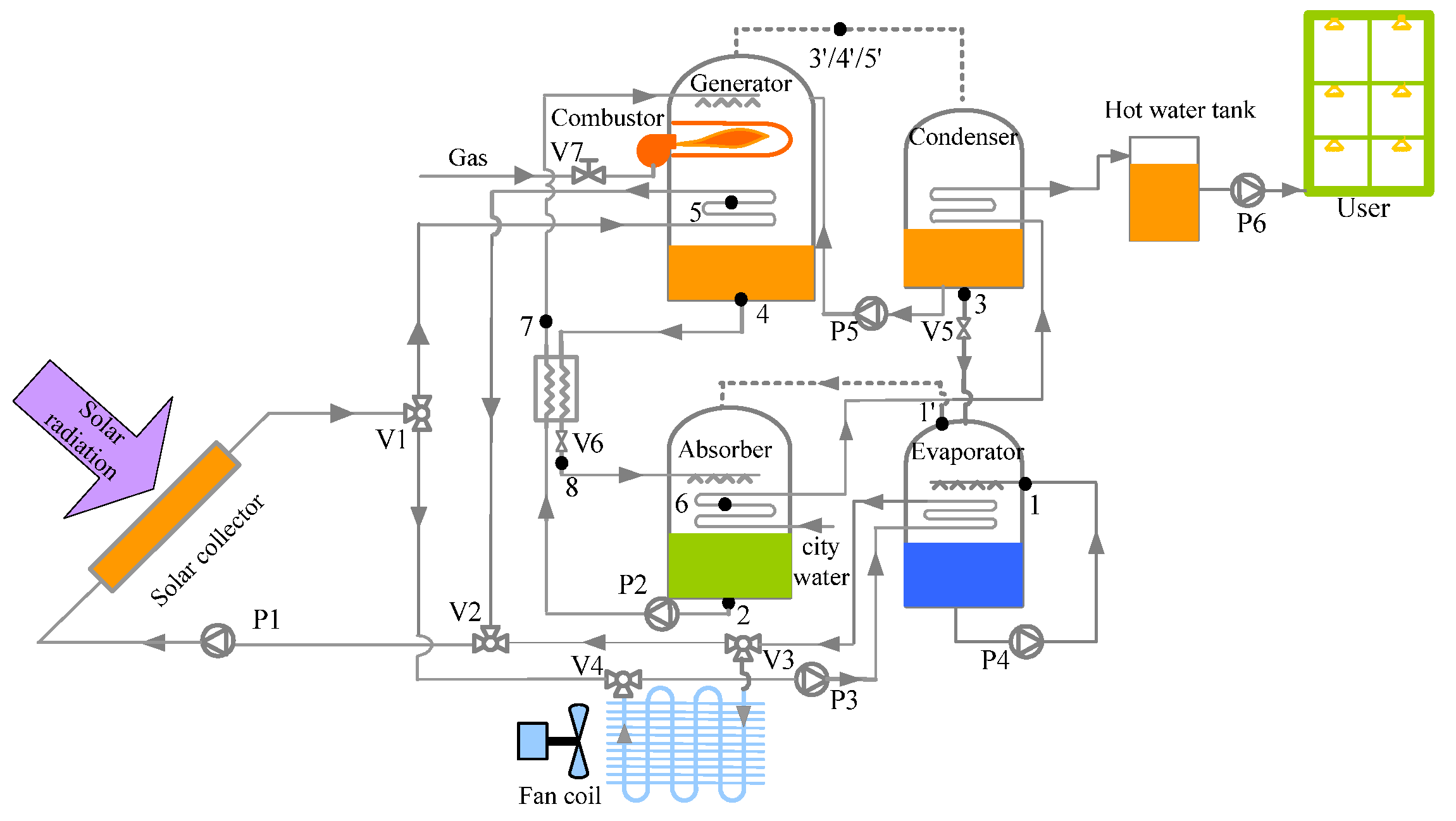
4. Results and Discussion
Based on the above models, the performance of SAAHP with LiBr/H
2O and LiNO
3/H
2O was simulated under the given operating conditions shown in
Table 9. Considering the influence of the flow ratio to the power consumption of solution pump and the COP of absorption heat pump, the performance of SAAHP with LiBr/H
2O and LiNO
3/H
2O was compared under the same flow ratio.
The main parameters, heat load and COP of AHP with LiBr/H
2O and LiNO
3/H
2O at the refrigeration flow rate D=1 kg/s are listed in
Table 10. The required generator temperature of AHP using LiNO
3/H
2O was lower than that using LiBr/H
2O working fluid, and the former reached higher COP throughout a year. This is mainly because of the differences in the absorption property between the two working fluids.
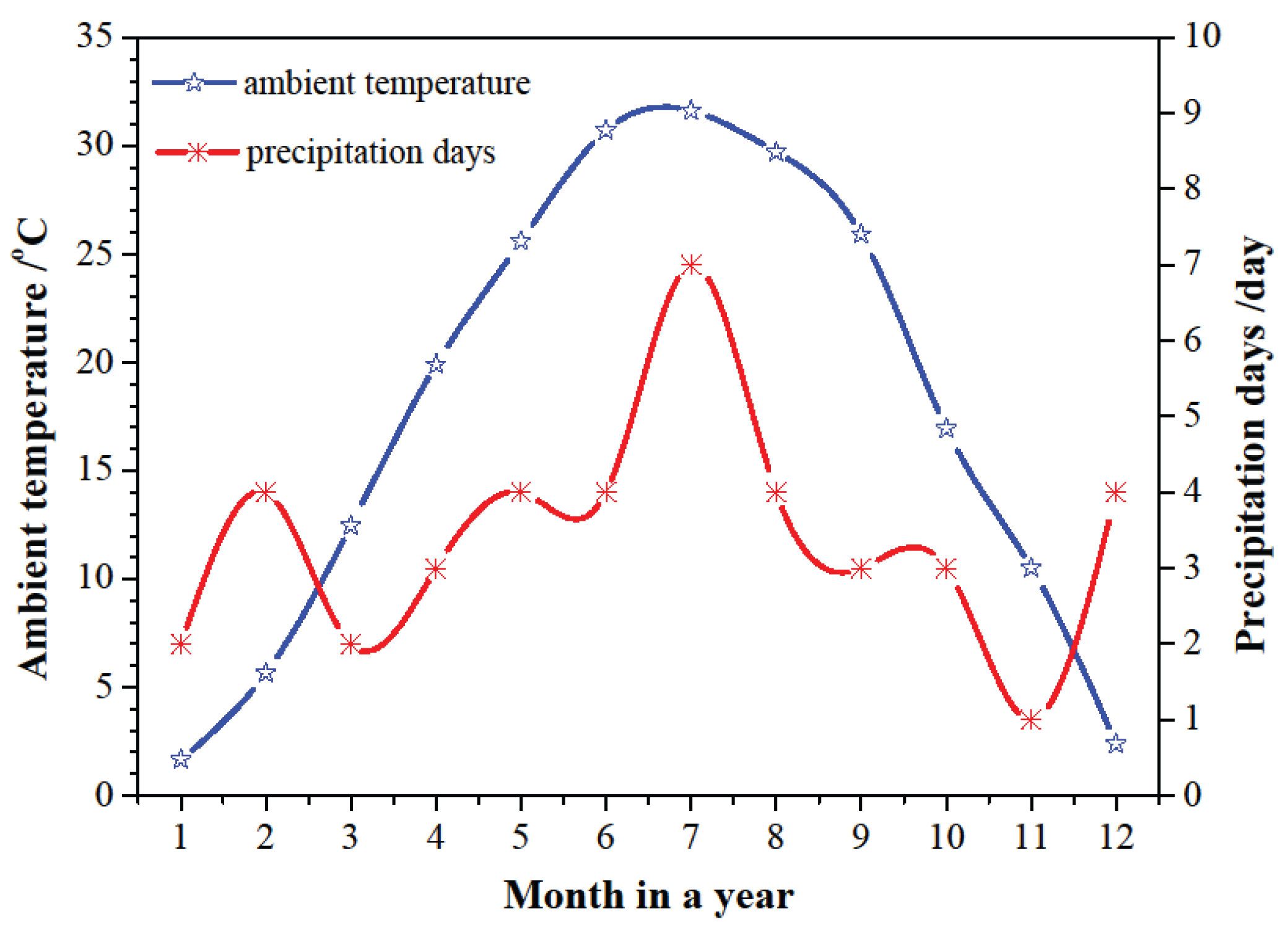
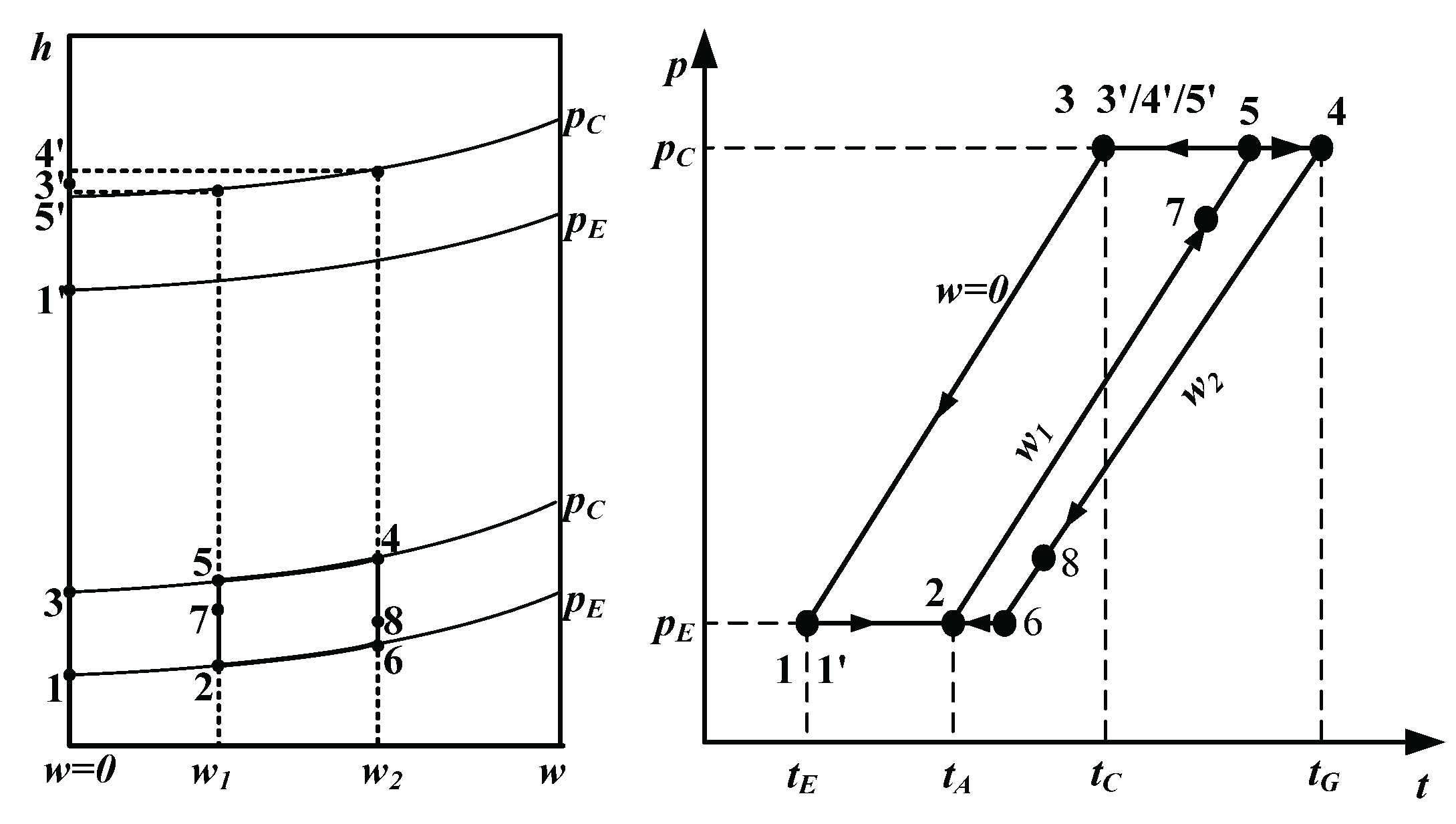
The switching time of the operating mode is determined with the ambient temperature and the heating capacity of SAAHP system. When SAAHP system runs in SD mode, the required ambient temperature in cold, moderate and hot seasons should be above 21.2
oC, 24.2
oC and 26.2
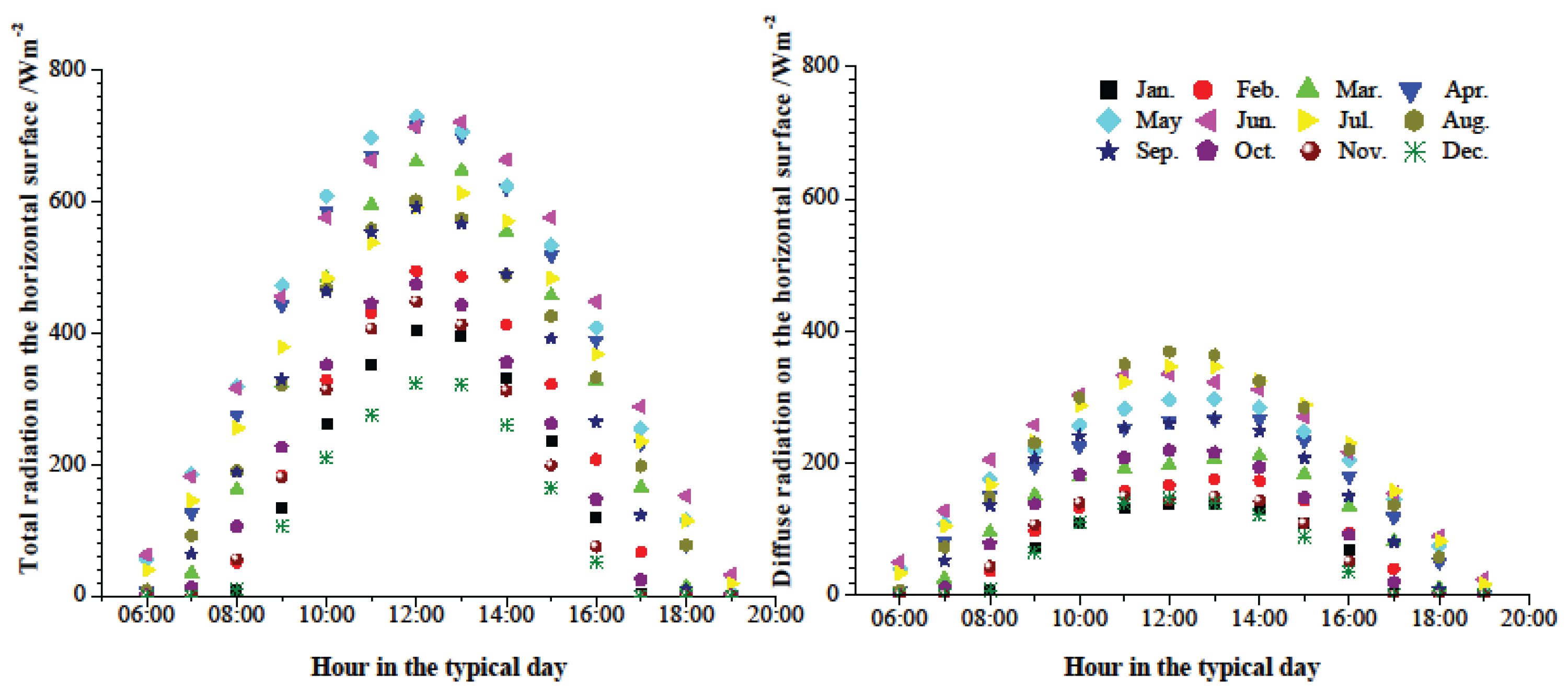
oC, respectively, under the given operating conditions. Based on the highest ambient temperature in a typical day shown in
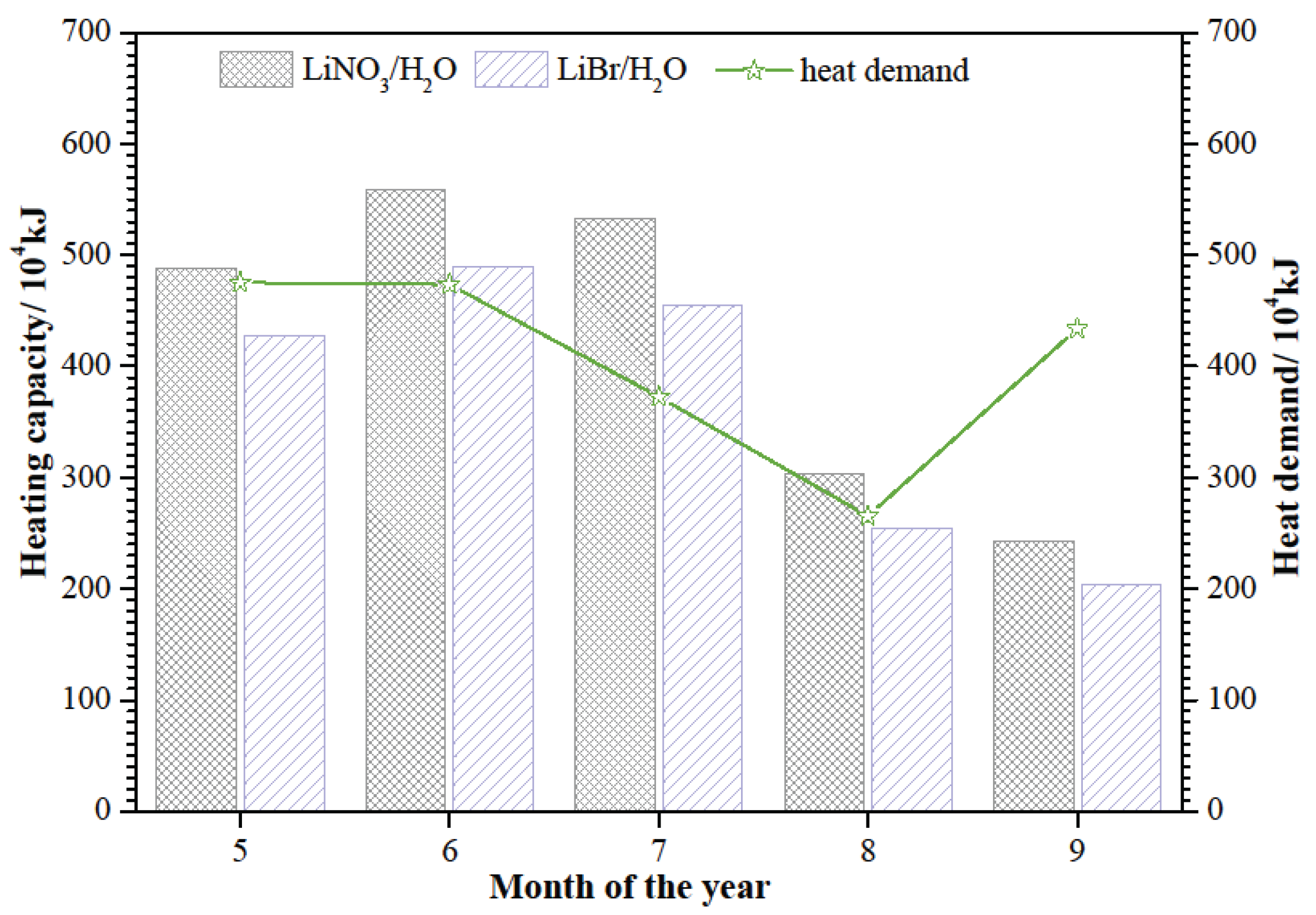
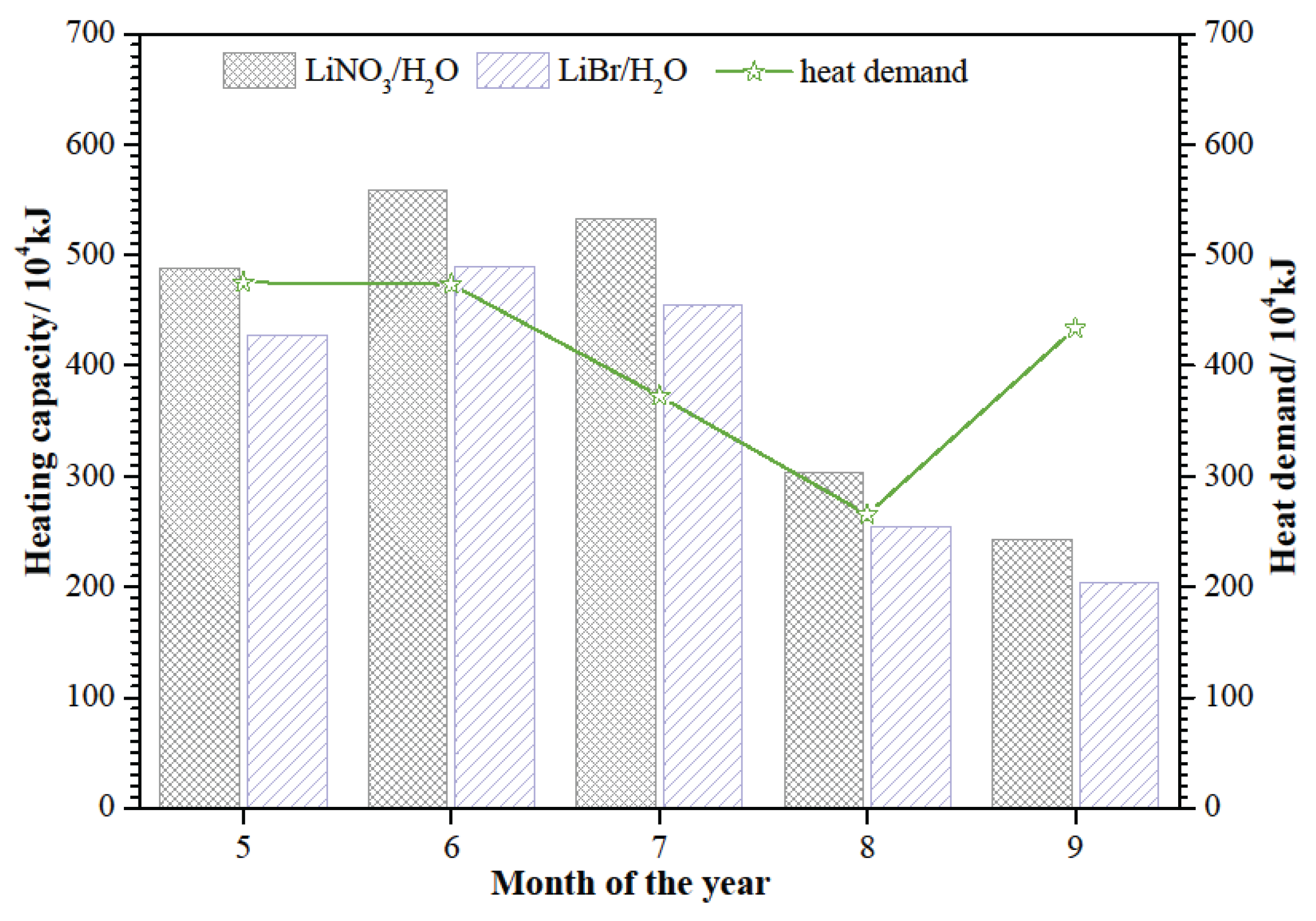
Figure 3, it is clear that the requirements can be meted in May to September. Beyond that, the heating capacity of SAAHP driven by solar energy in the above five months should meet the heat demand of the building. The heating capacity of SAAHP from May to September is presented in
Figure 6. The heating capacity of SAAHP with LiNO
3/H
2O can meet the heat demand from May to August, so SAAHP with LiNO
3/H
2O shall be operated in SD mode during this period. However, the heating capacity of SAAHP with LiBr/H
2O just meets the heat demand in June and July, so SAAHP with LiBr/H
2O shall be operated in SD mode in the two months. It is indicated that LiNO
3/H
2O is more suitable for SD mode for the reason that SAAHP running in SD mode based on LiNO
3/H
2O is able to utilize more solar energy. As seen in
Figure 7, when the solar energy is used as the driving heat source in SD mode, the outlet temperature of the solar collector based on LiNO
3/H
2O is below 88
oC, which is about 4
oC lower than that based on LiBr/H
2O. The reduction of the outlet temperature can improve the collector efficiency.
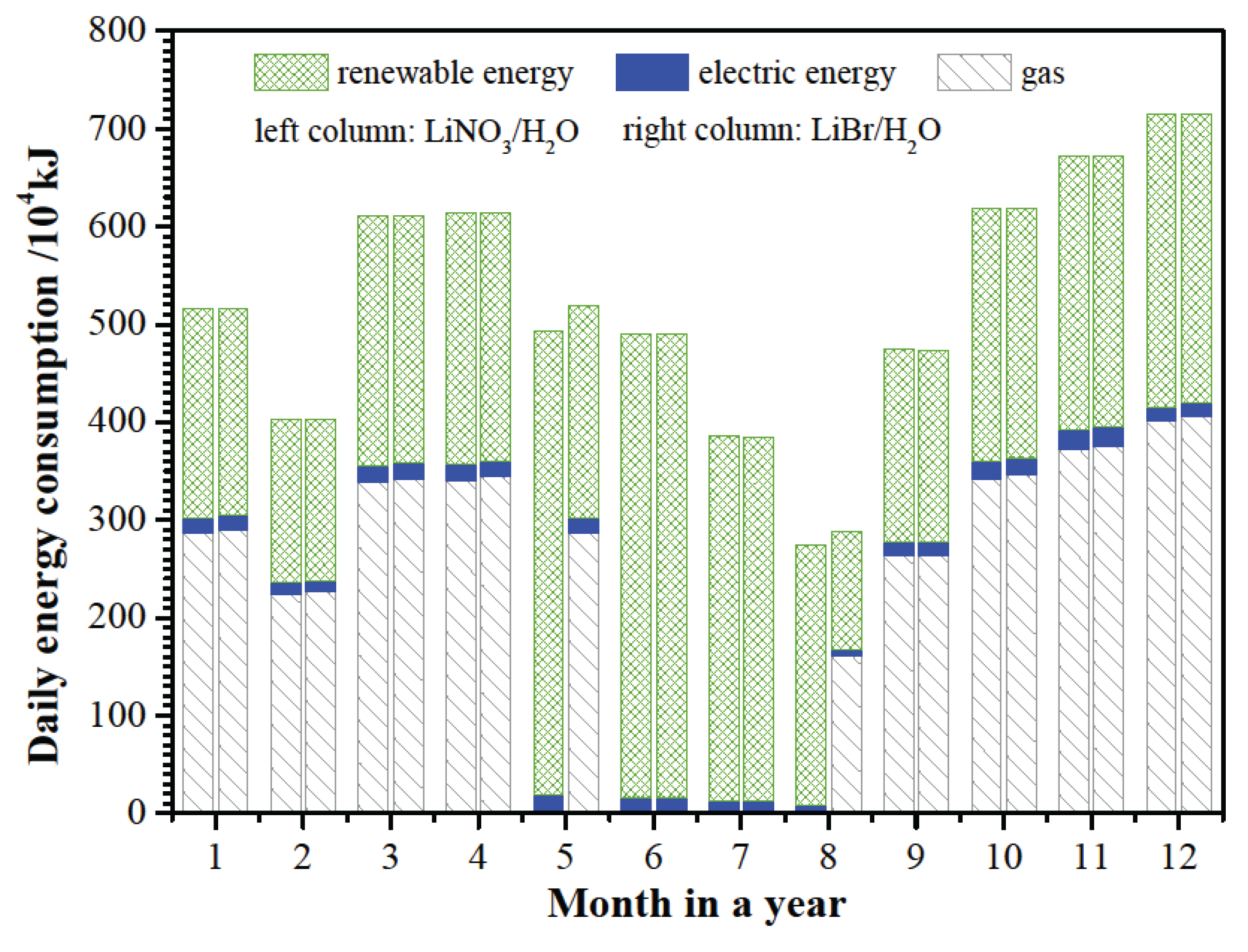
Figure 8 shows the daily energy consumption of SAAHP based on LiBr/H
2O and LiNO
3/H
2O working fluids. Results indicated that the daily energy consumption of SAAHP running in GD mode based on different working fluids has no significant difference. However, SAAHP based on LiNO
3/H
2O is able to utilize more renewable energy than that based on LiBr/H
2O since the former can be operated in SD mode for a longer period. Compared with the renewable energy and nature gas, the electric energy consumption by solution pump and fan is very small throughout a year.
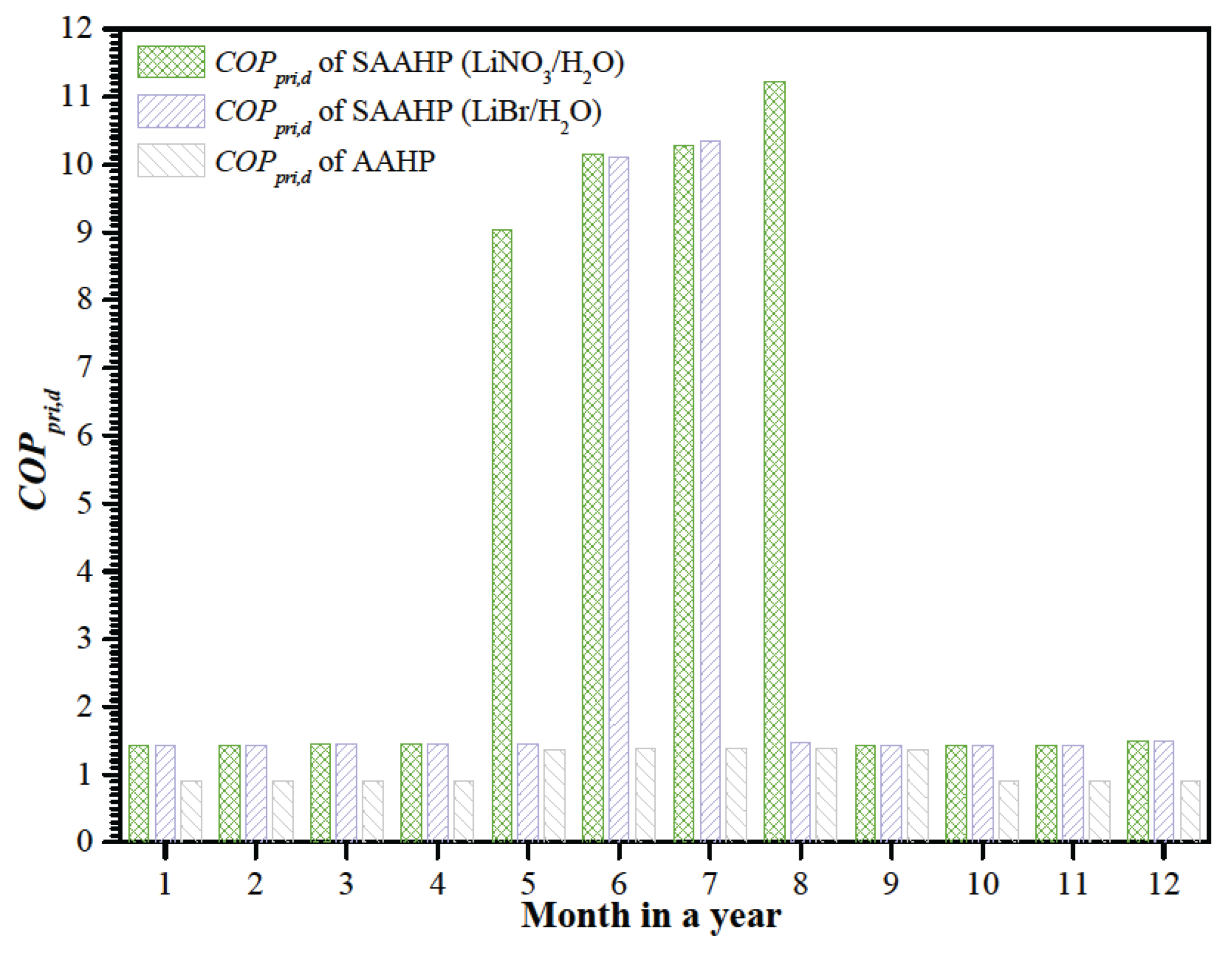
The daily primary energy
COP (
COPpri,d) of SAAHP based on LiBr/H
2O and LiNO
3/H
2O are presented in
Figure 9, and compared with that of a LiBr/H
2O AAHP with 300
kW heating capacity under the same operating conditions. Results indicated SAAHP running in SD mode achieves the highest
COPpri,d of 1.12 due to simultaneously utilizing solar and air energy, but AAHP just achieves the highest
COPpri,d of 1.40 since it misses the utilization of high grade solar energy in hot season.
COPpri,d of AAHP decreases with the decreasing ambient temperature. When the ambient temperature is too low to be utilized as the low temperature heat source,
COPpri,d of AAHP drops to the efficiency ( 90%) of the gas-fired combustor installed in AAHP. In contrast, SAAHP can utilize solar energy instead of air energy at a low ambient temperature, and still achieve a higher COPpri,d above 1.43 in cold regions.
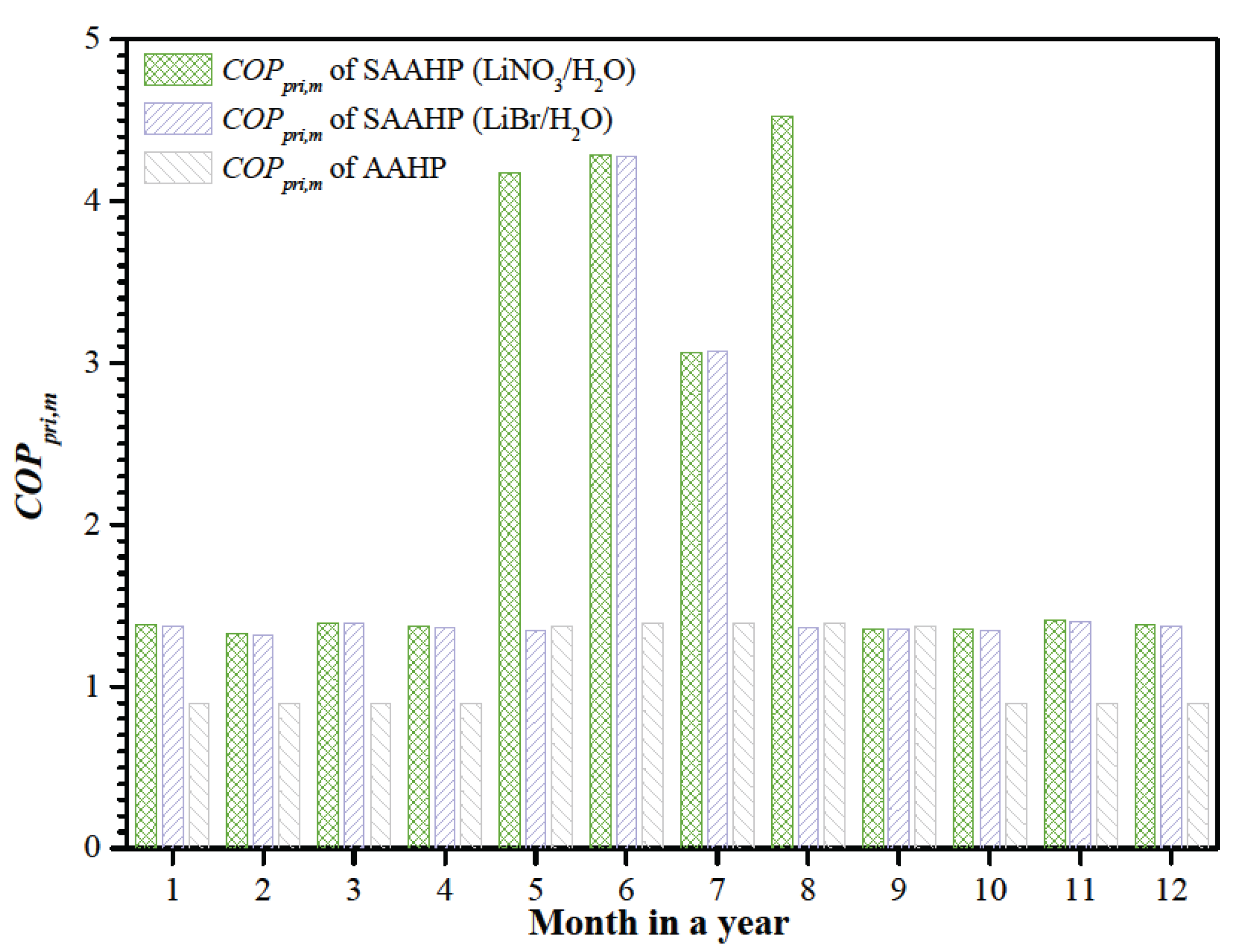
Figure 10 shows the monthly primary energy
COP (
COPpri,m) of SAAHP and AAHP. Considering the monthly precipitation days,
COPpri,m of SAAHP running in GD mode has no obvious change, but that in SD mode is reduced relative to
COPpri,d, especially in the rainy season of July. Though the precipitation day has a great influence on the primary energy consumption in SD mode,
COPpri,m of SAAHP based on LiNO
3/H
2O working fluids is still above 3.07. By comparing
COPpri,m of SAAHP with AAHP, it is seen that the former has a significant advantage throughout a year.
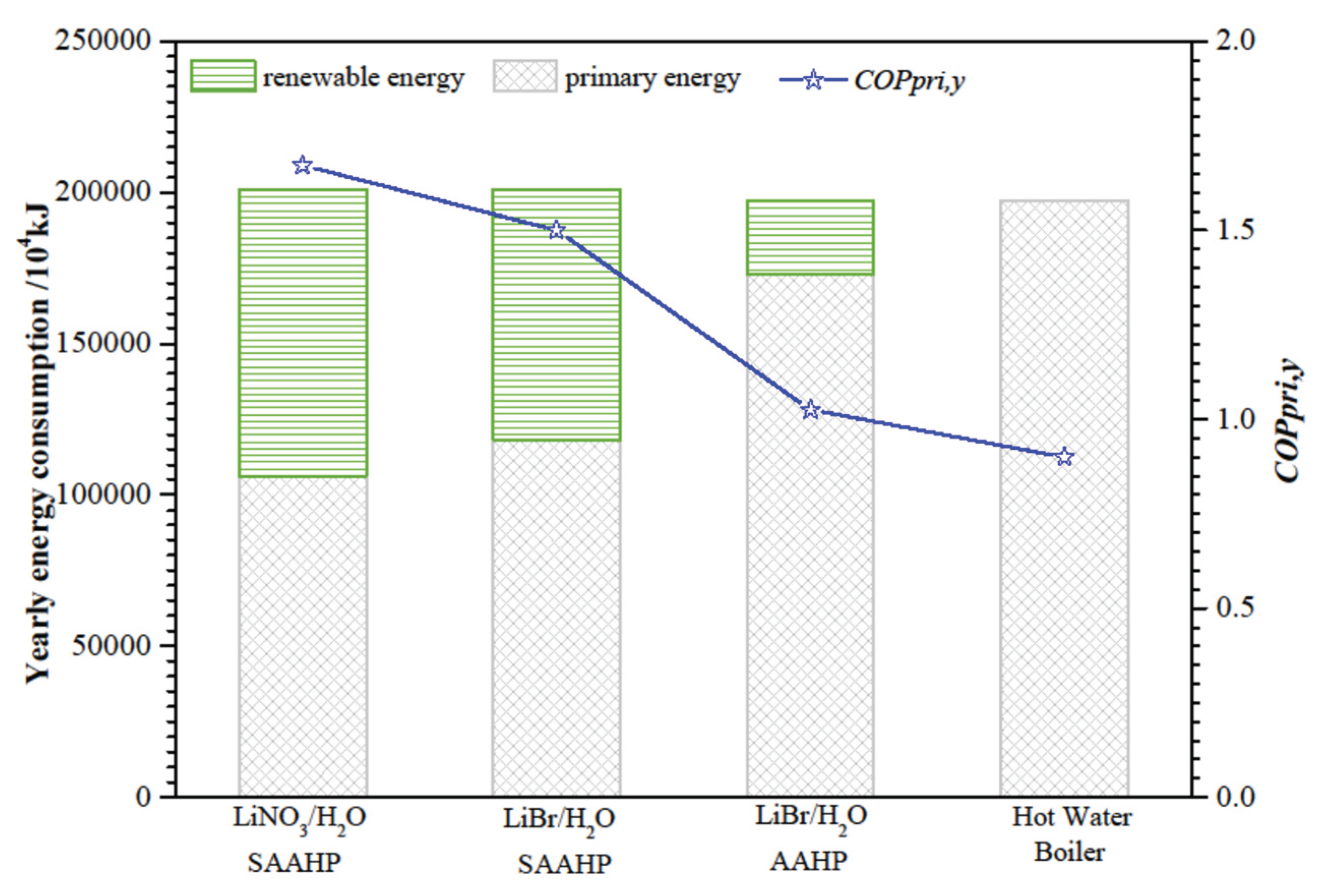
The yearly energy consumption and yearly primary energy
COP (
COPpri,y) of SAAHP and AAHP are shown in
Figure 11.
COPpri,y of SAAHP based on LiNO
3/H
2O and LiBr/H
2O are 1.67 and 1.50, respectively, which are obviously larger than that of AAHP. Relative to a common gas-fired hot water boiler, SAAHP based on LiNO
3/H
2O and LiBr/H
2O save 25631 Nm
3 and 22213 Nm
3 nature gas per year, whereas AAHP just saves 6766 Nm
3 nature gas per year. It is clear that SAAHP has obvious advantage in primary energy saving over the gas-fired hot water boiler and AAHP.
In order to further investigate the primary energy-saving effect of SAAHP and AAHP, the yearly primary energy saving rate (YPESR) is defined as:
where
Eboiler is the yearly primary energy consumption of the gas-fired hot water boiler,
Epri,y is the yearly primary energy consumption of SAAHP or AAHP. Relative to the gas-fired boiler, SAAHP based on LiNO
3/H
2O and LiBr/H
2O achieve yearly primary energy saving rates of 46.2% and 40.0%, respectively, while AAHP just achieves a yearly primary energy saving rate of 12.2%. Obviously compared to AAHP, SAAHP shows a great advantage in building energy saving, and its primary energy-saving effect can be further improved by using LiNO
3/H
2O instead of LiBr/H
2O.
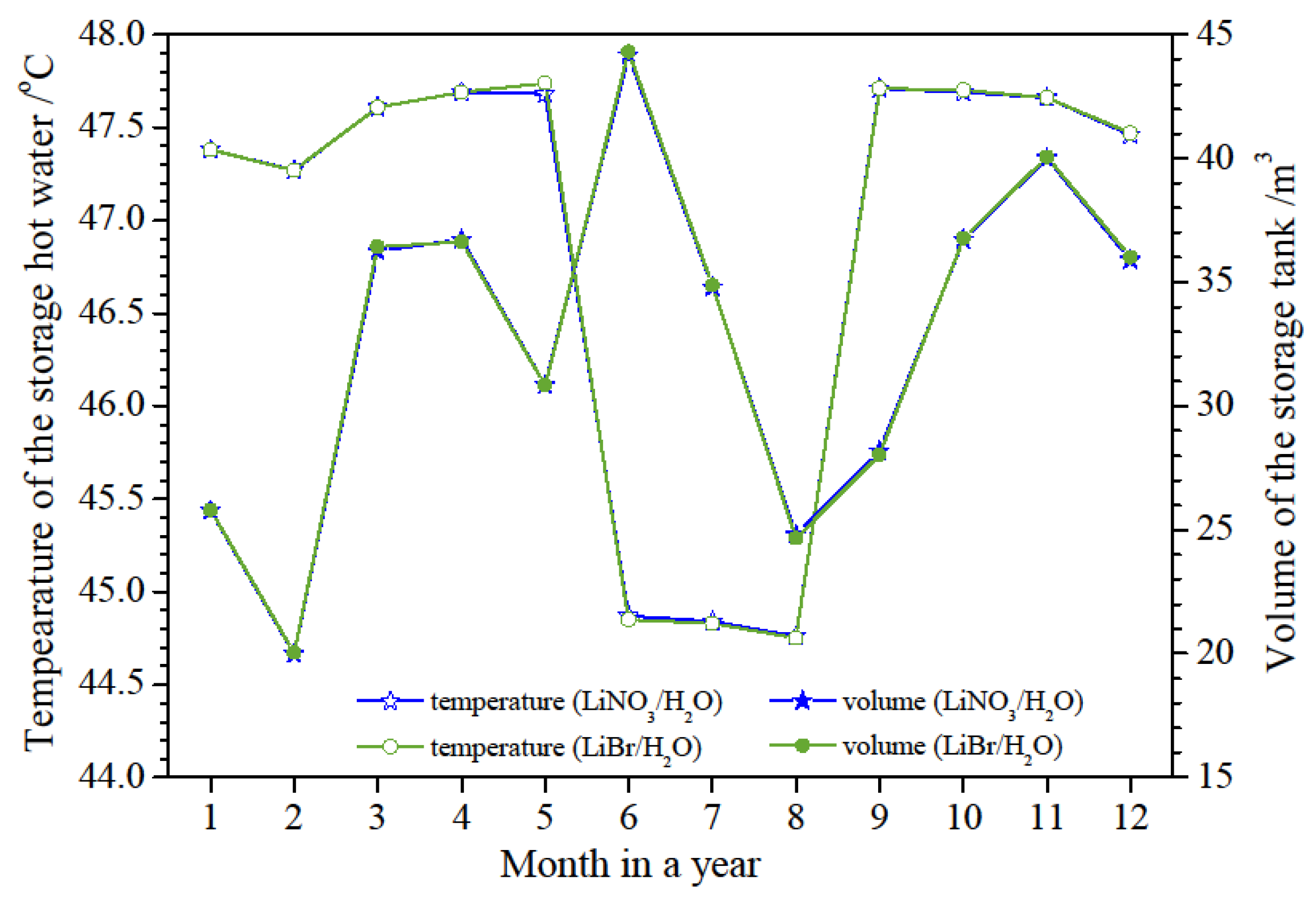
The temperature and volume of the storage hot water before bathing peak time in different months are shown in
Figure 12. It can be seen that the hot water temperatures and the storage volumes based on LiBr/H
2O and LiNO
3/H
2O working fluids have no significant differences throughout a year. The temperature of the storage hot water is above 44.8
oC in hot season and 47.3
oC in other seasons, respectively. The storage volume in the tank varies from 20 m
3 to 44 m
3 throughout a year due to the influences of the building heat demand and the temperature of city water. The storage tank used in this system should be more than 44 m
3 to meet the storage requirement through the all months.