Discussion
Interpretation of Elemental Anomalies
The elemental deviations identified in this study place strong constraints on the biochemical nature of anomalous intravascular casts (AICs) without, by themselves, specifying molecular identity. In particular, the combination of sulfur depletion and relative phosphorus enrichment provides insight into bulk matrix composition that is independent of histological appearance and resistant to analytical artefact.
Implications of Sulfur Depletion for Protein Composition
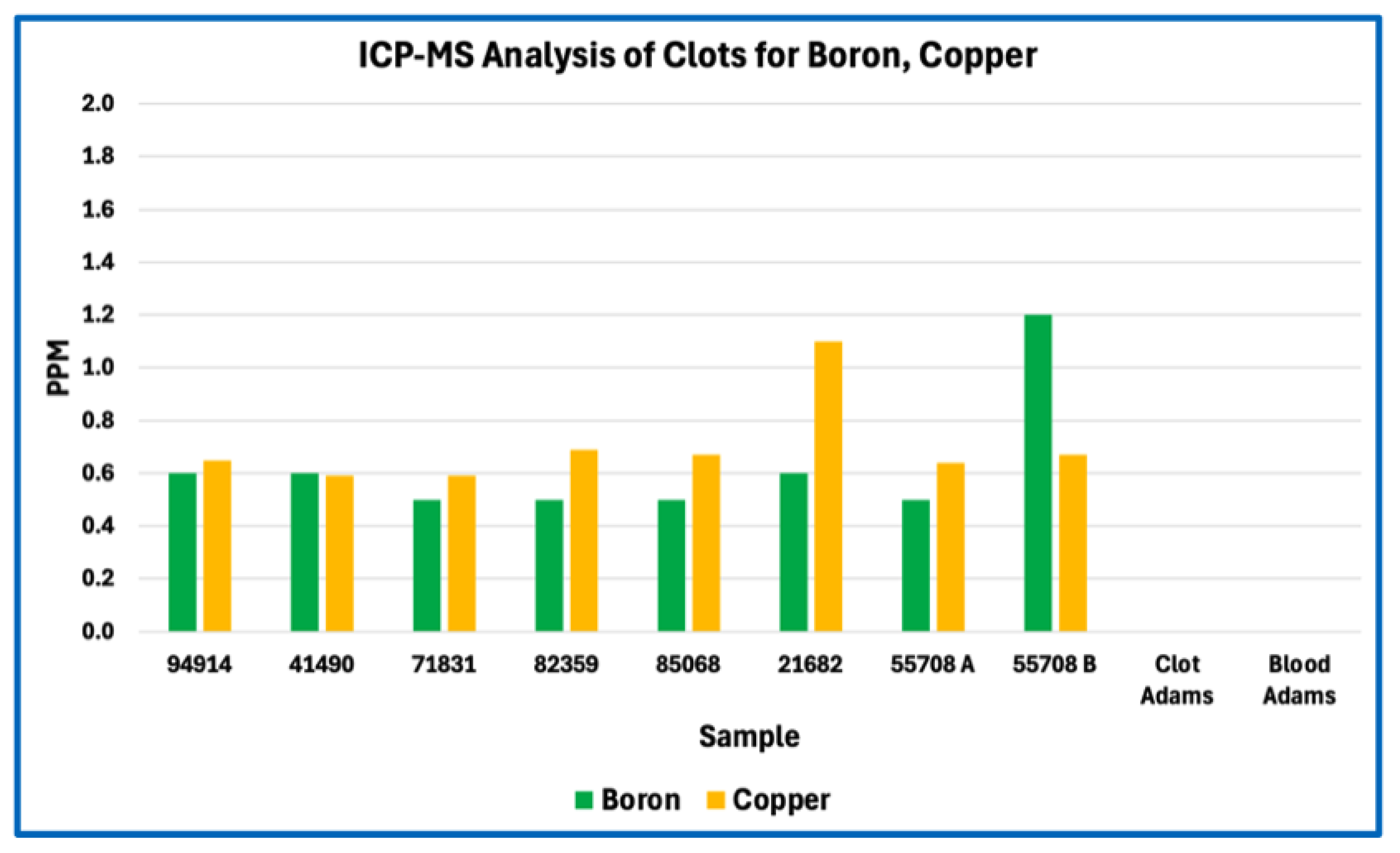
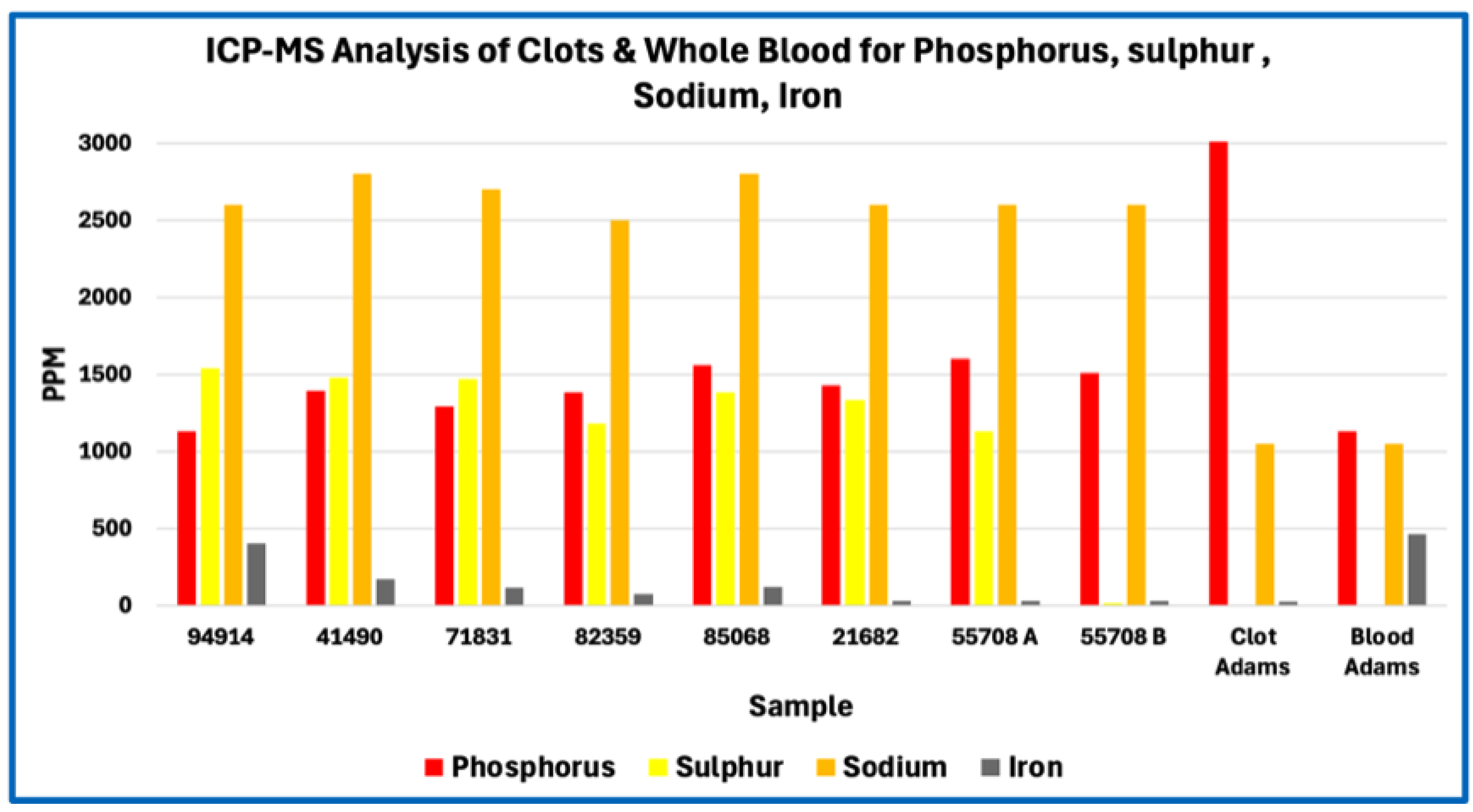
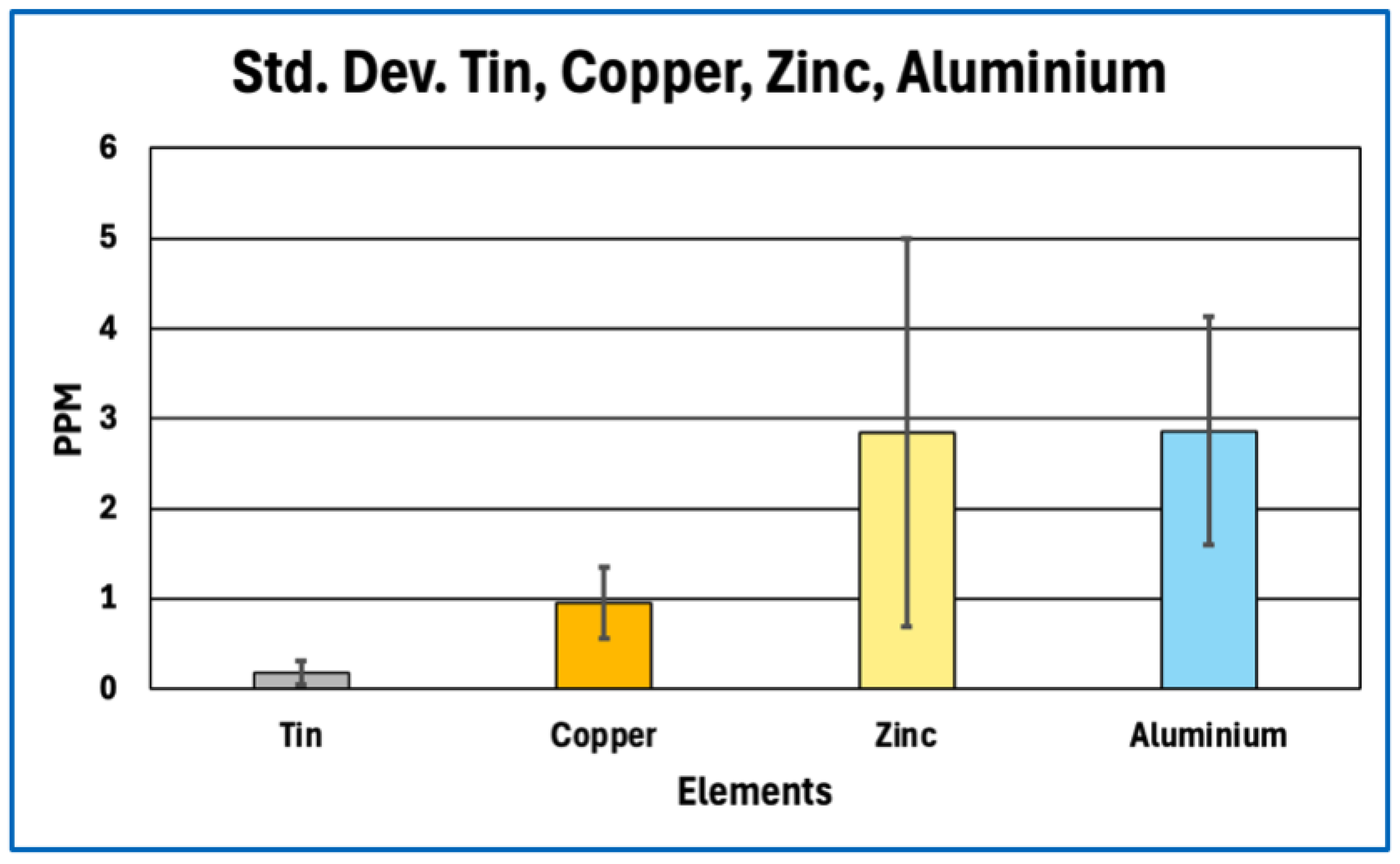
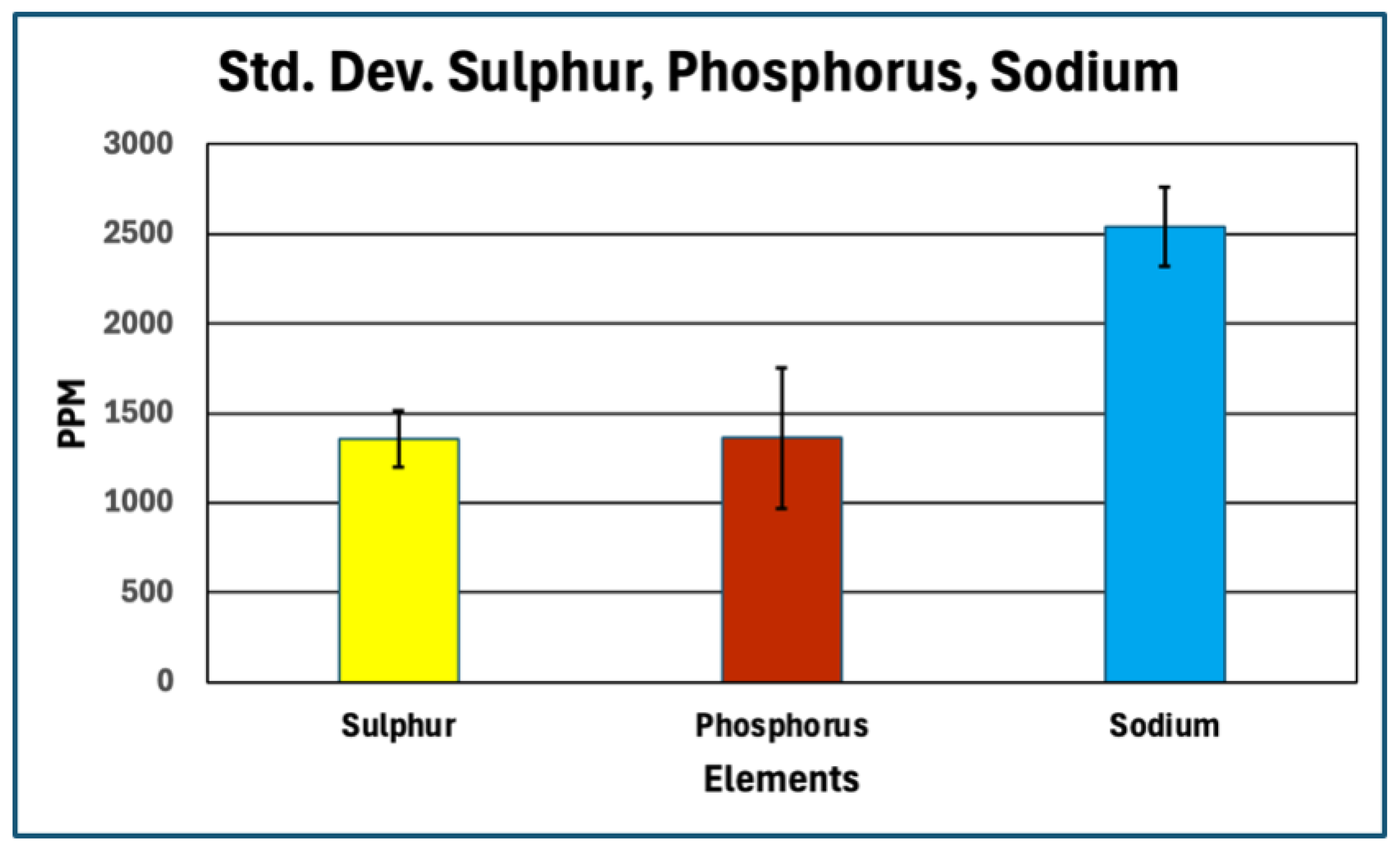
Sulfur content offers a robust proxy for total protein fraction because sulfur-bearing amino acids—cysteine and methionine—are intrinsic to polypeptide backbones and occur at relatively fixed proportions in fibrinogen and related clotting proteins. First-principles stoichiometric calculations therefore define a narrow expected range for sulfur abundance in fibrin-dominant matrices. The marked sulfur depletion observed in AICs relative to this expectation implies that proteinaceous material constitutes only a minor fraction of total dry mass. Even allowing for surface effects, partial analytical under-recovery, or postmortem modification, the magnitude and consistency of sulfur depletion across samples exceed what can reasonably be attributed to methodological bias alone.
Importantly, this finding does not indicate absence of protein, nor does it contradict the fibrinous architecture observed histologically. Rather, it constrains the bulk composition of the casts, indicating that while proteins are present and contribute to structural features, they do not dominate the mass of the matrix. This distinction resolves the apparent tension between fibrin-like morphology and non-fibrin-like elemental composition and cautions against equating histological fibrin appearance with a predominantly proteinaceous material.
Interpretation of Phosphorus Enrichment Without Molecular Attribution
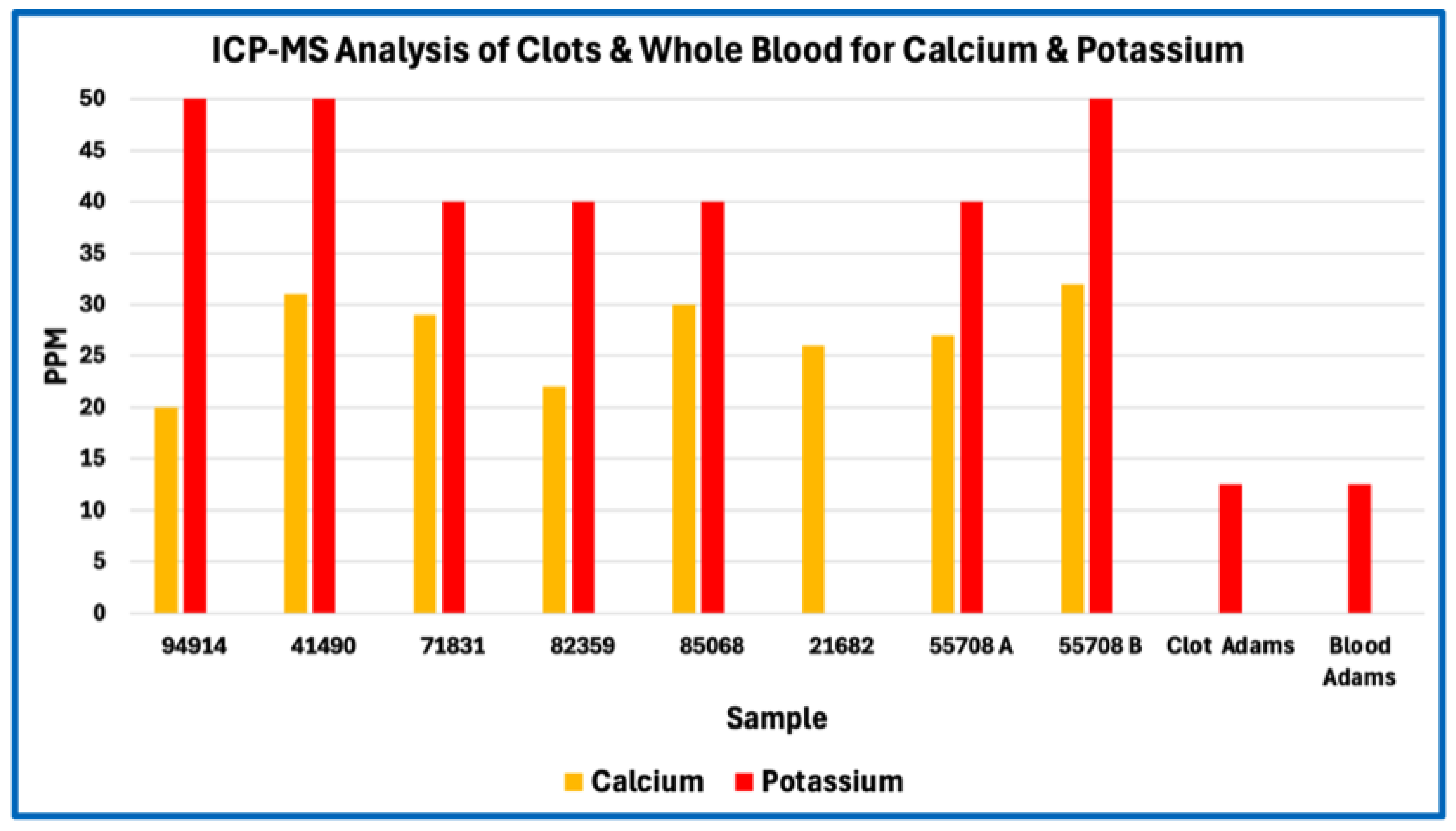
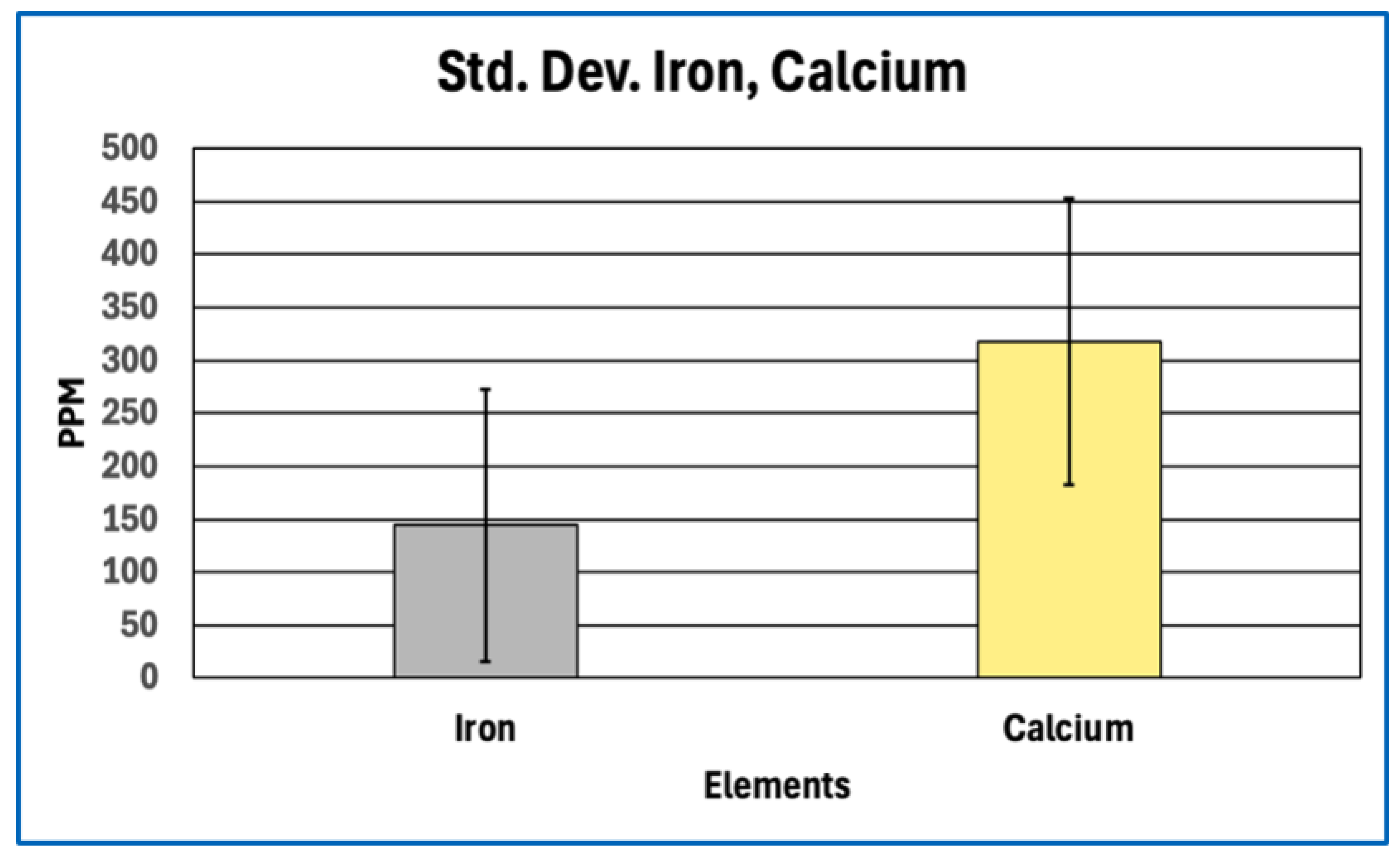
Phosphorus enrichment in AICs, observed both in absolute concentration and relative to other bulk elements, further distinguishes their composition from canonical fibrin-based thrombi. Phosphorus is not a major constituent of protein backbones and therefore serves as an indicator of phosphate-bearing components that are extrinsic to simple polypeptide matrices. The absence of commensurate calcium enrichment sufficient to indicate bulk apatite formation suggests that the detected phosphorus does not primarily reflect extensive mineralization.
At the same time, elemental analysis cannot resolve the specific chemical form of phosphorus present. The observed enrichment is therefore interpreted conservatively as evidence for incorporation or association of phosphate-bearing species within the matrix, rather than as proof of any particular molecular entity or bonding mechanism. This phosphorus signal, when considered alongside sulfur depletion and nitrogen–carbon imbalance, supports the inference that AICs comprise a hybrid matrix in which protein is structurally important but compositionally diluted by non-protein constituents.
Taken together, sulfur depletion and phosphorus enrichment define a compositional profile that is incompatible with a simple fibrin-dominant clot yet remains consistent with a biologically derived intravascular material. These elemental constraints establish the need for molecular-level analysis to determine the identities and relative abundances of the protein and non-protein components involved, which is addressed in the subsequent proteomic study.
Integration with Morphological–Histological Findings
The elemental findings reported here reinforce and extend the morphological and histological conclusions established in the first paper of this trilogy. In Paper 1, anomalous intravascular casts (AICs) were shown to form elongated, lumen-conforming structures with elastic mechanical properties and fibrinous lamination indicative of formation under active blood flow, yet with sparse cellular inclusion atypical of conventional thrombi. Those observations established a distinctive structural phenotype but could not resolve the biochemical nature of the matrix giving rise to that architecture.
Elemental analysis provides an independent and orthogonal line of evidence that complements these structural findings. The systematic depletion of sulfur and relative enrichment of phosphorus demonstrate that the material comprising AICs is compositionally inconsistent with a simple fibrin-dominant clot, despite exhibiting fibrin-like morphology under histological examination. This decoupling of appearance from bulk composition explains how AICs can display organized fibrillar architecture while failing to conform chemically to canonical thrombi, and it resolves potential ambiguity arising from histological resemblance alone.
Taken together, the convergence of a reproducible structural phenotype (morphology and histology) and a reproducible elemental phenotype (bulk composition) provides a stronger basis for defining AICs as a distinct intravascular entity than either line of evidence could alone. Morphological and histological data establish that the structures are real, coherent, and formed in vivo, while elemental data constrain what they are made of and rule out classification as ordinary antemortem thrombi or routine postmortem clots. This multi-modal consistency reduces the likelihood that the observed features arise from artefact, sampling bias, or isolated pathological variation.
Accordingly, the integration of structural and elemental evidence supports the interpretation that AICs represent a materially and biologically distinct class of intravascular cast. Defining the molecular constituents responsible for this combined phenotype requires resolution beyond elemental composition alone, providing a clear rationale for the proteomic analyses presented in the final paper of this series.
What Elemental Analysis Rules Out
The elemental composition of anomalous intravascular casts (AICs) allows several common alternative explanations for their origin to be excluded with confidence. These exclusions arise not from interpretation of structure or appearance, but from bulk chemical constraints that are incompatible with specific proposed mechanisms.
Exclusion of Simple Fibrin Overload
A straightforward explanation for unusual intravascular material might be excessive fibrin deposition or hyper-coagulability leading to unusually large or dense clots. However, simple fibrin overload would necessarily produce a matrix enriched in protein and therefore elevated in sulfur and nitrogen relative to bulk mass. The marked sulfur depletion observed in AICs is incompatible with this scenario, as it constrains the total fibrin-like protein fraction to a minor component of the material. Even accounting for analytical and postmortem effects, the sulfur deficit is too large and too consistent to be reconciled with a clot composed predominantly of fibrin. Elemental analysis therefore rules out simple quantitative excess of an otherwise normal fibrin matrix as an explanation for AIC composition.
Exclusion of Cellular Aggregation
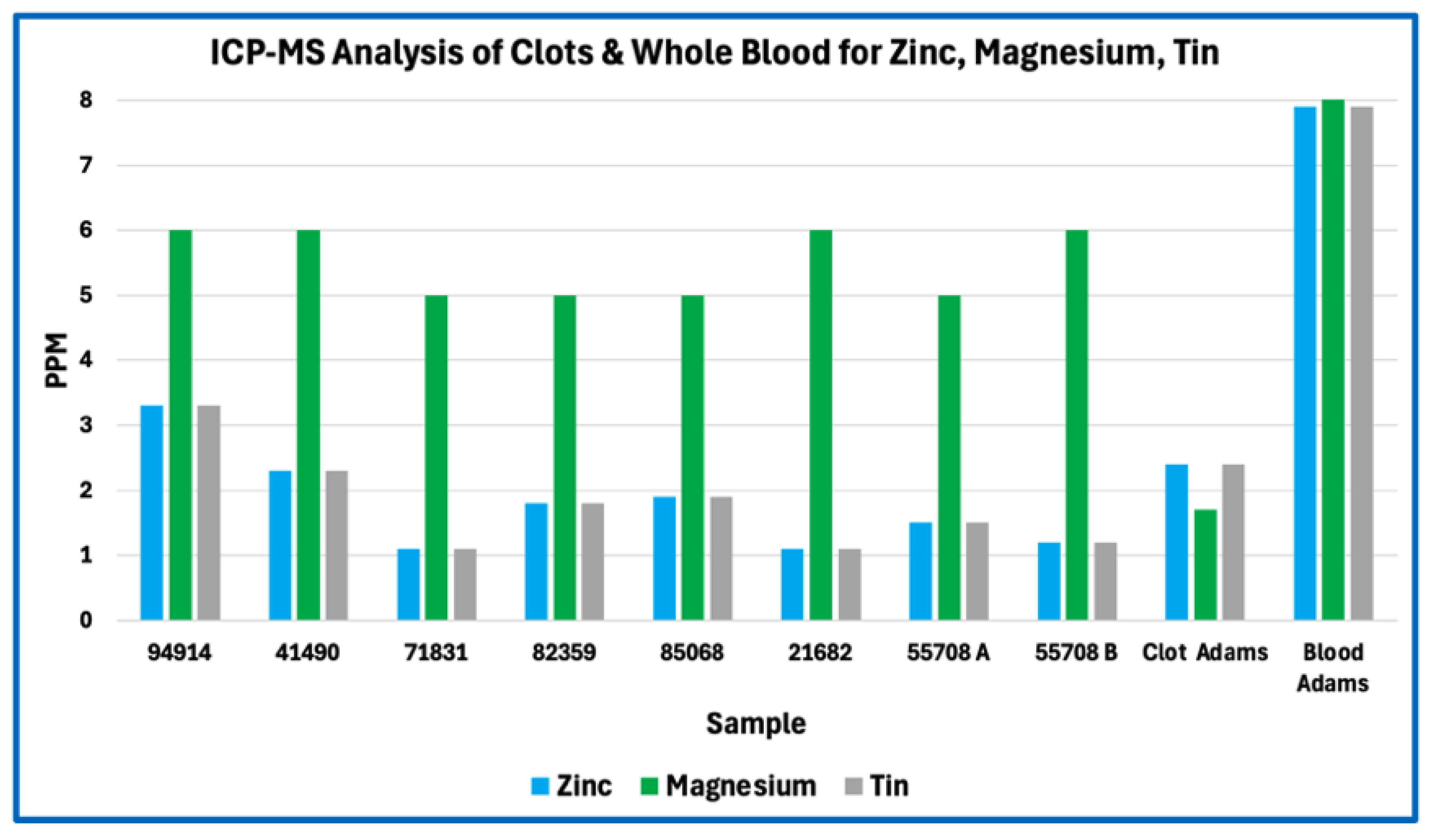
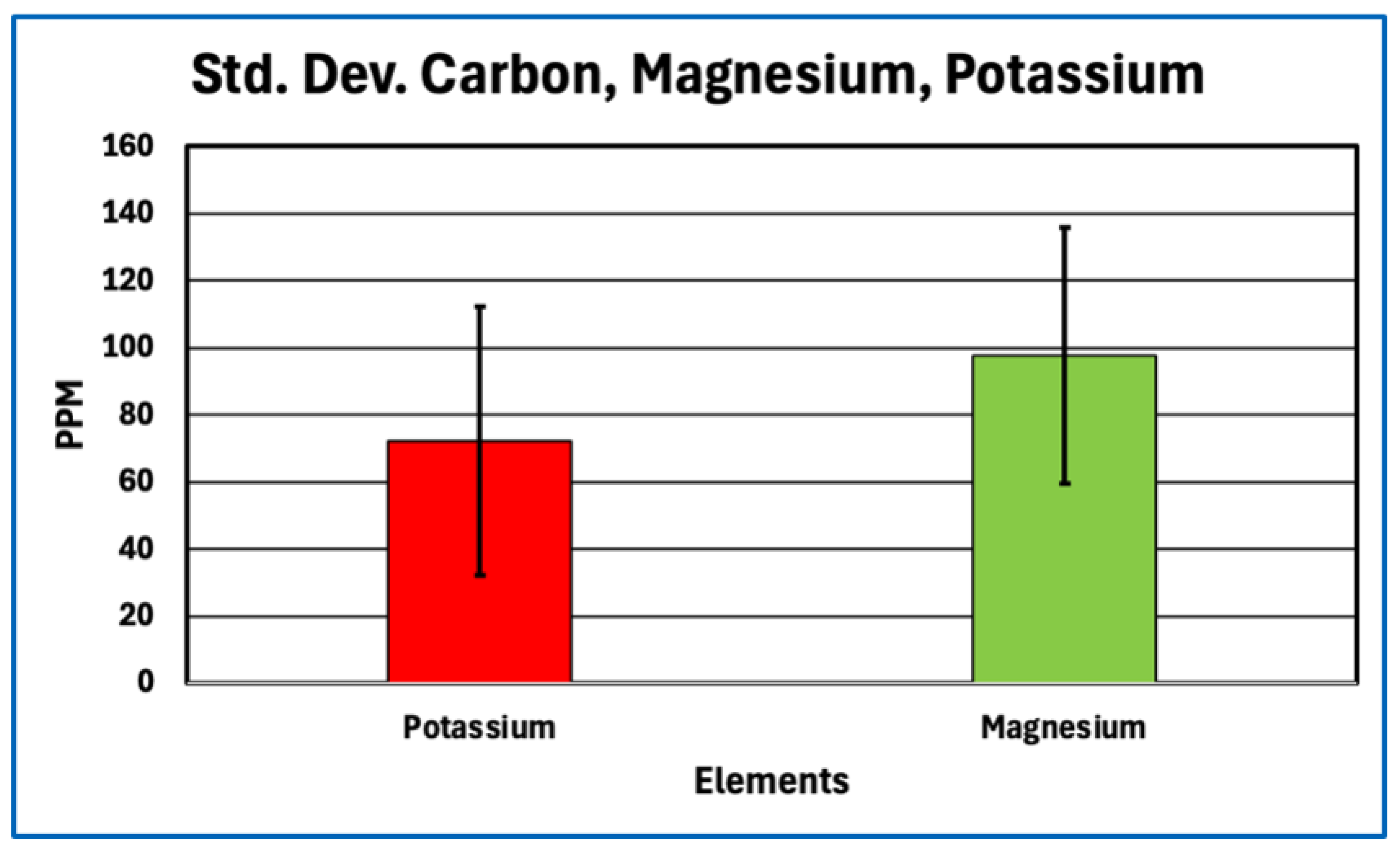
Aggregation or compaction of blood cells, platelets, or cellular debris could also produce coherent intravascular material without invoking novel chemistry. Such aggregates would be expected to retain elemental signatures characteristic of cellular biomass, including relatively high nitrogen and sulfur content from intracellular proteins, nucleoproteins, and membrane-associated enzymes, together with iron enrichment from hemoglobin where erythrocytes are involved. The elemental profiles of AICs do not display these features. Nitrogen and sulfur are depressed relative to protein expectations, and iron levels vary in a manner consistent with minor entrainment rather than bulk cellular composition. These findings are inconsistent with AICs being formed primarily by cellular aggregation or sedimentation.
Exclusion of Classical Coagulation Artifacts
Finally, classical coagulation artifacts arising from postmortem changes, fixation, dehydration, or handling would be expected to preserve the underlying elemental proportions of blood-derived proteins and cells, even if morphology were distorted. While such processes can redistribute soluble ions or modify surface chemistry, they do not selectively deplete sulfur-bearing amino acids or systematically enrich phosphorus relative to protein content. The reproducible elemental deviations observed in AICs across samples and laboratories therefore cannot be explained as artefacts of coagulation, fixation, or postmortem handling alone.
Collectively, these exclusions narrow the plausible interpretation of AICs to that of a biologically derived but compositionally abnormal intravascular matrix. Elemental analysis thus not only constrains what AICs are made of, but also rules out several conventional explanations that might otherwise account for their unusual appearance.
Limits of Elemental Inference
While elemental analysis provides strong constraints on bulk composition, it has intrinsic limitations that must be acknowledged. Elemental data describe the relative abundance of atoms within a material but do not specify how those atoms are organized into molecules, polymers, or complexes. As such, elemental analysis alone cannot resolve the identity, sequence, or relative abundance of specific proteins present within AICs, nor can it distinguish among protein isoforms or post-translationally modified variants.
In addition, elemental measurements do not define bonding states or molecular architecture. The presence of phosphorus, for example, cannot discriminate between inorganic phosphate, polyphosphate, phospholipids, nucleic acids, or other phosphate-bearing species, nor can sulfur measurements specify whether sulfur atoms reside in reduced thiols, disulfide bonds, or oxidised forms. Likewise, elemental ratios cannot determine whether non-protein constituents are covalently integrated into the matrix, weakly associated, or present as physically entrained phases.
Accordingly, elemental analysis should be understood as a constraint-setting tool rather than a molecular identification method. In the present study, elemental data establish that AICs are compositionally incompatible with canonical fibrin-dominant thrombi and require substantial non-protein contributions, but they do not identify the specific molecular constituents responsible for this abnormal matrix. Resolution of protein identity, relative abundance, and molecular interactions therefore requires complementary analytical approaches capable of operating at the molecular level.
These limitations define the scope boundary between elemental and proteomic analyses and provide the rationale for the molecular characterization presented in the final paper of this trilogy.
Rationale for Proteomic Analysis (Paper 3)
The elemental anomalies identified in this study define clear compositional constraints but simultaneously expose the limits of inference achievable without molecular resolution. Sulfur depletion constrains the bulk protein fraction, phosphorus enrichment indicates incorporation of phosphate-bearing components, and nitrogen–carbon imbalance confirms dilution of peptide backbone material; however, none of these elemental signatures can identify which proteins are present, which are absent, or how protein constituents are distributed within the anomalous intravascular cast (AIC) matrix. As a result, elemental analysis establishes that the matrix is abnormal but cannot determine what molecular species give rise to this composition.
Protein-level resolution is therefore required to reconcile the coexistence of fibrin-like morphology with a non-fibrin-dominant elemental profile. Proteomic analysis can determine whether fibrin chains are present in expected proportions, selectively depleted, modified, or accompanied by atypical proteins not characteristic of ordinary thrombi. It can also reveal whether the protein component of AICs represents a minor structural scaffold embedded within a broader non-protein matrix, as implied by sulfur-based mass constraints, or whether alternative protein assemblies contribute to the observed architecture.
Accordingly, the elemental findings presented here define the precise analytical gap that proteomics is uniquely positioned to fill. By identifying the protein constituents and their relative abundances, proteomic analysis provides the necessary molecular context to interpret the compositional anomalies revealed at the elemental level. This molecular characterization is addressed in the third and final paper of this trilogy, which builds directly on the structural and compositional constraints established in Papers 1 and 2.
Before molecular resolution is addressed directly, it is therefore useful to consider, in hypothesis-driven terms, the principal phosphate-bearing reservoirs that could plausibly account for the phosphorus enrichment constrained by elemental analysis.
Potential Sources of Phosphorus Enrichment in Anomalous Intravascular Casts
The consistent enrichment of phosphorus observed alongside marked sulfur depletion prompts consideration of potential non-fibrin sources of phosphate that could contribute to the elemental phenotype of anomalous intravascular casts (AICs).
Elemental analysis demonstrates that anomalous intravascular casts exhibit reproducible phosphorus enrichment concurrent with marked sulfur depletion relative to theoretical fibrinogen stoichiometry. Because sulfur provides a robust bulk proxy for protein content, this elemental imbalance constrains the clot matrix as protein-bearing but not protein-dominant. The excess phosphorus must therefore arise from non-fibrin phosphate reservoirs or from protein-associated phosphate contributions disproportionate to sulfur-containing amino acids. Several plausible sources merit consideration.
One potential contributor is membrane-derived phospholipid reservoirs associated with cellular disruption. Histological examination of AICs reveals extensive eosinophilic cytoplasmic material consistent with widespread erythrocyte lysis and fragmentation (Paper 1). While hemoglobin itself is not phosphorus-rich, erythrocyte and platelet membranes contain abundant phospholipids, each bearing phosphate head-groups. Fragmentation or persistence of membrane-derived material within the clot matrix could therefore elevate bulk phosphorus without restoring sulfur to levels expected for a fibrin-dominant protein scaffold. Such a mechanism would be consistent with phosphorus enrichment accompanied by depressed sulfur and nitrogen signals.
A second, non-mutually exclusive contributor is inorganic or polymeric phosphate incorporated during clot formation. Platelet activation releases inorganic polyphosphate from platelet dense granules, and this platelet-derived polyphosphate has been shown to associate with fibrin networks and modulate clot architecture by increasing fibrin fiber thickness and resistance to fibrinolysis (Smith & Morrissey, 2008; Mutch et al., 2010; Undas & Ariëns, 2011; Longstaff, 2015). Incorporation of polyphosphate-bearing reservoirs would elevate phosphorus content while contributing little sulfur or nitrogen, thereby shifting elemental ratios away from fibrinogen stoichiometry without requiring mineralization. The absence of calcium levels sufficient to indicate apatite formation supports the interpretation that phosphorus is present predominantly in non-mineralised forms.
While nucleic acids represent another class of potential phosphate-bearing reservoirs, their contribution is unlikely to dominate bulk phosphorus given the absence of corresponding nitrogen enrichment and the lack of histological features suggestive of nucleic acid–rich debris.
Additional phosphorus may also derive from phosphorylated proteins and other phosphate-bearing biomolecules present at low abundance. Proteomic analysis (Paper 3) indicates that AICs comprise a heterogeneous protein population, including fibrin family proteins, heme-associated proteins, and numerous additional constituents. Post-translational phosphorylation of proteins can contribute measurable phosphorus, and large-scale phosphoproteomic studies have demonstrated extensive phosphorylation of both host and viral proteins during SARS-CoV-2 infection (Bouhaddou et al., 2020). However, given the relatively small mass contribution of phosphosites—typically occurring on serine (Ser), threonine (Thr), and tyrosine (Tyr) residues via their hydroxyl groups—such modifications alone are unlikely to account for the magnitude of phosphorus enrichment observed, unless accompanied by substantial quantities of phosphate-bearing, non-protein material.
Finally, interaction between clot-associated proteins and specific ligands may further influence elemental balance and clot architecture. SARS-CoV-2 spike protein has been shown experimentally to bind fibrinogen and fibrin, inducing structural alterations and increased resistance to fibrinolysis in vitro (Grobbelaar et al., 2021). Additional studies have reported co-localization of spike protein with fibrin in experimental and pathological contexts, suggesting that spike–fibrin interactions may occur in vivo (Ryu et al., 2024). Detection of spike protein within retrieved thrombi from COVID-19 patients further supports the possibility that circulating spike fragments can associate with clot material (Pretorius et al., 2022). While spike protein itself is not a biochemical “phosphorus donor” in the enzymatic sense, it can carry phosphate groups via post-translational phosphorylation, raising the possibility that spike–fibrin interactions could contribute modestly to phosphate burden or alter clot composition indirectly. The quantitative contribution of such mechanisms to bulk elemental phosphorus, however, remains unresolved.
Taken together, these considerations suggest that phosphorus enrichment in AICs most plausibly reflects a composite contribution from membrane-derived phospholipid reservoirs, polyphosphate-bearing reservoirs, and other phosphate ester–rich reservoirs within a heterogeneous protein population rather than from fibrin alone.
Elemental analysis constrains the problem by excluding a purely proteinaceous or mineralised matrix, but it cannot resolve phosphate speciation or molecular origin. Targeted biochemical, lipidomic, and phosphate-speciation analyses will be required to discriminate among these candidate reservoirs and to determine their relative contributions to the anomalous elemental phenotype described here.