increases due to Ostwald ripening, where larger precipitates grow at the expense of smaller ones, as illustrated in
Figure 5d.
The dark appearance of these precipitates can be attributed to their interaction with trace amounts of gases in the observation chamber, which may result in the formation of oxide layers on the precipitate surfaces. Additionally, the high resolution and in-situ capabilities of HTCLSM allow for the real-time tracking of these microstructural changes, providing valuable insights into the kinetics of precipitation and growth mechanisms.
The evolution of the microstructure as the temperature is increased from 900°C to 1150°C, followed by a 5-minute hold, is shown in
Figure 6.
As the temperature rises from 900°C to 1150°C, the microstructural changes are continuously monitored. From 900°C to 1026°C, there is no significant change in the number of precipitates (Figures 6a and 6b), indicating that the existing precipitates remain stable within this temperature range. However, when the temperature reaches 1060°C, the precipitates begin to dissolve into the matrix, as indicated by the arrows in
Figure 6c. This dissolution process continues, and by 1131°C, the precipitates have completely dissolved into the matrix, as shown in
Figure 6d.
The dissolution of precipitates at high temperatures can be explained by the increased solubility of the precipitate-forming elements in the matrix at elevated temperatures. This process is critical for homogenizing the microstructure and eliminating potential sites for crack initiation, thereby enhancing the overall mechanical properties of the material.
These in-situ observations provide comprehensive insights into the phase transformations and precipitation behavior of the material at elevated temperatures. Understanding these transformations is essential for optimizing thermal processing parameters, such as heating rates, holding times, and cooling rates, to achieve desired microstructural characteristics and mechanical properties. The real-time data obtained from HTCLSM enables a detailed analysis of the dynamic processes occurring within the material, facilitating the development of advanced materials with tailored properties for specific applications.
3.2. SEM Analysis of Precipitates
Through in-situ observation using HTCLSM, it was evident that black precipitates formed during the holding process at 900°C. These precipitates persisted even when the temperature was increased to above 1000°C, indicating their thermal stability. The appearance of these precipitates during the heating process likely influences the mechanical properties of the material. To further investigate the nature and characteristics of these precipitates, a sample subjected to the same heating rate and soaked at 900°C for 5 minutes was rapidly cooled to room temperature to preserve the dark phase particles. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS) analyses were then conducted on the precipitates, as shown in
Figure 7 and
Figure 8.
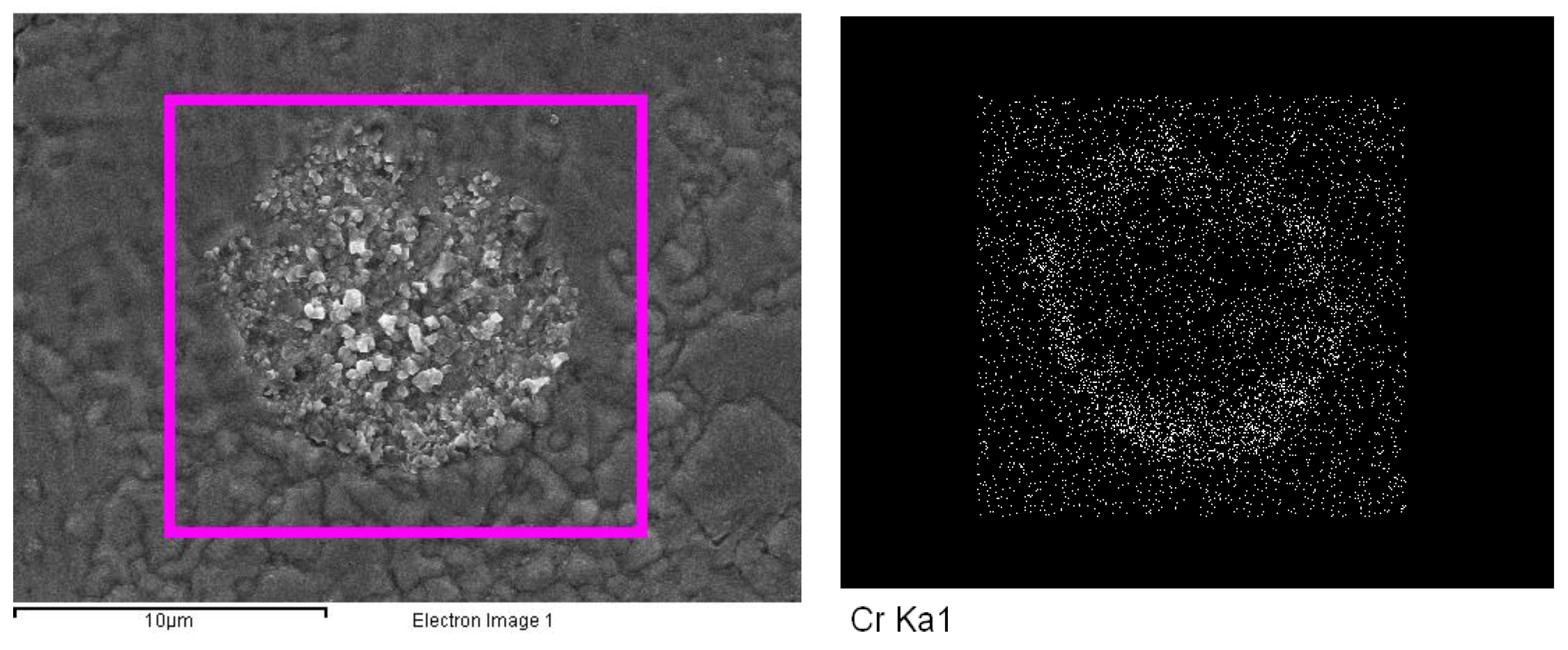
In
Figure 8, the mapping analysis of the alloying elements in the precipitated dark phases shows areas similar to those marked by arrows in
Figure 7, which contain dark particles. Initially, the precipitates appear circular, but as they grow, their shapes become irregular. EDS was employed to analyze the composition of these precipitated phases. The chemical compositions of the three positions indicated in
Figure 7 reveal that the dark phases contain significantly higher concentrations of alloying elements such as Cr, V, and Mn compared to the matrix. This suggests that the precipitation of the dark phases may be due to the segregation of these alloying elements during the thermal process.
The presence of precipitates, especially those enriched with alloying elements like Cr, V, and Mn, can have profound effects on the mechanical properties of the material. These elements can influence the hardness, strength, and toughness of the steel. For instance, vanadium and chromium are known to form carbides that can enhance the hardness and wear resistance of the material. Manganese can improve the hardenability and tensile strength. The thermal stability of these precipitates up to temperatures above 1000°C suggests they play a significant role in the high-temperature performance of the material. The segregation and subsequent precipitation of alloying elements at elevated temperatures could lead to localized strengthening or weakening of the microstructure, depending on the nature and distribution of the precipitates.
Furthermore, the morphology of these precipitates can also impact the material properties. The transition from spherical to irregular shapes as the precipitates grow suggests a complex precipitation mechanism, possibly involving multiple stages of nucleation and growth. This morphological evolution can influence the mechanical stability of the precipitates and their interaction with the surrounding matrix. To elucidate the impact of these precipitates on the material's properties, it is essential to correlate their presence with mechanical testing results. For instance, hardness tests, tensile tests, and impact tests can provide insights into how these precipitates affect the material's overall performance. By combining SEM and EDS analyses with mechanical testing, a comprehensive understanding of the precipitate behavior and its influence on the material properties can be achieved.
Understanding these precipitates' formation mechanisms and their impact on material properties is crucial for optimizing heat treatment processes and improving the performance of high-nickel steel. This knowledge aids in tailoring the microstructure to achieve desired mechanical properties, enhancing the material's applicability in demanding environments. The insights gained from this study can inform the development of advanced materials with superior high-temperature performance and reliability.
3.3. Determination of Precipitate Composition
To identify the types of precipitates formed during the heating process, Transmission Electron Microscopy (TEM) was employed to analyze the samples. The results are shown in
Figure 9.
From
Figure 9, it is evident that precipitates are formed and distributed within the grains, along the grain boundaries, and at the martensite lath boundaries, as illustrated in
Figure 9a. Figures 9b and 9d show TEM micrographs where the precipitation particles are clearly visible. These images indicate that the particles tend to form at the boundaries and dislocations, which act as nucleation sites for the precipitates. The diffraction pattern in
Figure 9c confirms that the particles are chromium carbides, specifically Cr7C3. The presence of Cr7C3, which is significantly harder than ordinary cementite, can substantially increase the steel's strength. This is due to the fact that Cr7C3 carbides have a higher hardness and wear resistance, providing enhanced reinforcement to the steel matrix. Additionally, Cr7C3 precipitates impede dislocation movement, thereby increasing the material's yield strength and hardness.
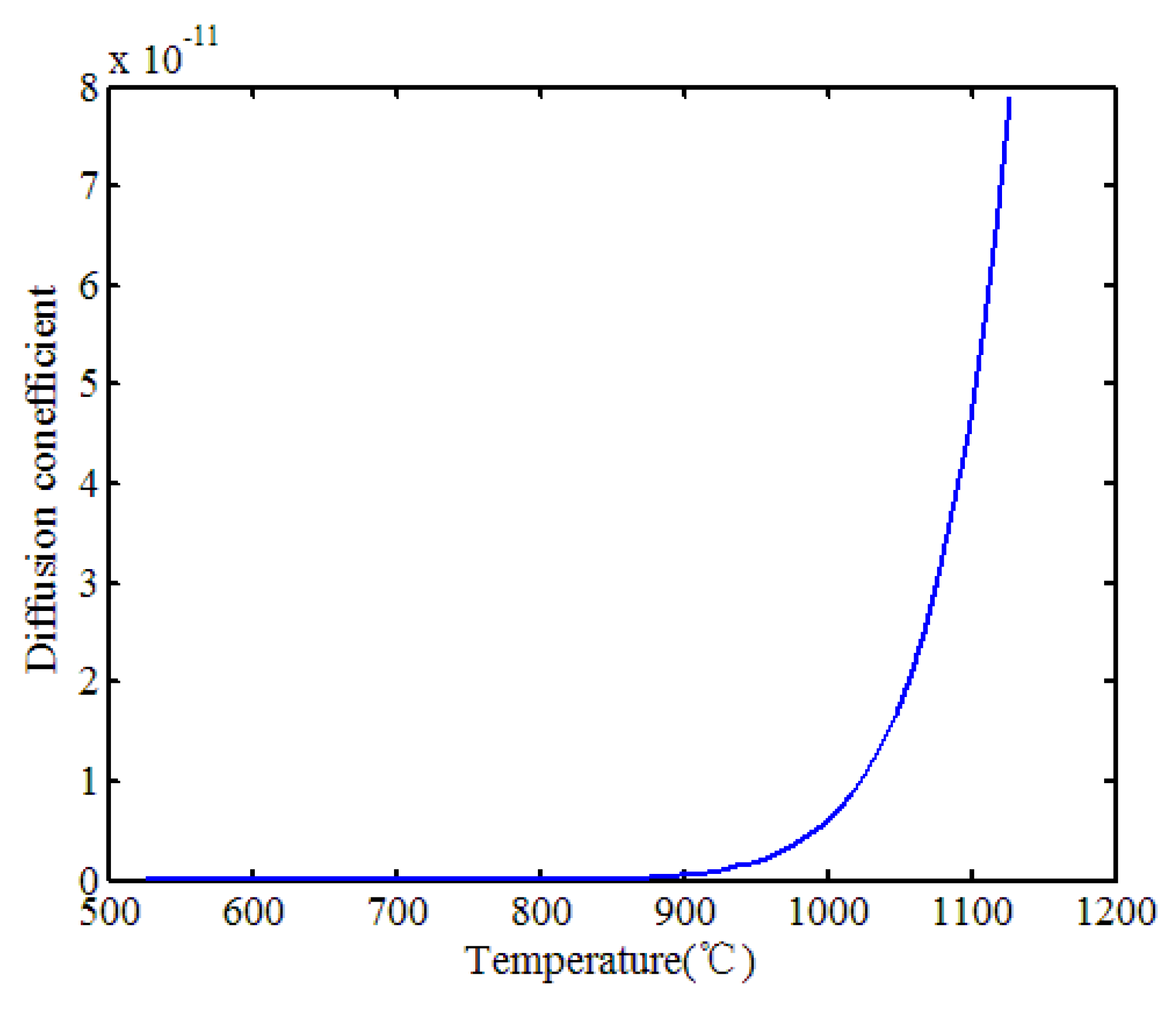
The importance of Cr7C3 in enhancing mechanical properties has been well-documented in the literature. Bowen A.W. and Leak G.M. have investigated the diffusion behavior of Cr7C3 in ferrite and austenite phases [27], as shown in
Figure 10.
The variation in the diffusion coefficient of Cr7C3 aligns with the in-situ observation results presented earlier. As seen from the diffusion coefficient curve, when the temperature exceeds 1000°C, the diffusion coefficient increases by at least two orders of magnitude. This explains why, at 1026°C, the particles begin to dissolve into the matrix and completely dissolve at around 1131°C, corroborating the previous findings.
The identification of chromium carbides (Cr7C3) as the primary precipitates provides significant insights into the thermal stability and mechanical enhancement of the high-nickel steel [28]. The formation of Cr7C3 at grain boundaries and dislocations suggests a strong interaction between these precipitates and the microstructural features of the steel, contributing to the observed increase in strength.
The diffusion behavior of Cr7C3, with a marked increase in the diffusion coefficient above 1000°C, highlights the dynamic nature of these precipitates during thermal processing. Understanding this behavior is crucial for optimizing heat treatment protocols to control the distribution and size of Cr7C3 precipitates, thereby tailoring the material's properties to specific applications.
In conclusion, the TEM analysis has provided a comprehensive understanding of the precipitate composition and its implications for the mechanical properties of high-nickel steel. The formation of Cr7C3 precipitates and their interaction with the microstructure play a pivotal role in enhancing the strength, thermal stability, and possibly the corrosion resistance of the material. These findings contribute to the broader knowledge required for developing advanced materials with superior performance, facilitating the design of more robust and reliable engineering solutions.
4. Conclusions
This research significantly advances the understanding of microstructural transformations in high-nickel steels and demonstrates the critical role of advanced microscopy techniques in material science. The comprehensive insights gained from HTCLSM, SEM, EDS, and TEM analyses provide a robust framework for optimizing the heat treatment processes of high-nickel shipbuilding steel. The key findings are summarized as follows:
(1)The study successfully utilized High Temperature Confocal Laser Scanning Microscopy (HTCLSM) to observe the in-situ microstructural evolution of high-nickel shipbuilding steel during heating and holding processes. It was determined that austenitization begins at approximately 700°C with nucleation primarily occurring at grain boundaries, leading to significant microstructural changes.
(2)At 900°C, the formation of black precipitates was observed, which persisted even when the temperature exceeded 1000°C. These precipitates were identified as chromium carbides (Cr7C3) through SEM and EDS analyses. The Cr7C3 precipitates contribute to the material's overall mechanical strength due to their hard, dispersed nature.
(3)The study found that the Cr7C3 precipitates begin to dissolve into the matrix at temperatures above 1026°C, with complete dissolution occurring around 1131°C. This dissolution behavior aligns with the increased diffusion coefficients of Cr7C3 at elevated temperatures.
Author Contributions
Conceptualization, Guojin Sun; methodology, Guojin Sun, Zhenggui Li and Qi Wang; formal analysis, Guojin Sun, Qi Wang and Zhenggui Li; investigation, Guojin Sun ,Qi Wang and Zhenggui Li; data curation, Guojin Sun and Qi Wang ; writing—original draft preparation, Guojin Sun; writing—review and editing, Guojin Sun and Zhenggui Li; supervision, Guojin Sun and Zhengui Li. All authors have read and agreed to the published version of the manuscript.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors acknowledge financial support from Kunlun Talent Project of Qinghai Province(2023-QLGKLYCZX-032).
References
- Okuda K , Yamamitsu K , Xu X ,et al.Change in NbC Precipitates during Cold Rolling of Nb-added IF Steel Sheet [J].Tetsu-to-Hagane, 2023, 109(4):323-332. [CrossRef]
- Zhao B , Ai F , Yuan Q ,et al.Effect of Normalizing on Impact and Corrosion Resistance of Low-temperature Service Seamless Steel Pipe [J].Journal of Physics: Conference Series, 2023, 2463(1). [CrossRef]
- Huang S , Li C , Li Z ,et al.Effect of Thermal Simulation Process on Microstructure of Seismic Steel Bars [J].Materials (Basel, Switzerland), 2022, 15(10). [CrossRef]
- Isobe K , Kumagai Y , Satou T .Effects of Cooling Conditions and γ Grain Size on Each Behavior of Transformations, 3-Dimensional Thermal Deformation and Stress Generation during Immersion Cooling of Steel Bloom [J].ISIJ International, 2023, 63(5):919-929. [CrossRef]
- Napal U G , Segarra M A , Lazcano B E ,et al.Thermal Mass Effect on the Solution Cooling Rate and on HIPped Astroloy Component Properties [J].Materials (Basel, Switzerland), 2022, 15(4). [CrossRef]
- Yan M , Sun J N , Huang H G ,et al.Effect of hot rolling and cooling process on microstructure and properties of 2205/Q235 clad plate [J].Journal of Iron and Steel Research International, 2018, 25. [CrossRef]
- Kuziak R , Kania Z , Pidvysots'Kyy V ,et al.Through Process Modelling of Rolling and Controlled Cooling of TRIP Assisted Bainitic Steel Rods and Prediction of the Retained Austenite in Products [J].Materials science forum, 2017, 892:23-33. [CrossRef]
- Liu G , Liu G , Liu J ,et al.Microstructure Evolution and Mechanical Properties of Medium Carbon Martensitic Steel during Warm Rolling and Annealing Process [J].Materials, 2021, 14(22):6900-. [CrossRef]
- Li X , Zhou X , Jiang Q ,et al.The Prediction of the Mechanical Properties for Hot-Rolled Nb Micro-Alloyed Dual-Phase Steel Based on Microstructure Characteristics [J].JOM, 2023. [CrossRef]
- Dige I , Nilsson H , Kilian M ,et al.In situ identification of streptococci and other bacteria in initial dental biofilm by confocal laser scanning microscopy and fluorescence in situ hybridization [J].European Journal of Oral Sciences, 2010, 115(6):459-467. [CrossRef]
- Reid M , Phelan D , Dippenaar R .Concentric Solidification for High Temperature Laser Scanning Confocal Microscopy [J].Isij International, 2004, 44(3):565-572. [CrossRef]
- Griesser S , Dippenaar R .Enhanced Concentric Solidification Technique for High-Temperature Laser-Scanning Confocal Microscopy [J].ISIJ International, 2014(54-3).
- Chen R , Zheng Z , Li N ,et al.In-situ investigation of phase transformation behaviors of 300M steel in continuous cooling process - ScienceDirect [J].Materials Characterization, 2018, 144:400-410. [CrossRef]
- Klein T , Niknafs S , Dippenaar R ,et al.High Temperature Laser-Scanning Confocal Microscopy for the in-situ Investigation of Grain Growth and Phase Transformations in Intermetallic γ-TiAl based Alloys [J].Praktische Metallographie, 2015, 52(5):259-269. [CrossRef]
- Chikama H , Shibata H , Emi T ,et al."In-situ" Real Time Observation of Planar to Cellular and Cellular to Dendritic Transition of Crystals Growing in Fe–C Alloy Melts [J].materials transactions, 2007, 37(4):620-626. [CrossRef]
- Shibata H , Arai Y , Suzuki M ,et al.Kinetics of peritectic reaction and transformation in Fe-C alloys [J].Metallurgical & Materials Transactions B, 2000, 31(5):981-991. [CrossRef]
- Yin H , Emi T , Shibata H .Morphological instability of δ-ferrite/γ-austenite interphase boundary in low carbon steels [J].Acta Materialia, 1999, 47(5):1523-1535. [CrossRef]
- Ryttberg K , Wedel M K , Dahlman P ,et al.Microstructural evolution during fracture induced by high strain rate deformation of 100Cr6 steel [J].Journal of Materials Processing Tech, 2009, 209(7):3325-3334. [CrossRef]
- Kozmel T , Tin S .Effects of Carbides on the Microstructural Evolution in Sub-micron Grain 9310 Steel During Isothermal Heat Treatment [J].Metallurgical&Materials Transactions A, 2015. [CrossRef]
- Wang B , Xu Y , Chen L ,et al.Insights into the microstructure evolution and wear resistance of Nano-TiC particles reinforced High-Cr hot work die steel [J].Journal of Materials Research and Technology, 2024, 30:8371-8381. [CrossRef]
- Benarrache S , Benchatti T , Benhorma H .Formation and Dissolution of Carbides and Nitrides in the Weld Seam of X70 Steel by the Effects of Heat Treatments [J].Annales de Chimie Science des Matériaux, 2019, 43(1):11-16. [CrossRef]
- Sai Gautam G , Hari Kumar K C .Elastic, thermochemical and thermophysical properties of rock salt-type transition metal carbides and nitrides: A first principles study [J].Journal of Alloys & Compounds, 2014, 587:380-386. [CrossRef]
- I,I,Gorbachev,et al.Analysis of the solubility of carbides, nitrides, and carbonitrides in steels using methods of computer thermodynamics: IV. Solubility of carbides, nitrides, and carbonitrides in the Fe-Nb-C, Fe-Nb-N, and Fe-Nb-C-N systems [J].The Physics of Metals and Metallography, 2010, 110(1). [CrossRef]
- Bowen A W , Leak G M .Solute diffusion in alpha- and gamma-iron [J].Metallurgical Transactions, 1970, 1(6):1695-1700. [CrossRef]
- Sun L , Ji X , Zhao L ,et al.First Principles Investigation of Binary Chromium Carbides Cr7C3, Cr3C2 and Cr23C6: Electronic Structures, Mechanical Properties and Thermodynamic Properties under Pressure [J].Materials, 2022, 15. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).