Submitted:
14 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Synthetic Oligonucleotides Samples
2.3. Genomic DNA Samples
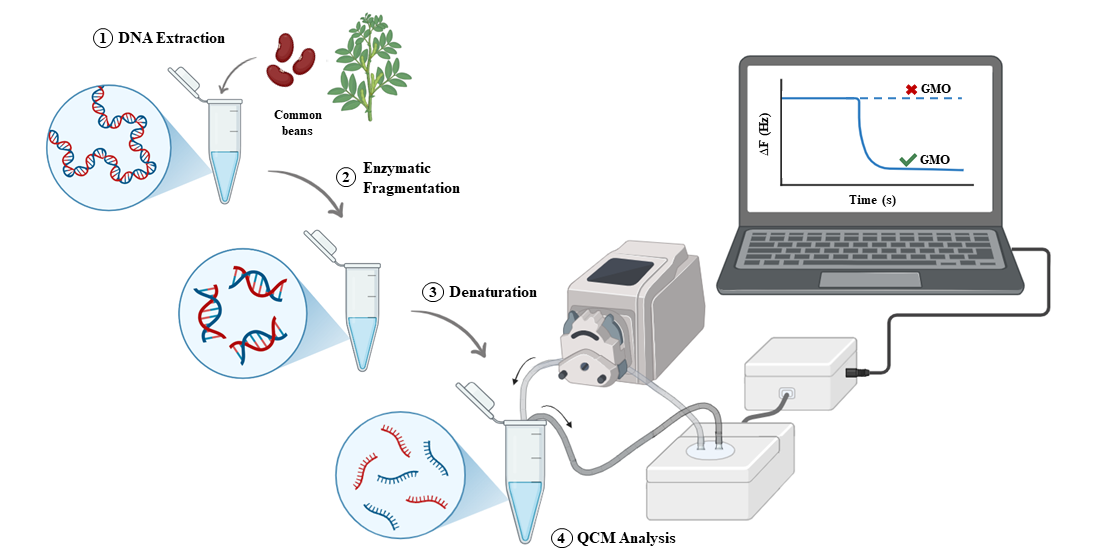
2.3.1. Genomic DNA Extraction
2.3.2. Enzymatic Digestion
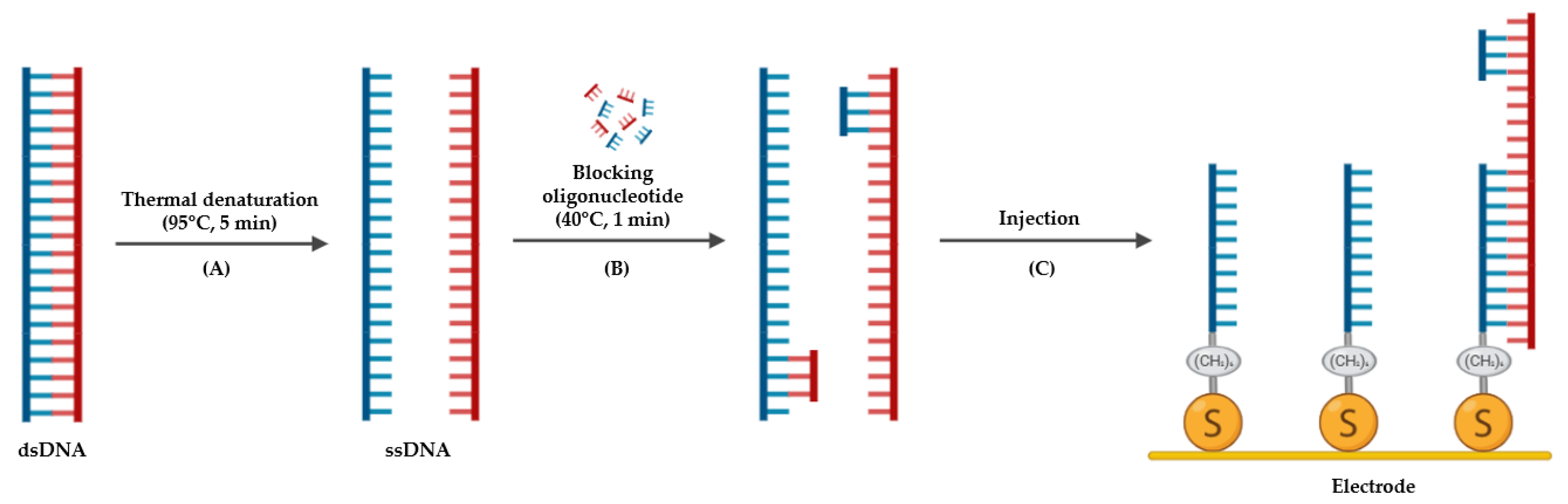
2.3.3. Thermal Denaturation with Blocking Oligonucleotides
2.4. QCM Crystals and the Experimental Set-Up
2.5. Immobilization of the Oligonucleotide Probe on Gold
2.6. Hybridization with Synthetic Oligonucleotides
2.7. Hybridization with Real Samples
3. Results and Discussion
3.1. Electrode Functionalization
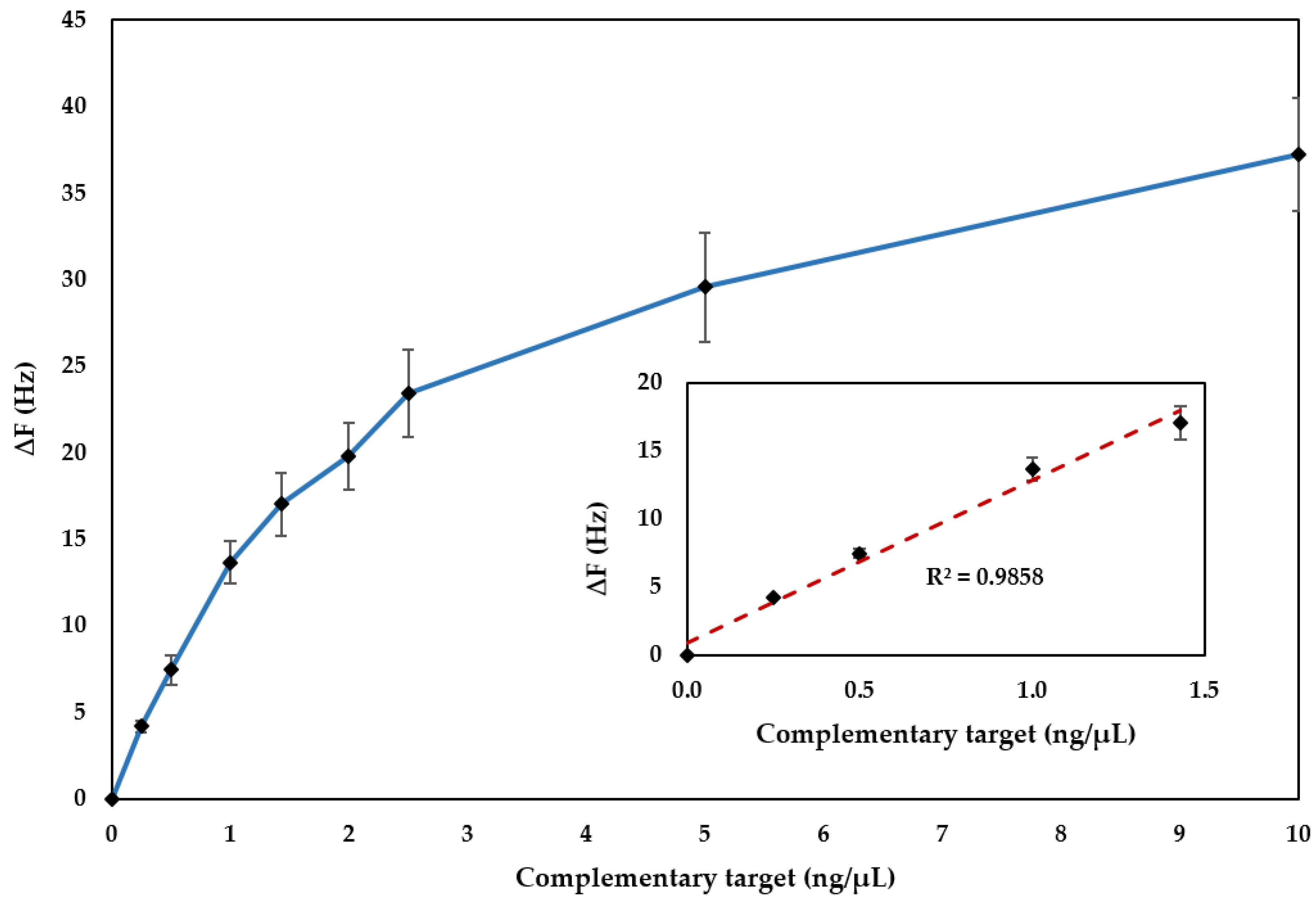
3.2. Analytical Performances of the Biosensor
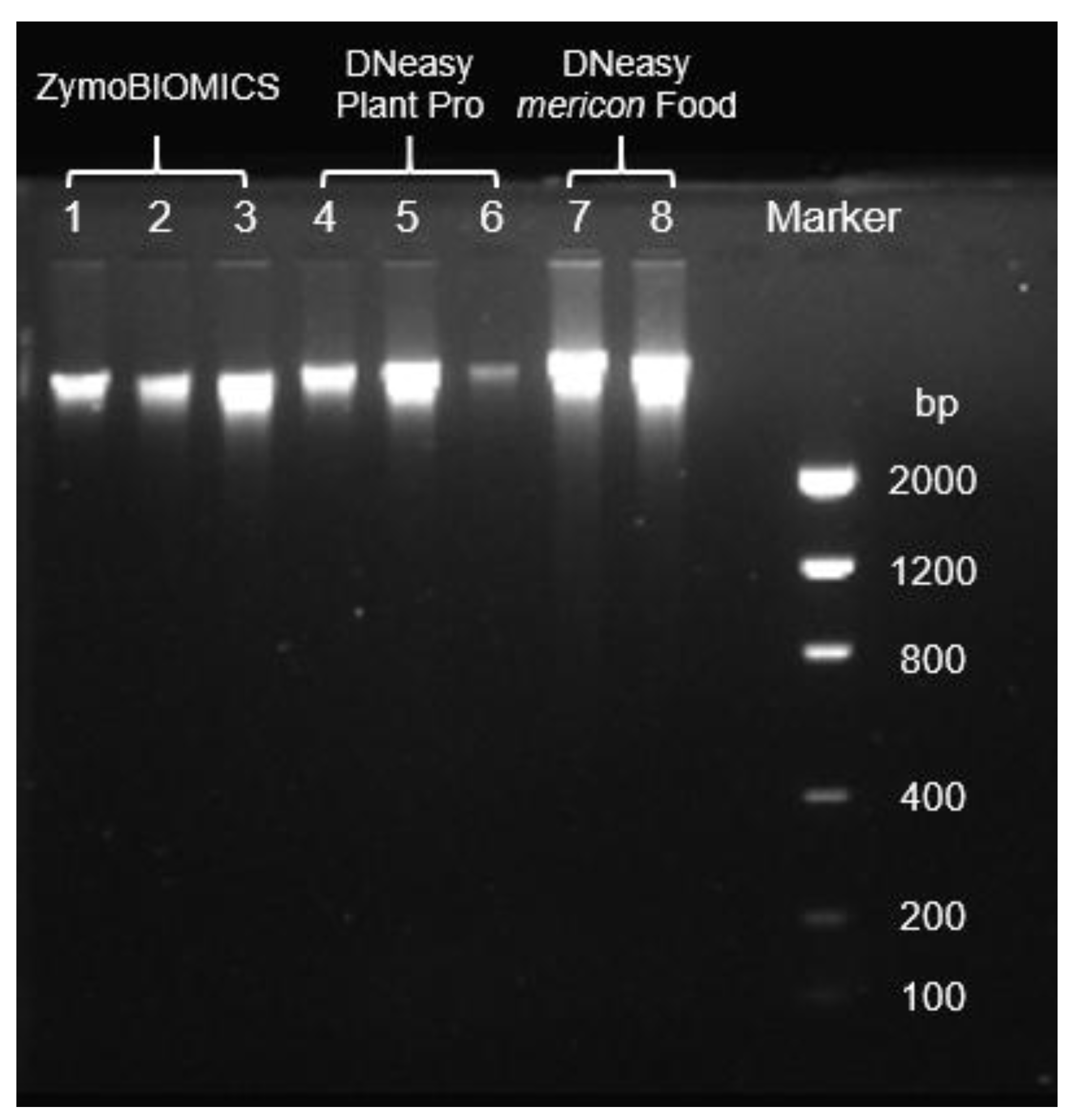
3.3. Genomic DNA Extraction
3.4. Sample Pretreatment: Enzymatic Fragmentation and Thermal Denaturation Combined with Oligonucleotides
3.5. Hybridization with Non-Amplified gDNA Samples
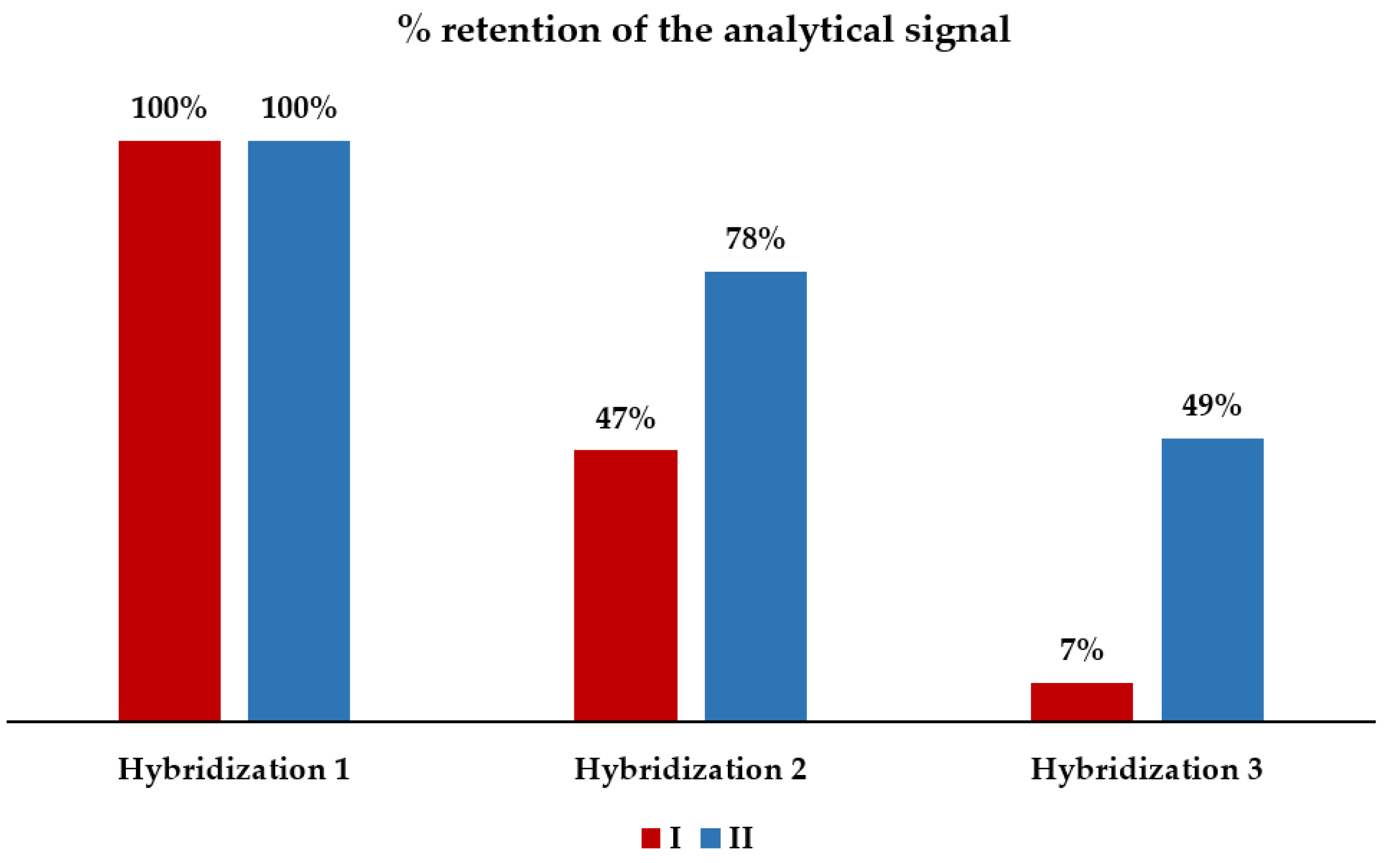
3.6. Surface Regeneration
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| BGMV | bean golden mosaic virus |
| dsRNA | double-stranded RNA |
| siRNA | small interfering RNAs |
| GMO | genetically modified organisms |
| PCR | polymerase chain reaction |
| gDNA | genomic DNA |
| ssDNA | single-stranded DNA |
| AFM | atomic force microscopy |
| XPS | x-ray photoelectron spectroscopy |
References
- Barbosa, F.R.; Souza, T.L.P.O.; Wendland, A.; Costa, A.G.; Quintela, E.D.; Aragão, F.J.L.; Faria, J.C.; Vivian, R. Feijão Resistente Ao Mosaico-Dourado; Embrapa: Goiás, 2021. [Google Scholar]
- Souza, T.L.P.O.; Faria, J.C.; Aragão, F.J.L.; Peloso, M.J.D.; de Faria, L.C.; Aguiar, M.S.; Wendland, A.; Quintela, E.D.; Díaz, J.L.C.; Magaldi, M.C. de S.; et al. BRS FC401 RMD: Cultivar de Feijão Carioca Geneticamente Modificada Com Resistência Ao Mosaico-Dourado; Embrapa, 2016. [Google Scholar]
- Souza, T.L.P.O.; Faria, J.C.; Aragão, F.J.L.; Del Peloso, M.J.; Faria, L.C.; Wendland, A.; Aguiar, M.S.; Quintela, E.D.; Melo, C.L.P.; Hungria, M.; et al. Agronomic Performance and Yield Stability of the RNA Interference-Based Bean Golden Mosaic Virus-Resistant Common Bean. Crop Science 2018, 58, 579–591. [Google Scholar] [CrossRef]
- Teixeira, N.C.; Wendland, A.; Oliveira, M.I. de S.; Brandão, L.T.D.; de Souza, T.L.P.O.; de Faria, J.C.; Del Peloso, M.J.; Cortes, M.V. de C.B. Rapid Identification of RNA-Interference-Based Resistance to Bean Golden Mosaic Virus in Transgenic Common Beans via Loop-Mediated Isothermal Amplification. Crop Science 2020, 60, 3004–3012. [Google Scholar] [CrossRef]
- Arugula, M.A.; Zhang, Y.; Simonian, A.L. Biosensors as 21st Century Technology for Detecting Genetically Modified Organisms in Food and Feed. Anal. Chem. 2014, 86, 119–129. [Google Scholar] [CrossRef]
- Brara, Z.; Madani, K.; Costa, J.; Taouzinet, L.; Djaoudene, O.; Amrane-Abider, M.; Bougherra, H.; Mouhoubi, K.; Brahimi, N.; Bouiche, C.; et al. Current Progress on the Detection of Genetically Modified Organisms in Food: From Transgenic Towards Genome-Edited Crops. Comprehensive Reviews in Food Science and Food Safety 2025, 24, e70243. [Google Scholar] [CrossRef]
- Han, C.; Park, S.; Park, S.; Lee, D.; Jo, H.; Seo, S.; Han, H.; Jeong, S.; Kwon, W. DNA-Functionalized Nanomaterials for Optical Biosensors: Mechanisms, Applications, and Design Perspectives. Sensors and Actuators Reports 2026, 11, 100443. [Google Scholar] [CrossRef]
- Pohanka, M. Overview of Piezoelectric Biosensors, Immunosensors and DNA Sensors and Their Applications. Materials 2018, 11, 448. [Google Scholar] [CrossRef]
- Gunasekaran, B.M.; Srinivasan, S.; Ezhilan, M.; Nesakumar, N. Nucleic Acid-Based Electrochemical Biosensors. Clinica Chimica Acta 2024, 559, 119715. [Google Scholar] [CrossRef]
- Babaei, A.; Pouremamali, A.; Rafiee, N.; Sohrabi, H.; Mokhtarzadeh, A.; de la Guardia, M. Genosensors as an Alternative Diagnostic Sensing Approaches for Specific Detection of Virus Species: A Review of Common Techniques and Outcomes. TrAC Trends in Analytical Chemistry 2022, 155, 116686. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M. Quartz Crystal Microbalance (QCM) Sensing Materials in Biosensors Development. International Journal of Electrochemical Science 2021, 16, 211220. [Google Scholar] [CrossRef]
- Barrias, S.; Fernandes, J.R.; Martins-Lopes, P. Newly Developed QCM-DNA Biosensor for SNP Detection in Small DNA Fragments: A Wine Authenticity Case Study. Food Control 2025, 169, 111036. [Google Scholar] [CrossRef]
- Sauerbrey, G. The Use of Quartz Oscillators for Weighing Thin Layers and for Microweighing. Zeitschrift für Physik 1959, 155, 206–222. [Google Scholar] [CrossRef]
- Karamollaoğlu, İ.; Öktem, H.A.; Mutlu, M. QCM-Based DNA Biosensor for Detection of Genetically Modified Organisms (GMOs). Biochemical Engineering Journal 2009, 44, 142–150. [Google Scholar] [CrossRef]
- Passamano, M.; Pighini, M. QCM DNA-Sensor for GMOs Detection. Sensors and Actuators B: Chemical 2006, 118, 177–181. [Google Scholar] [CrossRef]
- Stobiecka, M.; Cieśla, J.M.; Janowska, B.; Tudek, B.; Radecka, H. Piezoelectric Sensor for Determination of Genetically Modified Soybean Roundup Ready (R) in Samples Not Amplified by PCR. Sensors 2007, 7, 1462–1479. [Google Scholar] [CrossRef]
- Mannelli, I.; Minunni, M.; Tombelli, S.; Mascini, M. Quartz Crystal Microbalance (QCM) Affinity Biosensor for Genetically Modified Organisms (GMOs) Detection. Biosensors and Bioelectronics 2003, 18, 129–140. [Google Scholar] [CrossRef]
- Wang, R.; Minunni, M.; Tombelli, S.; Mascini, M. A New Approach for the Detection of DNA Sequences in Amplified Nucleic Acids by a Surface Plasmon Resonance Biosensor. Biosensors and Bioelectronics 2004, 20, 598–605. [Google Scholar] [CrossRef] [PubMed]
- iGEM Aachen Precise Peristaltic Pump. Available online: https://www.instructables.com/Open-Source-Peristaltic-Pump/ (accessed on 20 October 2025).
- Minunni, M.; Mannelli, I.; Spiriti, M.M.; Tombelli, S.; Mascini, M. Detection of Highly Repeated Sequences in Non-Amplified Genomic DNA by Bulk Acoustic Wave (BAW) Affinity Biosensor. Analytica Chimica Acta 2004, 526, 19–25. [Google Scholar] [CrossRef]
- Oberhaus, F.V.; Frense, D.; Beckmann, D. Immobilization Techniques for Aptamers on Gold Electrodes for the Electrochemical Detection of Proteins: A Review. Biosensors 2020, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.-K.; Yu, L.-K.; Kuo, C.-T.; Weng, J.-H.; Leïchlé, T.; Swami, N.S.; Chen, L.-C.; Wei, P.-K.; Chou, C.-F. Direct in Situ and Real-Time Visualization of Salt-Dependent Thiolated-DNA Immobilization and Biosensing on Gold in Nanofluidic Channels. Sensors and Actuators B: Chemical 2023, 394, 134303. [Google Scholar] [CrossRef]
- Optimisation of peptide nucleic acid (PNA) probe immobilisation by EIS for enhanced bioFET detection of miR-155. Microchemical Journal 2025, 217, 115074. [CrossRef]
- Lazerges, M.; Perrot, H.; Zeghib, N.; Antoine, E.; Compere, C. In Situ QCM DNA-Biosensor Probe Modification. Sensors and Actuators B: Chemical 2006, 120, 329–337. [Google Scholar] [CrossRef]
- Lazerges, M.; Perrot, H.; Rabehagasoa, N.; Compère, C. Thiol- and Biotin-Labeled Probes for Oligonucleotide Quartz Crystal Microbalance Biosensors of Microalga Alexandrium Minutum. Biosensors 2012, 2, 245–254. [Google Scholar] [CrossRef]
- Lino, C.; Barrias, S.; Chaves, R.; Adega, F.; Fernandes, J.R.; Martins-Lopes, P. Development of a QCM-Based Biosensor for the Detection of Non-Small Cell Lung Cancer Biomarkers in Liquid Biopsies. Talanta 2023, 260, 124624. [Google Scholar] [CrossRef]
- Camillone, N. Diffusion-Limited Thiol Adsorption on the Gold(111) Surface. Langmuir 2004, 20, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Tombelli, S.; Minunni, M.; Santucci, A.; Spiriti, M.M.; Mascini, M. A DNA-Based Piezoelectric Biosensor: Strategies for Coupling Nucleic Acids to Piezoelectric Devices. Talanta 2006, 68, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Prins, T.W.; Broothaerts, W.; Burns, M.; Demšar, T.; Edelmann, S.; Papazova, N.; Peterseil, V.; Taverniers, I. Guidance on the Selection and Use of DNA Extraction Methods. Available online: https://publications.jrc.ec.europa.eu/repository/handle/JRC134298 (accessed on 6 December 2025).
- Jia, Y.; Chen, S.; Wang, Q.; Li, J. Recent Progress in Biosensor Regeneration Techniques. Nanoscale 2024, 16, 2834–2846. [Google Scholar] [CrossRef]
- Minunni, M.; Tombelli, S.; Mascini, M. A Biosensor Approach for DNA Sequences Detection in Non-amplified Genomic DNA. Analytical Letters 2007. [Google Scholar] [CrossRef]
| Elements | Sequence |
|---|---|
| Probe | 5′ HS-(CH2)6 - ATGCGGCATCCGAAGCATTGAATGC 3′ |
| Complementary target | 5’ GCATTCAATGCTTCGGATGCCGCAT 3’ |
| Non-complementary 1 | 5’ TTGATGACATCGCACCGCACTATCT 3’ |
| Non-complementary 2 | 5’ AGCGGCACCTCTCCCGAAATAATCA 3’ |
| Blocking oligo 1 | 5’ ATATTCACAA 3’ |
| Blocking oligo 2 | 5’ TGCATGGCTT 3’ |
| N | gDNA Extraction Kit | ng/µL | A260/280 | A260/A230 |
|---|---|---|---|---|
| I | ZymoBIOMICS DNA Miniprep | 59 ± 9 | 1.95 ± 0.00 | 1.49 ± 0.07 |
| II | DNeasy Plant Pro | 52 ± 20 | 1.86 ± 0.03 | 0.13 ± 0.06 |
| III | DNeazy mericon Food | 127 ± 16 | 1.85 ± 0.04 | 1.28 ± 0.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




