Submitted:
22 October 2024
Posted:
24 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Statistical Analysis
3. Results
3.1. General Characteristic of the Diagnostic Risk Groups
| General characteristics | I n=88 (33,46%) |
II n=129 (49,05%) |
III n=46 (17,49%) |
p value |
|---|---|---|---|---|
| Gender [F] | 76 (86,4) | 106 (82,2) | 34 (73,9) | ns |
| SAR | 12 (13,6) | 20 (15,5) | 6 (13) | ns |
| PAR | 5 (5,7) | 10 (7,8) | 3 (6,5) | - |
| Asthma | 10 (11,4) | 11 (8,5) | 5 (10,9) | ns |
| Smoking | 15 (16,1) | 24 (18) | 10 (21,7) | ns |
3.2. Anamnestic Data
| 2 A | I n=88 (33,46%) |
II n=129 (49,05%) |
III n=46 (17,49%) |
p value - testχ 2 |
|---|---|---|---|---|
| PN hypersensitivity (n=198, 75.29%) | 77 (87,5) | 102 (79,1) | 19 (41,3) | p < 0,001 |
|
3 (3,4) | 2 (1,6) | 0 (0) | - |
|
1 (1,1) | 0 (0) | 1 (2,2) | - |
|
2 (2,3) | 1 (0,8) | 0 (0) | - |
|
62 (70,5) | 90 (69,8) | 18 (39,1) | p < 0,001 |
|
15 (17) | 18 (14) | 3 (6,5) | - |
| CF hypersensitivity (n=105, 39.92%) | 28 (31,8) | 47 (36,4) | 30 (65,2) | p < 0,001 |
ceftriaxone (n=5) cefixime (n=1) |
23 (26,1) | 38 (29,5) | 22 (47,8) | p < 0,001 |
cefaclor (n=4) cephalexine (n=3) |
5 (5,7) | 6 (4,7) | 1 (2,2) | - |
cefamandole (n=1) |
1 (1,1) | 4 (3,1) | 7 (15,2) | - |
|
2 (2,3) | 1 (0,8) | 0 (0) | - |
| Hypersensitivity to >1 BL antibiotic (n=62, 23.57%) | 24 (27,3) | 34 (26,3) | 4 (8,7) | p <0,05 |
| Hypersensitivity to non-BL antibiotics (n=80, 30.42%) | 27 (30,7) | 42 (32,6) | 11 (23,9) | ns |
|
11 (12,5) | 22 (17,1) | 6 (13) | ns |
|
3 (3,4) | 3 (2,3) | 0 (0) | - |
|
3 (3,4) | 5 (3,9) | 2 (4,3) | - |
|
7 (8) 0 (0) |
6 (4,7) 1 (0,8) |
3 (6,5) 0 (0) |
- - |
|
9 (10,2) | 11 (8,5) | 4 (8,7) | ns |
|
4 (4,5) | 5 (3,9) | 1 (2,2) | - |
| 2 B |
I n=88 (33,46%) |
II n=129 (49,05%) |
III n=46 (17,49%) |
p value - test 2 |
| Hypersensitivity to other drugs (n=107, 40,68%) |
39 (44,3) | 52 (40,3) | 16 (34,8) | ns |
|
28 (31,8) | 37 (28,7) | 10 (21,7) | ns |
|
3 (3,4) | 5 (3,9) | 3 (6,5%) | - |
|
1 (1,1) | 2 (1,6) | 1 (2,2) | - |
|
3 (3,4) | 1 (0,8) | 2 (4,3) | - |
|
1 (1,1) | 1 (0,8) | 0 (0) | - |
| Time from BL antibiotic intake to onset of symptoms | ||||
|
11 (12,5) | 46 (35,7) | 40 (87) | p < 0,001 |
|
15 (17) | 33(25,6) | 2 (4,3) | - |
|
21 (23,9) | 8 (6,2) | 0 (0) | - |
|
36 (40,9) | 38 (29,5) | 4 (8,7) | p <0,001 |
| >1 episode of hypersensitivity to BL antibiotic (n=72, 27.38%) | 33 (37,5) | 32 (24,8) | 7 (15,2) | p < 0,001 |
| Medical intervention (n=156, 59,32%) |
25 (28,7) | 85 (66,4) | 46 (100) | p < 0,001 |
| Documented anaphylactic reaction (n=76, 28.9%) | 2 (2,3) | 34 (26,4) | 40 (87) | p < 0,001 |
| Time from hypersensitivity episode to diagnosis | ||||
|
4 (4,5) | 10 (7,8) | 7 (15,2) | ns |
|
12 (13,6) | 20 (15,5) | 19 (41,3) | p < 0,001 |
|
19 (21,6) | 52 (40,3) | 12 (26,1) | p < 0,05 |
|
13 (14,8) | 13 (10,1) | 3 (6,5) | - |
|
15 (17) | 12 (9,3) | 2 (4,3) | - |
|
23 (26,1) | 22 (17,1) | 2 (4,3) | - |
3.3. De-Labeling in the Risk Groups
3.4. Analysis of a Low-Risk Group Hypersensitivity to Beta-Lactam Antibiotics
3.4.1. Univariate Analysis of a Low-Risk Group
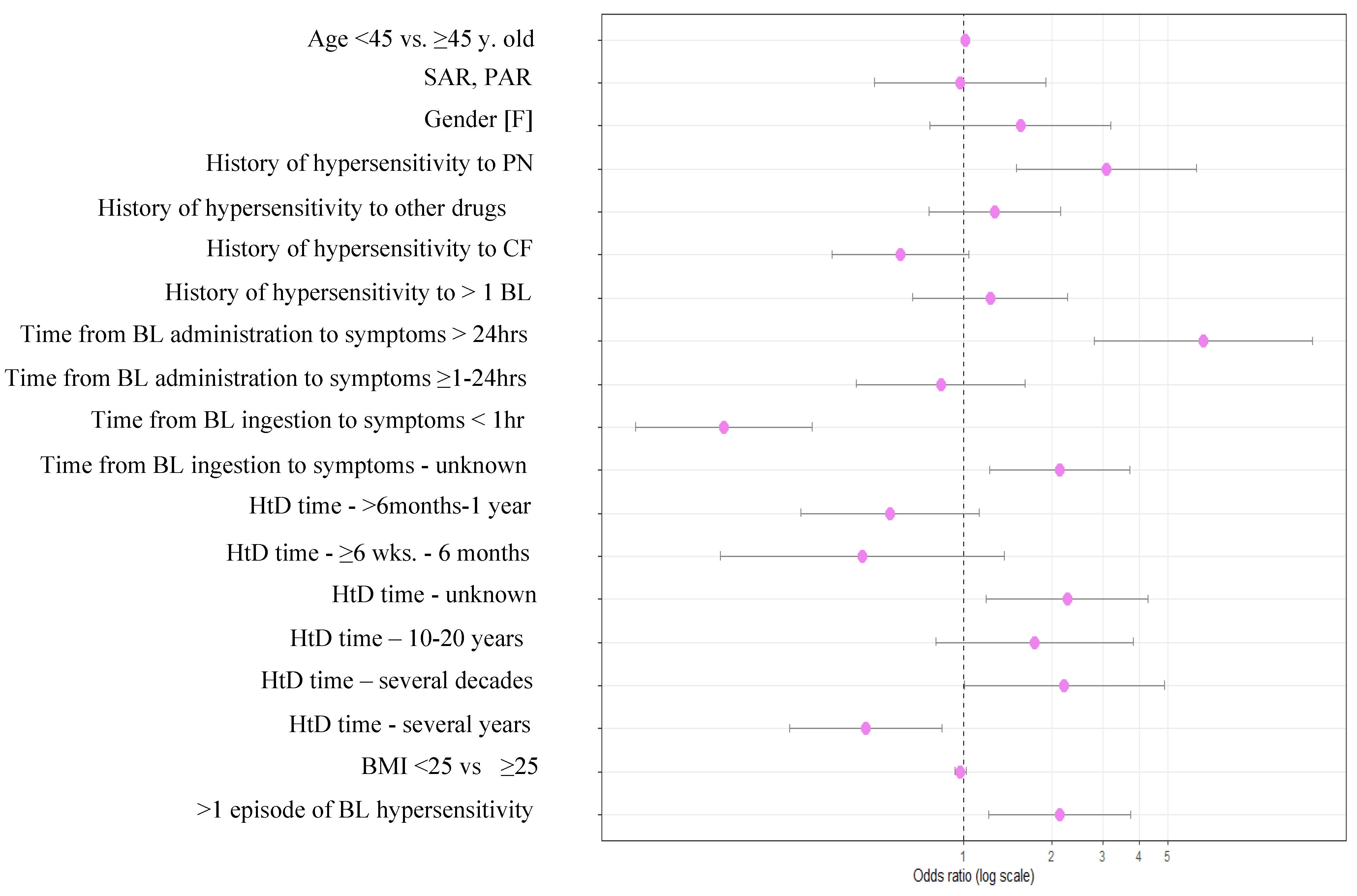
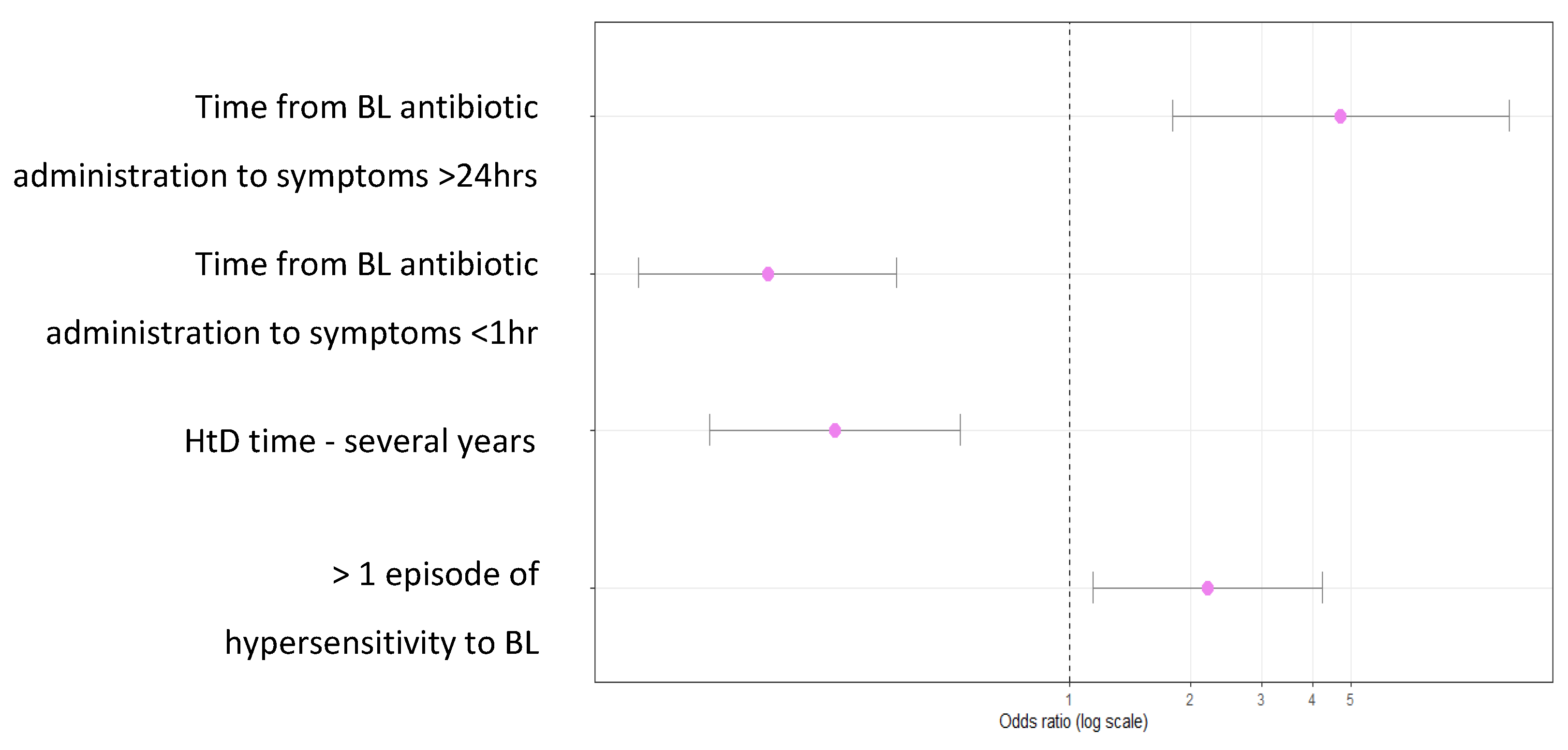
3.4.2. Multivariate Analysis of a Low-Risk Diagnostic Group
| Multivariate model | ||||
| Variable | p | OR | 95% CI | |
| HtD time- several years | < 0,01 | 0,26 | 0,13 | 0,54 |
| >1 episode of hypersensitivity to BL antibiotic | 0,02 | 2,20 | 1,15 | 4,23 |
| Time from BL antibiotic administration to symptoms <1hr | < 0,01 | 0,18 | 0,09 | 0,37 |
| Time from BL antibiotic administration to onset of symptoms >24hrs. | < 0,01 | 4,71 | 1,80 | 12,32 |
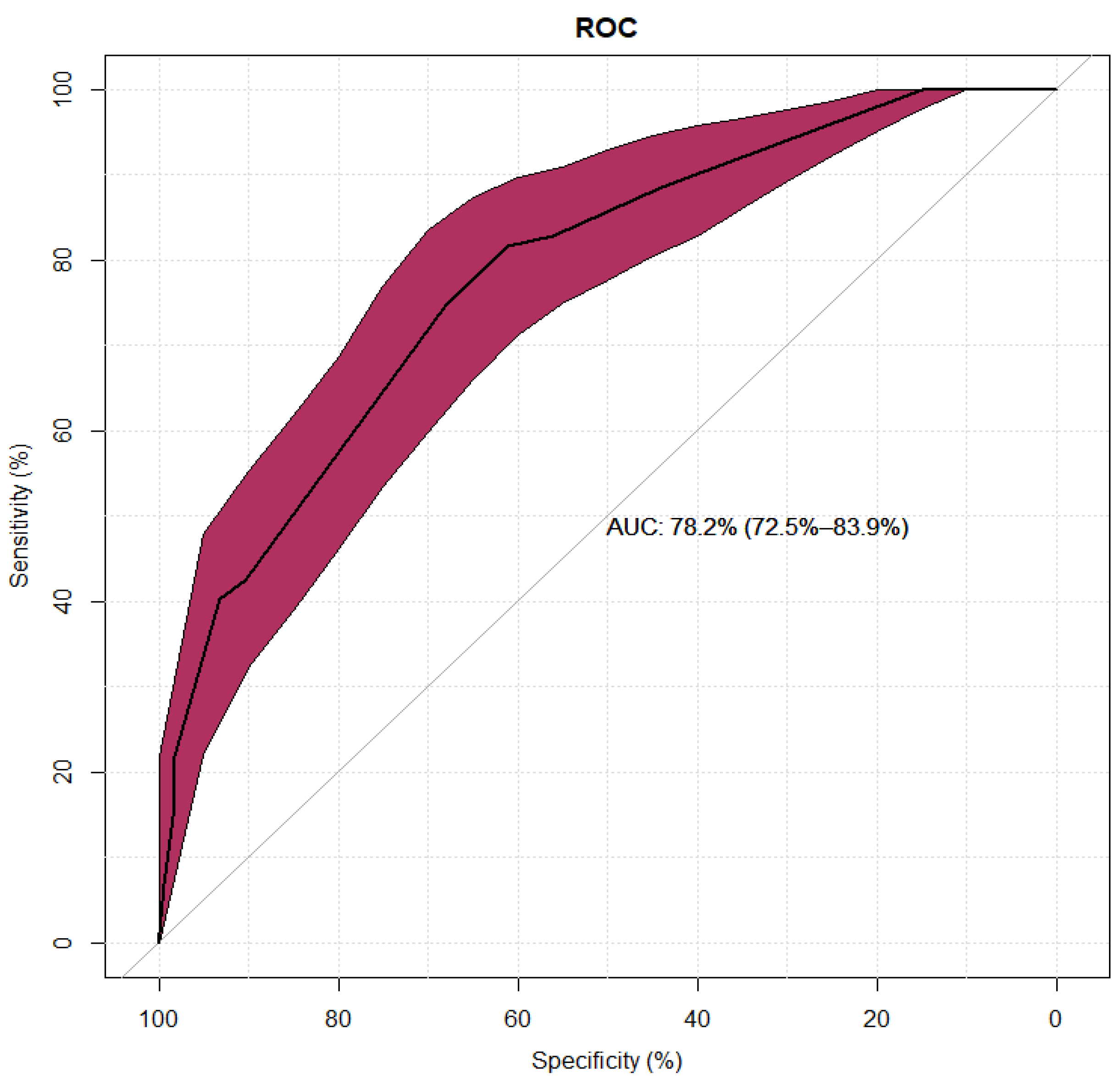
4. Discussion
4.1. Characteristics of the Low Diagnostic Risk Group
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Doña, I.; Caubet, J.C.; Brockow, K.; Doyle, M.; Moreno, E.; Terreehorst, I.; Torres, M.J. An EAACI task force report: recognising the potential of the primary care physician in the diagnosis and management of drug hypersensitivity. Clin. Transl. Allergy 2018, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Dhopeshwarkar, N.; Blumenthal, K.G.; Goss, F.; Topaz, M.; Slight, S.P.; Bates, D.W. Drug allergies documented in electronic health records of a large healthcare system. Allergy 2016, 71, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Peter, J.G.; Trubiano, J.A.; Phillips, E.J. Antibiotic allergy. Lancet 2019, 393, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Khan, D.A.; Banerji, A.; Blumenthal, K.G.; Phillips, E.J.; Solensky, R.; White, A.A.; et al. Drug allergy: A 2022 practice parameter update. J. Allergy Clin. Immunol. 2022, 150, 1333–1393. [Google Scholar] [CrossRef]
- Trubiano, J.A.; Adkinson, N.F.; Phillips, E.J. Penicillin Allergy Is Not Necessarily Forever. JAMA 2017, 318, 201–220. [Google Scholar] [CrossRef]
- Sacco, K.A.; Bates, A.; Brigham, T.J.; Imam, J.S.; Burton, M.C. Clinical outcomes following inpatient penicillin allergy testing: A systematic review and meta-analysis. Allergy 2017, 72, 1288–1296. [Google Scholar] [CrossRef]
- West, R.M.; Smith, C.J.; Pavitt, S.H.; Butler, C.C.; Howard, P.; Bates, C.; et al. “Warning: allergic to penicillin”: association between penicillin allergy status in 2.3 million NHS general practice electronic health records, antibiotic prescribing and health outcomes. J. Antimicrob. Chemother. 2019, 74, 2075–2082. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Shenoy, E.S.; Varughese, C.A.; Hurwitz, S.; Hooper, D.C.; Banerji, A. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann. Allergy Asthma Immunol. 2015, 115, 294–300.e2. [Google Scholar] [CrossRef]
- Lee, Y.; Bradley, N. Overview and Insights into Carbapenem Allergy. Pharm. 2019, 7, 110. [Google Scholar] [CrossRef]
- Trubiano, J.A.; Stone, C.A.; Grayson, M.L.; Urbancic, K.; Slavin, M.A.; Thursky, K.A.; et al. The Three C’s of Antibiotic Allergy – Classification, Cross-Reactivity and Collaboration. J. Allergy Clin. Immunol. Pract. 2017, 5, 1532. [Google Scholar] [CrossRef]
- Stone, C.A.; Trubiano, J.A.; Phillips, E.J. Testing Strategies and Predictors for Evaluating Immediate and Delayed Reactions to Cephalosporins. J. Allergy Clin. Immunol. Pract. 2021, 9, 435–444.e13. [Google Scholar] [CrossRef] [PubMed]
- Zagursky, R.J.; Pichichero, M.E. Cross-reactivity in β-Lactam Allergy. J. Allergy Clin. Immunol. Pract. 2018, 6, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Valluzzi, R.L.; Caruso, C.; Maggioletti, M.; Quaratino, D.; Gaeta, F. Cross-Reactivity and Tolerability of Cephalosporins in Patients with IgE-Mediated Hypersensitivity to Penicillins. J. Allergy Clin. Immunol. Pract. 2018, 6, 1662–1672. [Google Scholar] [CrossRef]
- Dhopeshwarkar, N.; Sheikh, A.; Doan, R.; Topaz, M.; Bates, D.W.; Blumenthal, K.G.; Zhou, L. Drug-Induced Anaphylaxis Documented in Electronic Health Records. J. Allergy Clin. Immunol. Pract. 2019, 7, 103–111. [Google Scholar] [CrossRef]
- Macy, E.; Contreras, R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: A cohort study. J. Allergy Clin. Immunol. 2014, 133, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Lu, N.; Zhang, Y.; Li, Y.; Walensky, R.P.; Choi, H.K. Risk of meticillin resistant Staphylococcus aureus and Clostridium difficile in patients with a documented penicillin allergy: population based matched cohort study. BMJ 2018, 361, k2400. [Google Scholar] [CrossRef]
- Huang, K.H.G.; Cluzet, V.; Hamilton, K.; Fadugba, O. The Impact of Reported Beta-Lactam Allergy in Hospitalized Patients With Hematologic Malignancies Requiring Antibiotics. Clin. Infect. Dis. 2018, 67, 27–33. [Google Scholar] [CrossRef]
- Li, M.; Krishna, M.T.; Razaq, S.; Pillay, D. A real-time prospective evaluation of clinical pharmaco-economic impact of diagnostic label of “penicillin allergy” in a UK teaching hospital. J. Clin. Pathol. 2014, 67, 1088–1092. [Google Scholar] [CrossRef]
- Picard, M.; Bégin, P.; Bouchard, H.; Cloutier, J.; Lacombe-Barrios, J.; Paradis, J.; Roches, A.D.; Laufer, B.; Paradis, L. Treatment of patients with a history of penicillin allergy in a large tertiary-care academic hospital. J. Allergy Clin. Immunol. Pract. 2013, 1, 252–257. [Google Scholar] [CrossRef]
- Jeffres, M.N.; Narayanan, P.P.; Shuster, J.E.; Schramm, G.E. Consequences of avoiding β-lactams in patients with β-lactam allergies. J. Allergy Clin. Immunol. 2016, 137, 1148–1153. [Google Scholar] [CrossRef]
- Stone, C.A.; Trubiano, J.; Coleman, D.T.; Rukasin, C.R.F.; Phillips, E.J. The challenge of de-labeling penicillin allergy. Allergy. 2020, 75, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Wolfson, A.R.; Huebner, E.M.; Blumenthal, K.G. Acute care beta-lactam allergy pathways: approaches and outcomes. Ann. Allergy Asthma Immunol. 2019, 123, 16–34. [Google Scholar] [CrossRef]
- Torres, M.J.; Blanca, M.; Fernandez, J.; Romano, A.; Weck, A.; Aberer, W.; Brockow, K.; Pichler, W.J.; Demoly, P.; Enda, F.; et al. Diagnosis of immediate allergic reactions to beta-lactam antibiotics. Allergy 2003, 58, 961–972. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Blanca, M.; Torres, M.J.; Bircher, A.; Aberer, W.; Brockow, K.; et al. ENDA; EAACI. Diagnosis of nonimmediate reactions to β-lactam antibiotics. Allergy 2004, 59, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Demoly, P.; Adkinson, N.F.; Brockow, K.; Castells, M.; Chiriac, A.M.; Greenberger, P.A.; Khan, D.A.; Lang, D.M.; Park, H.; Pichler, W.; et al. International Consensus on drug allergy. Allergy 2014, 69, 420–437. [Google Scholar] [CrossRef]
- Romano, A.; Atanaskovic-Markovic, M.; Barbaud, A.; Bircher, A.J.; Brockow, K.; Caubet, J.C.; Celik, G.; Cernadas, J.; Chiriac, A.; Demoly, P.; et al. Towards a more precise diagnosis of hypersensitivity to beta-lactams — an EAACI position paper. Allergy 2020, 75, 1300–1315. [Google Scholar] [CrossRef]
- Stone, C.A., Jr.; Stollings, J.L.; Lindsell, C.J.; Dear, M.L.; Buie, R.B.; Rice, T.W.; Phillips, E.J. Risk-stratified Management to Remove Low-Risk Penicillin Allergy Labels in the ICU. Am. J. Respir. Crit. Care Med. 2020, 201, 1572–1575. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Huebner, E.M.; Fu, X.; Li, Y.; Bhattacharya, G.; Levin, A.S.; Mancini, C.M.; Slawski, B.R.; Banerji, A. Risk-based pathway for outpatient penicillin allergy evaluations. J. Allergy Clin. Immunol. Pract. 2019, 7, 2411–2414.e1. [Google Scholar] [CrossRef] [PubMed]
- Koo, G.; Yu, R.; Phillips, E.J.; Stone, C.A. Retrospective stratification of cephalosporin allergy label risk using validated penicillin allergy frameworks. J. Allergy Clin. Immunol. Pract. 2022, 10, 2472–2475.e1. [Google Scholar] [CrossRef]
- Trubiano, J.A.; Vogrin, S.; Chua, K.Y.L.; Bourke, J.; Yun, J.; Douglas, A.; Stone, C.A.; Yu, R.; Groenendijk, L.; Holmes, N.E.; et al. Development and Validation of a Penicillin Allergy Clinical Decision Rule. JAMA Intern. Med. 2020, 180, 1. [Google Scholar] [CrossRef]
- Devchand, M.; Urbancic, K.F.; Khumra, S.; Douglas, A.P.; Smibert, O.; Cohen, E.; Sutherland, M.; Phillips, E.J.; Trubiano, J.A. Pathways to improved antibiotic allergy and antimicrobial stewardship practice: The validation of a beta-lactam antibiotic allergy assessment tool. J. Allergy Clin. Immunol. Pract. 2019, 7, 1063–1065.e5. [Google Scholar] [CrossRef] [PubMed]
- Phillips, E.J.; Demoly, P.; Torres, M.J. Addressing beta-lactam allergy: A time for action. Allergy 2022, 77, 1091–1093. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, E.S.; Macy, E.; Rowe, T.; Blumenthal, K.G. Evaluation and Management of Penicillin Allergy: A Review. JAMA 2019, 321, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Gaeta, F.; Valluzzi, R.L.; Maggioletti, M.; Zaffiro, A.; Caruso, C.; Quaratino, D. IgE-mediated hypersensitivity to cephalosporins: Cross-reactivity and tolerability of alternative cephalosporins. J. Allergy Clin. Immunol. 2015, 136, 685–691.e3. [Google Scholar] [CrossRef] [PubMed]
- Inglis, J.M.; Caughey, G.E.; Smith, W.; Shakib, S. Documentation of penicillin adverse drug reactions in electronic health records: inconsistent use of allergy and intolerance labels. Intern. Med. J. 2017, 47, 1292–1297. [Google Scholar] [CrossRef]
- Albin, S.; Agarwal, S. Prevalence and characteristics of reported penicillin allergy in an urban outpatient adult population. Allergy Asthma Proc. 2014, 35, 489. [Google Scholar] [CrossRef]
- Vyles, D.; Chiu, A.; Simpson, P.; Nimmer, M.; Adams, J.; Brousseau, D.C. Parent-Reported Penicillin Allergy Symptoms in the Pediatric Emergency Department. Acad. Pediatr. 2017, 17, 251–255. [Google Scholar] [CrossRef]
- Zembles, T.; Zhan, Y.; Chiu, A.; Brousseau, D.C.; Vyles, D. Cephalosporin allergy symptoms in children presenting to a pediatric emergency department. Ann. Allergy Asthma Immunol. 2021, 127, 259–260. [Google Scholar] [CrossRef]
- Touati, N.; Cardoso, B.; Delpuech, M.; Bazire, R.; El Kara, N.; Ouali, D.; Demoly, P.; Chiriac, A.-M. Cephalosporin Hypersensitivity: Descriptive Analysis, Cross-Reactivity, and Risk Factors. J. Allergy Clin. Immunol. Pract. 2021, 9, 1994–2000.e5. [Google Scholar] [CrossRef]
- Macy, E.; Contreras, R. Adverse reactions associated with oral and parenteral use of cephalosporins: A retrospective population-based analysis. J. Allergy Clin. Immunol. 2015, 135, 745–752.e5. [Google Scholar] [CrossRef]
- Macy, E.M.; Chen, L.H. The Incidence of Anaphylaxis Associated with Oral and Parenteral Penicillin-Class Antibiotic Exposures. J. Allergy Clin. Immunol. 2017, 139, AB33. [Google Scholar] [CrossRef]
- Yang, M.S.; Kang, D.Y.; Seo, B.; Park, H.J.; Park, S.Y.; Kim, M.; Park, K.H.; Koo, S.; Nam, Y.; Kim, S.; et al. Incidence of cephalosporin-induced anaphylaxis and clinical efficacy of screening intradermal tests with cephalosporins: A large multicenter retrospective cohort study. Allergy 2018, 73, 1833–1841. [Google Scholar] [CrossRef] [PubMed]
- Bogas, G.; Doña, I.; Dionicio, J.; Fernández, T.D.; Mayorga, C.; Boteanu, C.; Montañez, M.I.; Al-Ahmad, M.; Rondón, C.; Moreno, E.; et al. Diagnostic Approach of Hypersensitivity Reactions to Cefazolin in a Large Prospective Cohort. J. Allergy Clin. Immunol. Pract 2021, 9, 4421–4430.e4. [Google Scholar] [CrossRef]
- Jagpal, P.K.; Alshareef, S.; Marriott, J.F.; Thirumala Krishna, M. Characterization, epidemiology and risk factors of multiple drug allergy syndrome and multiple drug intolerance syndrome: A systematic review. Clin. Transl. Allergy. 2022, 12. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Li, Y.; Acker, W.W.; Chang, Y.; Banerji, A.; Ghaznavi, S.; Camargo, C.A.; Zhou, L. Multiple drug intolerance syndrome and multiple drug allergy syndrome: Epidemiology and associations with anxiety and depression. Allergy 2018, 73, 2012–2023. [Google Scholar] [CrossRef]
- Voelker, D.; D’Netto, M.; Park, M. Multiple drug allergies as a risk factor for IgE mediated penicillin drug allergy in a pediatric population. J. Allergy Clin. Immunol. 2021, 147, AB5. [Google Scholar] [CrossRef]
- Park, C.S.; Yang, M.S.; Kang, D.Y.; Park, H.J.; Park, S.Y.; Nam, Y.H.; Drug Allergy Work Group of, K.A.A.A.C.I.; et al. Risk factors of beta-lactam anaphylaxis in Korea: A 6-year multicenter retrospective adult case-control study. World Allergy Organ. J. 2021, 14. [Google Scholar] [CrossRef]
- Piotin, A.; Godet, J.; Trubiano, J.A.; Grandbastien, M.; Guénard-Bilbault, L.; de Blay, F.; Metz-Favre, C. Predictive factors of amoxicillin immediate hypersensitivity and validation of PEN-FAST clinical decision rule. Ann. Allergy Asthma Immunol. 2022, 128, 27–32. [Google Scholar] [CrossRef]
- Courtemanche, J.; Baril, L.; Clément, A.; Bédard, M.A.; Plourde, M.; Émond, M. Is it possible to identify patients at low risk of having a true penicillin allergy? CJEM 2022, 24, 366–368. [Google Scholar] [CrossRef]
- Chiriac, A.M.; Wang, Y.; Schrijvers, R.; Bousquet, P.J.; Mura, T.; Molinari, N.; Demoly, P. Designing Predictive Models for Beta-Lactam Allergy Using the Drug Allergy and Hypersensitivity Database. J. Allergy Clin. Immunol. Pract. 2018, 6, 139–148.e2. [Google Scholar] [CrossRef]
- Stevenson, B.; Trevenen, M.; Klinken, E.; Smith, W.; Yuson, C.; Katelaris, C.; Perram, F.; Burton, P.; Yun, J.; Cai, F.; et al. Multicenter Australian Study to Determine Criteria for Low-and High-Risk Penicillin Testing in Outpatients. J. Allergy Clin. Immunol. Pract. 2019, 8, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Siew, L.Q.C.; Li, P.H.; Watts, T.J.; Thomas, I.; Ue, K.L.; Caballero, M.R.; Rutkowski, K.; Till, S.J.; Pillai, P.; Haque, R. Identifying Low-Risk Beta-Lactam Allergy Patients in a UK Tertiary Centre. J. Allergy Clin. Immunol. Pract. 2019, 7, 2173–2181.e1. [Google Scholar] [CrossRef]
- Soria, A.; Autegarden, E.; Amsler, E.; Gaouar, H.; Vial, A.; Francès, C.; Autegarden, J.-E. A clinical decision-making algorithm for penicillin allergy. Ann. Med. 2017, 49, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Devchand, M.; Kirkpatrick, C.M.J.; Stevenson, W.; Garrett, K.; Perera, D.; Khumra, S.; Urbancic, K.; Grayson, M.L.; A Trubiano, J. Evaluation of a pharmacist-led penicillin allergy de-labelling ward round: a novel antimicrobial stewardship intervention. J. Antimicrob. Chemother. 2019, 74, 1725–1730. [Google Scholar] [CrossRef] [PubMed]
- Owens, R.; Sandoe, J.; Whyte, A.F.; Wilcock, M.; West, R.; Stonell, R.; Slatter, M.; Powell, N. A randomized evaluation of an antibiotic allergy assessment tool for supporting penicillin allergy de-labelling by non-allergists. Clin. Exp. Allergy. 2021, 51, 1246–1249. [Google Scholar] [CrossRef]
- Dunham, T.B.; Gardner, R.M.; Lippner, E.A.; Fasani, D.E.; Moir, E.; Halpern-Felsher, B.; Sundaram, V.; Liu, A.Y. Digital Antibiotic Allergy Decision Support Tool Improves Management of β-Lactam Allergies. J. Allergy Clin. Immunol. Pract. 2023, 11, 1243–1252.e6. [Google Scholar] [CrossRef]
- Wells, D.; DeNiro, K.L.; Ramsey, A. Telemedicine in Drug Hypersensitivity. Immunol. Allergy Clin. North. Am. 2022, 42, 323–333. [Google Scholar] [CrossRef]
- Allen, H.I.; Gillespie, P.; Vazquez-Ortiz, M.; Murphy, A.W.; Moylett, E.M. A cost-analysis of outpatient paediatric penicillin allergy de-labelling using telemedicine. Clin. Exp. Allergy 2021, 51, 495–498. [Google Scholar] [CrossRef]
| Single factor model | ||||
|---|---|---|---|---|
| Variable | p | OR | 95% CI | |
| Gender [F] | 0,22 | 1,56 | 0,77 | 3,19 |
| Age <45 vs ≥45 years old | 0,26 | 1,01 | 0,99 | 1,03 |
| BMI <25 vs ≥25 | 0,24 | 0,97 | 0,93 | 1,02 |
| SAR, PAR | 0,92 | 0,97 | 0,49 | 1,91 |
| Hypersensitivity to PN | < 0,001 | 3,08 | 1,52 | 6,27 |
| Hypersensitivity to CF | 0,07 | 0,60 | 0,35 | 1,04 |
| Hypersensitivity to >1 BL antibiotic | 0,5103 | 1,229 | 0,666 | 2,267 |
| Hypersensitivity to other drugs | 0,35 | 1,28 | 0,76 | 2,15 |
| Time from BL administration to symptoms <1hr | < 0,001 | 0,15 | 0,07 | 0,30 |
| Time from BL administration too symptoms ≥1h-24h | 0,59 | 0,83 | 0,43 | 1,63 |
| Time from BL administration to symptoms >24hrs | < 0,001 | 6,64 | 2,80 | 15,74 |
| Time from BL antibiotic administration to symptoms - unknown | 0,01 | 2,13 | 1,23 | 3,70 |
| >1 episode of hypersensitivity to BL antibiotic | 0,01 | 2,13 | 1,22 | 3,73 |
| HtD time ≥6 wks. - 6 months. | 0,16 | 0,45 | 0,15 | 1,37 |
| HtD time >6 months-1 year | 0,11 | 0,56 | 0,28 | 1,13 |
| HtD time – several years | 0,01 | 0,46 | 0,25 | 0,84 |
| HtD time – 10-20 years | 0,16 | 1,75 | 0,80 | 3,82 |
| HtD time – several decades | 0,05 | 2,21 | 1,00 | 4,86 |
| HtD time – unknown | 0,01 | 2,26 | 1,19 | 4,30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





