Submitted:
21 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients and Stimulation Protocols
2.2. MDS-UPDRS-8 Assessment
2.3. Resting Tremor Detection
2.4. Quantification and Analysis of Resting Tremor
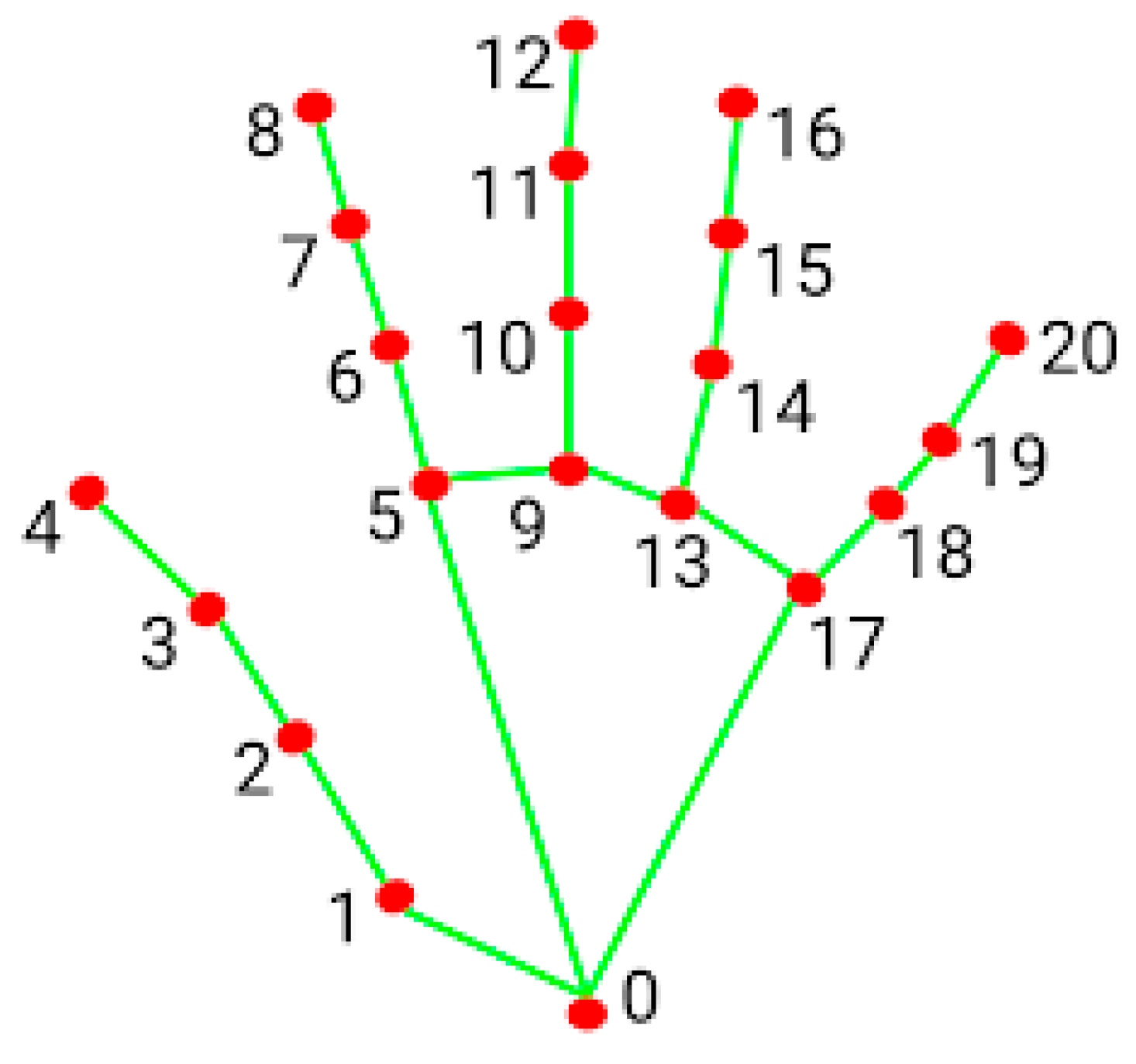
2.5. Analysis of MDS-UPDRS-8 Scores
3. Results
3.1. Tremor Analysis
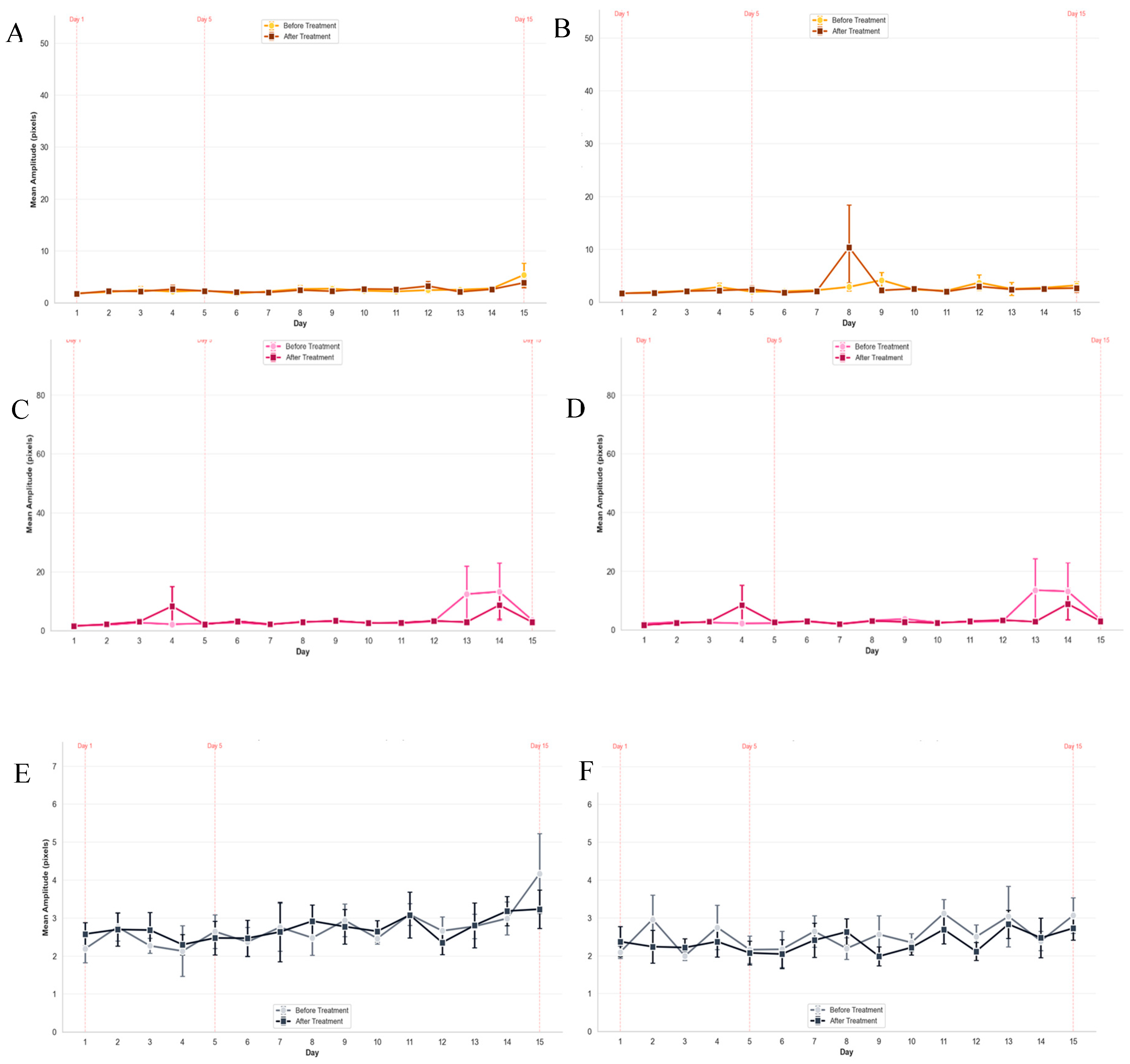
3.1.1. Mean Resting Tremor Amplitude Across Treatment Days by Neurostimulation Protocol
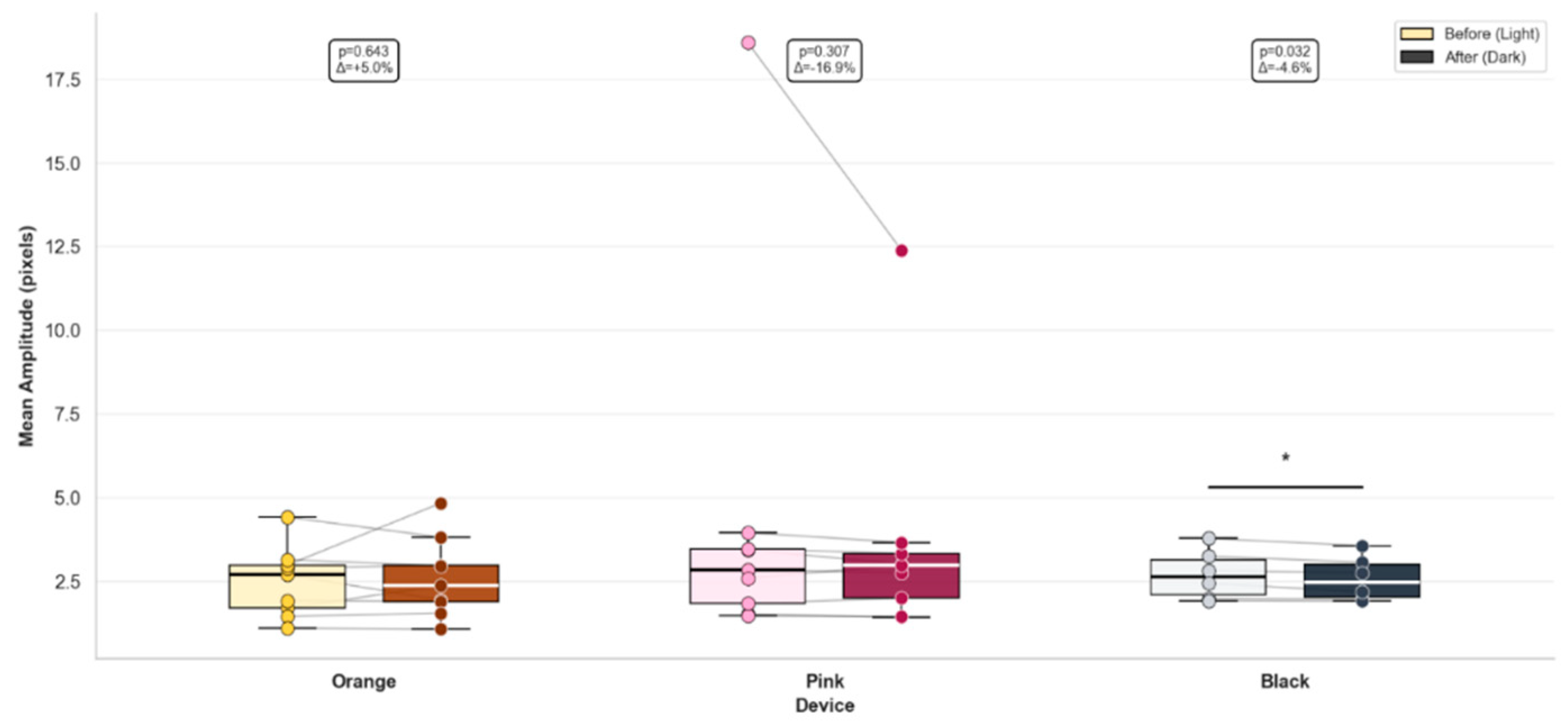
3.1.2. Effect of Treatment Type on Mean Resting Tremor Amplitude (3–12 Hz)
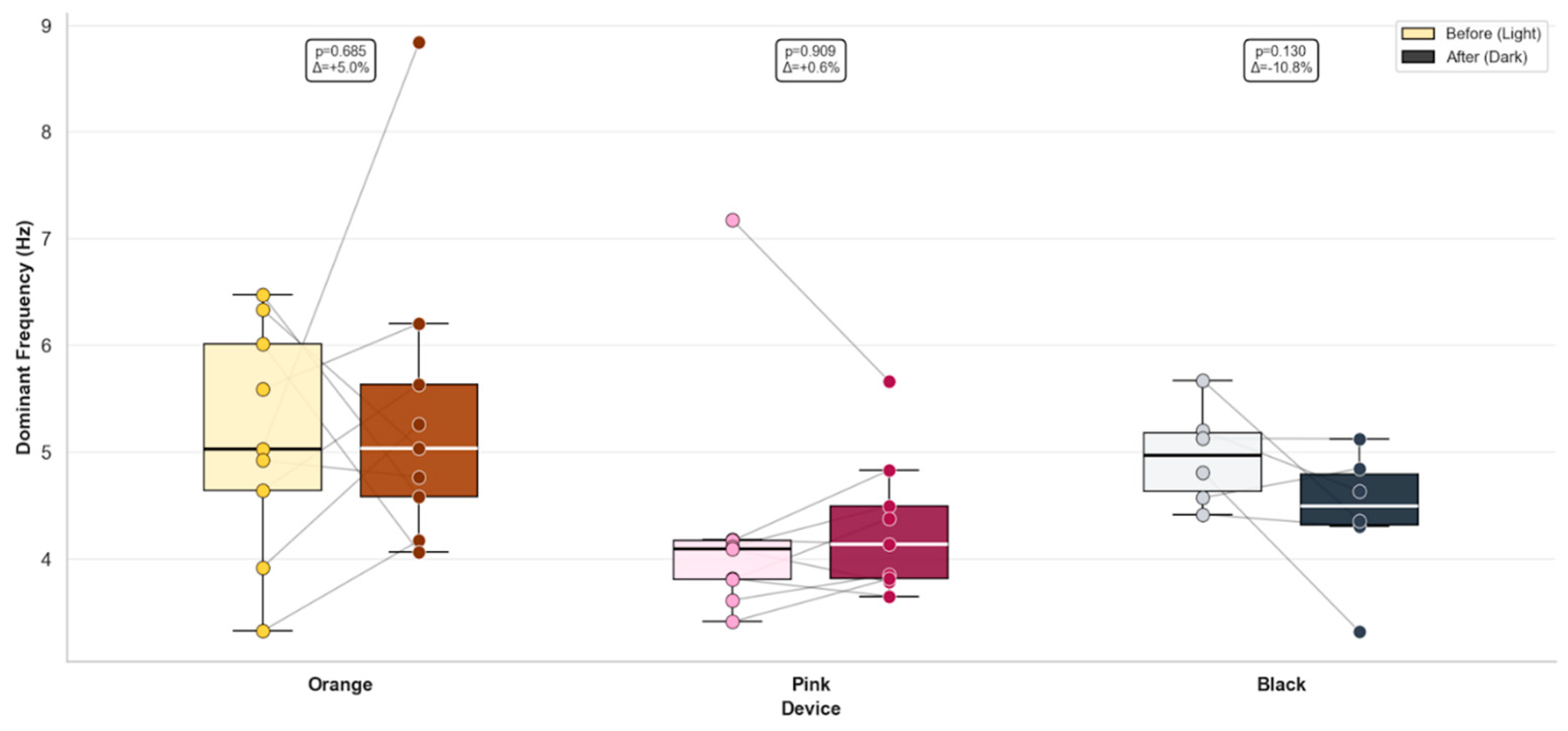
3.1.3. Effect of Treatment Type on Dominant Tremor Frequency
| Metric | Device | % Change (Mean ± SEM) | p-value |
|---|---|---|---|
| Mean Amplitude | MP-am-tPRNS | +4.96 ± 10.30 | 0.64 |
| am-tPRNS | −16.89 ± 15.49 | 0.31 | |
| am-tPCS | −4.58 ± 1.56 | 0.03* | |
| Dominant Frequency | MP-am-tPRNS | +5.00 ± 11.89 | 0.69 |
| am-tPRNS | −0.61 ± 5.19 | 0.91 | |
| am-tPCS | −10.79 ± 5.97 | 0.13 |
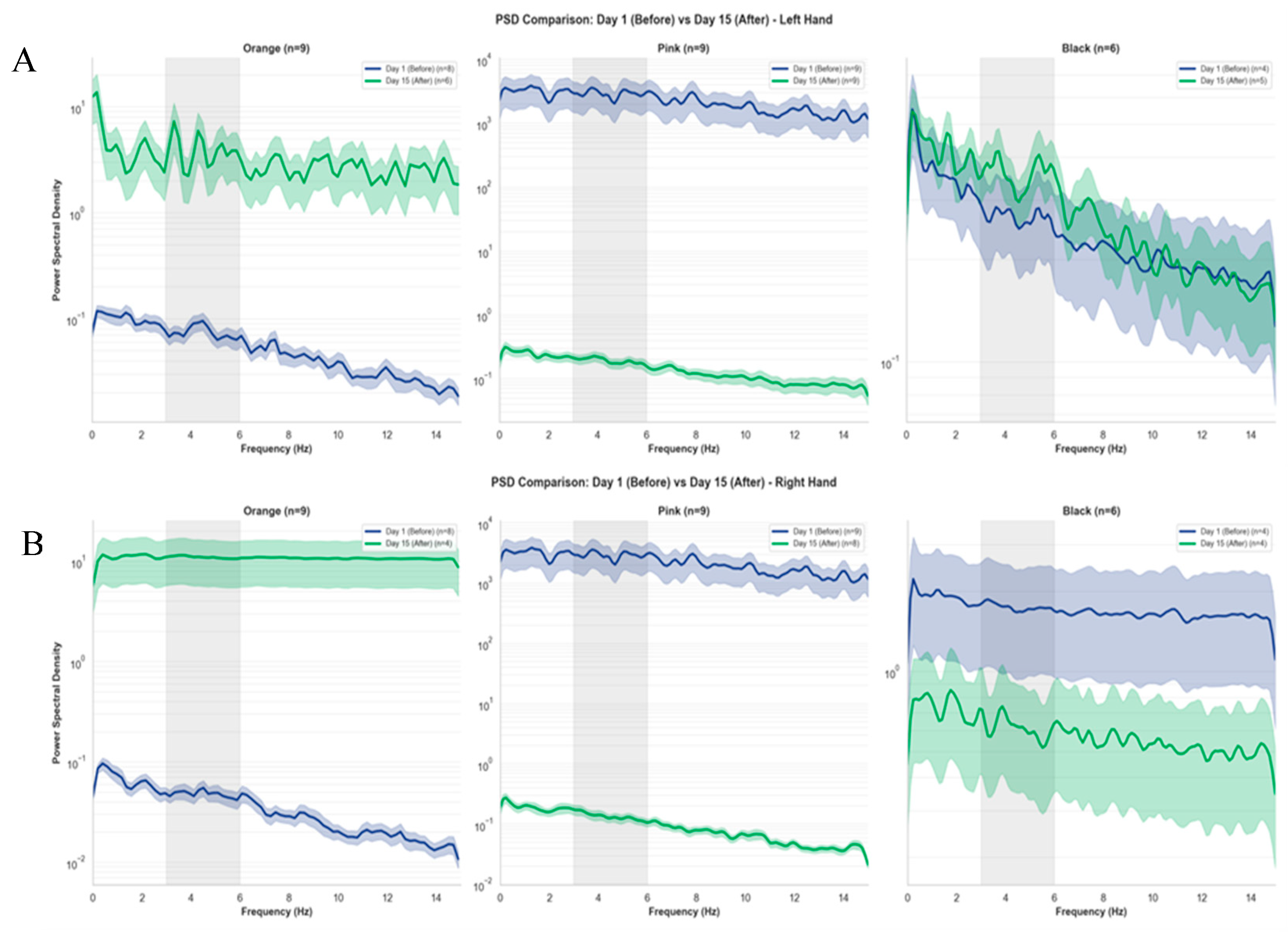
3.1.4. PSD Representations of Tremor Frequency Across Device Groups
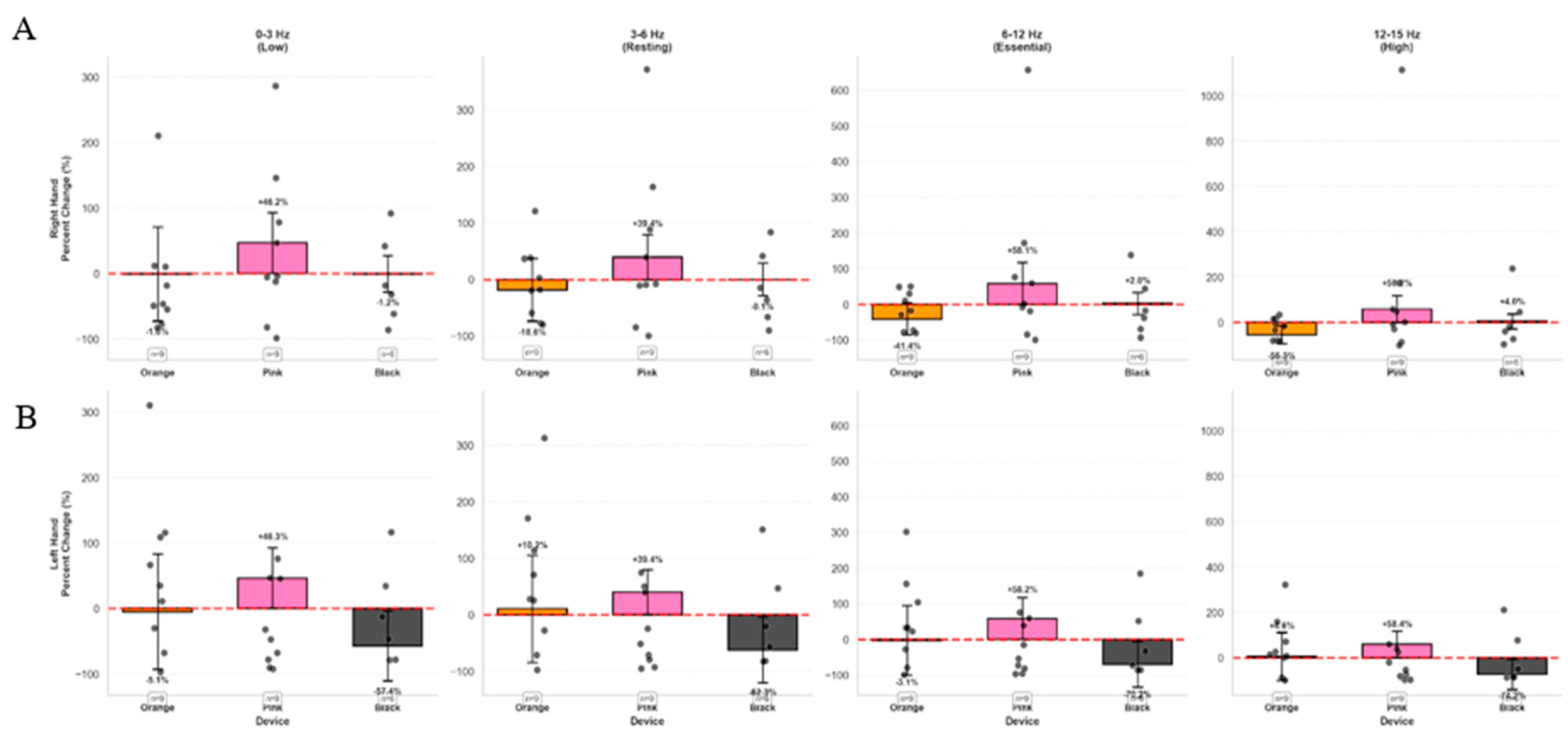
3.1.5. Percentage Changes in Spectral Band Power Across Device Groups
3.2. MDS-UPDRS-8 Analysis
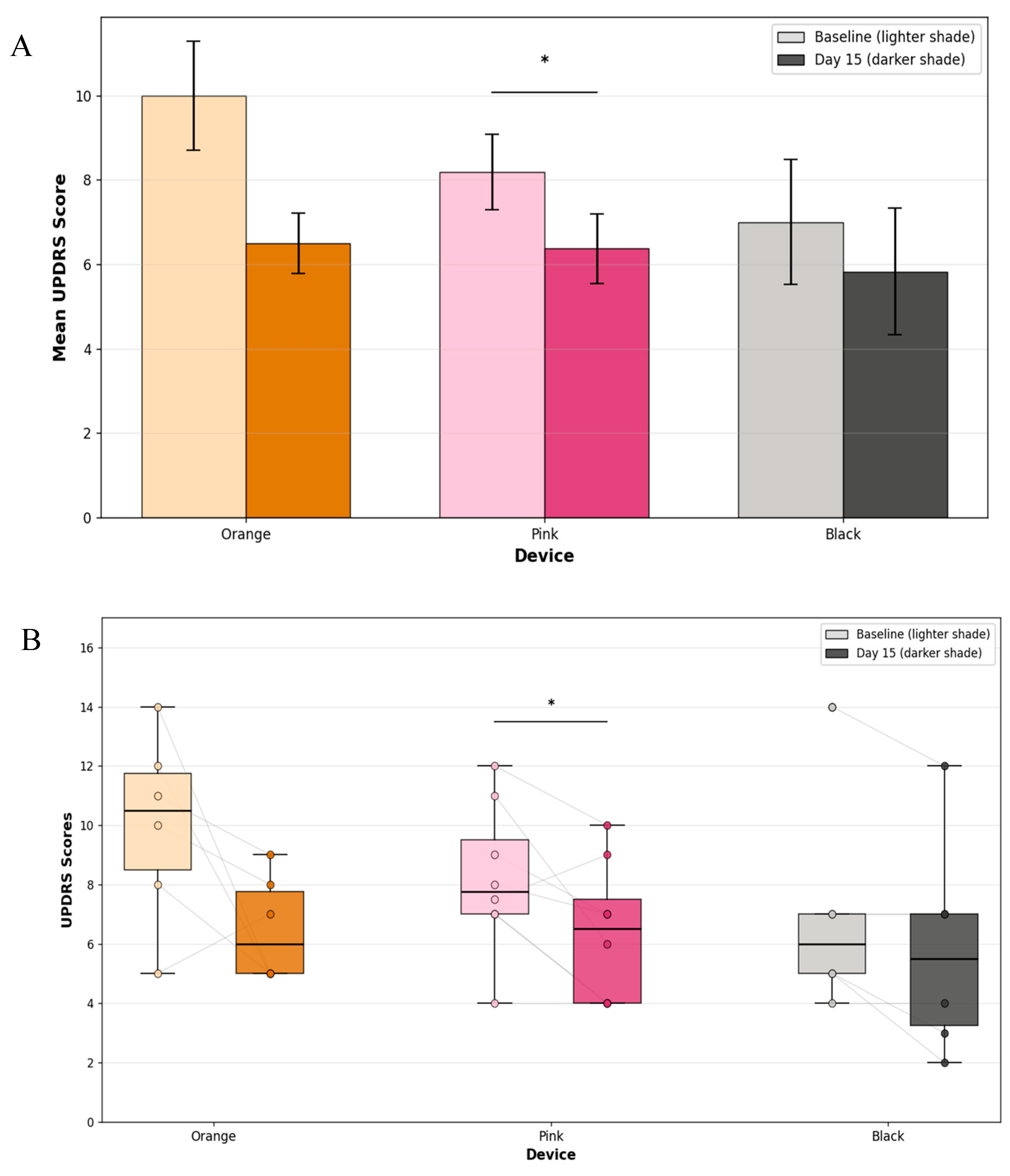
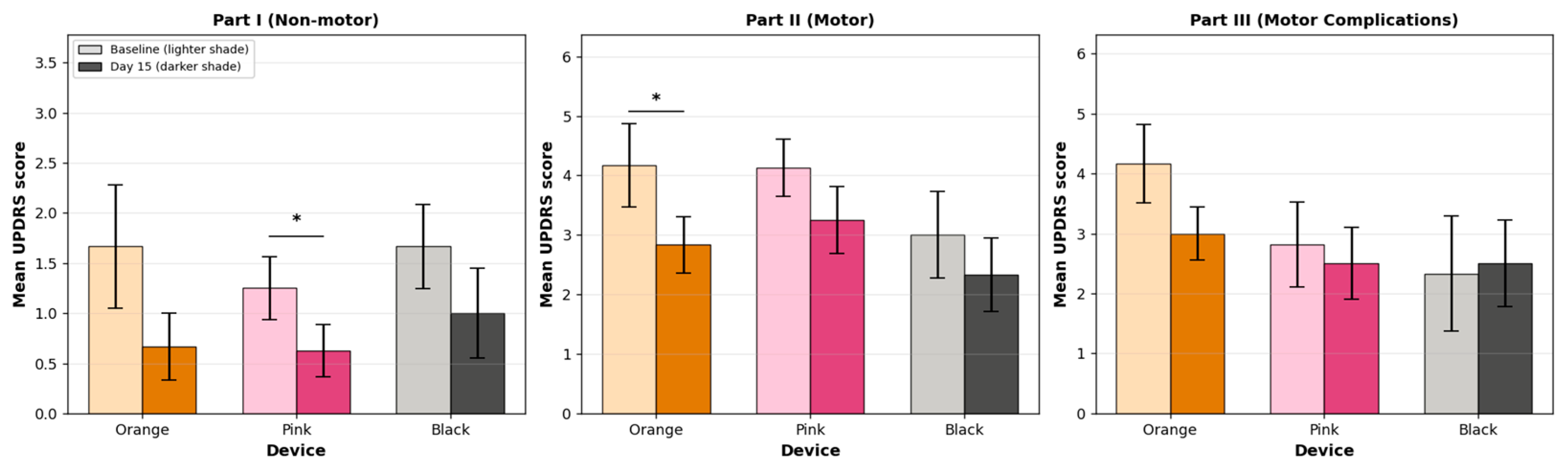
3.2.1. Mean Baseline and Endline MDS-UPDRS-8 Scores by Section and Device
3.2.2. Baseline and Endline Overall MDS-UPDRS-8 Scores by Device
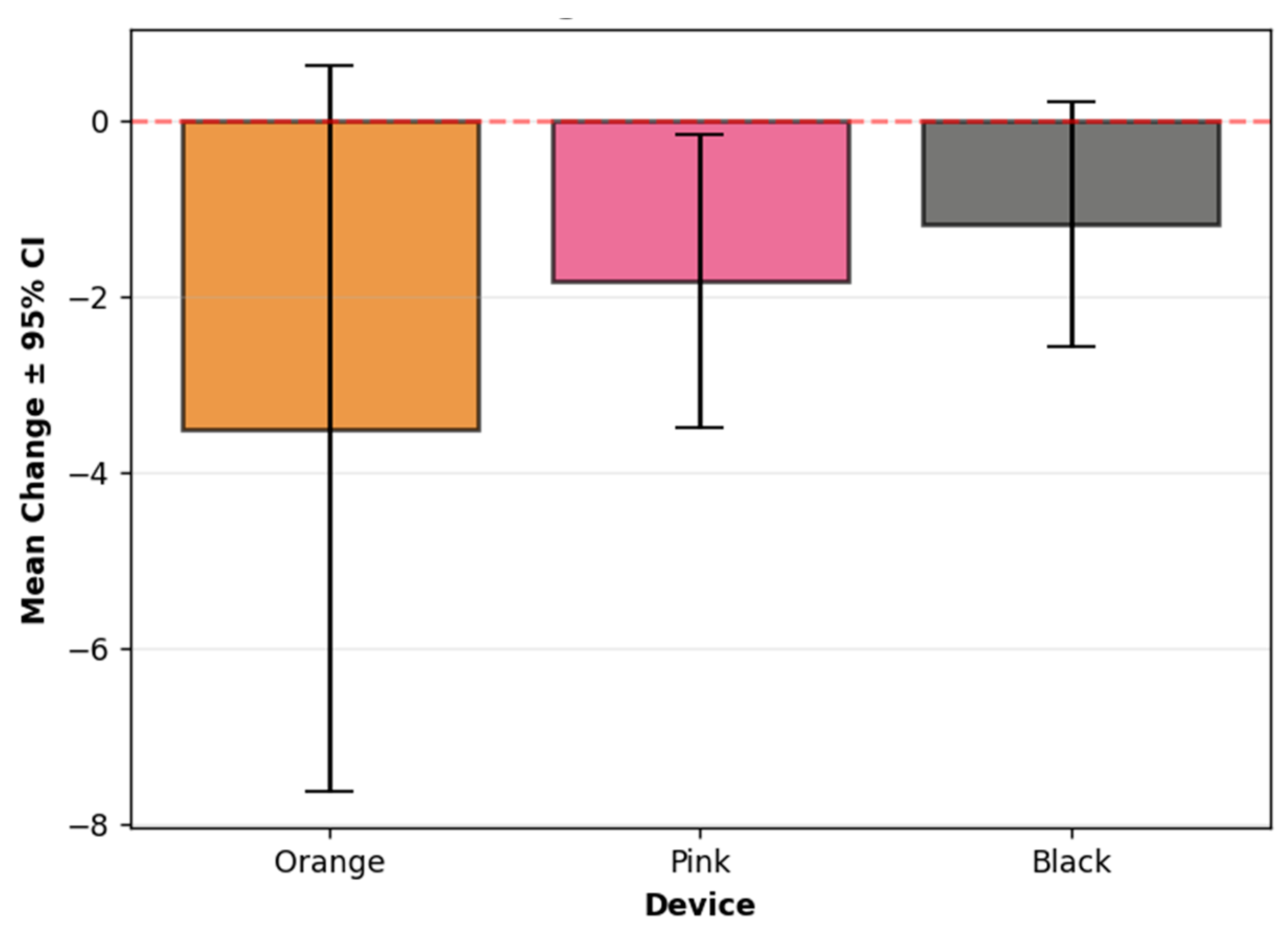
3.2.3. Effect of Stimulation on the Change in Absolute Overall MDS-UPDRS-8 Scores
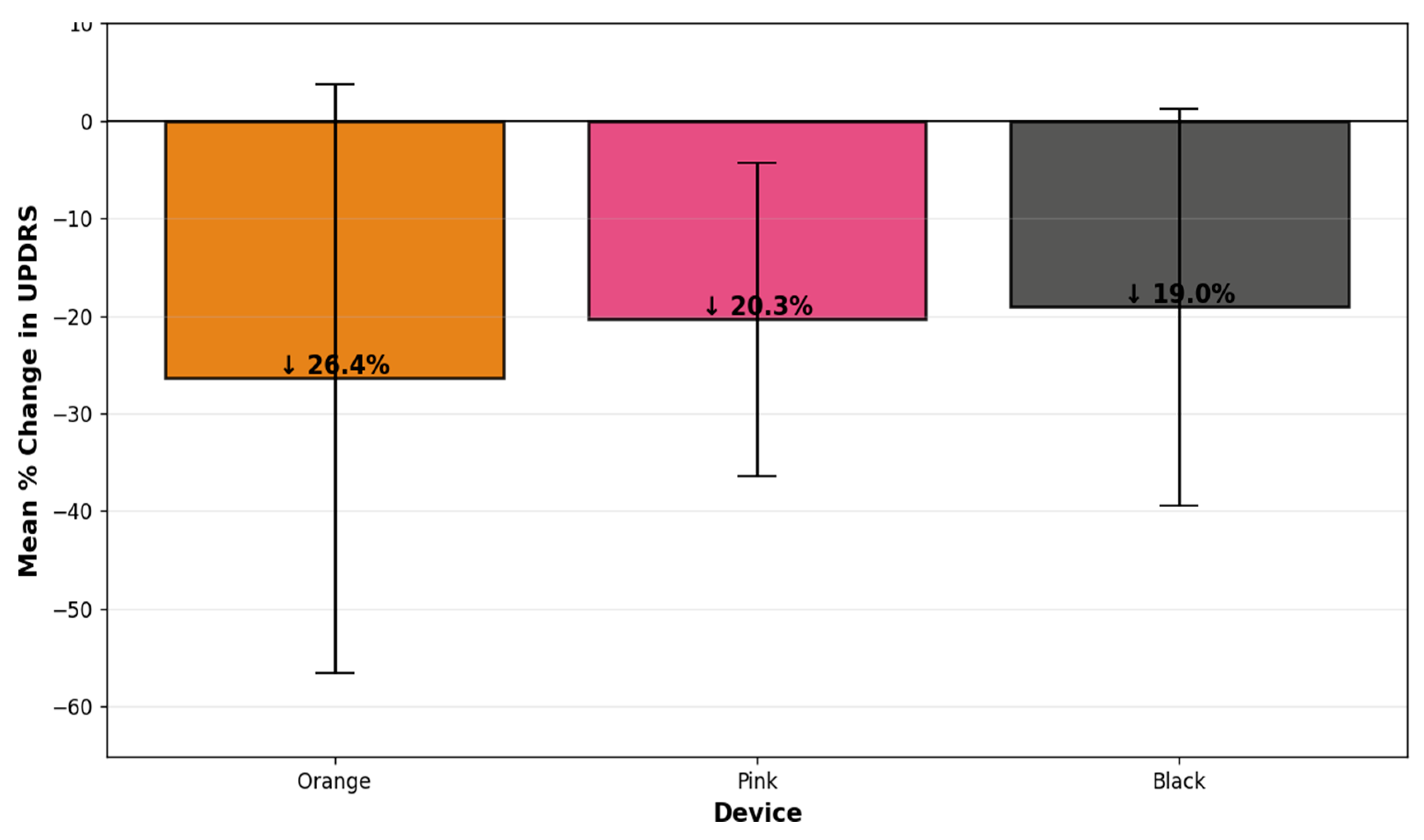
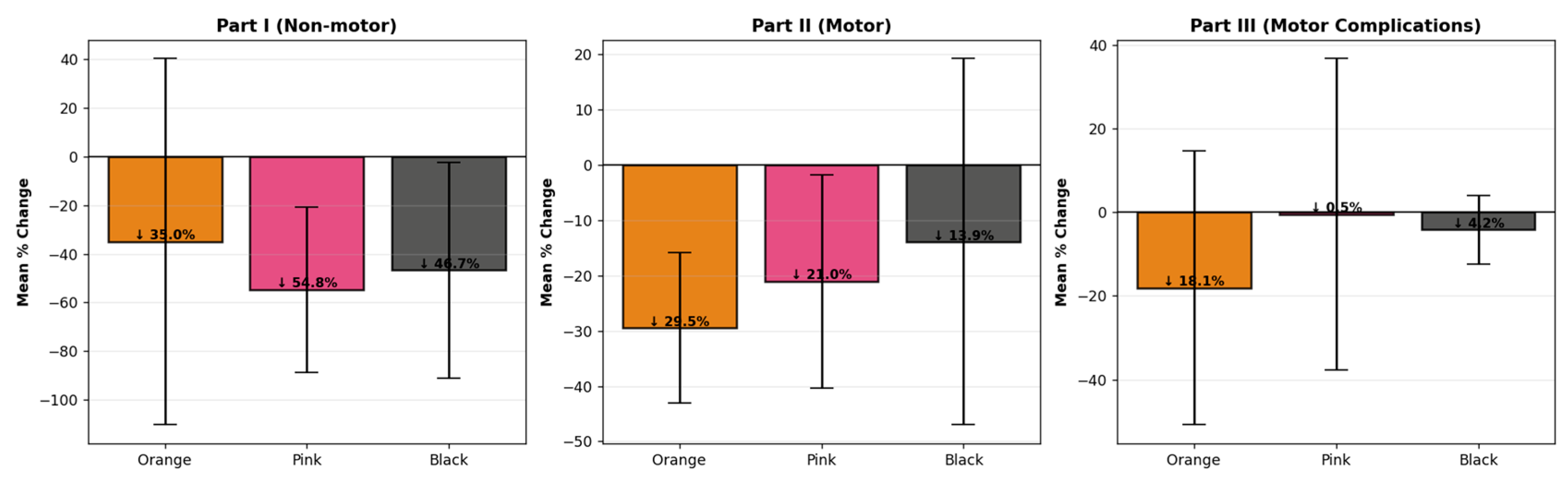
3.2.4. Mean Percentage Change in Overall MDS-UPDRS-8 Scores by Stimulation Type
3.2.5. Mean Percentage Change in MDS-UPDRS-8 Component Scores by Stimulation Type
4. Discussion
5. Conclusions
Conflicts of Interest
Informed consent statement
Acknowledgments
References
- Moustafa, Ahmed A et al. “Motor symptoms in Parkinson’s disease: A unified framework.” Neuroscience and biobehavioral reviews vol. 68 (2016): 727-740. [CrossRef]
- Fabbri, Margherita et al. “Response of non-motor symptoms to levodopa in late-stage Parkinson’s disease: Results of a levodopa challenge test.” Parkinsonism & related disorders vol. 39 (2017): 37-43. [CrossRef]
- Hariz, Marwan, and Patric Blomstedt. “Deep brain stimulation for Parkinson’s disease.” Journal of internal medicine vol. 292,5 (2022): 764-778. [CrossRef]
- Ni, Rui et al. “Novel Non-invasive Transcranial Electrical Stimulation for Parkinson’s Disease.” Frontiers in aging neuroscience vol. 14 880897. 12 Apr. 2022. [CrossRef]
- Hauser, R. A., Lyons, K. E., & Pahwa, R. (2012). The UPDRS-8: a brief clinical assessment scale for Parkinson’s disease. The International journal of neuroscience, 122(7), 333–337.
- Zhang, Fan, et al. “MediaPipe Hands: On-Device Real-Time Hand Tracking.” arXiv, 18 June 2020, arXiv:2006.10214.
- Salkhori, F., et al. “Frontal Lobe Electrical Stimulation Enhances Connectivity in Alzheimer’s Disease Networks: Evidence from rs-fMRI.” New Insights in Brain-Computer Interface Systems, edited by N. H. Kashou, IntechOpen, 2024. [CrossRef]
| Device | Protocol | Parameters | n |
|---|---|---|---|
| Orange | Multi-Path am-tPRNS | 1000 Hz base, 2 Hz main frequency, 4-electrode rotation at 10-second intervals | 10 |
| Pink | am-tPRNS | 1000 Hz base, random frequency distribution, stochastic modulation, 4-electrode array | 10 |
| Black | am-tPCS | 1000 Hz base, 50% duty cycle, biphasic polarity switching, 2-electrode bipolar | 6 |
| Score | Part 1 | Part 2 | Part 3 | |||||
|---|---|---|---|---|---|---|---|---|
| Intellectual impairment | Mood | Rest tremor upper extremity | Finger taps | Gait walking | Off time | Dyskinesia duration | Dyskinesia disability | |
| 0 | None | No depression | None | Normal | Normal | None | None | Not disabling |
| 1 | Mild. Consistent forgetfulness with partial recollection of events | Periods of sadness or guilt greater than normal, never sustained for days or weeks. | Slight and infrequently present | Mild slowing and/or reduction in amplitude. | Walks slowly, may shuffle with short steps. | 1-25% of day. | 1-25% of day. | Mildly disabling |
| 2 | Moderate memory loss, with disorientation and moderate difficulty handling complex problems. Mild but definite | Sustained depression (1 week or more). | Mild in amplitude and persistent. Or moderate in amplitude, but only intermittently present. | Moderately impaired. Definite and early fatiguing. May have occasional arrests in movement. | Walks with difficulty, but requires little or no assistance; may have some festination, short steps, or propulsion. | 26-50% of day. | 26-50% of day. | Moderately disabling |
| 3 | Moderate in amplitude and present most of the time. | Severely impaired. Frequent hesitation in initiating movements or arrests in ongoing movement. | 51-75% of day. | 51-75% of day. | Severely disabling | |||
| 4 | Marked in amplitude and present most of the time. | 76-100% of day. | 76-100% of day. | |||||
| Metric | Max Reduction | Notes |
|---|---|---|
| Mean Euclidean amplitude | −39.8% (Patient 5, Left, MP-am-tPRNS ) | Largest single-patient reduction observed |
| Peak-to-peak amplitude | −67.2% (Patient 1, Left, MP-am-tPRNS ) | Peak-to-peak analysis; individual result |
| Dominant frequency | −24.4% (Patient 10, Right, MP-am-tPRNS ) | Largest frequency reduction observed |
| Patients with no change | 4 patients (Pts 19, 22, 23, 25) | Across all device groups |
| Metric | Device | Hand | % Change (Mean ± SEM) | p-value |
|---|---|---|---|---|
| Spectral Power (0–3 Hz) | MP-am-tPRNS | Left | −5.09 ± 88.20 | 0.96 |
| MP-am-tPRNS | Right | −1.58 ± 72.14 | 0.98 | |
| am-tPRNS | Left | +46.26 ± 46.43 | 0.35 | |
| am-tPRNS | Right | +46.20 ± 46.29 | 0.35 | |
| am-tPCS | Left | −57.43 ± 53.49 | 0.33 | |
| am-tPCS | Right | −1.17 ± 28.01 | 0.97 | |
| Spectral Power (3–6 Hz)ᵃ | MP-am-tPRNS | Left | +10.24 ± 94.90 | 0.92 |
| MP-am-tPRNS | Right | −18.58 ± 55.55 | 0.75 | |
| am-tPRNS | Left | +39.45 ± 39.68 | 0.35 | |
| am-tPRNS | Right | +39.45 ± 39.57 | 0.35 | |
| am-tPCS | Left | −62.34 ± 57.97 | 0.33 | |
| am-tPCS | Right | −0.07 ± 28.99 | 1.00 | |
| Spectral Power (6-12 Hz)ᵃ | MP-am-tPRNS | Left | −3.08 ± 97.26 | 0.98 |
| MP-am-tPRNS | Right | −41.42 ± 44.52 | 0.38 | |
| am-tPRNS | Left | +58.15 ± 58.51 | 0.35 | |
| am-tPRNS | Right | +58.08 ± 58.27 | 0.35 | |
| am-tPCS | Left | −70.21 ± 63.60 | 0.32 | |
| am-tPCS | Right | +2.01 ± 30.68 | 0.95 | |
| Spectral Power (12–15 Hz) | MP-am-tPRNS | Left | +4.64 ± 105.06 | 0.97 |
| MP-am-tPRNS | Right | −55.27 ± 39.82 | 0.20 | |
| am-tPRNS | Left | +58.15 ± 58.51 | 0.33 | |
| am-tPRNS | Right | +58.37 ± 58.90 | 0.35 | |
| am-tPCS | Left | −74.22 ± 66.97 | 0.32 | |
| am-tPCS | Right | +4.05 ± 32.20 | 0.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




