1. Introduction
The life expectancy of SARS-CoV-2 survivors remains a subject of critical inquiry, affected by various factors such as age, pre-existing health conditions, and the severity of the initial infection [
1]. While much attention has been directed towards understanding the acute phase of COVID-19, there is a growing recognition of the long-term health implications for those who have recovered. It has become increasingly evident that the impact of SARS-CoV-2 extends beyond the immediate symptoms experienced during the acute phase of the disease. The COVID-19 pandemic has markedly elevated global mortality [
2], with a significant proportion of COVID-19 survivors facing lingering health challenges and increased morbidity in the months and years following recovery from acute infection.
Among those at risk for severe outcomes, older individuals with pre-existing health conditions such as cardiovascular disease, diabetes, and respiratory disorders are particularly vulnerable [
3,
4]. Studies have shown that male sex and ethnicity, specifically Black or South Asian backgrounds, as well as the severity of the initial infection, correlate with higher rates of COVID-19-related death. These factors contribute to a complex interplay of risks that affect the long-term prognosis of COVID-19 survivors.
Despite the growing body of research on acute COVID-19, long-term mortality among COVID-19 survivors is poorly understood, with specific underlying mechanisms remaining elusive. This study seeks to address this knowledge gap by examining mortality rates among patients who have survived the acute phase of the illness and by identifying specific urinary peptides that may be associated with future death. Urinary peptides are short sequences of amino acids that can reflect pathological processes in the body, and their identification could potentially serve as biomarkers for predicting long-term health outcomes in COVID-19 survivors.
By investigating mortality rates among patients who have survived the acute phase of COVID-19, we aim to identify potential predictors of long-term mortality, including demographic factors, clinical characteristics, and laboratory parameters. In a further approach, we focus on the role of urinary peptides as biomarkers of future death, leveraging recent advances in proteomic technology to explore their predictive value. Detection of urinary peptides associated with future mortality could have significant implications for risk stratification and personalized care among COVID-19 survivors.
Incorporating these biomarkers into clinical practice could allow healthcare providers to better identify individuals at high risk of adverse outcomes and tailor interventions accordingly. This would ultimately improve long-term outcomes and quality of life for survivors of COVID-19, contributing to more effective management of the ongoing pandemic. By integrating advanced biomarker detection with traditional clinical assessments, we can enhance our ability to predict, monitor, and manage the long-term health impacts of COVID-19, ensuring that survivors receive the comprehensive care they need to mitigate future health risks.
2. Materials and Methods
Study Population
Survival data of 651 unvaccinated patients were gathered from the "Prospective Validation of a Proteomic Urine Test for Early and Accurate Prognosis of Critical Course Complications in Patients with SARS-CoV-2 Infection" (CRIT-COV-U) study, spanning until December 2023 across six countries and 9 centers (
supplementary table 1) [
5]. These patients were enrolled during the initial and subsequent waves of the pandemic in 2020-2021, predominantly infected with the wild-type virus, and had survived the acute phase of COVID-19. This cohort was stratified into discovery (n=324) and validation (n=327) sets through random partitioning. The project complied with the Helsinki declaration. The Ethics Committee of the German-Saxonian Board of Physicians (Dresden, Germany; number EK-BR-70/23-1) and the Institutional Review Boards of the recruiting sites provided ethical approval. To assess the impact of age on mortality within this cohort, comparisons were made against age- and sex-matched data from non-SARS-CoV-2 infected individuals (n=5192), sourced from the Human Urinary Database [
6].
Urinary Peptidomics
Data were extracted from the Human Urinary Proteome Database, which contains datasets acquired using capillary electrophoresis coupled with mass spectrometry (for details on the CE-MS analysis, please see [
7]) as described previously [
6]. Data were evaluated using MosaFinder software and normalized based on the abundance of 29 collagen peptides [
8]. Of the 5071 sequenced peptides identified to date, only those present in at least 50% of the entire discovery cohort of 324 individuals (923 peptides) were retained for further analyses.
Statistical Analysis
As descriptive statistics for the samples, shown in
Table 1, median and interquartile range (IQR) were used for continuous variables and absolute (N) and relative frequencies (%) for categorical variables. Hypotheses of no differences in scale or distribution of patient characteristics between the death and non-death groups were tested with Wilcoxon–Mann–Whitney tests for continuous and with χ2-homogeneity tests for categorical variables. Adjustment for multiple testing was implemented according to Benjamini Hochberg [
9]. Kernel density estimates of the distribution of the scores split by mortality groups are displayed in
Figure 2B. Mortality per person-time stratified by age and DP201 groups, seen in
Figure 2 A, C, and D, is estimated as the ratio of the number of the deceased to the sum of all patients` observation times within each group scaled to 100 person-years. The corresponding mortality probabilities and their 95% confidence intervals (CI) for each group represent estimates from a logistic regression including all 651 patients.
Classifier Development
A classifier combining multiple features (peptides) into a single variable was developed using support vector machine modelling as described [
10]. All peptides demonstrating a significant difference (adjusted for the false-discovery rate set at 0.05) between cases and controls were included in the classifier. Classification is performed by determining the Euclidian distance (classification score) of the vector to a separating hyperplane. The optimal parameters for C (cost of misclassification) and gamma (flexibility of the separating hyperplane) are determined via leave one out cross-validation error estimation, as described in more detail in [
11].
3. Results
3.1. Assessment of Mortality in Acute COVID-19 Survivors
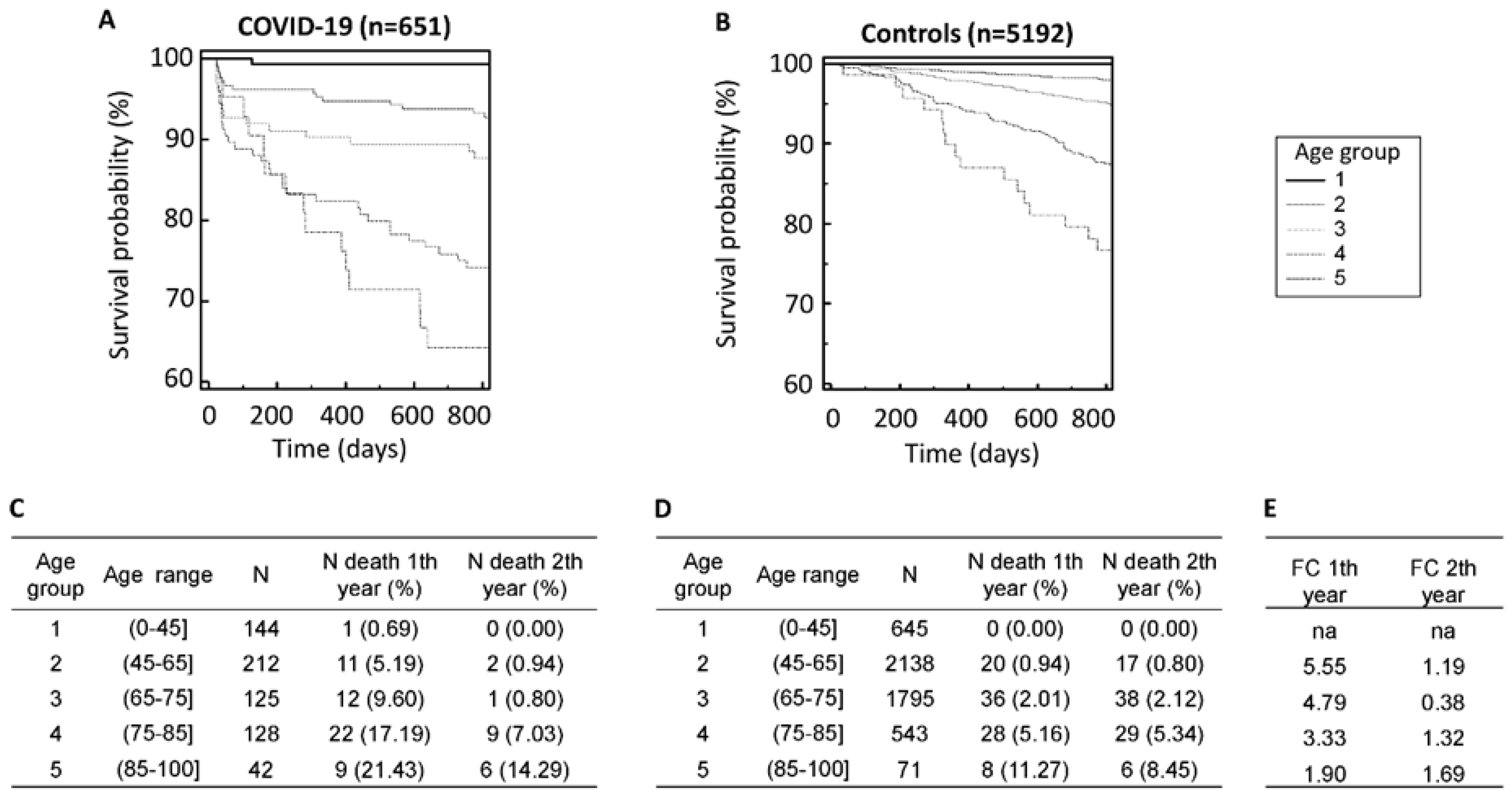
Of the 893 patients from the CRIT-COV-U study surviving the acute COVID-19, follow-up data from 651 patients could be obtained (
Table 1). At the time of inclusion in the CRIT-COV-U study (and urine sampling), the median age of the 651 patients was 63 years (IQR: 48-76)), with a male predominance of 53.5%. The median body mass index (BMI) recorded was 27.0 (IQR: 24.4-30.3) kg/m
2, and the estimated glomerular filtration rate (eGFR) was 90.0 (IQR: 70-111) ml/min/1.73m². The majority of patients, 56.4%, had no recorded comorbidities. The entry WHO scores were 1–3 in 311 (48%) participants, 4–5 in 317 (49%) participants, and 6 in 23 (4%) participants. Throughout the follow-up period, spanning a median of 2.92 years (IQR: 2.67-3.09), pertinent data was collected to assess mortality outcomes in survivors of the acute phase of COVID-19.
Among the 651 patients who survived the acute phase of COVID-19 and could be followed up on, 91 individuals (13.98%) succumbed during the follow-up duration, with 55 (8.45%) of these fatalities occurring within the first year post-infection. In stark contrast, among the age and sex-matched controls totaling 5192 individuals, a markedly lower proportion of 92 (1.77%) deaths were recorded within the same timeframe. Notably, mortality displayed an age-dependent pattern across both cohorts, with significantly elevated rates observed among those who had survived COVID-19 compared to their non-infected counterparts (
Figure 1A–D). Specifically, within the first year post-infection, mortality rates surged up to 4.7 times higher in patients younger than 65 years compared to the non-infected controls.
3.2. Identification of Biomarkers Associated with Post-Acute COVID-19 Mortality
For the identification of biomarkers potentially associated with mortality after surviving the initial acute phase of COVID-19, the previously acquired urinary peptidomics datasets from baseline samples of COVID-19-diagnosed patients within the CRIT-COV-U study were utilized. These datasets were stratified into discovery (n=324) and validation cohort (n=327) sets through random partitioning. Urinary peptides potentially associated with mortality were defined by applying the Mann-Whitney test to compare 44 deceased and 280 surviving patients within the discovery set. Subsequently, adjustments for multiple testing were implemented to ensure statistical robustness.
The analysis of urinary peptidome datasets within the discovery set enabled the identification of 201 peptides (listed in
supplementary table 2) as significantly associated with mortality when comparing deceased and surviving patients. These peptides encompassed upregulated fragments of albumin, alpha-2-HS-glycoprotein, apolipoprotein A-I, and beta-2-microglobulin, alongside with downregulated fragments of CD99 antigen, various collagens, fibrinogen alpha, polymeric immunoglobulin receptor, sodium/potassium-transporting ATPase, and uromodulin. Among these peptides, 14 overlapped with the previously established Cov50 classifier designed for prognosticating unfavorable COVID-19 outcomes during the acute phase[
12].
3.3. Establishment and Validating a Classifier Predicting Post-Acute COVID-19 Mortality
The 201 peptides significantly associated with
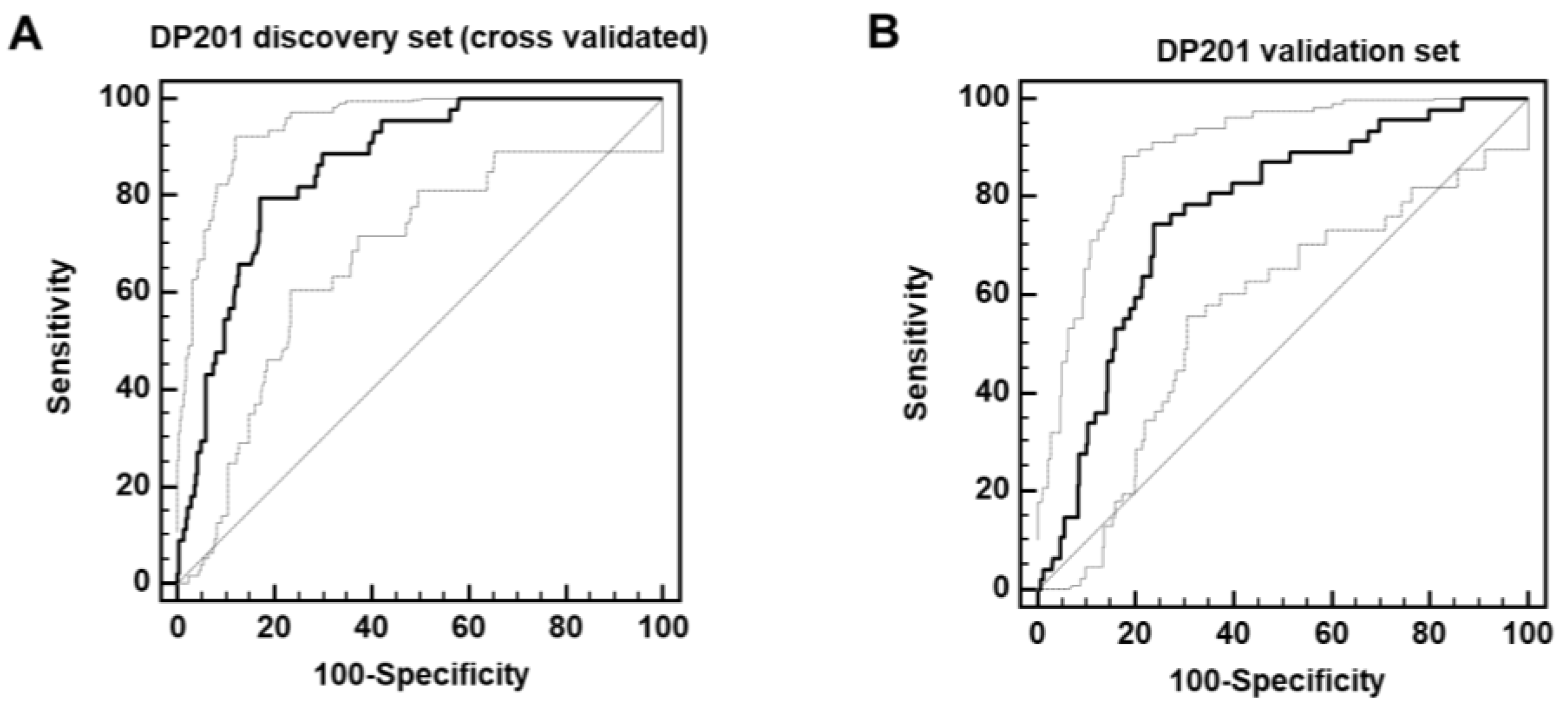
post-acute COVID-19 mortality (PACM) were combined to form a support-vector-machine-based classifier (DP201). This classifier enabled separating the discovery set with 80% sensitivity and 83% specificity upon complete take-one-out cross validation (area under the curve (AUC) = 0.86,
Figure 2A). Subsequently, this classifier was applied to the independent validation cohort, consisting of 47 deceased and 280 surviving patients, what resulted in significant separation of the groups with an AUC of 0.78 as shown in
Figure 2B.
Figure 2.
ROC curves for the classification of the deceased and surviving patients in the complete take-one-out cross-validated discovery cohort (A) and independent validation cohort (B).
Figure 2.
ROC curves for the classification of the deceased and surviving patients in the complete take-one-out cross-validated discovery cohort (A) and independent validation cohort (B).
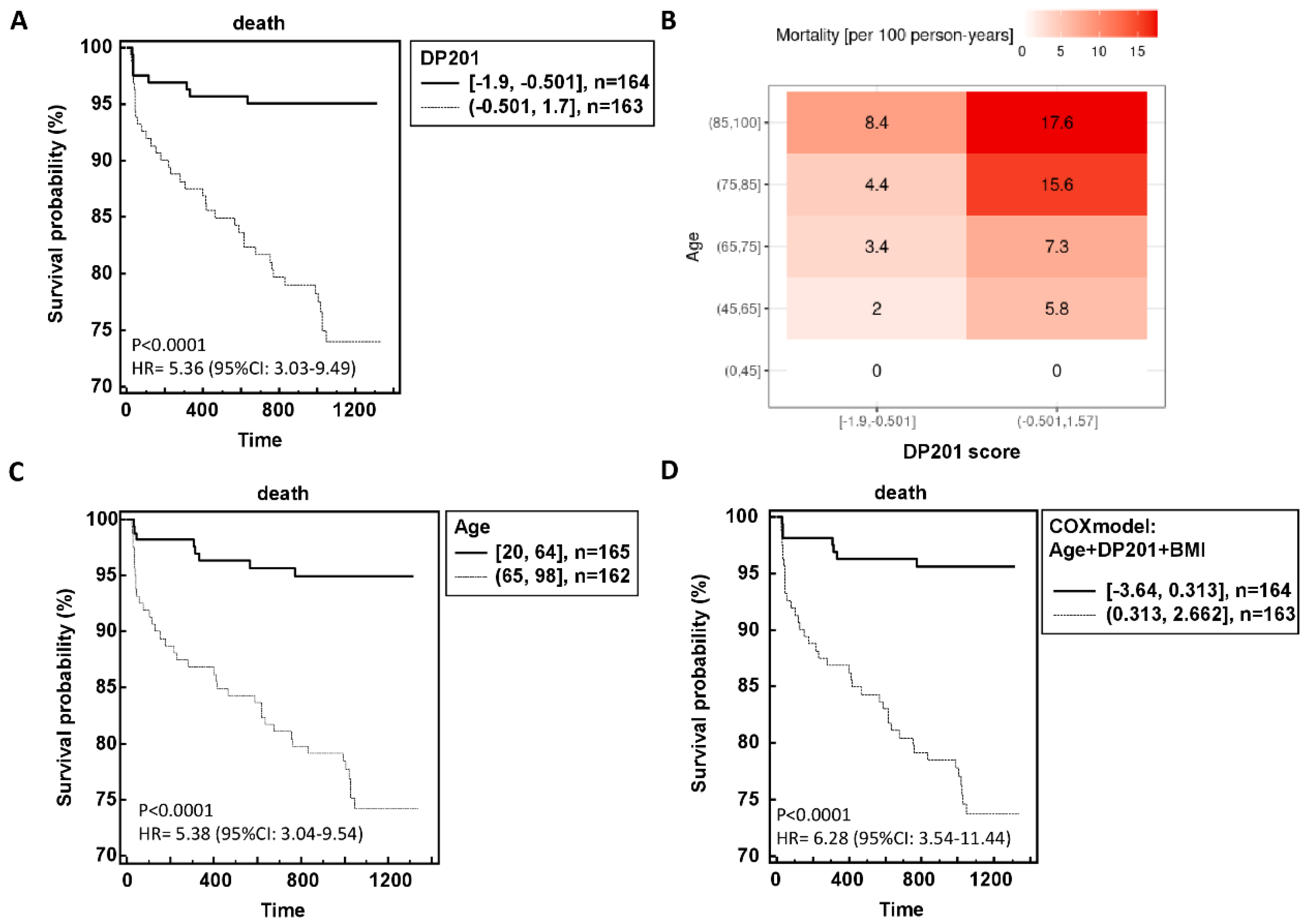
The resultant outcomes in relation to follow-up time are depicted in Figure 3A,B, illustrating a clear correlation between higher classification scores and heightened mortality risk. Further Cox regression analysis revealed that age, BMI, and DP201 were significantly associated with mortality, while sex, number of comorbidities, eGFR, and COVID-19 WHO score did not exhibit statistical significance. Integration of these three parameters into a Cox model yielded a hazard ratio of 6.28 (95%CI: 3.54-11.44) compared to age and DP201 alone (Figure 3C,D).
Figure 3.
Performance of the urinary peptide-based death prediction classifier in the independent validation data. The risk of death is significant dependent of the DP201 score (A) although the age dependency can still be observed (B). The hazard ration for survival probability DP201 classifier (A), and age (C) could be increased using a model including DP201, age and BMI (D).
Figure 3.
Performance of the urinary peptide-based death prediction classifier in the independent validation data. The risk of death is significant dependent of the DP201 score (A) although the age dependency can still be observed (B). The hazard ration for survival probability DP201 classifier (A), and age (C) could be increased using a model including DP201, age and BMI (D).
4. Discussion
Our findings first and foremost demonstrate that COVID-19 is associated with a highly significantly increased risk of mortality even after the acute phase of the disease. To date, this issue was apparently not well covered by the studies, which either investigated the immediate outcome of the acute infection, or the long-term effect in the context of post-acute sequelae of SARS-CoV-2 infection (PASC). However, PASC was typically only assessed in patients still alive. Our data indicate that a significant number of patients may have died as a result of the consequences associated with the previous SARS-CoV-2 infection. The mortality was significantly higher than in age and sex-matched controls and the deaths could be potentially labeled as PASC-related deaths.
In a retrospective analysis of 13638 patients with a COVID-19 hospitalization documented a significantly increased risk for future mortality. Increased 12-month mortality was observed in those patients with severe COVID-19 compared to COVID-19 negative patients, which was concluded to be an under-investigated sequela of COVID-19 [
13].
Another large retrospective analysis of long-term outcomes of 22.571 adult patients hospitalized due to COVID-19 in Austria in the year 2020 found an increased mortality compared to 217295 propensity score matched controls (Graf, Wendt et al., unpublished). Similar to our results, the difference between patients and controls remained significant in the younger age groups (41-64 years and 65-74 years, p<0.001) but not in the oldest age group (p=0.078). (Graf, Wendt et al., unpublished).
An investigation on long-term risks in over 800.000 COVID-19 patients compared the risk of post-discharge death with 56.409 Influenza patients as a historical control. Patients who were discharged alive from a COVID-19 related hospitalization admission had nearly twice the risk of post discharge death compared to historical controls admitted to hospital with influenza [
14].
Data from a large study with 47,780 english patients discharged alive after COVID-19 hospitalisation showed an increased risk of readmission and mortality during a follow-up of 140 days. The post-discharge mortality risk was eight times greater than in matched controls and with the largest differences in the age group < 70 years [
15].
A report from the US investigated the mortality after recovery from the initial episode of COVID-19 and reported a significantly higher 24-month adjusted all-cause mortality risk for patients with severe COVID-19 compared to uninfected comparators (HR 2.01). The risk of excess death was highest during days 0 to 90 after infection (aHR 6.36) and still elevated during days 91 to 180 (aHR 1.18). Beyond 180 days after infection there was no excess mortality during the next 1.5 years [
16].
SARS-CoV-2 infection obviously poses a persistent threat to individuals even beyond the acute phase. Importantly, among those who successfully navigate the acute phase of COVID-19, the risk of mortality escalates significantly during the subsequent follow-up period. Particularly noteworthy is the observation that within the first year following infection, mortality rates surge dramatically among individuals who have survived the acute phase of the illness when compared to a non-infected control cohort. What is striking is that this increase in mortality risk is most pronounced among younger individuals, highlighting a concerning trend that defies conventional assumptions regarding age-related vulnerability to severe outcomes. The most abundant significantly changed peptides in patients experiencing death during follow-up are derived from β2-microglobulin (B2M). Higher B2M serum concentrations are associated with higher mortality in the general population, non-dialyzed chronic kidney disease patients and patients receiving hemodialysis (HD) [
17].
The data also show a consistent higher level of uromodulin peptides in patients without event. This is in very good agreement with a recent study presented by Vasquez-Rios and colleagues, where increased levels of uromodulin were found associated with a lower risk of cardiovascular death [
18]
Thymosin beta4 (TB4) is an abundant actin sequestering protein that has been described in the context of multiple (patho)physiological processes, among others in wound healing, angiogenesis, migration, to name just a few. It has also been described as being increased in kidney disease, with highest levels detected in patients with end stage kidney disease [
19]. Drum and colleagues found TB4 significantly increased in women with heart failure with preserved ejection fraction, and associated with mortality. The strict association with female sex may be the result of TB4 being an X-linked gene product, which consequently is also found increased in women [
20].
The Sodium/potassium-transporting ATPase subunit gamma (FYXD2) is found highly expressed in the kidney distal tubulus. In previous studies reduced abundance of peptides derived from FYXD2 has been observed associated with progression of CKD, specifically of IgAN [
21].
Reduced levels of a peptide derived from S100A9, consequently reduced degradation of this protein likely results in increased levels of calprotectin, which was described associated with increased risk of mortality [
22].
Reduced abundance of peptides from the polymeric immunoglobulin (PIGR) receptor was previously found associated with acute COVID-19 mortality [
23]. Similarly, increased complement activation, which result in an increase of complement derived urine peptides, was also described associated with increased COVID-19 mortality [
24].
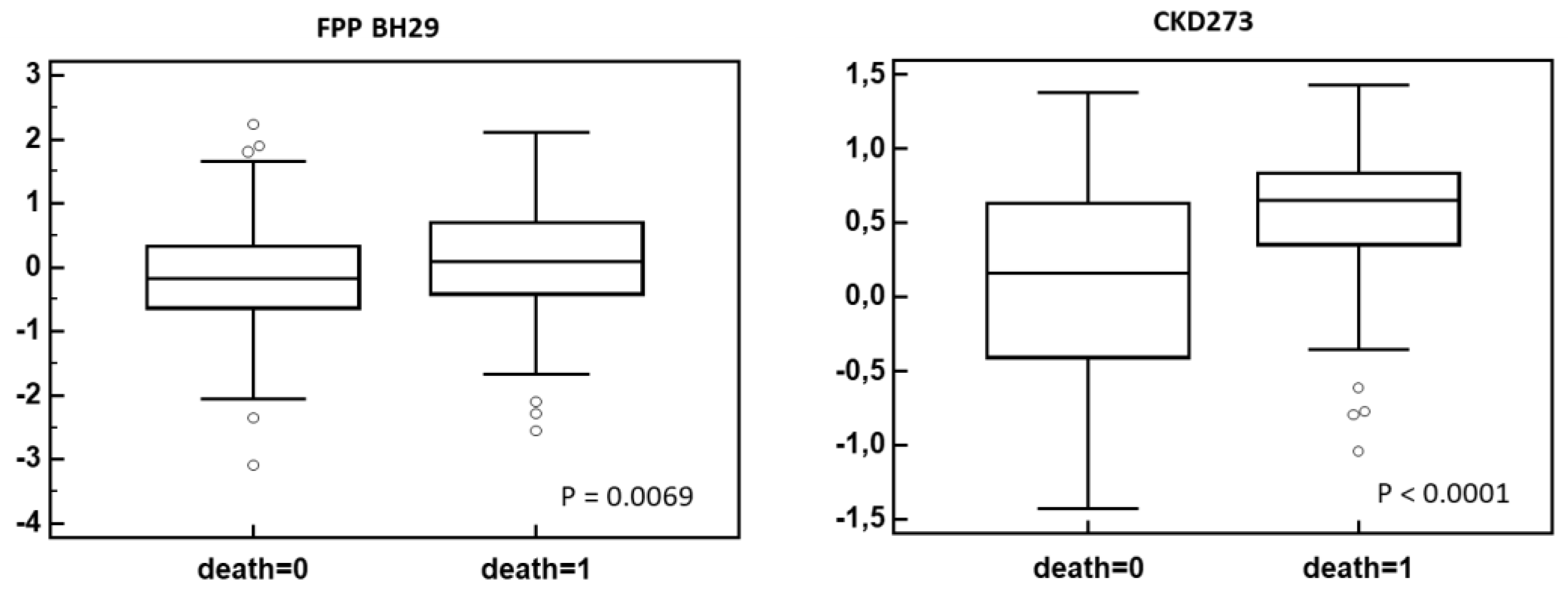
The most pronounced effect is on collagen fragments, with both, increase and decrease of specific collagen-derived peptides being observed. A change in collagen peptides has been described for multiple diseases and was also found associated with mortality, including mortality in the context of COVID-19 [
25,
26]. As observed here, both, up and downregulation of collagen fragments were observed. This was interpreted as disruption of collagen degradation, leading to increased fibrosis. Two urinary peptide-based classifiers, CKD273 [
27] and FPP_BH29 [
28], both based on multiple specific collagen peptides, were presented as highly significantly associated with fibrosis. To investigate if the changes observed in this study are associated with increased fibrosis, we applied these two classifiers onto the data and compared the scoring in the survivors vs. patients experiencing death in follow-up. As shown in
Figure 4, we observe a highly significantly increase in both scores, indicating increased fibrosis, in the case group.
Also, the consistent reduction in CD99 was observed in severe cases of COVID-19, where a significant reduction of CD99 also was found on the surface of peripheral blood lymphocytes [
29]. Based on the data the authors hypothesized that reduction of CD99 may have a negative impact on the endothelial barrier integrity, a well-known phenomenon in severe COVID-19.
The increase in urinary albumin, fetuin, and apolipoprotein A1 may all be consequences of a similar underlying mechanism: endothelial dysfunction resulting in a loss of functionality of the glomerular filtration barrier. In fact, increase of albuminuria is well known associated with increased risk of mortality.
Increase in alpha 1 antitrypsin, a major plasma inflammatory protein, was found associated with increased mortality in the Nagahama study based on 9682 subjects [
30]. This is in line with the observed increase associated with PACM in our study, which may be further exacerbated by the proteinuria, as mentioned above.
Furthermore, the identification of specific urinary peptides capable of predicting heightened mortality risk at the outset of SARS-CoV-2 infection underscores the intricate interplay between molecular biomarkers and clinical outcomes. These peptides serve as early indicators of the likelihood of mortality, providing valuable insights into the underlying pathophysiological mechanisms driving adverse outcomes in COVID-19 patients. By leveraging these predictive biomarkers, healthcare professionals can proactively identify individuals at elevated risk of mortality and implement targeted interventions aimed at mitigating this risk, thereby potentially altering the trajectory of the disease course, like already showed for chronic kidney and heart diseases [
31].
A shortcoming of the study may be that the patients investigated were not immunized against SARS-CoV-2, at the time of the initial study vaccine was generally not available. While mortality due to acute COVID-19 has been reduced dramatically as a result of immunization, it is not certain that immunity equally well protects from PACM, this needs to be investigated in a subsequent study.
5. Conclusions
Our findings underscore the multifaceted nature of SARS-CoV-2 infection, extending far beyond the acute phase and exerting a lasting impact on mortality outcomes. The acute phase of COVID-19 appears to initiate a complex disease trajectory in many survivors. Apparently, recovery from the acute phase does not necessarily equate to a return to pre-infection health. Instead, many survivors experience ongoing health challenges, often referred to as "long COVID" or post-acute sequelae of SARS-CoV-2 infection (PASC), which include persistent fatigue, cardiovascular complications, cognitive impairments, and other chronic conditions. Our study adds mortality risk in COVID-19 survivors to the list of symptoms. The risk is driven by the interplay between demographic factors (such as age), pre-existing health conditions (like cardiovascular disease, diabetes mellitus, and respiratory disorders), and molecular changes, detectable in the urinary peptidome. Understanding these interactions may help identifying individuals who are at higher risk of adverse outcomes, enabling more precise and effective healthcare interventions.
The identified predictive biomarkers represent a significant advancement in our ability to foresee and manage long-term health risks in COVID-19 survivors. These biomarkers offer a window into the biological processes that continue to affect patients long after the initial infection has resolved. The approach applied here not only enhances our understanding of the disease but also holds the promise of improving monitoring and treatment of COVID-19 survivors. By integrating predictive biomarkers into clinical practice, a more effective risk stratification and personalized interventions can be achieved. Personalized interventions, informed by a patient’s unique biomarker profile [
31], can improve management strategies and optimize treatment plans, ultimately leading to better health outcomes.
This research lays the groundwork for improved clinical management and patient outcomes by providing a robust framework for predicting and mitigating long-term risks associated with SARS-CoV-2 infection, consequently offering a path towards more proactive and patient-centered care. By focusing on the long-term health of COVID-19 survivors, their quality of life can be enhanced and mortality rates could be reduced.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org, Table S1: Distribution of patients across participating enters; Table S2: urinary peptides significantly associated with PACM.
Author Contributions
Conceptualization, JS and RW; methodology, JS, RW, FK, and JB; software, HM; validation, JS,RW; formal analysis, FK; investigation and resources, JS, RW, FK, MB, BP, ED, AM, DG, GS, MSL, MR, ŁF, MDZ, MH; resources, data curation, JS, FK; writing—original draft preparation, JS, RW, JB; writing—review and editing, JS, RW, FK, BP, HM, JB; visualization, FK, JS; funding acquisition, JS, RW, MB, AM, BP, HM, MH, JB. All authors have read and agreed to the published version of the manuscript.
Funding
This project was supported by Federal Ministry of Health (BMG) via grant number 25323FSB114; by the German ministry for education and science (BMBF), via grant 01KU2309; by the Swedish governmental agency for innovation systems (Vinnova), via grant 2022-00542; by National Centre for Research and Development (Narodowe Centrum Badań i Rozwoju), via grant number: PerMed/V/162/UriCov/2023; by Austrian Science Fund (FWF) via Project number I 6464, Grant-DOI 10.55776/I6464; and by the French National Research Agency – Agence Nationale de la Recherche (ANR), under the grant ANR-22-PERM-0014 and in part by Austrian Science Fund (FWF) via Project number I 6471, Grant-DOI 10.55776/I6471 under the frame of ERA PerMed.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) the German-Saxonian Board of Physicians (Dresden, Germany; number EK-BR-70/23-1) and the Institutional Review Boards of the recruiting sites.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Acknowledgments
UriCoV Working Group: Justyna Siwy, Mosaiques Diagnostics GmbH, Hannover, Germany; Ralph Wendt, Department of Nephrology, St. Georg Hospital, Leipzig, Germany; Joachim Beige, Division of Nephrology, St. Georg Hospital, Leipzig, Department of Internal Medicine II, Martin-Luther-University Halle-Wittenberg, Halle, Germany and Kuratorium for Dialysis and Transplantation (KfH) Leipzig, Leipzig, Germany, Miroslaw Banasik, Department of Nephrology and Transplantation Medicine, Wrocław Medical University, Wroclaw, Poland; Björn Peters, Department of Molecular and Clinical Medicine, Institute of Medicine, the Sahlgrenska Academy at University of Gothenburg, Gothenburg, Sweden and Department of Nephrology, Skaraborg Hospital, Skövde, Sweden; Emmanuel Dudoignon, Hospital Saint Louis-Lariboisière, Paris, France; Dilara Gülmez, Lenka Grula, Amelie Kurnikowski, Manfred Hecking, all from Department of Epidemiology, Medical University of Vienna, Vienna, Austria; Magdalena Krajewska, Justyna Zachciał, Dorota Bartoszek, Patryk Wawrzonkowski, Krzysztof Wiśnicki, from Department of Nephrology and Transplantation Medicine, Wrocław Medical University, Wroclaw, Poland; Emelie Sarenmalm, Region Västra Götaland, Skaraborg Hospital, Department of Infectious Diseases, Skövde, Sweden; Åsa Nilsson, Region Västra Götaland, Skaraborg Hospital, Research, Education, Development and Innovation Department, Skövde, Sweden; Goce Spasovski, University Sts. Cyril and Methodius, Skopje, Republic of North Macedonia; Mercedes Salgueira Lazo, Virgen Macarena Hospital and University of Seville, Sevill; Maria Isabel García Sánchez, Biobank Node at Virgen Macarena Hospital, Seville, integrated in the Spanish National Biobanks Network (PT23/00134); Marek W Rajzer from First Department of Cardiology, Interventional Electrocardiology and Arterial Hypertension, Jagiellonian University Medical College, Kraków, Poland; Beata Czerwieńska, from Department of Nephrology, Endocrinology and Metabolic Diseases, Medical University of Silesia, Katowice, Poland; Magdalena Dzitkowska – Zabielska from Faculty of Physical Education, Gdańsk University of Physical Education and Sport and Centre of Translational Medicine, Medical University of Gdańsk, Gdańsk, Poland; Łukasz Fuławka from Molecular Pathology Centre Cellgen, Wrocław, Poland; Elena Nowacki, University of Patients-Sorbonne University, Paris, France; Catherine Tourette-Turgis, University of Patients, research chair "Compétences & vulnérabilités" Sorbonne University, France; Morgane Michel, Université Paris Cité, ECEVE, UMR 1123, Inserm, Paris, France; Assistance Publique-Hôpitaux de Paris, Hôpital Robert Debré, Unité d'épidémiologie clinique, Paris, France.
Conflicts of Interest
H.M. is the co-founder and co-owner of Mosaiques Diagnostics. J.S. is employed by Mosaiques-Diagnostics GmbH. All other authors declare no conflicts of interest. Authors must identify and declare any personal circumstances or interest that may be perceived as inappropriately influencing the representation or interpretation of reported research results. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Zhang X, Wang F, Shen Y, Zhang X, Cen Y, Wang B, Zhao S, Zhou Y, Hu B, Wang M et al. Symptoms and Health Outcomes Among Survivors of COVID-19 Infection 1 Year After Discharge From Hospitals in Wuhan, China. JAMA Netw Open 2021; 4: e2127403.
- Global age-sex-specific mortality, life expectancy, and population estimates in 204 countries and territories and 811 subnational locations, 1950-2021, and the impact of the COVID-19 pandemic: a comprehensive demographic analysis for the Global Burden of Disease Study 2021. Lancet 2024; 403: 1989-2056.
- Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, Curtis HJ, Mehrkar A, Evans D, Inglesby P et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020; 584: 430-436.
- Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, Norman L, Holden KA, Read JM, Dondelinger F, Carson G et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ 2020; 369: m1985.
- Staessen JA, Wendt R, Yu YL, Kalbitz S, Thijs L, Siwy J, Raad J, Metzger J, Neuhaus B, Papkalla A et al. Predictive performance and clinical application of COV50, a urinary proteomic biomarker in early COVID-19 infection: a prospective multicentre cohort study. Lancet Digit Health 2022; 4: e727-e737.
- Latosinska A, Siwy J, Mischak H, Frantzi M. Peptidomics and proteomics based on CE-MS as a robust tool in clinical application: The past, the present, and the future. Electrophoresis 2019; 40: 2294-2308.
- Mavrogeorgis E, Mischak H, Latosinska A, Siwy J, Jankowski V, Jankowski J. Reproducibility Evaluation of Urinary Peptide Detection Using CE-MS. Molecules 2021; 26: 7260.
- Jantos-Siwy J, Schiffer E, Brand K, Schumann G, Rossing K, Delles C, Mischak H, Metzger J. Quantitative Urinary Proteome Analysis for Biomarker Evaluation in Chronic Kidney Disease. J Proteome Res 2009; 8: 268-281.
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Royal Stat Soc B (Methodological) 1995; 57: 125-133.
- Good DM, Zürbig P, Argiles A, Bauer HW, Behrens G, Coon JJ, Dakna M, Decramer S, Delles C, Dominiczak AF et al. Naturally occurring human urinary peptides for use in diagnosis of chronic kidney disease. Mol Cell Proteomics 2010; 9: 2424-2437.
- Farmakis D, Koeck T, Mullen W, Parissis J, Gogas BD, Nikolaou M, Lekakis J, Mischak H, Filippatos G. Urine proteome analysis in heart failure with reduced ejection fraction complicated by chronic kidney disease: feasibility, and clinical and pathogenetic correlates. Eur J Heart Fail 2016; 18: 822-829.
- Wendt R, Thijs L, Kalbitz S, Mischak H, Siwy J, Raad J, Metzger J, Neuhaus B, Leyen HV, Dudoignon E et al. A urinary peptidomic profile predicts outcome in SARS-CoV-2-infected patients. EClinicalMedicine 2021; 36: 100883.
- Mainous AG, III, Rooks BJ, Wu V, Orlando FA. COVID-19 Post-acute Sequelae Among Adults: 12 Month Mortality Risk. Front Med (Lausanne) 2021; 8: 778434.
- Oseran AS, Song Y, Xu J, Dahabreh IJ, Wadhera RK, de Lemos JA, Das SR, Sun T, Yeh RW, Kazi DS. Long term risk of death and readmission after hospital admission with covid-19 among older adults: retrospective cohort study. BMJ 2023; 382: e076222.
- Ayoubkhani D, Khunti K, Nafilyan V, Maddox T, Humberstone B, Diamond I, Banerjee A. Post-covid syndrome in individuals admitted to hospital with covid-19: retrospective cohort study. BMJ 2021; 372: n693.
- Iwashyna TJ, Seelye S, Berkowitz TS, Pura J, Bohnert ASB, Bowling CB, Boyko EJ, Hynes DM, Ioannou GN, Maciejewski ML et al. Late Mortality After COVID-19 Infection Among US Veterans vs Risk-Matched Comparators: A 2-Year Cohort Analysis. JAMA Intern Med 2023; 183: 1111-1119.
- Maruyama Y, Nakayama M, Abe M, Yokoo T, Minakuchi J, Nitta K. Association between serum beta2-microglobulin and mortality in Japanese peritoneal dialysis patients: A cohort study. PLoS One 2022; 17: e0266882.
- Vasquez-Rios G, Katz R, Levitan EB, Cushman M, Parikh CR, Kimmel PL, Bonventre JV, Waikar SS, Schrauben SJ, Greenberg JH et al. Urinary Biomarkers of Kidney Tubule Health and Mortality in Persons with CKD and Diabetes Mellitus. Kidney360 2023; 4: e1257-e1264.
- Mina IK, Mavrogeorgis E, Siwy J, Stojanov R, Mischak H, Latosinska A, Jankowski V. Multiple urinary peptides display distinct sex-specific distribution. Proteomics 2023; e2300227.
- Drum CL, Tan WKY, Chan SP, Pakkiri LS, Chong JPC, Liew OW, Ng TP, Ling LH, Sim D, Leong KG et al. Thymosin Beta-4 Is Elevated in Women With Heart Failure With Preserved Ejection Fraction. J Am Heart Assoc 2017; 6:.
- Rudnicki M, Siwy J, Wendt R, Lipphardt M, Koziolek MJ, Maixnerova D, Peters B, Kerschbaum J, Leierer J, Neprasova M et al. Urine proteomics for prediction of disease progression in patients with IgA nephropathy. Nephrol Dial Transplant 2020;
- Didriksson I, Lengquist M, Spangfors M, Leffler M, Sievert T, Lilja G, Frigyesi A, Friberg H, Schiopu A. Increasing plasma calprotectin (S100A8/A9) is associated with 12-month mortality and unfavourable functional outcome in critically ill COVID-19 patients. J Intensive Care 2024; 12: 26.
- Wendt R, Thijs L, Kalbitz S, Mischak H, Siwy J, Raad J, Metzger J, Neuhaus B, Leyen HV, Dudoignon E et al. A urinary peptidomic profile predicts outcome in SARS-CoV-2-infected patients. EClinicalMedicine 2021; 36: 100883.
- Barratt-Due A, Pettersen K, Borresdatter-Dahl T, Holter JC, Gronli RH, Dyrhol-Riise AM, Lerum TV, Holten AR, Tonby K, Troseid M et al. Escalated complement activation during hospitalization is associated with higher risk of 60-day mortality in SARS-CoV-2-infected patients. J Intern Med 2024; 296: 80-92.
- Keller F, Beige J, Siwy J, Mebazaa A, An D, Mischak H, Schanstra JP, Mokou M, Perco P, Staessen JA et al. Urinary peptides provide information about the risk of mortality across a spectrum of diseases and scenarios. J Transl Med 2023; 21: 663.
- Staessen JA, Wendt R, Yu YL, Kalbitz S, Thijs L, Siwy J, Raad J, Metzger J, Neuhaus B, Papkalla A et al. Predictive performance and clinical application of COV50, a urinary proteomic biomarker in early COVID-19 infection: a prospective multicentre cohort study. Lancet Digit Health 2022; 10: e727-e737.
- Magalhães P, Pejchinovski M, Markoska K, Banasik M, Klinger M, Svec-Billa D, Rychlik I, Rroji M, Restivo A, Capasso G et al. Association of kidney fibrosis with urinary peptides: a path towards non-invasive liquid biopsies? Sci Rep 2017; 7: 16915.
- Catanese L, Siwy J, Mavrogeorgis E, Amann K, Mischak H, Beige J, Rupprecht H. A Novel Urinary Proteomics Classifier for Non-Invasive Evaluation of Interstitial Fibrosis and Tubular Atrophy in Chronic Kidney Disease. Proteomes 2021; 9: 32.
- Siwy J, Wendt R, Albalat A, He T, Mischak H, Mullen W, Latosinska A, Lubbert C, Kalbitz S, Mebazaa A et al. CD99 and polymeric immunoglobulin receptor peptides deregulation in critical COVID-19: A potential link to molecular pathophysiology? Proteomics 2021; 21: e2100133.
- Tabara Y, Setoh K, Kawaguchi T, Kosugi S, Nakayama T, Matsuda F. Association between serum alpha1-antitrypsin levels and all-cause mortality in the general population: the Nagahama study. Sci Rep 2021; 11: 17241.
- Jaimes Campos MA, Andujar I, Keller F, Mayer G, Rossing P, Staessen JA, Delles C, Beige J, Glorieux G, Clark AL et al. Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study. Pharmaceuticals (Basel) 2023; 16: 1298.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).