Submitted:
28 April 2025
Posted:
29 April 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
Material and Methods
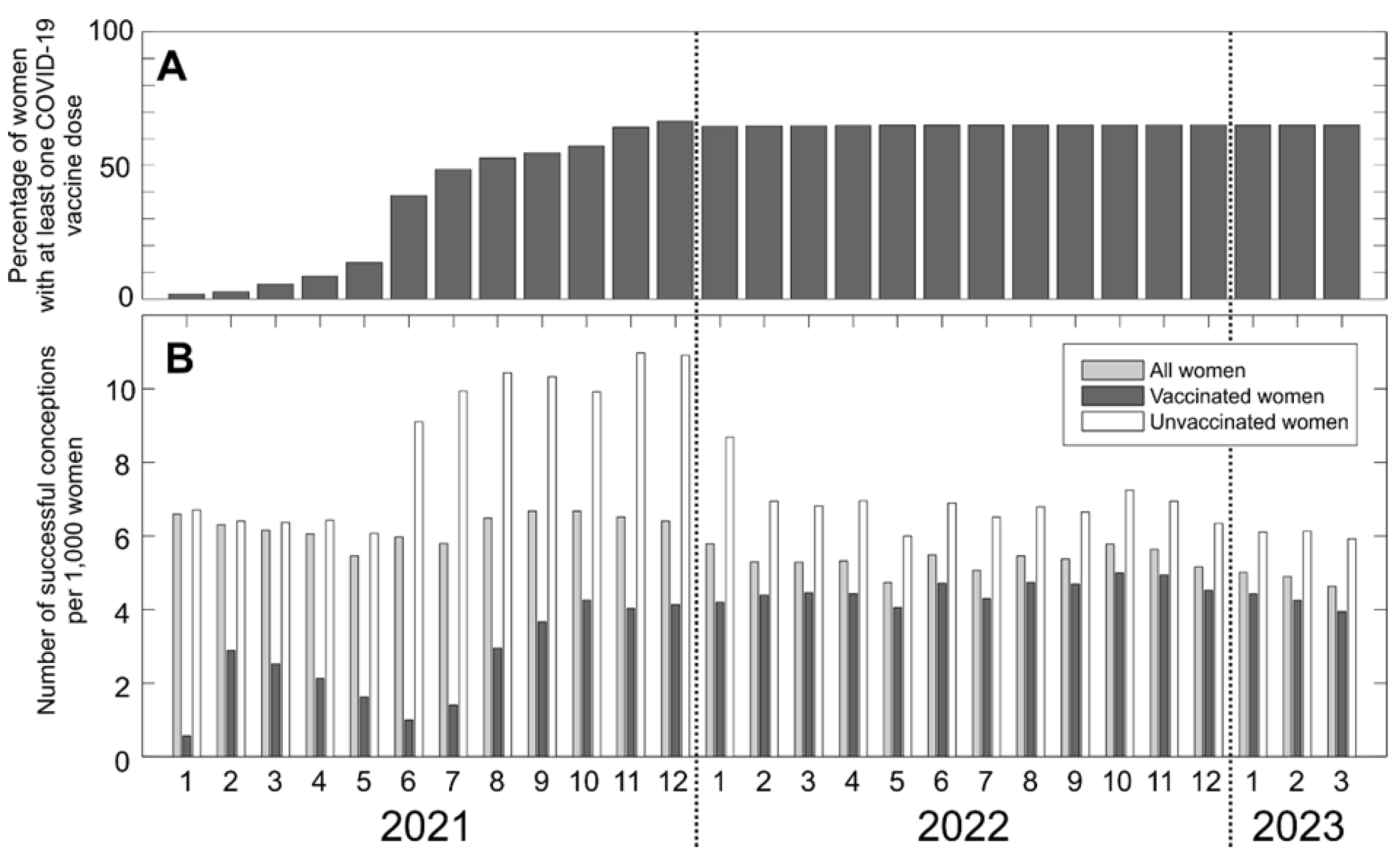
Results
Discussion
Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of interest
References
- Pomar L, Favre G, de Labrusse C, Contier A, Boulvain M, Baud D. Impact of the first wave of the COVID-19 pandemic on birth rates in Europe: a time series analysis in 24 countries. Hum Reprod 2022, 37, 2921–2931. [Google Scholar] [CrossRef] [PubMed]
- Bujard M, Andersson G. Fertility Declines Near the End of the COVID-19 Pandemic: Evidence of the 2022 Birth Declines in Germany and Sweden. Eur J Population 2024, 40, 4. [CrossRef] [PubMed]
- Winkler-Dworak M, Zeman K, Sobotka T. Birth rate decline in the later phase of the COVID-19 pandemic: the role of policy interventions, vaccination programmes, and economic uncertainty. Hum Reprod Open 2024, 2024, hoae052. [Google Scholar] [CrossRef] [PubMed]
- Wang C, Wang M, Li G, Song B, Xing Q, Cao Y. Effects of COVID-19 vaccination on human fertility: a post-pandemic literature review. Ann Med 2023, 55, 2261964. [Google Scholar] [CrossRef] [PubMed]
- Fernández-García S, Del Campo-Albendea L, Sambamoorthi D, Sheikh J, Lau K, Osei-Lah N, Ramkumar A, Naidu H, Stoney N, Sundaram P, Sengupta P, Mehta S, Attarde S, Maddock S, Manning M, Meherally Z, Ansari K, Lawson H, Yap M, Kew T, Punnoose A, Knight C, Sadeqa E, Cherian J, Ravi S, Chen W, Walker K, O'Donoghue K, van Wely M, van Leeuwen E, Kostova E, Kunst H, Khalil A, Brizuela V, Kara E, Kim CR, Thorson A, Oladapo OT, Mofenson L, Gottlieb SL, Bonet M, Moss N, Zamora J, Allotey J, Thangaratinam S; PregCOV-19 Living Systematic Review Consortium. Effectiveness and safety of COVID-19 vaccines on maternal and perinatal outcomes: a systematic review and meta-analysis. BMJ Glob Health 2024, 9, e014247. [Google Scholar] [CrossRef] [PubMed]
- Payne LA, Wise LA, Wesselink AK, Wang S, Missmer SA, Edelman A. Association between COVID-19 vaccination and menstruation: a state of the science review. BMJ Sex Reprod Health 2024, 50, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Bar-Joseph H, Raz Y, Eldar-Boock A, Michaan N, Angel Y, Saiag E, Nemerovsky L, Ben-Ami I, Shalgi R, Grisaru D. The direct effect of SARS-CoV-2 virus vaccination on human ovarian granulosa cells explains menstrual irregularities. NPJ Vaccines 2024, 9, Erratum in: NPJ Vaccines 2024, 9, 172. [Google Scholar]
- Regan AK, Fell DB, Wise LA, Vazquez-Benitez G, Håberg SE, Ogar C, Yland JJ, Wesselink AK, Zerbo O. Challenges & opportunities for the epidemiological evaluation of the effects of COVID-19 vaccination on reproduction and pregnancy. Vaccine 2023, 41, 5931–5935. [Google Scholar]
- SteelFisher GK, Blendon RJ, Caporello H. An Uncertain Public - Encouraging Acceptance of Covid-19 Vaccines. N Engl J Med 2021, 384, 1483–1487. [Google Scholar] [CrossRef] [PubMed]
- Diaz P, Reddy P, Ramasahayam R, Kuchakulla M, Ramasamy R. COVID-19 vaccine hesitancy linked to increased internet search queries for side effects on fertility potential in the initial rollout phase following Emergency Use Authorization. Andrologia 2021, 53, e14156.
- Wesselink AK, Hatch EE, Rothman KJ, Wang TR, Willis MD, Yland J, Crowe HM, Geller RJ, Willis SK, Perkins RB, Regan AK, Levinson J, Mikkelsen EM, Wise LA. A Prospective Cohort Study of COVID-19 Vaccination, SARS-CoV-2 Infection, and Fertility. Am J Epidemiol 2022, 191, 1383–1395. [Google Scholar] [CrossRef]
- Czech Statistical Office. Birth Statics. Available online: https://csu.gov.cz/births (accessed on day month year).
- Schmeling M, Manniche V, Hansen PR. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine. Eur J Clin Invest 2023, 53, e13998. [Google Scholar] [CrossRef] [PubMed]
- Fürst T, Šourek P, Krátká Z, Janošek J. Batch-dependent safety of COVID-19 vaccines in the Czech Republic and comparison with data from Denmark. Eur J Clin Invest 2024, 54, e14271. [Google Scholar] [CrossRef] [PubMed]
- Manniche V, Schmeling M, Gilthorpe JD, Hansen PR. Reports of Batch-Dependent Suspected Adverse Events of the BNT162b2 mRNA COVID-19 Vaccine: Comparison of Results from Denmark and Sweden. Medicina (Kaunas) 2024, 60, 1343. [Google Scholar] [CrossRef] [PubMed]
- Jablonowski K, Hooker B. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine in the United States. Science, Public Health Policy and the Law 2019, v5.2019-2024.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



