Submitted:
08 August 2024
Posted:
12 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
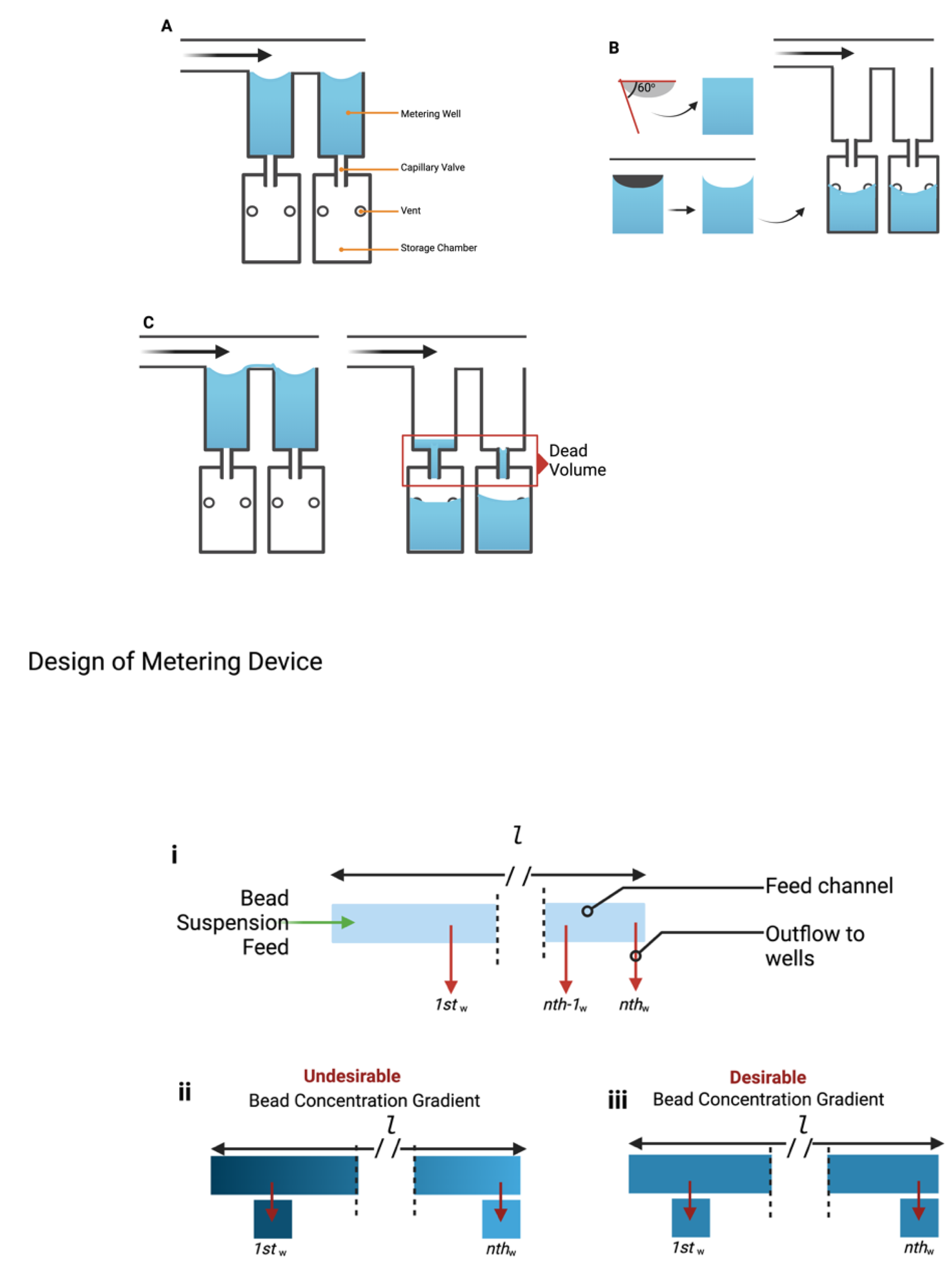
2.1. Design and Frication of Fluid-Metering Chip
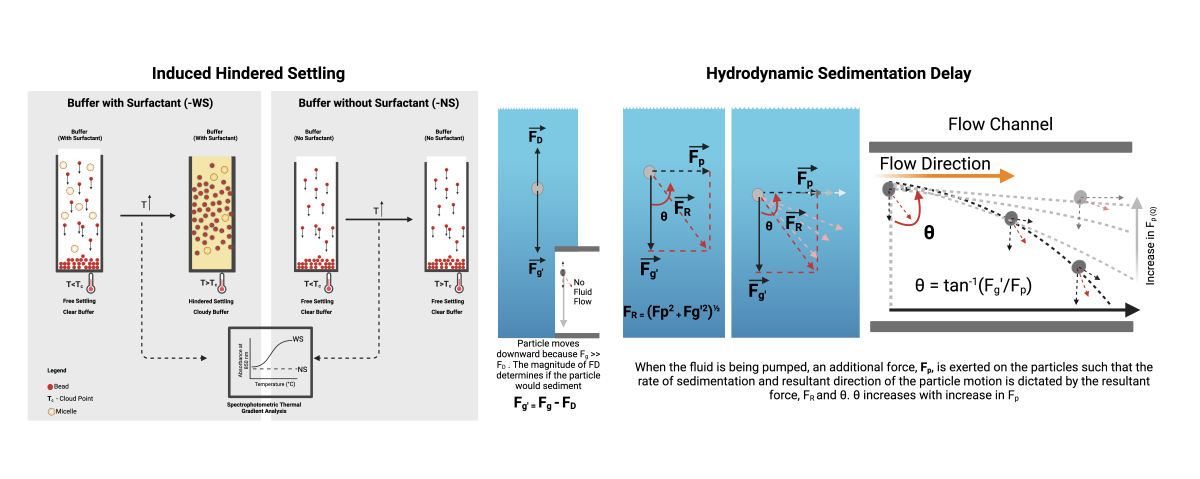
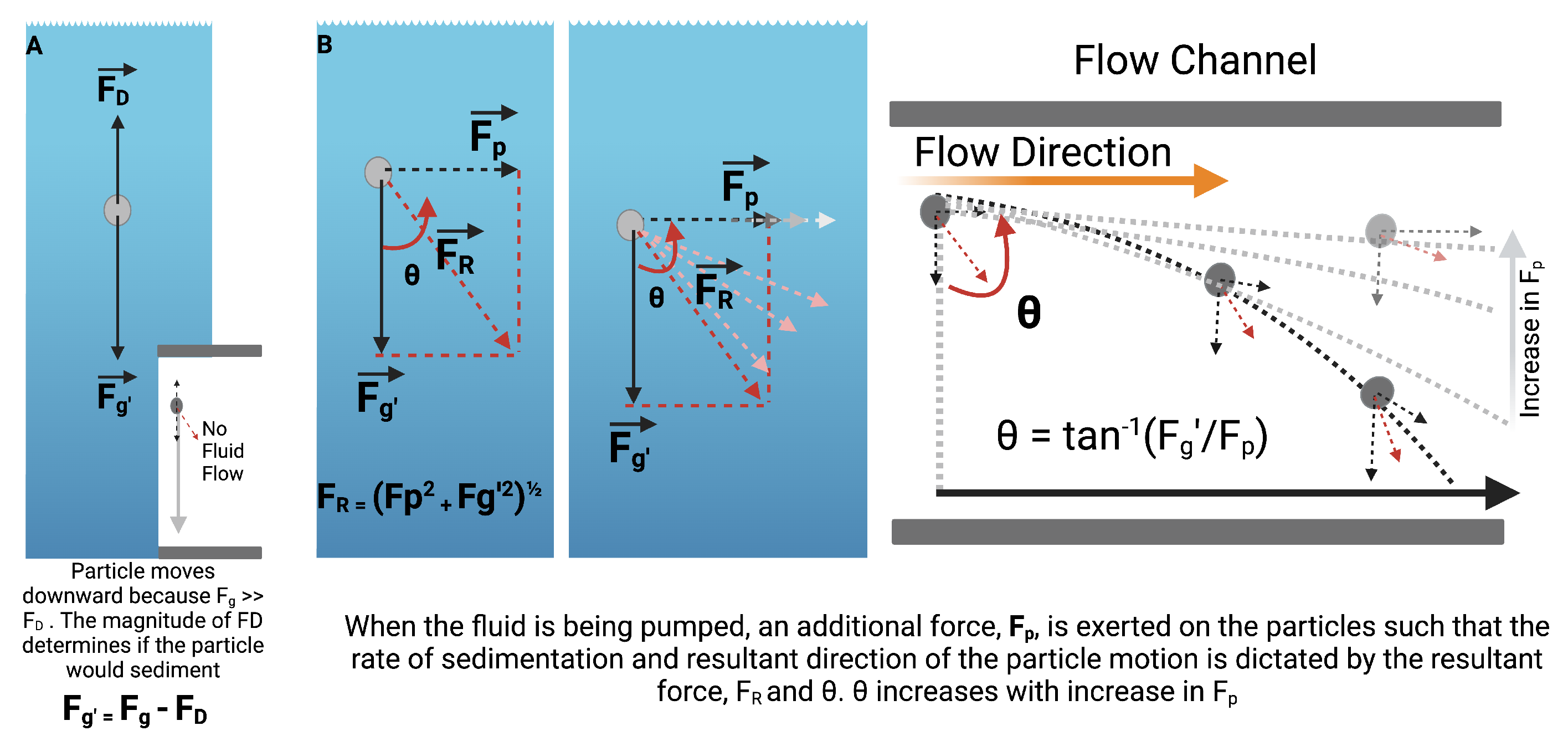
2.2. Hydrodynamic Interruption of Sedimentation
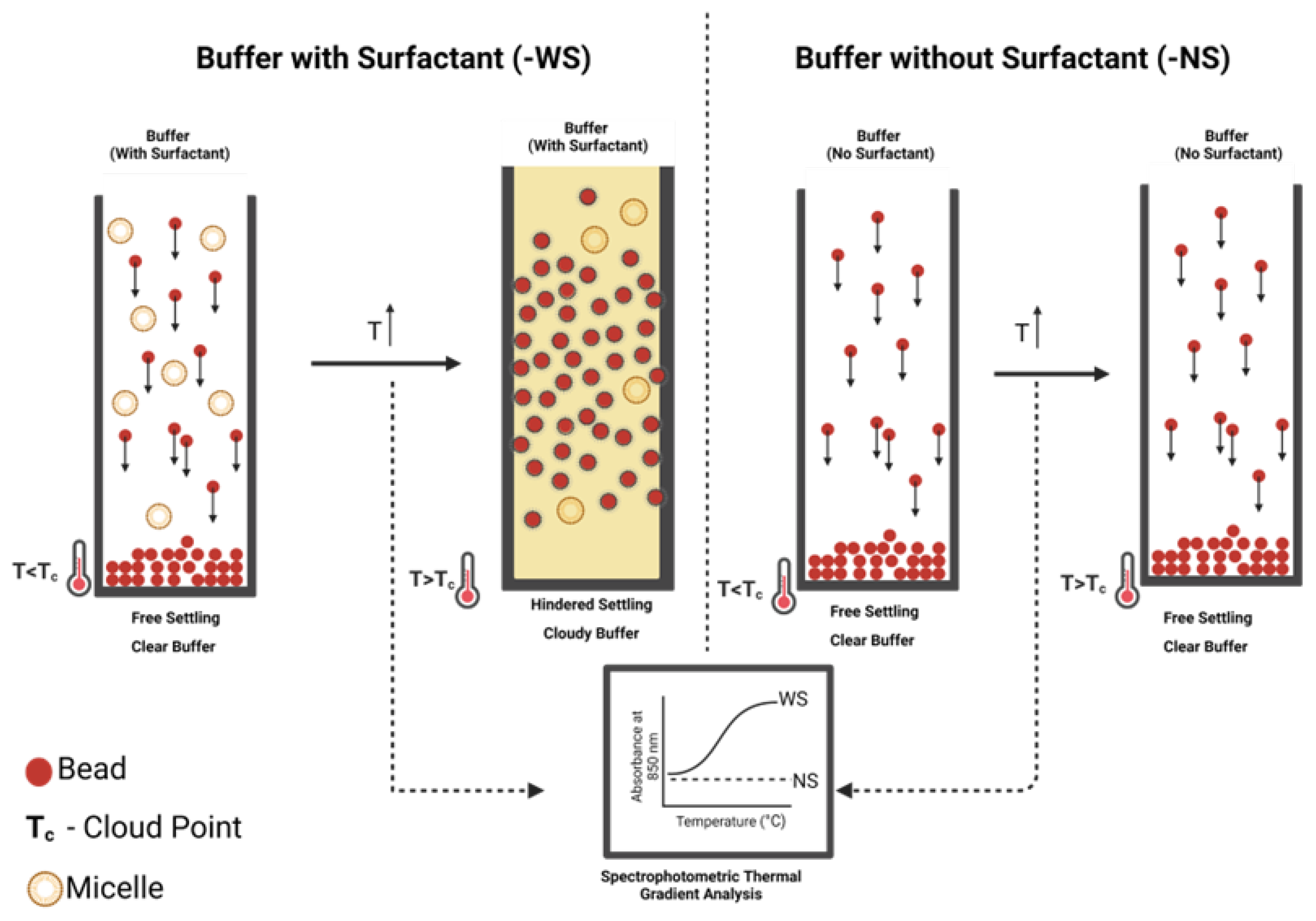
2.3. Induced Hindered Settling (i-HS)
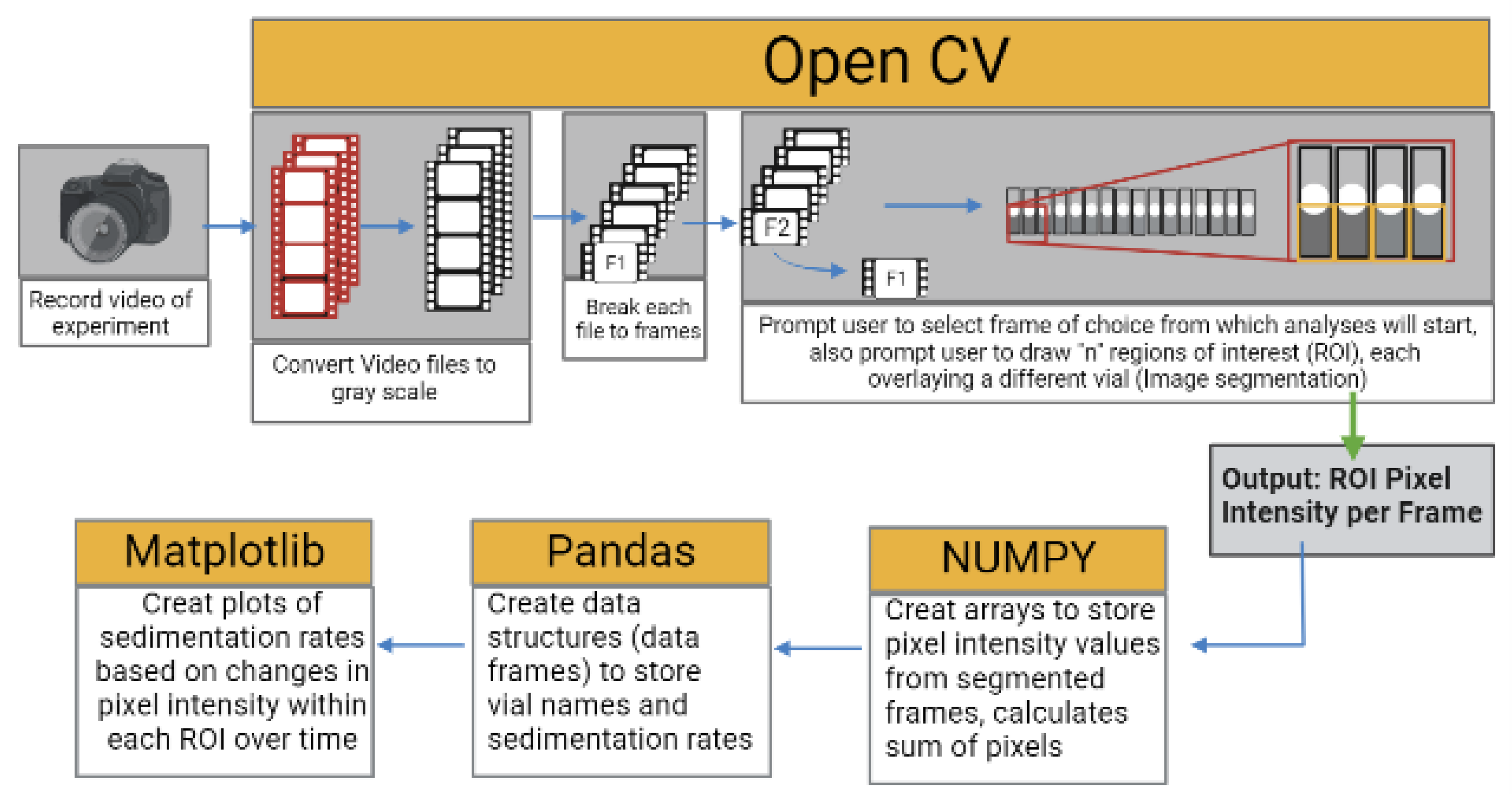
2.4. Data Acquisition and Analysis
3. Results
3.1. Fabrication of Particle Metering Chip
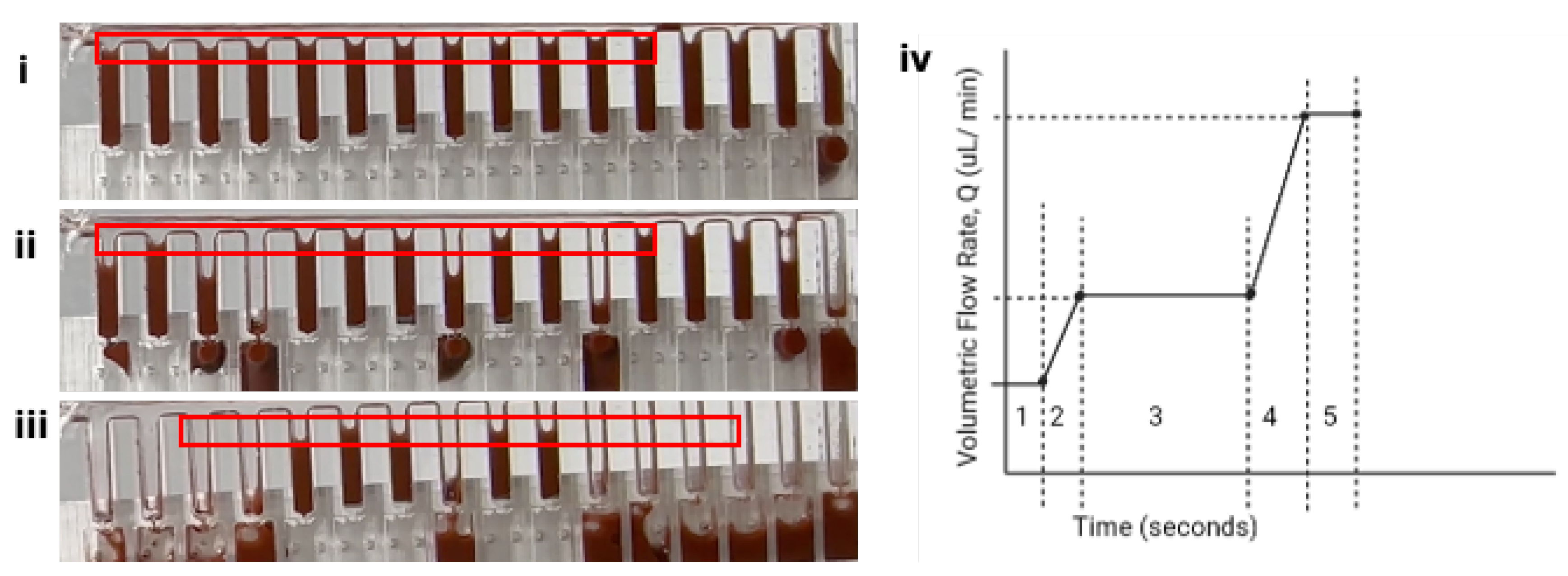
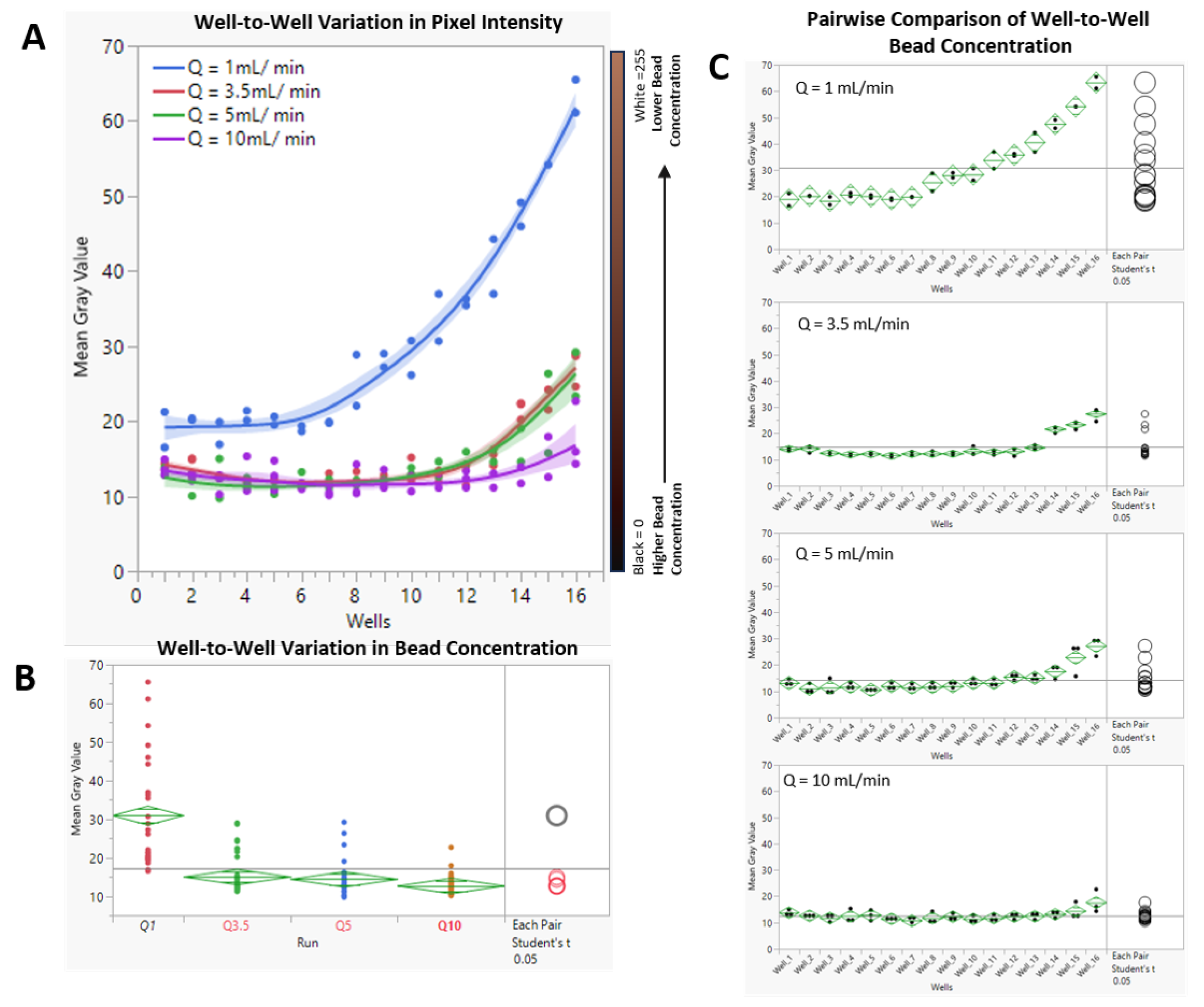
3.2. Hydrodynamic Interruption of Sedimentation
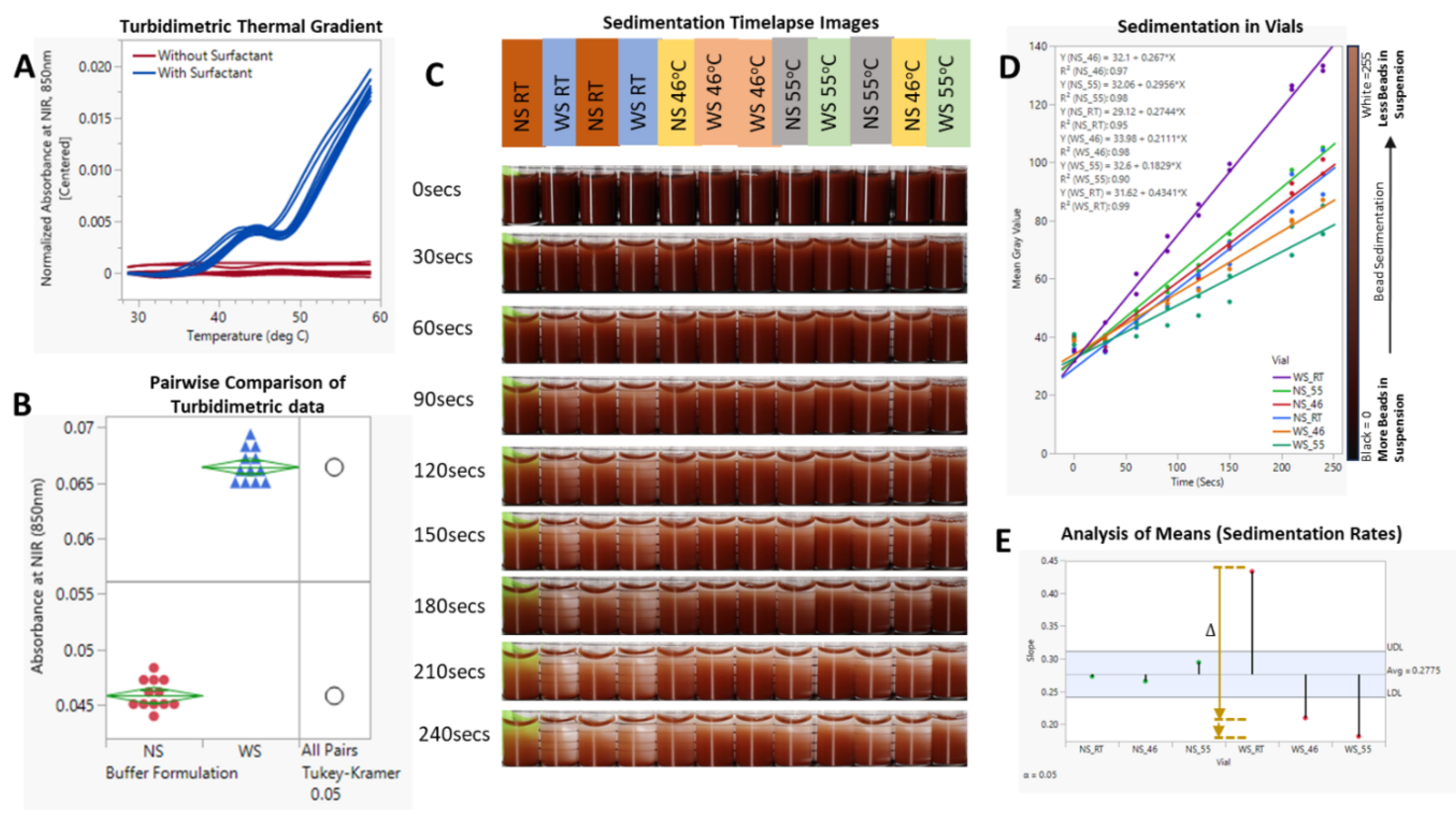
3.3. Sedimentation Offset Via Induced Hindered Settling
4. Discussion
4.1. Hydrodynamic Interruption of Particle Sedimentation
4.2. Induced Hindered Settling
5. Conclusions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iwai, K.; Sochol, D.R.; Lin, L. A bead-in-droplet solution exchange system via continuous flow microfluidic railing. 2013 IEEE 26th International Conference on Micro Electro Mechanical Systems (MEMS) 2013, pp. 1203–1206.
- Kim, H.; Choi, I.H.; Lee, S.; Won, D.J.; Oh, Y.S.; Kwon, D.; Sung, H.J.; Jeon, S.; Kim, J. Deterministic bead-in-droplet ejection utilizing an integrated plug-in bead dispenser for single bead-based applications. Scientific Reports 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Price, A.K.; Macconnell, A.B.; Paegel, B.M. Microfluidic bead suspension hopper. Analytical Chemistry 2014, 86, 5039–5044. [Google Scholar] [CrossRef] [PubMed]
- Myers, K.K.; Herich, J.P.; Chavez, J.E.; Berkey, K.G.; Loi, A.J.; Cleveland, P.H. A Novel Method to Gently Mix and Uniformly Suspend Particulates for Automated Assays. SLAS Technology 2021, 26, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Lien, K.Y.; Wu, J.J.; Lee, G.B. A magnetic bead-based assay for the rapid detection of methicillin-resistant Staphylococcus aureus by using a microfluidic system with integrated loop-mediated isothermal amplification. Lab on a Chip 2011, 11, 1521–1531. [Google Scholar] [CrossRef] [PubMed]
- Anyaduba, T.D.; Otoo, J.A.; Schlappi, T.S. Picoliter Droplet Generation and Dense Bead-in-Droplet Encapsulation via Microfluidic Devices Fabricated via 3D Printed Molds. Micromachines 2022, 13, 1946. [Google Scholar] [CrossRef] [PubMed]
- Sassolas, A.; Hayat, A.; Marty, J.L. Immobilization of enzymes on magnetic beads through affinity interactions. Methods in Molecular Biology 2013, 1051, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K.; Luong, J.H. Antibody Immobilization and Surface Functionalization Chemistries for Immunodiagnostics; Elsevier, 2018; pp. 19–46. [Google Scholar] [CrossRef]
- Poles, M.; Meggiolaro, A.; Cremaschini, S.; Marinello, F.; Filippi, D.; Pierno, M.; Mistura, G.; Ferraro, D. Shaking Device for Homogeneous Dispersion of Magnetic Beads in Droplet Microfluidics. Sensors 2023, 23. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, U.; Jain, S.K.; Sen, A.K. Particle encapsulation in aqueous ferrofluid drops and sorting of particle-encapsulating drops from empty drops using a magnetic field. Soft Matter 2021, 17, 6020–6028. [Google Scholar] [CrossRef] [PubMed]
- Anyaduba, T. Primer Payload System for Higher-Order Multiplex LAMP: Design and Development of Unit Processes. KGI Theses and Dissertations 2021. [Google Scholar]
- Shintaku, H.; Kuwabara, T.; Kawano, S.; Suzuki, T.; Kanno, I.; Kotera, H. Micro cell encapsulation and its hydrogel-beads production using microfluidic device. Microsystem Technologies 2007, 13, 951–958. [Google Scholar] [CrossRef]
- Klein, A.M.; Mazutis, L.; Akartuna, I.; Tallapragada, N.; Veres, A.; Li, V.; Peshkin, L.; Weitz, D.A.; Kirschner, M.W. Droplet barcoding for single-cell transcriptomics applied to embryonic stem cells. Cell 2015, 161, 1187–1201. [Google Scholar] [CrossRef] [PubMed]
- Zilionis, R.; Nainys, J.; Veres, A.; Savova, V.; Zemmour, D.; Klein, A.M.; Mazutis, L. Single-cell barcoding and sequencing using droplet microfluidics. Nature Protocols 2017, 12, 44–73. [Google Scholar] [CrossRef] [PubMed]
- Clark, I.C.; Abate, A.R. Microfluidic bead encapsulation above 20 kHz with triggered drop formation. Lab on a Chip 2018, 18, 3598–3605. [Google Scholar] [CrossRef] [PubMed]
- Ekanayake, N.I.; Berry, J.D.; Stickland, A.D.; Dunstan, D.E.; Muir, I.L.; Dower, S.K.; Harvie, D.J. Lift and drag forces acting on a particle moving with zero slip in a linear shear flow near a wall. Journal of Fluid Mechanics 2020, 904. [Google Scholar] [CrossRef]
- Riuxardson, J.F.; Zaki, W.N. The sedimentation of a suspension of uniform spheres under conditions of viscous flow.
- Zhu, Z.; Wang, H.; Peng, D.; Dou, J. Modelling the hindered settling velocity of a falling particle in a particle-fluid mixture by the Tsallis entropy theory. Entropy 2019, 21. [Google Scholar] [CrossRef] [PubMed]
- Ghaedi, M.; Shokrollahi, A.; Mehrnoosh, R.; Hossaini, O.; Soylak, M. Combination of cloud point extraction and flame atomic absorption spectrometry for preconcentration and determination of trace iron in environmental and biological samples. Central European Journal of Chemistry 2008, 6, 488–496. [Google Scholar] [CrossRef]
- Mariusz, R.; Ewelina, L. MODELING OF THE SEDIMENTATION PROCESS OF MONODISPERSE SUSPENSION. International Journal of Computational Methods and Experimental Measurements 2022, 10, 50–61. [Google Scholar] [CrossRef]
- Miyake, M.; Yamashita, Y. Molecular Structure and Phase Behavior of Surfactants. Cosmetic Science and Technology: Theoretical Principles and Applications 2017, pp. 389–414. [CrossRef]
- Cloud Point of Non-Ionic Surfactants â METTLER TOLEDO.
- Nazar, M.F.; Shah, S.S.; Eastoe, J.; Khan, A.M.; Shah, A. Separation and recycling of nanoparticles using cloud point extraction with non-ionic surfactant mixtures. Journal of Colloid and Interface Science 2011, 363, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Duester, L.; Fabricius, A.L.; Jakobtorweihen, S.; Philippe, A.; Weigl, F.; Wimmer, A.; Schuster, M.; Nazar, M.F. Can cloud point-based enrichment, preservation, and detection methods help to bridge gaps in aquatic nanometrology? Analytical and Bioanalytical Chemistry 2016, 408, 7551–7557. [Google Scholar] [CrossRef] [PubMed]
- Na, G.C.; Yuan, B.O.; Stevens, H.J.; Weekley, B.S.; Rajagopalan, N. Cloud point of nonionic surfactants: Modulation with pharmaceutical excipients. Pharmaceutical Research 1999, 16, 562–568. [Google Scholar] [CrossRef]
- Al-Saadi, M.R.; Al-Garawi, Z.S.; Thani, M.Z. Promising technique, cloud point extraction: Technology & applications. Journal of Physics: Conference Series, 2021. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




