Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Background and Objectives: Multiple sclerosis (MS) is a chronic autoimmune neuroinflammatory condition associated with significant disability. Given the increasing number of patients with MS, the growing number of available disease-modifying therapies (DMTs), and the substantial economic burden associated with MS, it is critically important to determine which treatment options are the most cost-effective. The economic burden of MS is considerable, and high-cost DMTs, whose prices continue to rise, represent the primary driver of health expenditures related to MS. The primary aim of this narrative review is to provide a brief review of the economic issues related to MS DMTs, including pricing trends, economic burden, and their impact on patient care, and to propose potential policy solutions. Materials and Methods: The aim of the study was to compare the cost effectiveness of The annual costs of DMTs from 1993 to 2023 in USA. An examination of DMT pricing trends suggests that pricing has been influenced in part by within-class competition and the availability of generic DMT options. Results: Over the past decade, the prices of DMTs have increased by more than 50%. Currently, the annual cost of many DMTs used in the treatment of MS has exceeded $100,000, and their economic value is widely debated. The high cost of DMTs and difficulties in timely access to medications can lead to psychological stress among many patients. Studies on cost-effectiveness indicate that the clinical benefits provided by DMTs do not fully justify their high costs, which further exacerbates issues related to economic accessibility. Conclusions: Collaborative neuropsychiatric care models, in which neurologists and mental health professionals work in coordination, may improve symptom recognition, optimize adherence to DMTs, and enhance overall functioning. Given that treatment non-adherence and reduced quality of life negatively affect cost-effectiveness outcomes, early psychiatric intervention may indirectly improve the economic value profile of high-cost DMTs.
Keywords:
1. Introduction
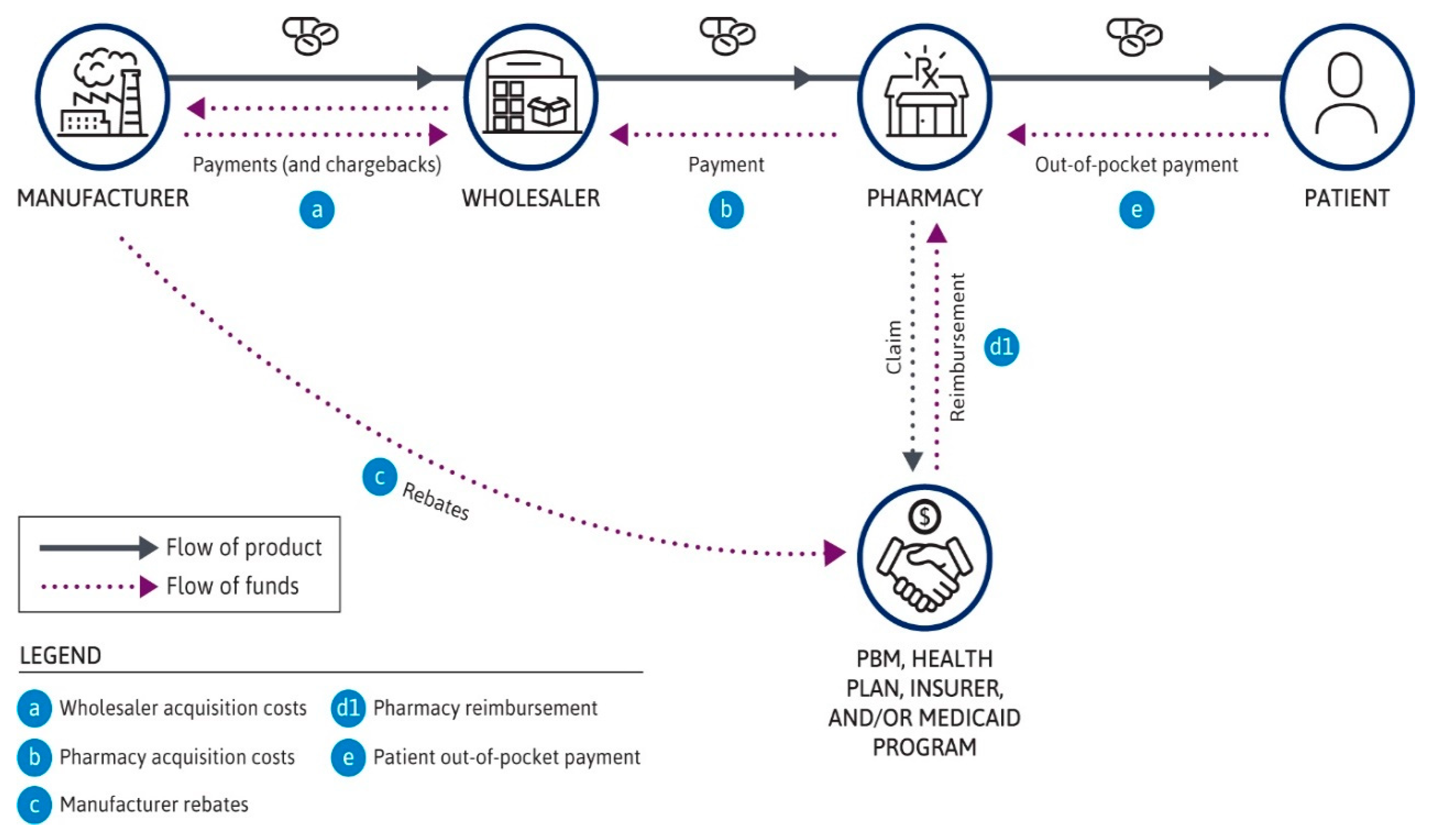
1.1. Pricing Strategy in the US Healthcare System
2. Materials and Methods
2.1. Pricing Policies for Medications Used in DMT Treatment
| Calendar Year 2012 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Brand Name | Generic Name | Total Spending |
Total Claims |
Total Patients |
Average Spending Per Dosage Unit (Weighted) | Average Spending Per Claim | Average Spending Per Beneficiary |
|
| Aubagio | Teriflunomide | $1.241.236,92 | 312 | 233 | $127,02 | $3.978,32 | $5.327,20 | |
| Avonex | Interferon Beta-1a | $390.201.432,00 | 98.657 | 11.901 | $1.108,80 | $3.955,13 | $32.787,28 | |
| Avonex | Interferon Beta-1a/Albumin | $36.195.551,33 | 9.256 | 1.326 | $921,94 | $3.910,50 | $27.296,80 | |
| Betaseron | Interferon Beta-1b | $174.898.129,99 | 44.045 | 5.432 | $268,65 | $3.970,90 | $32.197,74 | |
| Copaxone | Glatiramer Acetate | $911.468.903,08 | 208.429 | 25.119 | $4.150,19 | $4.373,04 | $36.286,03 | |
| Extavia | Interferon Beta-1b | $16.024.880,90 | 5.278 | 728 | $216,40 | $3.036,17 | $22.012,20 | |
| Gilenya | Fingolimod HCl | $144.758.245,70 | 29.217 | 4.019 | $155,03 | $4.954,59 | $36.018,47 | |
| Glatopa | Glatiramer Acetate | |||||||
| Plegridy | PEGinterferon Beta-1a | |||||||
| Rebif | Interferon Beta-1a/Albumin | $246.894.205,97 | 66.968 | 7.603 | $608,46 | $3.686,75 | $32.473,26 | |
| Rebif Rebidose | Interferon Beta-1a/Albumin | |||||||
| Tecfidera | Dimethyl Fumarate | |||||||
| Tysabri | Natalizumab | |||||||
| Total | $1.921.682.585,89 | 462.162 | 56.361 | |||||
| Calendar Year 2020 | ||||||||
| Brand Name | Generic Name |
Total Spending |
Total Claims |
Total Patients |
Average Spending Per Dosage Unit (Weighted) |
Average Spending Per Claim | Average Spending Per Beneficiary | |
| Aubagio | Teriflunomide | $778.201.329,31 | 78.610 | 10.386 | $265,08 | $9.899,52 | $74.927,92 | |
| Avonex | Interferon Beta-1a | $250.136.077,86 | 29.622 | 3.505 | $7.234,07 | $8.444,27 | $71.365,50 | |
| Avonex Pen | Interferon Beta-1a | $305.121.803,72 | 36.455 | 4.262 | $7.233,97 | $8.369,82 | $71.591,23 | |
| Betaseron | Interferon Beta-1b | $163.581.520,93 | 18.758 | 2.113 | $584,81 | $8.720,63 | $77.416,72 | |
| Copaxone | Glatiramer Acetate | $713.588.637,07 | 107.923 | 12.092 | $439,83 | $6.612,02 | $59.013,28 | |
| Extavia | Interferon Beta-1b | $12.471.387,29 | 2.080 | 300 | $382,92 | $5.995,86 | $41.571,29 | |
| Gilenya | Fingolimod HCl | $449.518.264,33 | 46.257 | 5.128 | $297,08 | $9.717,84 | $87.659,57 | |
| Glatiramer Acetate | Glatiramer Acetate | $212.422.737,82 | 54.545 | 7.519 | $287,79 | $3.894,45 | $28.251,46 | |
| Glatopa | Glatiramer Acetate | $88.246.254,82 | 25.645 | 3.825 | $194,12 | $3.441,07 | $23.070,92 | |
| Lemtrada | Alemtuzumab | $1.450.990,94 | 19 | 17 | $20.494,22 | $76.367,94 | $85.352,41 | |
| Plegridy Pen | PEGinterferon Beta-1a | $56.678.580,61 | 6.639 | 827 | $7.163,65 | $8.537,22 | $68.535,16 | |
| Plegridy* | PEGinterferon Beta-1a | $9.407.504,88 | 1.094 | 154 | $7.162,49 | $8.599,18 | $61.087,69 | |
| Rebif | Interferon Beta-1a/Albumin | $232.708.460,75 | 25.470 | 2.795 | $1.415,55 | $9.136,57 | $83.258,84 | |
| Rebif Rebidose | Interferon Beta-1a/Albumin | $153.166.653,12 | 16.341 | 1.950 | $1.423,93 | $9.373,15 | $78.547,00 | |
| Tecfidera | Dimethyl Fumarate | $1.054.984.601,40 | 108.629 | 14.663 | $142,02 | $9.711,81 | $71.948,76 | |
| Tysabri | Natalizumab | $41.553.342,14 | 5.732 | 749 | $475,77 | $7.249,36 | $55.478,43 | |
| Total | $4.523.238.146,99 | 563.819 | 70.285 | |||||
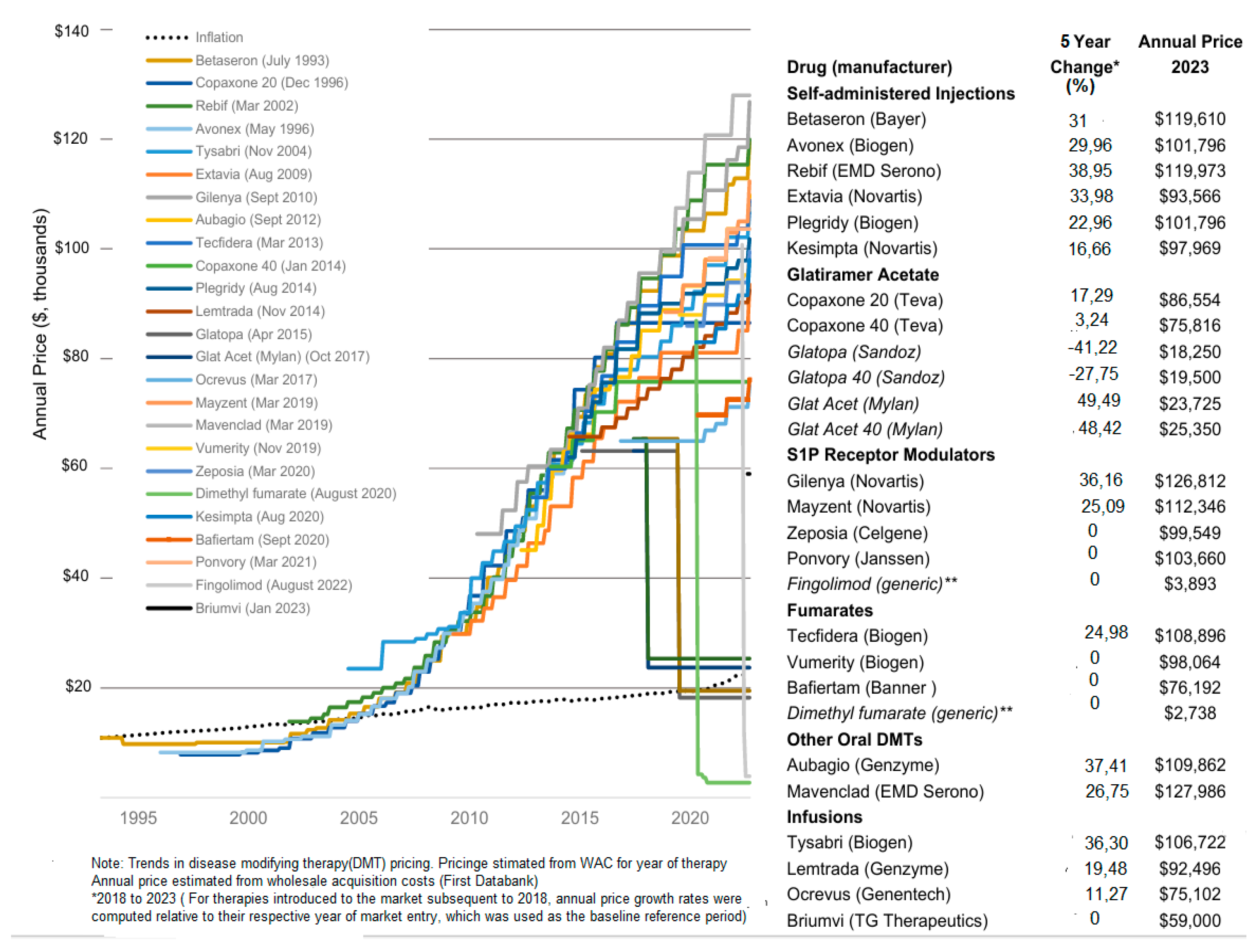
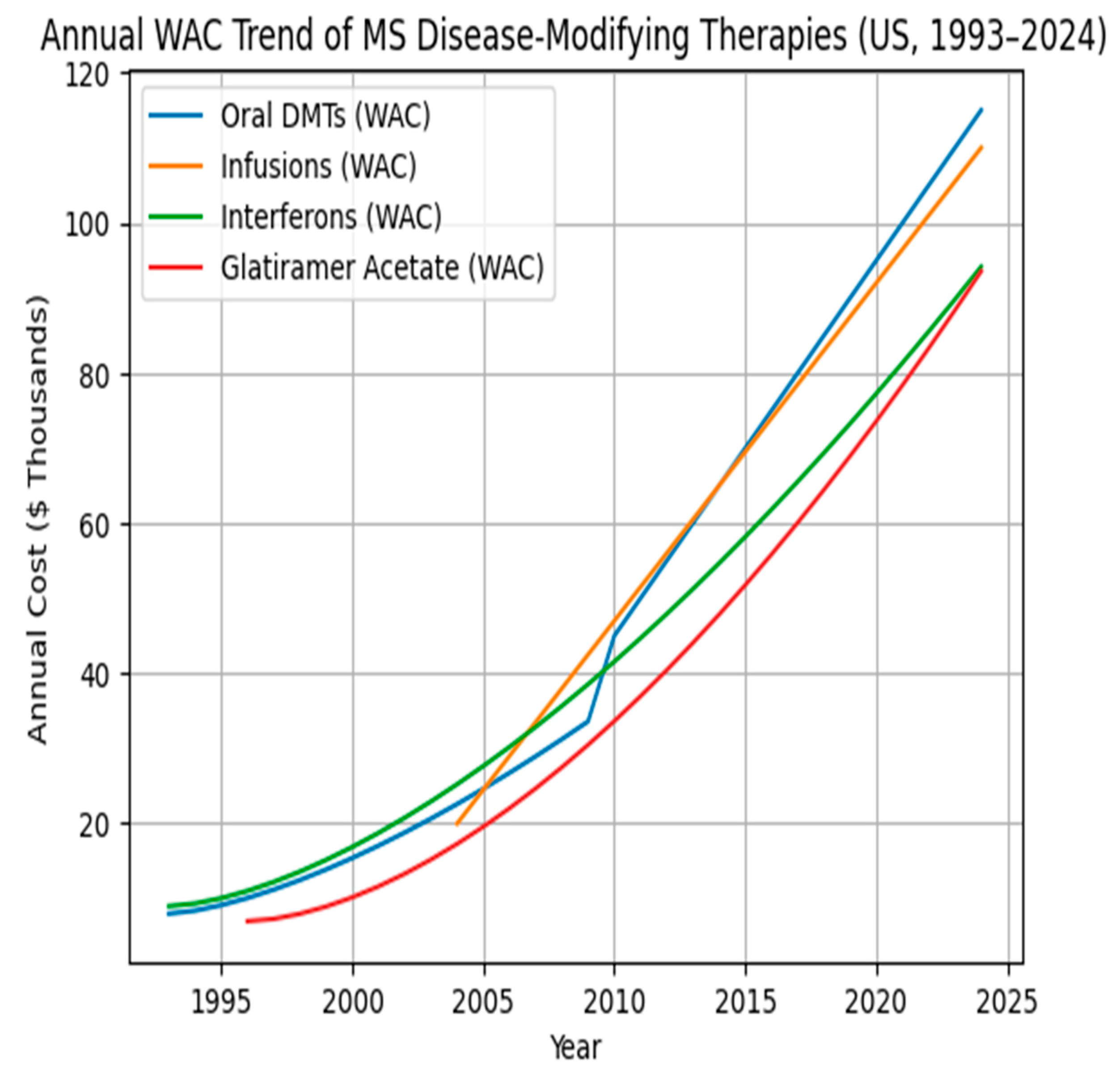
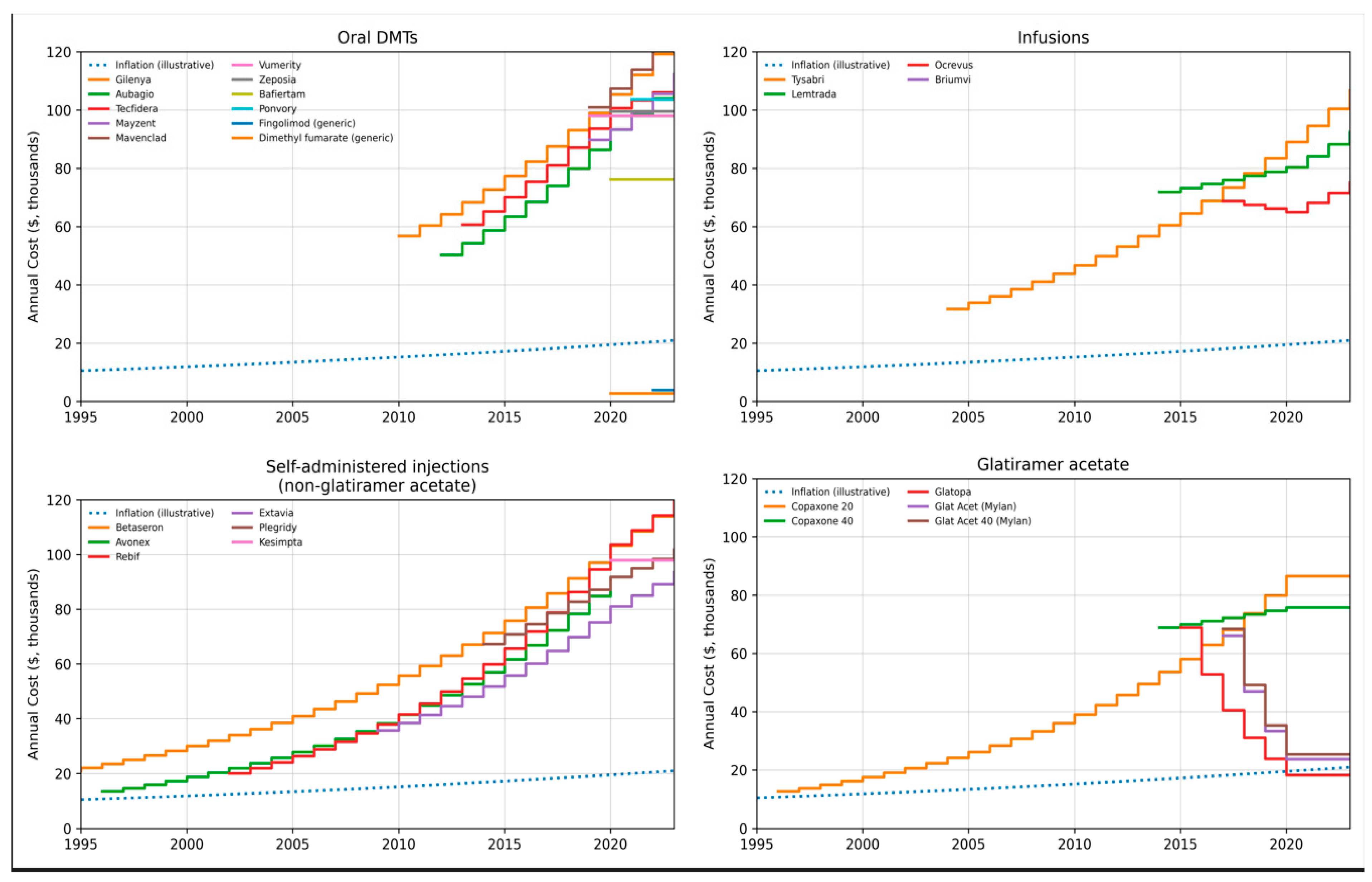
| Annual ıncreases (%) | 2023 Estimate Annual Cost | 2023 cost net standard medicaid rebate | ||||||
| DMT | Launch Year | Annual WAC at Introduction $ |
1998-2001 | 2002-2009 | 2010-2016 | 2018-2023 | ||
| IFN-β1b; Betaseron™ (1993) | 1993 | 10.988 | 0 | 16 | 15,2 | 31 | 119.610 | 91.980 |
| IFN-β1a IM; Avonex™ (1996) | 1996 | 8.301 | 2,5 | 18 | 16,1 | 29,96 | 101.796 | 78.281 |
| Glatiramer acetate;Copaxone™* (1996) | 1996 | 7.856 | 2,6 | 19 | 14,9 | 17,29 | 86.554 | 66.560 |
| IFN-β1a Subcutaneous İnjection; Rebif™ (2002) | 2002 | 13.965 | NA | NA | 12,6 | 38,95 | 119.973 | 92.259 |
| Natalizumab; Tysabri™ (2004) | 2004 | 23.595 | NA | NA | 11,30 | 36,3 | 106.722 | 82.069 |
| Interferon-β1b; Extavia™(2009) | 2009 | 29.965 | NA | NA | 14,4 | 33,98 | 93.566 | 71.952 |
| Fingolimod; Gilenya™ (2010) | 2010 | 48.203 | NA | NA | 9 | 36,16 | 126.812 | 97.518 |
| Teriflunomide; Aubagio™(2012) | 2012 | 45.226 | NA | NA | 16,6 | 37,41 | 109.862 | 84.484 |
| Dimethyl fumerate; Tecfidera™ (2013) | 2013 | 54.675 | NA | NA | 13,4 | 24,98 | 108.896 | 83.741 |
| Pegylated IFN-β1a; Plegridy™ (2014) | 2014 | 62.134 | NA | NA | 10,92 | 22,96 | 101.796 | 78.281 |
| Glatiramer acetate; Glatopa™(2015) | 2014 | 63.203 | NA | NA | 0 | -41,22 | 18.250 | 14.034 |
| Alemtuzumab; Lemtrada™ (2014) | 2015 | 65.961 | NA | NA | 2,5 | 19,48 | 92.496 | 71.129 |
| OcrevusTM (2017) | 2017 | 65.121 | NA | NA | NA | 11,27 | 75.102 | 57.753 |
| Glatiramer acetate; Generic™ (2017) | 2017 | 63.198 | NA | NA | NA | -166 | 23.725 | 18.245 |
| Siponimod (Mayzent) (2019) | 2019 | 89.812 | NA | NA | NA | 25,09 | 100.000 | 76.900 |
| Cladribine (Mavenclad) (2019) | 2019 | 100.975 | NA | NA | NA | 26,75 | 112.346 | 86.394 |
|
Notes: Annual price estimated from wholesale acquisition costs (First Databank) For therapies introduced to the market subsequent to 2018, annual price growth rates were computed relative to their respective year of market entry, which was used as the baseline reference period. | ||||||||
3. Results
3.1. The Importance of Drug Prices in Patient Care
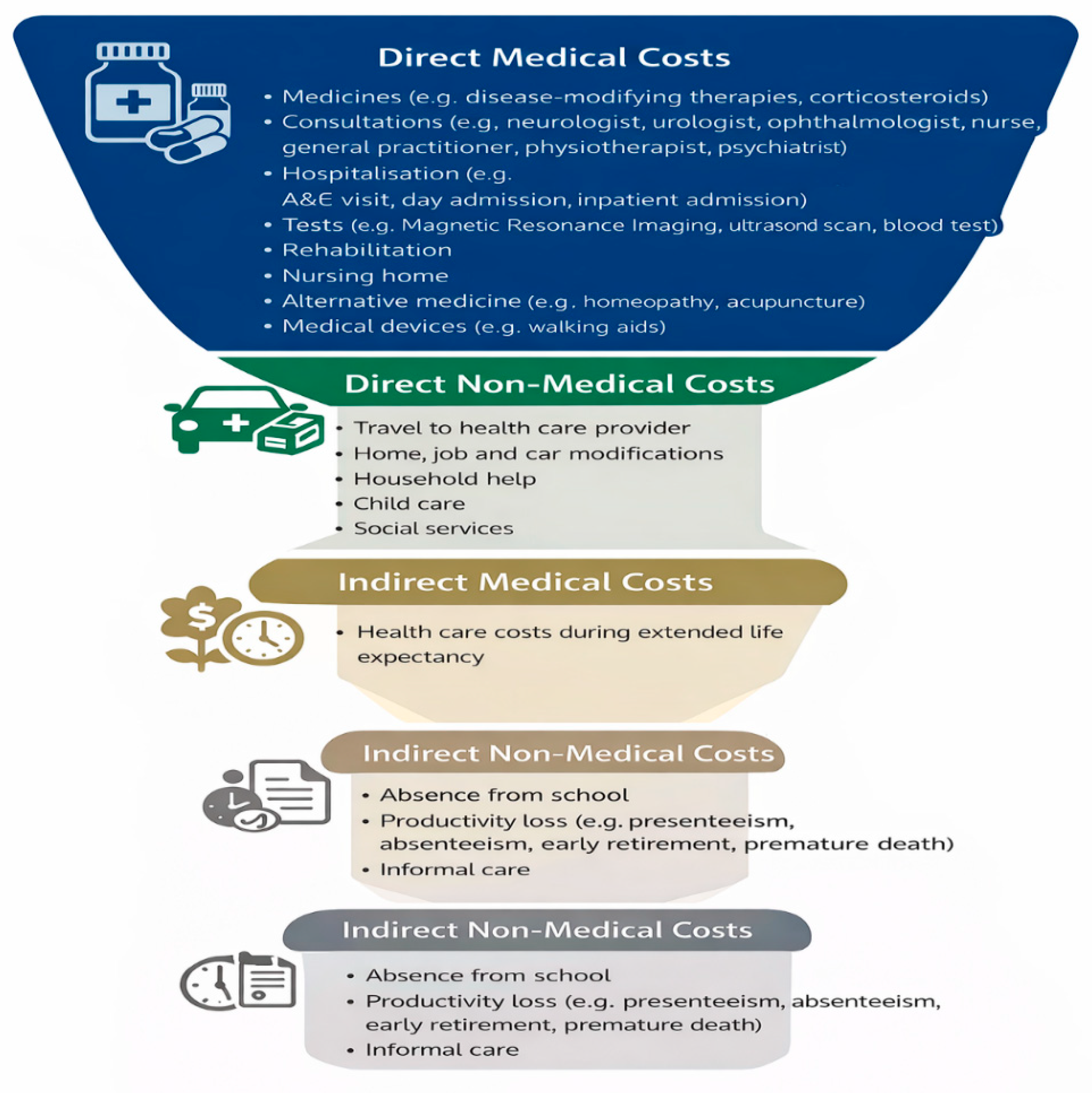
3.2. The Social and Economic Burden of DMTs
4. Discussion and Possible Solutions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MS | Multiple sclerosis |
| DMTs | disease-modifying therapies |
| CIS | Clinically Isolated Syndrome |
| RRMS | Relapsing-Remitting Multiple sclerosis |
| PPMS | Primary Progressive Multiple sclerosis |
| SSDI | Social Security Disability Insurance |
| DMF | dimethyl fumarate |
| MPPP | Medicare Prescription Payment Plan |
| IRA | Inflation Reduction Act |
| PBMs | Pharmacy Benefit Managers |
| VA | Veterans Affairs |
| AMP | Average Manufacturer Price |
| WAC | Wholesale Acquisition Cost |
| QALY | The Quality-Adjusted Life Year |
| ICER | the Incremental Cost-Effectiveness Ratio |
| OOP | out-of-pocket |
References
- Korsukewitz, C.; Wiendl, H. Emerging trends and challenges in multiple sclerosis in Europe: rethinking classification and addressing COVID-19 impact. Lancet Reg Health Eur. 2024, 44, 101017. [Google Scholar] [CrossRef]
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.A.; et al. Rising prevalence of multiple sclerosis worldwide: insights from the atlas of MS, third edition. Mult Scler J. 2020, 26(14), 1816–21. [Google Scholar] [CrossRef]
- Bebo, B.; et al. The economic burden of multiple sclerosis in the united States. Neurology 2022, 98(18), e1810–e7. [Google Scholar] [CrossRef]
- Available online: https://www.nationalmssociety.org/managing-ms/treating-ms/disease-modifying-therapies/cost-of-ms.
- Hartung, D.M. Economics of multiple sclerosis disease-modifying therapies in the USA. Curr Neurol Neurosci Rep. 2021, 21(7), 28. [Google Scholar] [CrossRef] [PubMed]
- Hartung, D.M.; Bourdette, D. Addressing the rising prices of disease-modifying therapies for multiple sclerosis. JAMA Neurol. 2019. Epub ahead of print 26 August 2019. [Google Scholar] [CrossRef] [PubMed]
- Whetten-Goldstein, K.; Sloan, F.A.; Goldstein, L.B.; Kulas, E.D. A comprehensive assessment of the cost of multiple sclerosis in the United States. Mult Scler. 1998, 4(5), 419–425. [Google Scholar] [CrossRef]
- Simoens, S. Societal economic burden of multiple sclerosis and cost-efectiveness of disease-modifying therapies. Front. Neurol. 2022, 13, 1015256. [Google Scholar] [CrossRef]
- Kister, I.; Corboy, J.R. Reducing costs while enhancing quality of care in MS. Neurology 2016, 87, 1617–1622. [Google Scholar] [CrossRef]
- Zimmermann, M.; Brouwer, E.; Tice, J.A.; et al. Disease-modifying therapies for relapsingremitting and primary progressive multiple sclerosis: a cost-utility analysis. CNS Drugs 2018, 32(12), 1145–1157. [Google Scholar] [CrossRef]
- McKenna, A.; Lin, G.A.; Whittington, M.D.; et al. Oral and monoclonal antibody treatments for relapsing forms of multiple sclerosis: effectiveness and value. J Manag Care Spec Pharm. 2023, 29(7), 857–861. [Google Scholar] [CrossRef] [PubMed]
- Lezzoni, L.I.; Ngo, L. Health; Disability, and life insurance experiences of working-age persons with multiple sclerosis. Mult Scler. 2007, 13, 534–546. [Google Scholar]
- Soini, E.; Joutseno, J.; Sumelahti, M.-L. Cost-utility of first-line disease-modifying treatments for relapsing–remitting multiple sclerosis. Clin Therapeut 2017, 39(3), 537–57.e10. [Google Scholar] [CrossRef]
- Huygens, S.; Versteegh, M. Modeling the cost-utility of treatment sequences for multiple sclerosis. Value Health 2021, 24(11), 1612–9. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Baranzini, S.E.; Geurts, J.; Hemmer, B.; Ciccarelli, O. Multiple sclerosis. Lancet (London, England) 2018, 391(10130), 1622–36. [Google Scholar] [CrossRef]
- Solaro, C.; Gamberini, G.; Masuccio, F.G. Depression in multiple sclerosis: epidemiology, aetiology, diagnosis and treatment. CNS Drugs 2018, 32(2), 117–33. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, J.I.; Birnbaum, H.G.; Samuels, S.; Davis, M.; Phillips, A.L.; Meletiche, D. The cost of disability and medically related absenteeism among employees with multiple sclerosis in the US. Pharmacoeconomics 2009, 27(8), 681–691. [Google Scholar] [CrossRef]
- Adelman, G.; Rane, S.G.; Villa, K.F. The cost burden of multiple sclerosis in the United States: a systematic review of the literature. J Med Econ 2013, 16(5), 639–647. [Google Scholar] [CrossRef]
- Hartung, D.H.; Gabriel, N.; Gellad, W.F.; Cameron, T.; Feeder, N.M.; Herhandez, I. Changes in List and Net Prices for Multiple Sclerosis Disease-Modifying Therapy, 2013 to 2021. Neurology Journal 2026, 16(2), e200597. [Google Scholar] [CrossRef]
- Daniel, H. Stemming the escalating cost of prescription drugs: a position paper of the american college of physicians. Ann Intern Med 2016. [Google Scholar] [CrossRef]
- Tag, T.; Rubinstein, E. AMCP Guide to Pharmaceutical Payment Methods, 2013 Update (Version 3.0). Academy of Managed Care Pharmacy 2013, 1083–4087. [Google Scholar]
- Hung, A.; Dickson, S. A primer on prescription drug pricing benchmarks in the United States. J Manag Care Spec Pharm 2025, 31(12), 1326–1335. [Google Scholar] [CrossRef]
- Dabora, M.C.; Turaga, N.; Schulman, K.A. Financing and distribution of pharmaceuticals in the United States. JAMA 2017, 381, 21–22. [Google Scholar] [CrossRef]
- Gusovsky, A.V.; Lin, C.C.; Kerber, K.; Reynolds, E.L.; Callaghan, B.C.; Burke, J.F. Costs are still on the rise for commonly prescribed branded neurologic medications. Neurology. 2024, 103(10), e210029. [Google Scholar] [CrossRef] [PubMed]
- Hartung, D.M.; Johnston, K.A.; Geddes, J.; Bourdette, D.N. Effect of generic glatiramer acetate on spending and use of drugs for multiple sclerosis. Neurology 2020, 94(13), e1407–e1414. [Google Scholar] [CrossRef]
- Rome, B.N.; Tessema, F.A.; Kesselheim, A.S. US spending associated with transition from daily to 3-times-weekly glatiramer acetate. JAMA Internal Medicine. 2020, 180(9), 1165–1172. [Google Scholar] [CrossRef]
- Hartung, D.M. Health economics of disease-modifying therapy for multiple sclerosis in the United States. Sage Journals 2021. [Google Scholar] [CrossRef]
- Hartung, D.M. Economics and Cost-Effectiveness of Multiple Sclerosis Therapies in the USA. Neurotherapeutics 2017, 14, 1018–1026. [Google Scholar] [CrossRef]
- American Academy of Neurology. AAN position statement: prescription drug prices. Available online: https://www.aan.com/policy-and-guidelines/policy/priority-issues/ (accessed on 4 March 2026).
- Banahan, B.F.; Datar, M.; Mendonca, C.M.; Bentley, J.P.; Phillips, A.L.; Stewart, M. Effect of medicare part d coverage on adherence with disease modifying drug therapy for multiple sclerosis. Annual Meeting of the Academy of Managed Care Pharmacy, San Francisco, 18– 20 April 2012. [Google Scholar]
- Reynolds, M.W.; Stephen, R.; Seaman, C.; Rajagopalan, K. Healthcare resource utilization following switch or discontinuation in multiple sclerosis patients on disease modifying drugs. J Med Econ 2010, 13(1), 90–98. [Google Scholar] [CrossRef]
- Ivanova, J.I.; Bergman, R.E.; Birnbaum, H.G.; Phillips, A.L.; Stewart, M.; Meletiche, D.M. Impact of medication adherence to diseasemodifying drugs on severe relapse, and direct and indirect costs among employees with multiple sclerosis in the US. J Med Econ 2012, 15(3), 601–609. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, S.C.; Faris, R.J.; Chang, C.F.; Chan, A.; Tankersley, M.A. Impact of adherence to interferons in the treatment of multiple sclerosis: a non-experimental, retrospective, cohort study. Clin Drug Investig 2010, 30(2), 89–100. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.S.; Porter, M.E. How to Solve the Cost Crisis in Health Care. Harv Bus Rev 2011, 89(9), 46–52. [Google Scholar] [PubMed]
- Egdom, L.S.E.; Lagendijk, M.; Kemp, M.H.; Dam, J.H.; Mureau, M.A.M.; Hazelzet, J.A.; Koppert, L.B. Implementation of Value Based Breast Cancer Care. European Journal of Surgical Oncology 2019, 45(7), 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Weiler; Ross, D. Transforming Health Care Through Value-Based Reimbursement; New England Employee Benefits Council, USA, 2019. [Google Scholar]
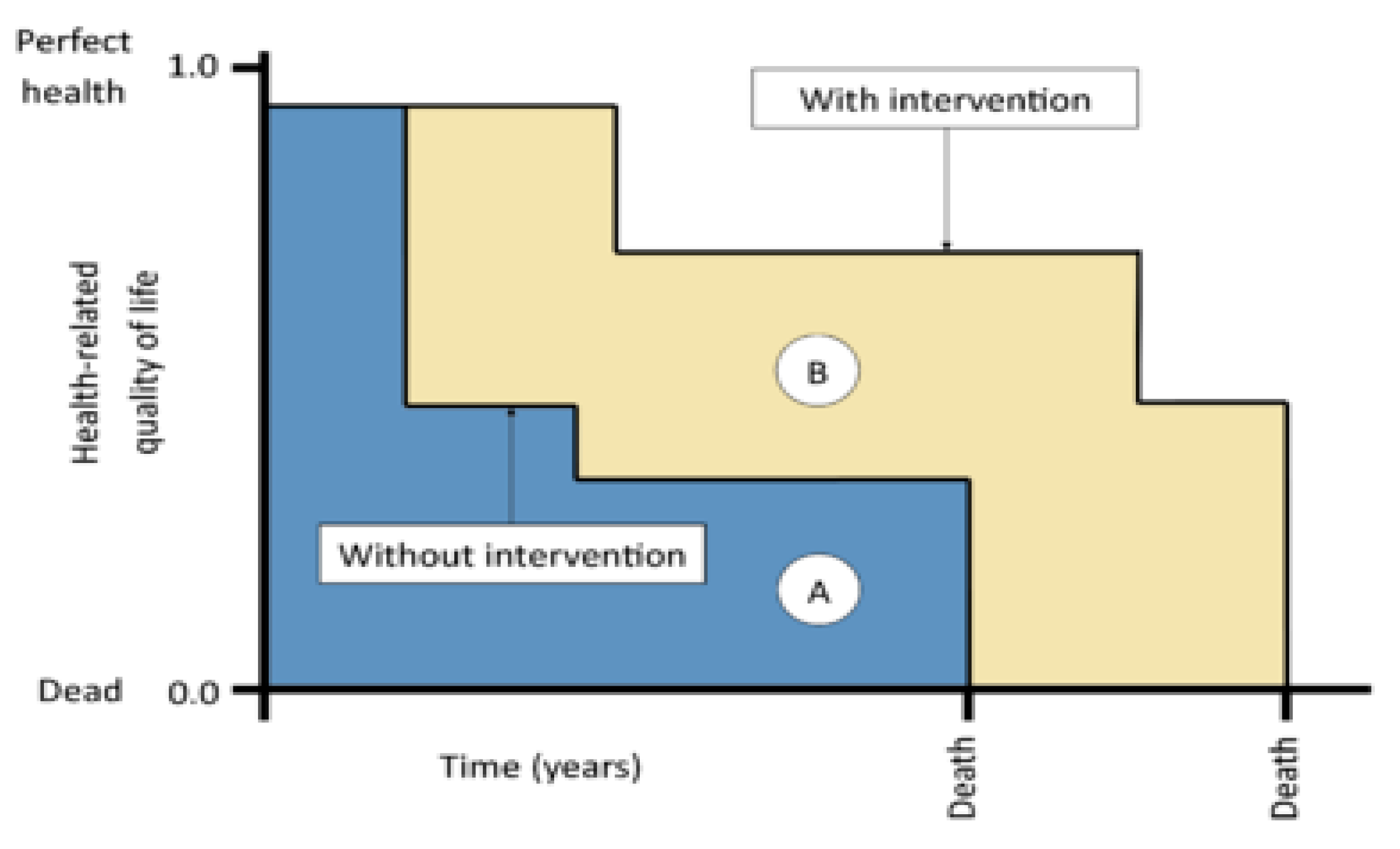
- Briggs, A.H.; Goldstein, D.A.; Kirwin, E.; Meacock, R.; Pandya, A.; Vanness, D.J.; et al. Estimating (quality-adjusted) life-year losses associated with deaths: With application to COVID-19. Health Economics 2020. online version ahead of print. [Google Scholar] [CrossRef]
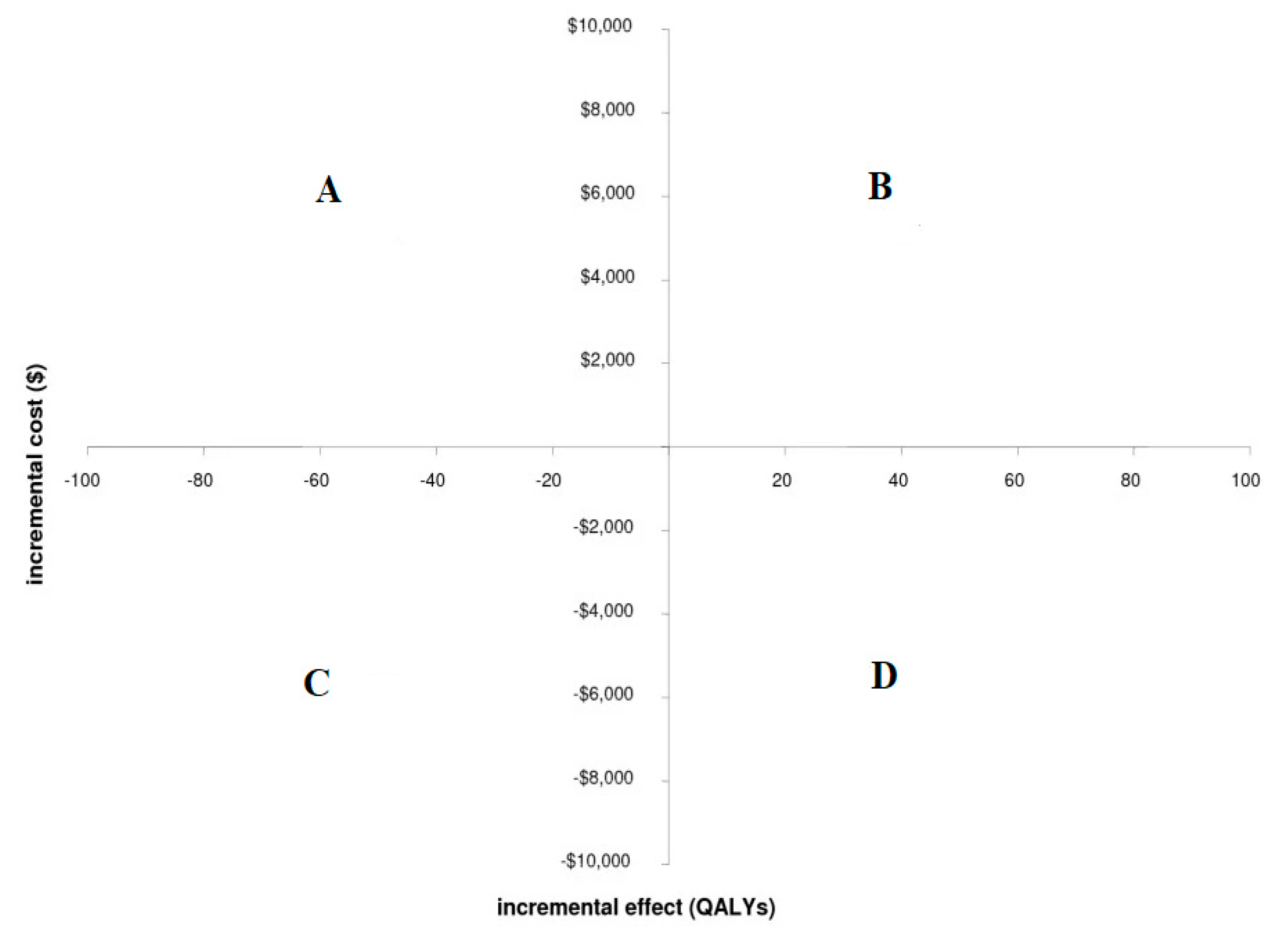
- Black, W. Maliyet-etkinlik düzlemi: Maliyet-etkinliğin grafiksel gösterimi. Tıbbi Karar Verme. 1990, 10, 212–214. [Google Scholar]
- Neumann, P.J.; Cohen, J.T.; Weinstein, M.C. Updating cost-effectiveness— the curious resilience of the $50,000-per-QALY threshold. N Engl J Med 2014, 371(9), 796–797. [Google Scholar] [CrossRef]
- Yamamoto, D.; Campbell, J.D. Cost-effectiveness of multiple sclerosis disease-modifying therapies: a systematic review of the literature. Autoimmun Dis 2012, 784364. [Google Scholar] [CrossRef]
- Noyes, K.; Bajorska, A.; Chappel, A.; et al. Cost-effectiveness of disease-modifying therapy for multiple sclerosis: a populationbased study. Neurology 2011, 77(4), 355–363. [Google Scholar] [CrossRef]
- Briggs, A.H.; Wonderling, D.E.; Mooney, C.Z. Pulling cost-effectiveness analysis up by its bootstraps: a non-parametric approach to confidence interval estimation. Health Economics 1997, 6, 327–340. [Google Scholar] [CrossRef]
- Lothgren, M.; Zethraeus, N. Definition, interpretation and calculation of cost-effectiveness acceptability curves. Health Economics 2000, 9, 623–630. [Google Scholar] [CrossRef]
- ICER. Disease-Modifying Therapies for Relapsing-Remitting and Primary-Progressive Multiple Sclerosis: Effectiveness and Value. Final Evidence Report; Institute for Clinical and Economic Review, 2017. [Google Scholar]
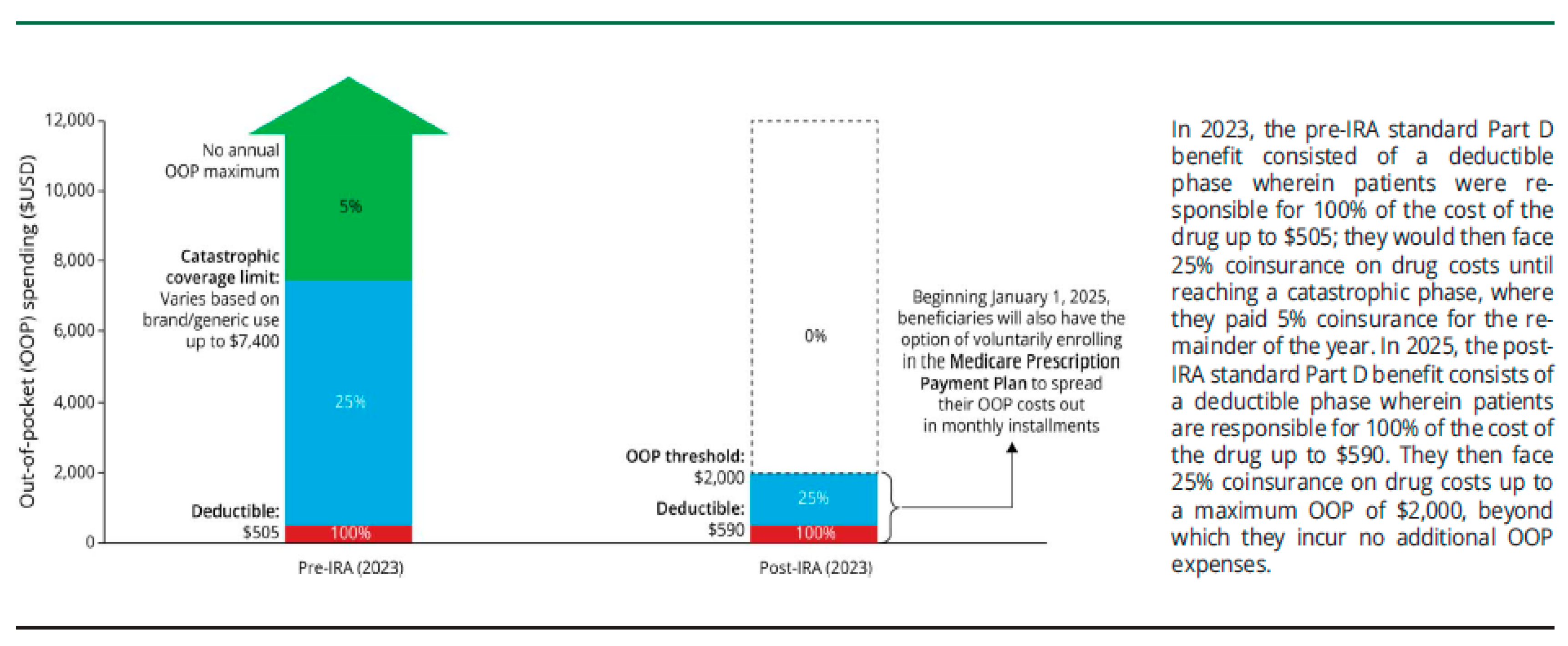
- Dusetzina, S.B.; Conti, R.M.; Yu, N.L.; Bach, P.B. Association of Prescription Drug Price Rebates in Medicare Part D With Patient Out-of-Pocket and Federal Spending. JAMA Intern Med 2017, 177, 1185–1188. [Google Scholar] [CrossRef]
- Aitken, M. Understanding Medicine Use and Expenditure in the U.S. 2025, IQVIA Institute for Human Data Science. Available online: https://www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/.
- Li, P.; Lin, J.K.; Klebanoff, M.J.; Palkar, R.; Chahin; S. Doshi, J.A. Reducing the Out-of-Pocket Costs of Disease-Modifying Therapies for Medicare Beneficiaries With Multiple Sclerosis. Neurol Clin Pract. 2025, 15, e200536. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.D. Medicare Drug Price Negotiation in the United States: Implications and Unanswered Questions. Value in Health 2023, 26(3), 394–399. [Google Scholar] [CrossRef] [PubMed]
- CMS. 2026. Available online: https://data.cms.gov/summary-statistics-on-use-and-payments/medicare-medicaid-spending-by-drug/medicare-part-d-spending-by-drug (accessed on 4 March 2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




