Submitted:
12 January 2026
Posted:
13 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
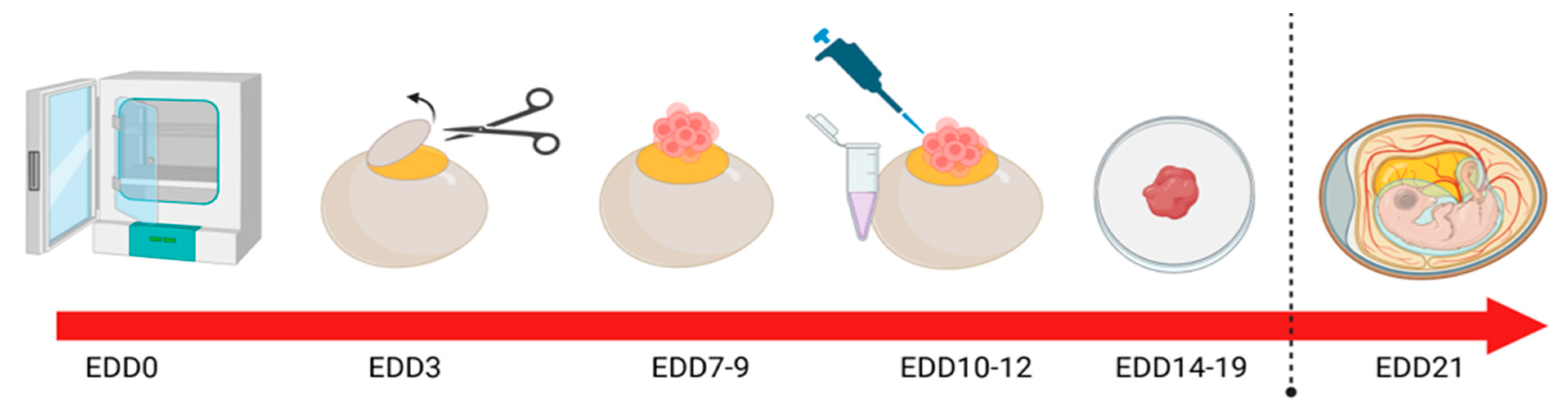
1.1. Introduction to the CAM Model
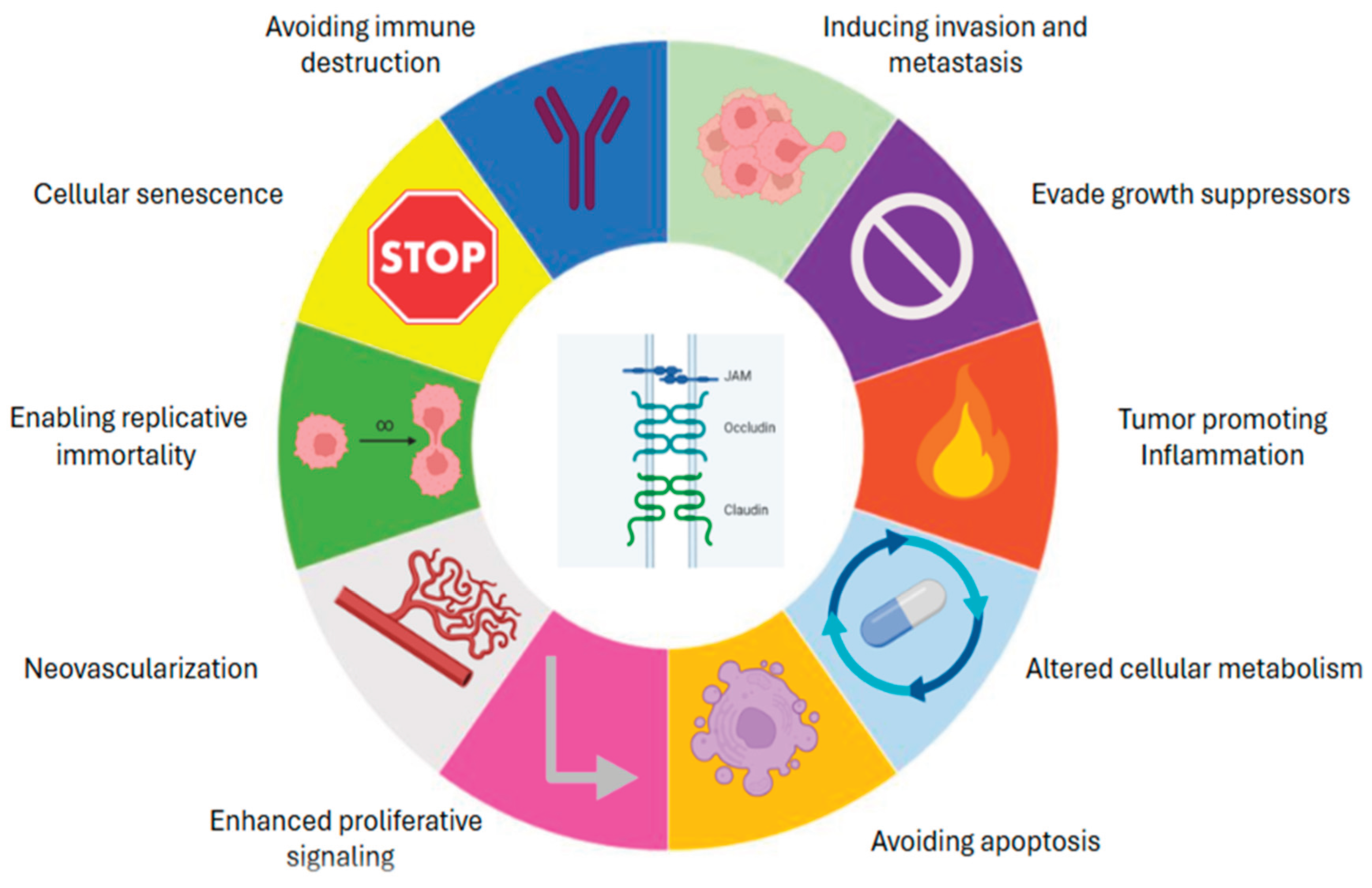
1.2. Studying the Hallmarks of Cancer in the CAM Model
1.3. Adhesion, Tight Junctions (TJs) and the CAM Model
2. Results
2.1. Integral Membrane TJ Proteins
2.2. Other Adhesion Complexes and Signaling TJ Proteins
3. Discussion
| Strengths | Limitations |
|---|---|
| Rapid tumour growth: Allows evaluation of tumour progression, angiogenesis, and invasion within 7–10 days. | Short experimental window: Limited to ~7–10 days post-engraftment, restricting long-term studies. |
| High vascularity: Enables real-time assessment of neovascularization and vascular targeting therapies. | Immature immune system: Lacks full adaptive immunity, limiting study of immunotherapy or tumour–immune interactions. |
| Cost-effective and low-maintenance: Inexpensive compared to murine models; requires minimal infrastructure. | Species differences: Avian host may not fully recapitulate mammalian tumour–stroma or ECM interactions. |
| No need for immunosuppression: Natural immune deficiency in the embryo supports xenograft engraftment. | Variable tumour take rates: Can suffer from inter-operator variability and inconsistent tumour establishment. |
| Ethically favourable: Not classified as an animal experiment in many jurisdictions (<ED14), aligning with the 3Rs. | Limited stromal complexity: Absence of fully developed mammalian stromal compartments may affect tumour behaviour. |
| Supports diverse tumour types: Compatible with cell lines, organoids, and increasingly, patient-derived xenografts (PDXs). | Lack of long-term metastatic model: While early invasion and dissemination can be studied, full metastatic colonisation is limited. |
| Accessible for imaging and manipulation: Translucent membrane allows for direct observation and microinjection. | Limited availability of chick-specific reagents: Antibodies and molecular tools for host–tumour interaction studies are less developed. |
| Amenable to genetic and pharmacological modulation: Supports siRNA, CRISPR, and small molecule screening. | Reproducibility challenges: Standardisation of protocols and quantification methods is still evolving. |
| Translational potential: Emerging role in personalised oncology through PDX and drug response studies. | Not suitable for chronic or immune-mediated diseases: Inadequate for studying long-term tumour–host dynamics. |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAM | Chorioallantoic Membrane |
| TJ | Tight Junction |
| JAM-A | Junctional Adhesion Molecule-A |
| PDX | Patient-Derived Xenograft |
| ECM | Extracellular Matrix |
| TME | Tumour Microenvironment |
| YAP | Yes-associated Protein |
| TAZ | Transcriptional Co-Activator with PDZ-binding Motif |
| RNAi | RNA Interference |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
References
- Kim, S.-Y.; van de Wetering, M.; Clevers, H.; Sanders, K. The future of tumor organoids in precision therapy. Trends in Cancer 2025. [Google Scholar] [CrossRef] [PubMed]
- Rous, P. A SARCOMA OF THE FOWL TRANSMISSIBLE BY AN AGENT SEPARABLE FROM THE TUMOR CELLS. Journal of Experimental Medicine 1911, 13, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Kunz, P.; Schenker, A.; Sahr, H.; Lehner, B.; Fellenberg, J. Optimization of the chicken chorioallantoic membrane assay as reliable in vivo model for the analysis of osteosarcoma. PLoS One 2019, 14, e0215312. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, S.; Sawosz, E.; Grodzik, M.; Kutwin, M.; Wierzbicki, M.; Włodyga, K.; Jasik, A.; Reichert, M.; Chwalibog, A. Comparison of tumour morphology and structure from U87 and U118 glioma cells cultured on chicken embryo chorioallantoic membrane. Bulletin of the Veterinary Institute in Pulawy 2013, 57, 593–598. [Google Scholar] [CrossRef]
- Ribatti, D.; Nico, B.; Vacca, A.; Roncali, L.; Burri, P.H.; Djonov, V. Chorioallantoic membrane capillary bed: a useful target for studying angiogenesis and anti-angiogenesis in vivo. Anat Rec 2001, 264, 317–324. [Google Scholar] [CrossRef]
- Steiner1, Normann; Ribatti4, Domenico; Willenbacher2, Wolfgang; Jöhrer3, Karin; Kern1, Johann; Marinaccio4, Christian; Aracil6, Miguel; García-Fernández6, Luis F.; Guenther Gastl2, G.U.; Gunsilius, Eberhard. Marine compounds inhibit growth of multiple myeloma in vitroand in vivo. Oncotarget 2015, Vol. 6. [Google Scholar] [CrossRef]
- Ranjan, R.A.; Muenzner, J.K.; Kunze, P.; Geppert, C.I.; Ruebner, M.; Huebner, H.; Fasching, P.A.; Beckmann, M.W.; Bauerle, T.; Hartmann, A.; et al. The Chorioallantoic Membrane Xenograft Assay as a Reliable Model for Investigating the Biology of Breast Cancer. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Andries Zijlstra, R.M.; Panzarella, Giano; Aimes, Ronald T.; Hooper, John D.; Marchenko, Natalia D.; Quigley, J.P. A Quantitative Analysis of Rate-limiting Steps in the Metastatic Cascade Using Human-specific Real-Time Polymerase Chain Reaction. Cancer Res 2002, 62. [Google Scholar]
- Fischer, D.; Fluegen, G.; Garcia, P.; Ghaffari-Tabrizi-Wizsy, N.; Gribaldo, L.; Huang, R.Y.; Rasche, V.; Ribatti, D.; Rousset, X.; Pinto, M.T.; et al. The CAM Model-Q&A with Experts. Cancers (Basel) 2022, 15. [Google Scholar] [CrossRef]
- Aslam, R.; Richards, C.E.; Fay, J.; Hudson, L.; Workman, J.; Lee, C.L.; Murphy, A.; O'Neill, B.; Toomey, S.; Hennessy, B.T. Synergistic Effects of the Combination of Alpelisib (PI3K Inhibitor) and Ribociclib (CDK4/6 Inhibitor) in Preclinical Colorectal Cancer Models. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Nagai, H.; Tanoue, Y.; Nakamura, T.; Chan, C.J.J.; Yamada, S.; Saitou, M.; Fukuda, T.; Sheng, G. Mesothelial fusion mediates chorioallantoic membrane formation. Philos Trans R Soc Lond B Biol Sci 2022, 377, 20210263. [Google Scholar] [CrossRef]
- Nowak-Sliwinska, P.; Segura, T.; Iruela-Arispe, M.L. The chicken chorioallantoic membrane model in biology, medicine and bioengineering. Angiogenesis 2014, 17, 779–804. [Google Scholar] [CrossRef]
- Ribatti, D. The chick embryo chorioallantoic membrane (CAM) assay. Reprod Toxicol 2017, 70, 97–101. [Google Scholar] [CrossRef]
- Chen, L.; Wang, S.; Feng, Y.; Zhang, J.; Du, Y.; Zhang, J.; Ongeval, C.V.; Ni, Y.; Li, Y. Utilisation of Chick Embryo Chorioallantoic Membrane as a Model Platform for Imaging-Navigated Biomedical Research. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Wang, Y.; Viallet, J.; Macek Jilkova, Z. The Chicken Embryo Model: A Novel and Relevant Model for Immune-Based Studies. Front Immunol 2021, 12, 791081. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D. The chick embryo chorioallantoic membrane (CAM). A multifaceted experimental model. Mech Dev 2016, 141, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Vacca, A.; Ribatti, D.; Presta, M.; Minischetti, M.; Iurlaro, M.; Ria, R.; Albini, A.; Bussolino, F.; Dammacco, F. Bone Marrow Neovascularization, Plasma Cell Angiogenic Potential, and Matrix Metalloproteinase-2 Secretion Parallel Progression of Human Multiple Myeloma. Blood 1999, 93, 3064–3073. [Google Scholar] [CrossRef]
- Pizon, M.; Schott, D.; Pachmann, U.; Schobert, R.; Pizon, M.; Wozniak, M.; Bobinski, R.; Pachmann, K. Chick Chorioallantoic Membrane (CAM) Assays as a Model of Patient-Derived Xenografts from Circulating Cancer Stem Cells (cCSCs) in Breast Cancer Patients. Cancers (Basel) 2022, 14. [Google Scholar] [CrossRef]
- Bencurova, K.; Tran, L.; Friske, J.; Bevc, K.; Helbich, T.H.; Hacker, M.; Bergmann, M.; Zeitlinger, M.; Haug, A.; Mitterhauser, M.; et al. An in vivo tumour organoid model based on the chick embryonic chorioallantoic membrane mimics key characteristics of the patient tissue: a proof-of-concept study. EJNMMI Res 2024, 14, 86. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Cai, L. Establishment of a transplantation tumor model of human osteosarcoma in chick embryo. The Chinese-German Journal of Clinical Oncology 2009, 8, 531–536. [Google Scholar] [CrossRef]
- Hu, J.; Ishihara, M.; Chin, A.I.; Wu, L. Establishment of xenografts of urological cancers on chicken chorioallantoic membrane (CAM) to study metastasis. Precis Clin Med 2019, 2, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D. Chicken Chorioallantoic Membrane Angiogenesis Model. In Methods in molecular biology; Clifton, N.J.), 2012; Volume 843, pp. 47–57. [Google Scholar] [CrossRef]
- Deryugina, E.I.; Zijlstra, A.; Partridge, J.J.; Kupriyanova, T.A.; Madsen, M.A.; Papagiannakopoulos, T.; Quigley, J.P. Unexpected effect of matrix metalloproteinase down-regulation on vascular intravasation and metastasis of human fibrosarcoma cells selected in vivo for high rates of dissemination. Cancer Res 2005, 65, 10959–10969. [Google Scholar] [CrossRef] [PubMed]
- Deryugina, E.I.; Quigley, J.P. Chick embryo chorioallantoic membrane model systems to study and visualize human tumor cell metastasis. Histochem Cell Biol 2008, 130, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Skowron, M.A.; Sathe, A.; Romano, A.; Hoffmann, M.J.; Schulz, W.A.; van Koeveringe, G.A.; Albers, P.; Nawroth, R.; Niegisch, G. Applying the chicken embryo chorioallantoic membrane assay to study treatment approaches in urothelial carcinoma. Urol Oncol 2017, 35, 544 e511–544 e523. [Google Scholar] [CrossRef]
- Shekatkar, M.; Kheur, S.; Deshpande, S.; Sakhare, S.; Sanap, A.; Kheur, M.; Bhonde, R. Critical appraisal of the chorioallantoic membrane model for studying angiogenesis in preclinical research. Mol Biol Rep 2024, 51, 1026. [Google Scholar] [CrossRef]
- Kakogiannos, N.; Ferrari, L.; Giampietro, C.; Scalise, A.A.; Maderna, C.; Rava, M.; Taddei, A.; Lampugnani, M.G.; Pisati, F.; Malinverno, M.; et al. JAM-A Acts via C/EBP-alpha to Promote Claudin-5 Expression and Enhance Endothelial Barrier Function. Circ Res 2020, 127, 1056–1073. [Google Scholar] [CrossRef]
- Zammarchi, I.; Santacroce, G.; Puga-Tejada, M.; Hayes, B.; Crotty, R.; O'Driscoll, E.; Majumder, S.; Kaczmarczyk, W.; Maeda, Y.; McCarthy, J.; et al. Epithelial neutrophil localization and tight junction Claudin-2 expression are innovative outcome predictors in inflammatory bowel disease. United European Gastroenterol J 2024, 12, 1155–1166. [Google Scholar] [CrossRef]
- Leech, A.O.; Cruz, R.G.; Hill, A.D.; Hopkins, A.M. Paradigms lost-an emerging role for over-expression of tight junction adhesion proteins in cancer pathogenesis. Ann Transl Med 2015, 3, 184. [Google Scholar] [CrossRef]
- Dionísio, M.R.; Vieira, A.F.; Carvalho, R.; Conde, I.; Oliveira, M.; Gomes, M.; Pinto, M.T.; Pereira, P.; Pimentel, J.; Souza, C.; et al. BR-BCSC Signature: The Cancer Stem Cell Profile Enriched in Brain Metastases that Predicts a Worse Prognosis in Lymph Node-Positive Breast Cancer. Cells 2020, 9, 2442. [Google Scholar] [CrossRef]
- Anderson, J.M.; Van Itallie, C.M. Physiology and function of the tight junction. Cold Spring Harb Perspect Biol 2009, 1, a002584. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov 2022, 12, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Wan, Z.; Hirche, C.; Fricke, F.; Dragu, A.; Will, P.A. Chick Chorioallantoic Membrane as an in vivo Model for the Study of Angiogenesis and Lymphangiogenesis. J Vasc Res 2025, 62, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D. The chick embryo chorioallantoic membrane as an experimental model to study in vivo angiogenesis in glioblastoma multiforme. Brain Res Bull 2022, 182, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Lokman, N.A.; Elder, A.S.F.; Ricciardelli, C.; Oehler, M.K. Chick chorioallantoic membrane (CAM) assay as an in vivo model to study the effect of newly identified molecules on ovarian cancer invasion and metastasis. Int J Mol Sci 2012, 13, 9959–9970. [Google Scholar] [CrossRef]
- Smith, Y.E.; Wang, G.; Flynn, C.L.; Madden, S.F.; MacEneaney, O.; Cruz, R.G.B.; Richards, C.E.; Jahns, H.; Brennan, M.; Cremona, M.; et al. Functional Antagonism of Junctional Adhesion Molecule-A (JAM-A), Overexpressed in Breast Ductal Carcinoma In Situ (DCIS), Reduces HER2-Positive Tumor Progression. Cancers (Basel) 2022, 14. [Google Scholar] [CrossRef]
- Naik, M.U.; Naik, T.U.; Suckow, A.T.; Duncan, M.K.; Naik, U.P. Attenuation of junctional adhesion molecule-A is a contributing factor for breast cancer cell invasion. Cancer Res 2008, 68, 2194–2203. [Google Scholar] [CrossRef]
- Brennan, K.; McSherry, E.A.; Hudson, L.; Kay, E.W.; Hill, A.D.K.; Young, L.S.; Hopkins, A.M. Junctional adhesion molecule-A is co-expressed with HER2 in breast tumors and acts as a novel regulator of HER2 protein degradation and signaling. Oncogene 2013, 32, 2799–2804. [Google Scholar] [CrossRef]
- Murakami, T.; Takasawa, A.; Takasawa, K.; Akimoto, T.; Aoyama, T.; Magara, K.; Saito, Y.; Ota, M.; Kyuno, D.; Yamamoto, S.; et al. Aberrant expression of junctional adhesion molecule-A contributes to the malignancy of cervical adenocarcinoma by interaction with poliovirus receptor/CD155. Cancer Sci 2021, 112, 906–917. [Google Scholar] [CrossRef]
- McSherry, E.A.; Brennan, K.; Hudson, L.; Hill, A.D.K.; Hopkins, A.M. Breast cancer cell migration is regulated through junctional adhesion molecule-A-mediated activation of Rap1 GTPase. Breast Cancer Research 2011, 13, R31. [Google Scholar] [CrossRef]
- Lathia, Justin D.; Li, M.; Sinyuk, M.; Alvarado, Alvaro G.; Flavahan, William A.; Stoltz, K.; Rosager, Ann M.; Hale, J.; Hitomi, M.; Gallagher, J.; et al. High-Throughput Flow Cytometry Screening Reveals a Role for Junctional Adhesion Molecule A as a Cancer Stem Cell Maintenance Factor. Cell Reports 2014, 6, 117–129. [Google Scholar] [CrossRef]
- Leech, A.O.; Vellanki, S.H.; Rutherford, E.J.; Keogh, A.; Jahns, H.; Hudson, L.; O'Donovan, N.; Sabri, S.; Abdulkarim, B.; Sheehan, K.M.; et al. Cleavage of the extracellular domain of junctional adhesion molecule-A is associated with resistance to anti-HER2 therapies in breast cancer settings. Breast Cancer Res 2018, 20, 140. [Google Scholar] [CrossRef]
- Vellanki, S.H.; Cruz, R.G.B.; Jahns, H.; Hudson, L.; Sette, G.; Eramo, A.; Hopkins, A.M. Natural compound Tetrocarcin-A downregulates Junctional Adhesion Molecule-A in conjunction with HER2 and inhibitor of apoptosis proteins and inhibits tumor cell growth. Cancer Letters 2019, 440-441, 23–34. [Google Scholar] [CrossRef] [PubMed]
- VELLANKI, S.H.; CRUZ, R.G.B.; RICHARDS, C.E.; SMITH, Y.E.; HUDSON, L.; JAHNS, H.; HOPKINS, A.M. Antibiotic Tetrocarcin-A Down-regulates JAM-A, IAPs and Induces Apoptosis in Triple-negative Breast Cancer Models. Anticancer Research 2019, 39, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.E.; Sheehan, K.M.; Kay, E.W.; Hedner, C.; Borg, D.; Fay, J.; O'Grady, A.; Hill, A.D.K.; Jirstrom, K.; Hopkins, A.M. Development of a Novel Weighted Ranking Method for Immunohistochemical Quantification of a Heterogeneously Expressed Protein in Gastro-Esophageal Cancers. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Kummer, D.; Ebnet, K. Junctional Adhesion Molecules (JAMs): The JAM-Integrin Connection. Cells 2018, 7, 25. [Google Scholar] [CrossRef]
- Ebnet, K. Junctional Adhesion Molecules (JAMs): Cell Adhesion Receptors With Pleiotropic Functions in Cell Physiology and Development. Physiol Rev 2017, 97, 1529–1554. [Google Scholar] [CrossRef]
- Shu, F.; Lu, J.; Zhang, W.; Huang, H.; Lin, J.; Jiang, L.; Liu, W.; Liu, T.; Xiao, S.; Zheng, Y.; et al. JAM-A Overexpression in Human Umbilical Cord-Derived Mesenchymal Stem Cells Accelerated the Angiogenesis of Diabetic Wound By Enhancing Both Paracrine Function and Survival of Mesenchymal Stem Cells. Stem Cell Rev Rep 2023, 19, 1554–1575. [Google Scholar] [CrossRef]
- Solimando, A.G.; Da Via, M.C.; Leone, P.; Borrelli, P.; Croci, G.A.; Tabares, P.; Brandl, A.; Di Lernia, G.; Bianchi, F.P.; Tafuri, S.; et al. Halting the vicious cycle within the multiple myeloma ecosystem: blocking JAM-A on bone marrow endothelial cells restores angiogenic homeostasis and suppresses tumor progression. Haematologica 2021, 106, 1943–1956. [Google Scholar] [CrossRef]
- Solimando, A.G.; Brandl, A.; Mattenheimer, K.; Graf, C.; Ritz, M.; Ruckdeschel, A.; Stühmer, T.; Mokhtari, Z.; Rudelius, M.; Dotterweich, J.; et al. JAM-A as a prognostic factor and new therapeutic target in multiple myeloma. Leukemia 2018, 32, 736–743. [Google Scholar] [CrossRef]
- Solimando, A.G.; Da Via', M.C.; Borrelli, P.; Leone, P.; Di Lernia, G.; Tabares Gaviria, P.; Brandl, A.; Pedone, G.L.; Rauert-Wunderlich, H.; Lapa, C.; et al. Central Function for JAM-a in Multiple Myeloma Patients with Extramedullary Disease. Blood 2018, 132, 4455–4455. [Google Scholar] [CrossRef]
- Ren, T.; Zheng, Y.; Liu, F.; Liu, C.; Zhang, B.; Ren, H.; Gao, X.; Wei, Y.; Sun, Q.; Huang, H. Identification and Validation of JAM-A as a Novel Prognostic and Immune Factor in Human Tumors. Biomedicines 2024, 12, 1423. [Google Scholar] [CrossRef]
- Miebach, L.; Berner, J.; Bekeschus, S. In ovo model in cancer research and tumor immunology. Front Immunol 2022, 13, 1006064. [Google Scholar] [CrossRef] [PubMed]
- Nipper, A.J.; Warren, E.A.K.; Liao, K.S.; Liu, H.-C.; Michikawa, C.; Porter, C.E.; Wells, G.A.; Villanueva, M.; Brasil da Costa, F.H.; Veeramachaneni, R.; et al. Chick Embryo Chorioallantoic Membrane as a Platform for Assessing the In Vivo Efficacy of Chimeric Antigen Receptor T-cell Therapy in Solid Tumors. ImmunoHorizons 2024, 8, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Hartle, S.; Sutton, K.; Vervelde, L.; Dalgaard, T.S. Delineation of chicken immune markers in the era of omics and multicolor flow cytometry. Front Vet Sci 2024, 11, 1385400. [Google Scholar] [CrossRef] [PubMed]
- Vellanki, s.h.; Richards, C.; Smith, Y.; Hopkins, A. The Contribution of Ig-Superfamily and MARVEL D Tight Junction Proteins to Cancer Pathobiology. Current Pathobiology Reports 2016, 4. [Google Scholar] [CrossRef]
- Gerardo-Ramirez, M.; Lazzarini-Lechuga, R.; Hernandez-Rizo, S.; Jimenez-Salazar, J.E.; Simoni-Nieves, A.; Garcia-Ruiz, C.; Fernandez-Checa, J.C.; Marquardt, J.U.; Coulouarn, C.; Gutierrez-Ruiz, M.C.; et al. GDF11 exhibits tumor suppressive properties in hepatocellular carcinoma cells by restricting clonal expansion and invasion. Biochim Biophys Acta Mol Basis Dis 2019, 1865, 1540–1554. [Google Scholar] [CrossRef]
- Zhang, P.; Zhong, S.; Wang, G.; Zhang, S.Y.; Chu, C.; Zeng, S.; Yan, Y.; Cheng, X.; Bao, Y.; Hocher, B.; et al. N-Acetylcysteine Suppresses LPS-Induced Pathological Angiogenesis. Cell Physiol Biochem 2018, 49, 2483–2495. [Google Scholar] [CrossRef]
- Panciera, T.; Citron, A.; Di Biagio, D.; Battilana, G.; Gandin, A.; Giulitti, S.; Forcato, M.; Bicciato, S.; Panzetta, V.; Fusco, S.; et al. Reprogramming normal cells into tumour precursors requires ECM stiffness and oncogene-mediated changes of cell mechanical properties. Nat Mater 2020, 19, 797–806. [Google Scholar] [CrossRef]
- Glienke, J.; Schmitt, A.O.; Pilarsky, C.; Hinzmann, B.; Weiss, B.; Rosenthal, A.; Thierauch, K.H. Differential gene expression by endothelial cells in distinct angiogenic states. Eur J Biochem 2000, 267, 2820–2830. [Google Scholar] [CrossRef]
- Wang, X.; Freire Valls, A.; Schermann, G.; Shen, Y.; Moya, I.M.; Castro, L.; Urban, S.; Solecki, G.M.; Winkler, F.; Riedemann, L.; et al. YAP/TAZ Orchestrate VEGF Signaling during Developmental Angiogenesis. Dev Cell 2017, 42, 462–478 e467. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Liu, P.; Xu, H.; Liang, D.; Fang, K.; Du, S.; Cheng, W.; Ye, L.; Liu, T.; Zhang, X.; et al. SASH1 suppresses triple-negative breast cancer cell invasion through YAP-ARHGAP42-actin axis. Oncogene 2020, 39, 5015–5030. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jia, Y.; Pan, P.; Zhang, X.; Jia, Y.; Zhu, P.; Chen, X.; Jiao, Y.; Kang, G.; Zhang, L.; et al. α5-nAChR associated with Ly6E modulates cell migration via TGF-β1/Smad signaling in non-small cell lung cancer. Carcinogenesis 2022, 43, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Strusi, G.; Suelzu, C.M.; Weldon, S.; Giffin, J.; Munsterberg, A.E.; Bao, Y. Combination of Phenethyl Isothiocyanate and Dasatinib Inhibits Hepatocellular Carcinoma Metastatic Potential through FAK/STAT3/Cadherin Signalling and Reduction of VEGF Secretion. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Han, J.M.; Jung, H.J. Synergistic Anticancer Effect of a Combination of Berbamine and Arcyriaflavin A against Glioblastoma Stem-like Cells. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Huang, Y.Q.; Yuan, J.D.; Ding, H.F.; Song, Y.S.; Qian, G.; Wang, J.L.; Ji, M.; Zhang, Y. Design, synthesis and pharmacological evaluation of a novel PEG-cRGD-conjugated irinotecan derivative as potential antitumor agent. Eur J Med Chem 2018, 158, 82–90. [Google Scholar] [CrossRef]
- Richards, C. Exploring New and Emerging Mechanisms to Target Difficult to Treat Cancers; Royal College of Surgeons in Ireland, 2020. [Google Scholar]
- Sharma, S.; Alizadeh, M.; Pratt, S.; Stamatikos, A.; Abdelaziz, K. Differential Expression of Key Immune Markers in the Intestinal Tract of Developing Chick Embryos. Vet Sci 2025, 12. [Google Scholar] [CrossRef]
- Martowicz, A.; Kern, J.; Gunsilius, E.; Untergasser, G. Establishment of a Human Multiple Myeloma Xenograft Model in the Chicken to Study Tumor Growth, Invasion and Angiogenesis 2015, 1940-087X 99, e52665.
- Schulze, J.; Librizzi, D.; Bender, L.; Jedelska, J.; Yousefi, B.H.; Schaefer, J.; Preis, E.; Luster, M.; Mahnken, A.H.; Bakowsky, U. How to Xenograft Cancer Cells on the Chorioallantoic Membrane of a Fertilized Hen's Egg and Its Visualization by PET/CT and MRI. ACS Appl Bio Mater 2023, 6, 2435–2445. [Google Scholar] [CrossRef]
- Herrmann, A.; Taylor, A.; Murray, P.; Poptani, H.; See, V. Magnetic Resonance Imaging for Characterization of a Chick Embryo Model of Cancer Cell Metastases. Mol Imaging 2018, 17, 1536012118809585. [Google Scholar] [CrossRef]
- Sommerfeld, S.; Mundim, A.V.; Silva, R.R.; Queiroz, J.S.; Rios, M.P.; Notário, F.O.; Medeiros Ronchi, A.A.; Beletti, M.E.; Franco, R.R.; Espindola, F.S.; et al. Physiological Changes in Chicken Embryos Inoculated with Drugs and Viruses Highlight the Need for More Standardization of this Animal Model. Animals 2022, 12, 1156. [Google Scholar] [CrossRef]
- Ahmed, T.A.E.; Cordeiro, C.M.M.; Elebute, O.; Hincke, M.T. Proteomic Analysis of Chicken Chorioallantoic Membrane (CAM) during Embryonic Development Provides Functional Insight. Biomed Res Int 2022, 2022, 7813921. [Google Scholar] [CrossRef]
- McGrew, M.J.; Holmes, T.; Davey, M.G. A scientific case for revisiting the embryonic chicken model in biomedical research. Developmental Biology 2025, 522, 220–226. [Google Scholar] [CrossRef]
- Handl, V.; Waldherr, L.; Arbring Sjöström, T.; Abrahamsson, T.; Seitanidou, M.; Erschen, S.; Gorischek, A.; Bernacka-Wojcik, I.; Saarela, H.; Tomin, T.; et al. Continuous iontronic chemotherapy reduces brain tumor growth in embryonic avian in vivo models. Journal of Controlled Release 2024, 369, 668–683. [Google Scholar] [CrossRef]
- Faihs, L.; Firouz, B.; Slezak, P.; Slezak, C.; Weissensteiner, M.; Ebner, T.; Ghaffari Tabrizi-Wizsy, N.; Schicho, K.; Dungel, P. A Novel Artificial Intelligence-Based Approach for Quantitative Assessment of Angiogenesis in the Ex Ovo CAM Model. Cancers (Basel) 2022, 14. [Google Scholar] [CrossRef]
| Hallmark being evaluated | Protocol | Reference |
|---|---|---|
| Evaluating cell state and morphology | Immunohistochemistry | [44] |
| Western blotting | [25] | |
| Haematoxylin and eosin | [10,26] | |
| Assessing neovascularization | Gross imaging of vasculature | [49],[22] |
| Evans blue dye | [58] | |
| Yolk sac membrane | [65] | |
| Proliferation and inflammation | Weighing tumour mass | [25] |
| Ki67 staining | [68] | |
| ELISA | [6,69,70] | |
| Fluorescent-tagged cells | [24] | |
| Cytokeratin staining | [23], [42], [57] | |
| MRI for secondary tumour sites | [71,72] | |
| Alu sequence PCRs | [8] | |
| Altered metabolism | Analysis of proteome | [73,74] |
| Immune response of the embryo | Flow cytometry | [9] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




