Submitted:
12 August 2023
Posted:
14 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Electrochemical Biosensors
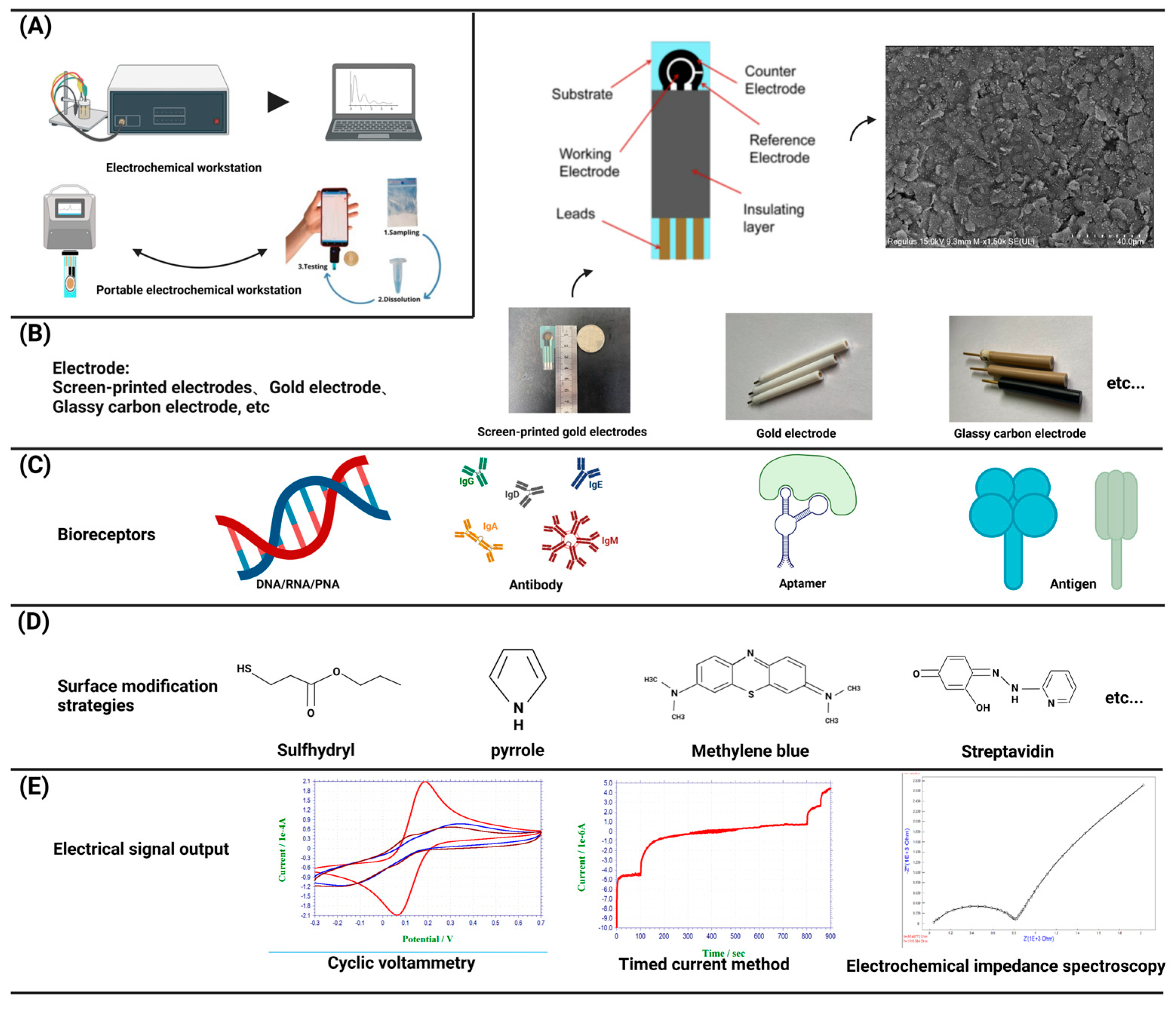
2.1. Components of electrochemical biosensor
2.1.1. Electrode
2.1.2. working station
2.2. Signal analysis and output
2.2.1. Voltammetry
2.2.2. Ampere method
2.2.3. Impedance method
3. The electrochemical biosensor model
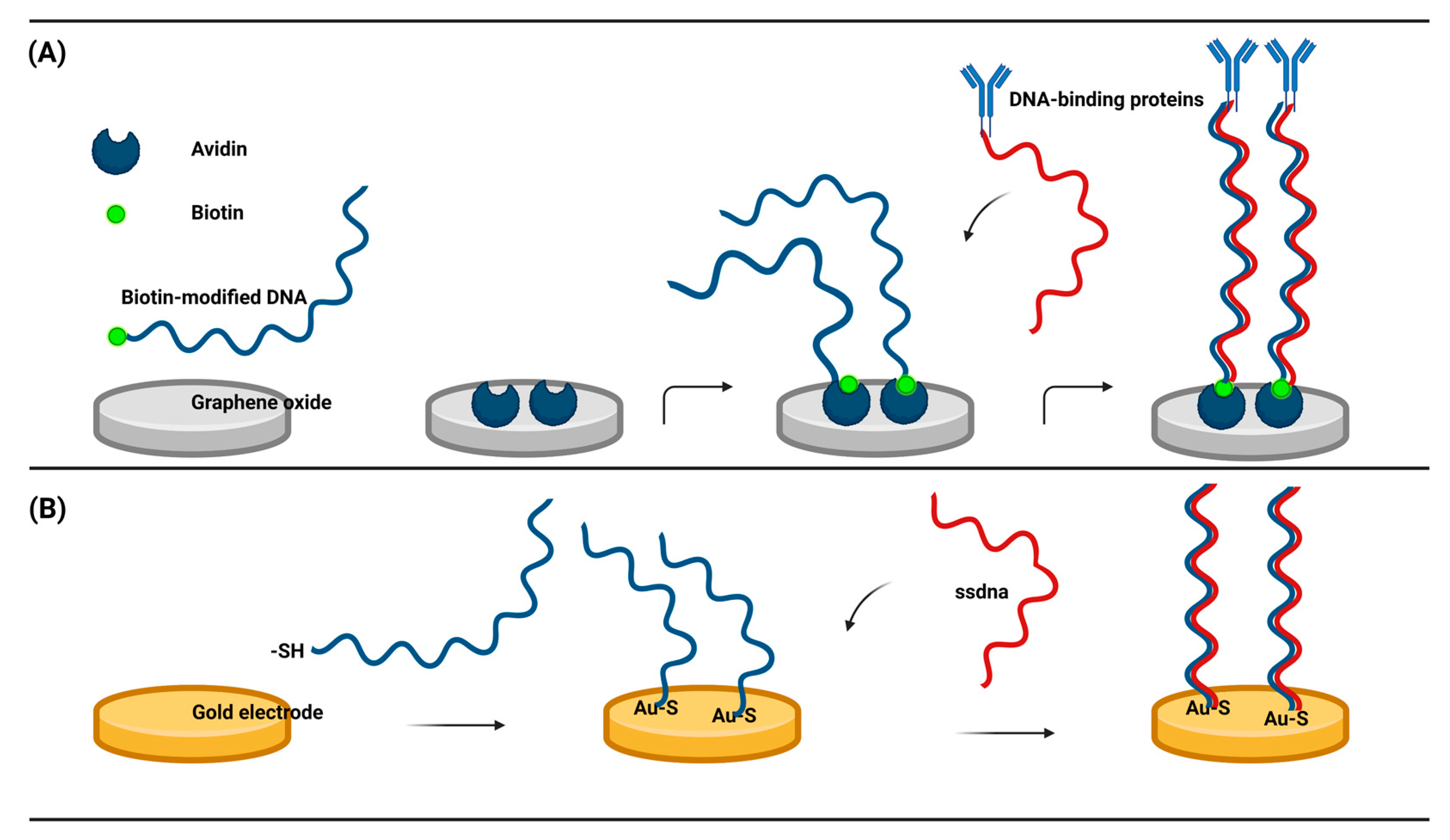
3.1. Nucleic acid based electrochemical biosensor model
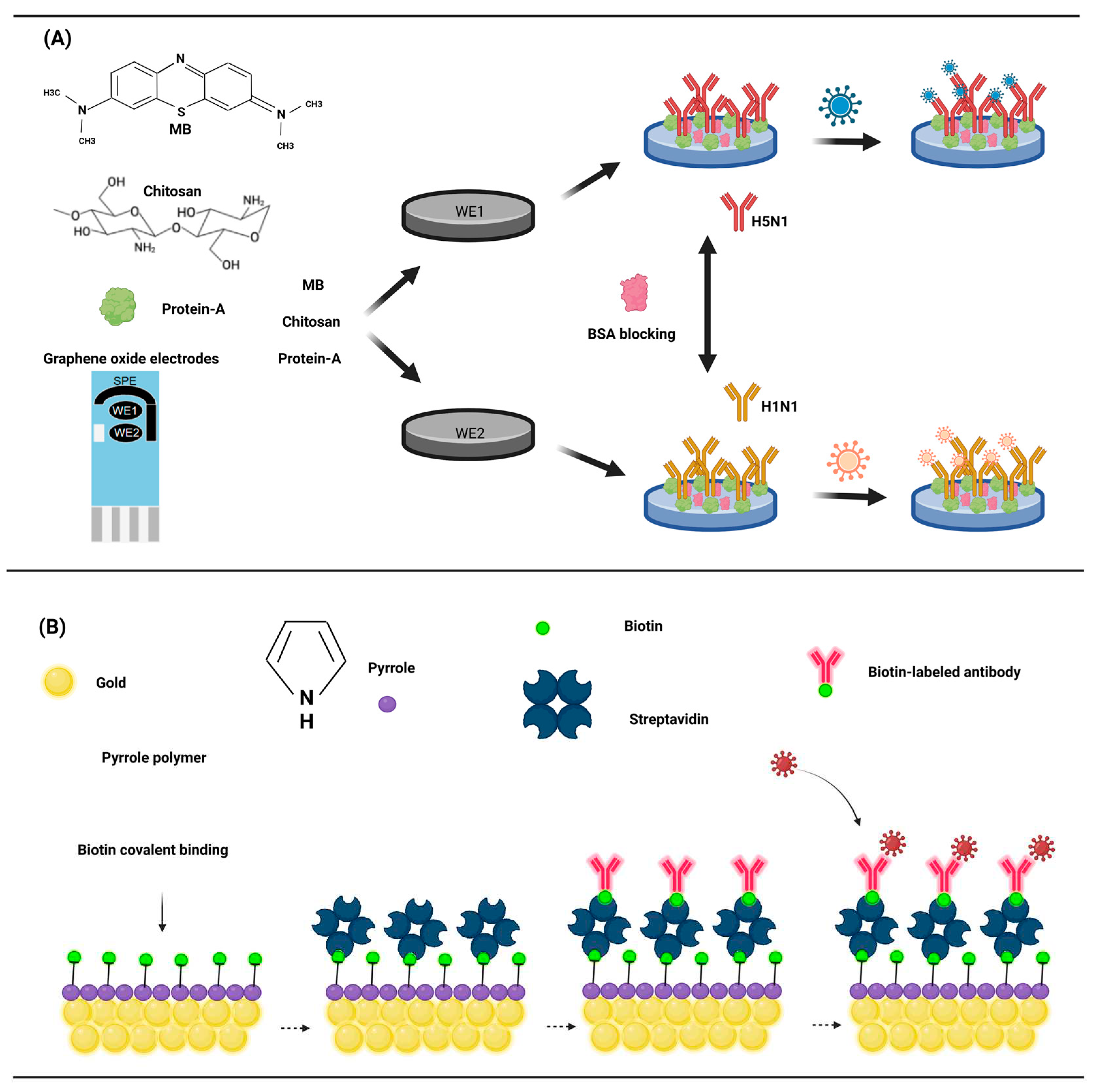
3.2. Antibodies and antigens based electrochemical biosensors model
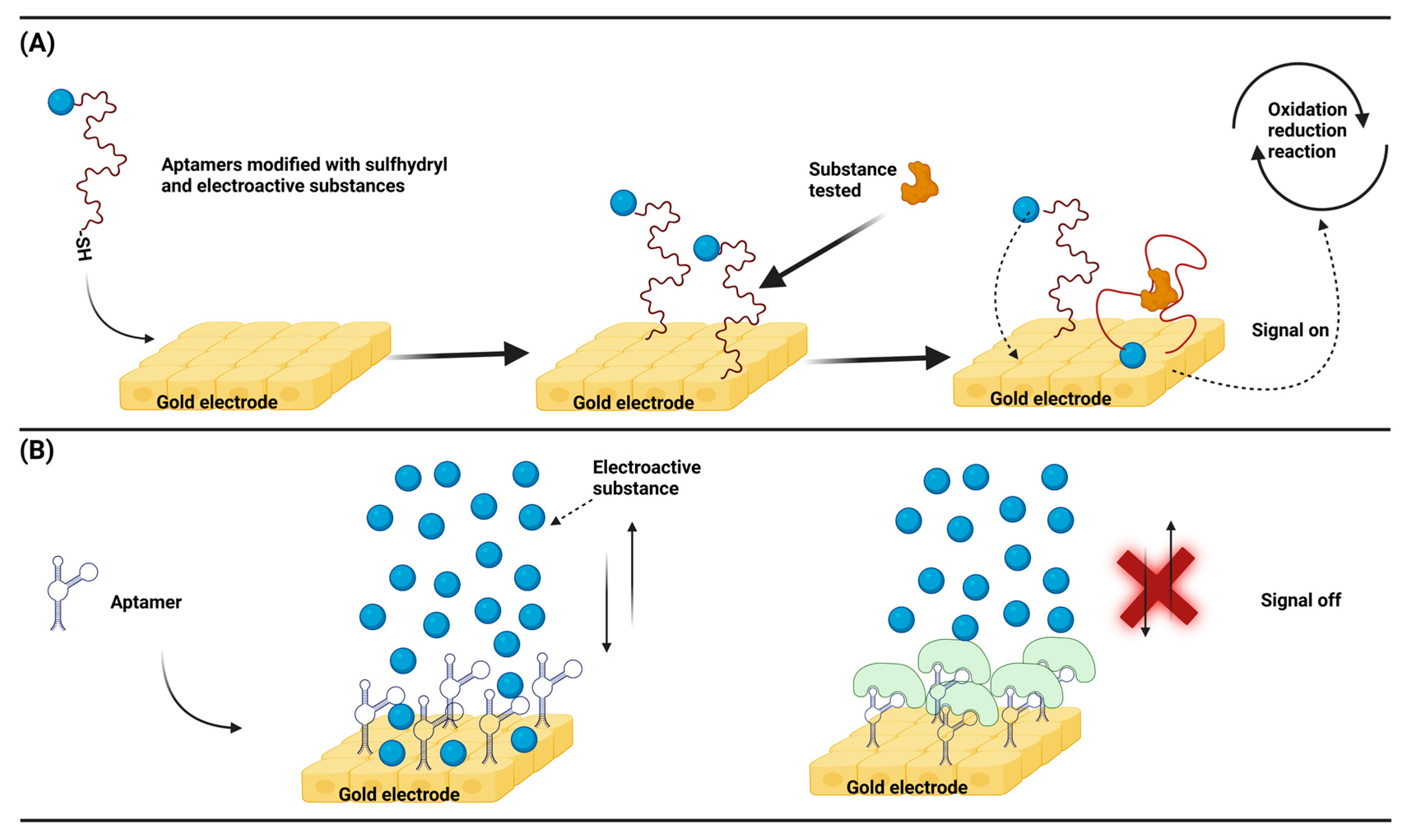
3.3. Nucleic acid aptamer-based electrochemical biosensor model
4. Advantages and limitation of electrochemical biosensors for virus detection
4.1. Advantages
4.2. Limitation
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Pellett, P.E.; Mitra, S.; Holland, T.C. Basics of virology. Handb Clin Neurol 2014, 123, 45–66. [Google Scholar] [PubMed]
- San, M.C. Virus maturation. Adv.Exp.Med.Biol. 2019, 1215, 129–158. [Google Scholar]
- Kalia, M.; Jameel, S. Virus entry paradigms. Amino Acids 2011, 41, 1147–1157. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, H.Y. Porcine enteric coronaviruses: an updated overview of the pathogenesis, prevalence, and diagnosis. Vet. Res. Commun. 2021, 45, 75–86. [Google Scholar] [CrossRef]
- Abd, E.M.; El-Saadony, M.T.; Alqhtani, A.H.; Swelum, A.A.; Salem, H.M.; Elbestawy, A.R.; Noreldin, A.E.; Babalghith, A.O.; Khafaga, A.F.; Hassan, M.I.; El-Tarabily, K.A. The relationship among avian influenza, gut microbiota and chicken immunity: an updated overview. Poult. Sci. 2022, 101, 102021. [Google Scholar]
- Labadie, T.; Batejat, C.; Leclercq, I.; Manuguerra, J.C. Historical discoveries on viruses in the environment and their impact on public health. Intervirology 2020, 63, 17–32. [Google Scholar] [CrossRef]
- Vidic, J.; Manzano, M.; Chang, C.M.; Jaffrezic-Renault, N. Advanced biosensors for detection of pathogens related to livestock and poultry. Vet. Res. 2017, 48, 11. [Google Scholar] [CrossRef]
- Neumann, E.J.; Kliebenstein, J.B.; Johnson, C.D.; Mabry, J.W.; Bush, E.J.; Seitzinger, A.H.; Green, A.L.; Zimmerman, J.J. Assessment of the economic impact of porcine reproductive and respiratory syndrome on swine production in the united states. JAVMA-J. Am. Vet. Med. Assoc. 2005, 227, 385–392. [Google Scholar] [CrossRef]
- Liu, S.; Zhuang, Q.; Wang, S.; Jiang, W.; Jin, J.; Peng, C.; Hou, G.; Li, J.; Yu, J.; Yu, X.; Liu, H.; Sun, S.; Yuan, L.; Chen, J. Control of avian influenza in china: strategies and lessons. Transbound. Emerg. Dis. 2020, 67, 1463–1471. [Google Scholar] [CrossRef]
- Nakada, S.; Fujimoto, Y.; Kohara, J.; Makita, K. Economic losses associated with mastitis due to bovine leukemia virus infection. J. Dairy Sci. 2023, 106, 576–588. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, X.; Qi, W.; Yang, Y.; Liu, Z.; An, T.; Wu, X.; Chen, J. Prevention and control strategies of african swine fever and progress on pig farm repopulation in china. Viruses-Basel 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Thi, T.; Pham-Thi-Ngoc, L.; Nguyen-Ngoc, Q.; Dang-Xuan, S.; Lee, H.S.; Nguyen-Viet, H.; Padungtod, P.; Nguyen-Thu, T.; Nguyen-Thi, T.; Tran-Cong, T.; Rich, K.M. An assessment of the economic impacts of the 2019 african swine fever outbreaks in vietnam. Front. Vet. Sci. 2021, 8, 686038. [Google Scholar] [CrossRef] [PubMed]
- Stegeman, A.; Bouma, A.; Elbers, A.R.; de Jong, M.C.; Nodelijk, G.; de Klerk, F.; Koch, G.; van Boven, M. Avian influenza a virus (h7n7) epidemic in the netherlands in 2003: course of the epidemic and effectiveness of control measures. J. Infect. Dis. 2004, 190, 2088–2095. [Google Scholar] [CrossRef] [PubMed]
- Aral, Y.; Yalcin, C.; Cevger, Y.; Sipahi, C.; Sariozkan, S. Financial effects of the highly pathogenic avian influenza outbreaks on the turkish broiler producers. Poult. Sci. 2010, 89, 1085–1088. [Google Scholar] [CrossRef] [PubMed]
- Mcorist, S.; Khampee, K.; Guo, A. Modern pig farming in the people's republic of china: growth and veterinary challenges. Rev. Sci. Tech. Off. Int. Epizoot. 2011, 30, 961–968. [Google Scholar] [CrossRef]
- Poonsuk, K.; Gimenez-Lirola, L.; Zimmerman, J.J. A review of foot-and-mouth disease virus (fmdv) testing in livestock with an emphasis on the use of alternative diagnostic specimens. Anim. Health Res. Rev. 2018, 19, 100–112. [Google Scholar] [CrossRef]
- Kennedy, D.A.; Cairns, C.; Jones, M.J.; Bell, A.S.; Salathe, R.M.; Baigent, S.J.; Nair, V.K.; Dunn, P.A.; Read, A.F. Industry-wide surveillance of marek's disease virus on commercial poultry farms. Avian Dis. 2017, 61, 153–164. [Google Scholar] [CrossRef]
- Thornton, P.K. Livestock production: recent trends, future prospects. Philos. Trans. R. Soc. B-Biol. Sci. 2010, 365, 2853–2867. [Google Scholar] [CrossRef]
- Monis, P.T.; Giglio, S. Nucleic acid amplification-based techniques for pathogen detection and identification. Infect. Genet. Evol. 2006, 6, 2–12. [Google Scholar] [CrossRef]
- Day, M.J. Introduction to antigen and antibody assays. Top. Companion Anim. Med. 2015, 30, 128–131. [Google Scholar] [CrossRef]
- Leland, D.S.; Ginocchio, C.C. Role of cell culture for virus detection in the age of technology. Clin. Microbiol. Rev. 2007, 20, 49–78. [Google Scholar] [CrossRef] [PubMed]
- Babaei, A.; Rafiee, N.; Taheri, B.; Sohrabi, H.; Mokhtarzadeh, A. Recent advances in early diagnosis of viruses associated with gastroenteritis by biosensors. Biosensors-Basel 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Aydin, E.B.; Aydin, M.; Sezginturk, M.K. Advances in electrochemical immunosensors. Advan. Clin. Chem. 2019, 92, 1–57. [Google Scholar]
- Manring, N.; Ahmed, M.; Tenhoff, N.; Smeltz, J.L.; Pathirathna, P. Recent advances in electrochemical tools for virus detection. Anal. Chem. 2022, 94, 7149–7157. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Lu, Y. Portable and quantitative detection of protein biomarkers and small molecular toxins using antibodies and ubiquitous personal glucose meters. Anal. Chem. 2012, 84, 4174–4178. [Google Scholar] [CrossRef] [PubMed]
- Banakar, M.; Hamidi, M.; Khurshid, Z.; Zafar, M.S.; Sapkota, J.; Azizian, R.; Rokaya, D. Electrochemical biosensors for pathogen detection: an updated review. Biosensors-Basel 2022, 12. [Google Scholar] [CrossRef]
- Eom, H.; Kang, J.; Jang, S.; Kwon, O.; Choi, S.; Shin, J.; Nam, I. Evaluating the electrochemical properties of supercapacitors using the three-electrode system. J. Vis. Exp. 2022. [Google Scholar] [CrossRef]
- Lereau, M.; Fournier-Wirth, C.; Mayen, J.; Farre, C.; Meyer, A.; Dugas, V.; Cantaloube, J.F.; Chaix, C.; Vasseur, J.J.; Morvan, F. Development of innovative and versatile polythiol probes for use on elosa or electrochemical biosensors: application in hepatitis c virus genotyping. Anal. Chem. 2013, 85, 9204–9212. [Google Scholar] [CrossRef]
- El-Said, W.A.; Abdelshakour, M.; Choi, J.H.; Choi, J.W. Application of conducting polymer nanostructures to electrochemical biosensors. Molecules 2020, 25. [Google Scholar] [CrossRef]
- Ronkainen, N.J.; Halsall, H.B.; Heineman, W.R. Electrochemical biosensors. Chem. Soc. Rev. 2010, 39, 1747–1763. [Google Scholar] [CrossRef]
- Inkpen, M.S.; Liu, Z.F.; Li, H.; Campos, L.M.; Neaton, J.B.; Venkataraman, L. Non-chemisorbed gold-sulfur binding prevails in self-assembled monolayers. Nat. Chem. 2019, 11, 351–358. [Google Scholar] [CrossRef]
- Lawal, A.T. Synthesis and utilisation of graphene for fabrication of electrochemical sensors. Talanta 2015, 131, 424–443. [Google Scholar] [CrossRef]
- Costa-Rama, E.; Fernandez-Abedul, M.T. Paper-based screen-printed electrodes: a new generation of low-cost electroanalytical platforms. Biosensors-Basel 2021, 11. [Google Scholar] [CrossRef]
- Antuna-Jimenez, D.; Gonzalez-Garcia, M.B.; Hernandez-Santos, D.; Fanjul-Bolado, P. Screen-printed electrodes modified with metal nanoparticles for small molecule sensing. Biosensors-Basel 2020, 10. [Google Scholar] [CrossRef]
- Ji, D.; Liu, L.; Li, S.; Chen, C.; Lu, Y.; Wu, J.; Liu, Q. Smartphone-based cyclic voltammetry system with graphene modified screen printed electrodes for glucose detection. Biosens. Bioelectron. 2017, 98, 449–456. [Google Scholar] [CrossRef]
- Mohamad, N.N.; Ridhuan, N.S.; Abdul, R.K. Progress of enzymatic and non-enzymatic electrochemical glucose biosensor based on nanomaterial-modified electrode. Biosensors-Basel 2022, 12. [Google Scholar] [CrossRef]
- Gruber, P.; Marques, M.; Szita, N.; Mayr, T. Integration and application of optical chemical sensors in microbioreactors. Lab Chip 2017, 17, 2693–2712. [Google Scholar] [CrossRef]
- Venton, B.J.; Cao, Q. Fundamentals of fast-scan cyclic voltammetry for dopamine detection. Analyst 2020, 145, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, K.; Naik, A.A.; Chaudhary, S.; Parvatalu, D.; Santhanam, V. Prudent practices in ex situ durability analysis using cyclic voltammetry for platinum-based electrocatalysts. Chem.-Asian J. 2021, 16, 3311–3325. [Google Scholar] [CrossRef] [PubMed]
- Yashin, Y.I.; Nemzer, B.V.; Ryzhnev, V.Y.; Yashin, A.Y.; Chernousova, N.I.; Fedina, P.A. Creation of a databank for content of antioxidants in food products by an amperometric method. Molecules 2010, 15, 7450–7466. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Rani, R. Development of biosensors for the detection of biological warfare agents: its issues and challenges. Sci. Prog. 2013, 96, 294–308. [Google Scholar] [CrossRef] [PubMed]
- Magar, H.S.; Hassan, R.; Mulchandani, A. Electrochemical impedance spectroscopy (eis): principles, construction, and biosensing applications. Sensors 2021, 21. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Hasan, M.R.; Hossain, S.I.; Ahommed, M.S.; Daizy, M. Ultrasensitive detection of pathogenic viruses with electrochemical biosensor: state of the art. Biosens. Bioelectron. 2020, 166, 112431. [Google Scholar] [CrossRef]
- Wehmeyer, K.R.; White, R.J.; Kissinger, P.T.; Heineman, W.R. Electrochemical affinity assays/sensors: brief history and current status. Annu. Rev. Anal. Chem. 2021, 14, 109–131. [Google Scholar] [CrossRef] [PubMed]
- Mahato, K.; Kumar, A.; Maurya, P.K.; Chandra, P. Shifting paradigm of cancer diagnoses in clinically relevant samples based on miniaturized electrochemical nanobiosensors and microfluidic devices. Biosens. Bioelectron. 2018, 100, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Yoo, E.H.; Lee, S.Y. Glucose biosensors: an overview of use in clinical practice. Sensors 2010, 10, 4558–4576. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.D.; Turner, A.P. Home blood glucose biosensors: a commercial perspective. Biosens. Bioelectron. 2005, 20, 2435–2453. [Google Scholar] [CrossRef]
- Wanjari, V.P.; Reddy, A.S.; Duttagupta, S.P.; Singh, S.P. Laser-induced graphene-based electrochemical biosensors for environmental applications: a perspective. Environ. Sci. Pollut. Res. 2023, 30, 42643–42657. [Google Scholar] [CrossRef]
- Hernandez-Vargas, G.; Sosa-Hernandez, J.E.; Saldarriaga-Hernandez, S.; Villalba-Rodriguez, A.M.; Parra-Saldivar, R.; Iqbal, H. Electrochemical biosensors: a solution to pollution detection with reference to environmental contaminants. Biosensors-Basel 2018, 8. [Google Scholar] [CrossRef]
- Li, Z.; Mohamed, M.A.; Vinu, M.A.; Zhu, Z.; Sharma, V.; Mishra, G.K.; Mishra, R.K. Application of electrochemical aptasensors toward clinical diagnostics, food, and environmental monitoring: review. Sensors 2019, 19. [Google Scholar] [CrossRef]
- Reta, N.; Saint, C.P.; Michelmore, A.; Prieto-Simon, B.; Voelcker, N.H. Nanostructured electrochemical biosensors for label-free detection of water- and food-borne pathogens. ACS Appl. Mater. Interfaces 2018, 10, 6055–6072. [Google Scholar] [CrossRef]
- Cesewski, E.; Johnson, B.N. Electrochemical biosensors for pathogen detection. Biosens. Bioelectron. 2020, 159, 112214. [Google Scholar] [CrossRef] [PubMed]
- Goud, K.Y.; Reddy, K.K.; Khorshed, A.; Kumar, V.S.; Mishra, R.K.; Oraby, M.; Ibrahim, A.H.; Kim, H.; Gobi, K.V. Electrochemical diagnostics of infectious viral diseases: trends and challenges. Biosens. Bioelectron. 2021, 180, 113112. [Google Scholar] [CrossRef] [PubMed]
- Abi, A.; Mohammadpour, Z.; Zuo, X.; Safavi, A. Nucleic acid-based electrochemical nanobiosensors. Biosens. Bioelectron. 2018, 102, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Palecek, E. Oscillographic polarography of highly polymerized deoxyribonucleic acid. Nature 1960, 188, 656–657. [Google Scholar] [CrossRef]
- Yang, I.V.; Thorp, H.H. Modification of indium tin oxide electrodes with repeat polynucleotides: electrochemical detection of trinucleotide repeat expansion. Anal. Chem. 2001, 73, 5316–5322. [Google Scholar] [CrossRef]
- Macwan, I.; Khan, M.; Aphale, A.; Singh, S.; Liu, J.; Hingorani, M.; Patra, P. Interactions between avidin and graphene for development of a biosensing platform. Biosens. Bioelectron. 2017, 89, 326–333. [Google Scholar] [CrossRef]
- Gorodetsky, A.A.; Buzzeo, M.C.; Barton, J.K. Dna-mediated electrochemistry. Bioconjugate Chem. 2008, 19, 2285–2296. [Google Scholar] [CrossRef]
- Cajigas, S.; Alzate, D.; Orozco, J. Gold nanoparticle/dna-based nanobioconjugate for electrochemical detection of zika virus. Microchim. Acta 2020, 187, 594. [Google Scholar] [CrossRef]
- Ilkhani, H.; Farhad, S. A novel electrochemical dna biosensor for ebola virus detection. Anal. Biochem. 2018, 557, 151–155. [Google Scholar] [CrossRef]
- Fan, Y.; Li, X.; Zhang, L.; Zong, Z.; Wang, F.; Huang, J.; Zeng, L.; Zhang, C.; Yan, H.; Zhang, L.; Zhou, F. Sumoylation in viral replication and antiviral defense. Adv. Sci. 2022, 9, e2104126. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Shetti, N.P.; Jagannath, S.; Aminabhavi, T.M. Electrochemical sensors for the detection of SARS-cov-2 virus. Chem. Eng. J. 2022, 430, 132966. [Google Scholar] [CrossRef] [PubMed]
- Bukkitgar, S.D.; Shetti, N.P.; Aminabhavi, T.M. Electrochemical investigations for covid-19 detection-a comparison with other viral detection methods. Chem. Eng. J. 2021, 420, 127575. [Google Scholar] [CrossRef] [PubMed]
- Imran, S.; Ahmadi, S.; Kerman, K. Electrochemical biosensors for the detection of SARS-cov-2 and other viruses. Micromachines 2021, 12. [Google Scholar] [CrossRef]
- Veerapandian, M.; Hunter, R.; Neethirajan, S. Dual immunosensor based on methylene blue-electroadsorbed graphene oxide for rapid detection of the influenza a virus antigen. Talanta 2016, 155, 250–257. [Google Scholar] [CrossRef]
- Yi, J.; Lee, S.; Lee, J.Y. Biomimetic polypyrrole/hyaluronic acid electrodes integrated with hyaluronidase inhibitors offer persistent electroactivity and resistance to cell binding. J. Mat. Chem. B 2022, 10, 1591–1600. [Google Scholar] [CrossRef]
- Huang, H.; Wu, J.; Lin, X.; Li, L.; Shang, S.; Yuen, M.C.; Yan, G. Self-assembly of polypyrrole/chitosan composite hydrogels. Carbohydr. Polym. 2013, 95, 72–76. [Google Scholar] [CrossRef]
- Le HQ, Sauriat-Dorizon, H; Korri-Youssoufi, H. Investigation of spr and electrochemical detection of antigen with polypyrrole functionalized by biotinylated single-chain antibody: a review. Anal. Chim. Acta 2010, 674, 1–8. [CrossRef]
- Wink, T.; van Zuilen, S.J.; Bult, A.; van Bennkom, W.P. Self-assembled monolayers for biosensors. Analyst 1997, 122, 43R–50R. [Google Scholar] [CrossRef]
- Frasconi, M.; Mazzei, F.; Ferri, T. Protein immobilization at gold-thiol surfaces and potential for biosensing. Anal. Bioanal. Chem. 2010, 398, 1545–1564. [Google Scholar] [CrossRef]
- Rusmini, F.; Zhong, Z.; Feijen, J. Protein immobilization strategies for protein biochips. Biomacromolecules 2007, 8, 1775–1789. [Google Scholar] [CrossRef] [PubMed]
- Jonkheijm, P.; Weinrich, D.; Schroder, H.; Niemeyer, C.M.; Waldmann, H. Chemical strategies for generating protein biochips. Angew. Chem.-Int. Edit. 2008, 47, 9618–9647. [Google Scholar] [CrossRef] [PubMed]
- Jarocka, U.; Sawicka, R.; Gora-Sochacka, A.; Sirko, A.; Zagorski-Ostoja, W.; Radecki, J.; Radecka, H. Electrochemical immunosensor for detection of antibodies against influenza a virus h5n1 in hen serum. Biosens. Bioelectron. 2014, 55, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. In vitro selection of rna molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: rna ligands to bacteriophage t4 dna polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Colas, P.; Cohen, B.; Jessen, T.; Grishina, I.; Mccoy, J.; Brent, R. Genetic selection of peptide aptamers that recognize and inhibit cyclin-dependent kinase 2. Nature 1996, 380, 548–550. [Google Scholar] [CrossRef]
- Darmostuk, M.; Rimpelova, S.; Gbelcova, H.; Ruml, T. Current approaches in selex: an update to aptamer selection technology. Biotechnol. Adv. 2015, 33, 1141–1161. [Google Scholar] [CrossRef]
- Wu, Y.X.; Kwon, Y.J. Aptamers: the "evolution" of selex. Methods 2016, 106, 21–28. [Google Scholar] [CrossRef]
- Cai, S.; Yan, J.; Xiong, H.; Liu, Y.; Peng, D.; Liu, Z. Investigations on the interface of nucleic acid aptamers and binding targets. Analyst 2018, 143, 5317–5338. [Google Scholar] [CrossRef]
- Yu, Z.G.; Lai, R.Y. A reagentless and reusable electrochemical aptamer-based sensor for rapid detection of ampicillin in complex samples. Talanta 2018, 176, 619–624. [Google Scholar] [CrossRef]
- Das, R.; Dhiman, A.; Mishra, S.K.; Haldar, S.; Sharma, N.; Bansal, A.; Ahmad, Y.; Kumar, A.; Tyagi, J.S.; Sharma, T.K. Structural switching electrochemical dna aptasensor for the rapid diagnosis of tuberculous meningitis. Int. J. Nanomed. 2019, 14, 2103–2113. [Google Scholar] [CrossRef] [PubMed]
- Lum, J.; Wang, R.; Hargis, B.; Tung, S.; Bottje, W.; Lu, H.; Li, Y. An impedance aptasensor with microfluidic chips for specific detection of h5n1 avian influenza virus. Sensors 2015, 15, 18565–18578. [Google Scholar] [CrossRef] [PubMed]
- Karash, S.; Wang, R.; Kelso, L.; Lu, H.; Huang, T.J.; Li, Y. Rapid detection of avian influenza virus h5n1 in chicken tracheal samples using an impedance aptasensor with gold nanoparticles for signal amplification. J. Virol. Methods 2016, 236, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Gattani, A.; Singh, S.V.; Agrawal, A.; Khan, M.H.; Singh, P. Recent progress in electrochemical biosensors as point of care diagnostics in livestock health. Anal. Biochem. 2019, 579, 25–34. [Google Scholar] [CrossRef]
- Brazaca, L.C.; Dos, S.P.; de Oliveira, P.R.; Rocha, D.P.; Stefano, J.S.; Kalinke, C.; Abarza, M.R.; Bonacin, J.A.; Janegitz, B.C.; Carrilho, E. Biosensing strategies for the electrochemical detection of viruses and viral diseases - a review. Anal. Chim. Acta 2021, 1159, 338384. [Google Scholar] [CrossRef]
- Yousefi, H.; Mahmud, A.; Chang, D.; Das, J.; Gomis, S.; Chen, J.B.; Wang, H.; Been, T.; Yip, L.; Coomes, E.; Li, Z.; Mubareka, S.; Mcgeer, A.; Christie, N.; Gray-Owen, S.; Cochrane, A.; Rini, J.M.; Sargent, E.H.; Kelley, S.O. Detection of SARS-cov-2 viral particles using direct, reagent-free electrochemical sensing. J. Am. Chem. Soc. 2021, 143, 1722–1727. [Google Scholar] [CrossRef]
- Couto, R.A.; Lima, J.L.; Quinaz, M.B. Recent developments, characteristics and potential applications of screen-printed electrodes in pharmaceutical and biological analysis. Talanta 2016, 146, 801–814. [Google Scholar] [CrossRef]
- Torre, R.; Costa-Rama, E.; Nouws, H.; Delerue-Matos, C. Screen-printed electrode-based sensors for food spoilage control: bacteria and biogenic amines detection. Biosensors-Basel 2020, 10. [Google Scholar] [CrossRef]
- Perez-Fernandez, B.; Costa-Garcia, A.; Muniz, A.E. Electrochemical (bio)sensors for pesticides detection using screen-printed electrodes. Biosensors-Basel 2020, 10. [Google Scholar] [CrossRef]
- Rebelo, P.; Costa-Rama, E.; Seguro, I.; Pacheco, J.G.; Nouws, H.; Cordeiro, M.; Delerue-Matos, C. Molecularly imprinted polymer-based electrochemical sensors for environmental analysis. Biosens. Bioelectron. 2021, 172, 112719. [Google Scholar] [CrossRef]
- Rishpon, J. Electrochemical biosensors for environmental monitoring. Rev. Environ. Health 2002, 17, 219–247. [Google Scholar] [CrossRef]
- Yunus, M.H.; Yusof, N.A.; Abdullah, J.; Sulaiman, Y.; Ahmad, R.N.; Md, N.S. Simultaneous amperometric aptasensor based on diazonium grafted screen-printed carbon electrode for detection of cfp10 and mpt64 biomarkers for early tuberculosis diagnosis. Biosensors-Basel 2022, 12. [Google Scholar] [CrossRef]
- Pothipor, C.; Aroonyadet, N.; Bamrungsap, S.; Jakmunee, J.; Ounnunkad, K. A highly sensitive electrochemical microrna-21 biosensor based on intercalating methylene blue signal amplification and a highly dispersed gold nanoparticles/graphene/polypyrrole composite. Analyst 2021, 146, 2679–2688. [Google Scholar] [CrossRef]
- Couto, R.A.; Lima, J.L.; Quinaz, M.B. Recent developments, characteristics and potential applications of screen-printed electrodes in pharmaceutical and biological analysis. Talanta 2016, 146, 801–814. [Google Scholar] [CrossRef]
- Wang, C.; Xu, Y.; Zhao, X.; Li, S.; Qian, Q.; Wang, W.; Mi, X. A double-tetrahedral dna framework based electrochemical biosensor for ultrasensitive detection and release of circulating tumor cells. Analyst 2021, 146, 6474–6481. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.; Lu, D.; Senecal, A.; Kurup, P. Voltammetric detection of arsenic (iii) using gold nanoparticles modified carbon screen printed electrodes: application for facile and rapid analysis in commercial apple juice. Food Chem. 2021, 352, 129327. [Google Scholar] [CrossRef] [PubMed]
- Abd-Rabboh, H.; Amr, A.; Naglah, A.M.; Almehizia, A.A.; Kamel, A.H. Effective screen-printed potentiometric devices modified with carbon nanotubes for the detection of chlorogenic acid: application to food quality monitoring. RSC Adv. 2021, 11, 38774–38781. [Google Scholar] [CrossRef]
- Spring, S.A.; Goggins, S.; Frost, C.G. Ratiometric electrochemistry: improving the robustness, reproducibility and reliability of biosensors. Molecules 2021, 26. [Google Scholar] [CrossRef] [PubMed]
- Kaya, H.O.; Cetin, A.E.; Azimzadeh, M.; Topkaya, S.N. Pathogen detection with electrochemical biosensors: advantages, challenges and future perspectives. J. Electroanal. Chem. 2021, 882, 114989. [Google Scholar] [CrossRef]
- Soares, R.; Hjort, R.G.; Pola, C.C.; Parate, K.; Reis, E.L.; Soares, N.; Mclamore, E.S.; Claussen, J.C.; Gomes, C.L. Laser-induced graphene electrochemical immunosensors for rapid and label-free monitoring of salmonella enterica in chicken broth. ACS Sens. 2020, 5, 1900–1911. [Google Scholar] [CrossRef]
| Methods | Time required | Convenience | On site detection |
|---|---|---|---|
| Virus isolation | 2-3 Days | Not to be carried | No |
| PCR | 1-2 Hours | Not to be carried | No |
| ELISA | 6-8 Hours | Not to be carried | No |
| Electrochemical biosensor | 10-30 min | Be portable | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




