Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Standard Protocol Approvals, Registrations, and Patient Consents
Participants
Clinical and Demographic Variables
Health-Related Quality of Life
Fatigue, Mood, and Personality/Psychological Variables
Neuropsychological Assessment
Statistical Analyses
Results
Discussion
Author Contributions
Data Availability Statement
Acknowledgments
References
- Okuda, DT; Mowry, EM; Beheshtian, A. Incidental MRI anomalies suggestive of multiple sclerosis: the radiologically isolated syndrome. Neurology 2009. Available online: https://pubmed.ncbi.nlm.nih.gov/19073949/. [CrossRef]
- Bourdette, D; Simon, J. The radiologically isolated syndrome: is it very early multiple sclerosis? Neurology 2009. Available online: https://pubmed.ncbi.nlm.nih.gov/19073944/. [CrossRef] [PubMed]
- Granberg, T; Martola, J; Bergendal, G. Radiologically isolated syndrome—incidental MRI findings suggestive of multiple sclerosis: a systematic review. Multiple Sclerosis 2013. Available online: https://pubmed.ncbi.nlm.nih.gov/22760099/. [CrossRef] [PubMed]
- Okuda, DT; Siva, A; Kantarci, O; Inglese, M; Katz, I; Tutuncu, M. Radiologically isolated syndrome: 5-year risk for an initial clinical event. PLoS One 2014. Available online: https://pubmed.ncbi.nlm.nih.gov/24598783/. [CrossRef]
- Labiano-Fontcuberta, J. Benito-León, Radiologically isolated syndrome: An update on a rare entity. Mult Scler 2016. [Google Scholar] [CrossRef] [PubMed]
- Lebrun-Frenay; Kantarci, O.; Siva, A.; Sormani, M.P.; Pelletier, D.; Okuda, D.T. 10-year RISC study group on behalf of SFSEP, OFSEP. Radiologically Isolated Syndrome: 10-Year Risk Estimate of a Clinical Event. Ann Neurol 2020. [Google Scholar] [CrossRef]
- Kantarci, O; Lebrun-Frénay, C. Radiologically isolated syndrome: 10-year risk estimate of a clinical event. Annals of Neurology 2020. Available online: https://pubmed.ncbi.nlm.nih.gov/32500967/.
- Lebrun-Frénay, C; Rollot, F; Mondot, L. Risk factors and time to clinical symptoms of multiple sclerosis among patients with radiologically isolated syndrome. JAMA Network Open 2021. Available online: https://pubmed.ncbi.nlm.nih.gov/34698934/. [CrossRef]
- Lebrun-Frénay, C; Kantarci, O; Siva, A. The radiologically isolated syndrome: revised diagnostic criteria. The Lancet Neurology 2023. Available online: https://pubmed.ncbi.nlm.nih.gov/36864688/. [CrossRef]
- Lebrun-Frénay, C. Teriflunomide and time to clinical multiple sclerosis in radiologically isolated syndrome (TERIS). JAMA Neurology 2023. Available online: https://pubmed.ncbi.nlm.nih.gov/37466238/. [CrossRef]
- Benito-León, J.; Morales, J.M.; Rivera-Navarro, J.; Mitchell, A. A review about the impact of multiple sclerosis on health-related quality of life. Disabil Rehabil 2003, 25, 1291–1303. [Google Scholar] [CrossRef]
- Morales-Gonzáles, J.M.; Benito-León, J.; Rivera-Navarro, J.; Mitchell, A.J. GEDMA Study Group, A systematic approach to analyse health-related quality of life in multiple sclerosis: the GEDMA study. Mult Scler 2004, 10, 47–54. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Benito-León, J.; González, J.-M.M.; Rivera-Navarro, J. Quality of life and its assessment in multiple sclerosis: integrating physical and psychological components of wellbeing. Lancet Neurol 2005, 4, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Labiano-Fontcuberta; Aladro, Y.; Martínez-Ginés, M.L.; Ayuso, L.; Mitchell, A.J.; Puertas, V.; Cerezo, M.; Higueras, Y.; Benito-León, J. Psychiatric disturbances in radiologically isolated syndrome. J Psychiatr Res 2015. [Google Scholar] [CrossRef] [PubMed]
- Labiano-Fontcuberta, A. Cognitive deficits in radiologically isolated syndrome compared with multiple sclerosis and healthy controls. Multiple Sclerosis 2016. Available online: https://pubmed.ncbi.nlm.nih.gov/26084350/. [CrossRef] [PubMed]
- Joly J, Alexithymia and quality of life in radiologically isolated syndrome. Brain and Behavior 2024. Available online: https://pubmed.ncbi.nlm.nih.gov/39341200/.
- Lebrun, C. Evaluation of quality of life and fatigue in radiologically isolated syndrome. Revue Neurologique (Paris) 2016. Available online: https://pubmed.ncbi.nlm.nih.gov/27158044/. [CrossRef]
- Oh, J.; Arbour, N.; Giuliani, F.; Guenette, M.; Kolind, S.; Lynd, L.; Marrie, R.A.; Metz, L.M.; Prat, A.; Schabas, A.; Smyth, P.; Tam, R.; Traboulsee, A.; Yong, V.W.; Patten, S.B. The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics. Ther Adv Neurol Disord 2024, 17, 17562864241273045. [Google Scholar] [CrossRef]
- Thompson, AJ; Banwell, BL; Barkhof, F. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. The Lancet Neurology 2018. Available online: https://pubmed.ncbi.nlm.nih.gov/29275977/. [CrossRef]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale. Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef]
- Rivera-Navarro, J.; Benito-León, J.; Morales-González, J.M. Searching for more specific dimensions for the measurement of quality of life in multiple sclerosis. Rev Neurol 2001, 32, 705–713. [Google Scholar]
- Morales-Gonzáles, J.M.; Benito-León, J.; Rivera-Navarro, J.; Mitchell, A.J. GEDMA Study Group, A systematic approach to analyse health-related quality of life in multiple sclerosis: the GEDMA study. Mult Scler 2004, 10, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Benito-León, J.; Mitchell, A.J.; Rivera-Navarro, J.; Morales-González, J.M. Impaired health-related quality of life predicts progression of disability in multiple sclerosis. Euro J of Neurology 2013, 20, 79–86. [Google Scholar] [CrossRef]
- Labiano-Fontcuberta; Mitchell, A.J.; Moreno-García, S.; Puertas-Martín, V.; Benito-León, J. Impact of anger on the health-related quality of life of multiple sclerosis patients. Mult Scler 2015, 21, 630–641. [Google Scholar] [CrossRef] [PubMed]
- Cella, DF; Dineen, K; Arnason, B. Validation of the functional assessment of multiple sclerosis quality of life instrument. Neurology 1996. Available online: https://pubmed.ncbi.nlm.nih.gov/8710066/. [CrossRef] [PubMed]
- EuroQol - a new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G. Beck Depression Inventory–II; 2011. [Google Scholar] [CrossRef]
- Spielberger, C.D.; Gonzalez-Reigosa, F.; Martinez-Urrutia, A.; Natalicio, L.F.; Natalicio, D.S. The State-Trait Anxiety Inventory. Revista Interamericana de Psicología/Interamerican Journal of Psychology 2017, 5. [Google Scholar] [CrossRef]
- Busse, M.; Whiteside, D.; Waters, D.; Hellings, J.; Ji, P. Exploring the reliability and component structure of the personality assessment inventory in a neuropsychological sample. Clin Neuropsychol 2014, 28, 237–251. [Google Scholar] [CrossRef]
- Rao, S.M. A manual for the brief repeatable battery of neuropsychological tests in multiple sclerosis; Medical College of Wisconsin: Milwaukee, 1990; p. 1696. [Google Scholar]
- Lebrun, C.; Blanc, F.; Brassat, D.; Zephir, H.; de Seze, J. behalf of CFSEP, Cognitive function in radiologically isolated syndrome. Mult Scler 2010, 16, 919–925. [Google Scholar] [CrossRef]
- Buschke, H.; Fuld, P.A. Evaluating storage, retention, and retrieval in disordered memory and learning. Neurology 1974, 24, 1019–1025. [Google Scholar] [CrossRef]
- Portaccio, E.; Goretti, B.; Zipoli, V.; Siracusa, G.; Sorbi, S.; Amato, M.P. A short version of Rao’s Brief Repeatable Battery as a screening tool for cognitive impairment in multiple sclerosis. The Clinical Neuropsychologist 2009, 23, 268–275. [Google Scholar] [CrossRef]
- Scarpina, F.; Tagini, S. The Stroop Color and Word Test. Front. Psychol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Peña-Casanova, J.; Casals-Coll, M.; Quintana, M.; Sánchez-Benavides, G.; Rognoni, T.; Calvo, L.; Palomo, R.; Aranciva, F.; Tamayo, F.; Manero, R.M. Estudios normativos españoles en población adulta joven (Proyecto NEURONORMA jóvenes): métodos y características de la muestra. Neurología 2012, 27, 253–260. [Google Scholar] [CrossRef]
- Bauer, K.; Malek-Ahmadi, M. Meta-analysis of Controlled Oral Word Association Test (COWAT) FAS performance in amnestic mild cognitive impairment and cognitively unimpaired older adults. Applied Neuropsychology: Adult 2021, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Benito-León, J.; Del Pino, A.B.; Aladro, Y.; Cuevas, C.; Domingo-Santos, Á.; Galán Sánchez-Seco, V.; Labiano-Fontcuberta, A.; Gómez-López, A.; Salgado-Cámara, P.; Costa-Frossard, L.; Monreal, E.; Sainz De La Maza, S.; Matías-Guiu, J.A.; Matías-Guiu, J.; Delgado-Álvarez, A.; Montero-Escribano, P.; Martínez-Ginés, M.L.; Higueras, Y.; Ayuso-Peralta, L.; Malpica, N.; Melero, H. Abnormal functional connectivity in radiologically isolated syndrome: A resting-state fMRI study. Mult Scler 2023, 29, 1393–1405. [Google Scholar] [CrossRef]
- Harrison, A.M.; Safari, R.; Mercer, T.; Picariello, F.; Van Der Linden, M.L.; White, C.; Moss-Morris, R.; Norton, S. Which exercise and behavioural interventions show most promise for treating fatigue in multiple sclerosis? A network meta-analysis. Mult Scler 2021, 27, 1657–1678. [Google Scholar] [CrossRef] [PubMed]
- Boda, M.R.; Rees, A.E.; Goldsworthy, M.R.; Evangelou, N.; Sidhu, S.K.; Smith, A.E. Scoping review: The effects of diet, physical activity and sleep on motor and physical fatigue in people with multiple sclerosis. Neuroscience 2025, 568, 166–194. [Google Scholar] [CrossRef]
- Cruz Rivera, S.; Buxhoeveden, S.; Aiyegbusi, O.L.; Bozinov, N.; Kamudoni, P.; McBurney, R.; Calvert, M. The importance of patient-reported outcomes: A call for their integration in the routine care of patients with multiple sclerosis. Mult Scler 2025, 31, 1518–1530. [Google Scholar] [CrossRef]
| Variable | Radiologically isolated syndrome (n=30) | Relapsing–remitting multiple sclerosis (n=29) | Healthy controls (n=30) | p value |
| Age (years), mean ± SD | 41.7 ± 8.1 | 41.4 ± 7.2 | 40.9 ± 7.8 | 0.925* |
| Female sex, n (%) | 26 (86.7%) | 20 (69.0%) | 24 (80.0%) | 0.247† |
| Education (years),median[Q1, Q3] | 15 [12–17.2] | 15 [11.5–17.5] | 15 [13-7–17.0] | 0.662‡ |
| Time since diagnosis (years), median[Q1, Q3] | 2.5 [1.0–8.7] | 4.0 [2.0–10.5] | — | 0.399** |
| Disability (Expanded Disability Status Scale), median [Q1, Q3] | — | 2.0 [1.0–3.0] | — | — |
| Fatigue impact (Daily Fatigue Impact Scale total score), median [Q1, Q3] | 3.0 [0.0, 13.0] | 7.0 [2.0, 15.0] | 2.0 [0.0, 8.2] | 0.051‡ |
| Depressive symptoms (Beck Depression Inventory total score), median [Q1, Q3] | 6.5 [3.0, 13.5] | 7.0 [2.7, 15.2] | 6.0 [1.5, 8.0] | 0.257‡ |
| State anxiety (State-Trait Anxiety Inventory-State total score), median [Q1, Q3] | 15.0 [8.0, 22.5] | 14.0 [5.0, 20.0] | 11.5 [5.7, 17.0] | 0.240‡ |
| Cognitive performance (component score; z), mean ± SD | -0.37 ± 1.12 | -0.04 ± 0.93 | 0.39 ± 0.81 | 0.013* |
| Psychological distress (component score; z), median [Q1, Q3] | -0.12 [-0.55, 0.96] | -0.10 [-0.59, 0.91] | -0.54 [-0.86, -0.14] | 0.014‡ |
| Outcome | Radiologically isolated syndrome | Relapsing–remitting multiple sclerosis | Healthy controls | p value† |
| Functional Assessment of Multiple Sclerosis) | ||||
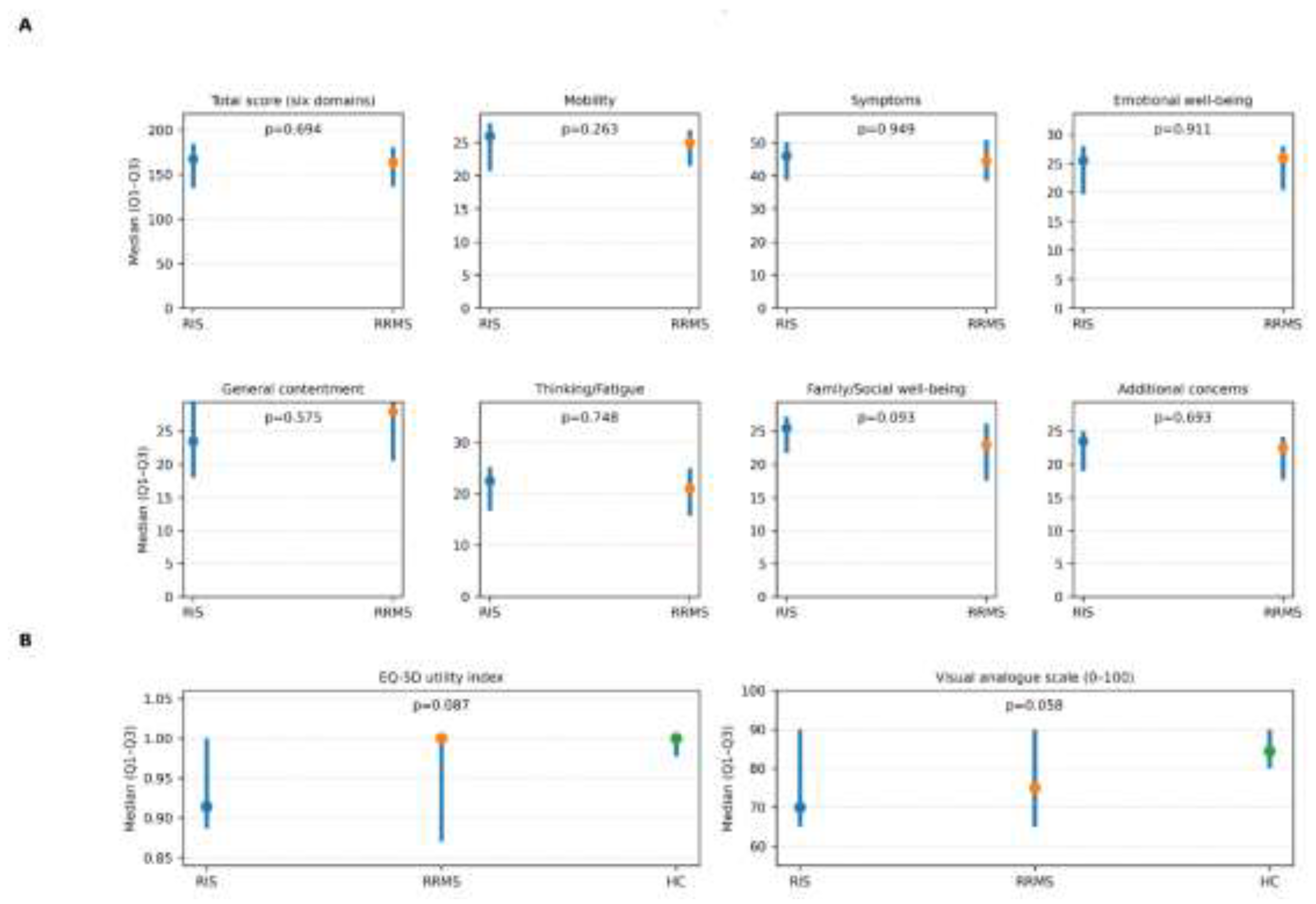
| Total score (six domains), median [Q1, Q3] | 167.5 [135.2, 185.0] | 164.0 [136.2, 181.0] | 0.694 | |
| Mobility, median [Q1, Q3] | 26.0 [20.7, 28.0] | 25.0 [21.5, 27.0] | — | 0.263 |
| Symptoms, median [Q1, Q3] | 46.0 [38.7, 50.2] | 44.5 [38.5, 51.0] | — | 0.949 |
| Emotional well-being, median [Q1, Q3] | 25.5 [19.7, 28.0] | 26.0 [20.5, 28.0] | — | 0.911 |
| General contentment, median [Q1, Q3] | 23.5 [18.0, 30.2] | 28.0 [20.5, 30.2] | — | 0.575 |
| Thinking/Fatigue, median [Q1, Q3] | 22.5 [16.7, 25.2] | 21.0 [15.7, 25.0] | — | 0.748 |
| Family/Social well-being, median [Q1, Q3] | 25.5 [21.7, 27.2] | 23.0 [17.5, 26.2] | — | 0.093 |
| Additional concerns, median [Q1, Q3] | 23.5 [19.0, 25.0] | 22.5 [17.7–24.2] | — | 0.693 |
| Generic HRQoL (EuroQol-5D) | ||||
| EQ-5D utility index, median [Q1–Q3] | 0.914 [0.887, 1.000] | 1.000 [0.870, 1.000] | 1.000 [0.978, 1.000] | 0.087 |
| Visual analogue scale (0–100), median [Q1–Q3] | 70.0 [65.0, 90.0] | 75.0 [65.0, 90.0] | 84.5 [80.0, 90.0] | 0.058 |
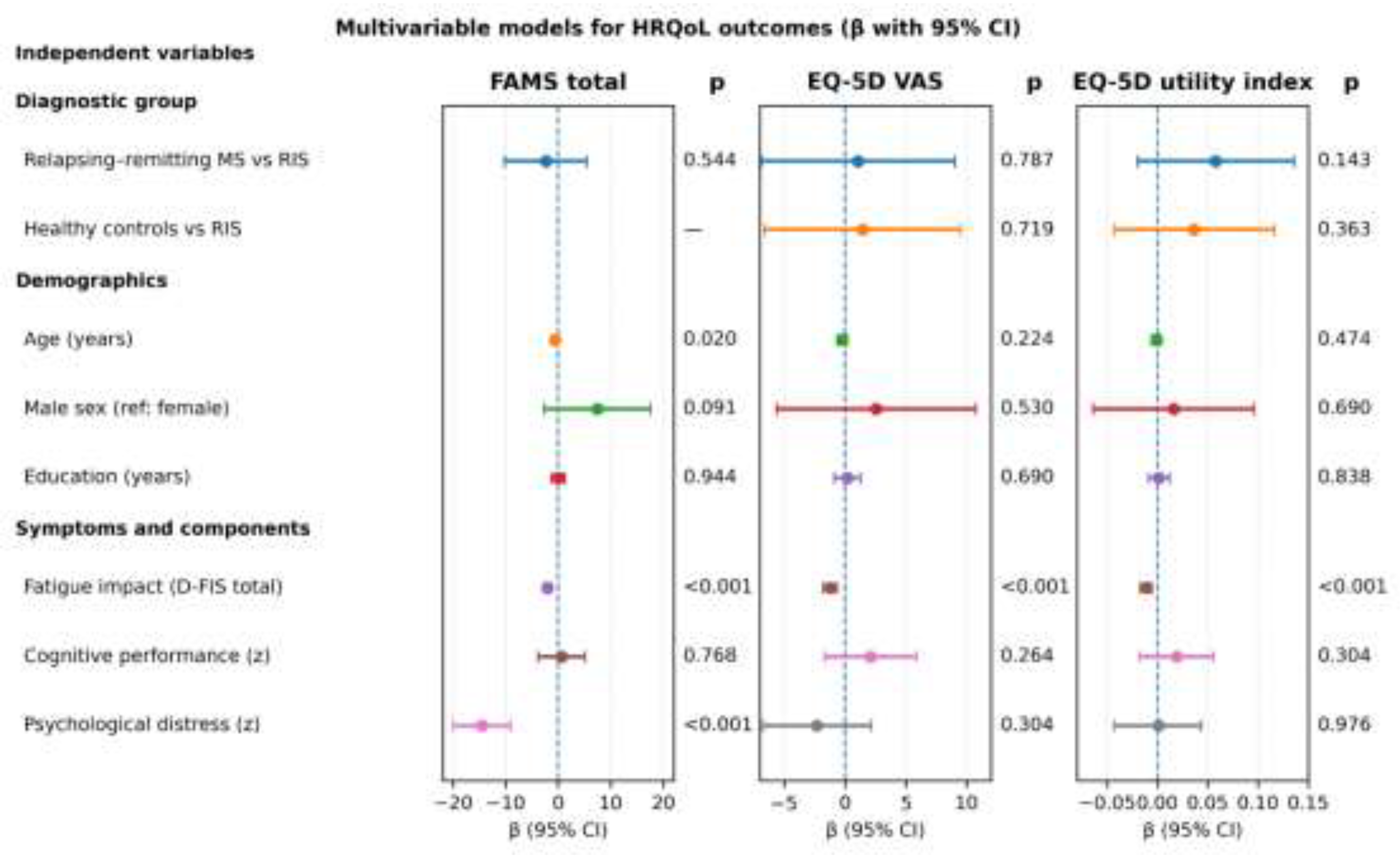
| Predictor | Functional Assessment of Multiple Sclerosis total β (95% CI) | p value | EuroQol-5D visual analogue scale β (95% CI) | p value | EQ-5D utility index β (95% CI) | p value |
| Relapsing–remitting multiple sclerosis vs. radiologically isolated syndrome | -2.37 (-10.18, 5.44) | 0.544 | 1.08 (-6.86, 9.01) | 0.787 | 0.058 (-0.020, 0.136) | 0.143 |
| Healthy controls vs. radiologically isolated syndrome | — | — | 1.46 (-6.60, 9.52) | 0.719 | 0.036 (-0.043, 0.116) | 0.363 |
| Age (years) | -0.60 (-1.09, -0.10) | 0.020 | -0.26 (-0.68, 0.16) | 0.224 | -0.001 (-0.006, 0.003) | 0.474 |
| Male sex (reference: female) | 7.44 (-2.71, 17.59) | 0.091 | 2.58 (-5.58, 10.75) | 0.530 | 0.016 (-0.064, 0.096) | 0.690 |
| Education (years) | 0.04 (-1.17, 1.25) | 0.944 | 0.22 (-0.86, 1.29) | 0.690 | 0.001 (-0.009, 0.012) | 0.838 |
| Fatigue impact (Daily Fatigue Impact Scale total score) | -2.04 (-2.66, -1.43) | <0.001 | -1.21 (-1.76, -0.67) | <0.001 | -0.011 (-0.017, -0.006) | <0.001 |
| Cognitive performance (component score; z) | 0.66 (-3.82, 5.13) | 0.768 | 2.12 (-1.64, 5.88) | 0.264 | 0.019 (-0.018, 0.056) | 0.304 |
| Psychological distress (component score; z) | -14.53 (-19.98, -9.08) | <0.001 | -2.30 (-6.75, 2.14) | 0.304 | 0.001 (-0.043, 0.044) | 0.976 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




