Submitted:
28 October 2024
Posted:
30 October 2024
You are already at the latest version
Abstract
Purpose: To evaluate the impact of intravitreal bevacizumab (IVB) therapy on anatomical and visual outcomes in patients with macular neovascularization (MNV) secondary to chronic cen-tral serous chorioretinopathy (cCSC). Methods: This retrospective observational study reviewed the medical records of treatment-naïve patients diagnosed with cCSC complicated by MNV, treated with IVB injections over a 5-year period. The presence of MNV was confirmed using op-tical coherence tomography angiography (OCTA). Best-corrected visual acuity (BCVA), subfoveal choroidal thickness (SFCT), and subretinal fluid (SRF) were recorded pre- and post-IVB treat-ment. Results: Twenty-two eyes of 22 patients (mean age, 68 ± 11 years) were included. After a mean follow-up of 21.0 ± 14.6 months, SRF significantly decreased from baseline (176.86 ± 115.62 µm) to the final follow-up (80.95 ± 87.32 µm, P = 0.003). Greater SRF reduction was associated with more injections (>7) (P = 0.047). However, no significant changes were observed in BCVA (P > 0.05) or SFCT (P > 0.05), irrespective of follow-up duration or injection frequency. Complete res-olution of SRF was achieved in 9 patients (40.9%), and a significantly greater reduction in SFCT was observed in complete responders compared to non-responders (P = 0.03). Conclusion: IVB therapy significantly reduced SRF in cCSC patients with secondary MNV, though it did not lead to visual improvement or significant changes in SFCT. However, greater choroidal thinning in patients with complete fluid resorption may suggest distinct underlying mechanisms or alterna-tive sources of subretinal fluid beyond the MNV itself.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study setting, duration and participants recruitment
2.2. Data collection
2.3. Image acquisition and analysis
2.4. Anti-VEGF Treatment
2.5. Outcome Measures
2.6. Statistical analysis
3. Results
3.1. Baseline Characteristics
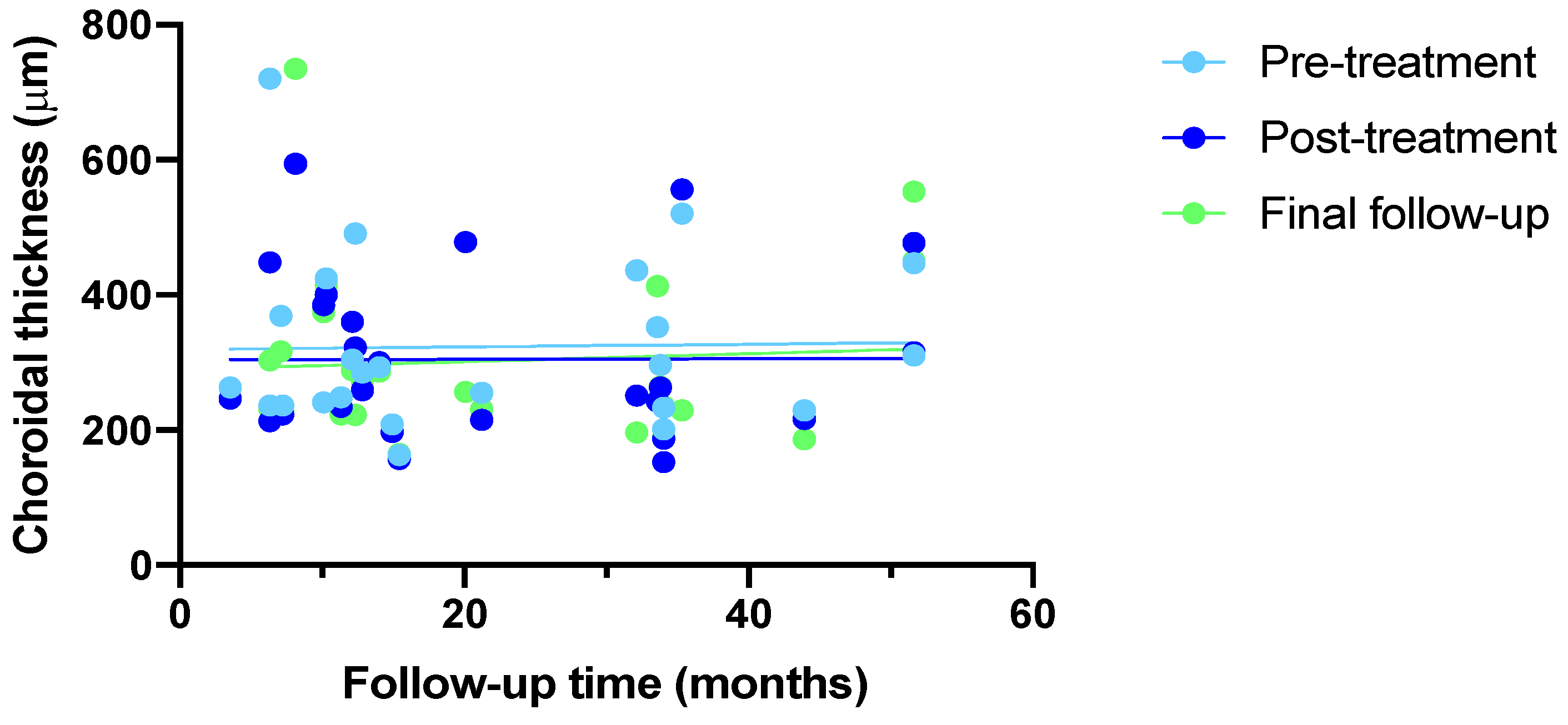
3.1. Anatomic and Visual Outcomes Over Time
3.2. Anatomic and Visual Outcomes Relative to Number of Injections
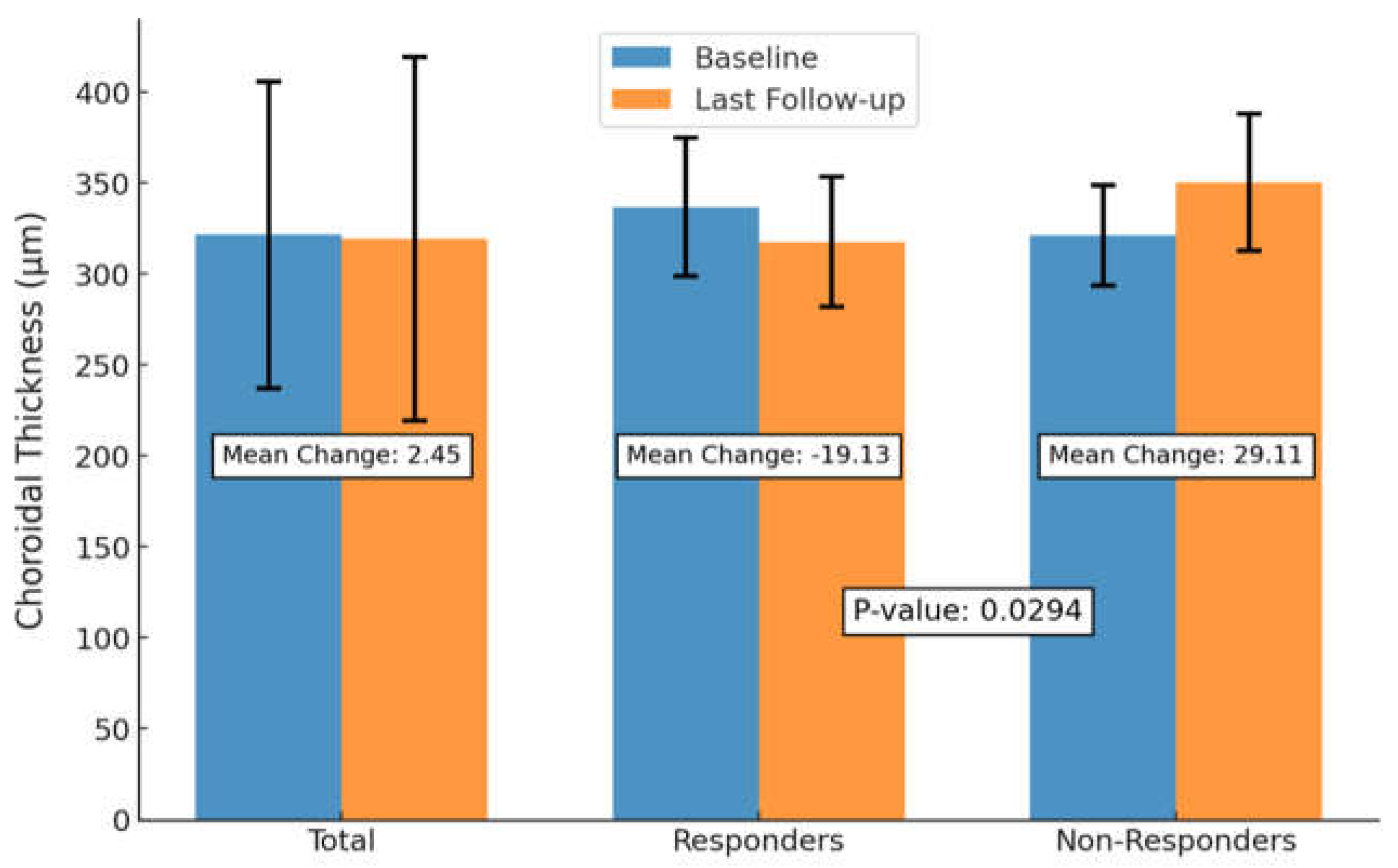
3.3. Anatomic and Visual Outcomes Between SRF Responders and Non-Responders
3.4. Anatomic and Visual Outcomes Relative to Number of Injections
3.5. Anatomic and Visual Outcomes Between SRF Responders and Non-Responders
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nicholson B, Noble J, Forooghian F, Meyerle C. Central serous chorioretinopathy: update on pathophysiology and treatment. Surv Ophthalmol Mar-Apr. 2013;58(2):103-126. doi:10.1016/j.survophthal.2012.07.004.
- Yannuzzi LA. Central serous chorioretinopathy: a personal perspective. Am J Ophthalmol Mar. 2010;149(3):361-363. doi:10.1016/j.ajo.2009.11.017.
- Central serous chorioretinopathy: Towards an evidence-based treatment guideline - ScienceDirect. Accessed October 11, 2024. https://www.sciencedirect.com/science/article/pii/S1350946218300946#sec3.
- Lee GI, Kim AY, Kang SW. Risk Factors and Outcomes of Choroidal Neovascularization Secondary to Central Serous Chorioretinopathy. Sci Rep Mar. 2019;9(1). doi:10.1038/s41598-019-40406-y.
- Daruich A, Matet A, Dirani A. Central serous chorioretinopathy: Recent findings and new physiopathology hypothesis. Prog Retin Eye Res. Sep. 2015;48:82-118. doi:10.1016/j.preteyeres.2015.05.003.
- Cheung CMG, Lee WK, Koizumi H, Dansingani K, Lai TYY, Freund KB. Pachychoroid disease. Eye Lond Engl. 2019;33(1):14-33. doi:10.1038/s41433-018-0158-4.
- Spaide RF, Gemmy Cheung CM, Matsumoto H, et al. Venous overload choroidopathy: A hypothetical framework for central serous chorioretinopathy and allied disorders. Prog Retin Eye Res. 2022;86:100973. doi:10.1016/j.preteyeres.2021.100973.
- Imamura Y, Fujiwara T, Margolis R, Spaide RF. Enhanced depth imaging optical coherence tomography of the choroid in central serous chorioretinopathy. Retina Nov-Dec. 2009;29(10):1469-1473. doi:10.1097/IAE.0b013e3181be0a83.
- Zarnegar A, Ong J, Matsyaraja T, Arora S, Chhablani J. Pathomechanisms in central serous chorioretinopathy: A recent update. Int J Retina Vitr. 2023;9(1):3. doi:10.1186/s40942-023-00443-2.
- Lai TYY, Staurenghi G, Lanzetta P, et al. EFFICACY AND SAFETY OF RANIBIZUMAB FOR THE TREATMENT OF CHOROIDAL NEOVASCULARIZATION DUE TO UNCOMMON CAUSE: Twelve-Month Results of the MINERVA Study. Retina Phila Pa. 2018;38(8):1464-1477. doi:10.1097/IAE.0000000000001744.
- Razavi S, Souied EH, Darvizeh F, Querques G. Assessment of Choroidal Topographic Changes by Swept-Source Optical Coherence Tomography After Intravitreal Ranibizumab for Exudative Age-Related Macular Degeneration. Am J Ophthalmol. 2015;160(5):1006-1013. doi:10.1016/j.ajo.2015.08.009.
- Sacconi R, Tomasso L, Corbelli E. Early response to the treatment of choroidal neovascularization complicating central serous chorioretinopathy: a OCT-angiography study. Eye Lond Nov. 2019;33(11):1809-1817. doi:10.1038/s41433-019-0511-2.
- Schworm B, Luft N, Keidel LF. Response of neovascular central serous chorioretinopathy to an extended upload of anti-VEGF agents. Graefes Arch Clin Exp Ophthalmol May. 2020;258(5):1013-1021. doi:10.1007/s00417-020-04623-w.
- Kim GA, Rim TH, Lee SC, et al. Clinical characteristics of responders to intravitreal bevacizumab in central serous chorioretinopathy patients. Eye. 2015;29(6):732-741. doi:10.1038/eye.2015.58.
- Koizumi H, Kano M, Yamamoto A, et al. Subfoveal Choroidal Thickness during Aflibercept Therapy for Neovascular Age-Related Macular Degeneration: Twelve-Month Results. Ophthalmology. 2016;123(3):617-624. doi:10.1016/j.ophtha.2015.10.039.
- Ting DSW, Ng WY, Ng SR, et al. Choroidal Thickness Changes in Age-Related Macular Degeneration and Polypoidal Choroidal Vasculopathy: A 12-Month Prospective Study. Am J Ophthalmol. 2016;164:128-136.e1. doi:10.1016/j.ajo.2015.12.024.
- Inan S, Baysal Z, Inan UU. Long-Term Changes in Submacular Choroidal Thickness after Intravitreal Ranibizumab Therapy for Neovascular Age-Related Macular Degeneration: 14-Mo Follow-Up. Curr Eye Res Apr. 2020;45(4):527-528. doi:10.1080/02713683.2019.1675174.
- van Rijssen TJ, van Dijk EHC, Yzer S, et al. Central serous chorioretinopathy: Towards an evidence-based treatment guideline. Prog Retin Eye Res. 2019;73:100770. doi:10.1016/j.preteyeres.2019.07.003.
- Shiragami C, Takasago Y, Osaka R. Clinical Features of Central Serous Chorioretinopathy With Type 1 Choroidal Neovascularization. Am J Ophthalmol Sep. 2018;193:80-86. doi:10.1016/j.ajo.2018.06.009.
- Mrejen S, Balaratnasingam C, Kaden TR. Long-term Visual Outcomes and Causes of Vision Loss in Chronic Central Serous Chorioretinopathy. Ophthalmol Apr. 2019;126(4):576-588. doi:10.1016/j.ophtha.2018.12.048.
- Lejoyeux R, Behar-Cohen F, Mantel I. Type one macular neovascularization in central serous chorioretinopathy: Short-term response to anti-vascular endothelial growth factor therapy. Eye. Published online September 28, 2021.
- Su Y, Zhang X, Gan Y, Ji Y, Wen F. Characteristics and Associated Factors of Flat Irregular Pigment Epithelial Detachment With Choroidal Neovascularization in Chronic Central Serous Chorioretinopathy. Front Med. 2021;8:687023. doi:10.3389/fmed.2021.687023.
- A pilot study of enhanced depth imaging optical coherence tomography of the choroid in normal eyes - PubMed. Accessed February 3, 2024. https://pubmed.ncbi.nlm.nih.gov/19232559/.
- Lim SJ, Roh MI, Kwon OW. INTRAVITREAL BEVACIZUMAB INJECTION FOR CENTRAL SEROUS CHORIORETINOPATHY. RETINA. 2010;30(1):100. doi:10.1097/IAE.0b013e3181bcf0b4.
- Cozzupoli GM, Sacconi R, Tombolini B, et al. Long-term predictors of anti-VEGF treatment response in patients with neovascularization secondary to CSCR: a longitudinal study. Graefes Arch Clin Exp Ophthalmol. 2024;262(1):73-80. doi:10.1007/s00417-023-06255-2.
- Romdhane K, Zola M, Matet A. Predictors of treatment response to intravitreal anti-vascular endothelial growth factor (anti-VEGF) therapy for choroidal neovascularisation secondary to chronic central serous chorioretinopathy. Br J Ophthalmol Jul. 2020;104(7):910-916. doi:10.1136/bjophthalmol-2019-314625.
- Padron-Perez N, Arias L, Rubio M. Changes in Choroidal Thickness After Intravitreal Injection of Anti-Vascular Endothel ial Growth Factor in Pachychoroid Neovasculopathy. Invest Ophthalmol Vis Sci Feb. 2018;59(2):1119-1124. doi:10.1167/iovs.17-22144.
- Intravitreal Brolucizumab for Pachychoroid Neovasculopathy Associated With Chronic Central Serous Chorioretinopathy - PMC. Accessed October 14, 2024. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10732086/.
- Song YY, Yu HY, Baek SK, Lee YH, Lee MW. Short-term effect of anti-VEGF for chronic central serous chorioretinopathy according to the presence of choroidal neovascularization using optical coherence tomography angiography. PLOS ONE. 2021;16(1):e0245342. doi:10.1371/journal.pone.0245342.
- Hagag AM, Chandra S, Khalid H, et al. Multimodal Imaging in the Management of Choroidal Neovascularization Secondary to Central Serous Chorioretinopathy. J Clin Med. 2020;9(6):1934. doi:10.3390/jcm9061934.
- CLINICAL FEATURES OF FLAT IRREGULAR PIGMENT EPITHELIAL DETACHMENT ASSOCIATED WITH CHOROIDAL NEOVASCULARIZATION IN CHRONIC CENTRAL SEROUS CHORIORETINOPATHY - PubMed. Accessed October 14, 2024. https://pubmed.ncbi.nlm.nih.gov/32251241/.
- J M, C Z, S Z, et al. Predictors of anti-VEGF efficacy in chronic central serous chorioretinopathy based on intraocular cytokine levels and pigment epithelium detachment subtypes. Acta Ophthalmol (Copenh). 2022;100(7). doi:10.1111/aos.15109.
- Chen YC, Chen SN. Three-year follow-up of choroidal neovascularisation in eyes of chronic central serous chorioretinopathy. Br J Ophthalmol. 2020;104(11):1561-1566. doi:10.1136/bjophthalmol-2019-315302.
- Correlation between Choroidal Neovascularization Shown by OCT Angiography and Choroidal Thickness in Patients with Chronic Central Serous Chorioretinopathy - PMC. Accessed February 2, 2024. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5646334/.
- Quantitative Analysis of the Activity in Choroidal Neovascularizations after a Single Anti-VEGF Injection: OCT Versus OCT Angiography - PubMed. Accessed June 27, 2024. https://pubmed.ncbi.nlm.nih.gov/33784223/.
- Chhablani J, Kozak I, Pichi F, et al. OUTCOMES OF TREATMENT OF CHOROIDAL NEOVASCULARIZATION ASSOCIATED WITH CENTRAL SEROUS CHORIORETINOPATHY WITH INTRAVITREAL ANTIANGIOGENIC AGENTS. RETINA. 2015;35(12):2489. doi:10.1097/IAE.0000000000000655.
- Chhablani J, Pichi F, Silva R, et al. ANTIANGIOGENICS IN CHOROIDAL NEOVASCULARIZATION ASSOCIATED WITH LASER IN CENTRAL SEROUS CHORIORETINOPATHY. RETINA. 2016;36(5):901. doi:10.1097/IAE.0000000000000804.
- Peiretti E, Caminiti G, Serra R, Querques L, Pertile R, Querques G. ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR THERAPY VERSUS PHOTODYNAMIC THERAPY IN THE TREATMENT OF CHOROIDAL NEOVASCULARIZATION SECONDARY TO CENTRAL SEROUS CHORIORETINOPATHY. RETINA. 2018;38(8):1526. doi:10.1097/IAE.0000000000001750.
- Lee GW, Roh HC, Kang SW, Kim AY, Noh H, Choi KJ. The implications of subretinal fluid in pachychoroid neovasculopathy. Sci Rep. 2021;11(1):4066. doi:10.1038/s41598-021-83650-x.
- Optical Coherence Tomography Angiography Signs of Vascular Abnormalization With Antiangiogenic Therapy for Choroidal Neovascularization - American Journal of Ophthalmology. Accessed June 27, 2024. https://www.ajo.com/article/S0002-9394(15)00200-7/abstract.
- Wu J, Zhang J. Neovascular Remodeling and Subretinal Fibrosis as Biomarkers for Predicting Incomplete Response to Anti-VEGF Therapy in Neovascular Age-Related Macular Degeneration. Front Biosci Landmark Ed Apr. 2022;27(4). doi:10.31083/j.fbl2704135.
- Demirel S, Yanık Ö, Nalcı H, Batıoğlu F, Özmert E. The use of optical coherence tomography angiography in pachychoroid spectrum diseases: a concurrent comparison with dye angiography. Graefes Arch Clin Exp Ophthalmol Albrecht Von Graefes Arch Klin Exp Ophthalmol. 2017;255(12):2317-2324. doi:10.1007/s00417-017-3793-8.
- Carnevali A, Capuano V, Sacconi R, et al. OCT Angiography of Treatment-Naïve Quiescent Choroidal Neovascularization in Pachychoroid Neovasculopathy. Ophthalmol Retina. 2017;1(4):328-332. doi:10.1016/j.oret.2017.01.003.
- Bousquet E, Bonnin S, Mrejen S, Krivosic V, Tadayoni R, Gaudric A. OPTICAL COHERENCE TOMOGRAPHY ANGIOGRAPHY OF FLAT IRREGULAR PIGMENT EPITHELIUM DETACHMENT IN CHRONIC CENTRAL SEROUS CHORIORETINOPATHY. Retina Phila Pa. 2018;38(3):629-638. doi:10.1097/IAE.0000000000001580.
- Chhablani J, Cohen FB, Central Serous Chorioretinopathy International Group. Multimodal Imaging-Based Central Serous Chorioretinopathy Classification. Ophthalmol Retina. 2020;4(11):1043-1046. doi:10.1016/j.oret.2020.07.026.
- Weng S, Mao L, Yu S, Gong Y, Cheng L, Chen X. Detection of Choroidal Neovascularization in Central Serous Chorioretinopathy Using Optical Coherence Tomographic Angiography. Ophthalmol J Int Ophtalmol Int J Ophthalmol Z Augenheilkd. 2016;236(2):114-121. doi:10.1159/000448630.
- 3. Hage R, Mrejen S, Krivosic V, et al. Flat irregular retinal pigment epithelium detachments in chronic central serous chorioretinopathy and choroidal neovascularization. Am J Ophthalmol. 2015. - Google Search. Accessed February 2, 2024. https://www.google.com/search?q=3.+Hage+R%2C+Mrejen+S%2C+Krivosic+V%2C+et+al.+Flat+irregular+retinal+pigment+epithelium+detachments+in+chronic+central+serous+chorioretinopathy+and+choroidal+neovascularization.+Am+J+Ophthalmol.+2015.&rlz=1C5CHFA_enUS1069US1069&oq=3.%09Hage+R%2C+Mrejen+S%2C+Krivosic+V%2C+et+al.+Flat+irregular+retinal+pigment+epithelium+detachments+in+chronic+central+serous+chorioretinopathy+and+choroidal+neovascularization.+Am+J+Ophthalmol.+2015.&gs_lcrp=EgZjaHJvbWUyBggAEEUYOdIBBzI1NmowajeoAgCwAgA&sourceid=chrome&ie=UTF-8.
- Quaranta-El Maftouhi M, El Maftouhi A, Eandi CM. Chronic central serous chorioretinopathy imaged by optical coherence tomographic angiography. Am J Ophthalmol. 2015;160(3):581-587.e1. doi:10.1016/j.ajo.2015.06.016.
- Bonini Filho MA, de Carlo TE, Ferrara D, et al. Association of Choroidal Neovascularization and Central Serous Chorioretinopathy With Optical Coherence Tomography Angiography. JAMA Ophthalmol. 2015;133(8):899-906. doi:10.1001/jamaophthalmol.2015.1320.
- Costanzo E, Cohen SY, Miere A, et al. Optical Coherence Tomography Angiography in Central Serous Chorioretinopathy. J Ophthalmol. 2015;2015:134783. doi:10.1155/2015/134783.
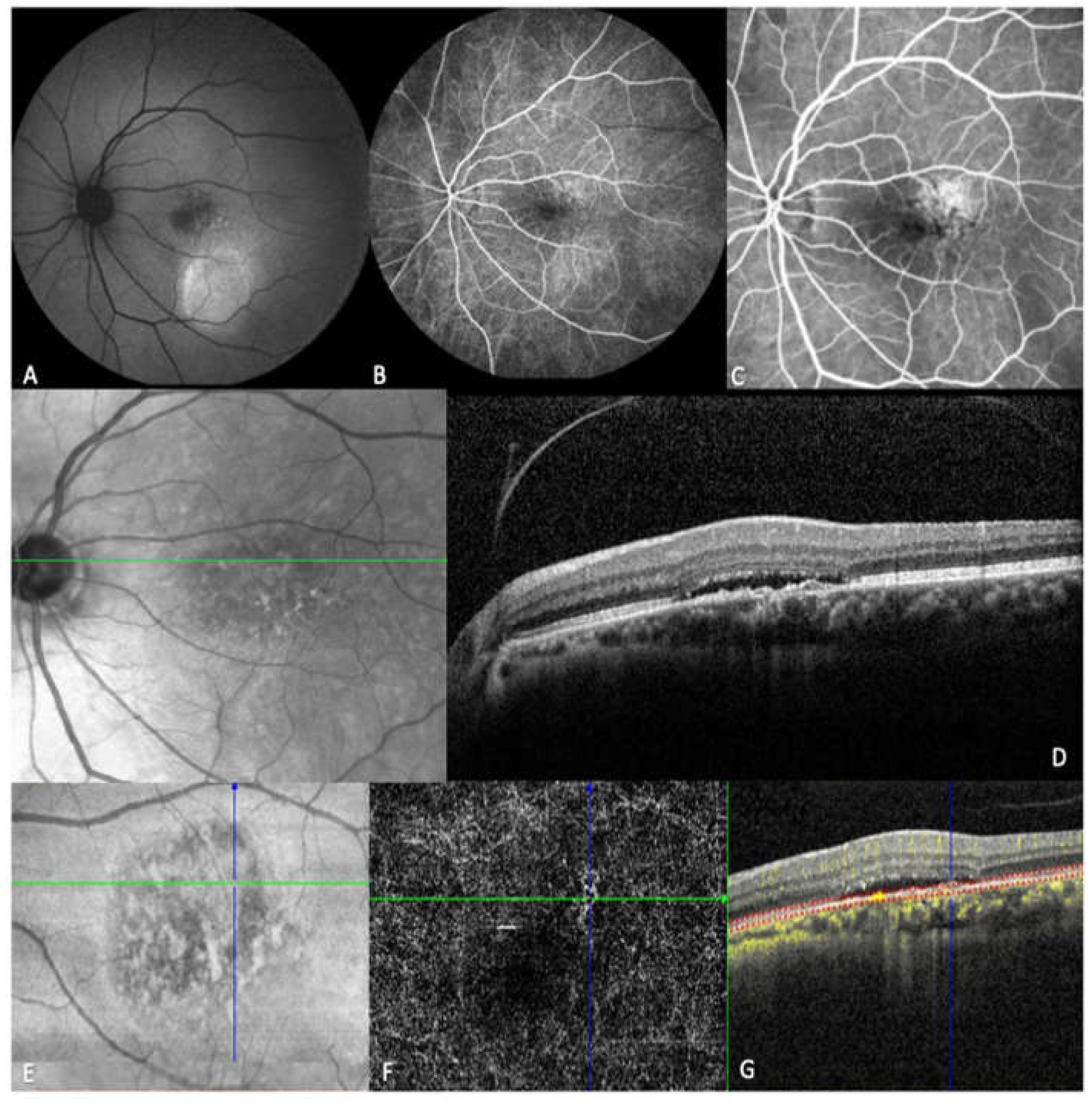
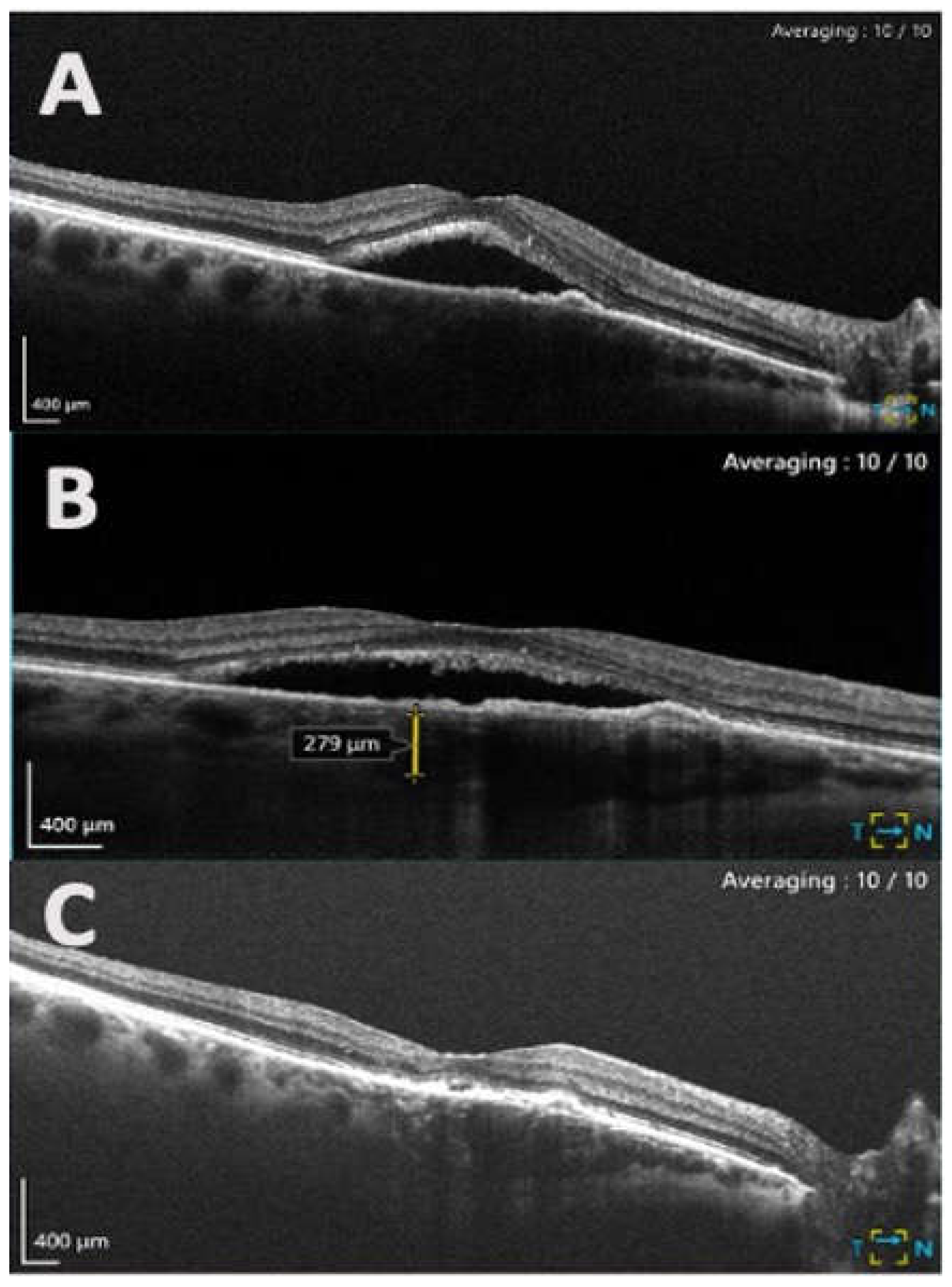
| Characteristics | |
| Total no. eyes (patients) | 22 |
| Age (years), mean ± SD | 67.61 ± 10.65 |
| Sex | |
| Female, no. (%) | 4 (18.00) |
| Male, no. (%) | 18 (82.00) |
| Eye | |
| OD, no. (%) | 14 (64.00) |
| OS, no. (%) | 8 (36.00) |
| MNV | |
| Type 1, no. (%) | 11 (50.00) |
| Mixed Type 1 + 2, no. (%) | 11 (50.00) |
| Time from diagnosis to first injection (Days), mean ± SD | 16.35 ± 13.80 |
| Total follow up period (Months), mean ± SD (IQR) | 21.0 ± 14.6 (3.5-51.0) |
| No. injections, mean ± SD (IQR) | 10 ± 8 (2.0-29.0) |
| BCVA (LogMAR), mean ± SD | 0.51 ± 0.47 |
| Anatomic Parameters | |
| SFCT, mean ± SD | 321.59 ± 84.32 |
| SRF, mean ± SD | 176.86 ± 115.62 |
| CMT, mean ± SD | 331.32 ± 130.49 |
| PED, no. (%) | 12 (54.55) |
| FIPED, no. (%) | 13 (59.09) |
| IRF, no. (%) | 11 (50.00) |
| Abbreviations: n: Total Number; SD: Standard Deviation; IQR: Interquartile Range; BCVA; Best-Corrected Visual Acuity: SFCT: Subfoveal Choroidal Thickness; CMT: Central Macular Thickness; SRF: Sub-Retinal Fluid; CT: Choroidal Thickness; FIPED: Flat Irregular PED. | |
| Baseline | Post injection | Last follow up | P Value | |
| SFCT, mean ± SD | 321.59 ± 84.32 | 317.45 ± 84.96 | 319.14 ± 99.96 | 0.850 |
| Nasal CT, mean ± SD | 318.23 ± 84.45 | 317.09 ± 83.57 | 319.68 ± 102.81 | 0.928 |
| Temporal CT, mean ± SD | 310.23 ± 80.04 | 304.50 ± 79.46 | 308.05 ± 79.07 | 0.779 |
| SRF, mean ± SD * | 176.86 ± 115.62 | 94.45 ± 83.24 | 80.95 ± 87.32 | 0.003 |
| CMT, mean ± SD ** | 331.32 ± 130.49 | 291.32 ± 106.70 | 284.77 ± 99.19 | 0.133 |
| BCVA (LogMAR), mean ± SD | 0.51 ± 0.47 | 0.56 ± 0.50 | 0.55 ± 0.54 | 0.336 |
| Abbreviations: No., Total Number; SD, Standard Deviation; SFCT, Subfoveal Choroidal Thickness; BCVA, Best-Corrected Visual Acuity; CMT, Central Macular Thickness; SRF, Subretinal Fluid; CT, Choroidal Thickness.Boldface values indicate significance with a threshold of P < 0.05 using repeated measures ANOVA (rmANOVA).* Significant reduction in SRF at presentation and post-injection (P = 0.007).** Significant reduction in CMT between presentation and post-injection (P = 0.049). | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




