1. Introduction
Human cytomegalovirus (CMV) is an ubiquitous DNA β-herpesvirus found worldwide. Global seroprevalence of CMV is estimated to be 56-94% with differences dependent on country of origin and socio-economic status of infected individuals1. HCMV infects many cell types, including epithelial cells in the oropharynx, vascular endothelial cells, hepatocytes, fibroblasts, smooth muscle cells, monocyte/macrophages, and dendritic cells. CMV enters the cell nucleus as an episomal element. Like other herpesviruses, HCMV establishes lifelong infection, including latent infection in myeloid cells of the bone marrow, where the outcome of infection depends on the cell type2, 3. Virally encoded proteins from virions or expressed in the cell during active infection, latency, or reactivation, control cellular functions necessary for further viral replication and spread 4. For example, CMV generally causes lytic infection in fibroblasts, while in myeloid progenitor cells, the initial burst of lytic gene expression is followed by viral latency, allowing infected monocytes to spread the virus systemically. In immunocompetent individuals, the host immune response controls acute CMV infection which is generally limited to asymptomatic or mild mononucleosis-like symptoms. In the absence of a fully functioning immune system, as in individuals following solid organ or bone marrow transplant (SOT or BMT) or with uncontrolled HIV infection, acute CMV infection or reactivation of latent virus can be associated with uncontrolled viral replication leading to end organ disease (EOD) and may contribute to rejection of transplanted organs5. Additionally, primary CMV infection in pregnancy poses a risk for congenital birth defects including neonatal hearing loss, microcephaly, and other neurodevelopmental conditions6.
Monocytes and macrophages are major components of the innate immune system and important targets for primary CMV infection and reactivation7. Here we will focus on reviewing the changes in gene expression and cell function associated with CMV infection/reactivation in monocytes and macrophages and the importance of these changes in the setting of SOT recipients.
2. Monocyte Functions
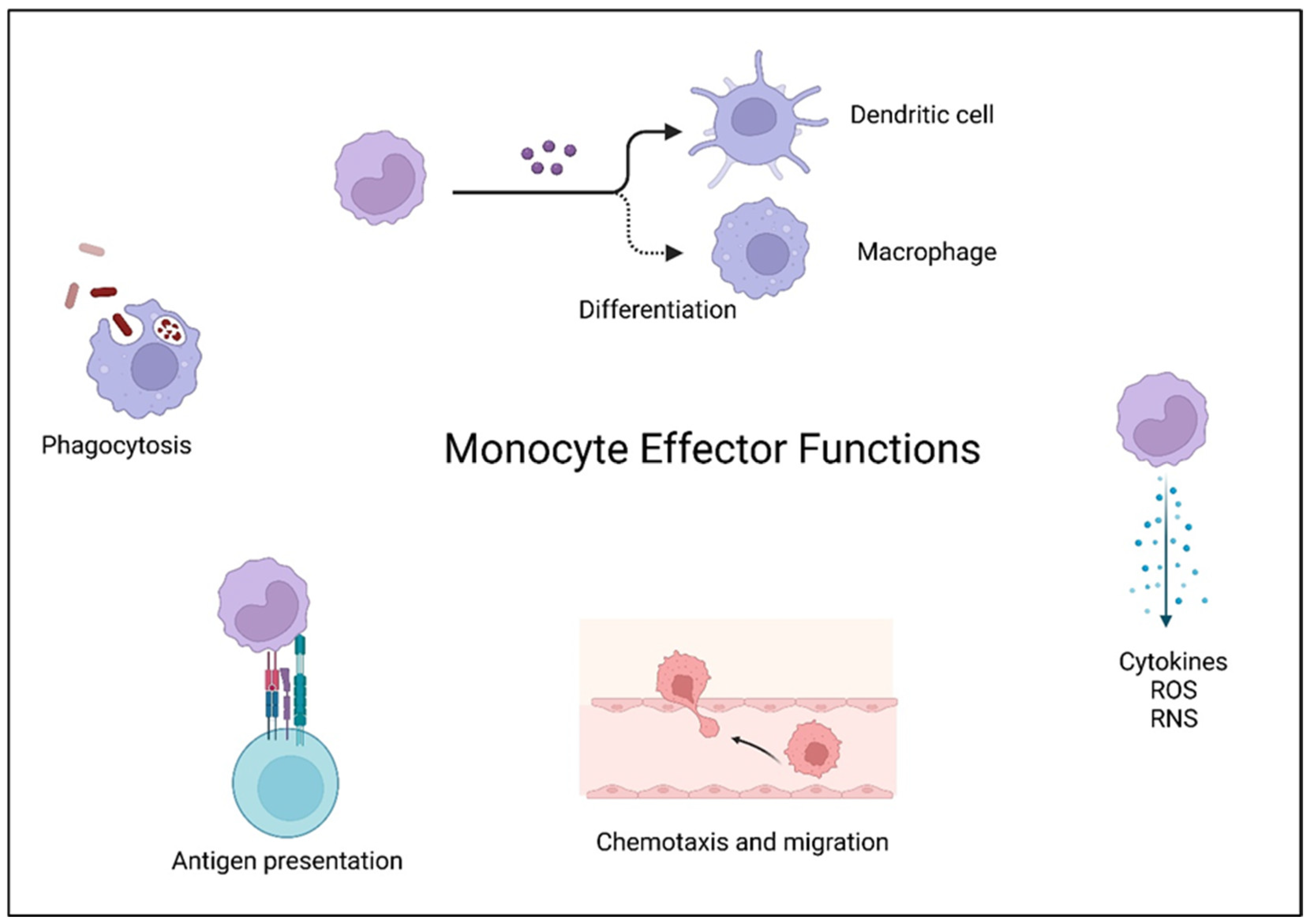
Monocytes are bone marrow-derived circulating immune cells responsible for many innate immune responses. There are three major subsets of human monocytes classified based on the relative expression of the cell surface markers CD14 and CD16: classical monocytes (CD14++, CD16-) intermediate (CD14+, CD16+), and nonclassical (CD14+, CD16++)8. Additionally, monocyte cell subsets are differentiated based on function (
Figure 1). Classical monocytes are the most prevalent subtype and are predominantly responsible for phagocytosis and antigen presentation. Intermediate monocytes are the next most prevalent subtype and mediate inflammation with generation of reactive oxygen and nitrogen species (ROS and RNS), antigen presentation for adaptive immune responses and T cell activation. Non-classical monocytes share functions with intermediate monocytes, secreting cytokines, activating T cells, and functioning in antigen presentation9, 10. A fourth subset with unknown function has been described in the literature based on single cell RNASeq data11.
Monocytes are crucial effectors in infection. In acute infections, monocytes migrate into tissues where they can differentiate into macrophages and dendritic cells9. Additionally, vascular and neural accumulation of monocytes plays a role in the pathogenesis of many chronic and inflammatory diseases including atherosclerosis, Alzheimer’s disease, and multiple sclerosis 12,13, 14. Monocytes are crucial in CMV pathogenesis in part due to their differentiation into macrophages within tissues. The lifespan of monocytes in circulation is short at approximately three weeks. However, differentiation into macrophages increases their life span to years15. The longevity of macrophages provides a stable reservoir for CMV persistence in multiple tissues and is responsible for clinical latency1.
Monocytes are versatile immune cells that perform several critical functions in the immune response (
Figure 1). These functions include:
1. Phagocytosis: Monocytes can engulf and digest self and non-self particles including pathogens and debris.
2. Antigen Presentation: Monocytes directly present antigens to T cells and can differentiate into macrophages and dendritic cells which are antigen presenting cells, initiating adaptive immune responses.
3. Cytokine Production: Monocytes produce cytokines and chemokines that modulate the immune response.
4. Chemotaxis: Monocytes migrate to sites of infection or injury in response to chemokine signals.
5. Tissue repair: Monocytes can differentiate into macrophages and dendritic cells, which have specialized functions in tissue repair and immune regulation.
6. ROS and RNS Production: Monocytes produce ROS and RNS involved in host defense and tissue damage.
3. Phagocytosis
Monocytes are among the first responders to infection or tissue damage. Phagocytosis is a fundamental function of monocytes with ingestion and destruction of invading pathogens such as bacteria, fungi and virus infected cells, as well as apoptotic cells and cellular debris.16, 17, 18. This action helps clear infections and initiates the process of healing and tissue repair. The engulfment of pathogens serves as a precursor to antigen presentation, a crucial step in activating the adaptive immune response. Monocytes recognize pathogens through pattern recognition receptors (PRRs),19, 20 Fc receptors,21 and complement receptors,22 and initiate phagocytosis. Once a particle is recognized and the phagocytic process is initiated, a specialized vacuole called phagosome forms which matures into a phagolysosome.23.
4. Antigen Presentation
After phagocytosis, monocytes act as antigen presenting cells (APCs), processing pathogens and presenting their antigens on the monocyte cell surface. Monocytes express major histocompatibility complex (MHC) class II molecules, which are essential for presenting exogenous antigens to CD4+ T helper cells. Additionally, monocytes can cross-present antigens on MHC class I molecules to CD8+ cytotoxic T cells, a function that is critical for antiviral immunity.24 By presenting antigens, monocytes help bridge the innate and adaptive immune systems, ensuring a more specific and sustained immune response against pathogens.
The antigen presentation capacity of monocytes is enhanced under inflammatory conditions and in response to certain stimuli. For instance, TLR7 stimulation has been shown to enhance cross-presentation of cell-associated antigens by Ly6C+ classical monocytes.25 In the context of cancer immunotherapy, antigen-loaded monocytes have demonstrated potential in inducing potent therapeutic antitumor T cell responses.26 These findings affirm monocytes' integral role in maintaining immune surveillance and orchestrating adaptive immunity.
In addition to directly presenting antigen to lymphocytes, after migrating to tissues, monocytes differentiate into macrophages and dendritic cells, both of which are proficient antigen-presenting cells (APCs). Similar to monocytes, macrophages and dendritic cells engulf pathogens, process their proteins, and present antigenic peptides on their surface via major histocompatibility complex (MHC) molecules.27, 28. However, it's important to note that while monocytes can present antigens, their efficiency may be lower compared to specialized dendritic cells. The role of monocytes in antigen presentation appears to be particularly significant in inflammatory conditions and may serve as a complementary mechanism to dendritic cell-mediated antigen presentation.
5. Cytokine and Chemokine Production
In addition to their phagocytic and antigen presenting capabilities monocytes have an important role in cytokine and chemokine production, which is crucial for orchestrating immune responses. Upon activation, monocytes produce a wide array of cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), IL-6, and IL-10, which modulate inflammation and immune cell recruitment. For instance, TNF-α and IL-1β are key pro-inflammatory cytokines that promote the activation and recruitment of other immune cells to sites of infection or injury.29 IL-6 plays a dual role by supporting both pro-inflammatory and anti-inflammatory pathways, depending on the context of its release.30
Chemokines are a family of small cytokines that activate G-protein-coupled receptors on monocytes. Chemokines produced by monocytes, such as Monocyte chemoattractant protein-1 (CCL2/MCP-1), are essential for the recruitment of monocytes and other leukocytes to inflamed tissues. CCL2 binds to chemokine receptor 2 (CCR2) on monocytes, facilitating their migration from the bone marrow to sites of inflammation.31 Additionally, monocytes can produce CCL5 (RANTES) and CXCL10 (IP-10), which further enhance the recruitment of T cells and other immune cells, thereby amplifying the immune response.32
The production of cytokines and chemokines by monocytes is tightly regulated and can be influenced by various factors, including pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs). This regulatory mechanism ensures a balanced immune response, preventing excessive inflammation that could lead to tissue damage. Understanding the specific pathways and triggers for cytokine and chemokine production in monocytes is crucial for developing targeted therapies for inflammatory and infectious diseases.33.
6. Chemotaxis
Chemotaxis is a crucial effector function of monocytes, enabling their directed migration towards sites of infection or inflammation. This process is primarily mediated by MCP-1/CCL2, a key chemokine regulating monocyte migration and infiltration through its interaction with its receptor CCR2.34 Upon activation, monocytes undergo a series of coordinated events involving upregulation of adhesion molecules expression, cytoskeletal rearrangements, and polarization to facilitate their movement along chemokine gradients.35 The chemotactic response involves the activation of intracellular signaling cascades, including the mobilization of intracellular calcium and the activation of kinases such as PI3K and MAPK.36 These signaling events lead to the activation of integrins, which mediate firm adhesion to the endothelium, and the formation of specialized structures like focal adhesions and the uropod, essential for directional movement.35 Importantly, chemotaxis is not only crucial for monocyte recruitment to inflammatory sites but also plays a role in their normal physiological trafficking and immune surveillance.37 Defects in monocyte chemotaxis have been associated with various pathological conditions, including acquired immune deficiency syndrome (AIDS), where impaired monocyte migration contributes to the compromised immune response 37 and Alzheimer’s disease.38, 39 Understanding the molecular mechanisms underlying monocyte chemotaxis is essential for developing targeted therapies for inflammatory disorders and improving our comprehension of the immune system's functionality.
7. Tissue Repair
Monocytes play a role in tissue repair through their ability to differentiate into macrophages and their direct contributions to the repair process. Recent studies have elucidated the complex interplay between monocyte subsets and their functions in various stages of tissue repair. Classical CD14++CD16- monocytes recruited to sites of injury differentiate into inflammatory macrophages that promote debris clearance and initiate the repair cascade. These cells secrete pro-inflammatory cytokines and growth factors that stimulate angiogenesis and fibroblast activation. As the repair process progresses, there is a shift towards non-classical CD14+CD16++ monocytes, which can differentiate into reparative macrophages that support resolution of inflammation, tissue regeneration and remodeling. 40
The plasticity of monocytes and monocyte-derived macrophages is a key feature in tissue repair, allowing them to adapt their phenotype in response to environmental cues. This plasticity is particularly evident in chronic inflammatory diseases, where the balance between pro-inflammatory and pro-resolving monocyte functions can influence disease progression and tissue repair outcomes.41 Recent research has also highlighted the potential of biomaterial-based approaches to modulate monocyte behavior in tissue repair contexts, offering new avenues for therapeutic interventions.42
It is important to note that while monocytes are generally beneficial for tissue repair, their dysregulation can contribute to fibrosis and impaired healing in certain conditions. The specific roles of monocyte subsets can vary depending on the tissue type and nature of the injury, underscoring the need for context-specific investigations to fully elucidate their functions in tissue repair processes.40.
8. ROS Production
Monocytes produce reactive oxygen species (ROS) and reactive nitrogen species (RNS) as part of their effector functions43. ROS, including superoxide anion (O2−) and hydrogen peroxide (H2O2), are generated through the activation of the NADPH oxidase complex44, 45. This activation occurs in response to various stimuli, such as pathogen-associated molecular patterns (PAMPs) and cytokines45. Upon activation, NADPH oxidase transfers electrons from NADPH to oxygen, forming superoxide anions, which subsequently transform to hydrogen peroxide46. RNS, primarily nitric oxide (NO), are synthesized by the enzyme inducible nitric oxide synthase (iNOS), which is upregulated during inflammatory responses47. Nitric oxide can react with superoxide to form peroxynitrite (ONOO−), a potent oxidant with antimicrobial properties48.
The production of ROS and RNS by monocytes contributes to the elimination of pathogens through oxidative and nitrosative stress, damaging microbial DNA, proteins, and lipids. This antimicrobial activity is essential for controlling infections, particularly in the early stages of immune response. Moreover, ROS and RNS act as signaling molecules, modulating various immune functions such as cytokine production, cell proliferation, and apoptosis49. However, excessive or uncontrolled production of ROS and RNS can lead to tissue damage and contribute to the pathogenesis of inflammatory diseases, including atherosclerosis 50, rheumatoid arthritis 51, and sepsis 52.
In conclusion, the production of ROS and RNS by monocytes is a critical effector function that mediates pathogen clearance and regulates immune responses. Understanding the balance between their beneficial and detrimental effects is essential for developing therapeutic strategies to modulate immune responses in various diseases.
9. Effect of CMV on Monocyte Effector Functions
9.1. Phagocytosis
CMV infection has been shown to modulate the phagocytic ability of monocytes (
Figure 2) which may compromise the immune system’s ability to handle infection. One mechanism by which CMV impacts phagocytosis is through a paradoxical effect on classical (CD14++, CD16-) monocytes. RNA sequencing of monocytes post-CMV infection demonstrates upregulation of viral pathogen recognition receptors (PRRs) while also demonstrating down regulation of pathogen associated molecular pattern (PAMP)-related scavenger receptors. The specific scavenger receptors impacted include CD36 which is responsible for fungal pathogen recognition53. Decreased expression of key phagocytotic receptors for fungi leads to a marked decreased ability to phagocytose key fungal pathogens including C. albicans and C. neoformans which are major contributors to morbidity in immunocompromised hosts.53, 54. These findings provide a molecular explanation for the increased susceptibility to fungal infections during or following infection with CMV. It remains unclear whether low level CMV reactivation associated with acute illness of many types is similarly involved in risks for nosocomial fungal superinfections 55.
Other studies have demonstrated that complement receptors 3 and 4, CR3 and CR4, two important modulators of fungal and bacterial phagocytosis, are downregulated in both THP-1 cell line-derived macrophages and monocyte-derived human macrophages infected with HCMV56.
While published evidence demonstrates directly that CMV down-regulates phagocytosis through scavenger and complement receptors, the effect of CMV infection on Fcγ-receptor-mediated phagocytosis is not fully clear. There is evidence that CMV may modify Fcγ receptor-mediated phagocytosis through the expression of cmvIL-10, a homologous cytokine to human interleukin-10 (hIL-10)57. Monocytes cultured with cmvIL-10 demonstrated increased expression of Fcγ receptors CD32 and CD6457. cmvIL-10 increased Fcγ-receptor mediated phagocytosis by monocytes similar to hIL-1057. The role of enhanced Fcγ-mediated phagocytosis in monocytes in the pathogenesis of CMV disease is not clear but may play a role in viral dissemination. Similarly, HCMV infection induces the expression of viral FcγRs, such as gp34 and gp68, which can bind to the Fc region of immunoglobulins and inhibit the activation of host FcγRs (FcγRI, FcγRIIA, and FcγRIIIA) on monocytes and other immune cells 58. This prevents IgG-mediated triggering of activating host FcγRs, thereby impairing antibody-dependent cellular phagocytosis (ADCP) and other Fc-mediated effector functions58. Further studies on the impacts of CMV infection on Fcγ-mediated phagocytosis are necessary to clarify the biologic significance and implications of altering this phagocytic pathway on CMV disease and the innate immune response.
10. Effect of CMV on Antigen Presentation
CMV infection affects the antigen-presenting function of monocytes through multiple mechanisms (
Figure 3). First, CMV decreases the effectiveness of antigen presentation by inhibiting the differentiation of monocytes into dendritic cells (DCs)59, 60, 61. DCs are the major antigen presenting cells and play a key role in continuing immune activation via signaling to T cells.62 By blocking differentiation- inducing cytokines like IL-4 and GM-CSF, CMV prevents monocyte differentiation into CD1a-positive dendritic cells.59 Monocytes possess a much lower affinity for antigen presentation than dendritic cells. Thus, inhibiting monocyte differentiation decreases antigen presentation making the host more susceptible to infection.
Second, in the setting of acute infection, CMV downregulates the expression of major histocompatibility complex (MHC) class I and II molecules.60 CMV also can downregulate the co-stimulatory molecules CD40 and CD80 on the surface of monocytes and immature DCs60, 61. Downregulation of these co-stimulatory molecules may inhibit antigen loading of existing MHC molecules. Third, CMV expresses immune-evasins, glycoproteins that interrupt classical MHC class I pathways.63 Indeed CMV encoded proteins US3, US6, and US10 can retain MHC class I molecules in the ER, inhibit peptide transport by the transporter associated with antigen processing (TAP), or delay trafficking of MHC I to the cell surface.64, 65 66, 67, 68 The combination of these effects further undermines CD4+ and CD8+ T cell immunity and may help CMV go unchecked in the settings of acute infection and reactivation.
11. Effect of CMV on Cytokine Production
Cytokines are crucial players in regulating immune responses and inflammation. Monocytes infected with CMV produce a mixed phenotype of pro- and anti-inflammatory cytokines (
Figure 4); this may reflect incomplete infection of the cell population as well as various states of CMV infection. The resulting immune environment promotes pathways beneficial for viral persistence while allowing cellular CMV to evade the immune system. Pro-inflammatory cytokines IL-6 and IL-8 are increased when monocytes are incubated with UV-inactivated CMV virus, indicating cytokine production results from cellular contact with viral particles rather than new gene expression.69 Increases in cytokine signaling requires TLR2 and CD14, pattern recognition receptors responsible for recognizing CMV virions and inflammatory signaling pathways. Other pro-inflammatory cytokines including TNF are also increased through activation of p38 kinase and NF-kB signaling pathways in monocytes 70; this requires further investigation as some reports show decreased TNF production with acute CMV infection or reactivation.61.
In addition to increasing cytokine gene expression, further mechanisms are involved in the CMV-modified monocytic cytokine production. Binding of CMV to monocytes and the major CMV glycoproteins gB (UL55)- and gH (UL75)-mediated signaling upregulated secretion of IL-1β. This expression required activation of NF-kB, a transcription factor responsible for regulating IL-1β gene expression.71 Although more research is needed, the CMV-mediated increase in IL-1β expression could be linked to chronic inflammation associated with CMV infections and further complicate analysis of CMV infection in immunocompromised hosts.
While increased pro-inflammatory cytokines may be crucial to acute infection and the inflammatory diseases associated with CMV, CMV also modulates anti-inflammatory cytokines for immune evasion and latency. For example, CMV-upregulates production of IL-10, an anti-inflammatory cytokine shown to prevent the differentiation of monocytes into dendritic cells.59, 72 Furthermore, monocytes and DCs infected with CMV have decreased production of interleukin-12, a key cytokine in Th1-mediated immunity. Decreased cytokine signaling presents another barrier to T-cell activation and the cytotoxic killing of CMV.
In addition to utilizing viral proteins for downregulating cytokines, CMV utilizes microRNA (miRNA) to decrease the expression of pro-inflammatory cytokines during latency and later infection. Through these miRNA species, CMV downregulates IKKa and IKKB signaling factors, limiting production of pro-inflammatory cytokines, IL-6 and TNF-a to remain dormant and evade host immunity.73 .
12. Effect of CMV on Chemotaxis
CMV-infected monocytes exhibit decreased chemokine receptor expression, which can impair migration to sites of infection or injury (
Figure 5). These downregulated chemokine receptors include CCR1, CCR2, CCR5, and CXCR4. Such downregulation occurs even in the absence of viral gene expression and is believed to be due to increased internalization of these receptors, rather than decreased gene expression or protein degradation. 74 Functionally, monocytes were unable to migrate toward CCL2, CCL5, CXCl12, CCL19, or CX3CL1, key chemokine modulators of adaptive immunity and critical for monocyte migration and recruitment to sites of inflammation, thereby potentially compromising the host’s ability to mount an effective immune response.
In contrast to downregulated chemokine-mediated migration, CMV-infected monocytes demonstrate increased transendothelial migration. This increased migration is independent of viral gene expression, and occurs in monocytes incubated with both live CMV and UV-inactivated CMV strains.75 Several mechanisms are thought to be responsible for such an increase. First, cell motility is increased.75 Second, increased monocyte adhesion to endothelial cells, a key step in both early and late stages of transendothelial migration, is also increased. This is due to enhanced expression of β1 integrin, occludin, and ZO-1, proteins involved in monocyte adhesion and diapedesis75. Additional studies also suggest that CMV-infection of the endothelial cells in turn increases migration of monocytes.76
It has been speculated that CMV could also impact migration through chemokinesis, a motility process independent of chemokines.77 Chemokinesis is dependent on PI(3)K and the actin cytoskeleton and requires activation and signaling via integrin and EGFR pathways. These pathways are increased as a result of CMV infection 1. These secondary pathways allow CMV-infected monocytes to migrate more effectively, even without the presence of chemotactic signaling. Downregulating chemotaxis-dependent migration while maintaining chemotactic-independent migration may help CMV evade the adaptive immune system while using it to disseminate further.
CMV can also influence chemotaxis and migration through the production of chemokine-mimics encoded in the CMV genome. Chemokine homologs are common to many viruses and provide a method of immune avoidance. In MCMV, a viral chemokine, MCK-2, a homolog of host chemokine IL-8, serves as a potent attractant for neutrophils and monocytes through binding of the CXCR2 receptor.78 In HCMV, a chemokine homolog is encoded by the UL128 gene promotes the migration of peripheral blood mononuclear cells, and may aid in viral dissemination during acute infection.79 Chemokine receptor homologs, like the one encoded by the CMV gene US28, may also promote monocyte chemotaxis through activation of G-protein signaling cascades.80, 81
Based on these observations, it is clear that CMV can regulate the migration of monocytes. Decreased chemokine-mediated monocyte migration can help CMV evade the immune response while increasing viral dissemination via increased CMV-induced monocyte chemokinesis and trans-endothelial migration and in response to CMV-induced chemokine mimics. Understanding the stages of CMV infection that regulate each of these migratory functions will clarify the role of this important monocyte effector function in CMV pathogenesis.
13. Effect of CMV on Tissue Repair and Immune Regulation
CMV infection can have a significant impact on tissue repair. On one hand, acute infection promotes the differentiation of monocytes into macrophages, which differentiation is often skewed towards an anti-inflammatory phenotype. CMV-infected monocytes have reduced phagocytotic abilities, altered migration, and improper signaling of T-cells. Dysregulation to these typical functions leads to improper tissue repair.82 In contrast, prolonged CMV infection can lead to a chronic inflammatory state caused by cytokines from CMV-infected monocytes. High levels of IL-6, IL-8, and IL-1β create a cellular environment that is less favorable to tissue regeneration.71 Improper adhesion to endothelial cells by CMV-infected monocytes may also contribute to excessive inflammation83. We propose that these apparent contrasting effects on the inflammatory process appear to have additive negative effects on tissue repair.
While the transition from monocytes to macrophages is upregulated through interactions with CMV, there is altered differentiation into CD1a-positive dendritic cells by IL-4 and GM-CSF59. Dendritic cells differentiated from CMV-infected monocytes demonstrate an inability to secrete IL-12 in response to LPS stimulation, decreased phagocytosis, decreased induction of TH-1 cell differentiation. There is also disruption of GM-CSF signaling in the entire population of dendritic cells, highlighting the widespread disruption by CMV. 84 Because CMV-infected dendritic cells exhibit decreased differentiation, we propose that their role in tissue repair is also dysregulated.
14. Effect of CMV on ROS Production
An important component of the monocytes host defense mechanisms is the generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS). ROS, including superoxide (O2͘-), hydrogen peroxide (H2O2), and hydroxyl radical (OH-).85 Within monocytes, engulfed material enters the phagolysosome where it encounters superoxide, proteases, and other molecules that can kill microbes. ROS are secreted extracellularly where they can perform their host defense functions86. This effector function of monocytes is important in the host’s response to bacterial, fungal and viral pathogens.87 ROS also play a major role as cellular signaling molecules during inflammation88
During acute infection or reactivation, CMV upregulates ROS generation even in the absence of DNA replication within monocytes. This modulation is thought to be associated with increased transduction of signaling cascades of ROS generation or through the augmentation of cytosolic factors involved in ROS generation 89. Similarly, when THP-1 cells, a human monocytic cell line, were infected with CMV, there was a significant increase in ROS generation during active CMV infection. 90. Therefore, in contrast to the role of ROS in bacterial and fungal killing, the increased levels of ROS are beneficial to CMV and enhance CMV gene expression, assisting with viral persistence during acute infections.91 Additionally, oxidative stress and inflammatory responses to increased ROS may create a cellular environment more favorable to viral replication.92
In contrast to upregulation of ROS signaling and production observed with active CMV replication in monocytes, CMV evades the immune response during latency by downregulating ROS production by macrophages. This suppressive effect on ROS production during latency is believed to be regulated by a long non-coding RNA (lncRNA) called β2.7. CMV variants without the β2.7 gene were less effective at establishing latency within host monocytes and suppressing ROS production 93. These findings suggest that the ability to downregulate ROS production in the host allows CMV to remain latent and undetected by the host immune system. CMV’s dual effect on monocyte and macrophage ROS production links this important monocyte/macrophage effector function to two important paths involved in CMV pathogenesis in immunocompromised hosts: one path leading to replication and one leading to integration into myeloid cells and life-long latency.
15. Clinical Implications
The effects of cytomegalovirus (CMV) infection on monocyte effector functions have significant clinical implications, particularly in the immunosuppressed host. Solid organ transplant (SOT) patients, those with uncontrolled HIV/AIDS, and those with iatrogenic immunosuppression are at high risk of complications of CMV-related illness. Although CMV possesses many mechanisms to disrupt host immunity, modulation of monocyte effector functions provides a paradoxical change to innate immune responses, leading to both immunosuppression (e.g., opportunistic infection) and immunostimulation (e.g., graft rejection). These dual and paradoxically opposed effects provide insight into the importance of CMV infection in active, reactivated, and latent states.
Phagocytosis: a crucial effector function in host defense against pathogens. Monocytes are responsible for phagocytosis, with disruption of this ability with CMV infection important in increased susceptibility to infection. The reduced phagocytic ability of monocytes is detrimental against clinically important and common yeasts including Candida albicans and Cryptococcus neoformans. This is associated with decreased expression of key pattern recognition receptors such as CD36, MRC1, and complement receptor 3.53 A higher risk of invasive fungal infections is amplified in immunocompromised individuals with decreased immunity against opportunistic fungi.
Increased infections with fungal pathogens create an issue in post-SOT management. The post-transplant regime is complex with a variety of immunosuppressive, antibacterial, antiviral, and antifungal agents.94 Impaired phagocytosis of C. albicans and C. neoformans, two of the three most common fungal infections post-transplant95, may necessitate more aggressive antifungal prophylaxis in CMV-infected transplant recipients. These fungal infections in patients following SOT may increase risk for allograft rejection (possibly associated with decreases in immunosuppression) and have a 12 month mortality of 34% and 27%, respectively.95 Strategies to restore phagocytic function in CMV-infected monocytes could potentially mitigate the indirect effects of CMV in transplantation.
Antigen Presentation: Decreased antigen presentation by monocytes in patients with CMV may have significant clinical implications. First, decreased antigen presentation will reduce the stimulation of T cell responses against both CMV and other opportunistic pathogens. CMV-infected monocytes may also exhibit increased expression of regulatory costimulatory molecules including CD86, potentially enhancing T cell activation in certain contexts.59 Expression of CD86 without proper antigen presentation may result in T-cell anergy, increasing risk for opportunistic (viral, fungal) infections.
Production of pro-inflammatory cytokines: A major impact on monocyte effector function is the production of pro-inflammatory cytokines during acute infection and with reactivation of CMV. Upregulation of key cytokines, IL-6, IL-1β, and TNF-α create a pro-inflammatory environment that is significant in both acute and chronic diseases. Of major concern is heightened levels of inflammation in SOT patients which may contribute to allograft dysfunction, rejection, and graft-versus-host disease in transplant recipients.
CMV is also associated with important organ-specific infections in immunocompromised hosts including CMV colitis, retinitis, hepatitis, pneumonitis, and encephalitis. There is evidence that the pro-inflammatory cellular environment may contribute to chronic inflammatory diseases including atherogenesis, accelerated atherosclerosis in cardiac transplant recipients, and chronic lung allograft dysfunction (CLAD) in lung recipients.96
Implications for therapeutics: The impact of CMV on monocyte differentiation and function has implications for development of therapeutic strategies. Targeting of pathways involved in monocyte differentiation and cytokine production could mitigate the impacts of CMV infection and improve clinical outcomes for affected patients. For example, therapies that inhibit the differentiation of monocytes into anti-inflammatory macrophages or enhance the phagocytic capacity of monocytes could reduce viral persistence and improve viral immune clearance.79
In conclusion, the complex modulation of monocyte effector functions by CMV has far-reaching clinical implications in transplantation. A nuanced understanding of these effects is crucial for optimizing management strategies, including antiviral therapy, immunosuppression, and the development of cellular therapies, to improve outcomes in CMV-infected transplant recipients.
Conversely, CMV infection also induces pro-inflammatory and immunostimulatory responses in monocytes. This includes upregulation of viral pattern recognition receptors, inflammasome components (e.g., AIM2, IFI16), and pro-inflammatory mediators associated with allograft rejection and graft-versus-host disease.97
These divergent effects of CMV on monocyte function suggest that the pro-inflammatory phenotype may require tailored immunosuppression strategies to mitigate the risk of allograft rejection. The heterogeneity in monocyte responses to CMV infection, as revealed by single-cell transcriptomics, suggests that personalized approaches may be needed to address the variable effects in individual patients. Understanding these CMV-mediated alterations in monocyte function opens avenues for targeted therapies. Strategies to restore phagocytic function or modulate inflammatory responses in CMV-infected monocytes could potentially mitigate the indirect effects of CMV in transplantation. Additionally, the immunostimulatory properties of CMV immunoglobulin (CMVIG) on innate immune cells, including monocytes, may be harnessed to enhance anti-CMV immunity in transplant recipients. 98
16. Conclusions
CMV infection has a profound impact on monocyte effector functions, impairing phagocytosis, antigen presentation, cytokine production, cell migration to sites of infection, and differentiation into effective immune cells. These alterations contribute to immune compromise and to susceptibility to infections, heightened inflammation, graft rejection, and long-term complications in immunocompromised individuals. Understanding the mechanisms by which CMV modulates monocyte functions is crucial for developing targeted therapies to mitigate the effects of CMV infection and improve clinical outcomes for affected patients.
References
- Min, C.-K.; et al. The Differentiation of Human Cytomegalovirus Infected-Monocytes Is Required for Viral Replication.
- Bruggeman, C.A. Cytomegalovirus and latency: an overview. Virchows Archiv B Cell Pathology Including Molecular Pathology 64, 325-333 (1993). [CrossRef]
- 3.Forte, E., Zhang, Z., Thorp, E.B. & Hummel, M. Cytomegalovirus Latency and Reactivation: An Intricate Interplay With the Host Immune Response. Front Cell Infect Microbiol 10, 130 (2020).
- Jean Beltran, P.M. & Cristea, I.M. The life cycle and pathogenesis of human cytomegalovirus infection: lessons from proteomics. Expert Rev Proteomics 11, 697-711 (2014).
- Fishman, J.A. & Rubin, R.H. Infection in organ-transplant recipients. N Engl J Med 338, 1741-1751 (1998). [CrossRef]
- Pesch, M.H., Saunders, N.A. & Abdelnabi, S. Cytomegalovirus Infection in Pregnancy: Prevention, Presentation, Management and Neonatal Outcomes. Journal of Midwifery & Women's Health 66, 397-402 (2021).
- Taylor-Wiedeman, J., Sissons, J.G., Borysiewicz, L.K. & Sinclair, J.H. Monocytes are a major site of persistence of human cytomegalovirus in peripheral blood mononuclear cells. J Gen Virol 72 ( Pt 9), 2059-2064 (1991).
- Ziegler-Heitbrock, L.; et al. Nomenclature of monocytes and dendritic cells in blood. Blood 116, e74-e80 (2010). [CrossRef]
- Williams, H. et al. Monocyte Differentiation and Heterogeneity: Inter-Subset and Interindividual Differences. Int J Mol Sci 24 (2023).
- Sampath, P., Moideen, K., Ranganathan, U.D. & Bethunaickan, R. Monocyte Subsets: Phenotypes and Function in Tuberculosis Infection. Frontiers in Immunology 9 (2018).
- Villani, A.-C.; et al. Single-cell RNA-seq reveals new types of human blood dendritic cells, monocytes, and progenitors. Science 356, eaah4573 (2017).
- Thériault, P., Elali, A. & Rivest, S. The dynamics of monocytes and microglia in Alzheimer’s disease. Alzheimer's Research & Therapy 7 (2015).
- Carstensen, M.; et al. Activated monocytes and markers of inflammation in newly diagnosed multiple sclerosis. Immunology & Cell Biology 98, 549-562 (2020). [CrossRef]
- El Khoury, J. Neurodegeneration and the neuroimmune system. Nat Med 16, 1369-1370 (2010).
- Collins-Mcmillen, D.; et al. HCMV Infection and Apoptosis: How Do Monocytes Survive HCMV Infection? Viruses 10, 533 (2018).
- Mikolajczyk, T.P.; et al. Interaction of human peripheral blood monocytes with apoptotic polymorphonuclear cells. Immunology 128, 103-113 (2009).
- Serbina, N.V., Jia, T., Hohl, T.M. & Pamer, E.G. Monocyte-mediated defense against microbial pathogens. Annu Rev Immunol 26, 421-452 (2008). [CrossRef]
- Westman, J., Grinstein, S. & Marques, P.E. Phagocytosis of Necrotic Debris at Sites of Injury and Inflammation. Front Immunol 10, 3030 (2019).
- Alquraini, A. & El Khoury, J. Scavenger receptors. Curr Biol 30, R790-R795 (2020).
- Wicherska-Pawlowska, K., Wrobel, T. & Rybka, J. Toll-Like Receptors (TLRs), NOD-Like Receptors (NLRs), and RIG-I-Like Receptors (RLRs) in Innate Immunity. TLRs, NLRs, and RLRs Ligands as Immunotherapeutic Agents for Hematopoietic Diseases. Int J Mol Sci 22 (2021).
- Nimmerjahn, F. & Ravetch, J.V. Fcgamma receptors as regulators of immune responses. Nat Rev Immunol 8, 34-47 (2008).
- Vandendriessche, S., Cambier, S., Proost, P. & Marques, P.E. Complement Receptors and Their Role in Leukocyte Recruitment and Phagocytosis. Front Cell Dev Biol 9, 624025 (2021).
- Uribe-Querol, E. & Rosales, C. Phagocytosis: Our Current Understanding of a Universal Biological Process. Front Immunol 11, 1066 (2020).
- Lauvau, G., Chorro, L., Spaulding, E. & Soudja, S.M. Inflammatory monocyte effector mechanisms. Cell Immunol 291, 32-40 (2014).
- 25.Larson, S.R. et al. Ly6C(+) monocyte efferocytosis and cross-presentation of cell-associated antigens. Cell Death Differ 23, 997-1003 (2016).
- Huang, M.N.; et al. Antigen-loaded monocyte administration induces potent therapeutic antitumor T cell responses. J Clin Invest 130, 774-788 (2020).
- Schlitzer, A., Zhang, W., Song, M. & Ma, X. Recent advances in understanding dendritic cell development, classification, and phenotype. F1000Res 7 (2018).
- Muntjewerff, E.M., Meesters, L.D. & van den Bogaart, G. Antigen Cross-Presentation by Macrophages. Front Immunol 11, 1276 (2020).
- Kany, S., Vollrath, J.T. & Relja, B. Cytokines in Inflammatory Disease. Int J Mol Sci 20 (2019).
- Choi, B.R., Johnson, K.R., Maric, D. & McGavern, D.B. Monocyte-derived IL-6 programs microglia to rebuild damaged brain vasculature. Nat Immunol 24, 1110-1123 (2023).
- Crane, M.J., Hokeness-Antonelli, K.L. & Salazar-Mather, T.P. Regulation of inflammatory monocyte/macrophage recruitment from the bone marrow during murine cytomegalovirus infection: role for type I interferons in localized induction of CCR2 ligands. J Immunol 183, 2810-2817 (2009).
- Ozga, A.J., Chow, M.T. & Luster, A.D. Chemokines and the immune response to cancer. Immunity 54, 859-874 (2021).
- Karki, R. & Kanneganti, T.D. The 'cytokine storm': molecular mechanisms and therapeutic prospects. Trends Immunol 42, 681-705 (2021).
- Deshmane, S.L., Kremlev, S., Amini, S. & Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res 29, 313-326 (2009).
- Sokol, C.L. & Luster, A.D. The chemokine system in innate immunity. Cold Spring Harb Perspect Biol 7 (2015).
- Heit, B., Tavener, S., Raharjo, E. & Kubes, P. An intracellular signaling hierarchy determines direction of migration in opposing chemotactic gradients. J Cell Biol 159, 91-102 (2002).
- Smith, P.D.; et al. Monocyte function in the acquired immune deficiency syndrome. Defective chemotaxis. J Clin Invest 74, 2121-2128 (1984).
- El Khoury, J.; et al. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat Med 13, 432-438 (2007).
- Hickman, S.E., Allison, E.K., Coleman, U., Kingery-Gallagher, N.D. & El Khoury, J. Heterozygous CX3CR1 Deficiency in Microglia Restores Neuronal beta-Amyloid Clearance Pathways and Slows Progression of Alzheimer's Like-Disease in PS1-APP Mice. Front Immunol 10, 2780 (2019).
- Kratofil, R.M., Kubes, P. & Deniset, J.F. Monocyte Conversion During Inflammation and Injury. Arterioscler Thromb Vasc Biol 37, 35-42 (2017).
- Das, A.; et al. Monocyte and macrophage plasticity in tissue repair and regeneration. Am J Pathol 185, 2596-2606 (2015).
- Italiani, P. & Boraschi, D. From Monocytes to M1/M2 Macrophages: Phenotypical vs. Functional Differentiation. Front Immunol 5, 514 (2014).
- Weiss, S.J., King, G.W. & LoBuglio, A.F. Evidence for hydroxyl radical generation by human Monocytes. J Clin Invest 60, 370-373 (1977).
- Johnston, R.B. & Kitagawa, S. Molecular basis for the enhanced respiratory burst of activated macrophages. Fed Proc 44, 2927-2932 (1985).
- Canton, M.; et al. Reactive Oxygen Species in Macrophages: Sources and Targets. Front Immunol 12, 734229 (2021).
- Puntarulo, S., Galleano, M., Sanchez, R.A. & Boveris, A. Superoxide anion and hydrogen peroxide metabolism in soybean embryonic axes during germination. Biochim Biophys Acta 1074, 277-283 (1991).
- Hazen, S.L.; et al. Formation of nitric oxide-derived oxidants by myeloperoxidase in monocytes: pathways for monocyte-mediated protein nitration and lipid peroxidation In vivo. Circ Res 85, 950-958 (1999).
- Crow, J.P. & Beckman, J.S. The role of peroxynitrite in nitric oxide-mediated toxicity. Curr Top Microbiol Immunol 196, 57-73 (1995).
- Lennicke, C. & Cocheme, H.M. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol Cell 81, 3691-3707 (2021).
- Batty, M., Bennett, M.R. & Yu, E. The Role of Oxidative Stress in Atherosclerosis. Cells 11 (2022).
- Wang, X.; et al. The Role of Reactive Oxygen Species in the Rheumatoid Arthritis-Associated Synovial Microenvironment. Antioxidants (Basel) 11 (2022).
- Jensen, I.J.; et al. Prolonged Reactive Oxygen Species Production following Septic Insult. Immunohorizons 5, 477-488 (2021).
- Sen, P.; et al. Linking indirect effects of cytomegalovirus in transplantation to modulation of monocyte innate immune function. Sci Adv 6, eaax9856 (2020).
- Dropulic, L.K. & Lederman, H.M. Overview of Infections in the Immunocompromised Host. Microbiol Spectr 4 (2016).
- Imlay, H. & Limaye, A.P. Current Understanding of Cytomegalovirus Reactivation in Critical Illness. J Infect Dis 221, S94-S102 (2020).
- Gafa, V.; et al. Human cytomegalovirus downregulates complement receptors (CR3, CR4) and decreases phagocytosis by macrophages. J Med Virol 76, 361-366 (2005).
- Jaworowski, A.; et al. Enhanced monocyte Fc phagocytosis by a homologue of interleukin-10 encoded by human cytomegalovirus. Virology 391, 20-24 (2009).
- Corrales-Aguilar, E.; et al. Human cytomegalovirus Fcgamma binding proteins gp34 and gp68 antagonize Fcgamma receptors I, II and III. PLoS Pathog 10, e1004131 (2014).
- Gredmark, S. & SöDerberg-NaucléR, C. Human Cytomegalovirus Inhibits Differentiation of Monocytes into Dendritic Cells with the Consequence of Depressed Immunological Functions. Journal of Virology 77, 10943-10956 (2003).
- Grigoleit, U.; et al. Human cytomegalovirus induces a direct inhibitory effect on antigen presentation by monocyte-derived immature dendritic cells. Br J Haematol 119, 189-198 (2002).
- Moutaftsi, M., Mehl, A.M., Borysiewicz, L.K. & Tabi, Z. Human cytomegalovirus inhibits maturation and impairs function of monocyte-derived dendritic cells. Blood 99, 2913-2921 (2002).
- Banchereau, J. & Steinman, R.M. Dendritic cells and the control of immunity. Nature 392, 245-252 (1998).
- Reddehase, M.J. Antigens and immunoevasins: opponents in cytomegalovirus immune surveillance. Nature Reviews Immunology 2, 831-844 (2002).
- Ahn, K.; et al. Human cytomegalovirus inhibits antigen presentation by a sequential multistep process. Proceedings of the National Academy of Sciences 93, 10990-10995 (1996).
- Jones, T.R.; et al. Human cytomegalovirus US3 impairs transport and maturation of major histocompatibility complex class I heavy chains. Proceedings of the National Academy of Sciences 93, 11327-11333 (1996).
- Furman, M.H., Dey, N., Tortorella, D. & Ploegh, H.L. The Human Cytomegalovirus US10 Gene Product Delays Trafficking of Major Histocompatibility Complex Class I Molecules. Journal of Virology 76, 11753-11756 (2002).
- Lehner, P.J., Karttunen, J.T., Wilkinson, G.W.G. & Cresswell, P. The human cytomegalovirus US6 glycoprotein inhibits transporter associated with antigen processing-dependent peptide translocation. Proceedings of the National Academy of Sciences 94, 6904-6909 (1997).
- Gewurz, B.E.; et al. Antigen presentation subverted: Structure of the human cytomegalovirus protein US2 bound to the class I molecule HLA-A2. Proc Natl Acad Sci U S A 98, 6794-6799 (2001).
- Compton, T.; et al. Human cytomegalovirus activates inflammatory cytokine responses via CD14 and Toll-like receptor 2. J Virol 77, 4588-4596 (2003).
- Smith, P.D., Saini, S.S., Raffeld, M., Manischewitz, J.F. & Wahl, S.M. Cytomegalovirus induction of tumor necrosis factor-alpha by human monocytes and mucosal macrophages. Journal of Clinical Investigation 90, 1642-1648 (1992).
- Yurochko, A.D. & Huang, E.-S. Human Cytomegalovirus Binding to Human Monocytes Induces Immunoregulatory Gene Expression. The Journal of Immunology 162, 4806-4816 (1999).
- Allavena, P.; et al. IL-10 prevents the differentiation of monocytes to dendritic cells but promotes their maturation to macrophages. European Journal of Immunology 28, 359-369 (1998).
- Hancock, M.H., Hook, L.M., Mitchell, J. & Nelson, J.A. Human Cytomegalovirus MicroRNAs miR-US5-1 and miR-UL112-3p Block Proinflammatory Cytokine Production in Response to NF-κB-Activating Factors through Direct Downregulation of IKKα and IKKβ. mBio 8, e00109-00117 (2017).
- Frascaroli, G.; et al. Human Cytomegalovirus Subverts the Functions of Monocytes, Impairing Chemokine-Mediated Migration and Leukocyte Recruitment. Journal of Virology 80, 7578-7589 (2006).
- Smith, M.S., Bentz, G.L., Alexander, J.S. & Yurochko, A.D. Human Cytomegalovirus Induces Monocyte Differentiation and Migration as a Strategy for Dissemination and Persistence. Journal of Virology 78, 4444-4453 (2004).
- Bentz, G.L.; et al. Human Cytomegalovirus (HCMV) Infection of Endothelial Cells Promotes Naïve Monocyte Extravasation and Transfer of Productive Virus To Enhance Hematogenous Dissemination of HCMV. Journal of Virology 80, 11539-11555 (2006).
- Sedgwick, A.D., Edwards, J.C. & Willoughby, D.A. Studies on the ability of inflammatory exudate obtained from acute and chronic phases of the inflammatory process to promote leukocyte locomotion in vitro. Inflammation 6, 13-20 (1982).
- Saederup, N., Lin, Y.C., Dairaghi, D.J., Schall, T.J. & Mocarski, E.S. Cytomegalovirus-encoded β chemokine promotes monocyte-associated viremia in the host. Proceedings of the National Academy of Sciences 96, 10881-10886 (1999).
- Baasch, S., Ruzsics, Z. & Henneke, P. Cytomegaloviruses and Macrophages—Friends and Foes From Early on? Frontiers in Immunology 11 (2020).
- McSharry, B.P., Avdic, S. & Slobedman, B. Human Cytomegalovirus Encoded Homologs of Cytokines, Chemokines and their Receptors: Roles in Immunomodulation. Viruses 4, 2448-2470 (2012).
- Billstrom, M.A., Johnson, G.L., Avdi, N.J. & Worthen, G.S. Intracellular Signaling by the Chemokine Receptor US28 during Human Cytomegalovirus Infection. Journal of Virology 72, 5535-5544 (1998).
- Gredmark, S., Tilburgs, T. & Soderberg-Naucler, C. Human cytomegalovirus inhibits cytokine-induced macrophage differentiation. J Virol 78, 10378-10389 (2004).
- Guetta, E., Guetta, V., Shibutani, T. & Epstein, S.E. Monocytes harboring cytomegalovirus: interactions with endothelial cells, smooth muscle cells, and oxidized low-density lipoprotein. Possible mechanisms for activating virus delivered by monocytes to sites of vascular injury. Circ Res 81, 8-16 (1997).
- Carlier, J.; et al. Paracrine inhibition of GM-CSF signaling by human cytomegalovirus in monocytes differentiating to dendritic cells. Blood 118, 6783-6792 (2011).
- Schieber, M. & Navdeep. ROS Function in Redox Signaling and Oxidative Stress. Current Biology 24, R453-R462 (2014).
- Chateau, M.T., Rabesandratana, H. & Caravano, R. Differentiated U937 cells and human monocytes exhibit a differential production of extracellular oxygen species: O2.- excretion versus H2O2 diffusion. FEMS Immunol Med Microbiol 13, 19-28 (1996).
- Nathan, C. antimicrobial mechanisms of macrophages. In: furth, r.v. (ed). mononuclear phagocytes. kluwer academic publishers: netherlands, 1992, pp 259-267.
- Thannickal, V.J. & Fanburg, B.L. Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol 279, L1005-1028 (2000).
- Suzuki, S.; et al. Superoxide generation by monocytes following infection with human cytomegalovirus. Immunopharmacology 37, 185-190 (1997).
- Xu, X.; et al. Human cytomegalovirus infection activates NLRP3 inflammasome by releasing mtDNA into the cytosol in human THP-1 cells. Microbiol Immunol 67, 303-313 (2023).
- Speir, E., Shibutani, T., Yu, Z.X., Ferrans, V. & Epstein, S.E. Role of reactive oxygen intermediates in cytomegalovirus gene expression and in the response of human smooth muscle cells to viral infection. Circ Res 79, 1143-1152 (1996).
- Monk, C.H. & Zwezdaryk, K.J. Host Mitochondrial Requirements of Cytomegalovirus Replication. Current Clinical Microbiology Reports 7, 115-123 (2020).
- Perera, M.R. & Sinclair, J.H. The Human Cytomegalovirus beta2.7 Long Non-Coding RNA Prevents Induction of Reactive Oxygen Species to Maintain Viral Gene Silencing during Latency. Int J Mol Sci 23 (2022).
- Papon, N.; et al. Fungal infections in transplant recipients: pros and cons of immunosuppressive and antimicrobial treatment. The Lancet Microbe 2, e6-e8 (2021).
- Papon, N.; et al. Invasive Fungal Infections among Organ Transplant Recipients: Results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clinical Infectious Diseases 50, 1101-1111 (2010).
- Wang, H.; et al. Cytomegalovirus Infection and Relative Risk of Cardiovascular Disease (Ischemic Heart Disease, Stroke, and Cardiovascular Death): A Meta-Analysis of Prospective Studies Up to 2016. Journal of the American Heart Association 6, e005025 (2017).
- Weigt, S.S., Palchevskiy, V. & Belperio, J.A. Inflammasomes and IL-1 biology in the pathogenesis of allograft dysfunction. J Clin Invest 127, 2022-2029 (2017).
- Deml, L.; et al. Stimulatory Effect of CMV Immunoglobulin on Innate Immunity and on the Immunogenicity of CMV Antigens. Transplant Direct 7, e781 (2021).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).